Abstract
Neospora caninum is an obligate intercellular protozoan parasite and considered one of the abortive pathogens in a wide range of ruminants. The present study aimed to investigate the antibodies against N. caninum in camels from three governorates in Egypt to understand the epidemiology of neosporosis and its associated abortion. The study was carried out on 282 camels using commercial enzyme-linked immunosorbent assay (ELISA) kit. The obtained results confirmed the presence of anti-Neospora caninum antibodies in 31 out of 282 camels. Moreover, no significant difference in seroprevalence was observed between different localities under the study and between males and females. On contrary, the number of seropositive camels was increased significantly in adult camels of 8–11 years old (OR = 9.1, 95% CI: 1.1–73.1), during the autumn (OR = 2.3, 95% CI: 0.79–7.1) and mainly in females with history of abortion (OR = 2.8, 95% CI: 1.2–6.5) or in camels raised with dogs (OR = 2.8, 95% CI: 1.1–7.08). The presence of antibodies against N. caninum in camels from Egypt increases the demand for further studies to determine its epidemiological role as a cause of abortion and reduce its economic impacts on camel’s industry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Camel (Camelus dromedarius) is called the ship of the desert as it is uniquely adapted to hot and arid environments and has an economic importance in Egypt as it could be used as a source of meat and milk, vehicle for transport, racing, and tourist rides as the image of the camel is tied to the history of the great Egyptian civilization. Fattening camel calves in Egypt by applying the intensive feeding system could solve the problem of red meat shortage. Additionally, its meat is rich of protein and iron, low of fat and cholesterol, and a healthy option because of its medical properties (El-Badawi 2018).
Neospora caninum, an obligate intracellular protozoan parasite, is one of them (Hilali et al. 1998). It was identified in 1984 in litter of dogs in Norway (Bjerkǻs et al. 1984). Today, neosporosis is recognized all over the world and the infection was detected naturally in domestic ruminants as intermediate hosts like cattle, buffaloes, sheep, goats, and camel (Amdouni et al. 2018).
Dogs are the final host N. caninum and neosporosis is mainly transmitted horizontally to intermediate hosts via ingestion of contaminated food with sporulated oocyst (Machado et al. 2011). Also, the infection could be transmitted vertically from the mother to the fetus through the placenta (Filho et al. 2017).
In ruminants, neosporosis causes abortion during the late stage of pregnancy, fetal death, and still birth. Furthermore, repeat breading could occur as sequelae of fetal death in early stage of gestation (Machado 2019; Semango et al. 2019). Moreover, the disease is characterized by neuromuscular disorder in dogs (Dubey et al. 1988).
In Egypt, previous sero-survey studies demonstrated that the prevalence rate of N. caninum was 3.6% in camels (Hilali et al. 1998), 68% in water buffaloes (Dubey et al. 1998), and 20.43% in cattle (Ibrahim et al. 2009). However, the level of prevalence of N. caninum in camels has not been properly evaluated in wide range in Egypt and the risk factors associated with infection were not considered.
Therefore, the present study was conducted to determine the seroprevalence of N. caninum in camels in some governorates from Egypt to overcome the gap of available information and estimate the risk factors related to infection.
Materials and methods
Study area
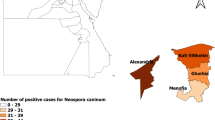
The sero-survey of neosporosis in camels was carried out in three Egyptian governorates as Qalyubia, Kafr ElSheikh, and Red Sea which situated geographically at 30° 25 N to 31° 13 E; 38° 18 N to 30° 56 E; and 25° 32′ 01 N to 33° 26′ 18 E, Fig. 1.
Both Qalyubia and Kafr ElSheikh governorates are located in the Nile Delta, North Egypt which is characterized by high temperature during the summer season and low temperature with little rain during the winter. In addition, Red Sea Governorate is situated at the Southeast of Egypt and shares in border with Sudan, and has a desert climate that is virtually no rainfall throughout the year.
Sample collection
The sample size was calculated using Win Episcope 2.0 (www.winepi.net) to investigate the prevalence level of neosporosis in camel based on previous reported rate 3.6% in Egypt during 1995 (Hilali et al. 1998) with 95% confidence interval and an expected error 5%.
A total of 282 blood samples were collected from individual camel during 2018–2019 from three governorates in Egypt. The sero-survey was performed on 65 males and 217 females. The age groups of the examined camels were divided into < 3 years (N = 31), 3–7 years (N = 106), 8–11 years (N = 60), and > 11 years (N = 85). Camels included in the present study had history of abortion (N = 150) while the rest of the camels were normal and some of them were raised in contact with dogs (N = 175).
The blood samples (5 ml) were collected from jugular vein using vacuum tubes, followed by centrifugation at 1500 rpm/min for 10 min to separate the serum and stored at − 20 °C for further serological analysis.
Serological analysis
The antibody titers against N. caninum in sera of examined camels were determined using CHEKIT Neospora Enzyme Immunoassay kit (Idexx Laboratories, Westbrook, ME, USA) following the manufacturer’s instructions except peroxidase-conjugated goat anti-camel IgG (Triple J. Farms, USA) which was used to detect positive camel sera. The optical density (OD) of each sample was measured using microplate ELISA reader at 450 nm and calculated by the formula, OD% of the test sample = 100 (OD of sample − OD of negative control)/(OD of positive control − OD of negative control). Samples with OD ≥ 40% were considered positive.
Statistical analysis
Data were analyzed using SPSS (ver16, USA) based on Chi-square test and P values < 0.05 were considered significant. Logistic regression model was applied to estimate the association between variables and prevalence of the disease.
Results
Overall, the antibodies against N. caninum among camels were detected in 31 out of 282 total examined camels. The obtained results demonstrated non-significant difference between prevalence of different governorates, and the prevalence was relatively high in Red Sea (15.4%), followed by Qalyubia (9%) and Kafr ElSheikh (6.2%) as in Table 1.
The logistic regression analysis for variables revealed important findings that effect on prevalence of neosporosis among camels, Table 1. Based on age of the examined camels, the seroprevalence was significantly highest (23.3%) in adult age (8–11 years), but it was 3.2%, 6.6%, and 10.6% in camels of < 3, 3-7 and > 11 years old of age, respectively.
Other findings have shown that the seroprevalence of the disease varied significantly according to season, and the disease was more prevalent during the autumn (24.4%, 95% CI: 14.2–38.6) and winter (20%, 95% CI: 10.9–33.8) when compared with the summer (12%, 95% CI: 5.6–23.8) and spring (3.5%, 95% CI: 1.5–7.9).
Interestingly, the number of seropositive animals was increased significantly in female camels (13.4%, 95%CI: 9.4–18.5) that had history of abortion or were raised with dogs, and the seropositive rate was 15.3% (95% CI: 10.4–21.9) and 14.3% (95% CI: 9.8–20.2), respectively.
Odds ratios for significant variables were estimated to determine the risk probability of prevalence of N. caninum among camels, Table 2, indicating that age, season, abortion, and presence of dogs in contact with camel have significant role in prevalence of neosporosis among camels. The present findings confirmed that 8–11-year-old camels could get the infection 9.1 times more than camels < 3 years of age. Camels could be infected 2.3 times during the autumn more than that of the spring season. In addition, female camels with history of abortion (OR = 2.8, 95% CI: 1.2–6.5) or in contact with dogs (OR = 2.8, 95% CI: 1.1–7.08) were more susceptible to N. caninum infection, Table 2.
Discussion
In the Middle East and Africa, the major causes of abortion among camels are brucellosis and trypanosomiasis (Tibary et al. 2006). Despite N. caninum being recognized as a cause of abortion among camels (Shaapan 2016), few studies addressed the prevalence of neosporosis in camel worldwide. The epidemiological information about N. caninum in domestic ruminants particularly in camels in Egypt is limited. Consequently, the present study aimed to investigate the seroprevalence of N. caninum among camels in three governorates that have high density of camel population.
The present findings confirmed the presence of antibodies (10.9%) against N. caninum in examined camels raised in different localities under the study. By comparing the obtained data with previous studies, we found that the reported rate was in line with another rates of 13.7% in the United Arab Emirates (Wernery et al. 2008) and 11.1% in central Punjab, Pakistan (Nazir et al. 2017). In contrast, the obtained prevalence was higher than those reported in Egypt, 3.7% (Hilali et al. 1998) in Mashhad, Iran, 5.83% (Sadrebazzaz et al. 2006) and in central of Iran, 3.9% (Hamidinejat et al. 2013) and lower than those reported in Canary Islands, Spain 86% (Mentaberre et al. 2013), in Czech Republic 24% (Bártová et al. 2017), in Saudi Arabia 16.6% (Mohammed et al. 2020), and in Sudan 38.5% (Ibrahim et al. 2014).
The variation between prevalences may be attributed to geographical factors or environmental patterns, applicable hygienic measures, animal model, and diagnostic test (Fereig et al. 2016; Selim and Ali 2020; Selim et al. 2018).
From the obtained results, it is obvious that the number of seropositive animals increased significantly in adult camels which ties well with previous study (Nazir et al. 2017). In contrast, other studies found that there is no association between prevalences of neosporosis and age of the examined camels (Aljumaah et al. 2018; Hamidinejat et al. 2013). This may be attributed to cumulative risk of repetitive exposure to infection throughout the life and indicates the substantial role of horizontal transmission in spreading of infection (Elhaig et al. 2018; Okumu et al. 2016).
The obtained results revealed that neosporosis is more prevalent (13.4%) in females than males. When comparing the present findings with previous studies, it must be pointed that the seroprevalence of N. caninum increased in female camels than males (Bártová et al. 2017; Mentaberre et al. 2013; Sadrebazzaz et al. 2006). Therefore, the sex has important role on the prevalence of neosporosis in camels that may be due to possibility of vertical transmission of N. caninum through either transplacental or transclostral routes (Filho et al. 2017; Nazir et al. 2017).
Furthermore, data of the present study demonstrated that the N. caninum infection was more prevalent during the autumn and winter when comparing with that of the other seasons. Such results come in accordance with previous research (Ansari-Lari et al. 2017) which found significant effect for season on prevalence of neosporosis in cow. This may be contributed to stressful effect of the temperature on the animals that play an important role in abortion (Selim et al. 2018a, b, c).
From the current results, it is clear that there is a strong association between abortion or presence of dogs and prevalence of N. caninum infection. A similar pattern of the results was obtained (Aljumaah et al. 2018; Nazir et al. 2017). It is important to highlight on fact that the dogs play an effective role in horizontal transmission of neosporosis via shedding of infective oocysts in feces (Machado et al. 2011; Semango et al. 2019) which is considered an important source for infection.
References
Aljumaah, R.S., Alshaikh, M.A., Jarelnabi, A., Abdelrahman, M.M., and Hussein, M.F., 2018. Serological Prevalence of Neospora caninum in Indigenous Dromedary Camels (Camelus dromedarius) in Saudi Arabia, Age, 25, 74.19
Amdouni, Y., Rjeibi, M.R., Awadi, S., Rekik, M., and Gharbi, M., 2018. First detection and molecular identification of Neospora caninum from naturally infected cattle and sheep in North Africa, Transboundary and emerging diseases, 65, 976-982
Ansari-Lari, M., Rowshan-Ghasrodashti, A., Jesmani, H., Masoudian, M., and Badkoobeh, M., 2017. Association of Neospora caninum with reproductive performance in dairy cows: A prospective study from Iran. Veterinary Research Forum, 2017, (Faculty of Veterinary Medicine, Urmia University, Urmia, Iran, 109)
Bártová, E., Kobédová, K., Lamka, J., Kotrba, R., Vodička, R., and Sedlák, K., 2017. Seroprevalence of Neospora caninum and Toxoplasma gondii in exotic ruminants and camelids in the Czech Republic, Parasitology Research, 116, 1925-1929
Bjerkǻs, I., Mohn, S., and Presthus, J., 1984. Unidentified cyst-forming sporozoon causing encephalomyelitis and myositis in dogs, Zeitschrift für Parasitenkunde, 70, 271-274
Dubey, J., Carpenter, J., Speer, C., Topper, M., and Uggla, A., 1988. Newly recognized fatal protozoan disease of dogs, Journal of the American Veterinary Medical Association, 192, 1269-1285
Dubey, J., Romand, S., Hilali, M., Kwok, O., and Thulliez, P., 1998. Seroprevalence of antibodies to Neospora caniuum and Toxoplasma gondii in water buffaloes (Bubalus bubalis) from Egypt, International Journal for Parasitology, 28, 527-529
El-Badawi, A., 2018. The present situation of animal protein in Egypt and the role of camels in providing cheap and healthy meat for people in poor greenery lands, International Journal of Avian & Wildlife Biology, 3, 319-322
Elhaig, M.M., Selim, A., Mandour, A.S., Schulz, C., Hoffmann, B., 2018. Prevalence and molecular characterization of peste des petits ruminants virus from Ismailia and Suez, Northeastern Egypt, 2014–2016. Small Ruminant Research, 169, 94-98
Fereig, R.M., AbouLaila, M.R., Mohamed, S.G., Mahmoud, H.Y., Ali, A.O., Ali, A.F., Hilali, M., Zaid, A., Mohamed, A.E.A., and Nishikawa, Y., 2016. Serological detection and epidemiology of Neospora caninum and Cryptosporidium parvum antibodies in cattle in southern Egypt, Acta tropica, 162, 206-211
Filho PCGA, Oliveira, J.M., Andrade, M.R., Silva, J.G., Kim, P.C., Almeida, J.C., Porto, W.J., and Mota, R.A., 2017. Incidence and vertical transmission rate of Neospora caninum in sheep, Comparative immunology, microbiology and infectious diseases, 52, 19-22
Hamidinejat, H., Ghorbanpour, M., Rasooli, A., Nouri, M., Hekmatimoghaddam, S., Namavari, M.M., Pourmehdi-Borojeni, M., and Sazmand, A., 2013. Occurrence of anti-Toxoplasma gondii and Neospora caninum antibodies in camels (Camelus dromedarius) in the center of Iran, Turkish Journal of Veterinary and Animal Sciences, 37, 277-281
Hilali, M., Romand, S., Thulliez, P., Kwok, O., and Dubey, J., 1998. Prevalence of Neospora caninum and Toxoplasma gondii antibodies in sera from camels from Egypt, Veterinary parasitology, 75, 269-271
Ibrahim, H.M., Huang, P., Salem, T.A., Talaat, R.M., Nasr, M.I., Xuan, X., and Nishikawa, Y., 2009. Prevalence of Neospora caninum and Toxoplasma gondii antibodies in northern Egypt, The American journal of tropical medicine and hygiene, 80, 263-267
Ibrahim, A.M., Ismail, A.A., Angara, T.E.E., and Osman, O.M., 2014. Seroprevalence of Neospora caninum in dairy cattle and the co-herdedcamels, sheep and goats in dairy farms in the Khartoum State, Sudan, Journal of Applied and Industrial Sciences, 2, 206-212
Machado, G.P., 2019. Neosporosis in Small Ruminants, Silva, 4, 211-214
Machado, G.P., Kikuti, M., Langoni, H., and Paes, A.C., 2011. Seroprevalence and risk factors associated with neosporosis in sheep and dogs from farms, Veterinary parasitology, 182, 356-358
Mentaberre, G., Gutiérrez, C., Rodríguez, N.F., Joseph, S., González-Barrio, D., Cabezón, O., de la Fuente, J., Gortazar, C., and Boadella, M., 2013. A transversal study on antibodies against selected pathogens in dromedary camels in the Canary Islands, Spain, Veterinary Microbiology, 167, 468-473
Mohammed, O.B., Amor, N., Omer, S.A., and Alagaili, A.N., 2020. Seroprevalence of Toxoplasma gondii and Neospora caninum in Dromedary camels (Camelus dromedarius) from Saudi Arabia, Revista Brasileira de Parasitologia Veterinária, 29
Nazir, M.M., Oneeb, M., Ayaz, M.M., Bibi, F., Ahmad, A.N., Waheed, A., Sajid, M.A., Sultan, M.T., Yasin, G., and Lindsay, D.S., 2017. Prevalence of antibodies to Neospora caninum in the serum of camels (Camelus dromedarius) from central Punjab, Pakistan, Tropical animal health and production, 49, 1081-1084
Okumu, T.A., Munene, J.N., Wabacha, J., Tsuma, V., and Van Leeuwen, J., 2016. Seroepidemiological survey of Neospora caninum and its risk factors in farm dogs in Nakuru district, Kenya, Veterinary world, 9, 1162
Sadrebazzaz, A., Haddadzadeh, H., and Shayan, P., 2006. Seroprevalence of Neospora caninum and Toxoplasma gondii in camels (Camelus dromedarius) in Mashhad, Iran, Parasitology Research, 98, 600-601
Selim, A., and Ali, A.-F., 2020. Seroprevalence and risk factors for C. burentii infection in camels in Egypt, Comparative Immunology, Microbiology and Infectious Diseases, 68, 101402
Selim, A., Ali, A.-F., Moustafa, S.M., and Ramadan, E., 2018a. Molecular and serological data supporting the role of Q fever in abortions of sheep and goats in northern Egypt, Microbial pathogenesis, 125, 272-275
Selim, A., Elhaig, M., and Moawed, S.A., 2018b. A Serological Survey of Four Abortifacient Infectious Agents among Small Ruminant in Egypt, Asian Journal of Animal and Veterinary Advances, 13, 114-121
Selim, A., Yang, E., Rousset, E., Thiéry, R., and Sidi-Boumedine, K., 2018c. Characterization of Coxiella burnetii strains from ruminants in a Galleria mellonella host-based model, New microbes and new infections, 24, 8-13
Semango, G., Hamilton, C.M., Kreppel, K., Katzer, F., Kibona, T., Lankester, F., Allan, K.J., Thomas, K.M., Claxton, J.R., and Innes, E.A., 2019. The sero-epidemiology of Neospora caninum in cattle in northern Tanzania, Frontiers in veterinary science, 6, 327
Shaapan, R.M., 2016. The common zoonotic protozoal diseases causing abortion, Journal of parasitic diseases, 40, 1116-1129
Tibary, A., Fite, C., Anouassi, A., and Sghiri, A., 2006. Infectious causes of reproductive loss in camelids, Theriogenology, 66, 633-647
Wernery, U., Thomas, R., Raghavan, R., Syriac, G., Joseph, S., and Georgy, N., 2008. Seroepidemiological studies for the detection of antibodies against 8 infectious diseases in dairy dromedaries of the United Arab Emirates using modern laboratory techniques-Part II, Journal of Camel Practice and Research, 15, 139-145
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Selim, A., Abdelhady, A. Neosporosis among Egyptian camels and its associated risk factors. Trop Anim Health Prod 52, 3381–3385 (2020). https://doi.org/10.1007/s11250-020-02370-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-020-02370-y