Abstract
Global warming is triggering some species to shift towards the poles or higher elevations, but spatial translocation is also influenced by land-use regime or intensity. The Himalayan climate is getting warmer and land use has changed, reducing in intensity in some areas. We estimated the upper species limit (USL) and tree limit of Abies spectabilis (D. Don) Spach and assessed whether these have changed over recent years. We hypothesise an upslope shift in response to enhanced temperature and changes in land-use intensity. Our four transects were located in treeline ecotones of two protected areas in Nepal, namely Manaslu Conservation Area (3 transects) and Gaurishankar Conservation Area (1 transect). Transects (20 m wide) ran from the USL of A. spectabilis down towards the treeline and beyond to the forest line. Length of each transect varied depending on local conditions. Co-ordinates, elevation, height and age of each A. spectabilis individual along the transects were recorded. We noted an upward shift of both the USL and the tree limit. The rate of shift was ca. 20 m per decade for the USL and 12 m per decade for the tree limit in the area of reduced land-use intensity and in the area with no change in land use, 5 m per decade for the USL, but almost nil for tree limit. The seedling density was higher below the treeline than above. Reduced intensity of land use was the dominant factor in upslope shift of A. spectabilis at both the USL and the tree limit.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The treeline in the mountains is one of the most fundamental ecological and conspicuous physiognomic boundaries along the elevation gradient (Harsch et al. 2009; Holtmeier 2009; Schickhoff 2005). The natural alpine treelines are the result of heat deficiency that adversely affects growth, regeneration and survival of trees, and they are therefore sensitive to climate change (Körner and Paulsen 2004).
The extent and limits of the treeline ecotone are easily confounded by different factors such as the presence of herbivores (Speed et al. 2012), forms of treeline (Harsch et al. 2009), land-use dynamics (Gehrig-Fasel et al. 2007), geomorphology (Leonelli et al. 2011; Resler 2006), moisture (Crimmins et al. 2011; Qiu 2015), as well as local temperature. Several recent studies document an upslope or poleward shift of species at the treeline and suggest that this is partly as a result of recent global warming (Bhatta and Vetaas 2016; e.g. Matteodo et al. 2013; Parmesan and Yohe 2003; Sturm et al. 2001; Telwala et al. 2013).
The impact of climate change is likely to be both species specific and site specific (e.g. Chen et al. 2011; Lenoir et al. 2008; Telwala et al. 2013), making it difficult to determine the role played by climate in any changed scenario. The upslope shift for different species ranged from 586 to 787 m on the Chimborazo volcano in Ecuador over a 210-year period (Morueta-Holme et al. 2015); in the Engadine valley of Swiss Alps, more than 100 m upslope shift has been reported during the last nine decades (Frei et al. 2010) and 87 percent of 124 endemic plants species have expanded their range in Sikkim Himalaya during the last ca. 150 years (Telwala et al. 2013). The treeline has shifted upwards in Barun valley in central Himalaya in the last 130 years (Chhetri and Cairns 2015), and A. spectabilis has shifted upslope in Manaslu region, central Himalaya in the last ca. 160 years (Gaire et al. 2014).
Not all species, however, exhibit a range expansion or upward shift (565 species, Grytnes et al. 2014; 92 species, Zhu et al. 2012). For instance, vascular plant species were found to be tracking their environmental niche by shifting downhill because of decreased water availability in California, although average temperature increased (Crimmins et al. 2011). Climatic warming increases water stress by increasing evapotranspiration. In such cases, temperature rises without an increase in precipitation counteracts the expected effects of warming. Picea crassifolia was found to be translocated downhill when suffering from such a phenomenon on the southern Tibetan Plateau (Qiu 2015).
Land-use change has also been identified as a driver in species-range dynamics. For example, plant species may expand into abandoned agropastoral areas due to reduced herbivory (Sharma et al. 2014b; Speed et al. 2012). Gehrig-Fasel et al. (2007) show that the treeline is moving upwards in the Swiss Alps due to land-use change, and in the French Maurienne valley, the upper elevational limit of Abies alba shifted upslope by more than 300 m in 50 years after land abandonment (Chauchard et al. 2010). Herbivores suppress the growth, and kill or uproot seedlings that are thriving in the harsh treeline ecotone (Hofgaard 1997). Thus the density of herbivores affects the species range (Grytnes et al. 2014). Grazing in the treeline ecotone impacts the position of the treeline (Holtmeier and Broll 2007; Speed et al. 2010) and plant species limit (Speed et al. 2012).
The range dynamics of a treeline species under different land-use regimes can be tested in the high mountain areas of Nepal that has experienced climatic warming of 0.6 °C per decade since 1970 s (between 1971 and 1994, Shrestha et al. 1999; between 1982 and 2006, Shrestha et al. 2012). We selected sites representative of two land-use regimes: one with a history of limited land use that has not changed as a control and the other with reduced land-use intensity (RLI) to study the upper-range dynamics of A. spectabilis (D. Don) Spach.
The aims of this study were (i) to compare the upper species limit and the tree-limit dynamics in two contrasting land-use scenarios and (ii) to analyse regeneration within the treeline ecotone.
Methodology
Study areas
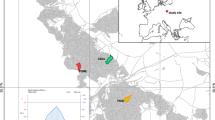
This study was carried out in two protected areas of Nepal, namely Manaslu Conservation Area (hereafter MCA; 84°39.50′E–84°41.81′E and 28°33.86′N–28°34.31′N) and Gaurishankar Conservation Area (henceforth GCA; 86°22.548′E–86°22.494′E and 27°53.865′N–27o53.914′N) (Fig. 1).
Climate
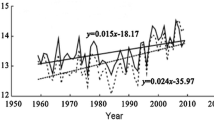
We used available climate data (1978–2009) from the nearest meteorological stations to the study areas. For the MCA, we used temperature data from Chame (2680 m a.s.l.; 28°33′N and 84°14′E; ca. 40 km west) and precipitation data from Larke Samdo (3650 m a.s.l.; 28°40′N and 84°37′E; ca. 8 km northwest). For the GCA, we used data for both precipitation and temperature from Jiri (2003 m a.s.l.; 27°38′N and 86o14′E; ca. 34 km south). Missing temperature and precipitation data were assigned the respective mean monthly values. Temperature data were extrapolated to treeline elevations assuming a mean annual lapse rate of 0.52 °C per 100 m elevation (Kattel et al. 2013). Temperature has been increasing at a rate of ca. 0.19 °C per decade in both the areas (p < 0.05) between 1978 and 2009. This warming rate is slower than the average warming in Nepal (between 1971 and 1994) (Shrestha et al. 1999) and in the Hindu Kush Himalayan region (between 1982 and 2006) (Shrestha et al. 2012).
Land use
In both our study areas, transhumance is still practiced, but in the GCA it does not affect the sampling area (Schickhoff et al. 2014). During summer, cattle herds are taken to the highland, while during autumn they are brought back to the lowland. In the MCA, land-use signs such as lopping, cut stumps, grazing, trampling and droppings were noticed. In the GCA, the practice of collecting dead tree branches and lopping signs were observed mainly in the dense forest at lower elevations. It is a near-natural or climatic treeline in this area (Müller et al. 2015; Schickhoff et al. 2014).
Vegetation
A. spectabilis is a coniferous tree species growing in cool temperate and subalpine regions in the central and western Himalayas. It forms monodominant forest and reaches a height of 50 m and trunk diameter of more than 1.5 m. Furrowed branchlets are densely leafy with yellowish grey, brown or reddish brown colour (eFloras 2008).
In the MCA, the lower part of the treeline ecotone is dominated by A. spectabilis and the upper part by Betula utilis and Rhododendron scrub. Juniperus recurva, Rhododendron companulatum, R. anthopogon, R. lepidotum, Delphinium spp. and Berberis spp. were also found in the forest of this area. In the treeline ecotone in the GCA, the lower part is dominated by A. spectabilis and the upper part by Rhododendron spp., Betula utilis, Rhododendron campanulatum, R. anthopogon, R. lepidotum, Rosa sericea, Potentilla fruticosa and Berberis spp. are found as co-occurring species.
Sampling
To be able to partially disentangle the effects of land-use intensity and climate change, we selected two study areas within the protected areas to circumvent contemporary high human disturbances. Then we filtered the areas based on the accessibility of the treeline ecotone.
One control transect was located in an area of negligible land use (transect GCA-1, control) and three transects (MCA-1, MCA-2, MCA-3) were located in an area which had undergone RLI. In all areas, we first looked for the uppermost individual (= Upper Species Limit, hereafter USL) in the treeline ecotone. Our horizontal search distance ranged from a few hundred metres to an 1800 m stretch of landscape. Transects were then drawn on the basis of the USL of A. spectabilis in a delimited area.
A transect of 20 m width running downhill towards the treeline [highest elevation of trees at least 2 m high in a patch comprising at least three individuals (Körner 2003)] was surveyed. From the treeline, a second half of the transect continued downhill into the forest. The second half of transect should ideally be as long as the first, but in two of the cases this was not possible due to topographical constraints.
Altogether four transects of variable lengths (see Table 1) were sampled. In each transect, trees (height ≥2 m), saplings (15–199 cm) and seedlings (≤15 cm) of A. spectabilis were recorded (Hofgaard and Rees 2008). Each tree was cored at the base of the tree using an increment borer (Haglöf, Sweden) to determine its age by counting the rings. The age of a sapling or seedling was estimated by the branch whorl count method (Camarero and Gutierrez 2004; Ninot et al. 2008). The height of each individual was measured. Elevation (±1 m accuracy, using a digital altimeter (Silva)), latitude and longitude (Garmin GPS) were recorded for all individuals.
Analysis
The USL shift rate (metre per decade) was calculated based on the elevation, and the age of the uppermost individual and the oldest individual occurring within the transect is given in the following equation:
where EUI is the elevation of the uppermost individual; EOI the elevation of the oldest individual; AOI the age of the oldest individual and AUI is the age of the uppermost individual (modified method of Gamache and Payette 2005).
The tree-limit shift rate [the upper limit of woody plants ≥2 m tall (Wardle 1974)] (metre per decade) was calculated using the following equation:
where EUT is the elevation of the uppermost tree and AUT is the age of the uppermost tree.
Average shifting rate of ten individuals (metre per decade) was estimated using the following equation:
where AE10UI is the average elevation of 10 uppermost individuals; AE10OI the average elevation of 10 oldest individuals; AA10OI the average age of 10 oldest individuals and AA10UI is the average age of 10 uppermost elevational individuals.
The regeneration of A. spectabilis was assessed with a histogram for different age groups expressed as individuals per hectare. We compared seedlings and saplings above and below the treeline in all transects in terms of density per hectare.
Results
Species limit shift
We found that the USL ranges from ca. 4000 to 3750 m a.s.l. (Table 1; Fig. 2) in our transects. The length of the transects varied from 102 to 570 m, and the number of individuals per unit area also varied between the transects. The average USL and tree-limit shift rates in the RLI area were ca. 20 and 12 m per decade, respectively. In the control area with a climatically controlled treeline, the rate of USL shift was much less, only ca. 5 m per decade, and effectively nil for the tree-limit shift. The average of ten individuals had a shift rate in the RLI area of about 26 m per decade and in the control area the shift was only ca. 2 m per decade (Table 2).
Scatter plots of recruitment year and elevation for seedlings, saplings and trees in each of the four transects [A MCA-1, B MCA-2, C MCA-3 (reduced land-use intensity), D GCA-1 (no land-use change)]. The dashed lines show the upper species limit. The arrow indicates the position of the current treeline position and the vertical lines mark 1950 for comparison before and after 1950. Note: For better illustration of the plots, we hide five points in subplot C and two points in subplot D before 1900 AD
Regeneration and density in treeline ecotone
The age–frequency histogram is left skewed or inverse-J shaped (Fig. 3), except in one transect in Manaslu (MCA-1), implying a high density of seedlings and saplings. The density of seedlings and saplings was higher below the treeline (Fig. 4). The seedling density above the treeline was ca. 83 and ca. 19 individuals ha−1 in the RLI and control areas, respectively, while below the treeline it was ca. 301 and 800 individuals ha−1. Similarly, the sapling density above the treeline was ca. 110 and 76 individuals ha−1, while below the treeline it was ca. 245 and 150 individuals ha−1 in the RLI and control areas, respectively.
Discussion
The treeline in the eastern location (GCA) is at a higher elevation than in the western part (MCA), which matches the geographical trend in the Himalaya (Mani 1974; Miehe et al. 2015). All four treelines almost coincide with the 10 °C isotherm of the warmest month as noted by Körner (2003) and Holtmeier (2009).
We find that the USL has ascended between 63 and 327 m at the different sites, consistent with other studies in the Himalaya (Gaire et al. 2014; cf. Telwala et al. 2013). The shift rate was calculated based on two individuals in each transect and may differ from the average for the whole landscape. The USL is not static but very dynamic due to the uncertainty of lethal events at an early life stage. It records extreme events experienced by each individual rather than the average response to climate change (Kreyling et al. 2012). In an attempt to compensate for this, we used the mean measurements of 10 individuals to provide a more general expression. The mean shift rate of 10 individuals was ca. 26 m per decade in the RLI area, while it was nearly static in the control area (ca. 2 m per decade). This indicates no significant change because two metres is within the error in the estimation of elevation.
We find that the species population has expanded in the RLI area, a finding confirmed by the local people. Species are colonising new areas in the mountains and moving upslope. In the control area, however, there were only a few individuals above the treeline. The uppermost individual was recruited in 1990 and was isolated from the rest of the population. There were no other individuals of A. spectabilis until close to the treeline. A similar pattern was observed in the Barun valley in central Himalaya for the same species (Chhetri and Cairns 2015). Establishment of new seedlings of A. spectabilis in the control area may be inhibited by the monotonous thick Rhododendron scrub above the treeline (Baker and Van Lear 1998), and there may be an allelopathic effect (Fisher 1980; Nilsen et al. 1999) and light control (Clinton and Vose 1996) from the Rhododendron. The ground was almost devoid of herbaceous vegetation, indicating that it would not be easy for species to establish here.
The regeneration study clearly shows that there were many young individuals along our transects. We recorded, on an average, higher numbers of saplings along the RLI transects than along the control transect, suggesting that some level of disturbance provided opportunities for sapling growth (Vetaas 1997). The inverse-J-shaped age–frequency histograms (Fig. 3) depict that A. spectabilis was maintaining a sustainable population in the treeline ecotone. The density of seedlings was, as expected, higher than the density of saplings. The MCA-3 transect went through very frequent anthropogenic disturbances and only few individuals had opportunity to survive to mature age. The intense disturbances are also depicted by lower density of saplings and trees in this transect. For all our transects, we see that the densities of seedlings and saplings were higher below the treeline than above. This is in line with other studies (Ninot et al. 2008; Shrestha et al. 2007), although it contrasts with findings from Bell et al. (2014) who note an upshift of range for juveniles and from Shrestha et al. (2014) who found more seedlings of A. spectabilis above the treeline in Laurebinayak area in Langtang National Park, Nepal. There could be many factors to cause these patterns, such as orography, soil moisture, pH, community interaction and chance events; some are likely to be quite site specific.
Our comparison of the USL and tree-limit shift between a control area and an area of reduced land-use intensity shows that a greater shift rate occurred in the area with reduced land use. The type of land-use practice has not changed, i.e. transhumance, husbandry, agroforestry and grazing, but the intensity has reduced substantially due to outmigration of local people (CBS 2014; Bal Kumar 2003; Sharma et al. 2014a). In contrast, there has been no discernible shift of the tree limit in the control area (a change of 1 m is less than the error in the estimation of elevation). There has been a slight shift in the USL of ca. 5 m per decade, but this estimate is based on only two individuals. Reduced land-use intensity is therefore the most likely driver of range expansion in the MCA. The small increase in average atmospheric temperature has not yet impacted species distribution in this ecotone, although it has been noted in other regions (Matteodo et al. 2013; Parmesan and Yohe 2003; Telwala et al. 2013). However, reduced intensity of land use (Müller et al. 2015) may also have synergistic effects with increased warming in the RLI area (Morueta-Holme et al. 2015).
Comparing the trends of the USL of the four transects (Fig. 2), it can be seen that the limit has increased at all sites. In the eastern Himalaya, Telwala et al. (2013) reported 87 % of endemic plants (out of 124 species) shifted upslope in response to a temperature rise in mean summer temperature of 0.76 and 3.65 °C in mean winter temperature. Over the last nine decades, an average upslope shift of 145 m was reported in Engadine valley of Swiss Alps (Frei et al. 2010) and in the last half century a 70 m elevational shift in the Montseny Mountains (Catalonia, NE Spain) was reported (Penuelas et al. 2007) as a response to global warming.
In the last 50 years the local mean annual temperature has increased by about 1 °C based on data from the nearest meteorological station. To track this warming, species are predicted to need to move upslope by about 192 m, equivalent to 38 m per decade. The analysis of our species demonstrates that it is lagging behind the recent warming. Projected climatic warming for 2081–2100 is 1.0–3.7 °C higher than the 1986–2005 normal (IPCC 2014), so a shift of between about 24–90 m per decade would be required for species to maintain their current climatic niche. However, a species’ ability to respond is affected by local geomorphic processes (Macias-Fauria and Johnson 2013), grazing (Speed et al. 2012) and community interactions such as dense grassland (Rehm and Feeley 2015). Based on the predicted required shifts, it seems that the species will not be able to track the projected warming which may cause them to lose their potential habitat (Engler et al. 2011; Svenning et al. 2008) and reduce their population size (Feeley and Silman 2010).
Conclusions
In an area of reduced land-use intensity, we document the growth of A. spectabilis at higher elevations than the estimated upper species limit and tree limit for earlier years. The slight increase in mean annual temperature has not yet prompted a noticeable response in the species. The rate of upward shift is site specific and appears to increase after 1950.
References
Baker TT, Van Lear DH (1998) Relations between density of rhododendron thickets and diversity of riparian forests. Forest Ecol Manag 109:21–32. doi:10.1016/S0378-1127(98)00259-X
Bal Kumar KC (2003) International migration in Nepal. In: Population monograph of Nepal. Central Bureau of Statistics, National Planning Commission, Kathmandu
Bell DM, Bradford JB, Lauenroth WK (2014) Early indicators of change: divergent climate envelopes between tree life stages imply range shifts in the western United States. Glob Ecol Biogeogr 23:168–180
Bhatta KP, Vetaas OR (2016) Does tree canopy closure moderate the effect of climate warming on plant species composition of temperate Himalayan oak forest? J Veg Sci. doi:10.1111/jvs.12423
Camarero JJ, Gutierrez E (2004) Pace and pattern of recent treeline dynamics: response of ecotones to climatic variability in the Spanish Pyrenees. Clim Change 63:181–200. doi:10.1023/B:Clim.0000018507.71343.46
CBS (2014) National Population and Housing Census 2011: social characteristics tables. Part VI, vol 5. Central Bureau of Statistics, National Planning Commission, GoN, Kathmandu
Chauchard S, Beilhe F, Denis N, Carcaillet C (2010) An increase in the upper tree-limit of silver fir (Abies alba Mill.) in the Alps since the mid-20th century: a land-use change phenomenon. For Ecol Manag 259:1406–1415
Chen IC, Hill JK, Ohlemuller R, Roy DB, Thomas CD (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024–1026. doi:10.1126/science.1206432
Chhetri PK, Cairns DM (2015) Contemporary and historic population structure of Abies spectabilis at treeline in Barun valley, eastern Nepal Himalaya. J Mt Sci 12:558–570
Clinton BD, Vose JM (1996) Effects of Rhododendron maximum L. on Acer rubrum L. seedling establishment. Castanea 61:38–45
Crimmins SM, Dobrowski SZ, Greenberg JA, Abatzoglou JT, Mynsberge AR (2011) Changes in climatic water balance drive downhill shifts in plant species’ optimum elevations. Science 331:324–327
eFloras (2008) Missouri Botanical Garden, St. Louis, MO & Harvard University Herbaria, Cambridge, MA. www.efloras.org. Accessed 18 May 2016
Engler R et al (2011) 21st century climate change threatens mountain flora unequally across Europe. Glob Change Biol 17:2330–2341
Feeley KJ, Silman MR (2010) Land-use and climate change effects on population size and extinction risk of Andean plants. Glob Change Biol 16:3215–3222
Fisher RF (1980) Allelopathy: a potential cause of regeneration failure. J For 78:346–350
Frei E, Bodin J, Walther GR (2010) Plant species’ range shifts in mountainous areas-all uphill from here? Bot Helv 120:117–128. doi:10.1007/s00035-010-0076-y
Gaire NP, Koirala M, Bhuju DR, Borgaonkar HP (2014) Treeline dynamics with climate change at the central Nepal Himalaya. Clim Past 10:1277–1290. doi:10.5194/cp-10-1277-2014
Gamache I, Payette S (2005) Latitudinal response of subarctic tree lines to recent climate change in eastern Canada. J Biogeogr 32:849–862. doi:10.1111/j.1365-2699.2004.01182.x
Gehrig-Fasel J, Guisan A, Zimmermann NE (2007) Tree line shifts in the Swiss Alps: climate change or land abandonment? J Veg Sci 18:571–582
Grytnes J-A et al (2014) Identifying the driving factors behind observed elevational range shifts on European mountains. Glob Ecol Biogeogr 23:876–884. doi:10.1111/geb.12170
Harsch MA, Hulme PE, McGlone MS, Duncan RP (2009) Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol Lett 12:1040–1049. doi:10.1111/j.1461-0248.2009.01355.x
Hofgaard A (1997) Inter-relationships between treeline position, species diversity, land use and climate change in the central Scandes Mountains of Norway. Glob Ecol Biogeogr Lett 6:419–429
Hofgaard A, Rees G (2008) Common protocols for field measurements and handling of collected materials. PPS Arctic Group, Tromsø
Holtmeier F-K (2009) Mountain timberlines: ecology, patchiness, and dynamics, vol 36. Springer, New York
Holtmeier F-K, Broll G (2007) Treeline advance–driving processes and adverse factors. Landsc Online 1:1–33
IPCC (2014) Climate change 2014: synthesis report. Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. Intergovernmental panel on climate change, Geneva
Kattel D, Yao T, Yang K, Tian L, Yang G, Joswiak D (2013) Temperature lapse rate in complex mountain terrain on the southern slope of the central Himalayas. Theor Appl Climatol 113:671–682
Körner C (2003) Alpine plant life: functional plant ecology of high mountain ecosystems. Springer, Berlin
Körner C, Paulsen J (2004) A world-wide study of high altitude treeline temperatures. J Biogeogr 31:713–732
Kreyling J et al (2012) Cold hardiness of Pinus nigra Arnold as influenced by geographic origin, warming, and extreme summer drought. Environ Exp Bot 78:99–108
Lenoir J, Gegout JC, Marquet PA, de Ruffray P, Brisse H (2008) A significant upward shift in plant species optimum elevation during the 20th century. Science 320:1768–1771. doi:10.1126/science.1156831
Leonelli G, Pelfini M, di Cella UM, Garavaglia V (2011) Climate warming and the recent treeline shift in the European Alps: the role of geomorphological factors in high-altitude sites. Ambio 40:264–273
Macias-Fauria M, Johnson EA (2013) Warming-induced upslope advance of subalpine forest is severely limited by geomorphic processes. Proc Natl Acad Sci 110:8117–8122
Mani M (1974) Ecology and biogeography in India. Dr. W. Junk B.V., The Hague. doi:10.1007/978-94-010-2331-3
Matteodo M, Wipf S, Stöckli V, Rixen C, Vittoz P (2013) Elevation gradient of successful plant traits for colonizing alpine summits under climate change. Environ Res Lett 8:024043
Miehe G, Pendry CA, Chaudhary RP (2015) Nepla: an introduction to the natural history, ecology and human environment of the Himalayas. Royal Botanic Garden Edinburgh, Edinburgh
Morueta-Holme N, Engemann K, Sandoval-Acuña P, Jonas JD, Segnitz RM, Svenning J-C (2015) Strong upslope shifts in Chimborazo’s vegetation over two centuries since Humboldt. In: Proceedings of the National Academy of Sciences: 201509938
Müller M, Böhner J, Chaudhary R, Drollinger S, Schickhoff U, Scholten T (2015) How do soil properties affect alpine treelines? General principles in a global perspective and novel findings from Rolwaling Himal, Nepal. Prog Phys Geogr
Nilsen ET, Walker JF, Miller OK, Semones SW, Lei TT, Clinton BD (1999) Inhibition of seedling survival under Rhododendron maximum (Ericaceae): could allelopathy be a cause? Am J Bot 86:1597–1605
Ninot JM, Batllori E, Carrillo E, Carreras J, Ferre A, Gutierrez E (2008) Timberline structure and limited tree recruitment in the Catalan Pyrenees. Plant Ecol Divers 1:47–57. doi:10.1080/17550870802260764
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42. doi:10.1038/Nature01286
Penuelas J, Ogaya R, Boada M, Jump AS (2007) Migration, invasion and decline: changes in recruitment and forest structure in a warming-linked shift of European beech forest in Catalonia (NE Spain). Ecography 30:829–837
Qiu J (2015) High-altitude forests in the Himalayas harder hit by droughts. Why are plateau and mountain timberlines in Asia shifting downslope, despite global warming?, vol 2015. Scientific American Division of Nature America Inc, Berlin
Rehm EM, Feeley KJ (2015) The inability of tropical cloud forest species to invade grasslands above treeline during climate change: potential explanations and consequences. Ecography 38:1167–1175
Resler LM (2006) Geomorphic controls of spatial pattern and process at alpine treeline. Prof Geogr 58:124–138. doi:10.1111/j.1467-9272.2006.00520.x
Schickhoff U (2005) The upper timberline in the Himalayas, Hindu Kush and Karakorum: a review of geographical and ecological aspects. Springer Verlag, Berlin-Heidelberg-New York
Schickhoff U et al (2014) Do Himalayan treelines respond to recent climate change? An evaluation of sensitivity indicators. Earth Syst Dyn Discuss 5:1407–1461
Sharma LN, Vetaas OR, Chaudhary RP, Måren IE (2014a) Ecological consequences of land use change: forest structure and regeneration across the forest-grassland ecotone in mountain pastures in Nepal. J Mt Sci 11:838–849
Sharma LN, Vetaas OR, Chaudhary RP, Måren IE (2014b) Pastoral abandonment, shrub proliferation and landscape changes: a case study from Gorkha, Nepal. Landsc Res 39:53–69
Shrestha AB, Wake CP, Mayewski PA, Dibb JE (1999) Maximum temperature trends in the Himalaya and its vicinity: an analysis based on temperature records from Nepal for the period 1971-94. J Clim 12:2775–2786. doi:10.1175/1520-0442(1999)012<2775:Mttith>2.0.Co;2
Shrestha BB, Ghimire B, Lekhak HD, Jha PK (2007) Regeneration of treeline birch (Betula utilis D. Don) forest in a trans-Himalayan dry valley in central Nepal. Mt Res Dev 27:259–267. doi:10.1659/mrdd.0784
Shrestha UB, Gautam S, Bawa KS (2012) Widespread climate change in the Himalayas and associated changes in local ecosystems. PLoS One 7:e36741. doi:10.1371/journal.pone.0036741
Shrestha KB, Hofgaard A, Vandvik V (2014) Recent treeline dynamics are similar between dry and mesic areas of Nepal, central Himalaya. J Plant Ecol: rtu035
Speed JD, Austrheim G, Hester AJ, Mysterud A (2010) Experimental evidence for herbivore limitation of the treeline. Ecology 91:3414–3420
Speed JDM, Austrheim G, Hester AJ, Mysterud A (2012) Elevational advance of alpine plant communities is buffered by herbivory. J Veg Sci 23:617–625. doi:10.1111/j.1654-1103.2012.01391.x
Sturm M, Racine C, Tape K (2001) Climate change—increasing shrub abundance in the Arctic. Nature 411:546–547. doi:10.1038/35079180
Svenning JC, Normand S, Skov F (2008) Postglacial dispersal limitation of widespread forest plant species in nemoral Europe. Ecography 31:316–326
Telwala Y, Brook BW, Manish K, Pandit MK (2013) Climate-induced elevational range shifts and increase in plant species richness in a Himalayan biodiversity epicentre. PLoS One 8:e57103. doi:10.1371/journal.pone.0057103
Vetaas OR (1997) The effect of canopy disturbance on species richness in a central Himalayan oak forest. Plant Ecol 132:29–38
Wardle P (1974) Alpine timberlines. Arctic and alpine environments. Methuen, London, pp 371–402
Zhu K, Woodall CW, Clark JS (2012) Failure to migrate: lack of tree range expansion in response to climate change. Glob Change Biol 18:1042–1052
Acknowledgments
Our sincere thanks are due to Lila Nath Sharma, Kuber Prasad Bhatta, Man Kumar Dhamala, Kumar Prasad Mainali, Khem Raj Bhattarai, Raj Kumar Gautam, Mandhata Acharya, Narayan Prasad Gaire and Rosy Makaju who helped with suggestions, comments and field sampling. We acknowledge IDEA WILD, the Central Department of Botany (Tribhuvan University), the Department of National Parks and Wildlife, the National Trust for Nature Conservation, Nepal Academy of Science and Technology, Nepal Climate Change Knowledge Management Center (NAST-CDKN/NCCKMC), Norad’s Programme for Master Studies and the University of Bergen for their support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Dr. Jan Wunder.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Suwal, M.K., Shrestha, K.B., Guragain, L. et al. Land-use change under a warming climate facilitated upslope expansion of Himalayan silver fir (Abies spectabilis (D. Don) Spach). Plant Ecol 217, 993–1002 (2016). https://doi.org/10.1007/s11258-016-0624-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-016-0624-7