Abstract
Purpose
In order to describe epidemiological and pathological features of penile cancer in a high-risk area of Brazil.
Methods
We reviewed the experience (378 patients from 1997 to 2007) of Hospital Aristides Maltez from Salvador, Bahia—the main institution in the state which provides oncologic treatment for penile cancer in the public health system.
Results
The present series showed a high rate (17 %) of patients less than 40 years at the time of diagnosis. Cancer-specific death rate in this age group was 19 % (in contrast to 11 and 13 % in the 41–60 and >60 age groups). Squamous cell carcinomas in younger patients were also more likely to exhibit infiltrative growth pattern, perineural invasion, and recurrence.
Conclusion
Regardless of tumor subtypes, penile carcinoma in Northeastern Brazil had more aggressive features and behavior when presented at younger age. This observation should be confirmed in other large series from endemic areas of penile cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Penile cancer is a locally aggressive disease which leads to mutilating treatments. It is more common in developing countries and shows progressive reduction in incidence in developed regions—as a consequence, it can be considered a neglected disease [1–4]. Northeast and North of Brazil, the two less developed regions of the country, concentrates most cases [5]. The Brazilian Society of Urology estimates that 42 and 40 % of all cases in Brazil occur in Northeast and North regions, respectively (unpublished data). The Brazilian Ministry of Health estimates a thousand penile amputations per year with 363 annual deaths due to penile cancer (year 2010). The state of Bahia, in the Northeast region of Brazil, has one of the highest incidences of penile cancer in Brazil. It is a general assumption between physicians that the disease is heterogeneous on clinical evolution and outcome. In order to describe epidemiological and pathological features of penile cancer in a high-risk area of Brazil, we reviewed cases from Hospital Aristides Maltez at Salvador, Bahia—the main institution in the state which provides oncologic treatment for penile cancer in the public health system.
Materials and methods
Patients and study design
This is a cross-sectional study. It was based in Hospital Aristides Maltez: the main institution that practice oncologic assistance for penile cancer for 351 municipalities of the state of Bahia. All consecutive patients who sought medical assistance on the division of Urology from January 1997 to December 2007 with a diagnosis of penile squamous cell carcinoma were eligible to be included in the study. Exclusion criteria were as follows: primary tumors surgically resected in other institutions, no tumor staging information on medical records, no available histological slides for review, and available histological slides that could not be used to confirm diagnosis due to technical issues. Clinical follow-up was recorded, and patients were grouped in the following groups: alive free of disease, alive with disease, dead in consequence of the disease or complications of treatment, dead due to other cases, and lost from follow-up. The study was approved by the Research Ethics Committee of Hospital Aristides Maltez located in Salvador, Brazil (224/08).
Tumor subtyping and grading
Tumor slides were reviewed and subtypes were classified or reclassified based on the World Health Classification of Tumors (2004) [6]. Tumor grading was performed as follows: Grade 1 is an extremely well-differentiated carcinoma, with a minimal deviation from the morphology of normal/hyperplastic squamous epithelium; Grade 2 tumors show disorganized growth as compared to grade 1 lesions, higher nuclear-to-cytoplasmic ratio, evident mitoses, and, although present, less prominent keratinization; Grade 3 are tumors showing any proportion of anaplastic cells, identified as solid sheets or irregular small aggregates, cords or nests of cells with little or no keratinization, high nuclear-to-cytoplasmic ratio, thick nuclear membranes, nuclear pleomorphism, clumped chromatin, prominent nucleoli, and numerous mitosis [7, 8]. Cytological features (koilocytes) suggestive of HPV infection were evaluated. No method based on direct detection of viral DNA was employed.
Statistical analyses
Statistical analyses were performed using the SPSS 17.0 software package. Categorical data were compared by a Chi-square test for trend, and the numerical data were compared using a Student’s t test. Kaplan–Meier curves were established to compare survival from different groups. Curves were compared using log-rank (Mantel–Cox) test. A p value <0.05 was considered significant.
Results
From January 1997 to December 2007, 378 patients with diagnosis of penile cancer were referred to the Division of Urology of the Hospital Aristides Maltez. Lack of available data leads to exclusion of 30 cases. Among 348 cases of penile cancer, 32 were only biopsied in the referral center. Local excision/postectomy was performed in 28 (8 %), partial penectomy in 262 (75 %), total penectomy in 8 (2 %), and emasculation in 18 (5 %) cases. Seventy-seven patients underwent lymphadenectomy.
Thirty-four patients were excluded for different reasons (e.g., only biopsies available), remaining 314 patients for further clinicopathological analysis.
The mean age was 57 ± 16 years (range 25–91), and 53 (17 %) patients were less than 40 years by the time of diagnosis. There was a low rate of formal learning: 42 % had incomplete primary/elementary education, and 45 % were illiterate. Poor quality of medical records precluded analysis of sexual behavior. Cigarette smoking was reported by 137 (44 %) patients and alcohol consumption by 127 (40 %). Forty-one patients (13 %) had phimosis, and 29 (9 %) reported previous circumcision. These characteristics of patients enrolled in the study are summarized in Table 1. The mean interval between the onset of symptoms and seek for medical assistance was 9.8 ± 11 months.
Tumors had a mean size of 4.2 cm (±2.0 cm) ranging from 0.5 to 13.0 cm. The glans was the most common primary site of involvement (250 cases or 80 %). Most cases (183 or 58 %) showed intermediate histological grade, and 210 (67 %) had invasion deeper than 5 mm. There was a decreasing frequency of involvement from corpus spongiosum (243 or 77 %), corpus cavernosum (121 or 39 %) to distal urethra (47 or 15 %). Evaluation of tumor front revealed that in 168 (54 %), the growth pattern was infiltrative. Nodal and distant metastasis were present in 98 (31 %) and 7 (2 %) of all patients, respectively. Cytological features suggestive of HPV infection were observed in 239 (76 %) of all cases. Based on WHO classification, the frequency of squamous cell carcinomas subtypes was 241 (77 %) for usual, 26 (8 %) for warty, 11 (4 %) basaloid, 4 (1 %) papillary, 2 (1 %) verruciform, 1 (0.3 %) sarcomatoid, and 29 (9 %) of mixed subtypes. Morphologic features of this series are detailed in Table 2.
Clinical follow-up detected enlargement of inguinal lymph nodes in 109 patients (35 %): 98 cases (31 %) had clinical suspicious of metastatic disease and 77 (25 %) were submitted to lymphadenectomy in the institution. Metastases were confirmed in 66 cases (86 %), while in 11 cases (14 %), removed lymph nodes showed only reactive features.
Twenty-seven patients underwent chemotherapy, and 43 were treated with radiotherapy. Forty patients experienced local recurrence. The mean follow-up for the 314 patients was 22.4 ± 26.5 months. A total of 42 cases (13 %) were lost during follow-up. At the end of the study, 66 patients (21 %) were alive with persistent disease, while 42 (13 %) had died due to penile carcinoma. Clinical follow-up of 38 patients with more than 5 years of follow-up gave the following results: alive free of disease (n = 28), alive with disease (n = 5), dead in consequence of the disease or complications of treatment (n = 4), and dead due to other cases (n = 1).
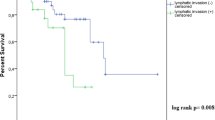
We were interested on how disease’s presentation and progression differed between younger (<40 years old) and older (≥40 years old) patients. Younger patients had higher frequency of some morphological features of worse prognosis such as infiltrative growth pattern (70 vs 50 %) and perineural invasion (26 vs 13 %) (p < 0.05). Younger patients experienced disease recurrence in 45 % of all cases with only 25 % of older patients showing recurrent disease at follow-up (p < 0.05). The frequency of death due to penile carcinoma was 19 % for patients <40 years old, 11 % for the age group between 40 and 60 years, and 13 % for older (>60 years old patients) (p < 0.05). Comparison of survival curves between patients less than 40 years and the age group between 40 and 60 years is shown in Fig. 1 (Mantel–Cox: 0.025). For the same age groups, the frequency of lymph node metastasis was 49, 34, and 21 %, respectively.
Survival curves showed no statistical difference between different carcinoma subtypes. At the 60th month of follow-up, disease-specific survival rate was 86 % for the usual subtype, 88 % for warty, 88 % for mixed subtypes, and 66 % for basaloid.
Discussion
A previous study, based on data from the same reference center for treatment of penile carcinoma in the state of Bahia, described 811 cases (803 cases of squamous cell carcinoma) from the period of 1952 to 1983 [9]. Importantly, 170 patients (21 %) were 40-year-old or younger, a frequency which is similar to the described herein (17 %). Of all patients, 20 % had already inguinal lymph node enlargement at the time of diagnosis. In this previous report, 65 % of affected patients had concurrent phimosis and none reported circumcision in childhood (125 or 15 % reported the procedure during adolescence and adulthood). In the present study, 41 (13 %) patients had phimosis and 29 (9 %) reported previous circumcision. Among the 811 cases from 1952 to 1983, the intervals between onset of symptoms and seeking medical assistance were less than 3 months, between 3 and 9 months, and longer than 9 months were 16, 43, and 41 %, respectively. In the present series, the mean interval between disease onset and medical assistance was 9.8 ± 11 months. No follow-up was reported in that previous study.
Despite economic growth, social development during the last decades in Brazil, particularly in Northeast region of Brazil and state of Bahia, the sociodemographic characteristics of patients with penile carcinoma seem to be unchanged. The illiteracy rate in Brazil dropped from 51 % in 1950 to 10 % in 2010 [10]; however, 45 % of all patients in this study were illiterate, and other 45 % had incomplete elementary education. Northeastern Brazil also experienced a sixfold rise in gross domestic product per capita from 1950 to 2010 [11]. In contrast, the present study covering the period from 1997 to 2007, in addition to the previous one covering the period from 1952 to 1983, did not detect a trend for reduction in diagnosis of new cases of penile cancer in this reference center. Poor formal education is a well-known factor associated with penile cancer, and recent studies from different settings show that the sociodemographic characteristics of affected patients seem to persist [2, 12] albeit the incidence of disease decreases in developed countries [1–4].
In the present series, the presence of cytological features suggestive of HPV infection had no impact on prognosis. Some previous studies are discordant on the possible impact of HPV infection on prognosis [13–15]. Survival curves also did not differ between different tumor subtypes.
The mean age in this case series, 57 years, is comparable with most series in the literature. However, the reported series from Brazil show a higher frequency of younger individuals (<45 years at the moment of diagnosis) [5, 16, 17]. The present series showed that 17 % of patients were less than 40 years at the time of diagnosis, and cancer-specific death rate in this age group was 19 % (in contrast to 11 and 13 % in the 41–60 and >60 age groups). Squamous cell carcinomas in younger patients were more likely to exhibit infiltrative growth pattern, perineural invasion, and recurrence. Distinct age groups did not differ on the frequency of specific subtypes of squamous cell carcinomas. The current literature and proposed prognostic indexes do not include age at cancer diagnosis as important information of prognostic value [18–21]. Recently, a multicenter study reported that zoophilia (sex with animals) was an independent factor for penile cancer, even though it did not impact on prognosis [22]. Although it is believed that zoophilia is common in Brazil, our medical records had no information on this behavior and precluded the evaluation on how patients from distinct age groups differed on this issue.
Conclusion
Regardless of squamous cell carcinoma subtypes, penile cancer in Northeastern Brazil seems to show more aggressive features and behavior when presented at younger age. If confirmed in other large series from endemic areas of penile cancer, younger age at diagnosis should be considered a prognostic factor in penile cancer.
References
Clark PE, Spiess PE, Agarwal N, Biagioli MC, Eisenberger MA, Greenberg RE, Herr HW, Inman BA, Kuban DA, Kuzel TM, Lele SM, Michalski J, Pagliaro L, Pal SK, Patterson A, Plimack ER, Pohar KS, Porter MP, Richie JP, Sexton WJ, Shipley WU, Small EJ, Trump DL, Wile G, Wilson TG, Dwyer M, Ho M, National Comprehensive Cancer Network (2013) Penile cancer: clinical practice guidelines in oncology. J Natl Compr Cancer Netw 11:594–615
Colon-Lopez V, Ortiz AP, Soto-Salgado M, Torres-Cintron M, Pettaway CA, Puras-Baez A, Martinez-Ferrer M, Suarez E (2012) Penile cancer disparities in Puerto Rican men as compared to the United States population. Int Braz J Urol 38:728–738
Verhoeven RH, Janssen-Heijnen ML, Saum KU, Zanetti R, Caldarella A, Holleczek B, Brewster DH, Hakulinen T, Horenblas S, Brenner H, Gondos A, Group ESW (2013) Population-based survival of penile cancer patients in Europe and the United States of America: no improvement since 1990. Eur J Cancer 49:1414–1421. doi:10.1016/j.ejca.2012.10.029
Held-Warmkessel J (2012) Penile cancer. Semin Oncol Nurs 28:190–201. doi:10.1016/j.soncn.2012.05.008
Favorito LA, Nardi AC, Ronalsa M, Zequi SC, Sampaio FJ, Glina S (2008) Epidemiologic study on penile cancer in Brazil. Int Braz J Urol 34:587–591 discussion 591–583
Cubilla AL, Dillner J, Schellhammer PF, Horenblas S (2004) Malignant epithelial tumors. In: Eble JN, Sauter G, Epstein J, Sesterhenn I (eds) Pathology and genetics of tumors of the urinary system and male genital organs. IARC press-World Health Organization Classification of Tumours, Lyon, France
Velazquez EF, Ayala G, Liu H, Chaux A, Zanotti M, Torres J, Cho SI, Barreto JE, Soares F, Cubilla AL (2008) Histologic grade and perineural invasion are more important than tumor thickness as predictor of nodal metastasis in penile squamous cell carcinoma invading 5 to 10 mm. Am J Surg Pathol 32:974–979. doi:10.1097/PAS.0b013e3181641365
Chaux A, Torres J, Pfannl R, Barreto J, Rodriguez I, Velazquez EF, Cubilla AL (2009) Histologic grade in penile squamous cell carcinoma: visual estimation versus digital measurement of proportions of grades, adverse prognosis with any proportion of grade 3 and correlation of a Gleason-like system with nodal metastasis. Am J Surg Pathol 33:1042–1048. doi:10.1097/PAS.0b013e31819aa4c9
Barbosa Junior Ade A, Athanazio PR, Oliveira B (1984) Cancer of the penis: study of its geographic pathology in the State of Bahia, Brazil. Rev Saude Publica 18:429–435
Brazil (2012) Instituto Brasileiro de Geografia e Estatística. Censo demográfico 1940/2010. Rio de Janeiro
Paraíba (2013) Brasil - um quadro de desigualdades. In: Federação das Indústrias do Estado da Paraíba - Desigualdes Regionais. Campina Grande
Koifman L, Vides AJ, Koifman N, Carvalho JP, Ornellas AA (2011) Epidemiological aspects of penile cancer in Rio de Janeiro: evaluation of 230 cases. Int Braz J Urol 37:231–240 discussion 240–233
Gregoire L, Cubilla AL, Reuter VE, Haas GP, Lancaster WD (1995) Preferential association of human papillomavirus with high-grade histologic variants of penile-invasive squamous cell carcinoma. J Natl Cancer Inst 87:1705–1709
Bezerra AL, Lopes A, Santiago GH, Ribeiro KC, Latorre MR, Villa LL (2001) Human papillomavirus as a prognostic factor in carcinoma of the penis: analysis of 82 patients treated with amputation and bilateral lymphadenectomy. Cancer 91:2315–2321
Fonseca AG, Soares FA, Burbano RR, Silvestre RV, Pinto LO (2013) Human papilloma virus: prevalence, distribution and predictive value to lymphatic metastasis in penile carcinoma. Int Braz J Urol 39:542–550. doi:10.1590/S1677-5538.IBJU.2013.04.12
Rippentrop JM, Joslyn SA, Konety BR (2004) Squamous cell carcinoma of the penis: evaluation of data from the surveillance, epidemiology, and end results program. Cancer 101:1357–1363. doi:10.1002/cncr.20519
Tsen HF, Morgenstern H, Mack T, Peters RK (2001) Risk factors for penile cancer: results of a population-based case-control study in Los Angeles County (United States). Cancer Causes Control 12:267–277
Chaux A, Caballero C, Soares F, Guimaraes GC, Cunha IW, Reuter V, Barreto J, Rodriguez I, Cubilla AL (2009) The prognostic index: a useful pathologic guide for prediction of nodal metastases and survival in penile squamous cell carcinoma. Am J Surg Pathol 33:1049–1057. doi:10.1097/PAS.0b013e31819d17eb
Spiess PE, Horenblas S, Pagliaro LC, Biagioli MC, Crook J, Clark PE, Greenberg RE, Ercole CE (2013) Current concepts in penile cancer. J Natl Compr Cancer Netw 11:617–624
Pond GR, Di Lorenzo G, Necchi A, Eigl BJ, Kolinsky MP, Chacko RT, Dorff TB, Harshman LC, Milowsky MI, Lee RJ, Galsky MD, Federico P, Bolger G, DeShazo M, Mehta A, Goyal J, Sonpavde G (2014) Prognostic risk stratification derived from individual patient level data for men with advanced penile squamous cell carcinoma receiving first-line systemic therapy. Urol Oncol 32:501–508. doi:10.1016/j.urolonc.2013.10.007
Jayaratna IS, Mitra AP, Schwartz RL, Dorff TB, Schuckman AK (2014) Clinicopathologic characteristics and outcomes of penile cancer treated at tertiary care centers in the Western United States. Clin Genitourin Cancer 12:138–142. doi:10.1016/j.clgc.2013.09.006
Zequi Sde C, Guimaraes GC, da Fonseca FP, Ferreira U, de Matheus WE, Reis LO, Aita GA, Glina S, Fanni VS, Perez MD, Guidoni LR, Ortiz V, Nogueira L, de Almeida Rocha LC, Cuck G, da Costa WH, Moniz RR, Dantas JH Jr, Soares FA, Lopes A (2012) Sex with animals (SWA): behavioral characteristics and possible association with penile cancer. A multicenter study. J Sex Med 9:1860–1867. doi:10.1111/j.1743-6109.2011.02512.x
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paiva, G.R., de Oliveira Araújo, I.B., Athanazio, D.A. et al. Penile cancer: impact of age at diagnosis on morphology and prognosis. Int Urol Nephrol 47, 295–299 (2015). https://doi.org/10.1007/s11255-014-0875-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-014-0875-y