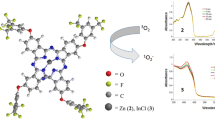
Abstract
In this study, new 4-chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile compound, containing bioactive thymoxy group, and its metal-free phthalocyanine and metallo-phthalocyanine derivatives were synthesized for the first time. Their structures were determined by spectroscopic methods such as FTIR, UV–Vis, 1H-, and 13C-NMR (for phthalonitrile derivative), MALDI-TOF mass spectrometry (for phthalocyanine derivatives) and elemental analysis as well. The phthalocyanines showed excellent solubility in polar and nonpolar solvents without aggregation and absorb at long wavelengths with their high molar coefficient. In N,N-dimethylformamide, the effects of the type of central metal ions [metal-free, indium(III) acetate, lutetium(III) acetate, magnesium(II) or zinc(II)] in the phthalocyanine, containing bioactive thymoxy group, cavity on the spectroscopic, photophysical, and photochemical properties of the phthalocyanines were determined. These features are compared with each other. Lutetium(III) acetate phthalocyanine did not show any fluorescence, while metal-free phthalocyanine and indium(III) acetate phthalocyanine showed low fluorescence. It was determined that magnesium phthalocyanine significantly enriched the fluorescence, and zinc phthalocyanine had appropriate and sufficient fluorescence. Lutetium(III) acetate and zinc(II), especially indium(III) acetate phthalocyanines, could produce large amounts of singlet oxygen. Metal-free and magnesium phthalocyanines had the capacity to produce sufficient singlet oxygen (it means production of enough amount of singlet oxygen by a photosensitizer candidate during PDT applications). All phthalocyanines have sufficient and suitable photostability (it means an ideal photosensitizer should be stable under light irradiation until complete its PDT activation, and it should be decomposed after its PDT activation so that it does not accumulate in the body). With these determined properties, magnesium(II), especially indium(III) acetate and zinc(II) phthalocyanines, may be suitable candidates as type II photosensitizers for photodynamic therapy applications. Lutetium(III) acetate phthalocyanine may be a photosensitizer candidate in photocatalytic applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Photodynamic therapy (PDT) is a well-known method based on the interaction of light, photoactive material, and oxygen, and commonly used to treat of many cancer (tumor) diseases and to control bacterial invasion in bacterial diseases [1,2,3,4]. In this method, the phototherapeutic agent, also called photosensitizer (PS), is firstly deposited in the tumor tissue, irradiated with light, and then, the oxygen in the cell reacts with this enriched agent (PS) and leads to the excitation of PS. Thus, the excited photosensitizer transfers spin-permissible energy to the cell, and the singlet oxygen occurs there. Last, highly reactive singlet oxygen can occur in reaction with the damaged tissue and cause the death of the tumor [5].
Despite the successes of first-generation PS such as Photofrin®, Photosan®, and some hematoporphyrin derivatives, scientists have been searched to new compounds that meet the demands of modern medicine. Important aspects for ideal photosensitizers are absorption in the long-wave range, high absorption coefficients, and stability during irradiation. In addition, second-generation PSs should have the features of rapid tumor accumulation, low dark toxicity, and a broad spectrum of activity [5,6,7,8]. A class of compounds that fulfills these properties in many ways is phthalocyanines (Pcs). Since the early 1980s, these macrocycles have been considered potential sensitizers for PDT and are the subject of intense research [9].
Pcs have been a major source of interest since the discovery of their unexpected synthesis at the beginning of the last century [10]. The reason for the intense interest in these macrocycles is because of their high stability, architectural flexibility, and unique spectral properties [11]. Due to the ease of the modifications on the Pc ring, optional adjustments can be made to improve their properties. For example, Pc aggregation can be decreased, and Pc solubility can be enhanced by introducing bulky groups with alkyl chains to the axial and peripheral positions of the Pcs. On the other hand, while the hydrogen atoms in the Pc core can be replaceable by more than 70 different metals and metalloids. This advantage offers additional features for synthetic organic chemists to optimize the physical responses of the Pcs [12].
Pcs have special photochemical properties due to the location of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels and their energy difference (ΔE). Since the molecular orbitals are quantized in terms of their energy, Pcs absorb the photons of certain energy (hν). The energy of photons is equal to the difference in energy (ΔE = hν) between the ground and excited states. Since the HOMO/LUMO energy difference of the Pcs is ~ 170 kJ/mol (~ 700 nm), Pcs can be excited by visible light [9, 13,14,15,16,17,18].
The photochemical generation of singlet oxygen takes place after the triplet singlet energy transfer due to the photosensitizers such as Pcs, porphyrins, rose Bengal, methylene blue, or chlorophyll [19].
The triplet energy for the porphyrins and Pcs is approx. 108–150 kJ/mol. The energies of the first excited state (1Δg) and the second excited state (1Σg +) of oxygen are 94.7 kJ/mol and 157.8 kJ/mol, respectively. The energy of the excited state of oxygen is therefore below the energy of the triplet state of these photosensitizer. This is one of the most important conditions for energy transfer [20].
Thymol and its derivatives have been studied extensively because of their biological activities. Thymol is a phenolic monoterpenoid compound obtained from essential oils of Thymus vulgaris, Monarda punctata [21], and various other kinds of plants. It shows wide biological activities against cancerogenic cells [22] and microbial diseases [23]. It has been known to exhibit anti-inflammatory [24] and anti-oxidative effects [25]. In fact, studies on alpha and beta substituted thymoxy substituted Pcs are also found in the literature [26, 27]. However, these species (alpha and beta tetra-substituted Pcs) form regioisomers during their synthesis process. It is extremely difficult to separate four obtained isomers from each other, and only HPLC method can most effectively be separate this isomer mixture [28]. Pc researchers who prefer these syntheses often and isolate the most abundant species in the mixture and can study its properties [27]. The properties of other isomers are often not determined due to lack of quantity [27]. However, octa thymoxy substituted Pcs obtained as a single isomer by using disubstituted starting phthalonitriles are not available in the literature. It was determined in the literature about the synthesis of octa thymoxy substituted Pcs that the starting material (4,5-bis(2-isopropyl-5-methylphenoxy)phthalonitrile) was synthesized, but no Pc synthesis was made using this substance [16]. Thus, syntheses of octa substituted Pcs, contain at peripheral positions a thymoxy and a chlorine groups, were planned by using 4-chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile starting compound in this study (Scheme 1). It is predicted that the addition of the electron-withdrawing chlorine groups to the molecular structure will show different chemical, physical, and photophysicochemical properties compared to alpha or beta tetra thymoxy substituted Pc in the literature. It is predicted that since electron-withdrawing chlorine groups added to the molecular structure, these Pcs will show different chemical, physical and photophysicochemical properties compared to alpha or beta tetra thymoxy substituted Pcs in the literature.
Experimental
Materials and method
In this study, thymoxy substituted phthalonitrile compound (1) was synthesized by a nucleophilic aromatic substitution reaction between thymol and 4,5-dichlorophthalonitrile [29] and its metal-free and metallo-Pcs (2–6) were obtained by using this compound. The materials and equipment are given in the supplementary information section.
Synthesis
4-Chloro-5-(2-isopropyl-5-methylphenoxy) phthalonitrile (1)
Solution of thymol (2-isopropyl-5-methylphenol) (1.5 g, 10 mmol) in 20 mL dry DMF was slowly added while stirring to the solution of 4,5-dichlorophthalonitrile (1.97 g, 10 mmol) in 30 mL dry DMF and the solution stirred further for 15 min. Afterward, finely powdered K2CO3 (2.76 g, 20 mmol) was added to this medium. The reaction solution was stirred at 60 °C for 3 days under a vacuum. The reaction was followed by thin-layer chromatography (TLC) and stopped after the disappearance of spots belonging to the reactants on the TLC plate. The reaction mixture was cooled, and K2CO3 was filtered. The filtrate was then poured into ice water. After the complete precipitation, the crude product was filtered with a sand funnel and washed several times with water. Compound 1 was purified by column chromatography (silica gel) using CHCl3 as an eluent. The collected fraction was dried to obtain pure compound 1 in a good yield (71%) [29].
Compound 1 is soluble in dichloromethane (DCM), CHCl3, tetrahydrofuran (THF), acetonitrile, acetone, toluene, DMF, and dimethyl sulfoxide (DMSO). Yield: 2.2 g (71%). Melting point: 124–126 °C. FTIR νmax (cm−1): 523, 610, 826, 959, 1020, 1069, 1094, 1179, 1246, 1364, 1420, 1486, 1514, 1588, 1607, 2233, 2848, 2965, 3046, 3080. 1H-NMR (500 MHz, DMSO-d6) δ, ppm: 8.54 (s, 1H), 7.44 (s, 1H), 7.34 (d, J = 7.8 Hz, 1H), 7.12 (d, J = 7.8 Hz, 1H), 6.88 (s, 1H), 2.96 (septet, J = 6.9 Hz, 1H), 2.28 (s, 3H), 1.14 (d, J = 6.9 Hz, 6H). 13C-NMR (500 MHz, CDCl3) δ, ppm: 158.5 (1C), 151.0 (1C), 139.0 (1C), 136.9 (1C), 135.5 (1C), 128.9 (1C), 128.0 (1C), 128.0 (1C), 120.9 (1C), 119.6 (1C), 116.0 (1C), 114.7 (1C), 114.6 (1C), 110.9 (1C), 27.3 (1C), 22.9 (1C), 22.8 (2C). Elemental analysis: % calculated for C18H15ClN2O: C, 69.57; H, 4.87; N, 9.01, obtained % results: C, 69.74; H, 4.69; N, 9.16[29].
2,9,16,23-tetrachloro-3,10,17,24-tetrakis(2-isopropyl-5-methylphenoxy)phthalocyanine (H2Pc) (2)
4-Chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile (1) (0.93 g, 3 mmol) and lithium (0.03 g, 4 mmol) in n-pentanol (20 mL) were mixed and stirred at 150 °C for 18 h under N2 atmosphere. This solution was cooled to room temperature. Then, the solvent was evaporated under a vacuum. Then, CH3COOH (30 mL) was added, and the suspension was stirred for 30 min. The product was extracted with dichloromethane and washed with water (3 × 30 mL). The organic phase is separated and dried with anhydrous magnesium sulfate.
Compound 2 is soluble in hexane, DCM, CHCl3, THF, acetonitrile, acetone, toluene, DMF, and DMSO. Yield: 0.24 g (26%). Melting point ˃300 °C. FTIR νmax (cm−1): 436, 460, 586, 688, 737, 811, 881, 937, 1011, 1084, 1147, 1246, 1417, 1502, 1575, 1603, 2014, 2161, 2554, 2867, 2924, 2959, 3028, 3286. UV–Vis (DCM, 1 × 10−5 M): λmax (nm) (log ε): 701 (5.05), 667 (5.03), 637 (4.71), 608 (4.56), 347 (4.91). Elemental analysis: % calculated for C72H62Cl4N8O4: C, 69.52; H, 5.06%; N, 14.45, obtained % results: C, 69.41; H, 5.13; N, 14.30. MS: (MALDI-TOF), m/z calc. 1245.14, found 1245.74 [M]+.
General synthesis procedure for the metallo-phthalocyanines
4-Chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile (1) (0.93 g, 3 mmol) and metal salt (0.58 g indium(III) acetate for In(OAc)Pc, 0.73 g lutetium(III) acetate hydrate for Lu(OAc)Pc, 0.43 g magnesium acetate tetrahydrate for MgPc, 0.37 g zinc acetate dihydrate for ZnPc), and 0.2 mL of DMF were evacuated into a Schlenk tube and stirred until all solids had dissolved. Then, 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) (0.35 mL, 2.34 mmol) was added dropwise to the reaction tube while stirring. The reaction mixture was stirred at 160 °C under an inert atmosphere overnight. After cooling to room temperature, the reaction solution was poured onto 450 mL of methanol/water (8: 1) and the formed solids were filtered. The crude product was washed with water overnight and methanol in a Soxhlet apparatus. The resulting solid was purified by column chromatography (silica gel) with CHCl3/MeOH (20:1) solvent system as an eluent. Green pure products were obtained.
2,9,16,23-Tetrachloro-3,10,17,24-tetrakis(2-isopropyl-5-methylphenoxy)phthalocyaninato indium(III)acetate (In(OAc)Pc) (3)
Compound 3 is soluble in DCM, CHCl3, THF, ethanol, acetone, toluene, DMF, and DMSO. Yield: 0.18 g (19%). Melting point ˃300 °C. FTIR νmax (cm−1): 439, 555, 586, 744, 814, 944, 993, 1060, 1084, 1147, 1246, 1330, 1389, 1435, 1505, 1572, 1603, 1649, 1718, 2851, 2867, 2924, 2959, 3052. UV–Vis (DMSO, 1 × 10−5 M): λmax (nm) (log ε): 693 (4.96), 626 (4.30), 366 (4.72). Elemental analysis: % calculated for C74H63Cl4N8O4In: C, 62.73; H, 4.48; N, 7.91, obtained % results: C, 62.61; H,4.33; N, 8.03. MS: (MALDI-TOF), m/z calc. 1416.99, found 1359.38 [M-OAc + H]+.
2,9,16,23-Tetrachloro-3,10,17,24-tetrakis(2-isopropyl-5-methylphenoxy)phthalocyaninato lutetium(III)acetate (Lu(OAc)Pc) (4)
Compound 4 is soluble in hexane, DCM, CHCl3, THF, ethanol, acetone, toluene, DMF, and DMSO. Yield: 0.20 g (21%). Mp: ˃300 °C. FTIR νmax (cm−1): 432, 474, 586, 678, 751, 811, 888, 944, 993, 1056, 1084, 1147, 1246, 1389, 1435, 1568, 1719, 1607, 1656, 1719, 2867, 2924, 2960, 3018. UV–Vis (DMSO, 1 × 10−5 M): λmax (nm) (log ε): 685 (5.06), 614 (4.33), 358 (4.77). Elemental analysis: % calculated for C74H63Cl4N8O4Lu: C, 60.17; H, 4.30; N, 7.59, obtained % results: C, 60.31; H,4.43; N, 7.73. MS: (MALDI-TOF), m/z calc. 1477.13, found 1418.07 [M-OAc + H]+.
2,9,16,23-Tetrachloro-3,10,17,24-tetrakis(2-isopropyl-5-methylphenoxy) phthalocyaninato magnesium (5)
Compound 5 is soluble in hexane, DCM, CHCl3, THF, ethanol, acetone, toluene, DMF, and DMSO. Yield: 0.30 g (32%). M.p. ˃ 300 °C. FTIR νmax (cm−1): 432, 492, 590, 695, 748, 807, 884, 941, 986, 1053, 1088, 1151, 1242, 1333, 1389, 1424, 1498, 1572, 1603, 1663, 2856, 2919, 2954, 3024. UV–Vis (DMSO, 1 × 10−5 M): λmax (nm) (log ε): 680 (4.96), 615 (4.42), 358 (4.71). Elemental analysis: % calculated for C72H60Cl4N8O4Mg: C, 68.23; H, 4.77; N, 8.84, obtained % results: C, 68.31; H,4.54; N, 8.73. MS: (MALDI-TOF), m/z calc. 1267.43, found 1266.57 [M-H]+.
2,9,16,23-Tetrachloro-3,10,17,24-tetrakis(2-isopropyl-5-methylphenoxy) phthalocyaninato zinc (6)
Compound 6 is soluble in hexane, DCM, CHCl3, THF, ethanol, acetone, toluene, DMF, and DMSO. Yield: 0.33 g (35%). FTIR νmax (cm−1): 446, 692, 741, 804, 881, 941, 990, 1056, 1084, 1147, 1246, 1333, 1382, 1428, 1481, 1568, 1603, 1656, 2863, 2919, 2961, 3038. UV–Vis (DMSO, 1 × 10−5 M): λmax (nm) (log ε): 681 (5.01), 628 (4.53), 354 (4.78). Elemental analysis: % calculated for C72H60Cl4N8O4Zn: C, 66.09; H, 4.62; N, 8.56, obtained % results: C, 66.27; H, 4.55; N, 8.72. MS: (MALDI-TOF), m/z calc. 1308.50, found 1308.81 [M]+.
Results and discussion
Synthesis and characterization
4-Chloro-5-(2-isopropyl-5-methylphenoxy)phthalonitrile (1) compound was obtained by the nucleophilic substitution reaction between thymol and 4,5-dichlorophthalonitrile[29]. In the 1H-NMR spectrum of this compound, aliphatic hydrogen peaks of isopropyl and methyl groups on the molecule can be seen clearly. Moreover, ortho and meta splitting of Ar–H peaks can be easily distinguished between 7 and 7.5 ppm (Fig. S1 in the SI). In the 13C-NMR spectrum of this compound, aliphatic carbons (4C) peaks, aromatic carbons (12C) peaks, and nitrile carbons (2C) peaks were observed at 22.8–27.3 ppm, 110.9–158.5 ppm, and 114.6 ppm, respectively (Fig. S2 in the SI). The functional groups of the phthalonitrile molecule were verified by FTIR spectroscopy, and it was observed peaks between 2869 and 2972 cm−1 for aliphatic C–H, 3036–3100 cm−1 for aromatic C–H, the sharp peak at 2234 cm−1 for nitrile (C≡N) vibration, and moreover, the aromatic O–H peak of thymol was disappeared in the FTIR spectrum of compound 1 (Fig. S3 in the SI) [29].
Metallo- and metal-free Pcs were synthesized from the anhydrous basic tetramerization reaction of the phthalonitrile compound 1. The structures of Pcs were checked by employing common spectroscopic techniques such as FTIR, UV–Vis, MALDI-TOF mass spectrometry and elemental analyses as well.
The nitrile stretching peak was disappeared in the FTIR spectra of the Pcs after the tetramerization reaction. The aromatic and aliphatic C–H stretching peaks were observed between 3052–3018 cm−1 and 2932–2907 cm−1 in the FTIR spectra, respectively. The characteristic N–H stretching peak of metal-free phthalocyanine (H2Pc) was observed at 3286 cm−1 as an extra peak (Figs. S4, S6, S8, S10, and S12 in the SI). In addition, C=O stretching bands were observed at 1718 and 1719 cm−1, respectively, in the FTIR spectra of In(OAc)Pc and Lu(OAc)Pc. This situation can be attributed to the fact that these metals with + 3 oxidation states attach acetate groups from their axial bonds in Pc molecules.
The MALDI-TOF mass spectra of the Pcs were accurate with the proposed structures. The molecular ion peaks for H2Pc (2), MgPc (5), ZnPc (6) and [M-OAc + H]+ fragmented ion peaks for In(OAc)Pc (3) and Lu(OAc)Pc(4) were easily identified at m/z: 1245.74 as [M+], 1266.57 as [M-H]+, 1308.81 as [M+], 1359.38 as [M-OAc + H]+, and 1418.07 as [M-OAc + H]+, respectively (Figs. S5, S7, S9, S11 and S13 in the SI). All results mentioned above confirmed that the synthesis of the compounds was accomplished. All the above-mentioned results confirmed that the synthesis of the compounds was successfully achieved.
Photophysical and photochemical properties
Electronic absorption and fluorescence spectra
To be able to penetrate deeper into the tissue with light, the photosensitizers must have long-wavelength absorption maxima and high molar extinction coefficients. A 600–800 nm absorption window is optimal for a photosensitizer for PDT treatment. Molecules with absorption maxima above 850 nm have triplet states that have too low energy and cannot be used for medical purposes, since the energy of light (λ = 850 nm) is not sufficient to generate singlet oxygen.
Thymol-substituted Pcs (2–6) showed two major characteristic absorption bands called as B and Q between 347–358 nm and 681–701 nm, respectively, caused by the π−π* transition. As a result of D2h symmetry, H2Pc exhibited typical metal-free Pc spectral behavior with two narrow peaks in the Q region band at 667 and 701 nm. While it shows monomeric behavior in chloroform, dichloromethane, DMF, THF, and toluene, it forms aggregates in acetone, and hexane (Fig. S14 in the SI).
The photophysical properties affect negatively when a photosensitizer forms aggregates. In addition, aggregated molecules have greatly altered solubility properties, which can cause difficulties at the cellular level. In particular, the formation of H-aggregates should be avoided. For this reason, the photosensitizer must be structurally modified so that aggregates in the solution are as unstable as possible. This problem can be overcome through the introduction of bulky substituents on the phthalocyanine ring. The formation of J aggregation instead of H-aggregates in a solution can be decisive for a photosensitizer for the potential application in PDT. However, the tendency to form aggregates in solution is difficult to predict and must always be examined very carefully.
In the aggregation studies of the synthesized metallo-Pcs (3–6), all the compounds showed quite similar behavior and tend not to aggregate in common organic solvents, except for MgPc, which showed J-aggregation in chloroform solutions, In(OAc)Pc and ZnPc which indicates H-aggregated species in ethanol solutions (Figs. S15, S16, S17 and S18 in the SI). DMF was chosen as a solvent for further photophysical and photochemical properties. The fact that the Q bands are very narrow suggests very little aggregation of the molecules in the concentration range investigated, which confirms the thymol-substituted Pcs (2–6) avoid π−π stacking interactions. The aggregation properties of the synthesized compounds were investigated at different concentrations ranging from 1 × 10−5 to 1 × 10−6 M in DMF (Figs. 1, 2, and S19, S20, and S21 in the SI). Any of the Pcs do not aggregate in DMF solutions. The small amount of DMF is not toxic in the biological environment, and its miscibility with water is another advantage of this solvent for PDT studies.
Fluorescence emission peaks maxima of the studied Pcs (2–6) are listed in Table 1. Figures 3,4, S22, and S23 in the SI show the absorption, fluorescence emission, and excitation spectra of peripherally thymol and chlorine-bearing Pcs (2–6) in DMF. The Stokes shifts of the studied Pcs (2–6) were observed to be slightly longer in comparison with unsubstituted ZnPc, and they were found in the range of 7–10 nm. The similarity between excitation and absorption spectra of the Pcs can be explained by the no effect of the used light during excitation.
Fluorescence quantum yields
An ideal photosensitizer should show some fluorescence behavior to be able to follow them in the body. Consequently, it is important to investigate fluorescence quantum yield (ФF) characteristics of photosensitization for PDT applications.
The ФF values of the thymol-substituted Pcs (2–6) were determined in DMF, and the results are provided in Table 1. ФF value of Std-ZnPc was also supplied as a reference in this table for comparison in fluorescence quantum yield determinations. It can be concluded from the similarities of the absorption and excitation spectra of the studied compounds that the nuclear configurations of ground and excited states of the compounds are also similar and not affected by excitation (Figs. 3 and 4). The fluorescence quantum yields of the newly synthesized Pcs were obtained to be lower than the value of Std-ZnPc except for MgPc, which has the highest ФF value with a yield of 0.345. The high ФF value of MgPc can be explained by the bivalency, monoatomic dication, and small ionic radius of the central metal, which diminishes the intersystem crossing to the triplet state. On the other hand, properties like high coordination number, big atomic radius, and trivalency of central metal of In(OAc)Pc make it less fluorescent compared to others. Due to the heavy atom effect of the lutetium(III) metal in the Pc cavity, no fluorescence emission was observed in the DMF solution of the compound Lu(OAc)Pc. Heavy metals increase intersystem crossing; therefore, less or no fluorescence is observed. Relatively low fluorescence quantum yield of H2Pc (ФF = 0.075) was observed compared to non-heavy metal Pcs (5 and 6) which might be the case of less fluorescence quenching for these metallo-Pcs 5 and 6.
Singlet oxygen quantum yields (Φ Δ)
The singlet oxygen formed in the tissue generates oxidative cellular damage, which leads to apoptosis or necrosis of the affected tumor cells. Irradiation with a specific wavelength of light converts the photosensitizer into an excited state. This exciting photosensitizer collides with molecular triplet oxygen contained in the surrounding tissue. Ultimately, the photosensitizer returns to its ground state, and the cycle can repeat itself with continued exposure to light.
Since the intracellular generation of 1O2 is the basis of PDT, it stands to reason that type II photoreaction is the focus of the further development of modern photosensitizers [30]. To obtain a high singlet oxygen quantum yield, the excited triplet state must be as long-lived as possible. This can be achieved by introducing heavy atoms, such as halogen atoms, or by introducing central atoms that stabilize the triplet state.
To calculate the singlet oxygen produced by the photosensitizer, 1,3-diphenylisobenzofuran (DPBF) is used as a singlet oxygen quencher. The quantum yield for the reaction between DPBF and singlet oxygen (ΦDPBF) is determined experimentally from the decrease in DPBF absorbance at 417 nm after each irradiation interval.
The ΦΔ values of the thymol-substituted Pcs (2–6) were determined in DMF, and the results are provided in Table 1. ΦΔ value of Std-ZnPc was also supplied in Table 1 for comparison in singlet oxygen quantum yields of the studied Pcs (2–6). From the measurements of all the studied compounds, a significant decrease in the maximum absorption bands of DPBF was observed but there was not any change in the Q bands of Pcs, which shows the production of the singlet oxygen and stabilities of the photosensitizer compounds during the photodynamic activity, respectively (Figs. 5, 6 and S24, S25 and S26 in the SI). Singlet oxygen quantum yields of H2Pc and MgPc were found lower than that of Std-ZnPc, but these values were found to be higher for ZnPc and the highest for In(OAc)Pc. The lowest singlet oxygen generation efficiency with the ΦΔ value of 0.13 was observed from the measurements of H2Pc in DMF solution due to the lack of metal ions in the cavity. Mg and Zn Pcs can form two different forms of coordination depending on the reaction conditions. The first one is the square planar coordination in which the central Mg(II) and Zn(II) cations coordinate with the four isoindole nitrogen atoms of the Pc ring. The second one is the distorted tetragonal bi-pyramid coordination in which the central Mg(II) and Zn(II) cations coordinate by binding extra two acetate anions belonging to the salts at the axial positions of the Pc ring(4 + 2 coordination) [31]. In this study, when the FTIR spectra of the MgPc and ZnPc were examined, the band of the C=O bond was not observed belonging to the acetate group at around 1700 cm−1. Thus, the result showed that the coordination of synthesized MgPc and ZnPc in this study is square planar. MgPc has a low ΦΔ value because of the central metal magnesium, which is an alkaline earth metal divalent metal cation. Therefore, magnesium metal in the core of the Pc decreases the efficiency of the generation of singlet oxygen. On the other hand, ZnPc has a high ΦΔ value of 0.70 compared to 0.56 of unsubstituted Std-ZnPc. It shows that both peripheral substituted thymol groups and the central metal zinc have an increasing effect on the singlet oxygen generation. The highest singlet oxygen generation efficiencies were observed for In(OAc)Pc and Lu(OAc)Pc, 0.84 and 0.80, respectively, which suggests the enhancing effect of heavy metals on the photochemical singlet oxygen production.
Photodegradation studies
In the case of PDT, substances are required that have rather low photooxidative stability and also dark stability. In this way, a photosensitizer can be broken down in the body more quickly after irradiation. Photostabilities of the studied compounds were examined by evaluation of changes in their Q bands when they were exposed to a 50 V light at certain intervals (Figs. 7, 8 and S27, S28, and S29 in the SI). Photodegradation quantum yield (Фd) values obtained from those spectra help us to interpret PS’s stability when exposed to light. Ideal photosensitizers should be in a certain range of stability because it is necessary to avoid their toxicity within the body after the completion of the photodynamic action. Photodegradation quantum yields of stable ZnPcs are around 10−6, and it is around 10−3 for unstable ones [17]. The observed Фd values of the studied thymol-substituted Pcs were higher than the unsubstituted Std-ZnPc (Φd = 0.23 × 10−4) in DMF which implies a decrease in stability because of peripheral substitution. Photodegradation quantum yield (Фd) values of the thymol-substituted Pcs were found between 1.10 × 10−4 and 4.32 × 10−4, which are acceptable values for their application in PDT.
Conclusion
In this study, new metal-free phthalocyanine [H2Pc2] and metallo-phthalocyanines [In(OAc)Pc 3, Lu(OAc)Pc 4, ZnPc 5 and MgPc 6] containing chlorine and biologically active thymol groups with oxo-bridges were synthesized. Structural characterizations were carried out with the common spectroscopic techniques including FTIR, 1H- and 13C-NMR (for phthalonitrile derivative), UV–Vis, MALDI-TOF spectrometry and elemental analysis as well. The Pcs (2–6) showed well solubility without aggregation in commonly known solvents, such as hexane, DCM, CHCl3, acetone, toluene, DMF, DMSO, and THF, and were also capable of absorbing at maximum intensity at long wavelengths. Photophysical and photochemical experiments of the Pcs have been carried out in DMF because the studied phthalocyanines do not exhibit any aggregates in this solvent. Fluorescence spectra of the studied compounds indicate lower quantum yield in comparison with the unsubstituted ZnPc except for MgPc. In(OAc)Pc, Lu(OAc)Pc, and ZnPc showed higher singlet oxygen yield compared to standard unsubstituted zinc phthalocyanine. In(OAc)Pc showed the highest singlet oxygen quantum yield. Photodegradation studies have shown that the Pcs (2–6) have suitable stability against to light irradiation for PDT applications. The singlet oxygen quantum yields, which give indication of the potential of the studied phthalocyanines as photosensitizers in applications where singlet oxygen is required, are suitable for PDT applications of the studied Pcs especially In(OAc)Pc.
Data and material availability
Not applicable.
References
Crucius G (2013) Synthesen nichtperipher glykokonjugierter Zink (II) phthalocyanine. Universitätsbibliothek Tübingen
Josefsen LB, Boyle RW (2012) Unique diagnostic and therapeutic roles of porphyrins and phthalocyanines in photodynamic therapy, imaging and theranostics. Theranostics 2:916
Hamblin MR, Hasan T (2004) Photodynamic therapy: a new antimicrobial approach to infectious disease? Photochem Photobiol Sci 3:436–450
Patrice T (2003) Photodynamic therapy. Royal Society of Chemistry, London
Hirth A, Michelsen U, Wöhrle D (1999) Photodynamische tumortherapie. Chem unserer Zeit 33:84–94
Whitacre CM, Feyes DK, Satoh T, Grossmann J, Mulvihill JW et al (2000) Photodynamic therapy with the phthalocyanine photosensitizer Pc 4 of SW480 human colon cancer xenografts in athymic mice. Clin Cancer Res 6:2021–2027
Allen CM, Sharman WM, Van Lier JE (2001) Current status of phthalocyanines in the photodynamic therapy of cancer. J Porphyr Phthalocyanines 5:161–169
Dolmans DE, Fukumura D, Jain RK (2003) Photodynamic therapy for cancer. Nat Rev Cancer 3:380–387
Brasseur N (2003) Sensitizers for PDT: phthalocyanines. In: Patrice T (ed) Photodynamic therapy, vol 2. The Royal Society of Chemistry, London, pp 105–118
Flom SR (2003) Nonlinear optical properties of phthalocyanines. In: Kadish KM, Smith KM, Guilard R (eds) The porphyrin handbook. Application of phthalocyanines. Elsevier Academic Press, California, pp 179–190
Tanaka M (2009) Phthalocyanines – high performance pigments and their applications. In: Faulkner EB, Schwartz RJ (eds) High performance pigments. John Wiley and Sons, Weinheim, pp 275–291
Wöhrle D, Schnurpfeil G (2003) Porphyrins and Phthalocyanines in Macromolecules-110. In: The Porphyrin Handbook. Academic Press, Amsterdam
Mckeown N, Weinreb S (2004) Science of Synthesis. Houben-Weyl Methods of Molecular Transformations–Hetarenes and Related Ring Systems. 17: 1237
Seikel E (2012) Axial funktionalisierte Metallophthalocyanine und-porphyrazine als Funktionsmoleküle für optoelektronische Anwendungen. Philipps-Universität Marburg
Wöhrle D, Eskes M, Shigehara K, Yamada A (1993) A Simple synthesis of 4, 5-disubstituted 1, 2-dicyanobenzenes and 2, 3, 9, 10, 16, 17, 23, 24-octasubstituted phthalocyanines. Synthesis 1993:194–196
Atalay Ş, Çoruh U, Akdemir N, Ağar E (2004) C–H⋯ π interactions in 4, 5-bis (2-isopropyl-5-methylphenoxy) phthalonitrile. Acta Crystallogr Sect E: Struct Rep Online 60:o303–o305
Nyokong T (2007) Effects of substituents on the photochemical and photophysical properties of main group metal phthalocyanines. Coord Chem Rev 251:1707–1722
Becker HG, Böttcher H, Dietz F, Rehorek D, Roewer G et al. (1983) Einführung in die Photochemie. Thieme Stuttgart
Klessinger M (1989) Physikalische organische Chemie. Verlag Chemie
Grofcsik A, Baranyai P, Bitter I, Csokai V, Kubinyi M et al (2004) Triple state properties of tetrasubstituted zinc phthalocyanine derivatives. J Mol Struct 704:11–15
Kumbhar PP, Dewang PM (2001) Eco-friendly pest management using monoterpenoids. I. Antifungal efficacy of thymol derivatives. J Sci Ind Res 60:645–648
Li Y, Wen J-m, Du C-j, Hu S-m, Chen J-x et al (2017) Thymol inhibits bladder cancer cell proliferation via inducing cell cycle arrest and apoptosis. Biochem Biophys Res Commun 491:530–536
Botelho M, Nogueira N, Bastos G, Fonseca S, Lemos T et al (2007) Antimicrobial activity of the essential oil from Lippia sidoides, carvacrol and thymol against oral pathogens. Braz J Med Biol Res 40:349–356
Braga PC, Dal Sasso M, Culici M, Bianchi T, Bordoni L, Marabini L (2006) Anti-inflammatory activity of thymol: inhibitory effect on the release of human neutrophil elastase. Pharmacology 77:130–136
Aeschbach R, Löliger J, Scott B, Murcia A, Butler J et al (1994) Antioxidant actions of thymol, carvacrol, 6-gingerol, zingerone and hydroxytyrosol. Food Chem Toxicol 32:31–36
Ma C, Tian D, Hou X, Chang Y, Cong F et al (2005) Synthesis and characterization of several soluble tetraphenoxy-substituted copper and zinc phthalocyanines. Synthesis 2005:741–748
Sobotta L, Lijewski S, Dlugaszewska J, Nowicka J, Mielcarek J, Goslinski T (2019) Photodynamic inactivation of Enterococcus faecalis by conjugates of zinc(II) phthalocyanines with thymol and carvacrol loaded into lipid vesicles. Inorg Chim Acta 489:180–190
Görlach B, Dachtler M, Glaser T, Albert K, Hanack M (2001) Synthesis and separation of structural isomers of 2(3),9(10),16(17),23(24)-tetrasubstituted phthalocyanines. Chem A Eur J 7:2459–2465
Atajanov R, Huraibat B, Odabaş Z, Özkaya AR (2023) Electrochemical, spectroelectrochemical, and electrocatalytic properties of novel soluble phthalocyanines containing peripheral thymoxy and chloride units. Inorg Chim Acta 547:121360
Fernandez JM, Bilgin MD, Grossweiner LI (1997) Singlet oxygen generation by photodynamic agents. J Photochem Photobiol B 37:131–140
Kubiak R, Janczak J, Śledź M, Bukowska E (2007) Comparative study of beryllium, magnesium and zinc phthalocyanine complexes with 4-picoline. Polyhedron 26:4179–4186
Acknowledgements
We are grateful to the Research Foundation of Marmara University, Commission of Scientific Research (BAPKO) for their support of this research as part of the project: FEN-C-YLP-120418-0164. In addition, we are very grateful to Assoc. Prof. Dr. Mehmet Pişkin for his skillful and fruitful collaboration.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
RA performed the synthesis, purification, and characterization of the starting compound and phthalocyanines in the article and write the manuscript. KK performed the photophysical and photochemical characterization of the phthalocyanines in the article. MD and ZO reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Atajanov, R., Khezami, K., Durmuş, M. et al. Synthesis, characterization, photophysical, and photochemical properties of novel phthalocyanines containing thymoxy groups as bioactive units. Transit Met Chem 48, 79–89 (2023). https://doi.org/10.1007/s11243-023-00525-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-023-00525-y