Abstract
The Mauritia flexuosa palm tree is widely distributed in South America, and is associated with flooded environments. It dominates the arboreal stratum in the highly threatened marsh ecosystems called “veredas” in the Cerrado biome (neotropical savanna), and its oleaginous fruits are important for the maintenance of the regional fauna and as a source of income for traditional human populations. The seeds of this palm demonstrate an unusual association of recalcitrance and dormancy, and information about its seed bank maintenance in natural environments is scarce. We measured the variations in water content, viability, and germination of seeds maintained over 1 year in four typical vereda microenvironments (edge, near the water, in the water, and at the lower zone of the vereda) and related that information to soil water contents and climatic data. Seed viability was best preserved under constantly flooded conditions (at the lower zone of the vereda), with the potential for maintaining a temporary seed bank in the aquatic environment that would favor dispersal. Although the seeds are intolerant of dehydration, they have a high capacity for maintaining their water contents under partially drained soil conditions, and overcoming dormancy is favored under those conditions. Mauritia flexuosa establishment strategies are driven by sophisticated interactions with climate and with vereda microenvironments, with a tendency for maintaining seed banks and seedling banks in localities with flooded or drained soils, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mauritia flexuosa L.f. (Arecaceae) (“buriti”) is a widely distributed palm tree in South America (Dransfield et al. 2008; Horn et al. 2012; Endress et al. 2013). The species produces large quantities of oleaginous fruits important for the maintenance of the regional fauna (Cymerys et al. 2005; Lorenzi et al. 2010) and that are used as food by traditional human populations (Holm et al. 2008; Manzi and Coomes 2009). Buriti palm trees show great economic potential for use as a food source and for manufacturing cosmetics and medicines (Bernal et al. 2011; Martins et al. 2012; Koolen et al. 2013). Certain aspects of M. flexuosa seed biology related to the rapid loss of their viability and low germinability, however, have limited seedling production for commercial purposes as well as the conservation of natural populations (Spera et al. 2001; Orozco-Segovia et al. 2003).
Recent works have demonstrated an association of recalcitrance and dormancy in M. flexuosa seeds (Silva et al. 2014; Veloso et al. 2016). This condition is rare, and there are still open questions about the behaviors of seeds showing those characteristics in the natural environment (Tweddle et al. 2003). Recalcitrance is related to desiccation intolerance, resulting in a low capacity for the maintenance of seed viability and the formation of soil seed banks (Hong and Ellis 1996; Berjak and Pammenter 2008, 2013). Dormancy is characterized by intrinsic germination restrictions caused by impediments related to the dispersal unit that must be overcome in order to complete germination—which usually requires a relatively long time interval (Finch-Savage and Leubner-Metzger 2006; Bewley et al. 2013). The apparent opposition of those conditions makes their study interesting from the point of view of our knowledge of plants’ reproductive strategies as well as their interactions with their habitats.
Despite the ecological and economic importance of M. flexuosa, little is currently known about the ecology of its populations, including recruitment and seedling establishment rates in the natural environment (Horn et al. 2012; Virapongse et al. 2017). M. flexuosa dominates the arboreal stratum of the vereda ecosystem in the Cerrado (neotropical savanna) biome (Dransfield et al. 2008; Lorenzi et al. 2010; Silva et al. 2014) in soils that usually show high water-saturation levels and abundant partially decomposed organic matter (Cymerys et al. 2005; Endress et al. 2013). Studies involving the behavior of those seeds in their natural environment will contribute to a better understanding of the establishment strategies of M. flexuosa and the perpetuation of its populations—favoring actions directed toward its in situ conservation.
Interactions between the anatomic and physiological characteristics of seeds and edaphoclimatic conditions determine seed bank dynamics (Bewley et al. 2013; Baskin and Baskin 2014; Dalling et al. 2011)—but are strongly modulated by microenvironments (Long et al. 2014; Lopez-Toledo et al. 2013). Preliminary laboratory studies have suggested that microenvironmental conditions are particularly important for seed bank formation by M. flexuosa, as those seeds, unlike the general pattern of recalcitrant behavior, have the potential to remain viable if stored with their water contents preserved (Silva et al. 2014; Veloso et al. 2016). However, as the water regimes of veredas have been highly affected by local anthropogenic impacts, and quite possibly by global climate change, studies on the dynamics of the seed banks of this key species may provide support for estimating the resilience of that ecosystem.
In order to test the hypothesis that the peculiarities of M. flexuosa seeds contribute to its adaptation to the natural environment, we evaluated the physiological aspects of seeds maintained for 1 year in four typical vereda microenvironments, to address the following questions: (i) what are the effects of seasonal microenvironmental and climatic conditions on seed viability? (ii) can seed banks or seedling banks be maintained in different microenvironments? We also sought to investigate the importance of the association of recalcitrance and dormancy to the reproduction of that species.
Materials and methods
Study area, seed bank establishment, and sample collections
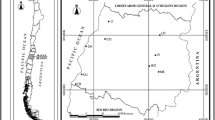
The study area was a vereda marsh located in the Pandeiros River Environmental Protection Area (APA—Pandeiros), in the municipality of Bonito de Minas, in northern Minas Gerais State, Brazil (44º55′21″W, 15º13′19″S). Approximately 9000 fruits of M. flexuosa were collected on the ground after natural abscission from more than 100 adult plants in December 2014. The integrity of the mesocarp and a yellowish abscission scar were considered indicative of recent abscission.
The fruits were manually de-pulped and the seeds selected, with discard of those with lesions indicative of predation by insects or necrosis; 7280 seeds were used in the experiments. The isolated seeds were placed (at 10 cm depth in the soil in polyethylene containers (15 × 22 × 12 cm) without their lids and with holes drilled in all of their sides) in three typical vereda microenvironments: (1) edge (the contact between the arboreal vegetation and the herbaceous vegetation zones) (ED); (2) near the water course (about 1 m from a small stream) (NW); (3) lower zone (an area with a predominant presence of M. flexuosa, with moist soil rich in organic matter) (LZ) (Araújo et al. 2002) (Fig. 1). Twenty seeds were placed in each container which were filled with soil from each microenvironment. The containers were distributed and buried, in each microenvironment, in four different places, 2 m apart. The seeds were also held in four permeable (raffia-like) plastic bags in the water course itself (at a water depth of 10 cm) (IW). Seed samples were analyzed at 0 days (initial condition) (December, 2014) and after 45, 90, 180, and 365 days. The seeds were kept submerged in water (which was changed daily) during transport and in the laboratory until examined.
Environmental assessments
At the time of establishment of each experimental seed bank, 2 kg soil samples (formed by combining four subsamples of 500 g) were collected at 0–20 cm depths in each of the three microenvironments for physicochemical analyses, following the methodology of Donagema et al. (2011); these were used to characterize the microenvironments (Table 1). The water contents of the soils of the three microenvironments were determined by collecting 50 g soil samples at 10 cm depths in the places where the fruits were buried, based on the ratio between the dry mass (after drying at 105 °C, for 24 h) and the initial fresh mass; these evaluations were carried out (with four replicates) at the beginning of the experiment (0 days) and after 90, 180, and 365 days (Donagema et al. 2011).
Daily rainfall data were obtained from the nearest climatology station of the National Meteorological Institute (Brasil 2016), located in the municipality of Januária, approximately 70 km from the study area; the sites have the same geomorphological (Sanfransciscana depression) and climatic (type Aw, Köppen) conditions.
Water contents of the seeds and embryos
The water content of the seeds was determined in the initial condition (before the establishment of the seed bank) and after 45, 90,180, and 365 days. At each time, from each microenvironment, four replicates of 10 seeds were obtained. Each replicate was represented by a container containing 20 seeds. In the case of seeds kept submerged, the replicates were obtained from four different bags kept inside the water. In each replicate, the germinated or predated seeds were discarded, and five seeds were randomly taken for the evaluations. The predation was identified by occurrence of typical perforations in the seeds caused by insects that had consumed the embryo.
For each treatment (combination between microenvironment and collection time), the water contents (fresh mass basis) of four replicates of five non-germinated seeds were determined from the dry mass (after drying at 105 °C for 24 h) to fresh mass ratio (Brasil 2009). These same evaluations were also performed with isolated embryos obtained by sectioning the seeds (using a machete). Evaluations were not performed at 365 days, as all of the seeds in all of the microenvironments showed predation by that time.
Embryo viability and germination
Embryo viability was evaluated using the tetrazolium test on four replicates of 10 embryos per microenvironment at 0, 45, 90, 180, and 365 days. Samples were obtained as previously described. The embryos were immersed in 1% triphenyl tetrazolium chloride, incubated in the dark at 30 °C for five hours, and then visually analyzed for staining (Spera et al. 2001; Brasil 2009). Evaluations were not performed at 365 days, as all of the seeds in all of the microenvironments showed predation by that time.
At each time (45, 90, 180, and 365 days), 200 seeds were removed from each microenvironment and germination was evaluated in the field, considering seeds as germinated if they showed protrusion of the cotyledonary petiole or developed seedlings (Ribeiro et al. 2015). Seeds were considered dead (by predation and/or microbial deterioration) if the embryo was absent or showed lesions or necrosis.
Statistical analyses
Evaluations of soil water contents considered a 3 (microenvironments) × 4 (collection times) factorial scheme. Evaluations of the water contents of the seeds and embryos and embryo viability considered a 4 (microenvironments) × 3 (collection times) factorial scheme; the initial condition (0 days) was considered an additional treatment. Data were submitted to the Kolmogorov–Smirnov normal distribution test, the Levene variance homogeneity test, and analysis of variance. The averages were compared by the Tukey test, at a 5% level of probability. Linear regression analysis was performed between the water contents of the embryos and viability. The analyses were performed using the software SAS System for Windows V8 (SAS Institute, Cary, USA).
Results
Environmental assessments
All of the soil samples had high percentages of sand (edge > near water > lower zone) and higher pH levels than commonly observed in Cerrado environments (the pH in Cerrado soils is usually below 5) (Ribeiro et al. 1999), associated with the absence of Al (Table 1). The levels of essential minerals (P, K, Ca, and Mg) and organic matter followed the order: near water > lower zone > edge.
The experiment was initiated in December, during the rainy season, and variations in soil water contents were observed throughout the year in the different microenvironments (F = 272.35; P < 0.0001) (Fig. 2a, b). The water content of the soil at the vereda edge (which was always less humid than the other microenvironments) showed a gradual drying up to 180 days, then increasing up to 365 days (associated with rainfall variations). Soil moisture near the water became reduced up to 90 days and elevated up to 180 days (even with the reduction in rainfall), indicating that this characteristic is influenced by the dynamics of the watercourse, which can experience small impoundments due to the accumulation of plant material. The soil at the lower zone of the vereda showed higher moisture in relation to the other microenvironments at times up to 90 days, and maintained saturation levels above 60% in all evaluations (despite reductions throughout the year). Comparisons of the rainfall data for the study year (total precipitation: 631 mm) with historical records (mean: 909 mm) indicated that 2015 was relatively dry, but nonetheless maintained the typical seasonal pattern of the Cerrado biome—with rainfall being concentrated in the summer.
Variations of the soil water contents of three vereda microenvironments along the year (a) and monthly rainfall in the municipality of Januária, Minas Gerais State, Brazil in 2015, with historical records (b). The same letters indicate the absence of significant differences using the Tukey test, at a 5% level of probability. Upper case letters represent the effects of time within each microenvironment; lower case letters reflect the effects of the microenvironments within each time. Vertical bars indicate the standard errors of the means
Seed and embryo water contents
The water contents of all of the seeds remained close to 50% in all of the microenvironments during the evaluation period, and did not decrease in relation to their initial conditions (Fig. 3a). The water contents of the seeds held in the water and near the water increased at 180 days in relation to their initial condition (F = 4.38; P < 0.0001). At 180 days, the water contents of the seeds kept at the edge were lower in relation to seeds kept in the water.
Water contents of Mauritia flexuosa seeds (a), and embryos (b), held in four vereda microenvironments for 180 days. The same letters indicate the absence of significant differences using the Tukey test, at a 5% level of probability. Upper case letters represent the effects of time within each microenvironment; lower case letters reflect the effects of the microenvironments within each time. Asterisks indicate significant differences in relation to the initial condition. The vertical bars indicate the standard errors of the means
The water contents of the embryos were higher than 75% at all evaluation times (Fig. 3b). At 45 and 180 days, the water contents of embryos from seeds kept submerged in water were higher than the initial condition (0 days) (F = 5.26; P < 0.0001). At 45 days, the water contents of the embryos of seeds kept in the water were higher than those kept close to the water or at the lower zone of the vereda. At 90 days, the embryos of seeds kept at the water’s edge had lower water contents in relation to the other microenvironments. There were no variations in the water contents of embryos from seeds kept at the lower zone of the vereda over time. At 180 days, the water contents of the embryos of seeds kept submerged in water and those kept near the water were similar to each other, and higher than those of the other treatments.
Embryo viability and germination
Embryo viability among seeds kept in the water was lower than the initial condition at 45 days, and showed important reductions during subsequent periods, and was lower than all of the other treatments after 90 days (F = 272.35; P < 0.0001) (Fig. 4). Viability reductions, in relation to the initial condition, were also observed among the embryos of seeds kept near the water, after 90 days. Embryo viability was preserved during the entire evaluation period at the lower zone of the vereda. The same occurred at the border of the vereda for up to 90 days; evaluations for longer periods of time than 180 days were not possible in that microenvironment, as all of the embryos had germinated or had suffered predation (the seeds showed typical perforations caused by insects). Regression analysis showed that the water content of the embryos was not directly related to viability.
Percentages of viable Mauritia flexuosa embryos obtained from seeds kept in four vereda microenvironments for 180 days, as evaluated using the tetrazolium test. The same letters indicate the absence of significant differences using the Tukey test, at a 5% level of probability. Upper case letters represent the effects of time within each microenvironment; lower case letters reflect the effects of the microenvironments within each time. Asterisks indicate significant differences in relation to the initial condition. Vertical bars indicate the standard errors of the means
Germination started at 45 days at the vereda edge and reached higher percentage at 180 days (when 43% of the evaluated seeds had germinated) (Fig. 5). All seeds that germinated showed live seedlings with developing roots and leaves. Seeds maintained submerged in the water did not germinate. The germination of seeds kept near the water or submerged in it reached only 3 and 1%, respectively, by the end of the evaluation period.
Discussion
Plant species adapted to humid environments commonly produce seeds that show recalcitrant germination and are intolerant of dehydration—with rapid germination—therefore tending to form seedling banks instead of seed banks (Tweddle et al. 2003; Long et al. 2014). Our results showed that M. flexuosa has an establishment strategy that varies according to each particular vereda microenvironment, with a tendency to form seedling banks at the vereda edge (where the soil is drier) while maintaining seed banks in the lower zone (where soils remain humid throughout the year)
Seed viability in different microenvironments
Although veredas can extend over large areas, they are usually associated with flowing water and are commonly elongated, which creates marked heterogeneity in their transversal profiles. The main differences between the vereda microenvironments considered in the present study were related to water availability. Soil water-retention capacities, whose ordering was lower zone > near the water > edge, were directly related to their clay contents and inversely related to their sand contents and their positions in the landscape profile (with the lower section of the vereda being at the lowest elevation), as previously described in other publications within that ecosystem (Sousa et al. 2011; Ramos et al. 2006). in addition, edges are subject to less shading and are more exposed to wind, which results in greater surface drying.
Initial seed viability was maintained in the soils of all of the different microenvironments for 45 days, and in the soil at the lower zone of the vereda for up to 180 days. Viability became reduced in the environment near the water over time, however, with only 40% of the seeds remaining viable after 180 days. Despite low soil moisture levels at the edge of the water course (less than 20% during the period without precipitation—June–September), the water contents of the seeds and embryos held there did not become reduced in relation to their initial conditions. Veloso et al. (2016) demonstrated, under laboratory conditions, that the embryos of M. flexuosa showed a tendency for slow dehydration (although they were intolerant of dehydration), which was associated with the presence of cells with mucilage-rich vacuoles that favored water retention. However, some studies have shown that although the maintenance of high water contents is essential for preserving the viability of recalcitrant seeds, loss of viability usually occurs due to factors such as the consumption of reserves, oxidative stress, and microbial deterioration (Berjak and Pammenter 2008, 2013; Dalling et al. 2011). Thus, although the water content of the embryos has been maintained in all evaluated microenvironments, it is probable that the other factors that control viability are more favorable for the maintenance of M. flexuosa seed banks in the lower zone of the vereda.
The chemical characteristics of a soil, as well as the quantities and the constitutions of its organic matter, commonly influence the maintenance of seed viability (Long et al. 2014; Galeano et al. 2015), although it was not possible to establish a direct relationship between those factors in the present study. pH levels, which may influence seed persistence (Long et al. 2009; Pakeman et al. 2012), did not show extreme values or large variations among the soils of the different microenvironments evaluated. The soils nearest the water had higher organic matter contents and P, Ca, and Mg concentrations than the other soils, but it was not clear if those conditions contributed to the lower viability observed in that microenvironment as compared to the lower portion of the vereda.
The increases in predation and/or seed deterioration rates after 180 days, coincided with the driest period of the year (June–September), and represented the main factor affecting seed persistence in the soil, as all non-germinated seeds were observed to be damaged at 365 days. A study by Johansson (2009) highlighted the vulnerability of M. flexuosa seeds exposed to drier soils—a condition that may have been particularly relevant during the present study year, as it was drier than the historical average.
Due to the association of populations of M. flexuosa with marsh environments, commonly crossed by streams, seed dispersal by water should be important for that species (Johansson 2009; Silva et al. 2014). Our results showed reduced viability with seed immersion time, and they did not germinate under those conditions. It is possible that the low oxygen availability imposed by immersion, and/or interactions with the aquatic biota, may have compromised germination. It is important to note, however, that the fruits and seeds of that species float, and will not remain fully immersed under natural conditions. Due to the difficulties encountered in maintaining large numbers of seeds on the surface of the water for long periods of time, however, we opted for examining seeds that had been fully immersed—which may have led to overestimations of viability losses. However, 54 and 10% of the immersed seeds were still viable after 45 and 180 days respectively, which points to the importance of dispersal by water for M. flexuosa.
The potential for maintaining seed and seedling banks
Although uncommon, associations of recalcitrance and dormancy may occur in the seeds of some species (Tweddle et al. 2003). While recalcitrance is considered a specialization factor in humid and stable environments, dormancy favors adaptation to unstable environments with seasonal climates (Baskin and Baskin 2014). It is interesting that veredas show associations of stable and unstable microenvironments in relation to soil moisture. While the soils at the lower end of the vereda remained constantly humid, the soils in the other microenvironments showed large fluctuations in their moisture levels during the year.
The recalcitrance of M. flexuosa seeds, which limits the occurrence of that species to humid environments, is modulated by the water-retention capacity of the seed, which allows the prolonged maintenance of viability under dry soil conditions until dormancy is overcome. Germination, on the other hand, is modulated by dormancy, and is restricted in humid, shady soil environments (where the densities of adult individuals are higher and seed bank maintenance is more feasible) and favored at the vereda edge (where competition with adult individuals is lower and the maintenance of seed banks is less viable over time). The ecological importance of the association of recalcitrance and dormancy in M. flexuosa seeds has been proposed in previous studies (Silva et al. 2014; Veloso et al. 2016), but without the evidence presented here.
The temporal variation of soil conditions in marshy environments is considered to be an important modulator of the dynamics of seed banks, and may favor the germination of certain species (Noe 2002). The overcoming of seed dormancy in M. flexuosa at the vereda edge (where soil water content is lower and more variable) is in agreement with the proposal that slight water stress conditions can stimulate recalcitrant seed germination (Eggers et al. 2007). in addition, oxygen availability, which is higher in drained soils, is considered important for palm seed germination (Pech y Aké et al. 2004; Ribeiro et al. 2012) and a determinant factor for the germination of M. flexuosa (Fernandes 2001). Interestingly, Resende et al. (2012) observed the proliferation of seedlings and young individuals of M. flexuosa in the edge environment after the passage of fire. We observed that seeds at the vereda edge germinated more rapidly and with greater percentage, and that all of the seedlings produced survived during the evaluation period. It is important to emphasize that long-term seed bank maintenance is unfeasible under those conditions due to the tendency for superficial drying of the soil and seed intolerance of desiccation. The maintenance of seedling banks may therefore be more feasible, since it would be possible for those plantlets to access to the subsurface water table after root development in that microenvironment. On the other hand, the interactions with the regional biota (an evaluation that was not an objective of the present work) may represent an important factor for the maintenance of seed banks of M. flexuosa, as observed by Johansson (2009) in the Amazon region. We observed that all of the ungerminated seeds at 365 days showed embryos that had suffered predation or deterioration. As insect and microorganism communities would be expected to vary among microenvironments, and seeds are very sensitive to predation/deterioration, the importance of site conditions on the successful maintenance of M. flexuosa seed banks is reinforced.
Studies carried out with palm trees Borassus aethiopum (Barot et al. 1999) Chamaedorea radicalis (Berry et al. 2007) and Brahea aculeata (Lopez-Toledo et al. 2013) revealed that palm species can interact with environmental heterogeneity or site factors, such as hebivory, in a complex way, which affects the pattern of spatial distribution of populations and the demography. In this context, the maintenance of both seed and seedlings banks of M. flexuosa in different microenvironments of the veredas seems to be a bet hedging strategy which favors the occupation and the resilience of the populations in a heterogenous environment. This possibility can be confirmed in the future, from results of studies on the ecophysiology of the seedlings (currently conducted by our research group) and broader studies to be performed with the ecology of populations of this species.
Conclusion
The unique strategy for the establishment of M. flexuosa seedlings (with recalcitrant seeds showing dormancy) is associated with the heterogeneity of the vereda environment. Seed viability maintenance is favored in soils that remain constantly moist, with the potential to form temporary seed banks, including in aquatic environments. Rapidly overcoming dormancy is associated with conditions observed at vereda edges (related to low soil moisture)—where seedling bank maintenance prevails.
References
Araújo GM, Barbosa AAA, Arantes AA, Amaral AF (2002) Composição florística de veredas no Município de Uberlândia. MG. Revista Brasil. Revista Brasil Bot 25:475–493
Barot SB, Gignoux J, Menaut J-C (1999) Demography of a savanna palm tree: predictions from comprehensive spatial pattern analyses. Ecology 80:1987–2005
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography and evolution of dormancy and germination. Academic Press, San Diego
Berjak P, Pammenter NW (2008) From Avicennia to Zizania: seed recalcitrance in perspective. Ann Bot 101:213–228
Berjak P, Pammenter NW (2013) Implications of the lack of desiccation tolerance in recalcitrant seeds. Front Plant Sci 4:478
Bernal R, Torres C, García N, Isaza C, Navarro J, Vallejo MI, Galeano G, Balslev H (2011) Palm management in South America. Bot Rev 77:607–646
Berry EJ, Gorchov DL, Endress BA, Stevens MHH (2008) Source-sink dynamics within a plant population: the impact of substrate and herbivory on palm demography. Popul Ecol 50:63–77
Bewley JD, Bradford KJ, Hilhorst HWM, Nonogaki H (2013) Seeds: physiology of development, germination and dormancy. Springer, New York
Brasil, Instituto Nacional de Metereologia (2016) Banco de Dados Meteorológicos para Ensino e Pesquisa http://www.inmet.gov.br/portal/index.php?r=bdmep/bdmep (Acesso em 27/12/2016)
Brasil, Ministério da Agricultura, Pecuária e Abastecimento (2009) Regras para análise de sementes. Mapa/ACS, Brasília
Cymerys M, Paula-Fernandes NM, Rigamonte-Azevedo NOC (2005) Buriti (Mauritia flexuosa L. f.). In: Shanley P, Medina G (eds) Fruit and useful plants in Amazonian life. Belém, Centro Internacional de Pesquisa Florestal-CIFOR, pp 175–182
Dalling JW, Davis AS, Schutte BJ, Arnold AE (2011) Seed survival in soil: interacting effects of predation, dormancy and the soil microbial community. J Ecol 99:89–95
Donagema GK, Campos DVB, Calderano SB, Teixeira WG, Viana JHM (2011) Manual de Métodos de Análise de Solo. Embrapa Solos, Rio de Janeiro
Dransfield J, Uhl NW, Asmussen CBA, Baker WJ, Harley MM, Lewis CE (2008) Genera Palmarum: the evolution and classification of palms. Kew Publishing, Richmond
Eggers S, Erdey D, Pammenter NW, Berjak P (2007) Storage and germination responses of recalcitrant seeds subjected to mild dehydration. In: Adkins S, Ashmore S, Navie SC (eds) Seeds: biology, development and ecology. CABI, Wallingford, pp 85–92
Endress BA, Horn CM, Gilmore MP (2013) Mauritia flexuosa palm swamps: composition, structure and implications for conservation and management. For Ecol Manag 302:346–353
Fernandes NMP (2001) Estratégias de produção de sementes e estabelecimento de plântulas de Mauritia flexuosa L. f. (Arecaceae) no Vale do Acre/Brasil. 205p. Thesis, Instituto Nacional de Pesquisas da Amazônia
Finch-Savage WE, Leubner-Metzger G (2006) Seed dormancy and the control of germination. New Phytol 171:501–523
Galeano A, Urrego LE, Sánchez M, Peñuela MC (2015) Environmental drivers for regeneration of Mauritia flexuosa L.f. in Colombian Amazonian swamp forest. Aquat Bot 123:47–53
Holm JA, Miller CJ, Crooper Jr WP (2008) Population dynamics of the dioecious Amazonian palm Mauritia flexuosa: simulation analysis of sustainable harvesting. Biotropica 40:550–558
Hong TD, Ellis RH (1996) A protocol to determine seed storage behaviour. In: Engels JMM, Toll J (eds) IPGRI Technical Bulletin Nº1. International Plant Genetic Resources Institute, Rome
Horn CM, Gilmore MP, Endress BA (2012) Ecological and socioeconomic factors influencing aguaje (Mauritia flexuosa) resource management in two indigenous communities in the Peruvian Amazon. For Ecol Manag 267:93–103
Johansson B (2009) Stay below water!—a strategy to avoid seed predators: seed survival and germination of Mauritia flexuosa in southeastern Peru. Dissertation, Linköpings Universitet
Koolen HHF, Silva FMA, Gozzo FC, Souza AQL, Souza ADL (2013) Antioxidant, antimicrobial activities and characterization of phenolic compounds from buriti (Mauritia flexuosa L. f.) by UPLC– ESI-MS/MS. Food Res Int 51:467–473
Long RL, Steadman KJ, Panetta FD, Adkins SW (2009) Soil type does not affect seed ageing when soil water potential and temperature are controlled. Plant Soil 320:131–140
Long RL, Gorecki MJ, Renton M, Scott JK, Colville L, Goggin DE, Commander LE, Westcott DA, Cherry H, Finch-Savage WE (2014) The ecophysiology of seed persistence: a mechanistic view of the journey to germination or demise. Biol Rev 90:31–59
Lopez-Toledo L, Portillo-Cruz Y, Pulido MT, Endress BA (2013) Seed dynamics of an endemic palm in a Northwestern Mexican tropical dry forest: implications for population spatial structure. Plant Ecol 214:1115–1125
Lorenzi H, Noblick LR, Kahn F, Ferreira E (2010) Flora Brasileira Lorenzi: Arecaceae (Palmeiras). Plantarum, Nova Odessa
Manzi M, Coomes OT (2009) Managing Amazonian palms for community use: a case of aguaje palm (Mauritia flexuosa) in Peru. For Ecol Manag 257:510–517
Martins RC, Filgueiras TS, Albuquerque UP (2012) Ethnobotany of Mauritia flexuosa (Arecaceae) in a Maroon Community in Central Brazil. Econ Bot 66:9–98
Noe GB (2002) Temporal variability matters: effects of constant vs. varying moisture and salinity on germination. Ecol Monogr 72:427–443
Orozco-Segovia A, Batis AI, Rojas-Aréchiga M, Mendoza A (2003) Seed biology of palms: a review. Palms 47:79–94
Pakeman RJ, Small JL, Torvell L (2012) Edaphic factors influence the longevity of seeds in the soil. Plant Ecol 213:57–65
Pech y Aké AE, Souza R, Maust B, Santamaria JM, Oropeza C (2004) Enhanced aerobic respiration improves in vitro coconut embryo germination and culture. In Vitro Cell Dev Biol Plant 40:90–94
Ramos MVV, Curi N, Motta PEF, Vitorino ACT, Ferreira MM, Silva MLN (2006) Veredas do triângulo mineiro: solos, água e uso. Ciênc Agrotec 30:283–293
Resende ILM, Santos FP, Chaves LJ, Nascimento JL (2012) Age structure of Mauritia flexuosa L. f. (Arecaceae) swamp populations in the central region of Goiás, Brazil. Rev Arv 36:103–112
Ribeiro AC, Guimarães PTG, Alvarez VVH (1999) Recomendações para o uso de corretivos e fertilizantes em Minas Gerais—5ª Aproximação. Editora UFV, Viçosa
Ribeiro LM, Oliveira DMT, Garcia QS (2012) Structural evaluations of zygotic embryos and seedlings of the macaw palm (Acrocomia aculeata, Arecaceae). Trees 26:851–863
Ribeiro LM, Garcia QS, Müller M, Munné-Bosch S (2015) Tissue-specific hormonal profiling during dormancy release in macaw palm seeds. Physiol Plant 153:627–642
Silva RS, Ribeiro LM, Mercadante-Simões MO, Nunes YRF, Lopes PSN (2014) Seed structure and germination in buriti (Mauritia flexuosa)—the swamp palm. Flora 209:674–685
Sousa RF, Nascimento JL, Fernandes EP, Leandro WM, Campos AB (2011) Matéria orgânica e textura do solo em veredas conservadas e antropizadas no bioma Cerrado. Rev Bras Eng Agric Ambient 15:861–866
Spera MRN, Cunha R, Teixeira JB (2001) Quebra de dormência, viabilidade conservação de sementes de buriti (Mauritia flexuosa). Pesqui Agropec Bras 36:1567–1572
Tweddle JC, Dickie JB, Baskin CC, Baskin JM (2003) Ecological aspects of seed desiccation sensitivity. J Ecol 91:294–304
Veloso VHS, Ribeiro LM, Mercadante-Simões MO, Nunes YRF (2016) Cytological aspects of recalcitrance in dormant seeds of Mauritia flexuosa (Arecaceae). Acta Physiol Plant 38:171
Virapongse A, Endress BA, Gilmore MP, Horn C, Romulo C (2017) Ecology, livelihoods, and management of the Mauritia flexuosa palm in South America. Glob Ecol Conserv 10:70–92
Acknowledgements
The authors thank the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for financial support to this project, and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for research productivity grants awarded to LMR and YRFN.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Anna R. Armitage.
Rights and permissions
About this article
Cite this article
Porto, K.C.N., Nunes, Y.R.F. & Ribeiro, L.M. The dynamics of recalcitrant seed banks of Mauritia flexuosa (Arecaceae) reveal adaptations to marsh microenvironments. Plant Ecol 219, 199–207 (2018). https://doi.org/10.1007/s11258-017-0788-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-017-0788-9