Abstract
Chest pain is one of the most common reasons for presentation to the Emergency Department and the ability to rapidly and correctly diagnose the minority of patients who have a myocardial infarction is of critical importance. We assessed the diagnostic performance of a multimarker strategy using heart-type fatty acid binding protein (H-FABP) in combination with a contemporary sensitive troponin (cTn) assay. We measured H-FABP (Randox) and a sensitive cTn (TnI-Ultra, Siemens) at baseline in 343 patients with chest pain enrolled in the prospective BWH-TIMI ED chest pain study. Final presenting diagnosis was adjudicated using all diagnostic data, including the local cTnI results, but reviewers were blinded to H-FABP and the sensitive cTn assays. The diagnostic accuracy of H-FABP and local cTn together (AUC 0.962) was superior to local cTn alone (AUC 0.910, p = 0.0009) with an especially marked improvement in early presenters (AUC 0.983 vs. 0.840, p = 0.0098). In contrast, when combined with the sensitive cTn assay, there was no significant difference in the AUC with H-FABP as compared with the sensitive cTn alone, either in the overall cohort (AUC 0.963 vs. 0.956, p = 0.23) or in early presenters (AUC 0.999 for both). In early presenters, the addition of H-FABP resulted in a NPV of 100 % when combined with either the local or sensitive cTn assay. In our study, the addition of H-FABP significantly enhanced the sensitivity and accuracy of diagnosis as compared to a prior-generation troponin assay alone, especially in patients who presented early. H-FABP but did improve overall diagnostic accuracy when added to a current-generation sensitive troponin assay; however, their combination offered the best NPV in early presenters. Further studies are needed to determine the utility a very rapid “rule out” of MI with a single blood draw of troponin and H-FABP at presentation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Acute non-traumatic chest pain is one of the most common reasons for presentation to the Emergency Department (ED), accounting for approximately 15 million visits per year in the United States and Europe [1]. The cost of chest pain evaluations in the United States has been estimated to exceed $3 billion annually [2]. While the most pressing concern is to rapidly and correctly diagnose the minority of patients with chest pain who have a myocardial infarction (MI) [3, 4], there is a growing imperative to avoid exposing patients to unnecessary testing and expense [5].
Cardiac troponin (cTn) is the cornerstone of diagnosis in this setting due to its tissue specificity, sensitivity, and established value for therapeutic decision-making [6, 7]. Emerging more sensitive troponin assays enable earlier detection of MI; [6, 8, 9] however, this increased sensitivity comes at cost of reduced clinical specificity, with more frequent elevation in patients without MI [10, 11]. In addition, since cTn assays appear unable to detect ischemia in the absence of necrosis, a multimarker strategy leveraging the diagnostic utility of confirmatory or complementary biomarkers, offers the potential to enhance diagnostic accuracy of acute coronary syndromes.
Heart-type fatty acid binding protein (H-FABP) is a small (15 kDA), stable protein that transports fatty acids from the cardiomyocyte cell membrane to mitochondria for oxidation [12]. Due to its small molecular size and cytoplasmic location, circulating levels of H-FABP are detectable as early as 30 min after an ischemic episode, potentially enabling very early recognition of myocardial damage [13].
The diagnostic performance of H-FABP has yet to be characterized in conjunction with a sensitive troponin assay in a general population of chest pain patients. We, therefore, assessed the diagnostic performance of H-FABP in combination with a contemporary sensitive troponin assay in a single center, prospective study of subjects presenting to the ED with chest pain suspicious for myocardial ischemia.
Methods
Study design and patient population
We designed a prospective, single center study coordinated by the Thrombolysis in Myocardial Infarction (TIMI) Study Group, Brigham and Women’s Hospital (BWH), Boston, MA. From July 2005 through May 2007, we enrolled 443 patients ≥18 years of age who presented within 24 h of onset of non-traumatic chest pain suspicious for acute coronary syndrome (ACS). Qualifying symptoms were to have occurred at rest and lasted at least 10 min or to have had a clear crescendo pattern culminating in symptoms occurring with minimal exertion. Exclusion criteria included recent hospitalization for ACS (with the previous 28 days), severe renal insufficiency (creatinine clearance <30 ml/min), and known chronic or acute systemic inflammatory disease or infection. All patients provided written informed consent and the study was approved by the hospital institutional review board.
Biochemical testing
Blood samples for determination of H-FABP and cTnI in serum were collected at enrollment, and serially (if not already discharged) at 4–6, 12–24, and 36–48 h and were stored at −70 °C until thawed for analysis. H-FABP was measured with a quantitative immunoturbidimetric assasy (Randox Laboratories Limited, Crumlin, Co. Antrim, UK) with a lower limit of detection 0.747 ng/ml. We used an optimal cut point of 5 ng/ml derived from receiver-operating characteristic (ROC) curves.
cTnI was measured using a commercially available sensitive assay (TnI-Ultra, Siemens Te(0.006 μg/l), an established 99th percentile reference limit of 40 ng/l (0.04 μg/l), and a total imprecision of 10 % at a concentration of 30 ng/l (0.03 μg/l) [14]. A prior-generation cTnI assay (Centaur cTnI, Siemens) was obtained locally as part of clinical care. This assay has a lower limit of detection of 30 ng/l (0.03 μg/l), an established 99th percentile reference limit of 100 ng/l (0.1 μg/l) and a total imprecision of 20 % at that concentration.
Adjudication of final diagnosis
Each patient’s final presenting diagnosis were adjudicated by a clinical end points committee (CEC) comprised of two independent reviewers using established criteria for myocardial infarction [7] and all available diagnostic data during the hospitalization, including the local cTnI results. Reviewers were blinded to the results of the H-FABP and sensitive cTnI assays. If there was disagreement about the final diagnosis, a third reviewer refereed.
Presenting diagnoses was adjudicated and categorized as follows: acute coronary ischemia (MI, UA, or stable angina); non-coronary acute cardiopulmonary processes (e.g. myocarditis, non-ischemic heart failure, pulmonary embolism); and non-cardiac chest pain. MI was further categorized into STEMI, NSTEMI or unknown, and by the Universal MI Classification system: type 1 (spontaneous), type 2 (demand), type 3 (sudden cardiac death), type 4a (PCI related), type 4b (stent thrombosis), type 5 (CABG related) [7]. ACS was defined as MI or UA.
Statistical analysis
Continuous variables are presented as medians (interquartile range) and categorical variables as numbers and percentages. Baseline characteristics were compared using the χ2 test for categorical variables and the Wilcoxon rank-sum test for continuous variables. Sensitivity, specificity, positive and negative predictive values were calculated using the 99th percentile cut point for each assay. The diagnostic accuracy of the assays at baseline were compared using the area under the curve derived from ROC curves [15]. McNemar’s test was used to compare sensitivity of the troponin assays alone compared with the troponin assay plus H-FABP. Two-tailed P values less than 0.05 were considered significant. All analyses were performed using STATA version 12.1 (StataCorp LP, College Station, Texas).
Results
Baseline characteristics
A total of 443 patients were enrolled; of these, the 343 (77 %) patients with complete samples available (H-FABP, local troponin, and cTnI) were included in these analyses. Patients were mostly male (60 %), non-smokers (83 %), and approximately one-third had a prior history of MI (30 %) or PCI (33 %). The final adjudicated diagnoses in the analysis cohort are listed in Table 1. A total of 91 (27 %) patients had a final adjudicated diagnosis of MI with the majority (90 %) being Type 1 (spontaneous) events, and very few of them (n = 13, 14 % of MIs) presenting with ST-elevation. There were 35 subjects (10 %) with a final adjudicated diagnosis of unstable angina. There were 155 (45 %) patients with non-cardiac diagnoses, the largest components being gastrointestinal (n = 38, 25 %) and musculoskeletal (n = 16, 10 %). There were 48 (14 %) patients with non-coronary acute cardiopulmonary chest pain processes (NCACP).
There were no significant differences in baseline characteristics between patients included in this cohort and those not included (Supplemental Table 1), with the exception of a slight difference in weight. The median concentration (25th and 75th percentile) of H-FABP at baseline was 3.54 ng/ml (2.2–5.69 ng/ml). Initial H-FABP levels were elevated (>5 ng/ml) at baseline in 111 (32 %) of the 343 patients (Table 2 ). H-FABP was more likely to be elevated in patients who were older, had a history of hypertension, CABG, impaired renal function, and in those who presented with ECG changes.
Diagnostic accuracy at presentation
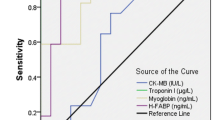
The baseline performance characteristics of individual assays for the diagnosis of MI are shown in Table 3. Troponin measured at baseline using the local assay had a sensitivity of 77 % (95 % CI 67–85) and a specificity of 97 % (95 % CI 94–99). The sensitive cTnI assay was substantially more sensitive for the diagnosis of MI than local troponin (92, 95 % CI 85–97) albeit less specific (92 %, 95 % CI 88–95), translating into a higher negative predictive value (NPV) (97 vs. 92 %) and a lower positive predictive value (PPV) (80 vs. 90 %). Considered alone, H-FABP was less sensitive (63 %, 95 % CI 52–73) and specific (79, 95 % CI 73–84) than either cTn assay.
The current-generation sensitive cTnI assay demonstrated the highest diagnostic accuracy (AUC of 0.956) followed by local troponin (AUC 0.910) and H-FABP (AUC 0.780) (Table 3). When restricted to patients presenting to the emergency department within 6 h of symptom onset (“early presenters”), the diagnostic performance of H-FABP (AUC 0.877) improved substantially and was comparable to local cTnI (p = 0.67) but was lower than that of the sensitive cTnI assay (p = 0.007).
Multimarker diagnostic accuracy at presentation
We also assessed whether there was additional diagnostic value in combining H-FABP and cTn (Table 4). As compared to the performance of local cTn alone, the addition of H-FABP significantly increased clinical sensitivity for MI (77–92 %, p = 0.008) and NPV (92–97 %). However, when compared to the performance of sensitive cTnI, the combined use of H-FABP and cTnI resulted in similar sensitivity (92–93 %, p = 0.78) and NPV (97 % for both). Among early presenters, the addition of H-FABP yielded a NPV of 100 % when combined with either local or the sensitive cTnI assay. The addition of H-FABP resulted in the detection of 14 additional MIs (20 % increase) when compared to local cTnI alone, and 1 additional MI (1 % increase) compared to the sensitive cTnI assay alone.
The diagnostic accuracy of H-FABP combined with the local cTn (AUC 0.962) was superior to that of local cTn alone (AUC 0.910, p = 0.0009; Table 4). This improvement was even more marked in early presenters (AUC 0.983 vs. 0.840, p = 0.0098). In contrast, there was no significant difference in accuracy when H-FABP was added to sensitive cTnI either in the overall cohort (AUC 0.963 vs. 0.956, p = 0.23) or in early presenters (AUC 0.999 for both). Use of either a positive H-FABP or sensitive cTnI to diagnose MI reduced clinical specificity compared with sensitive cTnI alone (73 vs. 92 %, p < 0.001). Moreover, clinical specificity was significantly increased by a strategy requiring both biomarkers to be positive (97 vs. 91 %, p = 0.02).
We performed additional sensitivity analyses on the diagnostic accuracy of H-FABP alone and in combination with troponin excluding STEMI patients, in whom clinical diagnosis is less reliant on circulating biomarkers (Supplemental Tables 2, 3). There were no qualitative differences observed.
Discussion
We evaluated the diagnostic performance of H-FABP in a broad population of patients presenting to the ED with chest pain. When used in combination with a prior generation assay for cTnI, H-FABP improved diagnostic accuracy, substantially enhancing the NPV over the prior generation cTn alone. However, when assessed in conjunction with a current-generation sensitive assay for cTnI, the negative predictive value of testing was modestly and non-significantly enhanced. Therefore, the findings from our cohort do not suggest a diagnostic advantage of testing for H-FABP in conjunction with contemporary sensitive assays for cTn. This study did not address the potential application of H-FABP for enhanced risk stratification as demonstrated in our previous work [16].
H-FABP in suspected or established acute coronary syndromes
Because of its small molecular size, H-FABP has been hypothesized to enable very early detection of myocardial [13]. Studies of H-FABP as an early marker of acute MI pre-date the widespread use of cTn [17–19] Subsequent studies comparing the performance of H-FABP in combination with prior generation assays for cTn suggested that H-FABP may enhance diagnostic accuracy for MI, particularly in patients presenting early [20–22].
In our study, the addition of H-FABP at presentation to the ED significantly enhanced the sensitivity and accuracy of diagnosis as compared to using a prior-generation troponin assay alone, markedly so in patients who presented early. However, H-FABP did not offer significant additional diagnostic information when added to a current generation sensitive assay for cTn, primarily due to the excellent performance of high-sensitivity troponin alone in a cohort that presented predominantly more than 3 h after onset of symptoms. Our results are consistent with the limited other data available comparing the addition of H-FABP to sensitive troponin assays for diagnosis of MI [23].
The ability to rapidly and correctly triage the majority of patients without MI for safe discharge to home is a priority to ensure efficient allocation of resources in the ED environment where there is a low tolerance for “missed MI’s” [24–26] Standard protocols have included serial cTn sampling over 6–12 h because the sensitivity of conventional assays for cTn was insufficient in patients presenting early after symptom onset. Recent studies suggest an approach using two closely spaced high-sensitivity troponin measurements that calculate an absolute change in concentration equal to 50 % of the 99th percentile as an additional diagnostic criterion along with a value >99th percentile [27], although there are several potential limitations that have to be addressed before widespread clinical adoption [28]. In early presenters, the addition of H-FABP resulted in an NPV of 100 % when used with either prior-generation troponin or a contemporary sensitive assay for troponin. This strategy is interesting, as it suggests a potential pathway to rule out MI with a single blood draw at presentation.
Our present study did not evaluate whether there is any incremental prognostic value of H-FABP when added to cTn. We have previously shown that elevated levels of H-FABP at baseline are associated with an increased risk of death and major cardiac events in patients with ACS independent of established clinical risk factors and biomarkers [16]. Moreover, a subsequent study using a contemporary sensitive assay for cTnI confirmed the finding that H-FABP, measured at presentation with ACS, is an independent predictor of both mortality and recurrent MI [29].
Limitations
There are several limitations of this analysis. We were unable to assess the very early performance of H-FABP in combination with troponin as we had very few patients who presented within 0–3 h from symptom onset. This study was focused on the diagnostic performance of H-FABP and thus we are unable to comment on the prognostic implications of H-FABP in this cohort. Because our study enrolled patients with a clinical suspicion for acute coronary syndrome, the prevalence of MI was higher than among unselected patients with non-traumatic chest pain in the US. Additionally, we used an FABP cut point that was optimized to our study population. However, these latter two limitations are more likely to have resulted in an overestimate rather than an underestimate of the diagnostic potential of H-FABP for MI.
Conclusions
The measurement of baseline H-FABP did not improve upon the overall diagnostic accuracy of a contemporary sensitive assay for cTn. Additional studies will clarify whether there is a role for H-FABP, leveraging its contribution for NPV, for a very rapid “rule out” of MI with a single blood draw at presentation.
References
Nawar EW, Niska RW, Xu J (2007) National hospital ambulatory medical care survey: 2005 emergency department summary. Adv Data 386:1–32
Amsterdam EA, Lewis WR, Yadlapalli S (1999) Evaluation of low-risk patients with chest pain in the emergency department: value and limitations of recent methods. Cardiol Rev 7:17–26
Antman EM, Hand M, Armstrong PW, Bates ER, Green LA, Halasyamani LK, Hochman JS, Krumholz HM, Lamas GA, Mullany CJ, Pearle DL, Sloan MA, Smith SC Jr., 2004 Writing Committee Members, Anbe DT, Kushner FG, Ornato JP, Jacobs AK, Adams CD, Anderson JL, Buller CE, Creager MA, Ettinger SM, Halperin JL, Hunt SA, Lytle BW, Nishimura R, Page RL, Riegel B, Tarkington LG, Yancy CW. (2008) 2007 Focused update of the ACC/AHA 2004 guidelines for the management of patients with st-elevation myocardial infarction: a report of the American college of cardiology/american heart association task force on practice guidelines: developed in collaboration with the canadian cardiovascular society endorsed by the American academy of family physicians: 2007 writing group to review new evidence and update the ACC/AHA 2004 guidelines for the management of patients with ST-elevation myocardial infarction, writing on behalf of the 2004 writing committee. Circulation 117: 296–329
Anderson JL, Adams CD, Antman EM, Bridges CR, Califf RM, E. CD Jr., Chavey WE, 2nd, Fesmire FM, Hochman JS, Levin TN, Lincoff AM, Peterson ED, Theroux P, Wenger NK, Wright RS, C. SS Jr., Jacobs AK, Adams CD, Anderson JL, Antman EM, Halperin JL, Hunt SA, Krumholz HM, Kushner FG, Lytle BW, Nishimura R, Ornato JP, Page RL, Riegel B. (2007) ACC/AHA 2007 guidelines for the management of patients with unstable angina/non-ST-Elevation myocardial infarction: a report of the American college of cardiology/American heart association task force on practice guidelines (writing committee to revise the 2002 guidelines for the management of patients with unstable angina/non-ST-elevation myocardial infarction) developed in collaboration with the American college of emergency physicians, the society for cardiovascular angiography and interventions, and the society of thoracic surgeons endorsed by the American association of cardiovascular and pulmonary rehabilitation and the society for academic emergency medicine. J Am Coll Cardiol 50: e1–e157
Polanczyk CA, Kuntz KM, Sacks DB, Johnson PA, Lee TH (1999) Emergency department triage strategies for acute chest pain using creatine kinase-MB and troponin I assays: a cost-effectiveness analysis. Ann Intern Med 131:909–918
Morrow DA, Antman EM (2009) Evaluation of high-sensitivity assays for cardiac troponin. Clin Chem 55:5–8
Thygesen K, Alpert JS, White HD (2007) Universal definition of myocardial infarction. Eur Heart J 28:2525–2538
Jaffe AS, Babuin L, Apple FS (2006) Biomarkers in acute cardiac disease: the present and the future. J Am Coll Cardiol 48:1–11
Apple FS, Jesse RL, Newby LK, Wu AH, Christenson RH, Cannon CP, Francis G, Morrow DA, Ravkilde J, Storrow AB, Tang W, Jaffe AS, Mair J, Ordonez-Llanos J, Pagani F, Panteghini M, Tate J (2007) National academy of clinical biochemistry and IFCC committee for standardization of markers of cardiac damage laboratory medicine practice guidelines: analytical issues for biochemical markers of acute coronary syndromes. Clin Chem 53:547–551
Keller T, Zeller T, Peetz D, Tzikas S, Roth A, Czyz E, Bickel C, Baldus S, Warnholtz A, Frohlich M, Sinning CR, Eleftheriadis MS, Wild PS, Schnabel RB, Lubos E, Jachmann N, Genth-Zotz S, Post F, Nicaud V, Tiret L, Lackner KJ, Munzel TF, Blankenberg S (2009) Sensitive troponin I assay in early diagnosis of acute myocardial infarction. N Engl J Med 361:868–877
Reichlin T, Hochholzer W, Bassetti S, Steuer S, Stelzig C, Hartwiger S, Biedert S, Schaub N, Buerge C, Potocki M, Noveanu M, Breidthardt T, Twerenbold R, Winkler K, Bingisser R, Mueller C (2009) Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. N Engl J Med 361:858–867
Glatz JF, van Bilsen M, Paulussen RJ, Veerkamp JH, van der Vusse GJ, Reneman RS (1988) Release of fatty acid-binding protein from isolated rat heart subjected to ischemia and reperfusion or to the calcium paradox. Biochim Biophys Acta 961:148–152
Kleine AH, Glatz JF, Van Nieuwenhoven FA, Van der Vusse GJ (1992) Release of heart fatty acid-binding protein into plasma after acute myocardial infarction in man. Mol Cell Biochem 116:155–162
Melanson SE, Morrow DA, Jarolim P (2007) Earlier detection of myocardial injury in a preliminary evaluation using a new troponin I assay with improved sensitivity. Am J Clin Pathol 128:282–286
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
O’Donoghue M, de Lemos JA, Morrow DA, Murphy SA, Buros JL, Cannon CP, Sabatine MS (2006) Prognostic utility of heart-type fatty acid binding protein in patients with acute coronary syndromes. Circulation 114:550–557
Ishii J, Wang JH, Naruse H, Taga S, Kinoshita M, Kurokawa H, Iwase M, Kondo T, Nomura M, Nagamura Y, Watanabe Y, Hishida H, Tanaka T, Kawamura K (1997) Serum concentrations of myoglobin vs human heart-type cytoplasmic fatty acid-binding protein in early detection of acute myocardial infarction. Clin Chem 43:1372–1378
Okamoto F, Sohmiya K, Ohkaru Y, Kawamura K, Asayama K, Kimura H, Nishimura S, Ishii H, Sunahara N, Tanaka T (2000) Human heart-type cytoplasmic fatty acid-binding protein (H-FABP) for the diagnosis of acute myocardial infarction. Clinical evaluation of H-FABP in comparison with myoglobin and creatine kinase isoenzyme MB. Clin Chem Lab Med 38:231–238
Seino Y, Ogata K, Takano T, Ishii J, Hishida H, Morita H, Takeshita H, Takagi Y, Sugiyama H, Tanaka T, Kitaura Y (2003) Use of a whole blood rapid panel test for heart-type fatty acid-binding protein in patients with acute chest pain: comparison with rapid troponin T and myoglobin tests. Am J Med 115:185–190
McMahon CG, Lamont JV, Curtin E, McConnell RI, Crockard M, Kurth MJ, Crean P, Fitzgerald SP (2012) Diagnostic accuracy of heart-type fatty acid-binding protein for the early diagnosis of acute myocardial infarction. Am J Emerg Med 30:267–274
McCann CJ, Glover BM, Menown IB, Moore MJ, McEneny J, Owens CG, Smith B, Sharpe PC, Young IS, Adgey JA (2008) Novel biomarkers in early diagnosis of acute myocardial infarction compared with cardiac troponin T. Eur Heart J 29:2843–2850
Haltern G, Peiniger S, Bufe A, Reiss G, Gulker H, Scheffold T (2010) Comparison of usefulness of heart-type fatty acid binding protein versus cardiac troponin T for diagnosis of acute myocardial infarction. Am J Cardiol 105:1–9
Kagawa Y, Toyofuku M, Masaoka Y, Muraoka Y, Okimoto T, Otsuka M, Tamekiyo H, Mito S, Kawase T, Yamane K, Senoo A, Amioka M, Shiode N, Hayashi Y (2011) Comparison of heart-type fatty acid binding protein and sensitive troponin for the diagnosis of early acute myocardial infarction. Int J Cardiol (in print)
Lindsell CJ, Anantharaman V, Diercks D, Han JH, Hoekstra JW, Hollander JE, Kirk JD, Lim SH, Peacock WF, Tiffany B, Wilke EK, Gibler WB, Pollack CV Jr. (2006) EMCREG-international i*trACS investigators. The internet tracking registry of acute coronary syndromes (i*trACS): a multicenter registry of patients with suspicion of acute coronary syndromes reported using the standardized reporting guidelines for emergency department chest pain studies. Ann Emerg Med 48: 666–77, 677. e1–9
Lee TH, Goldman L (2000) Evaluation of the patient with acute chest pain. N Engl J Med 342:1187–1195
Pope JH, Aufderheide TP, Ruthazer R, Woolard RH, Feldman JA, Beshansky JR, Griffith JL, Selker HP (2000) Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med 342:1163–1170
Reichlin T, Irfan A, Twerenbold R, Reiter M, Hochholzer W, Burkhalter H, Bassetti S, Steuer S, Winkler K, Peter F, Meissner J, Haaf P, Potocki M, Drexler B, Osswald S, Mueller C (2011) Utility of absolute and relative changes in cardiac troponin concentrations in the early diagnosis of acute myocardial infarction. Circulation 124:136–145
Apple FS, Morrow DA (2012) Delta cardiac troponin values in practice: are we ready to move absolutely forward to clinical routine? Clin Chem 58:8–10
Viswanathan K, Kilcullen N, Morrell C, Thistlethwaite SJ, Sivananthan MU, Hassan TB, Barth JH, Hall AS (2010) Heart-type fatty acid-binding protein predicts long-term mortality and re-infarction in consecutive patients with suspected acute coronary syndrome who are troponin-negative. J Am Coll Cardiol 55:2590–2598
Acknowledgments
The TIMI Study Group has received research Grant support from Abbott, Amgen, AstraZeneca, Beckman Coulter, BG Medicine, BRAHMS, Bristol-Myers Squibb, Buhlmann, Critical Diagnostics, CV Therapeutics, Daiichi Sankyo Co Ltd, Eli Lilly and Co, GlaxoSmithKline, Merck and Co, Nanosphere, Novartis Pharmaceuticals, Ortho-Clinical Diagnostics, Pfizer, Randox, Roche Diagnostics, Sanofi-Aventis, Siemens, and Singulex.
Conflict of interest
Dr. Ruff is an investigator and receives salary support from the TIMI Study Group. He has received remuneration for consulting from Alere, Beckman Coulter, Daiichi Sankyo, Boerhinger-Ingelheim. Dr. Bonaca is an investigator and receives salary support from the TIMI Study Group. Dr. Kosowsky, Mr. Conrad, Mrs. Murphy none to report. Dr. Jarolim has received research grant support from Beckman-Coulter, Amgen, BRAHMS, Daiichi Sankyo, Merck & Co., Abbott, Ortho-Clinical Diagnostics, Roche, and Siemens. Has received honoraria from Ortho-Clinical Diagnostics and served as a consultant to T2 Biosystems and Quanterix. Dr. Donahoe none to report. Dr. O’Donoghue is an investigator and receives salary support from the TIMI Study Group. She has received remuneration for consulting from Aegerion. Dr. Morrow is an investigator and receives salary from the TIMI Study Group. He has received remuneration for consulting from Beckman-Coulter, Boehringher Ingelheim, Critical Diagnostics, Genentech, Gilead, Instrumentation Laboratory, Johnson & Johnson, Merck, Roche Diagnostics, and Servier.
Author information
Authors and Affiliations
Corresponding author
Additional information
Christian T. Ruff, Marc P. Bonaca contributed equally to this work. Michelle L. O’Donoghue, David A. Morrow contributed equally to the oversight of this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ruff, C.T., Bonaca, M.P., Kosowsky, J.M. et al. Evaluation of the diagnostic performance of heart-type fatty acid binding protein in the BWH-TIMI ED chest pain study. J Thromb Thrombolysis 36, 361–367 (2013). https://doi.org/10.1007/s11239-013-0870-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-013-0870-7