Abstract
Background
Three–dimensional root architectural models emerged in the late 1980s, providing an opportunity to conceptualise and investigate that all important part of plants that is typically hidden and difficult to measure and study. These models have progressed from representing pre–defined root architectural arrangements, to simulating root growth in response to heterogeneous soil environments. This was done through incorporating soil properties and more complete descriptions of plant function, moving into the realm of functional-structural plant modelling. Modelling studies are often designed to investigate the relationship between root architectural traits and root distribution in soil, and the spatio–temporal variability of resource supply. Modelling root systems presents an opportunity to investigate functional tradeoffs between foraging strategies (i.e. shallow vs deep rooting) for contrasting resources (immobile versus mobile resources), and their dependence on soil type, rainfall and other environmental conditions. The complexity of the interactions between root traits and environment emphasises the need for models in which traits and environmental conditions can be independently manipulated, unlike in the real world.
Scope
We provide an overview of the development of three–dimensional root architectural models from their origins, to their place today in the world of functional–structural plant modelling. The uses and capability of root architectural models to represent virtual plants and soil environment are addressed. We compare features of six current models, RootTyp, SimRoot, ROOTMAP, SPACSYS, R-SWMS, and RootBox, and discuss the future development of functional-structural root architectural modelling.
Conclusion
Functional-structural root architectural models are being used to investigate numerous root–soil interactions, over a range of spatial scales. They are not only providing insights into the relationships between architecture, morphology and functional efficiency, but are also developing into tools that aid in the design of agricultural management schemes and in the selection of root traits for improving plant performance in specific environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Root system architecture (RSA) is a fundamental component of plant productivity. It determines the capacity for a plant to forage for, and acquire, resources in the dynamic and variable soil environment (Fitter 2002; Lynch 2005, 2007; Pagès 2011; Lynch and Brown 2012). How roots are spatially distributed through soil impacts on a range of plant functions including; anchorage, storage, transport, the acquisition of spatially variable resources (mobile and immobile), and competition for space, water and nutrients (Smith and De Smet 2012). While the notion of an ‘optimal’ root architecture is complex (Robinson 1988; Fitter 1991; Dunbabin et al. 2003b), the concept of ‘tailoring’ root systems to specific environments in order to improve crop productivity is gaining momentum (Dunbabin et al. 2003a, 2004; Wu et al. 2005; de Dorlodot et al. 2007; Lynch 2007; Gregory et al. 2009; Ao et al. 2010; Bingham and Wu 2011; Chen et al. 2011; Lynch and Brown 2012). World phosphate reserves are limited and agricultural management practices will need to be adapted to incorporate crop species that can produce yield under less phosphate fertilisation (Smith and de Smet 2012). Increased droughts and changes in climate will also increase stress on crops. Specifically adapted cultivars will be required in some areas in order to sustain yields. Much of this adaptation is due to the below ground parts, since they are the sites of soil-plant interactions.
A range of root traits have been linked to plant performance in specific environments (e.g. Gahoonia and Nielsen 2004; Lynch 2007; Lynch and Brown 2012). Root length and surface area play an important role in the uptake of both immobile and mobile soil resources (Brück et al. 1992; Lynch and van Beem 1993; Wiesler and Horst 1994; Brady et al. 1995; Dunbabin et al. 2003a, b, 2004; Zhu and Lynch 2004; Liao et al. 2006). Fine roots and fine root structures, including root hairs and cluster roots, define the precision with which plants forage, enabling small localised soil volumes to be exploited with high efficiency (Campbell et al. 1991; Grime et al. 1991; Eissenstat and Yanai 1997; Bates and Lynch 2000; Gilroy and Jones 2000; Lambers et al. 2006, 2011). Rooting depth plays an important role in reducing nitrogen losses through leaching and improving drought tolerance (Thorup-Kristensen 2001; Dunbabin et al. 2003a; Ho et al. 2005; Steele et al. 2007; Bernier et al. 2009), while shallow rooting is important for the acquisition of immobile resources such as phosphorus (P) (e.g. Lynch and Brown 2001; Zhu and Lynch 2004). Thus, for resource acquisition it is important that the distribution of roots through the soil profile coincides with the distribution of resources in the soil profile. The simulation of RSA reduces the root distribution problem to measurable phenes (phenotypic traits under genetic control) such as root angles, growth rates of individual root tips and branching frequencies, root traits that may be under tight genetic control, and therefore amenable to targeting in breeding programmes (Pagès 2011).
The capacity of root systems to respond to spatially and temporally variable resource supply plays a role in the efficient acquisition of water and nutrients (Robinson 1994, 1996; Lynch and Brown 2001, 2012; Dunbabin et al. 2004; Walk et al. 2006). Plants generally respond to low nutrient availability by reducing shoot growth, thereby reducing their leaf and stem mass fractions (Poorter et al. 2012b). Root growth rates are often less affected and may even increase, only reducing at the more severe stress levels. However, even when resource availability has no effect on the total length or biomass of the root system, there are often strong changes in RSA in response to resource availability (Smith and De Smet 2012). For example, common bean genotypes often increase the root surface area in the relatively P–rich topsoil layers by increasing basal root shallowness and adventitious rooting in response to low soil P availability (Liao et al. 2001; Lynch and Brown 2001; Miller et al. 2003). Shallow seminal rooting in maize also improved phosphorus acquisition efficiency in low–phosphorus soil (Zhu et al. 2005), whereas greater lateral root length and density in maize improved P acquisition and plant growth under low P availability (Zhu and Lynch 2004).
Plants can respond locally to the spatially and temporally variable soil environment through a number of mechanisms, including: root hair morphology, root exudation, cluster root growth, up–regulation of nutrient transport carriers, and locally enhanced root proliferation (Robinson 1994, 1996, 2005; Dinkelaker et al. 1995; Ryan et al. 2001; Vance et al. 2003; Gahoonia and Nielsen 2004; Lambers et al. 2006, 2008, 2011; Lynch 2007; Lynch and Brown 2008; Zhu et al. 2010b). These local plasticity responses allow plants to forage with precision in a spatially and temporally heterogeneous environment, minimising the metabolic cost of soil exploration by matching plant investment in root biomass and root function with resource supply in soil (Dunbabin et al. 2003b; Lynch 2007).
The diverse range of complex interactions between root systems and their soil environment, and the difficulties associated with visualising and measuring them, make studying the plant–soil continuum a challenge. This paper will elucidate the role that root architectural models play in investigating these interactions. Three–dimensional root architectural models hold particular promise because they allow the precise description of root architecture in space and time, down to the individual root level (Diggle 1996; Pagès 1999). The models provide a framework for studying the growth of root systems in response to supplies of soil resources that vary in space and time, and are a valuable tool for the visual communication of complex ideas (Diggle 1996; Doussan et al. 2003; de Dorlodot et al. 2007).
Development of three–dimensional root architecture models
Increases in computing power in the 80s and 90s enabled the development of more complex root models. Previous models relied upon one–dimensional functions of rooting depth vs time (Subbaiah and Rao 1993), or two–dimensional functional descriptions of root length densities with time and depth, such as diffusion–based models and percentage–distribution–with–depth models (Page and Gerwitz 1974). They were limited in their application and unable to accurately represent three–dimensional root systems (Hutchinson 2000). The root growth model of Lungley (1973) was the first true architectural model. It described, in two–dimensional radial coordinates, the growth of a tap root and two orders of laterals, with fixed growth rates and branch spacing, and a slope increment for the first order laterals. Even though this model was simple in its function, it was the precursor for the suite of three–dimensional (3D) root architectural models that were developed in the 80s and 90s (Deans and Ford 1983; Diggle 1988a, b; Pagès and Aries 1988; Pagès et al. 1989; Fitter et al. 1991; Clausnitzer and Hopmans 1993, 1994; Lynch et al. 1997; Spek 1997).
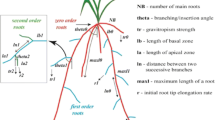
Root architectural simulation models explicitly simulate the architecture of root systems in 3D space (Figs. 1, 2 and 3). RSA is usually represented by connected points. Growth rates, growth direction and branching patterns typically need to be provided in order to simulate the development of the root system. The earliest 3D root architecture model is that of Deans and Ford (1983), developed for representing tree root systems. While few details of the model were provided, it was capable of representing excavated tree root systems, and considering the effect of wind pressure on tree development. Models for representing annual fibrous root systems quickly followed, and employed similar methods to each other for describing the basic growth of a root (Diggle 1988a; Pagès et al. 1989; Clausnitzer and Hopmans 1994). They used a set of growth rules, which are applied to a series of root types or classes, with each root type having its own characteristic set of growth parameters (Fig. 1). In ROOTMAP (Diggle 1988a, b), root elongation rate, branching density and branching delay time are the primary growth parameters.
Rendering of different root architectures simulated by the 3D root architectural model SimRoot at 40 days after germination. Roots have been dilated for better visibility, the deepest roots are at about 1.50 m. a Lupin, with a relatively small root system with strong primary root growth and relatively short first and second order laterals. b Bean, with a primary root in the middle, 8 basal roots coming from the base of the hypocotyl (these basal roots may grow longer than the primary root). The relatively fine hypocotyl-borne roots emerge later. All major root axes have first order laterals. Strongly developed first order laterals may have secondary order laterals. c Squash, with a strongly developed conical primary root. From the primary roots many lateral branches appear, and some may grow up to a meter long and have secondary growth. Lateral branching goes up to three orders. Total length is comparable to bean, despite the larger shoot size of the squash plant. d Maize, with the most extensive (~0.5 km) and complex root system of all four species shown, comprising a primary root, three seminal roots, successive whorls of crown roots increasing in diameter and the early development of brace roots. All major root axes have first order laterals of which some develop second and third order laterals
Three kinds of root systems simulated with RootTyp (Pagès et al. 2004). a Secondary root system, with a large number of adventitious roots originating from the base of the plant. It is typical of many monocotyledonous species (e. g. poacee family). Self-pruning has started at the base of the oldest nodal roots. b Primary root system, found in dicotyledonous species. The central taproot is apparent, and exhibits significant radial growth at this stage. The main lateral roots are partially self-pruned in their old proximal part. c Secondary root system, whose main roots originated from a horizontally growing rhizome. The particular shape and structure of this root system emerged from its specific developmental pattern
The ability to utilise both time and distance parameters allows root age factors to play a role in determining root growth and branching (Diggle 1988b, 1996). Random deviation is typically used in root architectural models to determine root deflection, combining a deflection index (tendency to deflect from the previous growth direction) with a geotropism index (tendency for a root to grow preferentially downward) (Fig. 1; Diggle 1988c; Pagès et al. 1989; Clausnitzer and Hopmans 1994; Lynch et al. 1997). The stochastic elements of these models allow simulated root systems to bend and deflect in a similar way to that observed in the field, without requiring knowledge of the complex phenomena that govern root deflections (Pagès 1999).
Using root architectural models to represent real root systems
Visual comparison of simulated root architectures reveals that the developmental architectural modelling approach can represent the root architecture of a diverse array of plant species (Figs. 2, 3 and 4; Pagès et al. 2004; Pierret et al. 2007; Leitner et al. 2010a; Chen et al. 2011), with larger architectural features often better represented than small–scale morphology. Root architectural models typically require a larger set of parameters compared to more commonly used root distribution models. The advantage is that those parameters are directly and independently measurable and thus the models are less reliant on calibration alone. This provides an opportunity to investigate and test at a mechanistic level, the processes and interactions occurring in the plant-soil system. These interactions are complex, and models provide an opportunity to reduce the number of experiments needed to investigate them, targeting experimentation to those combinations that, according to the simulation exercise, may be of special interest (Meyer et al. 2009; Lynch 2011). Such models can be also used in an inverse mode to estimate root parameters which are hardly measurable in situ. Garré et al. (2012) tried to optimize several of these parameters by inverse modelling based on mini-rhizotron measurements. Although their set-up was not optimal enough to get a good appraisal of these parameters, they nicely illustrated that combining these models with a powerful optimization algorithm could be highly relevant for future studies.
a Observed 2D Trifolium repens root system (15 days after seedling transplant) and b) simulated 3D root system using the SPACSYS model. From Wu et al. (2007)
Large numbers of parameters also bring about a limitation to models, with unintended interactions between model parameters and the uncertainties associated with each parameter due to measurement error. It is important to understand the degree to which individual parameters, measurement error associated with those parameters, and the choice of model algorithms, affect model behaviour. Unfortunately, these types of in-depth investigations are rarely done for individual crop and plant models. Sensitivity analysis approaches do exist, however, that enable parameter-intensive models to be investigated, and need to be applied not only for understanding individual models, but also for benchmarking across models (Morris 1991; Saltelli et al. 2000; Dunbabin 2007).
The representation of root architecture has been useful for studying those geometric aspects of root systems that are often difficult to determine experimentally. For example, the fractal dimensions of different root architectures might be genotype specific (Bohn et al. 2006) and potentially correlate with nutrient uptake under field conditions (Nielsen et al. 1998; Walk et al. 2004). However, less abstract measures can also be insightful. Virtual coring may explain how root length densities in cores from the field depend on the coring position relative to the plant (Miguel et al. 2012). Simulations may explain how root architecture affects the number of roots seen at different positions in minirhizotron tubes (Pagès and Bengough 1997), and for investigating field estimations of root length densities (Bengough et al. 1992). Root architectural models may also be used to understand how root length distribution with depth changes over time, and can thereby be used to generate input for crop models (Wu et al. 2007; Postma et al. 2008). With this approach, root architectural traits can be linked with crop performance in the field, combining the mechanistic elements of the root architectural models with the predictive power of crop models (Bingham and Wu 2011).
Root models are increasingly capable of representing root growth in response to objects and barriers in soil (Figs. 5 and 6; Table 1; Leitner et al. 2010b; Dunbabin et al. 2011). The ROOTMAP model, for example, has been modified to represent barriers, enabling the model to simulate the direction of root growth being affected by the presence of impenetrable objects such as pot walls, divisions in split–pot experiments and rocks (Dunbabin et al. 2011). Roots can also interact, through having their direction and rate of growth, and root thickness changed (Bengough et al. 2011; Bengough 2012), with objects of varying degrees of permeability such as harder layers in soils (Fig. 5). This provides an opportunity to understand the significance and relevance of plant behaviour in pots (Poorter et al. 2012a) and to scale up pot studies and other artificial environments in which root systems are confined, to unbounded field environments.
Seminal root systems of three wheat plants simulated with ROOTMAP. 1.) Left plant was grown with a band of phosphorus fertiliser placed at a depth of 6 cm below the seed. Proliferation of roots around the band of fertiliser can be seen in the top-soil. 2.) The middle plant was grown with no added fertiliser P, and with roots growing into a hard layer of soil (grey box, penetration probability 0.6) at a depth of 16–22 cm. This hard layer of soil has restricted subsoil exploration by the root system. 3.) Right plant was grown without P fertiliser and without changes in soil penetrability down the profile
a Root growth and (b) phosphate uptake by a growing root system as affected by inhomogeneous (split pot) initial phosphate distribution simulated by RootBox. The initial phosphate concentration in the right half of the pot was twice (1e-4 μmol cm−3) that of the left half (0.5e-4 μmol cm−3), resulting in a denser root system in the right half. Simulation time was 20 days. Spatial resolution along root axis was 0.2 m, resolution of soil model was 0.5 cm. Phosphate uptake was computed via a sink term in the CDE by averaging the P influxes into the root segments over the representative elementary volumes given by the cubes
The development of new non–destructive measurement techniques for imaging root architecture in situ has triggered renewed interest in the geometric properties of root systems. Techniques such as tomography in transparent gels (Clark et al. 2011), computed tomography (CT, Flavel et al. 2012), neutron radiography (Oswald et al. 2008) and magnetic resonance imaging (MRI, Jahnke et al. 2009; Schulz et al. 2012) make it possible to determine non–destructively the geometric properties of real root systems, allowing researchers to distinguish closely related genotypes, and to study plasticity responses to environmental factors. Interpretation of the acquired 3D datasets, however, remains difficult. Root architectural models may aid in the interpretation of the observed architectures. Schulz et al. (2012) used a root architectural model to test algorithms that were developed for segmenting roots from MRI images. Functional interpretation from such images was done by Pohlmeier et al. (2010), who used the root architecture of actual plants, imaged with MRI, to simulate water uptake from soil. This highlights another aspect of root architectural models, namely that they have moved beyond only representing root architecture, to providing a functional interpretation of root system architecture and describing benefits and tradeoffs of specific architectures in different environments (Dunbabin et al. 2003a, 2004; Ho et al. 2005). These models are being increasingly defined as functional-structural root models, or functional-structural plant models, for those models containing shoot modules.
Simulating root systems in their environment– soil, water and nutrient dynamics and carbon utilisation
The original root architecture models were restricted to growing pre–defined root architectures (Diggle 1988a; Pagès et al. 1989; Pagès 1999). It was, however, quickly identified that the real value of these models would be in the capacity to represent dynamic root systems that both modify, and are modified by, their environment, with individual roots responding to local conditions (Pagès 1999, 2000; Doussan et al. 2003). Experimental evidence has shown that the coordinated responses of root systems are an important aspect of plant function, particularly under heterogeneous resource supply (Drew et al. 1973; Drew and Saker 1975). In order to represent responsive root growth, root architecture models are coupled with a model of the soil environment. Water and nutrient uptake are important root functions, and the root system is an important sink for carbohydrates. The explicit nature of the models allows for precise accounting of carbon costs, and the relatively mechanistic implementation of nutrient and water uptake. However, the processes involved in plant resource acquisition occur at vastly different scales, from the size of uptake transporters at the molecular level to the scales of practical applications, i.e., root system and field scales. Suitable up-scaling methods can link the information across scales without losing relevant information (Roose and Schnepf 2008).
Up-scaling from the single root to the root system scale
For small root systems, direct solution of the three-dimensional problem using the root surfaces as geometric boundaries in a numerical simulation are feasible (see Fig. 7). For large root systems, this is mostly too computer intensive, and up-scaling methods provide suitable tools to develop simpler effective equations that still include all the relevant information from the smaller scale(s). Leitner et al. (2010c) modelled phosphate uptake by a small (12 days old) maize root system explicitly in three dimensions. The results compared well to the model presented in Roose et al. (2001), where phosphate uptake was computed by up-scaling an approximate analytical solution for the single root model to the root system scale.
In every up-scaling/averaging process information is lost. The art is to use up-scaling methods that keep just enough information to adequately describe the problem, while keeping the simulation numerically tractable. Leitner et al. (2010c) used the root surfaces of a three-dimensional root system as geometric boundaries in a soil model, which is the most accurate spatial description of the problem. However, this approach is limited by the large memory requirements, as well as the difficulties in the numerical implementation, and thus can only be used for small root systems with few side branches. Advantages of this approach include, first, the possibility of verifying simplifications that are made in the up-scaling process. Second, it is possible to extend existing models by including spatial heterogeneities. The proposed method can access inter-root competition, root dynamics, as well as differences in root function along the root axis.
Explicit three-dimensional simulation of water uptake by a 13 day old maize root system. The root system was computed using the RootBox model, the finite element mesh (a) was computed in Matlab and the dynamics of water movement in the soil were simulated with Comosol Multiphysics: (a) Transparent representation of the finite element mesh on which the water content was computed. The mesh is finer close to the roots than away from them.; (b) Effective saturation after two hours at initial effective saturation 0.2; (c) Effective saturation after two hours at initial effective saturation 0.4
Various mathematical up-scaling methods are available for dealing with problems that have complicated microscopic geometries such as root systems. The aim is to simplify those problems in order to reduce computational expense (Wallstrom et al. 2002). Recently, the method of homogenisation was applied to model phosphate uptake by a root with root hairs (Leitner et al. 2010c; Ptashnyk 2010; Zygalakis et al. 2011) and by cluster roots (Zygalakis and Roose 2012). For up-scaling from single root to root system scale, the most widely used method is to use a root architecture model coupled with a soil model and to average root uptake over a certain soil volume (Somma et al. 1998; Dunbabin et al. 2006; Javaux et al. 2008; Bear and Cheng 2010; Schnepf et al. 2012). Yet the question of the appropriate soil voxel size is still open (Schröder et al. 2009a) and may depend on the type of process being simulated.
For the purpose of up-scaling, less computer intensive density-based models were presented by Dupuy et al. (2010). Such models are continuous spatial models of root density that include more developmental parameters than root depth models. Dupuy et al. (2010) suggest that such models could have a wide range of applications, particularly where classic architectural models are faced with computational limitations, for example population models or breeding scenarios, or due to the difficulty in calibrating the models efficiently.
Simulation of water and nutrient uptake is challenging especially if we consider the soil conditions in the rhizosphere around the roots, which are often quite different from those in the bulk soil. Rhizosphere processes are known to play an important role in plant phosphate acquisition, for example; however they are still poorly accounted for in models of plant nutrition (Hinsinger et al. 2011). With respect to nutrients of low mobility such as phosphate, Hinsinger et al. (2011) discuss several rhizosphere processes that are still lacking in crop models. Strategies to increase phosphate availability in soil related to rhizosphere biophysics include root architecture, root hairs or root extensions by mycorrhizal hyphae. Strategies related to rhizosphere biogeochemistry are for example the exudation of protons, carboxylates or phosphatases. Other potential strategies could exploit traits related to the physiology of associated microorganisms, either symbiotic such as mycorrhizal fungi or free-living rhizosphere microorganisms such as phosphate-solubilising bacteria and fungi.
The problem partly arises from the different scales which easily vary by two to three orders of magnitude or, in three dimensions, 6–9 orders (Postma et al. 2008). While most root architectural models do poor justice to these scale differences, several solutions have been tried with R–SWMS. Schröder et al. 2009b) demonstrated that in their scenario, a fine finite–element grid gave a more accurate solution than a coarse grid, and that the grid refinement can be localized around the roots without loss in accuracy. A priori grid refinement can thereby significantly reduce the computational time of a simulation. However, most of the gain of a locally refined grid was lost when the grid could not be refined a priori, but had to be adapted to the (changing) architecture of the root system during simulation. The grid refinement method allowed Schröder et al. (2008) to estimate the effect of local soil drying around the roots on root water uptake; they concluded that extraction of water by the roots resulted in local drying of the rhizosphere, reducing the hydraulic conductivity of the rhizosphere and thereby limiting further water uptake by the roots (similar conclusions were reached by Van Lier et al. 2006; Metselaar and Van Lier 2007).
Simulating water uptake dynamics
Soil water dynamics are a fundamental component of the plant–soil system, directly affecting plant productivity and crop yields. A number of modelling approaches have been adopted to investigate the interaction between soil water dynamics and root architecture. Clausnitzer and Hopmans (1994) were the first to combine a root architectural model with a 3D hydrological model in order to simulate water uptake by root systems. The root water uptake term was computed by dividing transpiration over the root length. Somma et al. (1998) developed the code further by allowing the water uptake activity of the roots to change with root age. In the same year, Doussan et al. (1998) presented an alternative approach in which water flow transport equations were not only solved for the soil, but also for the plant. This approach allowed roots in wetter soil regions to compensate for roots in dryer soil regions; it required an understanding of the radial and lateral conductivities of the root system and was used to show that xylem development may, under specific conditions, limit water uptake from deeper soil layers (Pierret et al. 2007). Using this approach, Doussan et al. (2006) simulated water depletion from soil by contrasting lupin root architectures. The model represented contrasting extraction fronts for the two root architectures, simulated the zone of active water uptake moving across the root system with time, and water release from the roots into the top–soil through a process of hydraulic lift from moist sub–soil layers. This combination of root architecture, root system hydraulic properties and water transfer through soil was a significant advance in plant–soil water modelling, representing a complete soil–plant continuum driven by water potential gradients. This approach holds the potential for very detailed investigations of the soil water interactions that occur in real root systems, contributing to the understanding of the hydraulic architecture of root systems.
The models of Doussan et al. (2006) and Somma et al. (1998), were merged into one 3D root–soil water model named R–SWMS (Javaux et al. 2008 ; Draye et al. 2010; Pohlmeier et al. 2010; Table 1; Fig. 8; also see further detail below). Javaux et al. (2008) used the R–SWMS model to investigate further the relative contributions of root and soil hydraulic conductivities to water uptake by roots. The modelled scenarios showed the complexity of root–soil water interactions, and the inability of the traditional 1D sink terms for water uptake to represent water dynamics at the plant scale. Draye et al. (2010) elegantly demonstrated the potential for this modelling approach to investigate a wide range of root–soil–water interactions, with the potential for manipulating root hydraulic architecture as a means of improving water uptake efficiency of crop plants. The influence of soil hydraulic conductivity on soil water gradients across a root system, the effect of root hydraulic properties on water uptake with soil depth, and the effect of 3D root architecture on water uptake from soil were all investigated using this modelling approach.
Explicit three-dimensional simulation of root water uptake using R-SWMS (cyan color denotes removed soil water); roots are colored according to xylem pressure head from red (low) to blue (high); Detail shows particles within the roots; in this case, particles are generated at the root tips and transported via advection towards the root collar as would occur for instance with ABA
Even though many of the detailed 3D root modelling approaches are computationally intensive (e.g., Doussan et al. 2006), a number of models have recently been developed that use approximate approaches for representing the relatively coarse spatial and time scales associated with tree modelling (Kalliokoski et al. 2010; Mulia et al. 2010; Wagner et al. 2010, 2011). For example, Janott et al. (2011) detail an approach in which they combine a 1D tree hydrodynamics model with a 3D architectural representation of above– and below–ground tree elements to characterise the effect of root and shoot architecture on transpiration and water uptake at longer time–scales. This approach represented diurnal fluctuations in transpiration, daily water balance dynamics, daily fluctuations in xylem diameter, and hydraulic lift.
Simulating nutrient uptake dynamics
In the same year in which Clausnitzer and Hopmans (1994) published a root architectural model with water uptake, Nielsen et al. (1994) presented the first nutrient model, named SimRoot. The model calculated phosphorus uptake by root systems by estimating the volume of the depletion zones around the roots using the approach of Nye and Tinker (1977). This method was used to evaluate the P acquisition strategy for different root architectures (Lynch and Beebe 1995), for estimating how gravitropic responses effect competition between roots (Ge et al. 2000; Rubio et al. 2001) and to understand the architectural tradeoffs between root classes (Walk et al. 2006). Estimation of the volume of depletion zones in SimRoot, however, was done as a post–simulation analysis. Dynamic simulation of phosphorus uptake was developed later by implementing the Barber–Cushman model, which is a 1D radial model simulating the development of the depletion zones around the roots, in conjunction with the Michaelis–Menten kinetics of nutrient absorption by the roots (Postma and Lynch 2011b).
where C l is the nutrient concentration in soil solution, C l,0 is the initial nutrient concentration in soil solution, t is the time, r is the radial distance from the root axis, r 0 is the root radius, r 1 is the mean half distance between roots, θ is the volumetric water content, b is the buffer power, D l is the diffusion coefficient in water, f is the impedance factor, F max , K m and C min are Michaelis Menten parameters.
Roose et al. (2001) and Roose and Kirk (2009) provided approximate analytical solutions for the 1D radial nutrient uptake model with a Michaelis-Menten boundary condition at the root surface, for the cases when convective transport both can, and cannot, be regarded as negligibly small compared to diffusion. These solutions facilitate up-scaling to the root system scale, accounting for the dynamic development of the depletion zone (e.g. Roose and Fowler 2004). Baldwin et al. (1973) provided a steady-state approximation to this model, but showed that there is only good agreement between between the approximate solution and the numerical solution of the full problem when uptake is relatively small compared to diffusion.
Simulation of nitrate uptake is made difficult by the mobility of nitrate in the soil. The first nitrate model (Somma et al. 1998) solved the dispersion advection equation in the soil domain and computed uptake using Michaelis–Menten kinetics. Somma et al. (1998) were among the first to incorporate the nutrient status of the entire plant as a factor in the 3D modelling of root growth. In their model, root elongation was affected by temperature, soil strength and nutrient concentration when these properties fell outside an optimum range, and the resultant elongation was scaled according to the amount of biomass allocated to the root system. Over time, these approaches have not changed substantially, although different models have used somewhat different formulations for how the concentration in the finite element grid (used to solve water and solute transport in the soil) relates to the concentration at the root surface. These differences maybe of importance mostly when the finite element grid is relatively coarse.
ROOTMAP uses a particle tracer approach for visualising the bulk movement of nitrate solute in light, sandy soils (Dunbabin et al. 2002a, Table 1). The bulk movement of nitrate in water can be the result of rainfall–induced leaching, or water movement towards roots as a result of water uptake. The particle–tracking approach is based on an approximate solution to the convection–dispersion equation (Rose et al. 1982a, b). Small “packets” of nitrate (pseudo ions) represent the spread of nitrate ions through the profile (Diggle 1990), allowing the bulk movement of nitrate through sandy soils to be visualised.
Partitioning of resources for root growth
Roots not only acquire resources from the soil, but also require resources from the plant for growth and maintenance. The explicit nature of root architectural models allows relatively easy accounting of the construction and maintenance costs. Early development mostly focussed on carbon requirements of, and carbon allocation within, the root system. The first architectural models to include a carbon model were SimRoot (Nielsen et al. 1994) and the Clausnitzer and Hopmans model (Clausnitzer and Hopmans 1994). SimRoot was used to estimate the carbon cost of different root architectures relative to their phosphorus uptake capacity. Although cost–accounting is relatively easily done, the means by which carbon is allocated is less straight forward. Clausnitzer and Hopmans (1994) and Somma et al. (1998) incorporated a source/sink component into their model, using the amount of available carbohydrates within the plant to scale the growth of all roots. Thaler and Pagès (1998) developed a source/sink model that allocated carbohydrates to roots dependent upon their sink strength for assimilates, as determined by the apical diameter of the root. The apical diameter was initially based upon the root type and then had the potential to increase and decrease, along with the root elongation rate, according to the level of carbohydrate supply. The model was able to simulate the relationship between the periodicity of shoot and root growth, and so is a valuable tool for studying the growth coordination between roots and shoots. Bidel et al. (2000) developed a detailed model of carbon transport and partitioning, simulating photo-assimilate flow along phloem vessels to individual meristems, representing carbon transport around the root system and the subsequent growth of individual root tips. This approach was capable of investigating carbon concentration gradients across root systems over time, and the differences in the development of contrasting root architectural arrangements (Bidel et al. 2000).
From responsive root models to whole–plant models
Root models are being continuously developed to better represent roots as integrated, responsive components of a whole–plant and a whole plant–environment system (Pagès 2000). As a result, root architectural models are being increasingly promoted as functional–structural models. Functional–structural models have been described as virtual plants (Xu et al. 2011). They are spatially explicit, defining 3D plant architecture, and the effect of physiological processes and the external environment on plant development at the individual segment scale, and the whole–plant scale (Godin and Sinoquet 2005; Vos et al. 2010). Plants are divided into individual organs and individual organ segments, whose development, structure and functioning are modified by internal signalling between organs, as well as interactions with the external environment; this is an ideal format for investigating interactions and feedback processes in the plant–soil continuum (Vos et al. 2010).
The L–systems modelling structure (Prusinkiewicz and Lindenmayer 1990; Shibusawa 1994; Prusinkiewicz 2004) has been adopted in a number of functional–structural plant models. The L–system modelling approach of Han et al. (2011) showcases the capabilities of the modern functional–structural root architecture models. A functional–structural 3D root architectural model was used to investigate nodule production in soybean. Simulated root systems and nodulation patterns represented well those measured in the glasshouse, providing an opportunity to investigate the internal plant signalling processes that regulate nodule production in legumes. The authors highlight that this approach could be extended to simulate other signalling–based root–regulation activities such as lateral root initiation. Combining this signalling approach with resource supply to the plant and assimilate allocation within the plant holds the potential for investigating a range of as yet poorly understood signalling processes within root systems, and their affect on root system development across a range of environments.
Putting six current models under the spotlight – RootTyp, SimRoot, ROOTMAP, SPACSYS, R–SWMS, and RootBox
A broad range of root architectural modelling approaches exist, each of them varying in their parameterisation and functionality. In Table 1, we have listed the features of six functional-structural models of root architecture, RootTyp, SimRoot, ROOTMAP, SPACSYS, R–SWMS, and RootBox, which have been used for a range of root modelling studies. All six models can represent root architecture with high accuracy (Pagès et al. 2004; Bingham and Wu 2011; Chen et al. 2011). Although from a systems description point of view all six models are similar, they actually have quite different simulation approaches. ROOTMAP has a strong emphasis on root system plasticity and simulates root proliferation, whereas SimRoot emphasises resource acquisition and utilisation. SPACSYS has an emphasis on crop modelling, enabling predictions of crop biomass and yield to be coupled to root–soil dynamics. R–SWMS is a detailed root–soil hydrology model, capable of investigating root hydraulic architecture and the efficiency of water uptake by root systems. RootTyp is a detailed, dynamic root architecture simulator that has been designed to represent a wide range of plant species, and can be coupled with soil models for detailed studies of root-soil interactions. RootBox is an L-systems model of root growth and development implemented in a flexible Matlab structure that is publicly available, and allows individual modules and algorithms to be easily interchanged and tailored to specific simulations. This section highlights the similarities and differences between these six models, and highlights the range of modelling applications for which they were designed.
RootTyp
The RootTyp model (Pagès et al. 2004; Table 1; Fig. 3; email: Loic.Pages@avignon.inra.fr) has been developed to simulate the dynamics of root system architecture (RSA) for a large diversity of plant species (including tree species), as well as developmental stages and soil conditions. For this purpose, several concepts have been generalized and extended regarding root development and root-soil interactions, enabling flexibility. This model was originally focused on root development, and was a synthesis of the extensive knowledge on RSA dynamics. It contains only a simplified soil module (Table 1), but was designed to be merged with other more detailed models that provide a dynamic representation of the soil with relevant attributes (e. g. water transport: Doussan et al. 2006; Draye and Pagès 2007; Pierret et al. 2007; Javaux et al. 2008).
As suggested in its name, the RootTyp modelling approach relies on the definition of “root types”, which formalize the different root categories that can be generally distinguished from direct observations, and have different developmental behaviors of their root tip (e.g. elongation rate and duration, tropism, branching density). Such categories can represent either different branching orders, like in most original models (e. g. Diggle 1988a, b; Pagès and Aries 1988), or they can reproduce groups having a common morphogenetic origin. For example, seminal roots on one hand, and nodal roots on the other hand, can be separated into different types in monocotyledonous species, since they have different origins and growth characteristics. Similarly, “early lateral roots” have been distinguished in young rubber trees (Le Roux and Pagès 1994), as a distinct group of roots that appear around the taproot near the plant collar. When the user defines such root types, they must define the associated sets of parameters to quantify their main developmental traits. Hence, the number of types, which reflects the level of detail, directly impacts the number of parameters in the model. An interesting aspect of this type of model is that each parameter can be estimated independently from the others by simple observations on root systems. Thus, parameters have a straightforward biological significance. In the original presentation of the model (Pagès et al. 2004), the calibration was roughly achieved and illustrated on several species with various structures. Following papers have provided further detail on the calibration procedure for RootTyp using different datasets (Collet et al. 2006; Garré et al. 2012).
RootTyp is a stochastic model, including several stochastic processes. It is now common to include randomness in geometrical characteristics, like initial emergence angles, or root trajectories. In addition, more specifically in this model, numbers and locations of roots of different types, as well as their growth function, are also affected by some hazard. This stochasticity impacts the root system morphology, and may have a functional significance, as discussed by Forde (2009).
Another original feature of the RootTyp model is its ability to combine many developmental processes, which have been shown to notably contribute to the root system dynamics. In addition to root elongation and acropetal branching (commonly included in RSA models), RootTyp also considers different modalities of adventitious root emission, as well as radial growth, branching by reiteration and root decay - abscission. The actual importance of these processes depends on the considered species and on the considered stages. For example, radial growth is a very significant process for dicotyledonous species. It strongly influences the shape of their root system, it involves a significant amount of biomass and it deeply modifies the functional attributes of the root system. Namely, it allows the plant to reinforce its sap conducting capacities and anchorage power. Reiteration formalizes an interesting branching process, described mainly in tree root systems (Vercambre et al. 2003; Collet et al. 2006), in which an apical meristem is substituted at a given stage by several equivalent meristems. This process gives typical forks on the main roots (macrorhizae) of the system, and leads to a particular and significant space colonization. Root decay and abscission have also a considerable impact on aged root systems, and are obviously essential for the soil ecosystem.
Regarding environmental interactions, it is worth noting that two developmental processes (elongation and branching) are influenced by the soil medium during simulation with RootTyp. The soil is an explicit model component represented by at least a set of piled layers containing the information that is considered to be relevant in each particular situation. It includes the major determinants of soil constraints acting on root development, namely soil strength, temperature, oxygen or nutrient availability depending of the particular environmental conditions. Elongation is affected in both intensity and direction (randomly or vertically oriented constraint). Branching density is also potentially influenced by the local soil properties. This allows the model to represent, for instance, the effect of particular conditions prevailing in superficial layers that provide oxygen and significant amounts of nutrients. In the original version (as presented in Pagès et al. 2004), the soil was a fixed component. It was included primarily to represent the influence of major features of the soil morphology, such as contrasted horizons, strong layers, cracks, biopores, etc. It was also designed to be the explicit interface component between root and soil dynamic models. This capacity has been used, afterwards, by models focused on water movement in the soil-plant system (Doussan et al. 2006; Draye and Pagès 2007).
SimRoot
The SimRoot model of Lynch et al. (1997; Table 1; Fig. 2; http://plantscience.psu.edu/research/labs/roots/methods/computer/simroot; email: jpl4@psu.edu) was developed with the capacity for the kinematic description of root axis growth. It reconstructs the root architecture based on empirically derived growth rates, branching frequencies and growth angles. Any of the parameters can optionally be defined as time-dependent arrays with a stochastic component. Reconstruction of the root system architecture allows precise accounting of processes which are simulated at the root segment scale, for example root respiration, exudation and nutrient uptake. These processes are typically associated with root architectural attributes such as root class, diameter and age. Accounting of these processes has the potential to provide valuable data on factors such as the costs and benefits to a whole root system of old versus new, and rapid versus slowly growing roots. SimRoot makes few assumptions about how roots respond to the environment, with growth rates and branching typically simulated as a function of carbon status only. It is thereby assumed that growth responses, for example to soil impedance, are known before hand, and are accounted for in the parameter set.
The SimRoot model has been used to simulate the interaction between root systems and phosphorus acquisition efficiency at a range of spatial scales, focussing on the functional tradeoffs between root architectural arrangement and phosphorus uptake. Ge et al. (2000) and Walk et al. (2006) used SimRoot to investigate the effect of root architecture and P supply on the phosphorus acquisition efficiency of common bean. Modelling confirmed experimental studies, showing that shallower root systems (increased adventitious rooting and lower basal root angle) acquire more phosphorus due to increased root foraging in the relatively phosphorus–rich topsoil layer. Ma et al. (2001) used SimRoot to investigate the effect of root hair architecture on phosphorus acquisition efficiency of Arabidopsis. This approach simulated depletion zone overlap and hence competition between root hairs by varying hair length, density, distance from first hair to root tip, and number of trichoblast files. The modelling approach investigated not only the individual effects of varying individual root hair traits, but demonstrated that combining all four root hair traits had a greater combined effect on phosphorus acquisition than that predicted from the individual effects alone.
While earlier publications used predefined carbon budgets to compare different root architectures of similar cost, recent studies with SimRoot have simulated this using typical crop growth routines linking (nutrient dependent) shoot growth to photosynthesis. SimRoot represents plant shoots as leaf and stem pools that contribute to plant carbon through photosynthesis, and drive phosphorus uptake through phosphorus demand (Postma and Lynch 2011a, b, 2012). The carbon module in SimRoot balances carbon supply from the seed and photosynthesis, with growth of leaves (increase in leaf area) and the extension, thickening, respiration, N–fixation and exudation of roots (Postma and Lynch 2011a, 2012). Using the modified SimRoot model and incorporating a root cortical aerenchyma (RCA) module, Postma and Lynch (2011a) investigated the role that RCA plays in the efficient acquisition of phosphorus by maize and common bean. Using this approach, Postma and Lynch (2011a) were able to demonstrate theoretical support for the hypothesis that RCA are an adaptive trait for the acquisition of phosphorus and nitrogen in low–fertility soils, with the relative benefit of RCA being a function of both root architecture and root physiology (Postma and Lynch 2011a, b). Field experimentation seems to confirm the utility of RCA under drought (Zhu et al. 2010a).
Postma and Lynch (2012) used SimRoot to compare the soil foraging and resource acquisition of maize, bean and squash monocultures, with polycultures of the three. This type of study highlighted the true strengths of 3D functional–structural modelling. Resource acquisition by contrasting root architectures and plant functionality is a spatial problem involving interactions between differing plants, directly competing in a common soil environment. The capacity to investigate multiple planting schemes, multiple root architectures and multiple resource availability (nitrate, phosphorus, potassium), provided an insight into resource foraging by crops that cannot be readily obtained experimentally. This simulation study suggested that competition between root systems for mobile resources (nitrogen) was reduced in polycultures due to contrasting root architectures exploiting complementary parts of the rooting zone. This resulted in greater nitrate uptake and greater biomass production in polycultures (compared to monocultures) growing in low–nitrogen soils.
The inclusion of root morphological and anatomical features such as secondary root thickening, loss of root cortex, root hairs and root cortical aerenchyma, sets SimRoot apart from the other models (Table 1). Model development has focussed upon those traits that are likely to be under strong genetic control, and have the potential to be selected for in breeding programmes for enhanced phosphorus uptake efficiency. Current development of SimRoot continues to focus on the simulation of functional benefits and tradeoffs of root anatomical traits. RootScan (Burton et al. 2012) is software that semi–automates the analysis of root anatomical traits from cross sections, thereby enabling high throughput phenotyping for anatomical traits. Simulation of these traits, as for example done for the formation of root cortical aerenchyma (Postma and Lynch 2011a, b), may aid a better understanding of the functions and tradeoffs that lay behind the observed phenotypic variability. SimRoot has thereby developed into a computational tool for investigating the plant phenome, with special attention to anatomical and architectural phenes. SimRoot’s deficiency in simulating root plasticity (Table 1), as a root trait that is likely to be under genetic control, is an obvious draw back compared to several other models, but incorporation into the model of plasticity responses to both non–directional (e.g. concentrations) and directional (e.g. concentration gradients or soil impedance) soil conditions is currently underway.
SimRoot is parameterized from empirical data of actual plants grown in the field, in soil mesocosms, and in solution culture. Because of the need to consider the spatial and temporal distribution of parameters within and among root classes, many of these parameters were obtained from empirical research conducted in parallel with SimRoot modeling, including root anatomical and architectural phenotypes, growth rates, respiration, tissue nutrient content, and nitrate uptake kinetics (eg. Ge et al. 2000; Liao et al. 2001; Walk et al. 2006; Zhu et al. 2010a; Postma and Lynch 2011b). This empirical work encompasses phenotypic profiling of large numbers of genotypes; in the case of maize, anatomical and architectural data is available for over 7,000 wild and cultivated maize taxa (Burton et al. 2013). This permits a comprehensive range of phenotypes to be modelled. Other parameters were obtained from the published literature, as noted in Postma and Lynch (2011a,b, 2012). Parameters have not been estimated by fitting (calibrating) the model to an empirical dataset, a practice which can generate logical errors. It is important to note in this context that although SimRoot predicts the growth of maize and bean plants up to 42 days after planting fairly accurately (Ge et al. 2000; Walk et al. 2006; Postma and Lynch 2011a, b), SimRoot is intended to be a heuristic rather than a predictive model, which places emphasis on the validity of the underlying processes and parameters, rather than agreement with any specific growth measurements.
ROOTMAP
The ROOTMAP (Diggle 1988a,b; Table 1; Figs. 1 and 5; http://www.plants.uwa.edu.au/research/rootmap; email: art.diggle@agric.wa.gov.au) model of root growth and architecture was modified to include a 3D dynamic, heterogeneous soil environment and to represent root growth responses to that environment (Dunbabin et al. 2002a). Solutes are transported to the root surface by mass flow and diffusion (advection–dispersion equation) using a spatially variable grid. Michaelis–Menten kinetics describe plant capacity to take up ions at the root surface, and ions in solution phase can be transported (leached) after rainfall events by bulk water flow (Table 1; Dunbabin et al. 2002a). Water is lost from the soil surface via evaporation, and is taken up by the plant from each local soil volume as a function of pan evaporation, effective crop cover, local soil properties, and local root surface area (Dunbabin et al. 2009), and the redistribution of water in 3D space is described by Darcy’s law. The phosphate routine models the reactivity of the labile phosphate solid–liquid phases across 3D space at each time–step (Dunbabin et al. 2009).
ROOTMAP uses a black-box biomass provider to represent the shoot system, or uses an optional Woodruff–Hammer wheat module (Hammer et al. 1987) for estimating the biomass accumulation and grain yield of wheat (Dunbabin et al. 2009). This module can be substituted for other crop biomass simulators for feedback between the 3D root system and soil environment and crop development. All 6 models differ in how the shoot system is represented (see Table 1), ranging from not considered at all (RootBox) to black-box biomass providers (RootTyp, ROOTMAP), to shoot structural carbon pools (SimRoot) and crop shoot models (SPACSYS). One of the defining features of the SimRoot and SPACSYS models is the capacity to integrate root systems into a whole–plant model, where the role of carbon accumulation, shoot development and yield (in the case of SPACSYS) can be linked to soil water and nutrient dynamics and root architecture and growth. Simulations undertaken with ROOTMAP to date (with the exception of wheat modelling, Dunbabin et al. 2009), have assumed carbon supply to be non–limiting, and have focussed solely on root systems and root–soil dynamic interactions and plastic root growth responses. The modular structure of the ROOTMAP model lends itself to being used as a module in crop simulators, or incorporating an existing biomass simulator into it, enabling a greater representation of root–shoot interactions.
ROOTMAP can represent whole–root system responses to resource supply, as well as localised nutrient uptake and root proliferation responses to local nutrient patches (Fig. 5; Dunbabin et al. 2002a). The model simulates the feedback between plant demand for below–ground resources (nitrogen, phosphorus and water), and the capacity of individual root segments to supply those resources. This feedback mechanism drives the allocation of resources to root tips for growth, and to all root segments for branching, maintenance and nutrient uptake. This emphasis on root growth and nutrient uptake plasticity is one area in which the ROOTMAP model specialises, and differs from the other models. While nutrient supply has an effect on overall nutrient uptake and root growth for the other models, it does not influence root growth at the individual root tip level (Table 1).
ROOTMAP has been used to investigate root–soil interactions over a range of spatial and time scales (Dunbabin et al. 2002b, 2003a, b, 2004, 2006; Chen et al. 2008). Using contrasting wheat and lupin root architectures, Dunbabin et al. (2006) investigated the effect of phospholipid surfactant exudation on water and nutrient acquisition from soil, simulating interactions at both the rhizosphere scale (5 × 5 mm2 soil area, 10 mm root section, 12 h), and the whole plant scale (over 41 days). Comparison with a computationally expensive 2D Finite Element Method rhizosphere model confirmed the performance of the ROOTMAP model at the rhizosphere scale. Whole–plant simulations demonstrated the importance of scaling–up rhizosphere processes for investigating their impact on whole–plant function. At the whole–plant scale there was an interaction between phospholipid surfactant exudation, root architecture, soil nutrient status, and water and nutrient uptake (Dunbabin et al. 2006). ROOTMAP, SimRoot and RootBox have all been used to simulate root exudation and the role that root exudates play in nutrient acquisition (Table 1).
In a theoretical study of root form and function, Dunbabin et al. (2004) modelled the interaction between root architecture, the 3D supply of nitrate in soil (uniform, non–uniform, dynamic and static) and root plasticity responses to nitrate supply (local root proliferation and locally elevated nutrient uptake kinetics). This study highlighted that the efficiency of rooting strategy for nitrate uptake from soil is a strong function of how transitory and variable the supply of nitrate is. In a detailed sensitivity analysis study, Dunbabin (2007) used ROOTMAP to simulate crop and weed root systems growing in direct competition, investigating the impact of root traits upon the competitive success of crop plants. 30 parameters were included in the study, describing root architecture, the soil and seasonal environment, and crop management, providing a detailed study of individual parameters and their interactions across the parameter space (Dunbabin 2007). This type of study is not possible experimentally. This approach suggested a distinct separation in the root traits that maximise resource acquisition in weed–free monocultures compared to weedy crops, highlighting the potential for tailoring root traits to particular cropping conditions. The results need to be tested against field trials.
The ROOTMAP model has been parameterised with root architectural data from nutrient solution, pot and field experiments, along with data sets from literature (Dunbabin et al. 2001a,b, 2002a,b, 2009; Dunbabin 2007; Chen et al. 2011). Chen et al. (2011) detail the parameterisation of the ROOTMAP model using root architectural data from a semi-hydroponic phenotyping system. The simulations carried out to date using the model have focussed on representing root growth responses across growth media (nutrient solution, pot and field) for investigating interactions between crop root systems and their environment. To this end, a number of the simulation studies have been theoretical, focussing on understanding root:soil interactions, rather than precise prediction under specific conditions (Dunbabin et al. 2003a, 2004, 2006; Dunbabin 2007).
The ROOTMAP model currently distinguishes between root orders, but root age does not affect root function, with all sections of a root equally contributing to water and nutrient uptake regardless of distance from the root tip or root age. Future development work will focus on a more realistic representation of root function as roots age, as well as a more detailed representation of root exudation into the rhizosphere for investigating the interactions between rhizosphere processes and root system architecture in field soils. Future development work will also focus on the model software, with the aim of developing versions of the model that (with the aid of a user–interface) can be more readily used by other researchers.
SPACSYS
Wu et al. (2007) and Bingham and Wu (2011) developed SPACSYS (Table 1; Fig. 4; email: Ian.Bingham@sruc.ac.uk), a model that combines a 3D root architectural model with 1D and 2D processes representing water dynamics and C and N cycling between plants (leaf, stem, seed and root components), soil and microbes (microbial biomass pool). The model tracks the developmental stages of crop growth from sowing to maturity, and predicts grain yield or regrowth for perennial grasses. The root model uses the same root growth responses as those described in Clausnitzer and Hopmans (1994), with root extension being a function of soil temperature, strength and soil water solute concentration. At a whole–root system level, assimilate supply from the shoots determines root system growth and volume. Root death and decay and the subsequent contribution to nutrient cycling are included in the model (Table 1). Main root axes are considered to be indeterminate and will continue to extend according to the limitations set by soil conditions, plant phenology and the supply of carbon. Lateral roots, on the other hand, are determinate and cease extension after a specified maximum length is reached, the value of which is dependent on the order of lateral root. The mature branch then senesces and decays and dry matter is either added to the soil litter pool according to a first order relationship with the root biomass, or it is remobilised and transferred to living roots. Current understanding of the physiological and molecular control of root senescence is poor and data to parameterise this aspect of the model are limited. However, sensitivity analysis is being used to explore the relative impact of variation in maximum root axis length, death and decay on RSA and resource capture. In this regard, recent experiments have shown that although soil compaction can greatly reduce root length production, it may have little effect on the dynamics of root death and decay, which simplifies the modelling exercise considerably (Bingham et al. 2009). One important distinction between SPACSYS and the other models (Table 1), is that SPACSYS is a field–scale model running at a daily time–step, whereas the other models are principally plant–scale, or sub–plant scale models, running at finer temporal resolutions. The SPACSYS model, therefore, provides a link between detailed plant modelling and the traditional coarse–scale crop modelling.
SPACSYS was developed primarily to investigate the interactions between crop root systems and nutrient use efficiency under different soil, fertilizer and crop management regimes. A 3D modelling approach was adopted so that the potential effects of altering specific root architectural traits and trait combinations on root system growth, distribution and resource capture could be evaluated under contrasting soil conditions. SPACSYS deals with spatial variability in soil properties in the vertical direction as this is generally most relevant to questions about optimising root exploration of the soil profile and maximising the efficiency of water and nutrient uptake. Further, information on variation in soil properties with depth, required as model inputs, tends to be more readily available than that on heterogeneity across fields. The SPACSYS model is a detailed process-based model containing more than 400 parameters. The model is supplied with a reference, or default value for each of these parameters. These reference values have been primarily derived from literature, along (Wu et al. 2007; Wu and Bingham 2009) with experimental data collected by the authors of the model from their own laboratory or field experiments (Wu et al. 2007). Wu and Shepherd (2011) illustrated the procedures for parameterisation and validation of the SPACSYS model.
SPACSYS has been shown to produce realistic root architectural arrangements in microcosms (Fig. 7., Wu et al. 2007) and good predictions of root system size and distribution in field soils (Bingham and Wu 2011). Initial research focussed on the interplay between crop root architectural traits and nitrate leaching (Wu et al. 2005, 2008; Wu and Bingham 2009). The capacity to simulate nitrogen cycling interacting with developing root systems, provides insights into the mismatch between nitrate supply and demand that can occur over a season. This enables an assessment of the spatial and temporal root development needed to maximise fertiliser recovery and minimise nitrate leaching under crops, providing the potential for ‘designing’ root systems and cropping systems for improving nitrogen use efficiency in field soils. The description of plant phenological development, root and shoot biomass production, and grain yield, provides an opportunity to investigate direct relationships between root architectural traits and grain production (the only model of the 6 capable of doing this, Table 1). This presents an opportunity to study the interactions between root architectural traits, resource acquisition efficiency and grain yield, with the aim of providing advice to breeders, policy makers and growers on the potential economic and environmental benefits of developing root phenotypes for specific cropping environments (Bingham and Wu 2011).
R–SWMS
R–SWMS (Table 1; Fig. 8; email: mathieu.javaux@uclouvain.be; http://www.fz-juelich.de/ibg/ibg-3/EN/Research/Research%20Topics/Flow%20and%20Transport%20in%20Soil-Plant%20Systems/R-SWMS/_node.html) is a combination of the root simulation model in soil developed by (Somma et al. 1997, 1998) and based on the SWMS-3D flow and transport model (Simunek et al. 1995), and the model of Doussan et al. (1998) which solves water flow from the soil into the roots and through the roots up into the shoot. In essence, this is a similar model to those developed by Doussan et al. (2006) and Schneider et al. (2010).
In its latest version, R-SWMS contains different modules for solving specific processes: root water flow, soil water flow, solute transport in soils, solute transport in roots, root growth and plant growth. The root and plant growth models are similar to those used in SPACYS (Table 1), and use the same root growth responses as those described in Clausnitzer and Hopmans (1994). Water and solute/nutrient extraction from the soil are modelled through sink terms in the Richards and the convection–dispersion equations.
R–SWMS is possibly the root architectural model with the most advanced hydrology, capable of simulating soil and root water redistribution (eg. hydraulic lift) and compensatory water uptake (Couvreur et al. 2012). It can simulate a variety of drought or locally dry soil scenarios. Simulation studies have focused on sensitivity of water uptake to soil hydrological parameters (Javaux et al. 2008). The initial development goals for this model were not the simulation of the root architecture, but simulation of the water and associated solute transport through the soil–plant continuum. Several publications with this model used static root architectures, generated with RootTyp (Draye et al. 2010; De Willigen et al. 2012; Schröder et al. 2012) or from NMR images (Pohlmeier et al. 2010; Stingaciu et al. 2013). R-SWMS is parameterised with root hydraulic parameters from the literature, and soil hydraulic parameters are typically derived from soil databases or direct measurement.
Simulation of water and nutrient uptake is challenging especially if we consider the soil conditions in the rhizosphere around the roots, which are often quite different from those in the bulk soil. The problem partly arises from the different scales which easily vary by two to three orders of magnitude or, in three dimensions, 6–9 orders (Postma et al. 2008). Schröder et al. (2008) investigated this problem by assessing the impact of soil spatial resolution on the 3D root water uptake modelled by R-SWMS. They concluded that the local drying of the rhizosphere could not be neglected in order to properly simulate pressure head at the root soil interface and the onset of stress. They showed that a spatial resolution lower than 1 cm could ensure an error level lower than 5 %. Yet high spatial resolution demands high computational power. Several solutions have therefore been tried with R–SWMS to overcome this problem: the development of analytical solutions below the voxel scale and the grid refinement around roots. Schröder et al. (2009a) investigated a different methodology to implement a microscopic soil–root hydraulic conductivity drop function in R-SWMS. It allowed them to explicitly consider the pressure head drop around multiple roots at a scale lower than finite element grid resolution. In a second step, they implemented the possibility of generating a non-uniform grid with a finer resolution around the roots in R-SWMS (Schröder et al. 2009b). They demonstrated that in their scenario, a fine finite–element grid gave a more accurate solution than a coarse grid, and that the grid refinement can be localized around the roots without loss in accuracy. A priori grid refinement can thereby significantly reduce the computational time of a simulation. However, most of the gain of a locally refined grid could be lost when the grid could not be refined a priori, but had to be adapted to the (changing) architecture of the root system during simulation.
A particle tracker for solute transport in soils was also implemented in R-SWMS, allowing the analysis of steep solute concentration gradients in soils. The impact of solute transport type (active/passive/exclusion) on the apparent solute dispersivity length was investigated (Schröder et al. 2012).
Novel developments in R-SWMS consist of a particle tracker within the root system allowing the simulation for plant hormone (e.g. ABA) or nutrient transport within the xylem, change of the radial conductivity with environmental conditions and the implementation of a stomatal conductance model. Table 1 summarizes the current features of R-SWMS.
RootBox
Branching structures such as root systems possess a high degree of (topological) self-similarity (Prusinkiewicz 2004) that can well be modelled by a recursive or iterated approach. Leitner et al. (2010a) chose an L-systems approach to model root system growth. The 3D root growth model RootBox (Leitner et al. 2010a; Table 1; Figs. 6 and 7), includes growth of individual roots according to a growth function (e.g. Pagès et al. 1989), branching at predefined branching angles, and root death. The model can produce a variety of different root systems that compare well to observed images of root systems (Leitner et al. 2010a). Root system properties such as root length densities can be computed from the model output. The software is presented in the form of a publicly available Matlab-code (http://www.boku.ac.at/marhizo/simulations.html). Internal functions can readily be altered to specific requirements. This facilitates the coupling with different soil models and model adaptation for specific experimental designs. An advantage of RootBox over other models is that it does not provide a software package that simulates a fixed model of plant and soil interactions. Instead, it is implemented in Matlab in a way that keeps it open for any changes to the model structure. The root growth model can be coupled with any given soil model. For example, there is no fixed implementation for the solution of the convection dispersion-equation. Suitable numerical schemes are readily available in Matlab (e.g., built-in, from Comsol Multiphysics or C++/Fotran libraries).
Examples of coupling the root growth model with soil models are presented in Schnepf et al. (2011, 2012). Schnepf et al. (2011) simulated root growth as affected by chemotropism in soil of initially homogeneous and non-homogeneous phosphate distribution, in a bounded pot environment. See a sample simulation for chemotropism effects on root system growth in Fig. 6. Schnepf et al. (2012) simulated root system phosphate uptake from a rhizotron as affected by root exudation. In both cases, the root system model of Leitner et al. (2010b) was coupled with a different soil model. The soil model in Schnepf et al. (2011) contained a 3D form of the diffusion–reaction equation, while the soil model in Schnepf et al. (2012) modelled phosphate-citrate interactions with two coupled diffusion–reaction equations. Leitner et al. (2010c) demonstrated the use of the RootBox model to create a 3D tetrahedral mesh from the root system geometry (Fig. 7a). This enabled the explicit 3D simulation of water and nutrient transport in the soil with static root surfaces as boundaries (see Fig. 7).
This modelling approach facilitates the study of soil-root interactions at different levels of detail, corresponding to the underlying scientific questions. RootBox was first published in 2010 with the specific aim to study the effect of different root and rhizosphere traits at the root system scale. This capability was demonstrated by Schnepf et al. (2012), simulating the effect of the release of mobilising agents on phosphate mobility and uptake. This approach can be extended to model other root and rhizosphere traits, including root hairs and mycorrhizas.
Several features of the RootBox model are provided in Table 1. Contrary to some of the other models in Table 1, it was not developed for specific plants. Like most of the other models, except for SimRoot, RootBox considers the root system to consist of individual root segments of a given diameter without including secondary growth. Each root type can be defined with an individual set of parameters that does not necessarily coincide with the root order (similar to RootTyp). Several aspects of the other models, such as the affect of soil temperature or impedance on root architecture or shoot-root interactions, are not predefined in the RootBox model. However, the open implementation of the model in Matlab allows these affects to be easily included if required. Any confined or free root growth is possible. Confined containers are defined by signed distance functions that define the signed distances of points to the container boundaries. Root functions in the RootBox model are not predefined, but depend on the underlying soil model. Examples include water uptake, nutrient uptake and root exudation. Likewise, the soil model is not predefined, but RootBox can easily be coupled with different soil models, depending on the underlying scientific question. In the exudation case study, for example, the underlying soil model was not a simple convection dispersion equation but a coupled system of convection dispersion equations, one with a term for the decomposition of organic exudates (Schnepf et al. 2012). This also means that the numerical solution method can be freely chosen (Table 1). RootBox has been presented as a modelling framework and relies mainly on literature sources for parameterisation. In Schnepf et al. (2012), model parameterisation was based on 2D images from Lore Kutschera’s Root Atlas (Kutschera 1960), using manual measurements. Currently, RootBox is being used in an algorithm for semi-automated root tracking for the realistic recovery of root system parameters from 2D images (Leitner and Schnepf 2012).
The future development of functional-structural root models
Although current models include several important aspects of the plant–soil complex, many features are underdeveloped due to either a lack of an adequate conceptual model for representing the physiological processes, or a lack of quantitative data for parameterisation. Soil biology, rhizosphere chemistry, soil texture and mycorrhizas have influence both on root growth and on soil water and nutrient processes (e.g., Shu et al. 2007; Richardson et al. 2009; Feddermann et al. 2010; Rose et al. 2010; Marschner et al. 2011), but are not, or are rudimentarily included in functional-structural root models.
Plant physiology is mostly reduced to a set of response curves in these models. While from a simulation perspective, the response curves are testable simplifications of otherwise complex processes, these simulations do not yield major increases in understanding the underlying complexity; hence, it might be useful to break these curves down into smaller steps, moving towards regulatory networks that better mimic plant physiology and maybe even include gene–based approaches (Postma et al. 2008).
Root anatomy is largely overlooked by the root modelling community. Postma and Lynch (2011a,b,) simulated the function of root cortical aerenchyma formation on low fertility soil, but other anatomical traits may also influence nutrient and water uptake and the metabolic cost of roots.
One of the big challenges facing root architectural models is how to increase adoption and application in plant research (Tardieu 2010). While many of the models under development demonstrate substantial promise, there are still relatively few modelling studies that have led directly to the selection of improved plant genomes, or have explicitly predicted plant behaviour in the field. Further development of root architectural models down the path of functional–structural plant modelling will provide insights into the internal signalling and control mechanisms within plants. This will provide an opportunity to better represent and predict phenotypic plasticity in root systems. The challenge of predicting the relationship between genetics and environment is complex (Tardieu 2010), but promises the reward of selecting root systems that are more functionally efficient in target environments (Lynch and Brown 2012), enabling root architectural models to play a more integrated role in plant breeding programmes.
One path to the greater adoption of root architectural models is the development of software more readily used and adapted by researchers to a range of applications. Advances in computing power, modelling platforms and visualisation software are making the task of plant modelling easier and more accessible. Many studies can be performed with existing software, and the development of open platforms for the easy development of functional–structural plant models, such as CrossTalk (Draye and Pagès 2007), OpenALea (Pradal et al. 2008), PlantGL (Pradal et al. 2009) and GroImp (Kurth and Lanwert 2011), will enable many more researchers to contribute to root and plant architectural modelling. Pagès et al. (2004) have started down this path by developing the generic 3D root architectural model RootTyp. As a generic platform coupled to an external soil model, RootTyp can be used for investigating a range of root–soil interactions (Collet et al. 2006; Javaux et al. 2008; Draye et al. 2010; Couvreur et al. 2012). The generic MatLab format in which the RootBox model was developed (Leitner et al. 2010a) also provides a flexible structure in which soil models can be readily interchanged.
Conclusions and outlook
In the short history of root architectural models, they have evolved into functional–structural models capable of representing virtual plants by incorporating carbon fixation in leaves and partitioning into roots, combined with nutrient demand driving root architecture and root functional responses to variability in soil environment. As such, functional-structural root models have become valuable tools, contributing to understanding of the plant phenome, elucidation of signalling sequences governing development of root structures, and selection of optimal root architectures for specific environments. They play an important role in targeting and focussing experimental studies that investigate the complexity of soil–root interactions in heterogeneous environments. It is expected that future development will result in greater usefulness of root architectural models in deciphering the genotype-phenotype relationships in interaction with variable environments.
It is also anticipated that functional root models will play an increasingly important role in rhizosphere research. Root and rhizosphere traits are of major importance for agricultural management and breeding. So-called rhizo-technologies take advantage of these traits by direct or indirect manipulation of one or more components of the system. Examples are the selection of crop cultivars with regard to their nutrient-efficiency and/or root branching pattern, soil microbial inoculation or improvement of agricultural management. Root architectural models will help to design such rhizo-technologies and to optimise soil and crop management. The following are examples of possible model applications as an outlook.
Mycorrhizal inoculation of agricultural crops
Arbuscular mycorrhizal fungi are potentially an important tool in sustainable agriculture (Douds et al. 2008). Inoculation could help to overcome stress situations and improve yield of crops and medical plants (Chandra et al. 2010; Ozdemir et al. 2010). Growth models for arbuscular mycorrhizal fungi (e.g. Schnepf et al. 2008) could be coupled with root architectural models. This could help to characterise cultivars with sufficient capacity to sustain root symbiosis with arbuscular mycorrhizal fungi. It could further be relevant for the development and management of mycorrhizal inocula by testing different hypotheses of fungal infection and hyphal growth on the overall mycorrhrisation of whole root systems as well as on the effect of mycorrhization on plant nutrition. Further model development could include a multispecies symbiotic complex (Tajini et al. 2009) as some crops have the potential to establish symbiosis with both rhizobia and arbuscular mycorrhizal fungi.
Quantifying carbon costs
There is considerable knowledge about different root and rhizosphere traits that benefit plant phosphate acquisition (Jansa et al. 2005; Hinsinger et al. 2011; Thonar et al. 2011). In contrast, little is known about the related carbon costs (Lynch and Ho 2005; Lendenmann et al. 2011). Carbon trading at the soil-root interface including mycorrhiza, root exudation and root hairs was reviewed by Jones et al. (2009). Root architectural models could be extended to perform a precise carbon cost assessment of different root and rhizosphere traits.
Yield enhancement through soil additives
Practical implications of root plasticity for yield enhancement methods using soil additives are discussed by Akay and Burke (2012). These soil additives contain more water and nutrients than the bulk soil. Thus, their efficiency depends on hydrotropism and chemotropism mechanisms of root growth. Coupling a root architectural model with a soil model and allowing for feedback loops between root growth and soil nutrient concentration and/or water content will assist in the development of such techniques. They will benefit from careful up-scaling methods from single root scale to root system scale.
References
Akay G, Burke D (2012) Agro-process intensification through synthetic rhizosphere media for nitrogen fixation and yield enhancement in plants. Am J Agric Biol Sci 7:150–172
Ao J, Fu J, Tian J, Yan X, Liao H (2010) Genetic variability for root morph-architecture traits and root growth dynamics as related to phosphorus efficiency in soybean. Funct Plant Biol 37:304–312
Baldwin JP, Nye PH, Tinker PB (1973) Uptake of solutes by multiple root systems from soil. III a model for calculating the solute uptake by a randomly dispersed root system developing in a finite volume of soil. Plant Soil 38:621–635
Bates TR, Lynch JP (2000) The efficiency of Arabidopsis thaliana (Brassicaceae) root hairs in phosphorus acquisition. Am J Bot 87:964–970
Bear J, Cheng A (2010) Modeling groundwater flow and contaminant transport. Springer
Bechtold M, Vanderborght J, Ippisch O, Vereecken H (2011) Efficient random walk particle tracking algorithm for advective-dispersive transport in media with discontinuous dispersion coefficients and water contents. Water Res 47:W10526
Bengough AG (2012) Root elongation is restricted by axial but not radial pressures: so what happens in field soil? Plant Soil 360:15–18
Bengough AG, Mackenzie CJ, Diggle AJ (1992) Relations between root length densities and root intersections with horizontal and vertical planes using root growth modelling in 3-dimensions. Plant Soil 145:245–252
Bengough AG, McKenzie BM, Hallett PD, Valentine TA (2011) Root elongation, water stress, and mechanical impedance: a review of limiting stresses and beneficial root tip traits. J Exp Bot 62:59–68
Bernier J, Serraj R, Kumar A, Venuprasad R, Impa S, Gowda RPV, Oane R, Spaner D, Atlin G (2009) The large–effect drought–resistance QTL qtl12.1 increases water uptake in upland rice. Field Crop Res 110:139–146
Bidel LPR, Pagès L, Riviere LM, Pelloux G, Lorendeau JY (2000) MassFlowDyn I: a carbon transport and partitioning model for root system architecture. Ann Bot 85:869–886
Bingham IJ, Wu L (2011) Simulation of wheat growth using the 3D root architecture model SPACSYS: validation and sensitivity analysis. Eur J Agron 34:181–189
Bingham IJ, Rees RM, Bengough AG (2009) Influence of soil compaction on the dynamics of root growth and mortality in spring barley. International symposium “root research and applications” RootRAP, 2–4 Sept 2009. Boku, Vienna, pp 148–4
Bohn M, Novais J, Fonseca R, Tuberosa R, Grift TE (2006) Genetic evaluation of root complexity in maize. Acta Agron Hung 54:291–303
Brady DJ, Wenzel CL, Fillery IRP, Gregory PJ (1995) Root growth and nitrate uptake by wheat (Triticum aestivum L.) following wetting of dry surface soil. J Exp Bot 46:557–564
Brück HH, Becker HC, Sattelmacher B (1992) Phosphate efficiencies of two maize inbred lines. In: Kutschera L, Hüble E, Lichtenegger E, Persson H, Sobotik M (eds) Root ecology and its practical applications. 3rd ISRR Symposium, Vienna, Austria, pp 193–196
Burton A, Williams M, Lynch J, Brown K (2012) RootScan: software for high-throughput analysis of root anatomical traits. Plant Soil 357:189–203
Burton AL, Brown KM, Lynch JP (2013) Phenotypic diversity of root anatomical and architectural traits in Zea species. Crop Sci. doi:10.2135/cropsci2012.07.0440
Campbell BD, Grime JP, Mackey JML (1991) A trade–off between scale and precision in resource foraging. Oecol 87:532–538
Chandra K, Kumar N, Chand G (2010) Studies on mycorrhizal inoculation on dry matter yield and root colonization of some medicinal plants grown in stress and forest soils. J Environ Biol 31:975–979
Chen W, Dunbabin V, Bell R, Brennan R, Bowden B (2008) Simulating and understanding root growth using ROOTMAP to guide phosphorus fertiliser placement in wide row lupin cropping systems. In: Lupins for health and wealth - Proceedings of the 12th International Lupin Conference, 14–18 Sept, 2008, Fremantle, WA. International Lupin Association, Canterbury, pp 368–372
Chen YL, Dunbabin VM, Postma JA, Diggle AJ, Palta JA, Lynch JP, Siddique KHM, Rengel Z (2011) Phenotypic variability and modelling of root structure of wild Lupinus angustifolius genotypes. Plant Soil 348:345–364
Clark RT, MacCurdy RB, Jung JK, Shaff JE, McCouch SR, Aneshansley DJ, Kochian LV (2011) Three-dimensional root phenotyping with a novel imaging and software platform. Plant Physiol 156:455–465
Clausnitzer V, Hopmans JW (1993) An algorithm for three–dimensional simultaneous modelling of root growth and transient water flow. Land, air and water resources paper no. 100022. Department of Land, Air and Water Resources, University of California, Davis, pp 1–108
Clausnitzer V, Hopmans JW (1994) Simultaneous modeling of transient three–dimensional root growth and soil water flow. Plant Soil 164:299–314
Collet C, Löf M, Pagès L (2006) Root system development of oak seedlings analysed using an architectural model. Effects of competition with grass. Plant Soil 279:367–383
Couvreur V, Vanderborght J, Javaux M (2012) A simple three–dimensional macroscopic root water uptake model based on the hydraulic architecture approach. Hydrol Earth Syst Sci 16:2957–2971
de Dorlodot S, Forster B, Pages L, Price A, Tuberosa R, Draye X (2007) Root system architecture: opportunities and constraints for genetic improvement of crops. Trends Plant Sci 12:474–481
de Willigen P, van Dam JC, Javaux M, Heinen M (2012) Root water uptake as simulated by three soil water flow models. Vadose Zone J 11(3). doi:10.2136/vzj2012.0018
Deans JD, Ford ED (1983) Modelling root structure and stability. Plant Soil 71:189–195
Diggle AJ (1988a) ROOTMAP – a model in three-dimensional coordinates of the growth and structure of fibrous root systems. Plant Soil 105:169–178
Diggle AJ (1988b) ROOTMAP: a root growth model. Math Comput Simul 30:175–180
Diggle AJ (1988c) ROOTMAP2.1 – a root growth simulation program. Department of Agriculture Western Australia, Western Australia
Diggle AJ (1990) Interaction between mineral nitrogen and growth of wheat roots in a leaching environment. PhD, The University of Western Australia, Perth
Diggle AJ (1996) Developments in architectural models of root systems and the potential for their application to intercropping. In: Ito O, Johansen C, Adu–Gyamfi JJ, Katayama K, Kumar Rao JVDK, Rego TJ (eds) Dynamics of roots and nitrogen in cropping systems of the semi–arid tropics. Japan International Research Center for Agricultural Sciences, Japan, pp 559–571
Dinkelaker B, Hengeler C, Marschner H (1995) Distribution and function of proteoid roots and other root clusters. Bot Acta 108:183–200
Douds D, Nagahashi G, Reider C, Hepperly P (2008) Choosing a mixture ratio for the on-farm production of AM fungus inoculum in mixtures of compost and vermiculite. Compost Sci Utiliz 16:52–60
Doussan C, Pagès L, Vercambre G (1998) Modelling of the hydraulic architecture of root systems: an integrated approach to water absorption – model description. Ann Bot 81:213–223
Doussan C, Pagès L, Pierret A (2003) Soil exploration and resource acquisition by plant roots: an architectural and modelling point of view. Agronomie 23:419–431
Doussan C, Pierret A, Garrigues E, Pagès L (2006) Water uptake by plant roots: II – modelling of water transfer in the soil root–system with explicit account of flow within the root system – comparison with experiments. Plant Soil 283:99–117
Draye X, Pagès L (2007) CrossTalk: a simulation platform for the linking of existing soil, plant and atmosphere models. In: Proceeding of international symposium on plant growth modeling and applications. IEEE Computer Society, Los Alamitos, pp 93–100
Draye X, Kim Y, Lobet G, Javaux M (2010) Model–assisted integration of physiological and environmental constraints affecting the dynamic and spatial patterns of root water uptake from soils. J Exp Bot 61:2145–2155
Drew MC, Saker LR (1975) Nutrient supply and the growth of the seminal root system in barley. II. Localised compensatory increases in lateral root growth and rates of nitrate uptake when nitrate supply is restricted to only one part of the root system. J Exp Bot 26:79–90
Drew MC, Saker LR, Ashley TW (1973) Nutrient supply and the growth of the seminal root system in barley. I. The effect of nitrate concentration on the growth of axes and laterals. J Exp Bot 24:1189–1202
Dunbabin V (2007) Simulating the role of rooting traits in crop–weed competition. Field Crop Res 104:44–51
Dunbabin V, Rengel Z, Diggle A (2001a) The root growth response to heterogeneous nitrate supply differs for Lupinus angustifolius and Lupinus pilosus. Australian Journal of Agricultural Research 52:495–503
Dunbabin V, Rengel Z, Diggle A (2001b) Lupinus angustifolius has a plastic uptake response to heterogeneously supplied nitrate while Lupinus pilosus does not. Australian Journal of Agricultural Research 52:505–512
Dunbabin V, Diggle A, Rengel Z, van Hugten R (2002a) Modelling the interactions between water and nutrient uptake and root growth. Plant Soil 239:19–38
Dunbabin V, Diggle A, Rengel Z (2002b) Simulation of field data by a three–dimensional model of interactive root growth. Plant Soil 239:39–54
Dunbabin V, Diggle A, Rengel Z (2003a) Is there an optimal root architecture for nitrate capture in leaching environments? Plant Cell Environ 26:835–844
Dunbabin V, Rengel Z, Diggle A (2003b) Root architecture and nutrient capture – the complex riddle of what constitutes optimality of root form and function. In: Lynch JM, Schepers JS, Unver I (eds) Innovative soil-plant systems for sustainable agricultural practices. Organisation for Economic Co–operation and Development (OECD), Paris, pp 2–16
Dunbabin V, Rengel Z, Diggle A (2004) Simulating form and function of root systems: efficiency of nitrate uptake is dependent on root system architecture and the spatial and temporal variability of nitrate supply. Funct Ecol 18:204–211
Dunbabin VM, McDermott S, Bengough AG (2006) Upscaling from rhizosphere to whole root system: modelling the effects of phospholipid surfactants on water and nutrient uptake. Plant Soil 283:57–72
Dunbabin VM, Armstrong RD, Officer SJ, Norton RM (2009) Identifying fertiliser management strategies to maximize nitrogen and phosphorus acquisition by wheat in two contrasting soils from Victoria, Australia. Aust J Soil Res 47:74–90
Dunbabin VM, Airey M, Diggle AJ, Renton M, Rengel Z, Armstrong R, Chen Y, Siddique KHM (2011) Simulating the interaction between plant root, soil water and nutrient flows, and barriers and objects in soil using ROOTMAP. In: Anderssen RS, Chan F, Marinova D (eds) 19th International Congress on Modelling and Simulation. Modelling and Simulation Society of Australia and New Zealand, Dec 2011, pp 975–981. http://mssanz.org.au/modsim2011
Dupuy L, Gregory PJ, Bengough AG (2010) Root growth models: towards a new generation of continuous approaches. J Exp Bot 61(8):2131–2143
Eissenstat DM, Yanai RD (1997) The ecology of root lifespan. Adv Ecol Res 27:1–60
Feddermann N, Finlay R, Boller T, Elfstrand M (2010) Functional diversity in arbuscular mycorrhiza– the role of gene expression, phosphorous nutrition and symbiotic efficiency. Fungal Ecol 3:1–8
Fitter AH (1991) The ecological significance of root system architecture: an economic approach. In: Plant root growth – an ecological perspective. Blackwell Scientific Publications, Oxford, pp 229–243
Fitter AH (2002) Characteristics and functions of root systems. In: Waisel Y, Eshel A, Kafkafi U (eds) Plant roots, the hidden half. Marcel Dekker Inc, New York, pp 15–32
Fitter AH, Stickland TR, Harvey ML, Wilson GW (1991) Architectural analysis of plant root systems. 1. Architectural correlates of exploitation efficiency. New Phytol 118:375–382
Flavel RJ, Guppy CN, Tighe M, Watt M, McNeill A, Young IM (2012) Non–destructive quantification of cereal roots in soil using high-resolution X-ray tomography. J Exp Bot 63:2503–2511
Forde BG (2009) Is it good noise? The role of developmental instability in the shaping of a root system. J Exp Bot 60:3989–4002
Gahoonia TS, Nielsen NE (2004) Root traits as tools for creating phosphorus efficient crop varieties. Plant Soil 260:47–57
Garré S, Pagès L, Laloy E, Javaux M, Vanderborght J, Vereecken H (2012) Parameterizing the root system development of spring barley using minirhizotron data. Vadose Zone J 11(4). doi: 10.2136/vzj2011.0179
Ge ZY, Rubio G, Lynch JP (2000) The importance of root gravitropism for inter-root competition and phosphorus acquisition efficiency: results from a geometric simulation model. Plant Soil 218:159–171
Gilroy S, Jones DL (2000) Through form to function: root hair development and nutrient uptake. Trends Plant Sci 5:56–60
Godin C, Sinoquet H (2005) Functional–structural plant modelling. New Phytol 166:705–708
Gregory PJ, Bengough AG, Grinev D, Schmidt S, Thomas WTB, Wojciechowski T, Young I (2009) Root phenomics of crops: opportunities and challenges. Funct Plant Biol 36:922–929
Grime JP, Campbell BD, Mackey JML, Crick JC (1991) Root plasticity, nitrogen capture and competitive ability. In: Atkinson D (ed) Plant root growth – an ecological perspective. Blackwell Scientific Publications, Oxford, pp 381–397
Hammer GL, Woodruff DR, Robinson JB (1987) Effects of climatic variability and possible climatic change on reliability of wheat cropping – a modelling approach. Agr For Meteor 41:123–142
Han L, Gresshoff PM, Hanan J (2011) A functional-structural modelling approach to auto regulation of nodulation. Ann Bot 107:855–863
Hinsinger P, Brauman A, Devau N, Gérard F, Jourdan C, Laclau J, Le Cadre E, Jaillard B, Plassard C (2011) Acquisition of phosphorus and other poorly mobile nutrients by roots. Where do plant nutrition models fail? Plant Soil 348:29–61
Ho MD, Rosas JC, Brown KM, Lynch JP (2005) Root architectural tradeoffs for water and phosphorus acquisition. Funct Plant Biol 32:737–748
Hutchinson JMC (2000) Three into two doesn’t go: two-dimensional models of bird eggs, snail shells and plant roots. Biol J Linn Soc 70:161–187
Jahnke S, Menzel MI, Van Dusschoten D, Roeb GW, Bühler J, Minwuyelet S, Blümler P, Temperton VM, Hombach T, Streun M, Beer S, Khodaverdi M, Ziemons K, Coenen HH, Schurr U (2009) Combined MRI–PET dissects dynamic changes in plant structures and functions. Plant J 59:634–644
Janott M, Gayler S, Gessler A, Javaux M, Klier C, Priesack E (2011) A one-dimensional model of water flow in soil-plant systems based on plant architecture. Plant Soil 341:233–256
Jansa J, Mozafar A, Frossard E (2005) Phosphorus acquisition strategies within arbuscular mycorrhizal fungal community of a single field site. Plant Soil 276:163–176
Javaux M, Schröder T, Vanderborght J, Vereecken H (2008) Use of a three–dimensional detailed modeling approach for predicting root water uptake. Vadose Zone J 7:1079–1088
Jones D, Nguyen C, Finlay R (2009) Carbon flow in the rhizosphere: carbon trading at the soil-root interface. Plant Soil 321:5–33
Kalliokoski T, Sievanen R, Nygren P (2010) Tree roots s self-similar branching structures: axis differentiation and segment tapering in coarse roots of three boreal forest tree species. Trees 24:219–236
Kurth W, Lanwert D (2011) Grammar-based models and fractals. In: Jopp F, Reuter H, Breckling B (eds) Modelling complex ecological dynamics. Springer, Berlin Heidelberg, pp 147–161
Kutschera L (1960) Wurzelatlas mitteleuropäischer Ackerunkräuter und Kulturpflanzen. DLG Verlag, Frankfurt am main
Lambers H, Shane MW, Cramer MD, Pearse SJ, Veneklaas EJ (2006) Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann Bot 98:693–713
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient–acquisition strategies change with soil age. Trends Ecol Evol 23:95–103
Lambers H, Finnegan P, Laliberté E, Pearse S, Ryan M, Shane M, Veneklaas E (2011) Phosphorus nutrition of Proteaceae in severely phosphorus–impoverished soils: are there lessons to be learned for future crops? Plant Physiol 156:1058–1066
Le Roux Y, Pagès L (1994) Développement et polymorphisme racinaire chez de jeunes semis d’hévéa (Hevea brasiliensis). Can J Bot 72:924–932
Leitner D, Schnepf A (2012) Image analysis of 2-dimensional root system architecture. ALGORITMY 2012, 19th Conference on Scientific Computing, Vysoké Tatry – Podbanské, Slovakia, September 9 –14, 2012. http://www.iam.fmph.uniba.sk/algoritmy2012/
Leitner D, Klepsch S, Knieß A, Schnepf A (2010a) The algorithmic beauty of plant roots - an L-system model for dynamic root growth simulation. Math Comput Model Dyn 16:575–587
Leitner D, Klepsch S, Bodner G, Schnepf A (2010b) A dynamic root system growth model based on L-systems. Tropisms and coupling to nutrient uptake from soil. Plant Soil 332:177–192
Leitner D, Schnepf A, Klepsch S, Roose T (2010c) Water uptake by a maize root system – an explicit numerical 3-dimensional simulation. In: Geophysical Research Abstracts, Vol. 12, EGU2010-8473, 2010, EGU General Assembly 2010, 2–7 March, Vienna, Austria
Lendenmann M, Thonar C, Barnard R, Salmon Y, Werner R, Frossard E, Jansa J (2011) Symbiont identity matters: carbon and phosphorus fluxes between medicago truncatula and different arbuscular mycorrhizal fungi. Mycorrhiza 21:689–702
Liao H, Rubio G, Yan XL, Cao AQ, Brown KM, Lynch JP (2001) Effect of phosphorus availability on basal root shallowness in common bean. Plant Soil 232:69–79
Liao M, Palta JA, Fillery IRP (2006) Root characteristics of vigorous wheat improve early nitrogen uptake. Aust J Agric Res 57:1097–1107
Lungley DR (1973) The growth of root systems – a numerical computer simulation model. Plant Soil 38:145–159
Lynch JP (2005) Root architecture and nutrient acquisition. In: BassiriRad H (ed) Nutrient acquisition by plants -an ecological perspective. Springer–Verlag, Berlin, pp 147–183
Lynch J (2007) Roots of the second green revolution. Aust J Bot 55:493–512
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: tools for future crops. Plant Physiol 156:1041–1049
Lynch JP, Beebe SE (1995) Adaptation of beans (Phaseolus vulgaris L.) to low phosphorus availability. Hortscience 30:1165–1171
Lynch JP, Brown KM (2001) Topsoil foraging – an architectural adaptation of plants to low phosphorus availability. Plant Soil 237:225–237
Lynch JP, Brown KM (2008) Root strategies for phosphorus acquisition. In: White PJ, Hammond JP (eds) The ecophysiology of plant–phosphorus interaction. Springer, the Netherlands, pp 83–116
Lynch JP, Brown KM (2012) New roots for agriculture: exploiting the root phenome. Phil Trans R Soc B 367:1598–1604
Lynch J, Ho M (2005) Rhizoeconomics: carbon costs of phosphorus acquisition. Plant Soil 269:45–56
Lynch J, van Beem J (1993) Growth and architecture of seedling roots of common bean genotypes. Crop Sci 33:1253–1257
Lynch JP, Nielsen KL, Davis RD, Jablokow AG (1997) SimRoot: modelling and visualization of root systems. Plant Soil 188:139–151
Ma Z, Walk TC, Marcus A, Lynch JP (2001) Morphological synergism in root hair length, density, initiation and geometry for phosphorus acquisition in Arabidopsis thaliana: a modelling approach. Plant Soil 236:221–235
Marschner P, Crowley D, Rengel Z (2011) Rhizosphere interactions between microorganisms and plants govern iron and phosphorus acquisition along the root axis – model and research methods. Soil Biol Biochem 43:883–894
Metselaar K, Van Lier QJ (2007) The shape of the transpiration reduction function under plant water stress. Vadose Zone J 6:124–139
Meyer KM, Mooij WM, Vos M, Hol WHG, van der Putten WH (2009) The power of simulating experiments. Ecol Model 220:2594–2597
Miguel MA, Postma JA, Lynch JP (2012) Functional role and synergystic effect of root traits for phosphorus acquisition efficiency and their genetic basis in common bean (Phaseolus vulgaris L.). PhD Thesis, The Pennsylvania State University, USA
Miller CR, Ochoa I, Nielsen KL, Beck D, Lynch JP (2003) Genetic variation for adventitious rooting in response to low phosphorus availability: potential utility for phosphorus acquisition from stratified soils. Funct Plant Biol 30:973–985
Morris MD (1991) Factorial sampling plans for preliminary computational experiments. Techinometrics 33:161–174
Mulia R, Dupraz C, van Noordwijk M (2010) Reconciling root plasticity and architectural ground rules in tree root growth models with voxel automata. Plant Soil 337:77–92
Nielsen KL, Lynch JP, Jablokow AG, Curtis PS (1994) Carbon cost of root systems: an architectural approach. Plant Soil 165:161–169
Nielsen KL, Miller CR, Beck D, Lynch JP (1998) Fractal geometry of root systems: field observations of contrasting genotypes of common bean (Phaseolus vulgaris L.) grown under different phosphorus regimes. Plant Soil 206:181–190
Nye PH, Tinker PB (1977) Solute movements in the root-soil system. Blackwell, Oxford
Oswald S, Menon M, Carminati A, Vontobel P, Lehmann E, Schulin R (2008) Quantitative imaging of infiltration, root growth, and root water uptake via neutron radiography. Vadose Zone J 7:1035–1047
Ozdemir G, Akpinar C, Sabir A, Bilir H, Tangolar S, Ortas I (2010) Effect of inoculation with mycorrhizal fungi on growth and nutrient uptake of grapevine genotypes (vitis spp.). Eur J Hortic Sci 75:103–110
Page ER, Gerwitz A (1974) Mathematical models, based on diffusion equations, to describe root systems of isolated plants, row crops, and swards. Plant Soil 41:243–254
Pagès L (1999) Root system architecture: from its representation to the study of its elaboration. Agronomie 19:295–304
Pagès L (2000) How to include organ interactions in models of the root system architecture? The concept of endogenous environment. Ann For Sci 57:535–541
Pagès L (2011) Links between root development traits and foraging performance. Plant Cell Environ 42:1749–1760
Pagès L, Aries F (1988) SARAH : modèle de simulation de la croissance, du développement, et de l’architecture des systèmes racinaires. Agronomie 8:889–896
Pagès L, Bengough AG (1997) Modelling minirhizotron observations to test experimental procedures. Plant Soil 189:81–89
Pagès L, Jordan MO, Picard D (1989) A simulation model of the three–dimensional architecture of the maize root system. Plant Soil 119:147–154
Pagès L, Vercambre G, Drouet JL, Lecompte F, Collet C, Le Bot J (2004) Root Typ: a generic model to depict and analyse the root system architecture. Plant Soil 258:103–119
Pierret A, Doussan C, Capowiez Y, Bastardie F, Pages L (2007) Root functional architecture: a framework for modeling the interplay between roots and soil. Vadose Zone J 6:269–281
Pohlmeier A, Vanderborght J, Haber–Pohlmeier S, Wienke S, Vereecken H, Javaux M (2010) Root water uptake and tracer transport in a lupin root system: integration of magnetic resonance images and the numerical model RSWMS. In: EGU General Assembly Conference Abstracts, pp 7217
Poorter H, Bühler J, van Dusschoten D, Climent J, Postma JA (2012a) Pot size matters: a meta–analysis of the effects of rooting volume on plant growth. Funct Plant Biol. doi:10.1071/FP12049
Poorter H, Niklas KJ, Reich PB, Oleksyn J, Poot P, Mommer L (2012b) Biomass allocation to leaves, stems and roots: meta–analyses of interspecific variation and environmental control. New Phytol 193:30–50
Postma JA, Lynch JP (2011a) Theoretical evidence for the functional benefit of root cortical aerenchyma in soils with low phosphorus availability. Ann Bot 107:829–841
Postma JA, Lynch JP (2011b) Root cortical aerenchyma enhances the growth of maize on soils with suboptimal availability of nitrogen, phosphorus, and potassium. Plant Physiol 156:1190–1201
Postma JA, Lynch JP (2012) Complementarity in root architecture for nutrient uptake in ancient maize/bean and maize/bean/squash polycultures. Ann Bot 110:521–534
Postma JA, Jaramillo RE, Lynch JP (2008) Towards modeling the function of root traits for enhancing water acquisition by crops. In: Ahuja LR, Reddy VR, Saseendran SA, Yu Q (eds) Advances in agricultural systems modelling -response of crops to limited water: understanding and modelling water stress effects on plant growth processes. ASA-CSSA-SSSA, Madison, pp 251–276
Pradal C, Dufour-Kowalski S, Boudon F, Fournier C, Godin C (2008) OpenAlea: a visual programming and component–based software platform for plant modelling. Funct Plant Biol 35:751–760
Pradal C, Boudon F, Nouguier C, Chopard J, Godin C (2009) PlantGL: a python–based geometric library for 3D plant modelling at different scales. Graph Models 71:1–21
Prusinkiewicz P (2004) Modeling plant growth and development. Curr Opin Plant Biol 7:79–83
Prusinkiewicz P, Lindenmayer A (1990) The algorithmic beauty of plants. Springer, Berlin
Ptashnyk M (2010) Derivation of a macroscopic model for nutrient uptake by hairy-roots. Nonlinear Anal: Real World Appl 11:4586–4596
Richardson AE, Barea JM, McNeill AM, Prigent-Combaret C (2009) Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil 321:305–339
Robinson D (1988) Optimal relations between root length and nutrient inflow rate in plant root systems. J Theo Biol 135:359–370
Robinson D (1994) The responses of plants to non–uniform supplies of nutrients. New Phytol 127:635–674
Robinson D (1996) Variation, co–ordination and compensation in root systems in relation to soil variability. Plant Soil 187:57–66
Robinson D (2005) Integrated root responses to variations in nutrient supply. In: BassiriRad H (ed) Nutrient acquisition by plants - an ecological perspective. Springer–Verlag, Berlin, pp 43–62
Roose T, Fowler A, Darrah P (2001) A mathematical model of plant nutrient uptake. Journal of Mathematical Biology 42:347–360
Roose T, Fowler A (2004) A mathematical model for water and nutrient uptake by plant root systems. J Theor Biol 228:173–184
Roose T, Schnepf A (2008) Mathematical models of plant-soil interaction. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 366:4597–4611
Roose T, Kirk G (2009) The solution of convection-diffusion equations for solute transport to plant roots. Plant Soil 316:257–264
Rose WC, Chichester FW, Williams JR, Ritchie JT (1982a) A contribution to simplified models of field solute transport. J Environ Qual 11:146–151
Rose WC, Chichester FW, Williams JR, Ritchie JT (1982b) Application of an approximate analytic method of computing solute profiles with dispersion in soils. J Environ Qual 11:151–155
Rose TJ, Bingah H, Rengel Z (2010) Wheat, canola and grain legume access to soil phosphorus fractions differs in soils with contrasting phosphorus dynamics. Plant Soil 326:159–170
Rubio G, Walk T, Ge Z, Yan X, Liao H, Lynch JP (2001) Root gravitropism and below-ground competition among neighbouring plants: a modelling approach. Ann Bot 88:929–940
Ryan PR, Delhaize E, Jones DL (2001) Function and mechanism of organic anion exudation from plant roots. Annu Rev Plant Physiol Plant MolBiol 52:527–560
Saltelli A, Scott EM, Chan K (2000) Sensitivity analysis. John Wiley & Sons, Ltd, Chichester
Schneider CL, Attinger S, Delfs JO, Hildebrandt A (2010) Implementing small scale processes at the soil-plant interface - the role of root architectures for calculating root water uptake profiles. Hydrol Earth Syst Sci 14:279–289
Schnepf A, Roose T, Schweiger P (2008) Growth model for arbuscular mycorrhizal fungi. J R Soc Interface 5:773–784
Schnepf A, Leitner D, Klepsch S, Pellerin S, Mollier A (2011) Modelling phosphorus dynamics in the soil-plant system. In: Bünemann E, Oberson A, Frossard E (eds) Phosphorus in action: biological processes in soil phosphorus cycling. Springer-Verlag Berlin, Heidelberg, pp 113–133
Schnepf A, Leitner D, Klepsch S (2012) Modeling phosphorus uptake by a growing and exuding root system. Vadose Zone J 11:318–327
Schröder T, Javaux M, Vanderborght J, Korfgen B, Vereecken H (2008) Effect of local soil hydraulic conductivity drop using a three–dimensional root water uptake model. Vadose Zone J 7:1089–1098
Schröder T, Javaux M, Vanderborght J, Koerfgen B, Vereecken H (2009a) Implementation of a microscopic soil-root hydraulic conductivity drop function in a soil-root architecture water transfer model. Vadose Zone J 8(3):783–792. doi:10.2136/vzj2008.0116
Schröder T, Tang L, Javaux M, Vanderborght J, Körfgen B, Vereecken H (2009b) A grid refinement approach for a three–dimensional soil–root water transfer model. Water Resour Res 45, W10412
Schröder N, Javaux M, Vanderborght J, Steffen B, Vereecken H (2012) Effect of root water and solute uptake on apparent soil dispersivity: a simulation study. Vadose Zone J. doi:10.2136/vzj2012.0009
Schulz H, Postma JA, van Dusschoten D, Scharr H, Behnke S (2012) 3D reconstruction of plant roots from MRI images. Proceedings of the International Conference on Computer Vision Theory and Applications (VISAPP), Rome, February 2012
Shibusawa S (1994) Modelling the branching growth fractal pattern of the maize root system. Plant Soil 165:339–347
Shu L, Shen J, Rengel Z, Tang C, Zhang F, Cawthray GR (2007) Formation of cluster roots and citrate exudation by Lupinus albus in response to localized application of different phosphorus sources. Plant Sci 172:1017–1024
Simunek J, Huang K, Van Genuchten M Th (1995) The SWMS_3D code for simulating water flow and solute transport in three-dimensional variably-saturated media. Version 1.0. Research Report n°139. Riverside, California. U. S. Salinity Laboratory. Agricultural Research Service. U.S. Department of Agriculture
Smith S, De Smet I (2012) Root system architecture: insights from Arabidopsis and cereal crops. Phil Trans R Soc B 367:1441–1452
Somma F, Clausnitzer V, Hopmans JW (1997) An algorithm for three-dimensional, simultaneous modeling of root growth, transient soil water flow, and solute transport and uptake. Version 2.1. Paper No. 100034. Dept of Land, Air, and Water Resources, University of California
Somma F, Hopmans JW, Clausnitzer V (1998) Transient three-dimensional modeling of soil water and solute transport with simultaneous root growth, root water and nutrient uptake. Plant Soil 202:281–293
Spek LY (1997) Generation and visualization of root-like structures in a three-dimensional space. Plant Soil 197:9–18
Steele KA, Virk DS, Kumar R, Prasad SC, Witcombe JR (2007) Field evaluation of upland rice lines selected for QTLs controlling root traits. Field Crops Res 101:180–186
Stingaciu L, Schulz H, Pohlmeier A, Behnke S, Zilken H, Javaux M, Vereecken H (2013) In situ root system architecture extraction from magnetic resonance imaging for application to water uptake modeling. Vadose Zone J. doi:10.2136/vzj2012.0019
Subbaiah R, Rao KA (1993) Root growth simulation model under specified environment. J Irrig Drain Eng 119:898–904
Tajini F, Suriyakup P, Vailhe H, Jansa J, Drevon JJ (2009) Assess suitability of hydroaeroponic culture to establish tripartite symbiosis between different amf species, beans, and rhizobia. BMC Plant Biol 9:73
Tardieu F (2010) Why work and discuss the basic principles of plant modelling 50 years after the first plant models? J Exp Bot 61:2039–2041
Thaler P, Pagès L (1998) Modelling the influence of assimilate availability on root growth and architecture. Plant Soil 201:307–320
Thonar C, Schnepf A, Frossard E, Roose T, Jansa J (2011) Traits related to differences in function among three arbuscular mycorrhizal fungi. Plant Soil 339:231–245
Thorup-Kristensen K (2001) Are differences in root growth of nitrogen catch crops important for their ability to reduce soil nitrate–N content, and how can this be measured. Plant Soil 230:185–195
Tsegaye T, Mullins CE, Diggle AJ (1995) Modelling pea (Pisum sativum) root growth in drying soil. A comparison between observations and model predictions. New Phytol 131:179–189
Van Lier QJ, Metselaar K, Van Dam JC (2006) Root water extraction and limiting soil hydraulic conditions estimated by numerical simulation. Vadose Zone J 5:1264–1277
Vance CP, Uhde-Stone C, Allen DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a non–renewable resource. New Phytol 157:423–447
Vercambre G, Pagès L, Doussan C, Habib R (2003) Architectural analysis and synthesis of the plum tree root system in an orchard using a quantitative modelling approach. Plant Soil 51:1–11
Vos J, Evers JB, Buck–Sorlin GH, Andrieu B, Chelle M, de Visser PHB (2010) Functional-structural plant modelling: a new versatile tool in crop science. J Exp Bot 61:2101–2115
Wagner B, Gärtner H, Ingensand H, Santini S (2010) Incorporating 2D tree-ring data in 3D laser scans of coarse–root systems. Plant Soil 334:175–187
Wagner B, Ingensand H, Santini S, Gärtner H (2011) A tool to model 3D coarse-root development with annual resolution. Plant Soil 346:79–96
Walk TC, van Erp E, Lynch JP (2004) Modelling applicability of fractal analysis to efficiency of soil exploration by roots. Ann Bot 94:119–128
Walk TC, Jaramillo R, Lynch JP (2006) Architectural tradeoffs between adventitious and basal roots for phosphorus acquisition. Plant Soil 279:347–366
Wallstrom T, Christie M, Durlofsky L, Sharp D (2002) Effective flux boundary conditions for upscaling porous media equations. Transp Porous Media 46:139–153
Wiesler F, Horst WJ (1994) Root growth and nitrate utilization of maize cultivars under field conditions. Plant Soil 163:267–277
Wu L, Bingham IJ (2009) Using modelling as a tool to explore resource use efficiency by crops. Asp Appl Biol 93:257–261
Wu L, Shepherd A (2011) Special features of the SPACSYS modeling package and procedures for parameterization and validation. In: Ahuja LR, Ma L (eds) Methods of introducing system models into agricultural research. ASA, CSSA & SSSA, Madison, pp 117–154
Wu L, Baddeley JA, Watson CA (2005) Designer root systems – the value of modelling to determine optimum root systems for different end uses. Asp Appl Biol 71:11–16
Wu L, McGechan MB, McRoberts N, Baddeley JA, Watson CA (2007) SPACSYS: Integration of a 3D root architecture component to carbon, nitrogen and water cycling - Model description. Ecol Model 200:343–359
Wu L, Bingham IJ, Baddeley JA, Watson CA (2008) The importance of 3D root architecture when simulating plant N uptake. In: Ma L, Ahuja LR, Bruulsema TW (eds) Quantifying and understanding plant nitrogen uptake systems modelling (9). CRC Press, Boca Raton, pp 197–218
Xu L, Henke M, Zhu J, Kurth W, Buck-Sorlin G (2011) A functional-structural model of rice linking quantitative genetic information with morphological development and physiological processes. Ann Bot 107:817–828
Yang HS, Janssen BH (2000) A mono-component model of carbon mineralization with a dynamic rate constant. European Journal of Soil Science 51:517–529
Zhu J, Lynch JP (2004) The contribution of lateral rooting to phosphorus acquisition efficiency in maize (Zea mays L.) seedlings. Funct Plant Biol 31:949–958
Zhu J, Kaeppler S, Lynch JP (2005) Topsoil foraging and phosphorus acquisition efficiency in maize (Zea mays L.). Funct Plant Biol 32:749–762
Zhu J, Brown KM, Lynch JP (2010a) Root cortical aerenchyma improves the drought tolerance of maize (Zea mays L.). Plant Cell Environ 33:740–749
Zhu J, Zhang C, Lynch JP (2010b) The utility of phenotypic plasticity of root hair length for phosphorus acquisition. Funct Plant Biol 37:313–322
Zygalakis K, Roose T (2012) A mathematical model for investigating the effect of cluster roots on plant nutrient uptake. Eur Phys J Spec Top 204:103–118
Zygalakis K, Kirk G, Jones D, Wissuwa M, Roose T (2011) A dual porosity model of nutrient uptake by root hairs. New Phytol 192:676–688
Acknowledgments
The authors would like to thank Dr Ian Bingham and Dr Davide Tarsitano SRUC, UK, for providing additional information on the SPACSYS model, and Prof Jonathan Lynch, Penn State University for providing additional information on the SimRoot model. This research was supported by the Australian Research Council and by the Austrian Science Fund FWF (Grant No.: V220-N13). Andrea Schnepf is an Elise-Richter research fellow. Daniel Leitner is recipient of an APART-fellowship of the Austrian Academy of Sciences at the Computational Science Center, University of Vienna.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tim Simon George.
Rights and permissions
About this article
Cite this article
Dunbabin, V.M., Postma, J.A., Schnepf, A. et al. Modelling root–soil interactions using three–dimensional models of root growth, architecture and function. Plant Soil 372, 93–124 (2013). https://doi.org/10.1007/s11104-013-1769-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-1769-y