Abstract
It is traditionally believed that genetic susceptibility and adult faulty lifestyle lead to type 2 diabetes, a chronic non-communicable disease. The “Developmental Origins of Health and Disease” (DOHaD) model proposes that the susceptibility to type 2 diabetes originates in the intrauterine life by environmental fetal programming, further exaggerated by rapid childhood growth, i.e. a biphasic nutritional insult. Both fetal under nutrition (sometimes manifested as low birth weight) and over nutrition (the baby of a diabetic mother) increase the risk of future diabetes. The common characteristic of these two types of babies is their high adiposity. An imbalance in nutrition seems to play an important role, and micronutrients seem particularly important. Normal to high maternal folate status coupled with low vitamin B12 status predicted higher adiposity and insulin resistance in Indian babies. Thus, 1-C (methyl) metabolism seems to play a key role in fetal programming. DOHaD represents a paradigm shift in the model for prevention of the chronic non-communicable diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Diabetes is fast becoming the leading cause of ill health in the world. Traditionally considered a disease of the affluent it is now spreading rapidly to the developing countries where more than 80% of the world population lives. There are an estimated 246 million diabetic patients in the world, of which 165 million are in the developing world [1]. India has the largest number of diabetic patients in any one country (∼40 million) and is therefore called the world’s diabetes capital. By the year 2025, there will be over 70 million diabetic patients in India and 59 million in China, thus one in three diabetic patients will be either Indian or Chinese [2].
Diabetic patients in developing countries, especially in India and Asia have a number of distinct characteristics compared to those in white European patients [3] (Fig. 1). Indian patients are diagnosed at a relatively younger age and this age is falling. In addition, they are shorter, thinner (lower body mass index, BMI), and have a lower lean mass. Despite their overall thinness Indian patients have a higher waist-hip ratio (central obesity) and have higher body fat percent (adiposity). Imaging studies have shown that Indian diabetic patients have higher central subcutaneous and intra-abdominal fat compared to European patients at a given BMI. [4] Thus, Indian diabetic patients are ‘thin but fat’. Biochemical and endocrine studies have shown that Indian patients have higher insulin resistance and higher prevalence of the metabolic syndrome variables (higher plasma triglycerides, lower HDL cholesterol, and higher blood pressure).
Comparison of characteristics of newly diagnosed Type 2 diabetes patients in India with those in the UK (not drawn to scale). Bars above the line indicate higher levels in Indian patients. Insulin resistance was calculated from the homeostatic model assessment (HOMA) model. Abbreviations and labels used: mid-arm mid-arm circumference; SS/TR sub-capsular-triceps skin-fold ratio; WHR waist-hip ratio; M males; F females [5]
The causes of the rapidly rising epidemic of diabetes in developing countries are not entirely clear. The conventional model suggests a genetic predisposition which is thought to have originated in the hunter-gatherer days (‘thrifty gene’) and modern day lifestyle factors (abundant food and physical inactivity) which bring on the disease [5] (Fig. 2). Until recently there were few genetic markers for type 2 diabetes, though in last two years more than 10 markers have been associated in the Genome Wide Association Studies in Europeans [6]. It is not yet clear if highly susceptible ethnic groups like south Asians have a stronger ‘genetic’ predisposition, or the susceptibility genes are unusually affected by environmental factors.
In 1991 Hales and Barker reported in the UK, that low birth weight and thinness at birth (low ponderal index) are risk factors for type 2 diabetes (‘thrifty phenotype’) [7, 8]. This focused attention on the importance of intrauterine life as a determinant of later health.
The association was confirmed in many other populations including different ethnic groups in the USA, in the Chinese, Afro Caribbean, in South Africa and in some but not all studies in India. In Pima Indians the relationship is U shaped, the contribution of large birth weight presumably reflecting the effects of maternal diabetes [9]. A study in south India showed that the risk of diabetes was not related to birth weight but to shorter length and higher ponderal index [10]. These studies have confirmed that size at birth is an important predictor of future diabetes. A number of terms like ‘fetal origins of adult disease’ (FOAD), ‘small baby syndrome’ were used in addition to the ‘thrifty phenotype’ to describe these new findings.
The link between intrauterine exposures and type 2 diabetes has been explained by the concept of fetal ‘programming’ [11, 12]. A developing system (embryo and fetus) is plastic i.e. capable of taking any of the many diverse routes and forms, but the intrauterine environment ‘programs’ it along certain pathways which help the fetus survive and develop. This affects structure and function, sometimes reflected in altered growth, body composition (adiposity), beta cell function, tissue response to hormones, and vascular reactivity. ‘Programming’ restricts the options for the fetus; if the environment in later life does not match the programming environment, the capabilities of the system are exceeded, and result in ‘disease’. The ‘programming’ stimuli are only beginning to be understood: nutritional factors, metabolic and hormonal milieu, infections and inflammation all seem to contribute [13]. Given the orchestrated nature of embryonic development there are ‘windows of opportunity’ for programming of different systems and functions and it is possible that there is some ‘specificity’ for certain exposures.
We began our research on ‘fetal origins’ in 1991 in collaboration with Prof David Barker and Caroline Fall. The first study was the Pune Children’s Study (PCS) in the King Edward Memorial Hospital where birth weights were available from the labour-room record. We followed up over 400 children at 4 years of age to study their anthropometry, glucose tolerance and circulating insulin concentrations. We demonstrated that after oral glucose load 30 min plasma glucose and insulin concentrations were inversely related to birth weight (Fig. 3) [14]. This provided the first proof for Barker’s hypothesis in a developing country.
Plasma glucose and insulin concentrations after oral glucose load in 4 years old Indian children. Significance of the trend is corrected for age, gender and current body weight. The results show that low birth weight is associated with higher glucose and insulin concentrations [14]
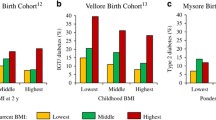
We followed up these children at 8 years of age to study their metabolic characteristics. In addition to confirming the association of low birth weight with increased insulin resistance, we made an interesting observation [15]. The levels of risk factors for diabetes and cardiovascular disease (glucose, insulin resistance, lipids, blood pressure, leptin concentrations etc.) were highest in children who were born the lightest but were heaviest at 8 years of age (Fig. 4). This study focused attention on rapid childhood growth as a powerful risk factor for type 2 diabetes in the developing countries. We also made one more interesting observation: children of short parents were more insulin resistant, and those who had grown taller at 8 years were the most insulin resistant. This finding provided a proof for intergenerational influences on metabolic risk, and showed that a discordance of size not only in one’s life time (birth weight and later weight) but across generations (parental height and childhood height) predicts higher risk. Further support for this observation was obtained in a study in Delhi where a large number of children have been followed from birth [16]. They were studied at 28 years of age. Those who were diabetic, were born lighter, had grown slower during infancy but had grown progressively faster from 3 years of age, and had an earlier adiposity rebound compared to those who were normal glucose tolerant (Fig. 5).
Mean levels of insulin resistance variable (HOMA), at 8 years of age by tertiles of birth weight and 8-year weight. Significance level for the trend in each row and column (adjusted for age and sex) is shown at the end [15] (ns not significant, *P < 0.05, **P < 0.01, ***P < 0.001)
Mean sex-specific unadjusted SD scores for body-mass index, according to age, for subjects in whom impaired glucose tolerance or diabetes developed. The mean SD scores (solid lines) are obtained by linear interpolation of yearly means, with one additional observation at 6 months. The dotted lines represent 95% confidence intervals. The dashed portions of lines indicate years in which there was no follow-up. The SD score for the cohort is set at zero (solid horizontal lines) [16]
2 Maternal nutrition and offspring risk
Nutrition is a major determinant of body size both in utero and later in life. The relationship between size at birth and later diabetes might be a reflection of nutrition of the fetus. [17] There are only a few studies which analysed offspring risk of diabetes in relation to maternal nutrition in pregnancy. The famous Dutch winter hunger study demonstrated that in utero exposure of the fetus to famine during mid and late gestation was associated with a higher plasma glucose concentration in middle age [18].
2.1 Pune maternal nutrition study
Convinced of the importance of intrauterine and childhood growth in influencing the risk of type 2 diabetes, we set up the Pune Maternal Nutrition Study in six villages near Pune in 1993. We aimed to investigate the influence of maternal size and her nutrition in pregnancy, on fetal growth and its future risk of diabetes.
We did a house-to-house survey to enroll all young (<35 years) women who were not sterilized (∼2,675 women). They were followed up every month to record their last menstrual date and every 3 months we measured their detailed anthropometry. Eight hundred and fourteen became pregnant during the study and 770 delivered a live, single, normal baby. The mothers were 21 years old, weighed 42 kg, were 1.52 m tall, with a BMI of 18.1 kg/m2. They ate ∼1,700 kcal/day and 45 g proteins/day during pregnancy. They were physically very active and worked at home and in the farms.
These mothers gave birth to babies 28% of whom were low birth weight (LBW, <2,500 g). [19] We compared these babies with those born in Southampton, UK (Fig. 6) and who were measured using comparable techniques. Indian babies were lighter (2.7 kg vs. 3.5 kg), shorter (47.3 cm vs. 50.2 cm) and thinner (ponderal index 24.1 vs. 27.3 kg/cm3) but the sub-scapular skin-fold measurements were comparable to those of the white babies. Thus, the Indian babies were short, thin and fat! In a subsequent study we demonstrated that the cord blood of Indian babies has higher glucose, insulin and leptin concentrations compared to those in the white British babies. [20]
Comparison of Indian and UK babies. UK measurements are used as a reference (0). The Indian babies were smaller than the British babies in all measurements of size. Cord plasma leptin concentration was similar and cord plasma glucose and insulin concentrations were higher in the Indian babies [5]
2.2 Maternal size and fetal growth
Maternal size was a strong determinant of baby’s size, and in general there was an association between ‘like’ measurements i.e. between length and fat. Short and fat mothers gave birth to fattest babies, highlighting that a nutritional transition in mothers’ lifetime reflected in adiposity in the baby [21]. Maternal macronutrient intake (calories and proteins) did not predict newborn size. However, the frequency of intake of green leafy vegetables, fruit and milk (foods rich in micronutrients) strongly predicted neonatal size. These results highlighted the importance of maternal micronutrient intake for fetal growth. We knew from other studies in Pune that vitamin B12 deficiency was quite prevalent in this population, due to vegetarian food habits [22]. We therefore measured vitamin B12 (B12), folate, total homocysteine (tHcy) and methyl malonic acid (MMA) in stored maternal blood samples.
We found low circulating concentrations of B12 in these mothers but folate concentrations were normal. A third of mothers had high tHcy concentrations but over 90% had higher MMA concentrations, suggesting that low B12 levels represented tissue deficiency. High maternal tHcy concentration (contributed by low B12 status rather than folate deficiency) predicted intrauterine growth retardation (IUGR) in the baby. [23] This highlights the importance of maternal micronutrient nutrition for fetal growth.
We assessed these women’s physical activity by a structured questionnaire. More than 80% of these women belonged to farming community and performed heavy physical work at home and in the farms. This included fetching water, cooking for the family, looking after cattle and heavy farm work. There was a strong and inverse association between maternal physical activity and neonatal size and heavy work increased the risk of low birth weight babies [24].
We measured circulating concentrations of glucose and lipids in the mothers during pregnancy. There were only a few women with gestational diabetes but maternal plasma glucose, cholesterol and triglyceride concentrations (within the normal range) were strong predictors of fetal size.
Our data highlights the importance of maternal pre-pregnancy size, her food intake and physical activity, and her metabolic status during pregnancy on fetal growth and body composition.
2.3 Follow up of the children
2.3.1 Size, body composition and metabolism
Children in the PMNS are measured every 6 months for their growth and every 6 years we evaluate their risk of diabetes and cardiovascular disease. The measurements include: detailed anthropometry, body composition (dual-energy X-ray absorptiometry, DXA), circulating glucose, insulin, lipids, insulin resistance [homeostatic model assessment of insulin resistance (HOMA-R)], and many novel risk factors.
At 6 years of age, these children were short and thin (BMI) by international standards but relatively adipose compared with the UK standards (skin-fold thicknesses) i.e. they continued to be short and thin and fat [25].
In the PMNS we are able to relate maternal characteristics during pregnancy with offspring development and body composition during childhood [26]. We found very interesting associations between maternal nutritional measurements during pregnancy and child’s adiposity and insulin resistance at 6 years of age. Frequency of maternal intake of green leafy vegetables and milk were associated with higher adiposity in the child, and higher maternal erythrocyte folate concentrations at 28 weeks predicted higher offspring adiposity. Higher maternal folate concentrations also predicted higher HOMA-R in the child. Low maternal B12 concentrations at 18 weeks gestation predicted higher HOMA-R in the children, and the children of mothers who had lowest B12 and highest folate concentrations were the most insulin resistant (Fig. 7).
Insulin resistance (HOMA-R) in the children at 6 years in relation to maternal vitamin B12 (18 weeks) and erythrocyte folate (28 weeks) [26]
Maternal B12 status was influenced by her dietary habits, especially intake of non-vegetarian foods and milk, both of which favorably affected plasma B12 concentrations. Forty percent of these women were lacto-vegetarian, and they were at a high risk of low B12 status. On the other hand folate status was uniformly good in these women; there was only one woman with low folate concentrations. In addition, all women received iron and folic acid at the first antenatal visit (18 weeks) as per the National Nutritional Anemia Prophylaxis Program. In clinical practice obstetricians in India give high doses of folic acid to prevent neural tube defects, though majority of women visit the antenatal clinic much later than the closure time for the neural tube (4 weeks from conception). Thus, in this population, both the natural food habits and iatrogenic practices seem to predispose these women to an imbalance between B12 and folate, which predicts higher adiposity and insulin resistance in the offspring. A common nutritional problem may thus contribute to the epidemic of adiposity and type 2 diabetes in India.
2.3.2 Bone mass
Many epidemiological studies have pointed out an association between intrauterine factors and bone mineral density in the child. Low birth weight is a risk factor for osteoporosis, and maternal body build, diet, vitamin D status, smoking, and physical activity have been shown to influence bone mass in the offspring [27–31]. Supplementation of pregnant mothers with vitamin D [32], calcium [33, 34], and other micronutrients [35] is associated with increased skeletal growth and bone mass in the offspring.
In the PMNS we found that children of mothers who had a higher frequency of intake of calcium-rich foods during pregnancy (milk, milk products, pulses, non-vegetarian foods, green leafy vegetables, and fruit) had higher total and spine bone mineral content and BMD (Fig. 8) [36]. In addition, higher maternal folate status at 28 week gestation also had similar effect. These effects were independent of the strong effects of parental size and DXA measurements.
Total body BMD (means and SE values) in the children according to the mother’s frequency of intake of milk, milk products, and calcium-rich foods at 28 wk gestation. (r = Pearson correlation coefficient and P value for continuous predictor and outcome variables) [36]
2.3.3 Neurocognitive function
Maternal single nutrient deficiency during pregnancy has been associated with abnormalities in the developing nervous system. Vitamin B12 is an important nutrient for development and functioning of the nervous system. [37, 38] In PMNS, we found an association between maternal B12 status during pregnancy and the child’s cognitive functioning (in press). Higher maternal plasma B12 concentration in pregnancy was an independent predictor of the child’s cognitive performance (attention and working) at 6 years of age.
In summary, PMNS results indicate that the intrauterine environment affects many aspects of the offspring’s health.
3 DOHaD and life-course model
Over last two decades the concept of ‘intrauterine origins’ has expanded to include post-natal influences on the risk of subsequent disease. It is now called ‘Developmental Origins of Health and Disease’. There is an international council with elected members and regular international conferences (http://www.mrc.soton.ac.uk/dohad/index.asp). The first conference was held in Mumbai in 2001 and the fifth in Perth in 2007. There is a resource unit in Southampton, UK.
The current concept of DOHaD is shown in Fig. 9. It is proposed that maternal characteristics before and during pregnancy influence fetal survival, growth, size and body composition, and function of various systems. Some of these effects are demonstrable at birth. Post-natal growth also makes important contribution to risk of later disease: rapid growth contributes to increased risk. These are manifest in risk factors related to structure (body composition) as well as function (beta cell function and insulin resistance) which contribute to disease and preterm mortality.
The concept of DOHaD has revolutionized the ideas in etiology of chronic non-communicable disease. It has taken us nearer to designing ‘real’ prevention strategies for type 2 diabetes and coronary heart disease. These new ideas are not yet part of the thinking of general medical profession and the policy makers. The WHO has endorsed these ideas by inviting various expert meetings and through its publications. An expert committee conceptualized the idea in the now famous ‘life-course’ evolution of the risk of chronic non-communicable disease [39] (Fig. 10). It proposes that the risk of these conditions originates in very early life, and sometimes intergenerationally. The risk progressively increases during life and the conditions are usually diagnosed in adult age at an arbitrarily defined cut point.
The World Health Organization’s life-course model of non-communicable disease. The model suggests that non-communicable diseases have their origins in early life. The risk progressively accumulates throughout the life course and the disease becomes manifest in later life [39]
4 Mechanisms of fetal programming: epigenetics and genetics
Fetal programming may be achieved through a number of mechanisms [40]. At the level of an organism it may be achieved by alterations in body composition (for example adiposity) or adjustments in set points of regulatory systems (for example neuro-endocrine axes like the insulin-IGF and the hypothalamic-pituitary-adrenal axis) [41]. At cellular level, programming affects aspects of cell cycle dynamics and apoptosis [42] which may reflect in numbers of cells. At sub-cellular level, telomerase shortening and altered function of different enzyme systems have been demonstrated [43]. A comparatively recent development is the recognition that ‘epigenetic’ changes are an important component of programming. These refer to changes in DNA other than a change in sequence of bases, and are mitotically heritable [44]. Silencing of genes through methylation of cytosine residues in the CpG islands of DNA is one major mechanism of epigenetic regulation. Acetylation of histones is another epigenetic mechanism that may alter tertiary structure of chromatin and expose the underlying genes to regulatory mechanisms. These mechanisms affect the expression of genes without affecting their base sequence and thus same genotype can give rise to different phenotypes. This is an important mechanism during development and differentiation, allowing different tissues and organs to develop from the same original genotype.
The importance of methylation in nutritional programming is well demonstrated in animal models. Feeding agouti mothers with a methylating cocktail (B12 + folate + betaine + choline) produced offspring of various coat colors and degree of obesity, despite inheriting the same mutation [45, 46]. This was linked to selective methylation in the promoter region of the gene. A sheep model of periconceptional methionine deficiency (induced by dietary deficiency of the amino acid and vitamins B12 and folate) demonstrated that the male offspring were adipose and insulin resistant, and these changes were associated with methylation of a number of genes [47]. These experiments have highlighted an important role for 1-C metabolism in nutritional programming and suggest that this could be achieved by dietary manipulation of methyl donors like vitamins B12 and folate. The PMNS results linking maternal vitamin B12 and folate nutrition with offspring adiposity and insulin resistance assume special significance [26].
A role for genetics in DOHaD has been suggested in the fetal insulin hypothesis. [48, 49] This hypothesis proposes that genes which increase susceptibility to diabetes also reduce birth weight by influencing insulin-mediated growth, i.e. low birth weight and type 2 diabetes are both phenotypes of the same genotype. The first evidence was from the rare mutation in glucokinase gene (GCK) that reduces insulin secretion in β-cells. Baby with GCK mutation inherited from father was born 500 g lighter if mother was normal. If the baby inherited GCK mutation from mother its weight was normal because maternal diabetes negated the effect of mutation, and 500 g heavier if it did not inherit the mutation. Other rare genetic syndromes those affect insulin secretion (transient neonatal diabetes) or function (Leprechaunism) are also associated with low birth weight. Fetal insulin hypothesis also predicted a low birth weight in children of diabetic fathers, this was first demonstrated in Pima Indians [50]. However, a direct relationship between paternal insulin resistance and offspring birth weight has also been demonstrated [51]. A number of common polymorphisms associated with diabetes have been shown to predict low birth weight, supporting genetic contribution to low birth weight and diabetes connection.
Maternal under-nutrition and lower socio-economic status remain the most substantive causes of low birth weight worldwide, and animal models of under-nutrition as well as micronutrient supplementation have confirmed the role of nutritional fetal programming. It would appear that gene-environment interactions are at the heart of intrauterine programming.
5 Concluding remarks
The conventional model of type 2 diabetes (Fig. 1) and other chronic non-communicable diseases (NCD) include a genetic susceptibility and precipitation of the disease state in adult age by affluent lifestyle. The rapidly changing epidemiology of the disease, including its rapid spread to the developing populations and its predilection for the young and the poor suggest that this model might be inadequate. The developmental origins (DOHaD) model (Fig. 9) has provided new exciting insights into the etiology of chronic NCD. It proposes that intrauterine environment ‘programs’ the developing fetus to be fit to face a similar post-natal environment (‘predictive adaptive response’). If the post-natal environment is discordant with intrauterine environment, it leads to disease. Thus babies born small but growing rapidly in childhood are at a very high risk of developing diabetes. Maternal nutrition has major effect on fetal growth and programming, and in populations undergoing rapid transition, early life under-nutrition and later life over-nutrition make major contribution to the ‘life course’ evolution of type 2 diabetes and other NCD. The obvious implication for diabetes prevention is that the efforts will have to start very early in life and continued throughout the life cycle. Improving the health of young girls will be of paramount importance. Controlling rapid childhood growth will be a challenge.
Recent research has highlighted the possible role epigenetics might play in fetal programming. Methyl donors like B12 and folate seem to play a major role in these phenomena. Maternal low B12 status along with normal to high folate status predicted adiposity and insulin resistance in Indian children. These results need to be confirmed and then acted upon. Maternal micronutrients also affect other systems in the fetus like brain and bones.
Further research in these areas is likely to provide exciting new leads in the prevention of many chronic NCD.
References
International Diabetes Federation. Diabetes atlas. 3rd ed. Belgium: World Diabetes Foundation; 2007.
King H, Aubert RE, Herman WH. Global burden of diabetes, 1995–2025: prevalence, numerical estimates and projections. Diabetes Care 1998;21:1414–31. doi:10.2337/diacare.21.9.1414.
Yajnik CS. The insulin resistance epidemic in India: fetal origins, later lifestyle, or both? Nutr Rev 2001;59:1–9.
Deurenberg P, Deurenberg Y, Guricci S. Asians are different from Caucasians and from each other in their body mass index/body fat percent relationship. Obes Rev 2002;3:141–6. doi:10.1046/j.1467-789X.2002.00065.x.
Yajnik CS. Early life origins of insulin resistance and type 2 diabetes in India and other Asian countries. J Nutr 2004;134:205–10.
Zeggini E, Weedon M, Lindgren C, Frayling T, Elliott K. Replication of genome-wide association signals in UK samples reveals risk loci for T2D. Science 2007;316:1336–41. doi:10.1126/science.1142364.
Barker DJP. Mothers, babies and health in later life. 2nd ed. Edinburgh: Churchill Livingstone; 1998.
Hales CN, Barker DJP. Type 2 (non-insulin dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia 1992;35:595–601. doi:10.1007/BF00400248.
McCance DR, Pettitt DJ, Hanson RL, Jacobsson LTH, Knowler WC, Bennett PH. Birth weight and non-insulin dependent diabetes: thrifty genotype, thrifty phenotype, or surviving small baby genotype? BMJ 1994;308:942–5.
Fall CHD, Stein CE, Kumaran K. Size at birth, maternal weight, and type 2 diabetes in south India. Diabet Med 1998;15:220–7. doi:10.1002/(SICI)1096-9136(199803)15:3<220::AID-DIA544>3.0.CO;2-O.
Lucas A. Programming by early nutrition in man. In: Bock GR, Whelan J, editors. The childhood environment and adult disease. CIBA Foundation Symposium 156. Chichester: Wiley; 1991. pp 38–55
Eriksson JG, Forsen TJ, Osmond C, Barker DJ. Pathways of infant and childhood growth that lead to type 2 diabetes. Diabetes Care 2003;26:3006–10. doi:10.2337/diacare.26.11.3006.
Hoet JJ, Hanson MA. Intrauterine nutrition: its importance during critical periods for cardiovascular and endocrine development. J Physiol 1999;514:617–27. doi:10.1111/j.1469-7793.1999.617ad.x.
Yajnik CS, Fall CH, Vaidya U, Pandit AN, Bavdekar A, Bhat DS. Fetal growth and glucose and insulin metabolism in four-year-old Indian children. Diabet Med 1995;12:330–6.
Bavdekar A, Yajnik CS, Fall CH, Bapat S, Pandit AN, Deshpande V, et al. Insulin resistance syndrome in 8-year-old Indian children: small at birth, big at 8 years, or both? Diabetes 1999;48:2422–9. doi:10.2337/diabetes.48.12.2422.
Bhargava SK, Sachdev HS, Fall CH, Osmond C, Lakshmy R, Barker DJP, et al. Relation of serial changes in childhood body-mass index to impaired glucose tolerance in young adulthood. N Engl J Med 2004;350:865–75. doi:10.1056/NEJMoa035698.
Shiell AW, Campbell-Brown M, Hall MH, Barker DJP. Diet in late pregnancy and glucose insulin metabolism of the offspring 40 years later. Br J Obstet Gynaecol 2000;107:890–5.
Ravelli AC, van der Meulen JH, Michels RP, Osmond C, Barker DJ, Hales CN, et al. Glucose tolerance in adults after prenatal exposure to famine. Lancet 1998;351:173–7. doi:10.1016/S0140-6736(97)07244-9.
Yajnik CS, Fall CHD, Coyaji KJ, Hirve SS, Rao S, Barker DJP, et al. Neonatal anthropometry: the thin-fat Indian baby: The Pune Maternal Nutrition Study. Int J Obes 2003;26:173–80. doi:10.1038/sj.ijo.802219.
Yajnik CS, Lubree HG, Rege SS, Naik SS, Deshpande JA, Deshpande SS, et al. Adiposity and hyperinsulinemia in Indians are present at birth. J Clin Endocrinol Metab 2002;87:5575–80. doi:10.1210/jc.2002-020434.
Rao S, Yajnik CS, Kanade A, Fall CHD, Margetts BM, Jackson AA, et al. Intake of micronutrient-rich foods in rural Indian mothers is associated with the size of their babies at birth: Pune Maternal Nutrition Study. J Nutr 2001;131:1217–24.
Yajnik CS, Deshpande SS, Lubree HG, Naik SS, Bhat DS, Uradey BS, et al. Vitamin B12 Deficiency and Hyperhomocysteinemia in Rural and Urban Indians. J Assoc Physicians India 2006;54:1–8.
Yajnik CS, Deshpande SS, Panchanadikar AV, Naik SS, Deshpande JA, Coyaji KJ, et al. Maternal total homocysteine concentration and neonatal size in India. Asia Pac J Clin Nutr 2005;14:179–81.
Rao S, Kanade A, Margetts BM, Yajnik CS, Lubree HG, Rege S, et al. Maternal activity in relation to birth Size in rural India: The Pune Maternal Nutrition Study. Eur J Clin Nutr 2003;57:531–42. doi:10.1038/sj.ejcn.1601582.
Joglekar C, Fall CHD, Deshpande VU, Joshi N, Bhalerao A, Solat V, et al. Newborn size, and childhood growth, and cardiovascular disease risk factors at the age of 6 years: The Pune Maternal Nutrition Study. Int J Obes 2007;31:1534–44. doi:10.1038/sj.ijo.0803679.
Yajnik CS, Deshpande SS, Jackson AA, Refsum H, Rao S, Fisher D, et al. Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: The Pune Maternal Nutrition Study. Diabetologia 2008;51:29–38. doi:10.1007/s00125-007-0793-y.
Jones G, Riley MD, Dwyer T. Maternal diet during pregnancy is associated with bone mineral density in children: a longitudinal study. Eur J Clin Nutr 2000;54:749–56. doi:10.1038/sj.ejcn.1601082.
Godfrey K, Walker-Bone K, Robinson S, Taylor P, Shore S, Wheeler T, et al. Neonatal bone mass: influence of parental birthweight and maternal smoking, body composition and activity during pregnancy. J Bone Miner Res 2001;16:1694–703. doi:10.1359/jbmr.2001.16.9.1694.
Tobias JH, Steer CD, Emmett PM, Tonkin RJ, Cooper C, Ness AR. Bone mass in childhood is related to maternal diet in pregnancy. Osteoporos Int 2005;16:1731–41. doi:10.1007/s00198-005-1912-6.
Pawley N, Bishop NJ. Prenatal and infant predictors of bone health: the influence of vitamin D. Am J Clin Nutr 2004;80:1748S–51S.
Javaid MK, Crozier SR, Harvey NC, Dennison EM, Boucher BJ, Arden NK, et al. Maternal vitamin D status during pregnancy and childhood bone mass at age nine years: a longitudinal study. Lancet 2006;367:36–43. doi:10.1016/S0140-6736(06)67922-1.
Brooke OG, Brown IRF, Bone CDM, Carter ND, Cleeve HJW, Maxwell JD, et al. Vitamin D supplements in pregnant Asian women: effects on calcium status and fetal growth. BMJ 1980;280:751–4.
Raman L, Rajalakshmi K, Krishnamachari KAVR, Sastry JG. Effect of calcium supplementation to undernourished mothers during pregnancy on the bone density of the neonates. Am J Clin Nutr 1978;31:466–9.
Koo WWK, Walters JC, Esterlitz J, Levine RJ, Bush AJ, Sibai B. Maternal calcium supplementation and fetal bone mineralization. Obstet Gynecol 1999;94:577–82. doi:10.1016/S0029-7844(99)00371-3.
Himes JH, Caulfield LE, Reynaldo M, Delgado H. Maternal supplementation and bone growth in infancy. Paediatr Perinat Epidemiol 1990;4:436–47. doi:10.1111/j.1365-3016.1990.tb00671.x.
Ganpule A, Yajnik CS, Fall CH, Rao S, Fisher DJ, Kanade A. Bone mass in Indian children; relationships to maternal nutritional status and diet during pregnancy; the Pune Maternal Nutrition Study. J Clin Endocrinol Metab 2006;91:2994–3001. doi:10.1210/jc.2005-2431.
Stabler SP. Vitamins, homocysteine and cognition. Am J Clin Nutr 2003;78:359–60.
Healton EB, Savage DG, Brust JC, Garett TJ, Lindenbaum J. Neurological aspects of cobalamin deficiency. Medicine (Baltimore) 1991;70:229–45. doi:10.1097/00005792-199107000-00001.
Aboderin I, Kalache A, Ben-Shlomo Y, Lynch JW, Yajnik CS, Kuh D, et al. (2001) Life course perspectives on coronary heart disease, stroke and diabetes: key issues and implications for policy and research. Geneva, World Health Organization (Summary report of a meeting of experts).
Fernandez-Twinn DS, Ozanne SE. Mechanisms by which poor early growth programs type-2 diabetes, obesity and the metabolic syndrome. Physiol Behav 2006;88:234–43. doi:10.1016/j.physbeh.2006.05.039.
Phillips DI. Programming of adrenocortical function and the fetal origins of adult disease. J Endocrinol Invest 2001;24:742–6.
Hill DJ, Duvillie B. Pancreatic development and adult diabetes. Pediatr Res 2000;48:269–74. doi:10.1203/00006450-200009000-00002.
Demerath EW, Cameron N, Gillman MW. Telomeres and telomerase in the fetal origins of cardiovascular disease: a review. Hum Biol 2004;76:127–46. doi:10.1353/hub.2004.0018.
Lederberg J. The meaning of epigenetics. Scientist 2001;15:6.
Waterland RA, Jirtle RL. Transposable elements: targets for early nutritional effects on epigenetic gene regulation. Mol Cell Biol 2003;23:5293–300. doi:10.1128/MCB.23.15.5293-5300.2003.
Lillycrop KA, Phillips ES, Jackson AA, Hanson MA, Burdge GC. Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J Nutr 2005;135:1382–6.
Sinclair KD, Allegrucci C, Singh R, Gardner DS, Sebastian S, Bispham J, et al. DNA methylation, insulin resistance, and blood pressure in offspring determined by maternal periconceptional B vitamin and methionine status. Proc Natl Acad Sci U S A 2007;104:19351–6. doi:10.1073/pnas.0707258104.
Hattersley AT, Tooke JE. The fetal insulin hypothesis: an alternative explanation of the association of low birth weight with diabetes and vascular disease. Lancet 1999;353:1789–92. doi:10.1016/S0140-6736(98)07546-1.
Frayling TM, Hattersley AT. The role of genetic susceptibility in the association of low birth weight with type 2 diabetes. Br Med Bull 2001;60:89–101. doi:10.1093/bmb/60.1.89.
Lindsay RS, Dabelea D, Roumain J, Hanson RL, Bennett PH, Knowler WC. Type 2 diabetes and low birth weight: the role of paternal inheritance in the association of low birth weight and diabetes. Diabetes 2000;49:445–9. doi:10.2337/diabetes.49.3.445.
Yajnik CS, Coyaji K, Joglekar CV, Kellingray S, Fall CHD. Paternal insulin resistance and fetal growth: problem for the ‘fetal insulin’ and the ‘fetal origins’ hypotheses. Diabetologia 2001;44:1197–8. doi:10.1007/s001250100622.
Acknowledgments
The author receives funding from the Wellcome Trust (London, UK), the Nestlé Foundation (Lausanne, Switzerland), the International Atomic Energy Agency (Vienna, Austria) and the Department of Biotechnology (DBT), Government of India (New Delhi, India). Thanks are due to colleagues, collaborators and participants in the studies mentioned in this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yajnik, C.S., Deshmukh, U.S. Maternal nutrition, intrauterine programming and consequential risks in the offspring. Rev Endocr Metab Disord 9, 203–211 (2008). https://doi.org/10.1007/s11154-008-9087-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-008-9087-z