Abstract
Although the overall prognosis for differentiated thyroid cancer (DTC) is excellent, a subset of patients will experience disease recurrence or may not respond to standard treatments. In recent years, DTC management has become more personalized in order to enhance treatment efficacy and avoid unnecessary interventions.
In this context, major guidelines recommend post-surgery staging to assess the risk of disease persistence, recurrence, and mortality. Consequently, risk stratification becomes pivotal in determining the necessity of postoperative adjuvant therapy, which may include radioiodine therapy (RIT), the degree of TSH suppression, additional imaging studies, and the frequency of follow-up.
However, the intermediate risk of recurrence is a highly heterogeneous category that encompasses various risk criteria, often combined, resulting in varying degrees of aggressiveness and a recurrence risk ranging from 5 to 20%. Furthermore, there is not enough long-term prognosis data for these patients. Unlike low- and high-risk DTC, the available literature is contradictory, and there is no consensus regarding adjuvant therapy.
We aim to provide an overview of intermediate-risk differentiated thyroid cancer, focusing on criteria to consider when deciding on adjuvant therapy in the current context of personalized approach, including molecular analysis to enhance the accuracy of patient management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Over the past decade or so, the management of thyroid cancer has undergone a significant transformation, primarily driven by the rising global incidence of thyroid cancer [1, 2]. This surge in incidence can be largely attributed to the increased accessibility of diagnostic tests like neck ultrasound (neck-US), computed tomography (CT), and magnetic resonance imaging (MRI). Additionally, ultrasound-guided aspiration punctures have played a pivotal role in the increased detection of low-risk papillary thyroid carcinomas (PTC) [3].
Although the overall prognosis for differentiated thyroid cancer (DTC) is excellent, a portion of DTC patients will experience disease recurrence or may not respond to standard treatments [4]. In order to improve treatment effectiveness and reduce unnecessary interventions, the management of DTC is evolving towards a more personalized approach.
In this context, the 2015 American Thyroid Association (ATA 2015) guidelines recommend evaluating the risk of disease persistence or recurrence, as well as the risk of mortality [1]. This assessment is crucial for determining the necessity of postoperative adjuvant therapy, which may include radioiodine therapy (RIT), the degree of TSH suppression, the need for additional imaging exams, and the frequency of follow-up [5].
The ATA recurrence risk stratification categorizes DTC tumors into low, intermediate, and high-risk groups (see Table 1 and Table 2). Among these, the intermediate-risk category is the second most prevalent, accounting for 25 to 35% of all cases. This group exhibits significant heterogeneity, with various risk criteria that can be combined, resulting in varying degrees of aggressiveness and a risk of recurrence that ranges from 5 to 20% [1]. Furthermore, there is not enough data on long-term prognosis for these patients. Unlike low- and high-risk DTC, the available literature is contentious, and there is no consensus regarding the prescription of adjuvant therapy [1, 5,6,7,8,9].
While currently accepted prognostic factors include patient's age, histological variant, initial disease extension, and the size of the primary tumor, a significant percentage of patients are not accurately classified solely based on these variables. Indeed, it is necessary to consider additional factors for a more individualized approach, including the experience of the surgeon and specialist physician, the geographical region, accessibility to healthcare services (including the availability of serum thyroglobulin (Tg) tests and the quality of neck-US), economic considerations, and the patient's values and preferences [10]. Molecular analysis also offers an effective way into a more personalized approach for patients in this category [11].
The aim of this publication is to review potential indicators that can aid in decision-making regarding the optimal surgical approach, the prescription of radioiodine therapy, and the recommended follow-up procedures, both initially and after restaging. It is important to note that this review is not applicable to children under 18 years of age.
2 Lobectomy or total thyroidectomy?
Due to the relatively low risk of persistent disease and recurrence of some thyroid cancers, lobectomy has emerged as a viable alternative to total thyroidectomy [1]. The primary objective is to optimize oncologic outcomes, specifically disease-specific and disease-free survival, while minimizing surgical morbidity and the long-term effects of treatment [12, 13]. Indeed, the complication rate associated with total thyroidectomy is higher than that of thyroid lobectomy, even when performed by experienced surgeons. Furthermore, this complication rate increases significantly for low-volume surgeons. In a study based on a dataset of 62,722 hospitalizations in the USA, the overall complication rate for lobectomy was 7.6% when performed by high-volume surgeons (those with over 99 cases per year) and 14.5% for total thyroidectomy. These numbers escalated to 11.8% and 24.1%, respectively, for low-volume surgeons [14].
Additionally, a prospective cohort study in patients with low to intermediate risk DTC showed that health-related quality of life (HRQOL) in the short-term postoperative period is better following lobectomy compared to total thyroidectomy, although there was no difference in long-term results (after 6–12 months) [15].
For the intermediate-risk group, the available data on patients' outcomes are conflicting, as summarized in Table 3. Some studies suggest that lobectomy does not result in any disadvantage in terms of recurrence-free survival (RFS) or disease-specific survival (DSS) [12, 13, 16,17,18]. In their respective studies, which included matched-pair analyses with propensity score matching for potential prognostic clinical and histological factors, Liu J et al. found that among 341 pairs of patients, there was no significant difference in the 10-year RFS rate between patients treated with lobectomy and total thyroidectomy (77.4% vs. 80.2%, p = 0.622), nor in the DSS rate (97.2% vs. 98.4%, p = 0.554) [12]. Similarly, Xu S et al. reported that among 265 pairs of patients, there was no significant difference in the 5-year RFS rate between patients treated with lobectomy and total thyroidectomy (92.3% vs. 93.7%; p = 0.77) [13].
On the other hand, a retrospective study, including 61,775 patients from the National Cancer Institute Database with DTC tumors ranging from 1.0 to 4.0 cm, indicated that when some histological features of intermediate risk such as extrathyroidal extension and lymph node metastases are present, patients tend to benefit from total thyroidectomy. In fact, prior to adjusting for these pathologic criteria, overall survival was slightly better for patients who underwent total thyroidectomy compared to those who had lobectomy at 5 years (97.2% vs. 96.9%), 10 years (92.9% vs. 91.4%), and 14 years (86.6% vs. 84.4%), with a p-value of 0.001 [14]. Colombo et al. also showed that structurally incomplete responses were significantly more frequent among patients treated with lobectomy (42% vs. 7% with total thyroidectomy; P = 0.010) [19]. Furthermore, an Italian multicentric retrospective study showed that 32% of intermediate-risk patients treated with lobectomy required completion thyroidectomy [20].
The decision regarding the need for total or completion thyroidectomy hinges on several critical factors, including the risk of cancer recurrence, the indication for adjuvant RIT, and the requirement for a highly sensitive tumor marker (Tg) to detect potential recurrences [21].
To better decide on the extent of thyroid surgery, it is important to consider both the preoperative characteristics of the tumors and the post-operative histological features. Pre-treatment characteristics that may warrant total thyroidectomy encompass the presence of a rapidly growing thyroid nodule, lymph node involvement, a history of radiation exposure and a family history of thyroid cancer, the both latter implying an increased risk of contralateral or multifocal disease. Additionally, if molecular testing is available and performed before surgery, the presence of BRAF and TERT promoter mutations may justify total thyroidectomy due to their aggressive nature [22]. For elderly patients, it is essential to account for comorbidities and assess the risk associated with a potential second surgery [23].
Intermediate-risk histological features that warrant considering adjuvant RIT often need a completion thyroidectomy (Table 4). These features encompass aggressive subtypes of PTC, such as hobnail, tall cell, or columnar carcinoma, as well as multicentric disease. In case of multicentric disease, there is a higher risk of residual disease in the contralateral lobe, making completion thyroidectomy more suitable [23, 24]. Specifically, the tall cell subtype of PTC is frequently associated with multifocal and bilateral disease, further underscoring the need for total thyroidectomy [25]. Conversely, microscopic extracapsular extension may not be an immediate indication for completion thyroidectomy. Numerous publications have shown that minimal extrathyroidal extension does not necessarily result in a worse prognosis [23, 26,27,28]. However, the presence of lymph node disease detected after lobectomy may justify completion thyroidectomy with neck dissection, depending on the size and number of lymph node metastases [23].
Despite careful preoperative patient selection and intraoperative evaluation to exclude extrathyroidal extension or lymph node metastases, approximately 30% of patients may ultimately require completion thyroidectomy. This is often due to higher-risk features that can only be confirmed after comprehensive histopathological and molecular analyses of the tumor [29]. Importantly, retrospective studies have shown that, if necessary, completion thyroidectomy does not result in significantly higher morbidity compared to an initial total thyroidectomy [23, 30, 31]
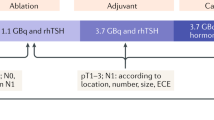
3 Radioiodine therapy (RIT) in intermediate risk: for whom? (Fig. 1)
The benefits of RIT for intermediate-risk thyroid cancer patients have been a subject of extensive discussion in the literature. The term "Radioiodine Therapy" broadly encompasses three main objectives: (I) ablation of remaining thyroid tissue to facilitate follow-up, (II) adjuvant treatment to eliminate suspicious neoplastic remnants undetected in imaging studies, with a focus on enhancing disease-free survival, and (III) treatment of macroscopic residual disease or known relapse, with the aim of improving overall survival and progression-free survival, either for curative or palliative purposes [3, 32].
Traditionally, most patients with DTC received postoperative RIT as standard care. However, the approach has changed in recent years into a more personalized management model, where RIT recommendations are based on clinical and pathological features, especially for the intermediate risk group. According to the ATA 2015 recommendations, RIT is not usually necessary for low-risk patients, highly recommended for high-risk patients, and should be considered on an individual basis for intermediate-risk patients.
The lack of standardized recommendations for intermediate-risk patients stems from the absence of prospective, multicenter, and randomized studies that could provide substantial evidence supporting adjuvant RIT in this group. The intermediate-risk category is notably diverse, incorporating a range of tumors with varying risk profiles and degrees of aggressiveness, making it challenging to establish universal RIT guidelines. Recurrence rates vary widely within this category, ranging from 5 to 20%. In a personalized approach, the decision to recommend adjuvant RIT and determine the appropriate radioactive iodine (131I) activity should consider several factors, including initial prognostic indicators, surgical reports, anatomopathological descriptions, post-operative Tg levels, and anti-thyroglobulin antibodies (ATg). Additional considerations include neck-US, other imaging exams, and the presence of relevant genetic markers.
Low to low-intermediate-risk patients do not appear to benefit from adjuvant RIT. Nonetheless, it can still be administered using lower doses of 131I, typically around 1.1 GBq after recombinant human TSH (rhTSH) stimulation, for ablative purposes [1, 33,34,35,36].
For instance, the impact of RIT remains a topic of debate for patients with intermediate risk and lymph node metastases, as the benefits of RIT appears to vary depending on the size and location of the involved lymph nodes. Some studies suggest potential benefits, even for micro papillary thyroid carcinoma (MPTC) with lymph node involvement, while others do not support this conclusion. Wang et al. [37] found no significant difference in 5-year central compartment nodal recurrence-free survival between PTC patients treated with RIT and those without RIT. A meta-analysis of 79 studies also failed to show a significant improvement in survival or reduced risk of persistent disease or recurrence in patients under 45 years old with microscopic central compartment lymph node metastases. Han K et al. [38] showed that the recurrence-free survival of intermediate-risk PTC patients did not significantly differ between those who received postoperative RIT and those who did not, especially for patients with negative extranodal extension and a low number of metastatic lymph nodes.
The role of minimal extrathyroidal extension (mETE) as a risk factor for persistent PTC in patients at the intermediate-risk category remains a subject of controversy. The British Thyroid Association (BTA) [39] and French Societies [40], recommend the use of RIT based on the presence of mETE. However, some studies suggest that although mETE may raise patients’ risk classification, it does not necessarily lead to a poor response to initial treatment [33, 41,42,43,44]. In fact, these studies have failed to demonstrate a discernible benefit of adjuvant RIT in reducing the risk of recurrence in case of mETE [33, 41,42,43,44]. A meta-analysis conducted by Diker-Cohen et al. [45] indicated that mETE does increase the risk of recurrence in patients with DTC, but the absolute increase in risk is relatively small, especially in patients with N0 disease, where the risk remains within the low-risk category for recurrence. They also showed that mETE has no impact on disease-related mortality and should not change tumor stage. Tran et al. [46] have shown that while mETE may not be an independent prognostic factor in multivariate analyses, it can provide valuable information about disease biology and serve as a surrogate marker for several closely associated adverse features.
For patients in the high-intermediate risk category (comprising patients aged over 55 years, those with tumors larger than 4 cm, a significant volume of lymph node disease, multiple lymph node involvement, lateral compartment lymph node metastases, and vascular invasion), adjuvant RIT with 3.7 GBq of 131I following rhTSH stimulation has demonstrated greater efficacy due to the increased risk of recurrence or distant metastasis, especially for cases with aggressive histology [1, 3, 47]. However, there may be exceptions for patients with aggressive histology, as available data can be inconclusive. In such cases, patients with BRAF mutations tend to benefit less from RIT, as they often have lower NIS expression, which affects their ability to concentrate 131I [48]. Nevertheless, some studies have indicated a reduction in the risk of recurrence after RIT for aggressive variants of PTC [41]. Kazaure et al. [49] and Regalbuto et al. [50] have shown that patients with diffuse sclerosing and tall cell histological subtypes who did not receive RIT had a 4.9- and 2.1-times higher likelihood of mortality compared to those who received RIT. For the time being, the overall risk–benefit assessment leans in favor of RIT.
The significance of vascular invasion (VI) as a predictor of distant metastasis and its potential role in RIT benefits are also subjects of debate, particularly in well-differentiated PTC. Different studies have yielded varying results. In their retrospective study Wreesmann et al. [51] found a significant association between VI and factors such as tumor size greater than 4 cm, extrathyroidal extension, distant metastasis, and radioiodine treatment. However, in the multivariate analysis, VI did not emerge as an independent predictor of distant recurrence-free survival. In contrast, Puga et al. [52] showed that lymphovascular invasion, including lymphatic invasion alone, was significantly linked to a higher likelihood of persistent or recurrent disease, especially among patients who did not undergo RIT, compared to those without lymph vascular invasion. This suggests that VI may have implications for the likelihood of disease recurrence. Creach et al. [53] also demonstrated the benefits of RIT for patients with VI, indicating a potential advantage in these cases. Furthermore, Al-Qahtani KH et al. [41] provided evidence that adjuvant RIT with a dose of 150 mCi or higher enhances Disease-Free Survival (DFS) in patients with thyroid microcarcinomas and VI. In summary, the potential benefit of adjuvant RIT in well-differentiated PTC cases characterized by the presence of VI and the absence of other aggressive features remains uncertain, with studies yielding mixed results [51]. Further research is needed to clarify the role of VI in guiding treatment decisions for these patients.
By reviewing the relevant literature, it becomes evident that clinicopathological features have primarily guided the RIT decision-making process. However, it is crucial to recognize that relying on these criteria alone may be insufficient to accurately predict patients' risks of recurrence and metastasis, as patients’ real-time status may be influenced by their initial treatment. This highlights the need to assess the postoperative status.
In this context, postoperative thyroglobulin (Tg) level plays a significant role in assessing the risk of recurrence and the effectiveness of RIT in patients with DTC. Several studies have consistently shown that elevated postoperative stimulated thyroglobulin (sTg) levels, typically above 1–2 ng/mL, are associated with an increased risk of recurrence [54,55,56]. Conversely, sTg levels below this threshold generally indicate a state of remission, suggesting a lower risk of recurrence [57, 58].
Tian et al. [59] conducted a propensity score matching (PSM) analysis to investigate the effectiveness of RIT in reducing the recurrence risk of intermediate-risk PTC in patients with low Tg levels. They found that RIT could decrease the recurrence risk of intermediate-risk PTC in patients with unstimulated Tg (usTg) levels of ≤ 1 ng/mL or sTg levels of ≤ 10 ng/mL. This suggests that RIT may be beneficial for these patients, even if their Tg levels are relatively low. A comprehensive meta-analysis [56] involving 3947 patients with DTC across fifteen studies, focusing on sTg values ranging from > 1–2 ng/mL and < 10 ng/mL, supported the notion that patients with postoperative sTg levels below 10 ng/mL, in the absence of anti-thyroglobulin antibodies (ATg), tend to have a significantly improved prognosis following RIT compared to those with higher sTg levels. Importantly, only 6% of these patients were found to have persistent disease after RIT, indicating a favorable response to treatment. Additionally, in a multicenter prospective study with a median follow-up of 10.6 months, it was observed that 80% of DTC patients with sTg levels ≥ 10 ng/mL, even when presenting more adverse clinicopathologic characteristics, maintained a non-structurally incomplete response after receiving 5.55 GBq (150 mCi) of 131I [35]. Gao et al. [60], in a retrospective analysis of 423 low- and intermediate-risk DTC patients, concluded that pretreatment sTg levels are essential indicators in RIT decision-making and efficacy evaluation. They found that DTC patients, particularly in the intermediate-risk group with sTg levels ≥ 10.0 ng/mL, would benefit from aggressive RIT. In summary, postoperative sTg level is a valuable indicator for assessing the risk of recurrence and guiding RIT decisions in patients with thyroid carcinoma. While elevated sTg levels are associated with a higher risk, some studies suggest that RIT can still be effective in cases with low to moderately elevated Tg levels, depending on the specific patients’ clinical characteristics. Although additional evidence is needed to establish an optimal sTg threshold for selecting patients who would benefit from RIT [36, 48], it is reasonable to consider omitting RIT for those with lower sTg levels below 1.0 ng/mL.
In addition to serum Tg levels and neck-US, radioiodine whole-body scintigraphy (WBS) performed with single-photon emission computed tomography-computed tomography (SPECT/CT) has emerged as a valuable tool for postoperative staging and patient selection for RIT. This approach offers several advantages, including enhanced image quality and improved capabilities for identifying foci of abnormal 131I uptake. These improvements make image interpretation and post-surgical staging more efficient, contributing to the assessment of risk of disease persistence and recurrence [61]. A study conducted by Avram et al. found residual disease or suspicious regional metastases in 35% of cases and distant metastases in 8% of cases among 320 patients, which modified the initial staging for 4% of younger patients and 25% of older patients [62]. In another study, pre-ablation WBS performed with SPECT/CT after surgery and before RIT, combined with serum thyroglobulin levels, modified the baseline risk stratification for 15% of patients, ultimately impacting the clinical management of 29.4% of the study's population [47]. Collectively, these modalities assist in patient selection for RIT and determining the most appropriate radioactive iodine activity for their treatment [62, 63]. A retrospective analysis conducted at a single center in Brazil, involving 301 intermediate-risk patients who underwent total thyroidectomy and RIT, showed that 17.3% of them had metastases detected through diagnostic 131I WBS (DxWBS) and/or post-therapy WBS (RxWBS). Specifically, DxWBS identified metastases in 10.6% of patients, including instances of unexpected distant metastases. When combined with single photon emission computed tomography-computer tomography (SPECT-CT), DxWBS more frequently detected 131I-avid metastases, especially in the case of lymph node metastases (13.1% vs. 4.2% for planar WBS, p = 0.015). These DxWBS findings led to modifications in patient management in 8.3% of cases. Importantly, a pretherapy sTg level below 1 ng/mL was associated with a low rate of false negatives for the presence of metastases (5.2%). This ability to exclude metastasis was further enhanced when combined with a negative DxWBS, as only 2.7% of patients with sTg levels below 1 ng/mL and a negative DxWBS showed metastases on post-therapy WBS [63].
In recent years, risk stratification based on molecular markers has emerged as a promising strategy for the personalized management of thyroid cancer [64]. This approach carries particular significance for DTC, given its high incidence and the well-established prognostic value of specific molecular markers [1,2,3, 5, 7, 32].
It has been observed that PTC carrying the BRAFV600E mutation often display a poor response to RIT [64,65,66,67]. This resistance to 131I treatment appears to be associated with the expression of genes involved in iodide uptake and metabolism, which are pivotal indicators of the differentiated state of follicular thyroid cells. In contrast, RAS-mutated PTCs exhibit a low MAPK-dependent transcriptional program due to negative feedback regulation. They also tend to maintain the expression of iodine metabolism genes and generally exhibit an affinity for radioiodine [68]. Nevertheless, a subset of BRAFV600E-mutated PTCs does respond to radioiodine therapy. In these cases, tumors still show relatively preserved expression of thyroid differentiation genes and higher expression of MicroRNAs targeting the transforming growth factor β (TGFβ) signaling pathway. Activation of this pathway suppresses the expression of thyroid-specific genes [69]. Moreover, in the ESTIMABL 2 trial, among patients initially categorized as "low-risk" DTC, over half of them carried the BRAFV600E mutation and 25% had the tall-cell variant, which reclassified some of them as intermediate-risk patients. Nevertheless, the trial demonstrated that a follow-up strategy without the use of 131I was noninferior to an ablation strategy [70]. An emerging and clinically significant question revolves around the reliability of molecular markers in predicting the resistance of thyroid carcinomas to 131I. Heterogeneous gene mutations may contribute to variations in tumor morphology, gene expression, and individual clinical characteristics. These markers have the potential to become pivotal prognostic factors for decision-making in radioiodine therapy. However, further evaluation is necessary to fully understand the impact of these mutations on disease progression.
Regarding the benefit of RIT in improving the survival of intermediate-risk patients, there is a lack of randomized clinical trials due to the indolent nature of DTC and its long-term survival. Conducting such trials would require an impractically large number of patients, extended follow-up periods, and significant associated costs [71]. In a nationally representative study using the Surveillance Epidemiology and End Results (SEER) database, Podnos et al.[72], and in a single-institution study with extensive patient follow-up conducted at the Mayo Clinic by Hay et al. [73], conclusive evidence supporting a survival benefit for RIT in the overall patient population was not found. It is important to note that neither study specifically examined the intermediate-risk patient subgroup. On the other hand, one study did demonstrate a survival benefit with RIT in a subset of intermediate-risk patients. Similarly, another study found a benefit in disease-free survival within a subset of patients with stage T2-T4 disease, but it was a smaller, single-center study [74]. Moreover, Ruel et al. [8] conducted a study involving 21,870 intermediate-risk patients defined by the American Thyroid Association risk criteria and American Joint Commission on Cancer staging (T3, N0, M0 or Mx, and T1–3, N1, M0, or Mx) over a mean follow-up of six years. They reported that RIT was associated with improved overall survival (OS) in all patients (p < 0.001). This benefit was also observed in subgroup analyses among patients younger than 45 years (n = 12,612, p = 0.002) and those 65 years and older (median OS 140 vs. 128 months, n = 2,122, p = 0.008). After adjusting for demographic and clinical factors, RIT was linked to a 29% reduction in the risk of death, with a hazard ratio of 0.71 (95% confidence interval 0.62–0.82, p < 0.001). For patients under 45 years, RIT was associated with a 36% reduction in the risk of death, with a hazard ratio of 0.64 (95% confidence interval 0.45–0.92, p = 0.016).
Table 4 provides a summary of the studies addressing controversial factors in recommending RIT for intermediate-risk DTC. Figure 1 illustrates the factors that can influence the decision to recommend or not RIT in intermediate-risk DTC patients.
4 Initial follow-up
Risk stratification for early disease persistence and recurrence should be used to guide management recommendations during the first 1–2 years of follow-up. However, it is crucial to adapt new recommendations based on the response to the initial therapy, aiming for a more precise and individualized long-term management and follow-up plan [5].
The most significant shift in risk estimation as a function of therapy response is observed within the intermediate-risk group. In this category, the estimated risk of recurrent or persistent structural disease, initially at 18%, diminishes significantly to just 2% in patients with an excellent treatment response. Similarly, a select few high-risk patients who respond exceptionally well to therapy experience a substantial reduction in the risk of recurrent or persistent structural disease, declining from 66 to 14%. Conversely, low-risk patients who exhibit an incomplete response to therapy see their risk of persistent structural disease increase from 3 to 13% [5]
Obtaining a clinical outcome with serum thyroglobulin (sTg) levels below 1 ng/mL and a negative imaging test is closely associated with a higher likelihood of showing no clinical evidence of disease. Furthermore, a combination of negative neck ultrasound (neck-US) and suppressed Tg levels below 1 ng/mL also presents a high probability of an excellent response, especially in low-risk (94%) and intermediate-risk (90%) patients within the initial 2 years of follow-up [5]. As a result, the necessity to conduct sTg testing should be approached with an individualized perspective, considering factors like the sensitivity and reliability of the available Tg assay, the quality of neck-US, and the cost associated with sTg testing.
Furthermore, several potentially crucial variables that could account for unexplained variations, beyond what current staging systems cover, include the impact of additional therapies administered after the 2-year re-stratification, complications stemming from therapy that may alter overall survival or affect the ability to detect recurrent disease, and the influence of the initial genotype on tumor aggressiveness, therapy response, or the ability to detect recurrent disease [75]After total thyroidectomy, the size of the presumably benign thyroid remnant is usually small, resulting in very low or often undetectable serum thyroglobulin (Tg) levels. Currently, even without postoperative radioiodine therapy, most patients can be followed up with serum Tg, with TSH suppression according to the stratification of risk of recurrence and disease persistence, and with imaging examination. For intermediate-risk patients, the TSH goal should be between 0.1–0.5 mU/L. An undetectable Tg level is comforting, as is a low but detectable level. In the latter case, the serum Tg should be monitored over time: a decline or stable Tg is reassuring, while an increase should prompt imaging to locate and treat the disease [1, 55]
5 Initial risk reassessment and prognosis
Numerous studies have consistently demonstrated that an excellent response to treatment, defined as the presence of a normal neck ultrasound (neck-US) and suppressed thyroglobulin (Tg) levels below 0.2 ng/mL or serum Tg (sTg) below 1 ng/mL [1], significantly reduces the initial risk of persistent or recurrent disease. This shift downgrades the risk level from intermediate to low, with an associated risk of only 1–2% [76]. Such patients, constituting 57–63% of intermediate-risk patients, can benefit from a less intensive follow-up regimen, which involves monitoring serum Tg and anti-thyroglobulin antibodies (ATg) every 12–24 months, undergoing cervical ultrasound (US) every 3–5 years, and having TSH levels maintained in the lower normal range (0.5 to 2 mUI/L) in the event of increasing tumor markers [77].
Conversely, patients exhibiting an incomplete structural response to treatment, accounting for 19–28% of intermediate-risk patients, face an increased risk of persistent disease at the conclusion of follow-up, despite additional treatment measures [76]. For these individuals, a comprehensive clinical and biological monitoring plan is recommended, involving frequent monitoring of serum Tg and ATg levels every 3–6 months. TSH suppression should be maintained at levels below 0.1 mUI/L, unless contraindicated [77]. Additionally, they should undergo more frequent cervical US assessments and other imaging staging procedures, including computed tomography (CT), contrast-enhanced magnetic resonance imaging (MRI), and [18F]2-fluoro-2-deoxy-D-glucose—positron emission tomography/computer tomography (18FDG-PET-CT), if there is suspicion of metastatic or progressive disease. For cases of persistent or recurrent disease, further treatment options such as reoperation for cervical lymph node dissection and/or a second round of Radioactive Iodine Therapy (RIT) may be necessary. The latter helps diagnose RIT-refractory disease, particularly when there is a lack of radioactive iodine (RAI) uptake in known secondary lesions or when 131I-positive lesions continue to progress after treatment [1, 77].
Patients with an indeterminate response to treatment, making up 8–23% of intermediate-risk patients, are typically defined by criteria such as suppressed thyroglobulin (Tg) levels at 0.2–1 ng/mL or serum Tg (sTg) at 1–10 ng/mL, positive stable/declining anti-thyroglobulin antibody (ATg) levels, or non-specific findings in imaging. Similarly, patients with an incomplete biochemical response to treatment, accounting for 21–22% of intermediate-risk patients, are characterized by criteria like negative imaging results, and suppressed Tg levels exceeding 1 ng/mL or sTg levels over 10 ng/mL, along with positive rising ATg levels. While these groups generally have a favorable prognosis [5, 78], the risk of recurrence can reach up to 20% [76]. Therefore, they should undergo clinical and biological monitoring, including serum Tg and ATg levels, every 6–12 months, with mild TSH suppression maintained within the range of 0.1 to 0.5 mUI/L. Additionally, more frequent cervical ultrasound (US) and other staging imaging should be considered when there is suspicion of metastatic or progressive disease [77].
Several risk factors have been associated with a non-excellent response to treatment in intermediate-risk Differentiated Thyroid Cancer (DTC). These factors include lateral cervical lymph node metastasis [79], the size of the largest focus of lymph node metastasis [80], larger tumor size [81], and post-operative stimulated serum thyroglobulin levels exceeding 10 ng/mL [28, 79]. The utility of BRAF status in dynamic risk stratification (DRS) analysis remains inconclusive due to conflicting data. While two studies suggest that BRAF mutation [82] and complete molecular testing [81] may enhance DRS, another study indicated that BRAF mutation did not significantly improve the differentiation of risk groups [83].
DRS offers a reliable prediction of patient status at the final follow-up in individuals treated with total thyroidectomy and Radioactive Iodine Therapy (RIT). Additionally, criteria for defining the response to treatment in patients treated with total thyroidectomy without RIT or lobectomy have been established, enabling the prediction of the risk of structural disease during the follow-up period [76, 77].
6 Prognostic role of the 18FDG-PET-CT and the molecular markers
Some intermediate-risk patients can benefit from undergoing 18FDG-PET-CT, not only when there is a well-established suspicion of persistent or recurrent disease during follow-up, but also at the time of the initial Radioiodine Therapy (RIT) [84]. This is particularly relevant for patients with aggressive histological subtypes, such as tall cell, columnar cell, squamous differentiation, diffuse sclerosing, hobnail, and solid/trabecular variants, including tumors harboring a BRAF V600E mutation.
Persistent disease identified on pre-ablation 18FDG-PET-CT scans has been observed in 13 to 33% of intermediate-risk patients [85, 86], with a higher frequency (53%) noted in cases with aggressive histology [87]. Pre- ablation 18FDG-PET-CT can be performed under TSH stimulation, resulting in a slight increase in sensitivity, though with limited clinical benefit. The results should be compared to Radioiodine Whole-Body Scintigraphy (RAI-WBS) findings. Tumors with aggressive histology are more likely to exhibit FDG-positive but iodine-negative lesions, indicating a refractory disease [84, 88].
18FDG-PET-CT also plays a prognostic role, both during the initial treatment and follow-up, especially when there is a rising trend in suppressed thyroglobulin (Tg) or anti-thyroglobulin antibody (ATg) levels. Its sensitivity increases with higher serum Tg levels and larger tumor foci. However, in cases of highly proliferative disease and those with a short Tg doubling time, the sensitivity can be very high, even in the presence of low Tg levels [88].
If available, molecular testing can provide valuable risk stratification for intermediate-risk patients. The BRAF mutation, which is the most frequent mutation in Differentiated Thyroid Cancer (DTC) (~ 60%) and present in almost 90% of the tall cell variant [89], is a marker of more aggressive tumor behavior, associated with lymph node metastasis, extrathyroidal extension (ETE), and higher recurrence rates. As a result, small tumors in the 1–4 cm range are considered at intermediate risk of recurrence when the BRAF mutation is present, and at high risk if there is a concomitant BRAF and TERT promoter mutation [1, 77].
Additionally, Yip et al. propose a molecular-based risk stratification of DTC using a commercially available molecular testing panel (ThyroSeq version 3). The intermediate-risk molecular-based group includes BRAF V600E, other BRAF-like alterations, and DNA copy number alterations, but lacks additional molecular features to further refine this cohort. In their case–control study, the molecular-based risk classification accurately stratified the risk of distant metastasis, which aligned with the expected risk of structural disease recurrence for the ATA risk groups [90].
Recently, Pizzimenti et al. suggested that in intermediate-risk patients, assessing the BRAF V600E mutation could be useful at the initial treatment stage to identify patients who might benefit from higher doses of 131I or other potential therapies, such as immunotherapy, particularly for those with higher expression of PD-L1 [82].
Finally, molecular testing can also predict tumor behavior and RIT refractoriness, as tumors harboring a BRAF V600E mutation exhibit significantly lower differentiation scores in The Cancer Genome Atlas (TCGA) cohort when compared to RAS tumors [91].
7 Conclusion
Intermediate-risk differentiated thyroid cancers encompass a highly diverse group of patients, characterized by varying combinations of risk factors, resulting in different levels of disease aggressiveness. Managing this patient category is a subject of debate, as there is no consensus regarding the recommendation and benefits of adjuvant therapy, given the limited availability of long-term prognosis data. In the current era of personalized medicine, decisions regarding treatment should consider multiple factors, including the patient's age, histological subtype, initial disease extent, and the size of the primary tumor. Additionally, the surgeon's experience, geographical location, access to healthcare services, the availability of serum thyroglobulin tests, the quality of neck ultrasound, economic considerations, and the patient's personal values and preferences all play a role in the decision-making process. Molecular analysis is emerging as a valuable tool for tailoring treatment to the individual needs of patients within this group.
Data availability statement
The contribution is original and unpublished, and is not being evaluated for publication by another journal.
References
Haugen BR, Alexander EK, Bible KC, et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The american thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133. https://doi.org/10.1089/thy.2015.0020.
Sherma SI. Thyroid carcinoma. The Lancet. 2003;361(9356):501–11. https://doi.org/10.1016/S0140-6736(03)12488-9.
Pacini F, Fuhrer D, Elisei R, et al. ETA Consensus Statement: What are the indications for post-surgical radioiodine therapy in differentiated thyroid cancer? Eur Thyroid J. 2022;11(1). https://doi.org/10.1530/ETJ-21-0046.
Rosário PW, Ward LS, Carvalho GA, et al. Thyroid nodules and differentiated thyroid cancer: update on the Brazilian consensus. Arq Bras Endocrinol Metabol. 2013;57(4):240–64. https://doi.org/10.1590/S0004-27302013000400002.
Tuttle RM, Tala H, Shah J, et al. Estimating Risk of Recurrence in Differentiated Thyroid Cancer After Total Thyroidectomy and Radioactive Iodine Remnant Ablation: Using Response to Therapy Variables to Modify the Initial Risk Estimates Predicted by the New American Thyroid Association Staging System. Thyroid. 2010;20(12):1341–9. https://doi.org/10.1089/thy.2010.0178.
Ballal S, Soundararajan R, Garg A, Chopra S, Bal C. Intermediate-risk differentiated thyroid carcinoma patients who were surgically ablated do not need adjuvant radioiodine therapy: long-term outcome study. Clin Endocrinol (Oxf). 2016;84(3):408–16. https://doi.org/10.1111/cen.12779.
Tuttle RM, Sabra MM. Selective use of RAI for ablation and adjuvant therapy after total thyroidectomy for differentiated thyroid cancer: A practical approach to clinical decision making. Oral Oncol. 2013;49(7):676–83. https://doi.org/10.1016/j.oraloncology.2013.03.444.
Ruel E, Thomas S, Dinan M, Perkins JM, Roman SA, Sosa JA. Adjuvant Radioactive Iodine Therapy Is Associated With Improved Survival for Patients With Intermediate-Risk Papillary Thyroid Cancer. J Clin Endocrinol Metab. 2015;100(4):1529–36. https://doi.org/10.1210/jc.2014-4332.
Kim SK, Woo JW, Lee JH, et al. Radioactive iodine ablation may not decrease the risk of recurrence in intermediate-risk papillary thyroid carcinoma. Endocr Relat Cancer. 2016;23(5):367–76. https://doi.org/10.1530/ERC-15-0572.
Padovani RDP, Chablani SV, Tuttle RM. Radioactive iodine therapy: multiple faces of the same polyhedron. Arch Endocrinol Metab. Published online May 13, 2022. https://doi.org/10.20945/2359-3997000000461.
Zafon C, Díez JJ, Galofré JC, Cooper DS. Nodular Thyroid Disease and Thyroid Cancer in the Era of Precision Medicine. Eur Thyroid J. 2017;6(2):65–74. https://doi.org/10.1159/000457793.
Liu J, Zhang Z, Huang H, et al. Total thyroidectomy versus lobectomy for intermediate-risk papillary thyroid carcinoma: A single-institution matched-pair analysis. Oral Oncol. 2019;90:17–22. https://doi.org/10.1016/j.oraloncology.2019.01.010.
Xu S, Huang H, Huang Y, et al. Comparison of Lobectomy vs Total Thyroidectomy for Intermediate-Risk Papillary Thyroid Carcinoma With Lymph Node Metastasis. JAMA Surg. 2023;158(1):73. https://doi.org/10.1001/jamasurg.2022.5781.
Adam MA, Pura J, Gu L, et al. Extent of Surgery for Papillary Thyroid Cancer Is Not Associated With Survival: An Analysis of 61,775 Patients. Ann Surg. 2014;260(4):601–7. https://doi.org/10.1097/SLA.0000000000000925.
Chen W, Li J, Peng S, et al. Association of Total Thyroidectomy or Thyroid Lobectomy With the Quality of Life in Patients With Differentiated Thyroid Cancer With Low to Intermediate Risk of Recurrence. JAMA Surg. 2022;157(3):200. https://doi.org/10.1001/jamasurg.2021.6442.
Park S, Kim WG, Song E, et al. Dynamic Risk Stratification for Predicting Recurrence in Patients with Differentiated Thyroid Cancer Treated Without Radioactive Iodine Remnant Ablation Therapy. Thyroid. 2017;27(4):524–30. https://doi.org/10.1089/thy.2016.0477.
Momesso DP, Vaisman F, Yang SP, et al. Dynamic Risk Stratification in Patients with Differentiated Thyroid Cancer Treated Without Radioactive Iodine. J Clin Endocrinol Metab. 2016;101(7):2692–700. https://doi.org/10.1210/jc.2015-4290.
Vaisman F, Shaha A, Fish S, Michael TR. Initial therapy with either thyroid lobectomy or total thyroidectomy without radioactive iodine remnant ablation is associated with very low rates of structural disease recurrence in properly selected patients with differentiated thyroid cancer. Clin Endocrinol (Oxf). 2011;75(1):112–9. https://doi.org/10.1111/j.1365-2265.2011.04002.x.
Colombo C, De Leo S, Di Stefano M, et al. Total Thyroidectomy Versus Lobectomy for Thyroid Cancer: Single-Center Data and Literature Review. Ann Surg Oncol. 2021;28(8):4334–44. https://doi.org/10.1245/s10434-020-09481-8.
Dobrinja C, Samardzic N, Giudici F, et al. Hemithyroidectomy versus total thyroidectomy in the intermediate-risk differentiated thyroid cancer: the Italian Societies of Endocrine Surgeons and Surgical Oncology Multicentric Study. Updat Surg. 2021;73(5):1909–21. https://doi.org/10.1007/s13304-021-01140-1.
Hartl DM, Guerlain J, Breuskin I, et al. Thyroid Lobectomy for Low to Intermediate Risk Differentiated Thyroid Cancer. Cancers. 2020;12(11):3282. https://doi.org/10.3390/cancers12113282.
Vuong HG, Altibi AMA, Duong UNP, Hassell L. Prognostic implication of BRAF and TERT promoter mutation combination in papillary thyroid carcinoma—A meta-analysis. Clin Endocrinol (Oxf). 2017;87(5):411–7. https://doi.org/10.1111/cen.13413.
Ronen O, Robbins KT, Olsen KD, et al. Case for staged thyroidectomy. Head Neck. 2020;42(10):3061–71. https://doi.org/10.1002/hed.26393.
Ibrahim B, Forest VI, Hier M, Mlynarek AM, Caglar D, Payne RJ. Completion thyroidectomy: predicting bilateral disease. J Otolaryngol - Head Neck Surg. 2015;44(1):23. https://doi.org/10.1186/s40463-015-0076-4.
Michels JJ, Jacques M, Henry-Amar M, Bardet S. Prevalence and prognostic significance of tall cell variant of papillary thyroid carcinoma. Hum Pathol. 2007;38(2):212–9. https://doi.org/10.1016/j.humpath.2006.08.001.
Shaha AR. Extrathyroidal extension—what does it mean. Oral Oncol. 2017;68:50–2. https://doi.org/10.1016/j.oraloncology.2017.03.008.
Tam S, Amit M, Boonsripitayanon M, et al. Effect of Tumor Size and Minimal Extrathyroidal Extension in Patients with Differentiated Thyroid Cancer. Thyroid. 2018;28(8):982–90. https://doi.org/10.1089/thy.2017.0513.
Almeida MFOD, Couto JS, Ticly ALT, et al. The impact of minimal extrathyroidal extension in the recurrence of papillary thyroid cancer patients. Arch Endocrinol Metab. 2020;64(3):251–6. https://doi.org/10.20945/2359-3997000000245.
Craig SJ, Bysice AM, Nakoneshny SC, Pasieka JL, Chandarana SP. The Identification of Intraoperative Risk Factors Can Reduce, but Not Exclude, the Need for Completion Thyroidectomy in Low-Risk Papillary Thyroid Cancer Patients. Thyroid. 2020;30(2):222–8. https://doi.org/10.1089/thy.2019.0274.
Nicholson KJ, Teng CY, McCoy KL, Carty SE, Yip L. Completion thyroidectomy: A risky undertaking? Am J Surg. 2019;218(4):695–9. https://doi.org/10.1016/j.amjsurg.2019.07.014.
Gulcelik MA, Dogan L, Akgul GG, Güven EH, Ersöz GN. Completion Thyroidectomy: Safer than Thought. Oncol Res Treat. 2018;41(6):386–90. https://doi.org/10.1159/000487083.
Van Nostrand D. Selected Controversies of Radioiodine Imaging and Therapy in Differentiated Thyroid Cancer. Endocrinol Metab Clin North Am. 2017;46(3):783–93. https://doi.org/10.1016/j.ecl.2017.04.007.
Mallick U, Harmer C, Yap B, et al. Ablation with Low-Dose Radioiodine and Thyrotropin Alfa in Thyroid Cancer. N Engl J Med. 2012;366(18):1674–85. https://doi.org/10.1056/NEJMoa1109589.
Castagna MG, Cevenini G, Theodoropoulou A, et al. Post-surgical thyroid ablation with low or high radioiodine activities results in similar outcomes in intermediate risk differentiated thyroid cancer patients. Eur J Endocrinol. 2013;169(1):23–9. https://doi.org/10.1530/EJE-12-0954.
Du P, Jiao X, Zhou Y, et al. Low versus high radioiodine activity to ablate the thyroid after thyroidectomy for cancer: a meta-analysis of randomized controlled trials. Endocrine. 2015;48(1):96–105. https://doi.org/10.1007/s12020-014-0333-8.
Shengguang Y, Ji-Eun C, Lijuan HL. I-131 for Remnant Ablation in Differentiated Thyroid Cancer After Thyroidectomy: A Meta-Analysis of Randomized Controlled Evidence. Med Sci Monit. 2016;22:2439–50. https://doi.org/10.12659/MSM.896535.
Wang LY, Palmer FL, Migliacci JC, et al. Role of RAI in the management of incidental N1a disease in papillary thyroid cancer. Clin Endocrinol (Oxf). 2016;84(2):292–5. https://doi.org/10.1111/cen.12828.
Han K, Noh HM, Jeong HM, Lim YC. Is Postoperative Adjuvant Radioactive Iodine Ablation Therapy Always Necessary for Intermediate-Risk Papillary Thyroid Cancer Patients With Central Neck Metastasis? Ann Surg Oncol. 2021;28(12):7533–44. https://doi.org/10.1245/s10434-021-10164-1.
Perros P, Boelaert K, Colley S, et al. Guidelines for the management of thyroid cancer. Clin Endocrinol (Oxf). 2014;81(s1):1–122. https://doi.org/10.1111/cen.12515.
Zerdoud S, Giraudet AL, Leboulleux S, et al. Radioactive iodine therapy, molecular imaging and serum biomarkers for differentiated thyroid cancer: 2017 guidelines of the French Societies of Nuclear Medicine, Endocrinology, Pathology, Biology, Endocrine Surgery and Head and Neck Surgery. Ann Endocrinol. 2017;78(3):162–75. https://doi.org/10.1016/j.ando.2017.04.023.
AL-Qahtani KH, Al Asiri M, Tunio MA, et al. Adjuvant Radioactive iodine 131 ablation in papillary microcarcinoma of thyroid: Saudi Arabian experience. J Otolaryngol - Head Neck Surg. 2015;44(1):51. https://doi.org/10.1186/s40463-015-0108-0.
Kim HJ, Kim SW. Radioactive iodine ablation does not prevent recurrences in patients with papillary thyroid microcarcinoma. Clin Endocrinol (Oxf). 2013;79(3):445–445. https://doi.org/10.1111/cen.12131.
Ahmaddy F, Wenter V, Ilhan H, et al. Effects of the Minimal Extrathyroidal Extension on Early Response Rates after (Adjuvant) Initial Radioactive Iodine Therapy in PTC Patients. Cancers. 2020;12(11):3357. https://doi.org/10.3390/cancers12113357.
Forleo R, Grani G, Alfò M, et al. Minimal Extrathyroidal Extension in Predicting 1-Year Outcomes: A Longitudinal Multicenter Study of Low-to-Intermediate-Risk Papillary Thyroid Carcinoma (ITCO#4). Thyroid. 2021;31(12):1814–21. https://doi.org/10.1089/thy.2021.0248.
Diker-Cohen T, Hirsch D, Shimon I, et al. Impact of Minimal Extrathyroid Extension in Differentiated Thyroid Cancer: Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2018;103(6):2100–6. https://doi.org/10.1210/jc.2018-00081.
Tran B, Roshan D, Abraham E, et al. An Analysis of The American Joint Committee on Cancer 8th Edition T Staging System for Papillary Thyroid Carcinoma. J Clin Endocrinol Metab. 2018;103(6):2199–2206. https://doi.org/10.1210/jc.2017-02551.
Chow SM, Yau S, Kwan CK, Poon PCM, Law SCK. Local and regional control in patients with papillary thyroid carcinoma: specific indications of external radiotherapy and radioactive iodine according to T and N categories in AJCC 6th edition. Endocr Relat Cancer. 2006;13(4):1159–1172. https://doi.org/10.1677/erc.1.01320.
Sawka AM, Brierley JD, Tsang RW, et al. An Updated Systematic Review and Commentary Examining the Effectiveness of Radioactive Iodine Remnant Ablation in Well-Differentiated Thyroid Cancer. Endocrinol Metab Clin North Am. 2008;37(2):457–80. https://doi.org/10.1016/j.ecl.2008.02.007.
Kazaure HS, Roman SA, Sosa JA. Aggressive Variants of Papillary Thyroid Cancer: Incidence, Characteristics and Predictors of Survival among 43,738 Patients. Ann Surg Oncol. 2012;19(6):1874–80. https://doi.org/10.1245/s10434-011-2129-x.
C. Regalbuto, P. Malandrino, F. Frasca, et al. The tall cell variant of papillary thyroid carcinoma: Clinical and pathological features and outcomes. J Endocrinol Invest. 2013;36(4). https://doi.org/10.3275/8515.
Wreesmann VB, Nixon IJ, Rivera M, et al. Prognostic Value of Vascular Invasion in Well-Differentiated Papillary Thyroid Carcinoma. Thyroid. 2015;25(5):503–8. https://doi.org/10.1089/thy.2015.0052.
Puga FM, Al Ghuzlan A, Hartl DM, et al. Impact of lymphovascular invasion on otherwise low-risk papillary thyroid carcinomas: a retrospective and observational study. Endocrine. Published online August 28, 2023. https://doi.org/10.1007/s12020-023-03475-8.
Creach KM, Siegel BA, Nussenbaum B, Grigsby PW. Radioactive Iodine Therapy Decreases Recurrence in Thyroid Papillary Microcarcinoma. ISRN Endocrinol. 2012;2012:1–6. https://doi.org/10.5402/2012/816386.
Lamartina L, Grani G, Biffoni M, et al. Risk Stratification of Neck Lesions Detected Sonographically During the Follow-Up of Differentiated Thyroid Cancer. J Clin Endocrinol Metab. 2016;101(8):3036–44. https://doi.org/10.1210/jc.2016-1440.
Rondeau G, Fish S, Hann LE, Fagin JA, Tuttle RM. Ultrasonographically Detected Small Thyroid Bed Nodules Identified After Total Thyroidectomy for Differentiated Thyroid Cancer Seldom Show Clinically Significant Structural Progression. Thyroid. 2011;21(8):845–53. https://doi.org/10.1089/thy.2011.0011.
Webb RC, Howard RS, Stojadinovic A, et al. The Utility of Serum Thyroglobulin Measurement at the Time of Remnant Ablation for Predicting Disease-Free Status in Patients with Differentiated Thyroid Cancer: A Meta-Analysis Involving 3947 Patients. J Clin Endocrinol Metab. 2012;97(8):2754–63. https://doi.org/10.1210/jc.2012-1533.
Kim TY, Kim WB, Kim ES, et al. Serum Thyroglobulin Levels at the Time of 131 I Remnant Ablation Just after Thyroidectomy Are Useful for Early Prediction of Clinical Recurrence in Low-Risk Patients with Differentiated Thyroid Carcinoma. J Clin Endocrinol Metab. 2005;90(3):1440–5. https://doi.org/10.1210/jc.2004-1771.
Piccardo A, Arecco F, Puntoni M, et al. Focus on High-Risk DTC Patients: High Postoperative Serum Thyroglobulin Level Is a Strong Predictor of Disease Persistence and Is Associated to Progression-Free Survival and Overall Survival. Clin Nucl Med. 2013;38(1):18–24. https://doi.org/10.1097/RLU.0b013e318266d4d8.
Tian T, Qi Z, Huang S, Wang H, Huang R. Radioactive Iodine Therapy Decreases the Recurrence of Intermediate-Risk PTC With Low Thyroglobulin Levels. J Clin Endocrinol Metab. 2023;108(8):2033–41. https://doi.org/10.1210/clinem/dgad045.
Gao H, Huang J, Dai Q, Su J. Radioiodine (131I) treatment decision-making for low- and intermediate-risk differentiated thyroid cancer. Arch Endocrinol Metab. Published online January 18, 2023. https://doi.org/10.20945/2359-3997000000538.
Gulec SA, Ahuja S, Avram AM, et al. A Joint Statement from the American Thyroid Association, the European Association of Nuclear Medicine, the European Thyroid Association, the Society of Nuclear Medicine and Molecular Imaging on Current Diagnostic and Theranostic Approaches in the Management of Thyroid Cancer. Thyroid. 2021;31(7):1009–19. https://doi.org/10.1089/thy.2020.0826.
Avram AM, Esfandiari NH, Wong KK. Preablation 131-I Scans With SPECT/CT Contribute to Thyroid Cancer Risk Stratification and 131-I Therapy Planning. J Clin Endocrinol Metab. 2015;100(5):1895–902. https://doi.org/10.1210/jc.2014-4043.
Danilovic DLS, Coura-Filho GB, Recchia GM, et al. Is there a role for diagnostic scans in the management of intermediate-risk thyroid cancer? Endocr Relat Cancer. 2022;29(8):475–83. https://doi.org/10.1530/ERC-22-0038.
Xing M, Haugen BR, Schlumberger M. Progress in molecular-based management of differentiated thyroid cancer. The Lancet. 2013;381(9871):1058–69. https://doi.org/10.1016/S0140-6736(13)60109-9.
Agrawal N, Akbani R, Aksoy BA, et al. Integrated Genomic Characterization of Papillary Thyroid Carcinoma. Cell. 2014;159(3):676–90. https://doi.org/10.1016/j.cell.2014.09.050.
Sabra MM, Dominguez JM, Grewal RK, et al. Clinical Outcomes and Molecular Profile of Differentiated Thyroid Cancers With Radioiodine-Avid Distant Metastases. J Clin Endocrinol Metab. 2013;98(5):E829–36. https://doi.org/10.1210/jc.2012-3933.
Liu J, Liu R, Shen X, Zhu G, Li B, Xing M. The Genetic Duet of BRAF V600E and TERT Promoter Mutations Robustly Predicts Loss of Radioiodine Avidity in Recurrent Papillary Thyroid Cancer. J Nucl Med. 2020;61(2):177–82. https://doi.org/10.2967/jnumed.119.227652.
Xing M, Westra WH, Tufano RP, et al. BRAF Mutation Predicts a Poorer Clinical Prognosis for Papillary Thyroid Cancer. J Clin Endocrinol Metab. 2005;90(12):6373–9. https://doi.org/10.1210/jc.2005-0987.
Boucai L, Seshan V, Williams M, et al. Characterization of Subtypes of BRAF -Mutant Papillary Thyroid Cancer Defined by Their Thyroid Differentiation Score. J Clin Endocrinol Metab. 2022;107(4):1030–9. https://doi.org/10.1210/clinem/dgab851.
Leboulleux S, Bournaud C, Chougnet CN, et al. Thyroidectomy without Radioiodine in Patients with Low-Risk Thyroid Cancer. N Engl J Med. 2022;386(10):923–32. https://doi.org/10.1056/NEJMoa2111953.
Morris LF, Romero Arenas MA, Cerny J, et al. Streamlining variability in hospital charges for standard thyroidectomy: Developing a strategy to decrease waste. Surgery. 2014;156(6):1441–9. https://doi.org/10.1016/j.surg.2014.08.068.
Podnos YD, Smith DD, Wagman LD, Ellenhorn JDI. Survival in patients with papillary thyroid cancer is not affected by the use of radioactive isotope. J Surg Oncol. 2007;96(1):3–7. https://doi.org/10.1002/jso.20656.
Hay ID, Thompson GB, Grant CS, et al. Papillary Thyroid Carcinoma Managed at the Mayo Clinic during Six Decades (1940–1999): Temporal Trends in Initial Therapy and Long-term Outcome in 2444 Consecutively Treated Patients. World J Surg. 2002;26(8):879–85. https://doi.org/10.1007/s00268-002-6612-1.
Jonklaas J, Sarlis NJ, Litofsky D, et al. Outcomes of Patients with Differentiated Thyroid Carcinoma Following Initial Therapy. Thyroid. 2006;16(12):1229–42. https://doi.org/10.1089/thy.2006.16.1229.
Knauf JA, Fagin JA. Role of MAPK pathway oncoproteins in thyroid cancer pathogenesis and as drug targets. Curr Opin Cell Biol. 2009;21(2):296–303. https://doi.org/10.1016/j.ceb.2009.01.013.
Pitoia F, Jerkovich F. Dynamic risk assessment in patients with differentiated thyroid cancer. Endocr Relat Cancer. 2019;26(10):R553–66. https://doi.org/10.1530/ERC-19-0213.
Filetti S, Durante C, Hartl D, et al. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30(12):1856–83. https://doi.org/10.1093/annonc/mdz400.
Vaisman F, Momesso D, Bulzico DA, et al. Spontaneous remission in thyroid cancer patients after biochemical incomplete response to initial therapy. Clin Endocrinol (Oxf). 2012;77(1):132–8. https://doi.org/10.1111/j.1365-2265.2012.04342.x.
Liu YQ, Li H, Liu JR, Lin YS. Unfavorable Responses to Radioiodine Therapy in N1b Papillary Thyroid Cancer: A Propensity Score Matching Study. Endocr Pract. 2019;25(12):1286–94. https://doi.org/10.4158/EP-2019-0155.
Deng Y, Zhu G, Ouyang W, et al. Size of the Largest Metastatic Focus to the Lymph Node is Associated with Incomplete Response of Pn1 Papillary Thyroid Carcinoma. Endocr Pract. 2019;25(9):887–98. https://doi.org/10.4158/EP-2018-0583.
Liu JB, Baugh KA, Ramonell KM, et al. Molecular Testing Predicts Incomplete Response to Initial Therapy in Differentiated Thyroid Carcinoma Without Lateral Neck or Distant Metastasis at Presentation: Retrospective Cohort Study. Thyroid. 2023;33(6):705–14. https://doi.org/10.1089/thy.2023.0060.
Pizzimenti C, Fiorentino V, Ieni A, et al. BRAF-AXL-PD-L1 Signaling Axis as a Possible Biological Marker for RAI Treatment in the Thyroid Cancer ATA Intermediate Risk Category. Int J Mol Sci. 2023;24(12):10024. https://doi.org/10.3390/ijms241210024.
Pérez-Fernández L, Sastre J, Zafón C, et al. Validation of dynamic risk stratification and impact of BRAF in risk assessment of thyroid cancer, a nation-wide multicenter study. Front Endocrinol. 2023;13:1071775. https://doi.org/10.3389/fendo.2022.1071775.
Giovanella L, Deandreis D, Vrachimis A, Campenni A, Petranovic OP. Molecular Imaging and Theragnostics of Thyroid Cancers. Cancers. 2022;14(5):1272. https://doi.org/10.3390/cancers14051272.
Iwano S, Kato K, Ito S, Tsuchiya K, Naganawa S. FDG-PET performed concurrently with initial I-131 ablation for differentiated thyroid cancer. Ann Nucl Med. 2012;26(3):207–13. https://doi.org/10.1007/s12149-011-0559-y.
Lee JW, Lee SM, Lee DH, Kim YJ. Clinical Utility of 18 F-FDG PET/CT Concurrent with 131 I Therapy in Intermediate–to–High-Risk Patients with Differentiated Thyroid Cancer: Dual-Center Experience with 286 Patients. J Nucl Med. 2013;54(8):1230–6. https://doi.org/10.2967/jnumed.112.117119.
Nascimento C, Borget I, Al Ghuzlan A, et al. Postoperative Fluorine-18-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography: An Important Imaging Modality in Patients with Aggressive Histology of Differentiated Thyroid Cancer. Thyroid. 2015;25(4):437–44. https://doi.org/10.1089/thy.2014.0320.
Klain M, Zampella E, Nappi C, et al. Advances in Functional Imaging of Differentiated Thyroid Cancer. Cancers. 2021;13(19):4748. https://doi.org/10.3390/cancers13194748.
Baloch ZW, Asa SL, Barletta JA, et al. Overview of the 2022 WHO Classification of Thyroid Neoplasms. Endocr Pathol. 2022;33(1):27–63. https://doi.org/10.1007/s12022-022-09707-3.
Yip L, Gooding WE, Nikitski A, et al. Risk assessment for distant metastasis in differentiated thyroid cancer using molecular profiling: A matched case-control study. Cancer. 2021;127(11):1779–87. https://doi.org/10.1002/cncr.33421.
Hamidi S, Hofmann MC, Iyer PC, et al. Review article: new treatments for advanced differentiated thyroid cancers and potential mechanisms of drug resistance. Front Endocrinol. 2023;14:1176731. https://doi.org/10.3389/fendo.2023.1176731.
Zhao H, Gong Y. Radioactive iodine in low- to intermediate-risk papillary thyroid cancer. Front Endocrinol. 2022;13:960682. https://doi.org/10.3389/fendo.2022.960682.
Sacks W, Fung CH, Chang JT, Waxman A, Braunstein GD. The Effectiveness of Radioactive Iodine for Treatment of Low-Risk Thyroid Cancer: A Systematic Analysis of the Peer-Reviewed Literature from 1966 to April 2008. Thyroid. 2010;20(11):1235–45. https://doi.org/10.1089/thy.2009.0455.
Funding
N/A.
Author information
Authors and Affiliations
Contributions
N/A.
Corresponding author
Ethics declarations
Ethical approval
N/A.
Conflict of interest
N/A.
Informed consent
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
do Prado Padovani, R., Duarte, F.B. & Nascimento, C. Current practice in intermediate risk differentiated thyroid cancer – a review. Rev Endocr Metab Disord 25, 95–108 (2024). https://doi.org/10.1007/s11154-023-09852-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-023-09852-y