Abstract
Lumenal extrinsic proteins PsbO, PsbP, and PsbQ of photosystem II (PSII) protect the catalytic cluster Mn4CaO5 of oxygen-evolving complex (OEC) from the bulk solution and from soluble compounds in the surrounding medium. Extraction of PsbP and PsbQ proteins by NaCl-washing together with chelator EGTA is followed also by the depletion of Ca2+ cation from OEC. In this study, the effects of PsbP and PsbQ proteins, as well as Ca2+ extraction from OEC on the kinetics of the reduced primary electron acceptor (QA −) oxidation, have been studied by fluorescence decay kinetics measurements in PSII membrane fragments. We found that in addition to the impairment of OEC, removal of PsbP and PsbQ significantly slows the rate of electron transfer from QA − to the secondary quinone acceptor QB. Electron transfer from QA − to QB in photosystem II membranes with an occupied QB site was slowed down by a factor of 8. However, addition of EGTA or CaCl2 to NaCl-washed PSII did not change the kinetics of fluorescence decay. Moreover, the kinetics of QA − oxidation by QB in Ca-depleted PSII membranes obtained by treatment with citrate buffer at pH 3.0 (such treatment keeps all extrinsic proteins in PSII but extracts Ca2+ from OEC) was not changed. The results obtained indicate that the effect of NaCl-washing on the QA − to QB electron transport is due to PsbP and PsbQ extrinsic proteins extraction, but not due to Ca2+ depletion.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Photosynthetic organisms have evolved enzymes that efficiently convert solar energy into useful and storable chemical energy. A multiprotein complex of photosystem II (PSII) that resides in thylakoid membranes of cyanobacteria and chloroplasts by using the energy from photons operates as a water:plastoquinone oxidoreductase. Photooxidation of the PSII reaction center accompanied by the formation of charge separation between the primary electron donor P680 and the primary quinone acceptor QA (P680+QA −) leads to two chemical events: oxidation of water to molecular oxygen (after four turnover of PSII) and reduction of plastoquinone to plastohydroquinone (after two turnovers of PSII) (see Wydrzynski and Satoh 2005; Renger 2007; Najafpour et al. 2016; Vinyard and Brudvig 2017 for details). The QB functions as a two-electron gate, after a sequential transfer of two electrons and the two protonation events plastoquinol is formed at the QB site on the acceptor side of PSII (Bouges-Bocquet 1973; Velthuys and Amesz 1974; Shinkarev and Wraight 1993) [reviewed in (Crofts and Wraight 1983; Müh et al. 2012)]. A Mn4CaO5 cluster together with its coordinating amino acids and four water molecules form a catalytic site where oxidation of water molecules occurs (Kok et al. 1970; Shinkarev 2004; Renger and Kuhn 2007; Dau and Haumann 2007; Barber 2008). In doing so, the oxygen-evolving complex (OEC), which is located on the donor side of the enzyme, is the most fragile site within PSII and is easily susceptible to oxidative damage.
Catalytic center Mn4CaO5 of oxygen-evolving complex in higher plants is covered by three luminal extrinsic proteins with molecular masses of 33, 24, and 17 kDa that are encoded by the psbO, psbP, and psbQ genes, respectively (Bricker and Ghanotakis 1996). Two of these, the PsbP and PsbQ polypeptides (ExP), can be removed by washing with 2 M NaCl salt [see Murata and Miyao (1985) for a review]. Removal of these polypeptides results in the inhibition of O2 evolution up to 15–20% of initial level (Kuwabara and Murata 1982). Recovery of this lost oxygen evolution capability can be accomplished up to 80% via reconstitution with large Ca2+ concentration (tens mM; Ghanotakis et al. 1984a). Such high, non-physiological concentrations of calcium can support reasonably the high rates of oxygen evolution in the absence of the PsbP and PsbQ components (Miyao and Murata 1984). Rebinding of the PsbQ and PsbP polypeptides restores oxygen-evolving activity only when the sample has not been pretreated with EGTA (Ghanotakis et al. 1984b). Removal of loosely bound Ca2+ from the salt-extracted PS II complex and from the polypeptide solution blocks reconstitution of oxygen evolution activity even though the two polypeptides do rebind; restoration of Ca2+ to EGTA-treated systems, after rebinding of the PsbQ and PsbP polypeptides, results in a strong reconstitution of oxygen evolution activity (Ghanotakis et al. 1984b). All these results indicate that O2 production reaction depends only on Ca2+ cation, but not on extrinsic proteins. The authors suggested that the effect of PsbQ and PsbP subunits is to promote high affinity binding of Ca2+ cation to the reconstituted membrane. Thus, NaCl-washing provides not only extraction of PsbQ and PsbP polypeptides, but also extraction of Ca2+ cation from Mn4CaO5 cluster due to the decrease of its binding affinity. Taking all these results into account (Ca2+ contamination can effect on restoration of O2 activity in the presence of PsbQ and PsbP), the treatment of NaCl-washed membranes with EGTA is further used to get Ca-depleted PSII membranes without or with extrinsic proteins after reconstruction (Boussac et al. 1989). PSII membranes prepared by washing with NaCl were named as NaCl-washed PSII or PsbQ, PsbP-depleted PSII membranes, whereas washing of PSII membranes with NaCl together with EGTA provides Ca-depleted PSII membranes.
NaCl-washing/Ca-depletion of PSII membranes has a number of effects besides inhibition of O2 evolution. One of the interesting effect is the influence of such treatment on the redox potential of QA (Krieger and Weiss 1992; Krieger et al. 1995) or the kinetics of QA − oxidation by QB (Andréasson et al. 1995; Roose et al. 2010). A number of published results have shown that the extraction of Ca (using NaCl-washing together with EGTA) or Mn from OEC shifts the midpoint redox potential (Em) of QA from about −140 to 120 mV to +60 to 40 mV (Krieger and Weiss 1992; Krieger et al. 1995; Ido et al. 2011; Allakhverdiev et al. 2011). At the same time, Andréasson et al. (1995) and Roose et al. (2010) have shown that the NaCl-washing/Ca-depletion of PSII membranes significantly slows the QA − oxidation kinetic in PSII membranes. According to Andréasson et al. (1995), slowing down of QA − oxidation is the result of Ca2+ extraction from OEC, whereas extraction of extrinsic proteins is ineffective. However, Bricker with coworkers published recently the work in which they have shown the effect of NaCl treatment on the kinetic of QA − oxidation after single flash using fluorescence decay measurements method (Roose et al. 2010). They observed significant slowing of QA − oxidation rate induced by NaCl treatment; however, effects of Ca2+ and Ca-related compounds like EGTA were not studied. Roose et al. (2010) suggested that retardation of electron transport on the acceptor side of PSII is induced by PsbP and PsbQ extraction.
In the present study, we investigated the effect of NaCl-washing on QA − oxidation kinetics with or without Ca-chelator, as well as the effects of Ca extraction from OEC of samples which contain all extrinsic proteins. The obtained results show that effect of NaCl treatment on the acceptor side is related to only the extraction of extrinsic proteins, but not Ca2+.
Materials and methods
PSII-enriched membrane fragments with an active OEC were prepared from market spinach according to Berthold et al. (1981), but with minor modifications (Ghanotakis and Babcock 1983). Oxygen evolution was measured polarographically under continuous saturating white light illumination at 25 °C with a Clark-type oxygen electrode. Source of light was fitted with a water filter. The O2-evolving activity of the PSII membranes ranged from 400 to 500 µmol O2 mg Chl−1 h−1, when 0.2 mM 2,6-dichloro-p-benzoquinone was used as the artificial electron acceptor. PSII membranes were suspended in the reaction medium (buffer A) containing 15 mM NaCl, 400 mM sucrose, and 50 mM MES/NaOH buffer (pH 6.5) at a chlorophyll concentration 10 µg ml−1. Chlorophyll (Chl) concentrations were determined in 80% acetone, according to the method of Porra et al. (1989).
PsbQ- and PsbP-depleted PSII membranes were prepared from intact PSII membranes using a buffer solution containing 2 M NaCl, 0.4 M sucrose, and 25 mM MES (pH 6.5) (Ono and Inoue 1990) in the absence of chelator. The PSII preparations were incubated in this buffer at 0.5 mg Chl ml−1 for 15 min under room light (4–5 µE m−2 s−1) and at 23 °C. The resulting material was washed twice and resuspended in a buffer A. Samples are designated as PSII(−ExP) membranes.
Ca-depleted PSII membranes lacking PsbQ and PsbP extrinsic proteins were prepared by washing intact PSII membranes with NaCl to obtain PSII(−ExP) membranes (Ono and Inoue 1990). PSII(−ExP) membranes were then resuspended in a buffer A and incubated for 5 min prior to use in the dark at 23 °C with 5 mM EGTA to remove Ca2+ cations. Samples are designated as PSII(−Ca,−ExP) membranes.
Ca-depleted PSII membranes containing PsbQ and PsbP extrinsic proteins were prepared according to Ono and Inoue (1988). PSII membrane preparations in a buffer A were centrifuged, and the pellets resuspended in citrate buffer (400 mM sucrose, 20 mM NaCl, 10 mM sodium citrate, pH 3.0) at 2 mg Chl ml−1. Further, the membranes were incubated for 5 min on ice in the dark. The samples were finally diluted (1:3 v/v) with buffer A (pH shifted to 6.5) and incubated for 20 min on ice in the dark. Samples are designated as PSII(−Ca,+ExP) membranes.
Mn-depleted PSII membranes [PSII(−Mn)]
Manganese depletion was accomplished by incubating thawed intact PSII membranes (0.5 mg of Chl ml−1) in a buffer composed of 1 M Tris–HCl buffer, pH 9.4, 0.4 M sucrose for 30 min at 5 °C under room light (Preston and Seibert 1991). Further, the membranes were centrifuged (16,000×g, 10 min), washed twice with buffer A for removal of any non-specific Mn(II), and finally the pellet was resuspended in a buffer A. These membranes did not contain any extrinsic proteins (including the PsbO polypeptide), Ca2+ cations, or the Mn catalytic cluster.
Fluorescence decay measurements
Fluorescence decays after single saturating flash were measured using an FL3000 fluorometer (Photon Systems Instruments, Czech Republic). The measuring and saturating flashes were produced by computer-controlled photodiode arrays. The duration of the flash was 10 μs. Samples were assayed in buffer A (50 mM MES, 400 mM sucrose, 15 mM NaCl, pH 6.5). All samples were dark incubated for 5 min prior to measurement. The fluorescence intensity recorded 50 μs after the saturating actinic flash was collected as the FM value, while the fluorescence intensity collected immediately before the flash was collected as the F0 value. Decays of 3–5 separate samples were averaged. Data were fit to a three-component exponential decay equation after normalization, using the Origin program package (Origin Lab Corporation, USA). Fluorescence experiments have been carried out at 23 °C.
Profiles of protein composition of spinach PSII membrane fragments resolved by SDS-PAGE according to Kashino et al. (2001). Sodium dodecyl sulfate–urea–polyacrylamide gel was prepared in the presence of 2 M urea, 2% sodium dodecyl sulfate, and 5% mercaptoethanol. Percentage of the resolving gel was 13%. Aliquots of PSII membranes (7 μg Chl) were loaded on the gel. The gels were stained with Coomassie blue R-250.
Results and discussion
Effect of extrinsic proteins PsbP and PsbQ extraction on the QA − oxidation kinetics
To separate the effects caused by ExP-depletion and Ca2+ extraction from the OEC, influences of extrinsic proteins PsbP and PsbQ extraction with 2 M NaCl on the QA − oxidation kinetics have been studied. It is known that depletion of calcium and/or ExP influences transfer of an electron both on the oxidizing and reducing sides of the PSII reaction centers (Andréasson et al. 1995; Roose et al. 2010). Donor-side effects are due to the damage of OEC. Therefore, PsbP, PsbQ-depleted PSII membranes show usual decrease of the O2 evolution rate. Without exogenous Ca2+, PSII(−ExP) membranes perform the light-driven oxidation of water to molecular oxygen only at a low rate (the activity is about 15%; Table 1), corresponding to the results described in Andréasson et al. (1995), Boussac and Rutherford (1988), Roose et al. (2010), Semin et al. (2008). Exogenous Ca2+ can restore O2-evolving activity up to 70–80% (a restoration factor of about 5×) of the original rate of O2-evolving function (Table 1) (see also Andréasson et al. 1995; Ghanotakis et al. 1984a; Ono and Inoue 1988; Semin et al. 2008).
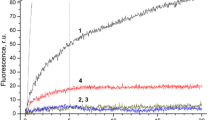
To study the effect of ExP-depletion on the acceptor side of PSII membrane fragments, we studied the kinetics of QA − oxidation rate using fluorescence decay measurement method. Results of these experiments are presented in Fig. 1 [the fluorescence decay observed in untreated PS II membranes both in the absence (curve 1) and in the presence (curve 2) of 40 μM DCMU] and Fig. 2 (the fluorescence decay observed in PsbP, PsbQ-depleted PSII membranes). Quantitative evaluation of these decay curves was performed by fitting to a three-exponential component decay equation (Reifarth et al. 1997; Roose et al. 2010). Results of fitting are presented in Table 2. There are three fitting exponential components for decay of fluorescence after single saturating flash in PSII samples in the absence of DCMU. The fastest component represents the electron transfer from QA − to QB (Bowes and Crofts 1980), while the intermediate decay component reflects QA − to QB electron transfer in centers with an unoccupied QB site (Renger et al. 1993). The slow decay component corresponds to QA − charge recombination with oxidizing-side components (Crofts and Wraight 1983). Our results demonstrate the good coincidence of the QA − oxidation kinetics in the intact PSII membranes without DCMU. For example, the fast decay phase determined in the present work is equal 0.9 ms (ca. 70%), whereas according to Roose et al. (2010), it is equal to 1.2 ms (64%). Note that, intermediate and slow decay phases are also similar (Table 2) (see also Roose et al. 2010).
Effect of PsbP and PsbQ extrinsic proteins removal on QA − reoxidation kinetics after a single saturating flash. (1) Intact PSII, (2) NaCl-washed PSII membranes without DCMU, (3) NaCl-washed PSII membranes with DCMU (40 µM). Concentration of PSII membranes was 10 µg Chl ml−1. Data were collected after dark incubation for 5 min. Note that, curve 1 (shown on in Fig. 1, curve 1) is presented on Fig. 2 for the convenience of comparing kinetic of QA − oxidation in the intact PSII membranes fragments with kinetics of such process in NaCl-treated PSII sample (curve 1, Fig. 2)
Effect of 30 mM Ca2+ (A) and 5 mM EGTA (B) on QA − oxidation kinetics in PsbP and PsbQ-depleted PSII membranes (10 µg Chl ml−1) after a single saturating flash. Data were collected after dark incubation for 5 min. A Curve 1 (rhombus)—NaCl-washed PSII membranes without addition; curve 2 (triangle)—with 30 mM Ca2+; B curve 1 (rhombus)—NaCl-washed PSII membranes without addition; curve 2 (triangle)—with 5 mM EGTA. Note that, curve 1 is shown in Fig. 2 and it is presented in Fig. 3 for convenience of comparison with kinetics of QA − oxidation affected by Ca2+ or EGTA treatment
Polyacrylamide gel electrophoretogram of PSII membranes fragments. Lane 1, intact PSII membranes; lane 2, NaCl-washed PSII membranes without PsbP and PsbQ extrinsic proteins; lane 3, Mn-depleted PSII membranes without all extrinsic proteins; lane 4, Ca-depleted PSII prepared using low pH (3.0) citrate buffer containing all extrinsic proteins; lane 5, ExP-depleted PSII prepared using low pH (3.7) citrate buffer; lane M, protein standards. The positions of the PSII extrinsic proteins (PsbO, PsbP, and PsbQ) are indicated by arrows on the left. LHC, light-harvesting, chlorophyll protein complex. One of the components of LHC locates close to PsbP protein (band above PsbP band). As example, close position of LHC and PsbP extrinsic protein was fixed in papers of Miller et al. (1987), Bricker (1992) and Kuntzleman and Haddy (2009)
In the presence of DCMU, fluorescence decay represents charge recombination between QA − and oxidizing-side components of PSII (Fig. 1, curve 2). This inhibitor significantly slows the oxidation of QA − that is determined by inhibition of forward electron transfer reaction from QA − to QB. Under these conditions, the fast fitting component of decay represents the fraction of PSII centers that lack a functional Mn4CaO5 cluster, in which QA − recombines with oxidized the redox-active tyrosine YZ (Weiss and Renger 1984). The origin of the middle component is unclear (see Reifarth et al. 1997 for a discussion). The slow phase is associated with charge recombination between QA − and the S2 state of manganese cluster (Debus 1992).
Extraction of PsbQ and PsbP polypeptides significantly slows the kinetics of QA − oxidation in samples (Fig. 2). The NaCl-treated PSII membranes displayed significantly slower time constants for the fast (τ = 7 ms) and intermediate (τ = 169 ms) decay phases relative to those of the intact membranes (τ = 0.9 and 23 ms correspondingly), indicating slower electron transfer from QA − to QB upon removal of the extrinsic proteins (Table 2). Thus, electron transfer from QA − to QB in PSII reaction centers with an occupied and unoccupied QB site was slowed by a factor of 8 and 7, respectively. These results correspond to data obtained by Roose et al. (2010), indicating that slowing factors are 12 and 6, respectively. Thus, our results demonstrate that extraction of PsbP and PsbQ extrinsic proteins from PSII membranes significantly slows down the electron transfer from QA − to QB and support data obtained by Roose et al. (2010).
Effect of Ca2+ and extrinsic proteins PsbP and PsbQ extraction on the QA − oxidation kinetics
Extraction of extrinsic proteins PsbP and PsbQ is accompanied by significant inhibition of O2 evolution activity (up to 15%) which can be restored up to 70–80% by exogenous Ca2+ in large concentration. Investigation of this effect has shown that the reason of inhibition is the damage of Ca2+-binding site in OEC, but not depletion of ExP (Ghanotakis et al. 1984b). It was suggested that ExP provides high efficiency of calcium cation binding with its site (Ghanotakis et al. 1984b). Without ExP, Ca2+ binds with low affinity that requires the high concentration of this cation. At the same time, the contamination of ExP-depleted PSII by Ca2+ can provide restoration of O2 activity upon reconstruction of ExP (Ghanotakis et al. 1984b). Therefore, to remove such Ca2+, the chelator (EDTA or EGTA) is used often during ExP extraction (Boussac et al. 1989; Ono and Inoue 1990). The obtained PSII membranes are designated as Ca2+-depleted PSII, whereas the samples prepared without EDTA are designated as ExP-depleted PSII.
In presented above experiments, we used ExP-depleted PSII membranes. Therefore, further investigation of the effect of EGTA and exogenous calcium cations on the electron-transport activity of ExP-depleted PSII samples could allow elucidate influences of Ca-depletion on the acceptor side of PSII. Addition of 5 mM EGTA to Exp-depleted PSII membranes with the subsequent incubation for 5 min in the dark at the room temperature slightly inhibits O2 evolving activity (without exogenous Ca2+). In doing so, the activity decreases from 15 to 8% (Table 1). This result demonstrates that ca. 7% of remaining O2 evolving activity in ExP-depleted PSII is determined by Ca2+ contamination of sample. Addition of exogenous Ca2+ to ExP-depleted PSII samples significantly (up to 70%) restores O2 evolution rate (Table 1). However, either incubation of ExP-depleted PSII with EGTA or with Ca2+ had no any effect on the decay kinetics of fluorescence in PSII membranes without PsbP and PsbQ extrinsic polypeptides (Fig. 3a, b). These results demonstrate that NaCl-washing effect on the acceptor side is determined by PsbP and PsbQ extraction, but not Ca2+ extraction from OEC. To prove this conclusion, we investigated QA − oxidation kinetics in Ca-depleted samples containing ExP polypeptides.
QA − oxidation kinetics in Ca-depleted PSII membranes containing all extrinsic proteins
There are two main methods of Ca2+ extraction from OEC of PSII membranes. The first method—the treatment of PSII samples with 1–2 M NaCl solution together with chelator like EDTA or EGTA (Ghanotakis et al. 1984b; Boussac et al. 1989; Ono and Inoue 1990) to get Ca-depleted PSII membranes. However, such samples also lose two extrinsic proteins PsbP and PsbQ. We used such method and corresponding samples above. Second method of Ca2+ extraction was developed by Ono and Inoue (1988) and resides in application of citrate buffer at low pH for treatment of membranes. It is important that the method allows extract Ca2+ cation from OEC without extraction of ExP. Therefore, we used this approach to investigate and distinguish the effects of Ca2+ and ExP extraction on the acceptor side of PSII. Samples prepared with this method is designated as PSII(−Ca,+ExP) membranes.
Extraction of Ca2+ at low pH with citrate is accompanied by the inhibition of oxygen evolution up to 12% which can be restored up to 69% by the addition of 30 mM Ca2+ (Table 1). In fact, these numbers are similar to corresponding ones for ExP-depleted PSII membranes (Table 1). EGTA (5 mM) decreases remaining O2-evolving activity to 6% as in the case of ExP-depleted PSII membranes. It is known that incubation of low pH citrate-washed PSII membranes with exogenous Ca2+ is accompanied by irreversible binding of Ca cations to its binding site in the OEC (Ono and Inoue 1988). This process needs several 10 min and bound Ca cation becomes resistant to chelator action (Ono and Inoue 1988). Investigation of the EGTA effect on the activity of PSII(−Ca,+ExP) membranes after incubation for 40 min with exogenous Ca2+ showed that remaining activity after EGTA addition was 33%, i.e., Ca2+ cation irreversibly bound to Ca-binding site of OEC, although there is pool of Ca2+ which can be extracted (Table 1). Ono and Inoue (1988) found 50% that is a little higher than ours. This difference can be explained by insignificant distinctions in a method—EGTA (5 mM) instead of EDTA (2 mM), 30 mM CaCl2 during reconstruction were used by us instead of 50 mM based on paper by Ono and Inoue. Thus, the results obtained by us are in line with data of Ono and Inoue (1988), demonstrating the extraction of Ca2+ cation from OEC by citrate.
Polyacrylamide gel electrophoresis in the presence of sodium dodecyl sulfate–urea has been used to study the composition of extrinsic proteins (PsbO, PsbP, and PsbQ) in PSII membranes treated with citrate buffer at pH 3.0 (Fig. 4). As controls, the electrophoretograms for intact PSII, NaCl-washed PSII, and Mn-depleted PSII membranes have been also obtained. Control sample lanes demonstrate that intact PSII sample contains all extrinsic proteins (lane 1); NaCl-washed membranes contain only PsbO polypeptide (lane 2) and PSII(−Mn) membranes do not contain extrinsic proteins (lane 3). All these results are in coincidence with knowledge about content of extrinsic proteins in such samples. Note that, based on the gel used by us, the PsbP band is localized very close to a band associated with the integral membrane protein of light-harvesting complex (Miller et al. 1987; Bricker 1992; Kuntzleman and Haddy 2009). Electrophoretogram of citrate-treated PSII membranes (Fig. 4, lane 4) has shown the presence in samples all extrinsic proteins that is in line with results of Ono and Inoue (1988). Thus, after citrate treatment according to a standard method (Ono and Inoue 1988), we have PSII membranes containing all ExP without Ca2+ cation in the OEC. Ca2+ extraction from the OEC by citrate buffer at low pH (3.0) is supported by inhibition of O2 evolution which can be restored by the addition of exogenous Ca2+ cation in large concentration (Table 1).
In the next experiments, we studied the kinetics of QA − oxidation in PSII(−Ca,+ExP) membrane fragments. Two kinetics of fluorescence decay after saturating flash are shown in Fig. 5a, namely fluorescence kinetics in (1) intact PSII membranes and in (2) Ca-depleted membranes containing all extrinsic proteins (low pH/citrate-washed samples with extrinsic proteins). Comparison of these kinetics clearly demonstrates that extraction from PSII only Ca cation does not change the rate of QA − oxidation [compare curve 1 (native PSII membranes) and curve 2 (Ca-depleted membranes containing ExP)], whereas extraction of ExP significantly delays the QA − oxidation [compare curve 1 (intact PSII membranes) on Fig. 5a and curve 1 (PsbP, PsbQ-depleted PSII) on Fig. 5b]. Quantitatively, the time constants of fast decay components reflecting electron transfer rate from QA − to QB are the same in PSII and PSII(−Ca,+ExP) membranes (compare 0.9 and 0.8 ms, respectively) (Table 2). However, citrate treatment slows the electron transfer from QA − to QB in centers with unoccupied QB site by factor 3 (Table 2), although it is significantly less than the effect of NaCl-washing on this parameter (factor of slowing 7).
QA − oxidation kinetics after a single saturating flash in low pH/citrate-treated PSII membranes with (A) or without (B) extrinsic (PsbP and PsbQ) proteins (10 µg Chl ml−1). Data were collected after dark incubation of sample for 5 min. A Curve 1 (square)—fluorescence decay in the intact PSII membranes [this kinetic is shown also in Fig. 1 (curve 1) and it is presented here for convenience to detect of citrate treatment effect]; curve 2 (circle)—fluorescence decay in pH 3.0/citrate-treated (Ca-depleted) PSII membranes with extrinsic proteins prepared according to Ono and Inoue (1988). B Curve 1 (rhombus)—fluorescence decay in the NaCl-washed PSII [this kinetic is shown also in Fig. 2 (curve 2) and it is presented here for convenience to detect of citrate treatment effect]; curve 2 (circle)—fluorescence decay in pH 3.7/citrate-treated PSII membranes without PsbP and PsbQ extrinsic proteins (see legend to Table 2 for method of preparation of sample)
Low pH method of Ca2+ extraction suggested by Ono and Inoue (1988) is based on the short-time treatment of PSII membranes with citrate at pH 3.0. After 5 min incubation, the sample is diluted by buffer to adjust pH to 6.5. Samples contain all extrinsic proteins, but not Ca2+. More detailed studies of low pH/citrate effect on extrinsic proteins have been carried out by Shen with coworkers (Shen and Katoh 1991; Shen and Inoue 1991). Shen and Katoh (1991) have shown that incubation of PSII membranes at pH 3.0 is accompanied by extraction of all extrinsic proteins, whereas adjusting of pH to 6.5 is accompanied by rebinding of extrinsic proteins, i.e., extrinsic proteins reconstruction takes place. Shen and Inoue (1991) also investigated the pH-dependent dissociation of extrinsic proteins and determined values of apparent pK for each protein. The pHs for half dissociation of 17, 23, and 33 kDa extrinsic proteins were determined to be 5.0, 4.1, and 3.6, respectively. These data are useful for preparation of low pH/citrate-treated samples without PsbP and PsbQ proteins. Such membrane fragments obtained before extrinsic proteins rebinding together with samples obtained after rebinding of ExP [PSII(−Ca,+ExP)] can be used to study the effect of reconstruction of ExP on the acceptor side of PSII. To prepare such samples, we suspended PSII membranes in citrate buffer (400 mM sucrose, 70 mM NaCl, 10 mM sodium citrate, pH 3.7) at 0.5 mg Chl ml−1. Such value of pH allows extract only PsbP and PsbQ proteins and prevent the extraction of Mn from OEC (Shen and Inoue 1991). The membranes were further incubated for 1 min at 4 °C in the dark and centrifuged for 5 min to remove dissociated ExP. The pellet was washed and then suspended in buffer A. The obtained samples do not contain PsbP and PsbQ extrinsic proteins according to electrophoresis (Fig. 4, lane 5) but retain PsbO polypeptide in accordance with the results obtained by Shen and Inoue (1991). Fluorescence kinetics (Fig. 5b, curve 2) correspond to kinetics of NaCl-treated PSII (Fig. 5b, curve 1) that also do not contain ExP. Kinetic parameters of fluorescence decay in low pH/citrate-treated PSII membranes without ExP are very similar to kinetic parameters of NaCl-treated PSII (Table 2). Thus, all these results show that during citrate treatment, extraction and reconstruction of extrinsic proteins restoring the kinetics of electron transfer between QA and QB occur.
X-ray crystallographic structures of PSII show that the extrinsic proteins directly interact with neither the Mn4CaO5 cluster nor the Cl− ions (Umena et al. 2011). Thus, extraction of PsbP and PsbQ extrinsic proteins from PSII should be accompanied by structural modification of core intrinsic proteins which provide the inactivation of OEC and translation of extraction effect on the acceptor side of PSII. Structural mechanism of the ExP interaction with OEC was recently studied using Fourier transform infrared spectroscopy (see for review Ifuku and Noguchi 2016; Roose et al. 2016). PSII membranes depleted of PsbP and PsbQ showed clear changes in amide I bands which reflect structural changes in polypeptide main chains in S2-minus-S1 FTIR difference spectra (Tomita et al. 2009). These results together with reconstruction data indicate that PsbP protein, but not PsbQ or PsbO, affects the protein conformation around the Mn4CaO5 cluster. No changes were observed in the carboxylate and imidazole CN stretching bands which had been assigned to manganese cluster ligands. However, the molecular mechanism of the interactions of individual extrinsic proteins which provide OEC stability remains to be clarified. This note also concerns to the mechanism of the translation of ExP effects on the reducing site of PSII.
Conclusions
NaCl washing of PSII membranes extracts the PsbP and PsbQ peripheral proteins and influences on the calcium-binding site in the OEC. Such treatment is accompanied by inhibition of the OEC and slowing down of electron transfer from QA − to QB on the donor and acceptor sides of PSII, respectively. We have attempted to answer the following questions: is this effect due to the extraction of ExP or is it determined by damage of Ca2+ function within OEC? Measurements of QA − decays in the NaCl-washed PSII membranes in the presence or absence of Ca2+/EGTA and in the Ca-depleted PSII membranes containing ExP clearly have shown that acceptor-side effect is determined by the extraction of PsbP and PsbQ extrinsic proteins, but not Ca2+ cation from OEC.
Abbreviations
- Chl:
-
Chlorophyll
- DCMU:
-
3-(3,4-Dichlorophenyl)-1,1-dimethylurea
- EGTA:
-
Ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic
- ExP:
-
PsbP and PsbQ extrinsic polypeptides
- LHC:
-
Light-harvesting chlorophyll protein complex
- MES:
-
2-(N-Morpholino)ethanesulfonic acid
- OEC:
-
Oxygen-evolving complex
- PSII:
-
Photosystem II
- PSII(−Mn):
-
Mn-depleted PSII samples
- PSII(−ExP):
-
PsbP and PsbQ extrinsic polypeptides-depleted PSII
- PSII(−Ca,−ExP):
-
Ca-depleted PSII membranes lacking PsbQ and PsbP proteins
- PSII(−Ca,+ExP):
-
Ca-depleted PSII membranes containing PsbQ and PsbP extrinsic proteins
References
Allakhverdiev SI, Tsuchiya T, Watabe K, Kojima A, Los DA, Tomo T, Klimov VV, Mimuro M (2011) Redox potentials of primary electron acceptor quinone molecule (QA)- and conserved energetics of photosystem II in cyanobacteria with chlorophyll a and chlorophyll d. Proc Natl Acad Sci USA 108:8054–8058
Andréasson LE, Vass I, Styring S (1995) Ca2+ depletion modifies the electron transfer on both donor and acceptor sides in photosystem II from spinach. Biochim Biophys Acta 1230:155–164
Barber J (2008) Photosynthetic generation of oxygen. Phil Trans R Soc B 363:2665–2674
Berthold DA, Babcock GT, Yocum CF (1981) A highly resolved oxygen-evolving photosystem II preparation from spinach thylakoid membranes. FEBS Lett 134:231–234
Bouges-Bocquet B (1973) Electron transfer between the two photosystems in spinach chloroplasts. Biochim Biophys Acta 314:250–256
Boussac A, Rutherford AW (1988) Nature of the inhibition of the oxygen-evolving enzyme of photosystem II induced by sodium chloride washing and reversed by the addition of calcium(2+) or strontium(2+). BioChemistry 27:3476–3483
Boussac A, Zimmermann J-L, Rutherford AW (1989) EPR signals from modified charge accumulation states of the oxygen evolving enzyme in Ca2+-deficient photosystem II. BioChemistry 28:8984–8989
Bowes J, Crofts AR (1980) Binary oscillations in the rate of reoxidation of the primary acceptor of photosystem II. Biochim Biophys Acta 590:373–389
Bricker TM (1992) Oxygen evolution in the absence of the 33-kilodalton manganese-stabilizing protein. Biochemistry 31:4623–4628
Bricker TM, Ghanotakis DF (1996) Introduction to oxygen evolution and the oxygen-evolving complex. In Ort DR, Yocum CF (eds) Oxygenic photosynthesis: the light reactions, vol 4. Kluwer Academic Publishers, Boston, pp 113–136
Crofts AR, Wraight CA (1983) The electrochemical domain of photosynthesis. Biochim Biophys Acta 726:149–185
Dau H, Haumann M (2007) Eight steps preceding O–O bond formation in oxygenic photosynthesis: a basic reaction cycle of the photosystem II manganese complex. Biochim Biophys Acta 1767:472–483
Debus RJ (1992) The manganese and calcium ions of photosynthetic oxygen evolution. Biochim Biophys Acta 1102:269–352
Ghanotakis DF, Babcock GT (1983) Hydroxylamine as an inhibitor between Z and P680 in photosystem II. FEBS Lett 153:231–234
Ghanotakis DF, Babcock GT, Yocum CF (1984a) Calcium reconstitutes high rates of oxygen evolution in polypeptide depleted Photosystem II preparations. FEBS Lett 167:127–130
Ghanotakis DF, Topper JN, Babcock GT, Yocum CF (1984b) Water-soluble 17and 23 kDa polypeptides restore oxygen evolution activity by creating a high-affinity binding site for Ca2 the oxidizing sideof photosystem II. FEBS Lett 170:169–173
Ido K, Gross CM, Guerrero F, Sedoud A, Lai TL, Ifuku K, Rutherford AW, Krieger-Liszkay A (2011) High and low potential forms of the QA quinone electron acceptor in photosystem II of Thermosynechococcus elongatus and spinach. J Photochem Photobiol B 104:154–157
Ifuku K, Noguchi T (2016) Structural coupling of extrinsic proteins with the oxygen-evolving center in photosystem II. Front Plant Sci 7:84
Kashino Y, Koike H, Satoh K (2001) An improved sodium dodecyl sulfate-polyacrylamide gel electrophoresis system for the analysis of membrane protein complexes. Electrophoresis 22:1004–1007
Kok B, Forbush B, McGloin M (1970) Cooperation of charges in photosynthetic O2 evolution. I. A linear four-step mechanism. Photochem Photobiol 11:467–475
Krieger A, Weis E (1992) Energy-dependent quenching of chlorophyll a fluorescence: the involvement of proton calcium exchange at photosystem II. Photosynthetica 27:89–98
Krieger A, Rutherford AW, Johnson GN (1995) On the determination of the redox midpoint potential of the primary quinone acceptor, QA, in photosystem II. Biochim Biophys Acta 1229:193–201
Kuntzleman T, Haddy A (2009) Fluoride inhibition of photosystem II and the effect of removal of the PsbQ subunit. Photosynth Res 102:7–19
Kuwabara T, Murata N (1982) Inactivation of photosynthetic oxygen evolution and concomitant release of three polypeptides in the photosystem II particles of spinach chloroplasts. Plant Cell Physiol 23:533–539
Miller A-F, de Paula JC, Brudvig GW (1987) Formation of the S2 state and structure of the Mn complex in photosystem II lacking the extrinsic 33 kilodalton polypeptide. Photosynth Res 12:205–218
Miyao M, Murata N (1984) Calcium ions can be substituted for the 24 kDa polypeptide in photosynthetic oxygen evolution. FEBS Lett 168:118–120
Müh F, Glöckner C, Hellmich J, Zouni A (2012) Light-induced quinone reduction in photosystem II. Biochim Biophys Acta 1817:44–65
Murata N, Miyao M (1985) Extrinsic membrane proteins in the photosynthetic oxygen-evolving complex. Trends Biochem Sci 10:122–124
Najafpour MM, Renger G, Hołyńska M, Moghaddam AN, Aro EM, Carpentier R, Nishihara H, Eaton-Rye JJ, Shen JR, Allakhverdiev SI (2016) Manganese compounds as water-oxidizing catalysts: from the natural water-oxidizing complex to nanosized manganese oxide structures. Chem Rev 116(5):2886–2936
Ono T, Inoue Y (1988) Discrete extraction of the Ca atom functional for O2 evolution in higher plant photosystem II by a simple low pH treatment. FEBS Lett 227:147–152
Ono T, Inoue Y (1990) Abnormal redox reactions in photosynthetic O2-evolving centers in NaCl/EDTA-washed PS II A dark-stable EPR multiline signal and an unknown positive charge accumulator. Biochim Biophys Acta 1020:269–277
Porra RJ, Tompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous-equations for assaying chlorophyll-A and chlorophyll-B extracted with 4 different solvents: verification of the concentration of chlorophyll standards by atomic-absorption spectroscopy. Biochim Biophys Acta 975:384–394
Preston C, Seibert M (1991) The carboxyl modifier 1-ethyl-3-[3-(dimethylamino) propyl] carbodiimide (EDC) inhibits half of the high-affinity manganese-binding site in photosystem II membrane fragments. Biochemistry 30:9615–9624
Reifarth F, Christen G, Seeliger AG, Dormann P, Benning C, Renger G (1997) Modification of the water oxidizing complex in leaves of the dgd1 mutant of Arabidopsis thaliana deficient in the galactolipid digalactosyldiacylglycerol. BioChemistry 36:11769–11776
Renger G (2007) Oxidative photosynthetic water splitting: energetics, kinetics and mechanism. Photosynth Res 92:407–425
Renger G, Kuhn P (2007) Reaction pattern and mechanism of light induced oxidative water splitting in photosynthesis. Biochim Biophys Acta 1767:458–471
Renger G, Gleiter HM, Haag E, Reifarth F (1993) Photosystem II: thermodynamics and kinetics of electron transport from QA- to QB and to QB- and deleterious effects of copper. Z Naturforsch 48c:234–240
Roose JL, Frankel LK, Bricker TM (2010) Documentation of significant electron transport defects on the reducing side of photosystem II upon removal of the PsbP and PsbQ extrinsic proteins. Biochemistry 49:36–41
Roose JL, Frankel LK, Mummadisetti MP, Bricker TM (2016) The extrinsic proteins of photosystem II: update. Planta 243:889–908
Semin BK, Davletshina LN, Ivanov II, Rubin AB, Seibert M (2008) Uncoupling of processes of molecular synthesis and electron transport in the Ca2+-depleted PSII membrane. Photosynth Res 98:235–249
Shen J-R, Inoue Y (1991) Low pH-induced dissociation of three extrinsic proteins from O2-evolving photosystem II. Plant Cell Physiol 32:453–457
Shen J-R, Katoh S (1991) Inactivation and calcium-dependent reactivation of oxygen evolution in photosystem II preparation treated at pH 3.0 or with high concentrations of NaCl. Plant Cell Physiol 32:439–446
Shinkarev VP (2004) Photosystem II: oxygen evolution and chlorophyll a fluorescence induced by multiple flashes. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence: a signature of photosynthesis. Advances in photosynthesis and respiration. Kluwer Academic Publishers, Dordrecht, pp 197–229
Shinkarev VP, Wraight CA (1993) Oxygen evolution in photosynthesis: from unicycle to bicycle. Proc Natl Acad Sci USA 90(5):1834–1838
Tomita M, Ifuku K, Sato F, Noguchi T (2009) FTIR evidence that the PsbP extrinsic protein induces protein conformational change saround the oxygen-evolving Mn cluster in photosystem II. BioChemistry 48:6318–6325
Umena Y, Kawakami K, Shen J-R, Kamiya N (2011) Crystal structure of oxygen evolving photosystem II at a resolution of 1.9 Å. Nature 473:55–60
Velthuys BR, Amesz J (1974) Charge accumulation at the reducing side of system 2 of photosynthesis. Biochim Biophys Acta 333:85–94
Vinyard DJ, Brudvig GW (2017) Progress toward a molecular mechanism of water oxidation in photosystem II. Annu Rev Phys Chem 68:4.1–4.16
Weiss W, Renger G (1984) UV spectral characterization in Tris-washed chloroplasts of the redox component D1 which functionally connects the reaction center with the water-oxidizing enzyme system Y in photosynthesis. FEBS Lett 169:219–223
Wydrzynski TJ, Satoh K (eds) (2005) Photosystem II: the light-driven water: plastoquinone oxidoreductase. Springer, Dordrecht
Acknowledgements
We want to thank prof. A. Yu. Semenov for valuable discussions and Dr. L. E. Petrovskaya for help with the SDS-PAGE analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Semin, B.K., Davletshina, L.N. & Mamedov, M.D. Effect of different methods of Ca2+ extraction from PSII oxygen-evolving complex on the QA − oxidation kinetics. Photosynth Res 136, 83–91 (2018). https://doi.org/10.1007/s11120-017-0441-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-017-0441-4