Abstract
Epstein–Barr virus (EBV) is a recognized oncogenic virus that is related to the occurrence of lymphoma, nasopharyngeal carcinoma (NPC), and approximately 10% of gastric cancer (GC). EBV is a herpesvirus, and like other herpesviruses, EBV has a biphasic infection mode made up of latent and lytic infections. It has been established that latent infection promotes tumorigenesis in previous research, but in recent years, there has been new evidence that suggests that the lytic infection mode could also promote tumorigenesis. In this review, we mainly discuss the contribution of the EBV lytic phase to tumorigenesis, and graphically illustrate their relationship in detail. In addition, we described the relationship between the lytic cycle of EBV and autophagy. Finally, we also preliminarily explored the influence of the tumorigenesis effect of the EBV lytic phase on the future treatment of EBV-associated tumors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
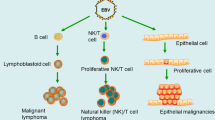
The Epstein–Barr virus (EBV), also known as human herpesvirus 4 (HHV4), is a large double-stranded DNA (dsDNA) virus belonging to the gamma-herpesvirus subfamily. EBV is a well-characterized oncovirus associated with several malignancies [1], including epithelial tumors like nasopharyngeal carcinoma (NPC), gastric cancer (GC) and diverse lymphoid malignancies such as Burkitt's lymphoma (BL), Hodgkin's lymphoma (HL) and NK/T cell tumors. Like other herpesviruses, EBV has a biphasic infection mode: latent infection and lytic infection.
The biphasic life cycle of EBV allows it to establish a latent period after primary infection. During the first infection, a short cracking program will be run temporarily, and then the latent infection will be established. The cracking program refers to the fact that when EBV is de novo infected, in addition to expressing the latent genes, it also expresses the lytic genes, and then enters the latent period (this stage is called the "pre-latent abortive lytic state") [2]. In the latent state, the genomic DNA exists in the form of episomes in the nucleus, in which the closed circular plasmid binds to histone proteins, so that only a limited number of viral latent genes can be produced [3]. The latent period of EBV can be separated into three types: latency I, II, and III. During latent infection, standard expression products are six EBV-encoded nuclear antigens (EBNAs: 1, 2, 3A, 3B, 3C and LP), two latent membrane proteins (LMPs: 1, 2A and 2B), two non-translated small RNAs (EBERs: 1 and 2), and more than 40 microRNAs (BHRF1 and BART miRNAs) [4, 5]. These limited latent viral genes are specifically responsible for tumorigenesis, apoptosis inhibition, immune escape and do not increase the number of viral particles. However, this silent mode of infection is conducive to the long-term existence of the virus as only a few gene products that can be targeted by the host's immune system are expressed. It has been well-documented that latent infection with EBV can promote tumorigenesis and is a cunning way to evade host attac k[6,7,8].
Only a small percentage of infected cells could transition from the latent to the lytic period and produce viral offspring. In the lytic phase, > 70 viral proteins are produced. Interestingly, EBV lytic genes are expressed in cascade in a time-regulated manner, including immediate-early (IE) genes, early (E) genes, and late (L) genes. We described the differences between the latent and lytic phases of EBV in Fig. 1. In the lytic cascade, the most critical step is encoding their protein products by the two IE genes, BZLF1 and BRLF1, which then activate the expression of other viral lytic genes to initiate the entire lysis cascade [9]. The promoters of BZLF1 and BRLF1 (encoded by Zp and Rp, respectively) are initially activated by transcription factors, and subsequently, BZLF1 and BRLF1 proteins activate their own and each other's promoters, greatly amplifying the induction of lysis [10]. They then cooperatively activate the promoters of the early lytic genes, which are mainly responsible for encoding the enzymes required for virus replication. Following viral genome replication, late viral genes are expressed, primarily responsible for encoding viral structural proteins, resulting in the generation of progeny virus particles and cell lysis and death. Although EBV in cancer cells is mostly in a latent state, the lytic cycle of the virus is also expected to play a crucial role in tumorigenesis and maintenance, as a small number of cells in the lytic cycle are associated with the secretion of cytokines or growth factors that promote carcinogenesis. We described the lytic genes of EBV in Table 1 [3, 11,12,13,14,15,16,17].
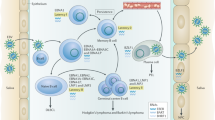
The differences between latent and lytic infection. Note The left half represents latent infection, mainly expressing some latent proteins, leading to tumorigenesis, and evading host attack. The right half represents lytic infection, expressing almost seventy proteins that can generate new viral particles by cascaded expression of lytic genes
Past and present understanding of lytic infection
After reactivation, EBV will briefly undergo three successive lysis stages, including IE, E, and L. The viral IE genes BZLF1 and BRLF1 are first transcribed, encoding the transactivators Zta and Rta, respectively, and then express the early genes required for EBV genome replication. EBV DNA expresses late genes after replication, mainly encoding viral structural proteins, including capsid antigens and membrane proteins, and then wraps the viral genome to produce mature viral particles. In the past, EBV latent phase was considered the main stage for promoting tumorigenesis, so induction of EBV lysis is one of the methods for treating EBV-related tumors.
During a complete lytic cycle, viral DNA replicates into large, intact molecules that are subsequently cleaved and packaged into the viral progeny, which are released to infect neighboring cells [18]. What's different now is with the in-depth study of EBV, more and more literatures have shown (explained in detail below) that the lytic phase of EBV could promote tumorigenesis by increasing viral particles horizontally and producing some binding effector proteins vertically. In other words, the latent phase of EBV mainly promotes tumorigenesis, while the main purpose of the lytic phase is to help EBV infect uninfected cells. The role of the lysis stage of EBV on tumorigenesis appears to be underestimated. The reason for the underestimation may be that the lytic cycle of EBV may lead to cell death [19]. Therefore, the action of lytic cycle proteins must either act in trans on other cells, or as a rare process, defined proteins of the lytic cycle prevent such cell death [20]. In other words, the involvement of EBV lytic proteins in tumorigenesis may be associated with very rare events that are masked by frequent and different main effects. It's a very complicated situation, but it's so interesting that it deserves a detailed discussion. Since the lytic stage of EBV can also promote tumorigenesis, in what way? What impact will it have on the treatment of EBV-related tumors?
The differences between primary EBV infection and reactivation concerning induction of malignancy
EBV primary infection and reactivation act differently in inducing and modulating malignancy. In primary infections, LMP1 and LMP2A have been studied in more detail. The C-terminus of LMP1 has three functional domains, called C-terminal activation regions 1–3(CTAR1, CTAR2 and CTAR3), which have strong signal transduction capabilities [21]. They can activate NF-κB [22], JNK/SAPK [23], PI3-K/Akt [24], ERK-MAPK [25], PLC/PKC [26] and JAK/STAT [26] signaling pathways, thereby affecting cell proliferation, invasion, apoptosis and other cellular processes. There are several motifs at the N-terminus of LMP2A that are docking sites for the tyrosine kinases Lyn, Syk and the ubiquitin ligase Nedd4/Itchy [27], which activate PI3-K/Akt [28], JNK/SAPK [29], ERK-MAPK [29] and Wnt/β-catenin [30] signaling pathway, promote cell growth, inhibit apoptosis and differentiation, and contribute to cell transformation. Differently, BZLF1 is a transcription factor, but also considered an enhancer [31]. Therefore, when EBV is reactivated, BZLF1 functions primarily as a DNA-binding protein to influence tumorigenesis in two ways: (1) BZLF1 can activate various promoters, such as transforming growth factor-β1 (TGF-β1), which may enhance viral escape responses to host immunity[32]; (2) BZLF1 can interact with cellular proteins to inhibit or synergize them, such as NF-κB and p53[33].
Some of the EBV lytic proteins with essential roles in promoting tumorigenesis
This section mainly studies the perspectives of more extensive lytic genes on tumorigenesis and development. However, some reviews have introduced the role of BZLF1 and BRLF1 in tumorigenesis in detail, so we will not go into detail here [34, 35].
BNLF2a
BNLF2a is an early lytic protein of EBV encoding 60 amino acids with a hydrophobic C-terminal and a hydrophilic N-terminal domain, which plays an important role in the immune evasion of the virus. Studies have shown that these two domains have different functions. Post-translationally, the hydrophobic C-terminus is mainly inserted into the endoplasmic reticulum (ER) membrane, while the hydrophilic N-terminus is exposed in the cytoplasmic matrix and binds directly to the core transporter associated with antigen processing (TAP) complex, which subsequently blocks TAP with antigen processing [36, 37]. In addition, the presence of BNLF2a interferes with the ability of host cells to recognize proteins at different stages of EBV lysis differently. In EBV-infected B cells, when BNLF2a is deficient, CD8 + T cells have significantly increased recognition of immediate early lytic proteins and early lytic proteins, but no significant difference for late lytic proteins [38].
BARF1
BARF1 plays a multifaceted role in tumorigenesis, including proliferation, apoptosis, and malignant transformation. BARF1 promotes cell proliferation in GC by activating the NF-κB/cyclinD axis and reducing cell cycle inhibitors p21WAF1 [39]. BARF1 could upregulate the anti-apoptotic proteins Bcl-2 and Bcl-xL through the MAPK/c-Jun signaling pathway [40]. Literature has shown that BARF1 plays a role in the malignant transformation of GC and NPC: in gastric epithelial cell GES, malignant transformation mainly includes increased cell growth activity, shortening the G1 phase in the cell cycle, prolongation of the S phase, enhanced colony-forming ability and enhanced tumorigenicity [41]. In the nasopharyngeal immortalized epithelial cell NP69, BARF1 cooperates with Ras to form denser foci, larger cell size and greater resistance to growth factor deprivation [42].
BHRF1
BHRF1 is mainly associated with autophagy and is described in detail in the sixth subsection of this paper.
Cell biological function of tumorigenesis in the lytic phase of EBV-induced carcinoma
More and more evidence indicateS that EBV lytic genes can promote EBV-induced tumorigenesis. EBV lytic genes can affect tumorigenesis by regulating tumor cells and tumor microenvironment. Next, we will discuss how the lytic phase of EBV contributes to tumorigenesis from seven aspects: immunomodulation and immune evasion, angiogenesis and invasion, apoptosis and cell cycle and genomic instability (GI).
Immunomodulation and immune evasion
EBV lytic genes can perform immune regulation and evasion by evading antiviral responses, reducing antigen presentation, and inducing interleukin (IL) production. IL refers to lymphokines that interact with white blood cells or immune cells. It transmits information, regulates immune cells, mediates T and B cell activation, proliferation and differentiation, and plays an important role in inflammation.
The EBV lytic phase can escape antiviral responses. RIG-I is a kind of pattern recognition receptors (PRRs) that senses endogenous and pathogenic RNAs [43]. When cells are initially infected with EBV, RIG-I promotes the secretion of inflammasome-dependent cytokines IL-1β and IL-18 by sensing EBERs, thereby activating T cells and NK cells [44, 45]. Long et al. showed that BRLF1 could inhibit the activation of inflammasome RIG-1 in the early stage of EBV primary infection and reactivation by binding to subunits of RNA polymerase III (POLR3F and POLR3G), and then escape the antiviral response of T cells and NK cells through inflammasome-dependent factors [45]. BNLF2a plays an essential role in evading immune surveillance by encoding a 60 amino acid protein that interferes with antigen presentation to CD8+T cells. In addition, BNLF2a can encode an inhibitor of the TAP, thereby reducing antigen presentation and EBV-specific CD8+T cell immune recognition to infected cells, reducing the immunogenicity of EBV-infected cells, and protecting the virus from immunity cell attack [46]. Studies have shown that Zta could directly target the promoter of BNLF2a to promote its expression [47]. EBV BGLF5 expression is sufficient to induce the shutdown of host gene expression, including HLA class I. This may be an important reason affecting antigen presentation [48]. BILF1 down-regulates MHC-I and affects the endocytosis and exocytosis pathways of MHC-I, thereby reducing the ability of T cell antigen recognition and inhibiting antigen presentation [49]. BDLF3 is a powerful protein in reducing antigen presentation. It can target both MHC-I molecules to impair the recognition of CD8 + T cells and MHC-II molecules to impair the recognition of CD4 +T cells. BDLF3 downregulates MHC mainly by ubiquitinating MHC [50].
Both BZLF1 and BRLF1 can induce the secretion of IL-6, and the production of IL-6 promotes early lymphoproliferative diseases in patients [51]. Katsumura et al. demonstrated that BZLF1 could induce the expression of IL-13 with LMP1, allowing EBV-infected cells to proliferate in the presence of T cells, thereby inducing tumorigenesis [52]. Tsai et al. found that BZLF1 could directly bind to the IL-13 promoter through the AP-1 binding site to induce IL-13 expression. The production of IL-13 promotes the proliferation of B cells, thereby contributing to the occurrence of EBV-associated lymphoproliferative disorders [53]. In NPC cells, BZLF1 can upregulate the expression of IL-8 and increase chemotactic activity, which may contribute to tumor growth, angiogenesis and metastasis and other oncogenic processes [54]. Lee et al. found that EBV-induced immunomodulators in NPC could upregulate IL-10 produced by monocytes and promote local immune suppression [55].
EBV has its IL-10 homolog: vIL-10, encoded by BCRF1. It can attenuate the NK cell-mediated killing effect on infected B cells, interfere with CD4 + T cell activity, and regulate cytokine response, thereby promoting immune escape during the lytic phase of EBV-infected cells [46]. In addition, The HLA-I antigen processing machinery (APM) plays a crucial role in the anticancer immune response. Ren et al. demonstrated that in NPC, vIL-10 could inhibit the transcription of some components in APM through the NF-κB pathway, thereby inhibiting antigen presentation [56]. The relationship between immunomodulation and immune evasion with the EBV lytic cycle is shown in Fig. 2.
The roles of lytic genes in immunodulation and immune evasion. Note This figure demonstrates that EBV lytic genes evade host attack in different ways. These methods include inhibiting the production of inflammasomes, inhibiting antigen presentation, and regulating the production of IL. As a result, the activity of CD8 + T cells, CD4 + T cells or NK cells is inhibited, leading to the escape of EBV
Angiogenesis and invasion
VEGF is a highly specific vascular endothelial growth factor that promotes increased vascular permeability and extracellular matrix degeneration, vascular endothelial cell migration, proliferation and blood vessel formation. Hong et al. showed that supernatants of early passage LCLs infected with WT EBV contained more VEGF than cells infected with BZLF1-deletion and BRLF1-deletion viruses. Interestingly, however, there was no significant difference in the amount of VEGF within the cells, suggesting that BZLF1 and BRLF1 increase VEGF secretion and release [57]. These phenomena indicated that the lysis of infected cells might promote tumorigenesis by enhancing angiogenesis.
In addition to promoting angiogenesis, the lytic genes of EBV can also promote tumorigenesis by promoting cell invasion in vitro. For all types of solid tumors, metastasis is critically dependent on matrix-metallo proteinases (MMPs) [58]. Lan et al. demonstrated that BZLF1-induced the expression of MMP3 and MMP9 in NPC cells, and that BZLF1-induced cell migration required MMP3, and cell invasion required MMP3 and MMP9 [59]. BZLF1 binds to the AP-1 element of DNA promoters and increases the transcription and protein expression levels of MMP3 and MMP9. In addition to MMP3 and MMP9, BZLF1 can upregulate MMP1 in NPC [60]. The relationship between angiogenesis and invasion with the EBV lytic cycle is shown in Fig. 3.
The roles of lytic genes in tumor angiogenesis and invasion. Note As seen from the picture, angiogenesis is mainly by increasing the secretion of VEGF. The lytic proteins can also increase the expression of MMPs from the transcriptional level and protein level, respectively, by binding to AP-1 on DNA, and finally improve the invasion ability of tumor cells
Apoptosis and cell cycle
Apoptosis is a highly regulated, energy-dependent form of cell suicide. The apoptotic machinery is conserved among metazoans from worms to humans, and choreographed cell death is required for proper development and tissue homeostasis [61]. BCL-2 family proteins can be divided into two types: pro-apoptosis and pro-survival. Pro-apoptotic BCL-2 proteins including BIM, PUMA, NOXA, BID, BMF, BIK, BAD, HRK, BAX, BAK and BOK, and pro-survival BCL-2 proteins include BCL-2, BCL-XL, BCL-W, MCL-1 and A1/BFL1. Whether or not cells undergo apoptosis is determined by the relative expression and interaction of pro-survival and pro-apoptotic BCL-2 proteins [62].
EBV encodes two viral homologs of the cellular Bcl-2 anti-apoptotic proteins: BHRF1 and BALF1. BHRF1 binds to a variety of pro-apoptotic proteins to inhibit cell apoptosis. Research has shown that BHRF1 may act either by binding to the most lethal form of BH3-only protein (BIM) or by working catalytically on BIM to block apoptosis [63]. In addition to BIM, Kvansakul et al. also concluded that BHRF1 could keep BAK inactivated by direct binding but must indirectly inhibit BAX. In addition to the above-mentioned apoptosis-related proteins, BHRF1 also inhibits apoptosis by binding to the BH3 domains of PUMA and BID. BALF2 is associated with BAX and BAK to play an anti-apoptotic effect. Because BALF1 inhibits cell apoptosis, BALF1 increases the survival rate of cells under low serum conditions. Mice injected with BALF1 transfectants had more tumors than mice injected with control transfectants [64]. However, there is also literature showing that BALF2 can antagonize the anti-apoptotic activity of BHRF1 [65].
The EBV lytic protein BZLF1 prevents cells in G0/G1 [66], G1/S and G2/M [67]. According to reports, BRLF1 cells re-enter the S phase [68]. Studies have shown that BRLF1 could interfere with cells during the G1/S transition and induce cellular senescence [69]. CCCTC-binding factor (CTCF) is a multifunctional protein that participates in gene expression and higher-order chromatin structure in cellular and viral genomes. CTCF occupancy and DNA methylation are mutually exclusive. By reducing CTCF overloading, BRLF1 could turn off the expression of MYC, CCND1 and JUN, thereby hindering cell cycle progression [70]. The relationship between apoptosis and cell cycle with the EBV lytic cycle is shown in Fig. 4.
The roles of lytic genes in apoptosis and cell cycle. Note This figure demonstrates that many lytic proteins can inhibit mitochondrial outer membrane permeabilization by binding to the BH3 domains of apoptosis-related proteins, ultimately inhibiting apoptosis. Lytic proteins can also affect cell growth by affecting different nodes in the cell cycle and related genes in cellular processes
GI
GI seems to be a hallmark of cancer. It is defined as an increase in the frequency of genetic changes, including subtle sequence changes and chromosomal changes and is considered the cause or result of carcinogenesis. In B cells, EBV particles induce centrosome amplification and chromosomal instability [71]. In NPC, repeated chemical reactivation of EBV promotes GI and enhances the tumor progression of NPC cells [72].
53BP1 and RNF8 DNA damage lesions are involved in DNA damage repair and cell cycle checkpoints, which can prevent DNA damage. BZLF1 can prevent the formation of these two lesions to induce DNA damage and increase the GI of NPC cells [73]. Huang et al. showed that BRLF1 could cause chromosome mis-segregation in NPC cells through the ERK pathway, accumulating GI and increasing tumorigenic characteristics [74]. Micronucleus is an indicator of GI. Wu et al. indicated that BGLF5 could increase micronuclei and DNA damage in human epithelial cells [75]. Chiu et al. found that BALF3 could induce micronuclei and DNA strand breaks. After repeated induction of BALF3 expression, the genome copy number of NPC cells was aberrated, and the tumorigenic characteristics also increased [76]. The relationship between GI with the EBV lytic cycle is shown in Fig. 5.
EBV lytic infection promotes tumorigenesis and autophagy
Autophagy is the process of engulfing one's cytoplasmic proteins or organelles, coating them into vesicles, and fusing with lysosomes to form autophagic lysosomes, degrading the contents of the lysosomes, thereby fulfilling the metabolic needs of the cell itself and the renewal of specific organelles [77]. Autophagy has a dual role in the development of cancer. It can not only promote the growth of tumor cells but also prevent the further development of the disease, the so-called “autophagy paradox” [78]. The complex relationship between autophagy and microorganisms can protect the body by activating the immune system. In addition, autophagy and microorganisms can communicate with each other in various ways to influence various physiological and pathological reactions involved in cancer progression. Various molecular mechanisms related to microbiota disorders and autophagy activation control the outcome of pro-tumor or anti-tumor responses, depending on the type of cancer, the tumor microenvironment, and the stage of the disease.
Hung et al. found that Rta could promote autophagy through ERK1/2, and autophagy could promote EBV replication [79]. Consistent with this evidence, autophagy is induced by the primary treatment that induces the EBV lytic cycle and confirms that the suppression of the first autophagy step counteracts viral replication. In addition, the final stage of autophagy is inhibited by EBV so that the virus can avoid being cleared by lysosomal proteases and usurp the autophagy mechanism of intracellular transport [80]. Blocking the last step of autophagy helps EBV replication, possibly due to further inhibition of the last step that has been induced by viral replication. However, using different experimental methods to silence the ATG protein and other types, these authors found that inhibiting the initial autophagy step can also enhance EBV replication [80]. EBV weakens the fusion of autophagosomes and lysosomes during the replication process and spreads in autophagic vesicles [81]. The lytic phase of EBV may cause autophagy to be dysregulated, thereby counteracting the antiviral immune response, promoting virus replication and tumorigenesis.
Shao et al. showed that BALF0 and BALF1 could modulate autophagy, which may be beneficial to the replication of virus particles [82]. Futhermore, BHRF1 interacts with BECN1/Beclin1 to induce mitochondrial autophagy, a cellular process that can specifically isolate and degrade mitochondria. Since mitochondrial autophagy plays a central role in innate immunity, the authors further explored the role of BHRF1 in innate immunity and found that BHRF1 could induce type I IFN-induced inhibition by inducing mitochondrial autophagy [81]. In addition, Song et al. also demonstrated that BHRF1 could enhance mitochondrial autophagy in NPC, and NPC may adapt to the hypoxia of the tumor microenvironment through the enhancement of autophagy, thereby promoting tumorigenesis [83].
Similarities and differences of lytic proteins in tumor initiation and tumorigenesis
The potential of EBV lytic cycle proteins in tumor initiation and tumorigenesis is similar in that lytic proteins could act as DNA-binding proteins that bind to effector molecules to promote tumorigenesis and progression. However, there are also different aspects: unlike tumor progression, at tumor initiation, (1) lytic proteins could promote tumor initiation in a paracrine manner, for example, in the presence of T cells, induction of IL13 by BZLF1 could enhance the transformation of EBV-infected B lymphocytes [84]; (2) it could also change the expression pattern of kinases, such as phosphorylation of tyrosine kinases (TKT) to initiate signaling cascades that promote tumor initiation [52]. The ways of lytic proteins at tumor initiation are more diverse, but whether the potential is more powerful still needs further literature verification and discussion.
Is there any difference in the effect of inhibiting EBV lysis in different therapies?
Previous studies believe that inducing EBV lysis is a way to treat EBV-associated malignancies. Reactivating recurrent EBV caused by dysregulation of BZLF1 or BRLF1 expression may also be a potential therapy for EBV-related tumors. However, EBV lysis can also promote tumorigenesis. Will this have any impact on future treatment methods? NPC is radioresistant, a problem that has long been an obstacle to NPC treatment. Studies have shown that the degree of EBV reactivation in NPC is positively correlated with the degree of radioresistance to treatment [85], thus reducing EBV reactivation may provide a benefit in the treatment of patients with radioresistant cancers. However, this study did not demonstrate whether EBV activation and radioresistance are causal or parallel. In the clinic, for the first time, Hu et al. generated a molecular subtype model for NPC based on 8-OHdG and EBV DNA levels. These dual markers can identify high-risk patients with poor prognoses but may benefit from sequential treatment of reactive oxygen species block followed by radiotherapy, which provides a new perspective for the precise treatment of NPC [85]. According to the continuous exploration of researchers, many small molecules have been found to have the function of inhibiting the lysis and replication of EBV. It mainly includes nucleoside analogs, valproic acid (VPA), maribavir (MBV), rapamycin and so on. Nucleoside analogs first acquire activity by phosphorylation to the triphosphate form and then inhibit viral DNA polymerase and incorporate into viral DNA, thereby preventing viral replication [86]. VPA is used to activate EBV in cell culture, but this needs to be in a specific cellular context and may require the assistance of other drugs. In most cases, VPA can antagonize other histone deacetylase (HDAC) inhibitors, thereby impairing their ability to reactivate EBV lytic cycle [87]. The mechanism of action of MBV is mainly to effectively inhibit EBV transcription, genome replication and infectivity by inhibiting BGLF4 [88]. It has been reported that rapamycin alters the lytic replication of EBV in B cells through the regulation of mTOR activity, and has different effects on different lysis periods. Notably, the inhibitory effect of rapamycin was dose-dependent, and rapamycin did not inhibit EBV lytic replication in epithelial cells [89]. Unfortunately, although drugs mentioned above have an effect in inhibiting the lytic cycle of EBV, they are not effective in treating EBV infection. And these drugs are toxic, so finding compounds from natural sources seems like a safer option. We still have a long way to go in the search for therapeutic drugs. Since the lytic period of EBV can also promote tumorigenesis, inducing EBV lysis may no longer be a single way for us to treat EBV-related malignant tumors. Different therapies are selected in different periods to inhibit or induce lysis or use them in combination. This leads us to new thinking.
Conclusions
The lytic phase of EBV promotes tumorigenesis mainly in two ways: (1) the production of infectious particles to infect more cells; (2) the encoded products of the lytic genes through immunomodulation, angiogenesis and invasion, apoptosis and cell cycle and induction of GI promote tumorigenesis. In addition to these, EBV can also lead to dysregulation of autophagy, and we believe that dysregulation of autophagy may also play a role in tumorigenesis. There are many aspects to be explored regarding the relationship between EBV lytic phase and autophagy and tumorigenesis. Although only a small number of cells in EBV-related tumors are in a lytic phase, promoting the lytic phase on tumorigenesis cannot be underestimated. Therefore, it is very important to explore the detailed mechanism of the EBV lytic period on tumorigenesis and the impact on subsequent treatment methods.
Abbreviations
- EBV:
-
Epstein–Barr virus
- NPC:
-
Nasopharyngeal carcinoma
- GC:
-
Gastric cancer
- HHV4:
-
Human herpesvirus 4
- dsDNA:
-
Double-stranded DNA
- BL:
-
Burkitt's lymphoma
- HL:
-
Hodgkin's lymphoma
- EBNAs:
-
Epstein–Barr virus nuclear antigens
- LMPs:
-
Latent membrane proteins
- IE:
-
Immediate early genes
- E:
-
Early
- L:
-
Late
- CSF-1:
-
Colony stimulating factor 1
- TAP:
-
Transporter associated with antigen processing
- GPCR:
-
G protein-coupled receptor
- GI:
-
Genomic instability
- LCLs:
-
Lymphoblastoid cell lines
- VEGF:
-
Vascular endothelial growth factor
- IL:
-
Interleukin
- PRRs:
-
Pattern recognition receptors
- APM:
-
Antigen processing machinery
- MMPs:
-
Matrix-metallo proteinases
- BIM:
-
BH3-only protein
- CTCF:
-
CCCTC-binding factor
- VPA:
-
Valproic acid
- MBV:
-
Maribavir
- HDAC:
-
Histone deacetylase
References
Bedri S, Sultan AA, Alkhalaf M, Al-moustafa AE, Vranic S (2019) Epstein-Barr virus (EBV) status in colorectal cancer: a mini review. Hum Vaccines Immunother 15:603–610. https://doi.org/10.1080/21645515.2018.1543525
Murata T (2018) Encyclopedia of EBV-encoded lytic genes: an update. Adv Exp Med Biol 1045:395–412. https://doi.org/10.1007/978-981-10-7230-7_18
Murata T et al (2021) Molecular basis of epstein-barr virus latency establishment and lytic reactivation. Viruses. https://doi.org/10.3390/v13122344
Thorley-Lawson DA (2015) EBV persistence-introducing the virus. Curr Top Microbiol Immunol 390:151–209. https://doi.org/10.1007/978-3-319-22822-8_8
Skalsky RL, Cullen BR (2015) EBV noncoding RNAs. Curr Top Microbiol Immunol 391:181–217. https://doi.org/10.1007/978-3-319-22834-1_6
Levitskaya J et al (1995) Inhibition of antigen processing by the internal repeat region of the Epstein-Barr virus nuclear antigen-1. Nature 375:685–688. https://doi.org/10.1038/375685a0
Middeldorp JM, Pegtel DM (2008) Multiple roles of LMP1 in Epstein-Barr virus induced immune escape. Semin Cancer Biol 18:388–396. https://doi.org/10.1016/j.semcancer.2008.10.004
Lin JH et al (2015) Epstein-Barr virus LMP2A suppresses MHC class II expression by regulating the B-cell transcription factors E47 and PU.1. Blood 125:2228–2238. https://doi.org/10.1182/blood-2014-08-594689
Flemington EK, Lytle JP, Cayrol C, Borras AM, Speck SH (1994) DNA-binding-defective mutants of the Epstein-Barr virus lytic switch activator Zta transactivate with altered specificities. Mol Cell Biol 14:3041–3052. https://doi.org/10.1128/mcb.14.5.3041-3052.1994
Kenney SC, Mertz JE (2014) Regulation of the latent-lytic switch in Epstein-Barr virus. Semin Cancer Biol 26:60–68. https://doi.org/10.1016/j.semcancer.2014.01.002
Jung YJ, Choi H, Kim H, Lee SK (2014) MicroRNA miR-BART20-5p stabilizes Epstein-Barr virus latency by directly targeting BZLF1 and BRLF1. J Virol 88:9027–9037. https://doi.org/10.1128/jvi.00721-14
Strockbine LD et al (1998) The Epstein-Barr virus BARF1 gene encodes a novel, soluble colony-stimulating factor-1 receptor. J Virol 72:4015–4021. https://doi.org/10.1128/jvi.72.5.4015-4021.1998
Cuconati A, White E (2002) Viral homologs of BCL-2: role of apoptosis in the regulation of virus infection. Genes Dev 16:2465–2478. https://doi.org/10.1101/gad.1012702
Strong MJ et al (2015) Latent expression of the Epstein-Barr virus (EBV)-encoded major histocompatibility complex class I TAP inhibitor, BNLF2a EBV-Positive Gastric Carcinomas. J Virol 89:10110–10114. https://doi.org/10.1128/jvi.01110-15
Chiu SH et al (2014) Epstein-Barr virus BALF3 has nuclease activity and mediates mature virion production during the lytic cycle. J Virol 88:4962–4975. https://doi.org/10.1128/jvi.00063-14
Paulsen SJ, Rosenkilde MM, Eugen-Olsen J, Kledal TN (2005) Epstein-Barr virus-encoded BILF1 is a constitutively active G protein-coupled receptor. J Virol 79:536–546. https://doi.org/10.1128/jvi.79.1.536-546.2005
Kurilla MG, Heineman T, Davenport LC, Kieff E, Hutt-Fletcher LM (1995) A novel Epstein-Barr virus glycoprotein gp150 expressed from the BDLF3 open reading frame. Virology 209:108–121. https://doi.org/10.1006/viro.1995.1235
Tsurumi T, Fujita M, Kudoh A (2005) Latent and lytic Epstein-Barr virus replication strategies. Rev Med Virol 15:3–15. https://doi.org/10.1002/rmv.441
Kawanishi M (1993) Epstein-Barr virus induces fragmentation of chromosomal DNA during lytic infection. J Virol 67:7654–7658. https://doi.org/10.1128/jvi.67.12.7654-7658.1993
Inman GJ, Binné UK, Parker GA, Farrell PJ, Allday MJ (2001) Activators of the Epstein-Barr virus lytic program concomitantly induce apoptosis, but lytic gene expression protects from cell death. J Virol 75:2400–2410. https://doi.org/10.1128/jvi.75.5.2400-2410.2001
Huen DS, Henderson SA, Croom-Carter D, Rowe M (1995) The Epstein-Barr virus latent membrane protein-1 (LMP1) mediates activation of NF-kappa B and cell surface phenotype via two effector regions in its carboxy-terminal cytoplasmic domain. Oncogene 10:549–560
Laherty CD, Hu HM, Opipari AW, Wang F, Dixit VM (1992) The Epstein-Barr virus LMP1 gene product induces A20 zinc finger protein expression by activating nuclear factor kappa B. J Biol Chem 267:24157–24160
Morris MA et al (2016) The Epstein-Barr virus encoded LMP1 oncoprotein modulates cell adhesion via regulation of activin A/TGFβ and β1 integrin signalling. Sci Rep 6:19533. https://doi.org/10.1038/srep19533
Thornburg NJ et al (2006) LMP1 signaling and activation of NF-kappaB in LMP1 transgenic mice. Oncogene 25:288–297. https://doi.org/10.1038/sj.onc.1209023
Dawson CW, Laverick L, Morris MA, Tramoutanis G, Young LS (2008) Epstein-Barr virus-encoded LMP1 regulates epithelial cell motility and invasion via the ERK-MAPK pathway. J Virol 82:3654–3664. https://doi.org/10.1128/jvi.01888-07
Zheng H, Li LL, Hu DS, Deng XY, Cao Y (2007) Role of Epstein-Barr virus encoded latent membrane protein 1 in the carcinogenesis of nasopharyngeal carcinoma. Cell Mol Immunol 4:185–196
Miller CL et al (1994) Epstein-Barr virus protein LMP2A regulates reactivation from latency by negatively regulating tyrosine kinases involved in sIg-mediated signal transduction. Infect Agents Dis 3:128–136
Portis T, Longnecker R (2004) Epstein-Barr virus (EBV) LMP2A mediates B-lymphocyte survival through constitutive activation of the Ras/PI3K/Akt pathway. Oncogene 23:8619–8628. https://doi.org/10.1038/sj.onc.1207905
Chen SY, Lu J, Shih YC, Tsai CH (2002) Epstein-Barr virus latent membrane protein 2A regulates c-Jun protein through extracellular signal-regulated kinase. J Virol 76:9556–9561. https://doi.org/10.1128/jvi.76.18.9556-9561.2002
Morrison JA, Klingelhutz AJ, Raab-Traub N (2003) Epstein-Barr virus latent membrane protein 2A activates beta-catenin signaling in epithelial cells. J Virol 77:12276–12284. https://doi.org/10.1128/jvi.77.22.12276-12284.2003
Ramasubramanyan S et al (2015) Epstein-Barr virus transcription factor Zta acts through distal regulatory elements to directly control cellular gene expression. Nucleic Acids Res 43:3563–3577. https://doi.org/10.1093/nar/gkv212
Cayrol C, Flemington EK (1995) Identification of cellular target genes of the Epstein-Barr virus transactivator Zta: activation of transforming growth factor beta igh3 (TGF-beta igh3) and TGF-beta 1. J Virol 69:4206–4212. https://doi.org/10.1128/jvi.69.7.4206-4212.1995
Dreyfus DH, Liu Y, Ghoda LY, Chang JT (2011) Analysis of an ankyrin-like region in Epstein Barr Virus encoded (EBV) BZLF-1 (ZEBRA) protein: implications for interactions with NF-κB and p53. Virology journal 8:422. https://doi.org/10.1186/1743-422x-8-422
Germini D et al (2020) Oncogenic properties of the EBV ZEBRA protein. Cancers. https://doi.org/10.3390/cancers12061479
Li H et al (2016) Epstein-Barr virus lytic reactivation regulation and its pathogenic role in carcinogenesis. Int J Biol Sci 12:1309–1318. https://doi.org/10.7150/ijbs.16564
Horst D et al (2011) EBV protein BNLF2a exploits host tail-anchored protein integration machinery to inhibit TAP. J Immunol 186:3594–3605. https://doi.org/10.4049/jimmunol.1002656
Wycisk AI et al (2011) Epstein-Barr viral BNLF2a protein hijacks the tail-anchored protein insertion machinery to block antigen processing by the transport complex TAP. J Biol Chem 286:41402–41412. https://doi.org/10.1074/jbc.M111.237784
Croft NP et al (2009) Stage-specific inhibition of MHC class I presentation by the Epstein-Barr virus BNLF2a protein during virus lytic cycle. PLoS Pathog 5:e1000490. https://doi.org/10.1371/journal.ppat.1000490
Chang MS et al (2013) Epstein-Barr virus-encoded BARF1 promotes proliferation of gastric carcinoma cells through regulation of NF-κB. J Virol 87:10515–10523. https://doi.org/10.1128/jvi.00955-13
Zhang Y, Xu M, Zhang X, Chu F, Zhou T (2018) MAPK/c-Jun signaling pathway contributes to the upregulation of the anti-apoptotic proteins Bcl-2 and Bcl-xL induced by Epstein-Barr virus-encoded BARF1 in gastric carcinoma cells. Oncol Lett 15:7537–7544. https://doi.org/10.3892/ol.2018.8293
Li S et al (2020) The role of the Epstein-Barr virus-encoded BARF1 gene expressed in human gastric epithelial cells. Turkish J Gastroenterol 31:775–781. https://doi.org/10.5152/tjg.2020.18827
Jiang R, Cabras G, Sheng W, Zeng Y, Ooka T (2009) Synergism of BARF1 with Ras induces malignant transformation in primary primate epithelial cells and human nasopharyngeal epithelial cells. Neoplasia (New York, N.Y.) 11:964–973. https://doi.org/10.1593/neo.09706
Brennan K, Bowie AG (2010) Activation of host pattern recognition receptors by viruses. Curr Opin Microbiol 13:503–507. https://doi.org/10.1016/j.mib.2010.05.007
Ansari MA et al (2013) Constitutive interferon-inducible protein 16-inflammasome activation during Epstein-Barr virus latency I, II, and III in B and epithelial cells. J Virol 87:8606–8623. https://doi.org/10.1128/jvi.00805-13
Long X et al (2021) BRLF1 suppresses RNA Pol III-mediated RIG-I inflammasome activation in the early EBV lytic lifecycle. EMBO Rep 22:e50714. https://doi.org/10.15252/embr.202050714
Jochum S, Moosmann A, Lang S, Hammerschmidt W, Zeidler R (2012) The EBV immunoevasins vIL-10 and BNLF2a protect newly infected B cells from immune recognition and elimination. PLoS Pathog 8:e1002704. https://doi.org/10.1371/journal.ppat.1002704
Almohammed R et al (2018) Mechanism of activation of the BNLF2a immune evasion gene of Epstein-Barr virus by Zta. J Gen Virol 99:805–817. https://doi.org/10.1099/jgv.0.001056
Rowe M et al (2007) Host shutoff during productive Epstein-Barr virus infection is mediated by BGLF5 and may contribute to immune evasion. Proc Natl Acad Sci USA 104:3366–3371. https://doi.org/10.1073/pnas.0611128104
Zuo J et al (2011) The Epstein-Barr virus-encoded BILF1 protein modulates immune recognition of endogenously processed antigen by targeting major histocompatibility complex class I molecules trafficking on both the exocytic and endocytic pathways. J Virol 85:1604–1614. https://doi.org/10.1128/jvi.01608-10
Quinn LL et al (2016) The missing link in Epstein-Barr virus immune evasion: the BDLF3 gene induces ubiquitination and downregulation of major histocompatibility complex class I (MHC-I) and MHC-II. J Virol 90:356–367. https://doi.org/10.1128/jvi.02183-15
Jones RJ et al (2007) Roles of lytic viral infection and IL-6 in early versus late passage lymphoblastoid cell lines and EBV-associated lymphoproliferative disease. Int J Cancer 121:1274–1281. https://doi.org/10.1002/ijc.22839
Katsumura KR, Maruo S, Takada K (2012) EBV lytic infection enhances transformation of B-lymphocytes infected with EBV in the presence of T-lymphocytes. J Med Virol 84:504–510. https://doi.org/10.1002/jmv.23208
Tsai SC et al (2009) EBV Zta protein induces the expression of interleukin-13, promoting the proliferation of EBV-infected B cells and lymphoblastoid cell lines. Blood 114:109–118. https://doi.org/10.1182/blood-2008-12-193375
Hsu M et al (2008) Epstein-Barr virus lytic transactivator Zta enhances chemotactic activity through induction of interleukin-8 in nasopharyngeal carcinoma cells. J Virol 82:3679–3688. https://doi.org/10.1128/jvi.02301-07
Lee CH et al (2011) Epstein-Barr virus Zta-induced immunomodulators from nasopharyngeal carcinoma cells upregulate interleukin-10 production from monocytes. J Virol 85:7333–7342. https://doi.org/10.1128/jvi.00182-11
Ren YX et al (2016) Viral IL-10 down-regulates the “MHC-I antigen processing operon” through the NF-κB signaling pathway in nasopharyngeal carcinoma cells. Cytotechnology 68:2625–2636. https://doi.org/10.1007/s10616-016-9987-9
Hong GK et al (2005) Epstein-Barr virus lytic infection is required for efficient production of the angiogenesis factor vascular endothelial growth factor in lymphoblastoid cell lines. J Virol 79:13984–13992. https://doi.org/10.1128/jvi.79.22.13984-13992.2005
Niland S, Riscanevo AX, Eble JA (2021) Matrix metalloproteinases shape the tumor microenvironment in cancer progression. Int J Mol Sci. https://doi.org/10.3390/ijms23010146
Lan YY et al (2013) Epstein-Barr virus Zta upregulates matrix metalloproteinases 3 and 9 that synergistically promote cell invasion in vitro. PLoS ONE 8:e56121. https://doi.org/10.1371/journal.pone.0056121
Lu J, Chua HH, Chen SY, Chen JY, Tsai CH (2003) Regulation of matrix metalloproteinase-1 by Epstein-Barr virus proteins. Can Res 63:256–262
Rodriguez-Nieto S, Zhivotovsky B (2006) Role of alterations in the apoptotic machinery in sensitivity of cancer cells to treatment. Curr Pharm Des 12:4411–4425. https://doi.org/10.2174/138161206779010495
Strasser A, Cory S, Adams JM (2011) Deciphering the rules of programmed cell death to improve therapy of cancer and other diseases. EMBO J 30:3667–3683. https://doi.org/10.1038/emboj.2011.307
Fitzsimmons L et al (2020) EBV BCL-2 homologue BHRF1 drives chemoresistance and lymphomagenesis by inhibiting multiple cellular pro-apoptotic proteins. Cell Death Differ 27:1554–1568. https://doi.org/10.1038/s41418-019-0435-1
Hsu WL, Chung PJ, Tsai MH, Chang CL, Liang CL (2012) A role for Epstein-Barr viral BALF1 in facilitating tumor formation and metastasis potential. Virus Res 163:617–627. https://doi.org/10.1016/j.virusres.2011.12.017
Marshall WL et al (1999) Epstein-Barr virus encodes a novel homolog of the bcl-2 oncogene that inhibits apoptosis and associates with Bax and Bak. J Virol 73:5181–5185. https://doi.org/10.1128/jvi.73.6.5181-5185.1999
Guo Q et al (2010) Transactivators Zta and Rta of Epstein-Barr virus promote G0/G1 to S transition in Raji cells: a novel relationship between lytic virus and cell cycle. Mol Immunol 47:1783–1792. https://doi.org/10.1016/j.molimm.2010.02.017
Rodriguez A, Jung EJ, Flemington EK (2001) Cell cycle analysis of Epstein-Barr virus-infected cells following treatment with lytic cycle-inducing agents. J Virol 75:4482–4489. https://doi.org/10.1128/jvi.75.10.4482-4489.2001
Swenson JJ, Mauser AE, Kaufmann WK, Kenney SC (1999) The Epstein-Barr virus protein BRLF1 activates S phase entry through E2F1 induction. J Virol 73:6540–6550. https://doi.org/10.1128/jvi.73.8.6540-6550.1999
Huang SY et al (2012) Epstein-Barr virus Rta-mediated transactivation of p21 and 14–3-3σ arrests cells at the G1/S transition by reducing cyclin E/CDK2 activity. J Gen Virol 93:139–149. https://doi.org/10.1099/vir.0.034405-0
Chen YJ et al (2017) Epstein-Barr virus Rta-mediated accumulation of DNA methylation interferes with CTCF binding in both host and viral genomes. J Virol. https://doi.org/10.1128/jvi.00736-17
Shumilov A et al (2017) Epstein-Barr virus particles induce centrosome amplification and chromosomal instability. Nat Commun 8:14257. https://doi.org/10.1038/ncomms14257
Fang CY et al (2009) Recurrent chemical reactivations of EBV promotes genome instability and enhances tumor progression of nasopharyngeal carcinoma cells. Int J Cancer 124:2016–2025. https://doi.org/10.1002/ijc.24179
Yang J et al (2015) Epstein-Barr virus BZLF1 protein impairs accumulation of host DNA damage proteins at damage sites in response to DNA damage. Lab Invest 95:937–950. https://doi.org/10.1038/labinvest.2015.69
Huang SY et al (2017) Epstein-Barr virus BRLF1 induces genomic instability and progressive malignancy in nasopharyngeal carcinoma cells. Oncotarget 8:78948–78964. https://doi.org/10.18632/oncotarget.20695
Wu CC et al (2010) Epstein-Barr virus DNase (BGLF5) induces genomic instability in human epithelial cells. Nucleic Acids Res 38:1932–1949. https://doi.org/10.1093/nar/gkp1169
Chiu SH et al (2014) Epstein-Barr virus BALF3 mediates genomic instability and progressive malignancy in nasopharyngeal carcinoma. Oncotarget 5:8583–8601. https://doi.org/10.18632/oncotarget.2323
Jiang J, Xu H, Wang Y, Lu H (2021) Identification and validation of autophagy-related gene nomograms to predict the prognostic value of patients with cervical cancer. J Oncol 2021:5583400. https://doi.org/10.1155/2021/5583400
Chmurska A, Matczak K, Marczak A (2021) Two Faces of Autophagy in the Struggle against Cancer. Int J Mol Sci. https://doi.org/10.3390/ijms22062981
Kim JE, Kang TC (2021) CDDO-me attenuates astroglial autophagy via Nrf2-, ERK1/2-SP1- and Src-CK2-PTEN-PI3K/AKT-mediated signaling pathways in the hippocampus of chronic epilepsy rats. Antioxidants. https://doi.org/10.3390/antiox10050655
Granato M et al (2014) Epstein-barr virus blocks the autophagic flux and appropriates the autophagic machinery to enhance viral replication. J Virol 88:12715–12726. https://doi.org/10.1128/jvi.02199-14
Vilmen G et al (2021) BHRF1, a BCL2 viral homolog, disturbs mitochondrial dynamics and stimulates mitophagy to dampen type I IFN induction. Autophagy 17:1296–1315. https://doi.org/10.1080/15548627.2020.1758416
Shao Z, Borde C, Quignon F, Escargueil A, Maréchal V (2019) Epstein-Barr virus BALF0 and BALF1 modulate autophagy. Viruses. https://doi.org/10.3390/v11121099
Song S et al (2020) BHRF1 enhances EBV mediated nasopharyngeal carcinoma tumorigenesis through modulating mitophagy associated with mitochondrial membrane permeabilization transition. Cells. https://doi.org/10.3390/cells9051158
Lu J et al (2000) Upregulation of tyrosine kinase TKT by the Epstein-Barr virus transactivator Zta. J Virol 74:7391–7399. https://doi.org/10.1128/jvi.74.16.7391-7399.2000
Hu J et al (2022) Erratum: Targeting Epstein-Barr virus oncoprotein LMP1-mediated high oxidative stress suppresses EBV lytic reactivation and sensitizes tumors to radiation therapy: Erratum. Theranostics 12:3609. https://doi.org/10.7150/thno.73630
Coen N, Duraffour S, Topalis D, Snoeck R, Andrei G (2014) Spectrum of activity and mechanisms of resistance of various nucleoside derivatives against gammaherpesviruses. Antimicrob Agents Chemother 58:7312–7323. https://doi.org/10.1128/aac.03957-14
Daigle D et al (2011) Valproic acid antagonizes the capacity of other histone deacetylase inhibitors to activate the Epstein-barr virus lytic cycle. J Virol 85:5628–5643. https://doi.org/10.1128/jvi.02659-10
Whitehurst CB et al (2013) Maribavir inhibits Epstein-Barr virus transcription through the EBV protein kinase. J Virol 87:5311–5315. https://doi.org/10.1128/jvi.03505-12
Adamson AL, Le BT, Siedenburg BD (2014) Inhibition of mTORC1 inhibits lytic replication of Epstein-Barr virus in a cell-type specific manner. Virol J 11:110. https://doi.org/10.1186/1743-422x-11-110
Acknowledgements
Thanks to everyone who helped with this article.
Funding
This work was supported by Natural Science Foundation of Shandong Province [ZR2020MH302; ZR2020MC020].
Author information
Authors and Affiliations
Contributions
Writing-original draft preparation, YL; writing-review and editing, YZ and BL.
Corresponding authors
Ethics declarations
Conflict of interest
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work, there is no professional or other personal interest of any nature or kind in any product, service and/or company that could be construed as influencing the position presented in, or the review of, the manuscript entitled.
Additional information
Edited by Hartmut Hengel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, Y., Zhang, Y. & Luo, B. The lytic phase of Epstein–Barr virus plays an important role in tumorigenesis. Virus Genes 59, 1–12 (2023). https://doi.org/10.1007/s11262-022-01940-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-022-01940-6