Abstract
Cytosolic free calcium ([Ca2+]cyt), which is essential during pollen germination and pollen tube growth, can be sensed by calmodulin-like proteins (CMLs). The Arabidopsis thaliana genome encodes over 50 CMLs, the physiological role(s) of most of which are unknown. Here we show that the gene AtCML24 acts as a regulator of pollen germination and pollen tube extension, since the pollen produced by loss-of-function mutants germinated less rapidly than that of wild-type (WT) plants, the rate of pollen tube extension was slower, and the final length of the pollen tube was shorter. The [Ca2+]cyt within germinated pollen and extending pollen tubes produced by the cml24 mutant were higher than their equivalents in WT plants, and pollen tube extension was less sensitive to changes in external [K+] and [Ca2+]. The pollen and pollen tubes produced by cml24 mutants were characterized by a disorganized actin cytoskeleton and lowered sensitivity to the action of latrunculin B. The observations support an interaction between CML24 and [Ca2+]cyt and an involvement of CML24 in actin organization, thereby affecting pollen germination and pollen tube elongation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Once a fully developed pollen grain has successfully adhered to a compatible pistil, it becomes hydrated, germinates, and develops a pollen tube (Taylor and Hepler 1997). Ca2+ acts as a prime regulatory signal in pollen germination and pollen tube growth (Jaffe et al. 1975; Schiefelbein et al. 1993). In vitro imaging of extending lily and tobacco pollen tubes has demonstrated the presence of Ca2+ concentration gradients and regular oscillations of the cytosolic free calcium concentration ([Ca2+]cyt) at the tip of the tube (Pierson et al. 1996). As both disruption of the endogenous Ca2+ gradient and application of high concentrations of exogenous Ca2+ are detrimental to pollen tube extension, it is evident that both the intra- and extracellular Ca2+ are important for pollen growth (Picton and Steer 1983; Obermeyer and Weisenseel 1991; Iwano et al. 2004; Wang et al. 2004; Lazzaro et al. 2005).
Actin dynamics and F-actin organization represent an additional important feature in pollen germination and tube growth. Actin cables direct the cytoplasmic streaming needed for rapid tube extension (Hepler et al. 2001; Ren and Xiang 2007; Xiang et al. 2007; Cheung and Wu 2008; Zhang et al. 2010). Pollen tube actin filaments are either longitudinally oriented (primarily present in the pollen tube shank), densely clustered at the subapex (the “actin collar”), or behave in a dynamic fashion in the distal region of the pollen tube (Lovy-Wheeler et al. 2005; Cheung et al. 2008; Chen et al. 2009; Vidali et al. 2009; Staiger et al. 2010). Time-course analyses have established that the actin collar structure alternates with F-actin dynamics, and that the peak of tip F-actin formation precedes tube growth (Fu et al. 2001). The quantity of tip F-actin is inversely related to both the rate of pollen tube elongation and the tip [Ca2+]cyt, while the dynamic polymerization and depolymerization of the microfilament cytoskeleton is a key regulator of the pollen grain plasma membrane Ca2+ channel (Wang et al. 2004).
Calmodulin-like proteins (CMLs) belong to a plant-specific family of Ca2+ sensors. They feature Ca2+-binding EF hands and share at least 16 % amino acid identity with calmodulin (McCormack and Braam 2003; Azimzadeh et al. 2008). The Arabidopsis thaliana genome encodes some 50 CML proteins (McCormack and Braam 2003). Calcium-dependent protein kinases (CDPKs) and calcineurin B-like proteins (CBLs) are further members of the Ca2+-binding EF hand superfamily. Calmodulins, CDPKs, and CBLs are all involved in a wide range of physiological processes (DeFalco et al. 2010; Landoni et al. 2010), but the function of most CMLs remains largely unknown. Accumulating data suggest that they have a role in plant growth and signaling (Delk et al. 2005). The A. thaliana CML family AtCML24 has been shown to participate in pathogen infection-associated nitric oxide production, as well as having a role in seed germination, the transition from vegetative to reproductive growth, and the sensing of photoperiod (Delk et al. 2005). The cml24-4 mutant flowers late, and its leaves contain a higher level of nitric oxide than wild type (WT; Tsai et al. 2007); in addition its innate immune response is compromised, as is its pathogen-mediated generation of nitric oxide (Ma et al. 2008). The elevation of [Ca2+]cyt is believed to be linked to nitric oxide generation via the action of CML24 (Tsai et al. 2007), and the protein appears able to bind with a cysteine protease involved in autophagy progression (Tsai et al. 2013). The wide-ranging activity of CML24 suggests that the CMLs may well be associated with many Ca2+-dependent processes. Here, we show that AtCML24 acts as a positive regulator of pollen germination and pollen tube development, and that it influences pollen physiology via an effect exerted on cytoskeleton dynamics signaling.
Materials and methods
Plant materials and growing conditions
A. thaliana ecotype Columbia-0 (WT) and cml24 mutants were grown in potting mix (Lu-Qing Plant Inc.) under an 8 h photoperiod (140 µmol m−2 s−1 light), 22/20 °C day/night temperature regime, and 60 % relative humidity. After 3–4 weeks, the photoperiod was lengthened to 16 h to initiate flowering.
cml24 mutants and transcription of CML24
The T-DNA insertion line cml24-T1 (CS815583) was obtained from the Nottingham Arabidopsis Stock Centre (http://arabidopsis.info), and homozygous segregants were identified via a genomic polymerase chain reaction (PCR) assay based on the primer pair cml24-T1 LP/RP, in conjunction with the left border primer LB2 (sequences given in Table S1). Reverse transcription (RT)-PCR was performed to assess the abundance of CML24 transcript, using two different primer pairs (Table S1). The template for these reactions was RNA extracted from leaves of 21-day-old plants using TriPure isolation reagent (Roche, Basel, Switzerland), followed by complementary DNA (cDNA) synthesis achieved with Moloney murine leukemia virus (MMLV) reverse transcriptase (Thermo, MA, USA). A segment of AtActin2 was used as the reference sequence. The cml24-4 mutant was isolated from a targeting-induced local lesions in genome (TILLING) population; it carries a K124E point mutation predicted to alter the functionality of the CML24 protein.
Pollen germination and pollen tube extension measurement
Pollen germination in vitro followed a modified version of the Fan et al. (2001) protocol. In brief, pollen harvested from newly fully opened flowers was placed on the surface of pollen germination medium [1 mM KCl, 10 mM CaCl2, 0.8 mM MgSO4, 1.5 mM boric acid, 1 % (w/v) agarose, 18 % (w/v) sucrose, 10 mM myo-inositol, 5 mM 2-(N-morpholino)ethanesulfonic acid (MES), pH 5.8] and incubated at 28 °C for 3–10 h. Pollen grains obtained from wild-type Col-0 and cml24 mutant plants were germinated for 10 h in vitro. Pollen germination and pollen tube growth were determined from digital micrographs acquired via light microscopy. Three independent replicates were performed, and for each replicate, at least 200 pollen grains or pollen tubes were assessed to obtain a mean value used for drawing the columns and statistical analysis. Student’s t test was used to test the statistical significance of differences between means.
Aniline blue staining assay
The method used for aniline blue staining of pollen tubes growing within the pistil followed a published protocol, with only minor modifications (Ishiguro et al. 2001). Briefly, pre-emasculated mature WT flowers were pollinated with either wild-type or cml24 mutant pollen. After 2, 6, and 10 h, the pollinated pistils were incubated in fixing solution containing ethanol:acetic acid (3:1) for 1 h at room temperature. The fixed pistils were washed three times with distilled water before an 8-h incubation step in 4 M NaOH softening solution. The pistils were washed three times with distilled water before subsequent staining in aniline blue solution (0.05 % aniline blue in 50 mM K3PO4 buffer, pH 7.5) for 3–5 h in the dark. After staining, micrographs of the pistils were obtained by fluorescence microscopy. To quantify the seed setting rate, siliques were harvested 10 days after flowering, then decolorized by immersion in 100 % EtOH to allow for seed set to be visualized.
Mutation complementation by transgenesis
A ~1.1-kb upstream sequence, along with a 486-bp genic fragment of CML24, was PCR-amplified using the primer pair CML24 fw/rev (Table S1). The fragment was first ligated into the pEASY-Blunt Cloning Vector (Transgen, Beijing, China) and subsequently into the HindIII and SacI cloning sites of the binary pCambia plasmid. The fusion product was introduced into Agrobacterium tumefaciens strain EHA105, then transformed into cml24-T1 plants using the floral dip method (Clough and Bent 1998), followed by hygromycin selection. For detection of the CML24 expression level in CML24 gene complementation, total RNA was extracted from mature pollen using TriPure isolation reagent (Roche, Basel, Switzerland) and cDNA was synthesized using MMLV reverse transcriptase (Thermo, MA, USA). Two complementation transgenic lines (C-1 and C-2) were obtained and used for further analyses.
Quantitative RT-PCR (qPCR)
qPCR was performed using the BioRad qPCR system (FastStart Universal SYBR Green Master, Roche) with three biological replicates. An Rn threshold of 0.2 was applied to obtain the threshold cycle number. Relative transcript abundances were determined using the 2−ΔΔCT method (Livak and Schmittgen 2001). qPCR primer sequences are listed in Table S1.
Measurement of [Ca2+]cyt in pollen and pollen tube
The fluorescent calcium indicator Fluo-3 AM was loaded into pollen grains following a published protocol (Qu et al. 2012). The estimation of [Ca2+]cyt in the pollen tube required in vitro germination of pollen for 2 h prior to Fluo-3 AM loading. The resulting fluorescence signal was detected by fluorescence microscopy. Three independent replicates were performed, and a mean based on at least 100 pollen grains or pollen tubes per replicate was calculated. Student’s t test was used to test the statistical significance of differences between means.
F-actin staining
The actin cytoskeleton was stained as described elsewhere (Wu et al. 2010). Pollen grains were dispersed over the surface of pollen germination medium (as above) and held at 28 °C for 3 h. The emerged pollen tubes were fixed for 1 h in liquid pollen germination medium supplemented with 300 mM 3-maleimidobenzoic acid N-hydroxysuccinimide ester, and subsequently extracted in 0.05 % v/v Nonidet P-40 in liquid germination medium for 10 min. After three 10-min washes in TBSS [50 mM Tris-HCl (pH 7.5), 200 mM NaCl, 400 mM sucrose] containing 0.05 % v/v Nonidet P-40, the pollen tubes were stained overnight at 4 °C in 200 nM Alexa-488 phalloidin (Invitrogen, CA, USA) dissolved in the wash solution. Micrographs were obtained using confocal laser scanning microscopy (excitation at 488 nm, emission at 515 nm).
Results
Loss of CML24 function delayed pollen germination and inhibited pollen tube elongation
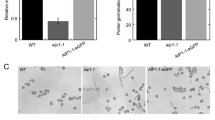
Transcription of CML24 in the WT pollen and pollen tube (but not in the leaf or flower) was confirmed using both semiquantitative RT-PCR (Fig. 1a) and qPCR (Fig. 1b). In both the T-DNA mutant cml24-T1 and the K124E point mutant cml24-4, the abundance of CML24 transcript was much lower than in the WT (Fig. 2b). The T-DNA insertion in the 3′-untranslated region (UTR) dramatically decreased the transcript accumulation, suggesting that the disrupted sequence may contain essential regulatory elements for the stability of the messenger RNA (mRNA) (Bashirullah et al. 2001; Winter et al. 2008). Both cml24-T1 and cml24-4 pollen germinated less readily in vitro than did WT pollen; the mean germination percentages of the two mutants were 26 % (cml24-T1) and 22 % (cml24-4), compared with the WT rate of 67 % (Fig. 3d). The mean length of the pollen tube after 10 h was also reduced in both mutants (Fig. 3a–c, e). Quantification of the growth rate of the pollen tube showed a delay in both mutants compared with WT (Fig. 3f–i). Analysis of in vivo germination based on pollinating WT stigma showed that, while WT pollen had successfully penetrated into the style and reached the top of the transmitting tract within 2 h of pollination (Fig. 4a), the mutant pollen tubes had only just begun to penetrate the style by this time (Fig. 4b, c). At 6 h after pollination, WT pollen tubes had grown much more strongly than those of the two mutants (Fig. 4d–f). And by 10 h after pollination, the WT pollen tube had reached the bottom of the transmitting tract (Fig. 4g), while those of the mutants had only reached the halfway point (Fig. 4h, i). Data from the pollination experiments suggest that pollen germination on the stigma is delayed and growth of the pollen tube in the style and transmitting tract is inhibited in cml24 mutants.
cml24 mutants are compromised with respect to both pollen germination and pollen tube extension. a WT, b cml24-T1 mutant, and c cml24-4 mutant pollen germinated in vitro for 10 h. Bar 100 µm. d Pollen germination rate after 10 h. e Pollen tube length after 10 h. f–h Time-lapse images of WT, cml24-T1, and cml24-4 pollen tubes captured at 5-min intervals. Bar 20 µm. i Mean growth rate of WT, cml24-T1, and cml24-4 pollen tubes. *, **P < 0.01, 0.005; error bars show the standard error (SE)
cml24 mutation results in slower pollen tube elongation when applied to WT stigma. Arrows indicate zones reached by most of the pollen tubes within the transmitting tract. Bar 100 µm. a–c WT, cml24-T1, and cml24-4 pollen germinated 2 h after pollination. d–f WT, cml24-T1, and cml24-4 pollen germinated 6 h after pollination. g–i WT, cml24-T1, and cml24-4 pollen germinated 10 h after pollination
Complementation of the cml24-T1 mutant
Transforming cml24-T1 with a copy of CML24 driven by its native promoter fusion restored the WT level of CML24 transcription (Fig. 5d). The average pollen germination rate, pollen tube length and growth rate of the transgenic lines C-1 and C-2 did not differ significantly from WT levels (Fig. 5e, f, h). Corresponding photos are shown in Fig. 5a–c, g. In the in vivo germination assay, when measured 6 h after pollination, the length of the pollen tube in C-1 and C-2 was equivalent to that of the WT pollen tube (Fig. 5i). These results confirm that defects in male gametophytes are indeed caused by knock-down expression of CML24.
Complementation of cml24. a WT pollen germinated for 10 h. b, c Pollen of complementation lines C-1 and C-2 germinated for 10 h. Bar 100 µm. d CML24 transcript abundance assayed by qPCR. e Germination rate of pollen and f pollen tube elongation in lines C-1 and C-2. g Time-lapse images of the growth of WT, C-1, and C-2 pollen tubes. Bar 10 µm. h Mean growth rate of WT, C-1, and C-2 pollen tubes. Error bars indicate the SE. i WT, C-1, and C-2 pollen applied to WT stigma, sampled after 6 h. Bar 100 µm. *, **P < 0.01, 0.005; error bars show the SE
Decreased seed set in cml24 mutants is caused by abnormal pollen tube growth
Analysis of self-fertilized WT, cml24-T1 and cml24-4 mutant plant siliques showed that the seed setting rate of the mutants was about 70 and 67 %, which is lower than the 100 % setting rate of WT (Fig. 6e). Photos of discolored siliques are shown in Fig. 6a, b. The transgenic complemented lines C-1 and C-2 had normal seed settings as for WT (Fig. S3a). The average seed number per silique was equivalent to that of the WT silique (Fig. S3b).
cml24 mutation results in a high proportion of nonfertilized ovaries. Siliques of a WT, b cml24-T1 mutant, c WT♀ × WT♂ hybrid, d WT♀ × cml24-T1♂ hybrid. Bar 0.1 cm. The seed setting rate of siliques in e selfed WT, cml24-T1, and cml24-4 plants, f pollinated WT, and cml24 mutant plants. *P < 0.01. Error bars indicate the SE
To further clarify if the seed defect in cml24 mutants was caused by male gametophytes, we performed reciprocal cross-pollinations between wild-type and cml24 mutant plants. WT stigmas were pollinated with pollen from Col-0 or cml24 mutants. The average seed setting rate was 72 and 71 % when cml24-T1 and cml24-4 was the pollen donor (Fig. 6f), which is consistent with the self-fertilized plants.
cml24 mutants are hyposensitive to extracellular Ca2+ and K+
Extracellular ions such as Ca2+ and K+ are important for pollen germination and tube growth, promoting both processes at low concentration and restraining them at high concentration (Wang et al. 2004; Fan et al. 2001). CML24 is a candidate downstream effector of calcium, and probably participates in Ca2+-related signal transduction. Also, calcium current by plasma membrane calcium channel is the key trigger in activating Ca2+ signal (Pandey et al. 2007; Zhang et al. 2007). A concentration gradient of calcium was employed for pollen germination bioassays. The growth of the WT pollen tube increased as the concentration of exogenous Ca2+ supplied rose from 0.1 to 10 mM, but was suppressed at 50 mM. The growth of both the cml24-T1 and the cml24-4 pollen tubes behaved quite similarly, although their level of sensitivity to Ca2+ was not so great (Fig. 7a). As the effect of extracellular Ca2+ on pollen tube growth is regulated by transmembrane cellular signals (Holdaway-Clarke et al. 1997), this suggests that CML24 may participate in the regulation of cellular Ca2+ signal.
Effect of externally applied Ca2+ and K+ on pollen tube extension. a Externally applied Ca2+. Pollen grains were germinated on solidified medium containing 1 mM K+ and the indicated concentration of Ca2+. b Externally applied K+. Pollen grains were germinated on solidified medium containing 10 mM Ca2+ and the indicated concentration of K+. Error bars indicate the SE. *P < 0.01
To keep rapid growth, germinated pollen tubes are equipped with an efficient ion transport system at the plasma membrane to sustain higher cellular osmotic pressure (Zhang et al. 2007). K+ is a crucial osmotic ion for pollen tube elongation. The extension of the pollen tube in response to exogenous K+ was also investigated. Supply of up to 10 mM K+ promoted the elongation of the WT pollen tube, but growth was suppressed once the concentration reached 20 mM. Both cml24-T1 and cml24-4 pollen tubes were less sensitive to exogenous K+ than were WT tubes (Fig. 7b). Pollen tube elongation in the complemented cml24-T1 lines behaved indistinguishably from WT ones (Fig. S4).
[Ca2+]cyt in the cml24 mutants was greater than in WT
The effect of extracellular Ca2+ and K+ on pollen tube growth may be mediated by intracellular signals, and then further alter transmembrane transportation of these ions. Intracellular Ca2+ has been proved to be one of the most important second messengers and is involved in regulating pollen germination and pollen tube growth (Konrad et al. 2011). Quantification of the Fluo-3 AM fluorescence generated in treated cml24 mutant pollen and pollen tubes showed that [Ca2+]cyt was higher than in WT (Fig. 8). The level present in the pollen and pollen tubes of the complemented cml24-T1 lines was the same as in WT plants (Fig. S5).
Disorganized actin cables are a characteristic feature in the cml24 mutants
Actin dynamics and F-actin patterns play critical roles in regulation of pollen germination and pollen tube growth (Fu et al. 2001), so it is worth checking whether the actin cytoskeleton organization is affected by CML24 mutation. Confocal microscopy imaging showed that, in WT pollen tubes, the actin filaments formed a dense structure at the subapex (Fig. 9a II), but were less abundant in the apical region (Fig. 9a III), and with numerous longitudinal actin cables throughout the shank of the WT pollen tube (Fig. 9a I). However, this normal pattern of actin cytoskeleton in WT pollen tubes was disrupted in the cml24-T1 pollen tubes (Fig. 9b–f); For example, some of the mutant tubes completely lacked the subapex actin structure (Fig. 9b ii), the actin filaments at the extreme tip also showed abnormal organization (Fig. 9c iii), or the actin cables were not oriented in a strictly longitudinal arrangement parallel to the growth axis (Fig. 9d i, e i). In WT tissue, most (>91 %) of the actin appeared normal, whereas in the cml24-T1 mutant, this proportion was slightly over 12 %. A similar level of disorganization was a feature of the cml24-4 pollen tubes as well (Fig. S6b–d). In the complemented cml24-T1 lines, the actin cytoskeleton was indistinguishable from that of WT (Fig. S6e, f).
Disorganization of actin cables in cml24-T1 pollen tube. a WT pollen tube: (I) longitudinal actin cables, (II) actin fringes at the subapex of WT pollen tube, (III) highly dynamic but less abundant actin filaments at the extreme tips. b–f Forms of actin disorganization (square brackets) in cml24-T1 pollen tubes. i, ii, and iii show abnormal actin organization compared with normal structures I, II, and III in WT pollen tubes, respectively. Bar 10 µm
cml24 mutant pollen germination and pollen tube extension are insensitive to LatB treatment
The above results suggest that the level of filamentous actin is affected in cml24 pollen tubes. We sought to determine the effects of the suggested alterations in cml24-T1 and cml24-4 actin levels using the actin-depolymerizing drug latrunculin B (LatB), which prevents actin polymerization by binding monomeric actin (Gibbon et al. 1999). A concentration gradient of LatB was employed for pollen germination bioassays. The germination of cml24 mutant pollen was unaffected by the presence of 0.5–1.5 nM LatB in the medium. In contrast, WT pollen was sensitive to the drug, especially at the highest concentration tested, at which hardly any pollen grains were able to germinate (Fig. 10a). The elongation of both WT and cml24 mutant pollen tubes was inhibited by LatB in a dose-dependent manner, although the effect was less marked in the mutants’ pollen (Fig. 10b). In the presence of 1.5 nM LatB, WT pollen tube growth was stopped, whereas those of cml24-T1 and cml24-4 continued to extend (Fig. 10b). Photographs are shown in Fig. S7. Meanwhile, the complemented cml24-T1 pollen responded in the same way as did WT pollen (Fig. S8).
cml24 mutation reduces the sensitivity of pollen germination and pollen tube elongation to LatB treatment. a WT, cml24-T1, and cml24-4 pollen germinating on a medium containing various concentrations of LatB for 10 h. b The insensitivity of pollen tube elongation to LatB treatment in the absence of CML24. *P < 0.01
Discussion
The A. thaliana CML genes encode putative Ca2+ sensors and feature a conserved Ca2+-binding domain (McCormack and Braam 2003). CML24 is a 161-residue 16-kDa protein which shares 66 % peptide similarity and about 40 % peptide identity with the A. thaliana calmodulins 1, 2, 6, and 7. CML24 binds Ca2+, and its conformation is altered following its binding with Ca2+ (Delk et al. 2005). The inference is that it probably functions as a Ca2+ sensor. Here, it has been demonstrated that CML24 plays a role in regulating pollen germination and pollen tube elongation, since mutations to the gene inhibited both processes both in vitro (Fig. 3) and in vivo (Fig. 4). The abundance of CML24 transcript appeared to be greater in cml24-4 than in cml24-T1 (Fig. 2), even though the phenotype was more severe in cml24-4. The assumption is that the K124E point mutation altered functions in addition to its signaling with respect to pollen tube development. 4′,6-Diamidino-2-phenylindole (DAPI) treatment revealed that the nuclei of both cml24-T1 and cml24-4 pollen took up as much of the stain as did WT nuclei (Fig. S1), which suggests that the lesion in the cml24 mutants was not one affecting pollen development, but rather one which compromised pollen germination and tube growth. The mutants also showed lower seed setting rates (Fig. 6), while the outcome of the reciprocal crosses proved that the compromised self-fertility of the mutants was due to a weakness in male gametophytes (Fig. 6).
Cytoplasmic free Ca2+ is a ubiquitously exploited messenger during episodes of both biotic and abiotic stress (Batistic and Kudla 2012), while the K+ ion is used by many plant tissues, including the pollen tube, to balance their osmotic environment and stabilize their membranes (Franklin-Tong 2010). K+ influx also promotes pollen tube growth (Fan et al. 2001; Mouline et al. 2002; Boavida and McCormick 2007). The expression of CML24-GFP in Arabidopsis leaf protoplasts demonstrates that CML24 is deposited in the cytoplasm (Fig. S2). It determines the sensitivity of the pollen and pollen tube to exogenously supplied K+ and Ca2+ (Fig. 7), and it is involved in the regulation of [Ca2+]cyt (Fig. 8). The behavior of the complemented cml24-T1 lines confirmed the function of CML24 in regulating pollen germination and pollen tube growth (Figs. 5, S4, S5). Raising [Ca2+]cyt has been implicated in the inhibition of inward K+ currents in the pollen tube (Zhao et al. 2013), so the higher [Ca2+]cyt level in the cml24 mutants may explain their observed hyposensitivity towards extracellular Ca2+ and K+ during pollen germination and tube growth.
There is a close interaction between intracellular Ca2+ and cytoskeleton in pollen tubes. High levels of [Ca2+]cyt are known to damage the actin cytoskeleton of the lily pollen tube (Cardenas et al. 2008). Here, the actin cables formed in the cml24 mutant pollen tubes appeared disorganized, especially the actin fringe (Fig. 9). In lily, the actin fringe is essential for the rapid growth of the pollen tube, and its density and length are both determinants of the tubes’ growth rate (Dong et al. 2012). The growth rate of the cml24 pollen tube was much slower than that of the WT tube (Fig. 3), and they exhibited a distinctly abnormal actin fringe. Apical F-actin dynamics are correlated with the [Ca2+]cyt at the distal end of the tube, and it is believed that some cross-talk occurs between actin dynamics and the oscillation of the Ca2+ gradient in this region during tube growth (Fu et al. 2001; Wang et al. 2004; Gu et al. 2005; Hwang et al. 2005). Ion fluxes regulate the structure and activity of F-actin through the medium of various actin binding proteins (Cardenas et al. 2008). According to Wang et al. (2004), Ca2+-activated actin depolymerization may further raise the [Ca2+]cyt, while actin depolymerization-induced elevation of [Ca2+]cyt may in turn accelerate actin depolymerization. Therefore, the disorganized actin filaments noted in the cml24-T1 and cml24-4 pollen tubes may reflect the outcome of a higher than normal [Ca2+]cyt. A high [Ca2+]cyt can elevate the amount of monomeric actin present, supplying additional targets for LatB to bind to, and thereby preventing an optimal rate of actin polymerization from occurring in the pollen tube. Consequently, the hyposensitivity of the cml24-T1 and cml24-4 mutants to LatB may result in a greater frequency of monomeric actin in the pollen tube (Fig. 10). After binding with LatB, the monomeric actin could subsequently be polymerized, in this way supporting the extension of the pollen tube in the cml24 mutant.
According to Tsai et al. (2013), CML24 interacts with the autophagy-related gene ATG4b and affects autophagy progression in Arabidopsis. Recycling the contents of the cytoplasm is a marker for cell autophagy, and is a process particularly required for pollen tube extension (Liu and Bassham 2012; Chebli et al. 2013). The A. thaliana protein ATG6 co-localizes with ATG8, a symbol of pre-autophagosome formation, and is essential for normal germination of the pollen (Fujiki et al. 2007; Harrison-Lowe and Olsen 2008). The known interaction of CML24 with ATG4b to regulate AGT8 accumulation and autophagy (Tsai et al. 2013) suggests a second possible regulatory pathway in which CML24 could be involved in pollen tube extension.
In line with the proposed function of all CML gene products, CML24 is likely to be a downstream component in [Ca2+]cyt signaling. Our hypothesis is that pollen development requires a robust intracellular signaling system to regulate plasma membrane Ca2+ channels, generate the appropriate Ca2+ concentration, and integrate K+ influx to modulate the cytoplasmic Ca2+ signal. Any blockage of downstream signaling in response to [Ca2+]cyt could activate a feedback response in order to raise the [Ca2+]cyt. Since mutations to CML24 inhibit pollen germination and slow the elongation of the pollen tube, the inference is that CML24 is a positive regulator of both of these processes. Understanding how CML24 modulates [Ca2+]cyt to facilitate K+ influx will require an electrophysiological approach based on cml24 mutants and transgenic overexpressors. A major mechanism for the regulation of ion channel activity is protein phosphorylation/dephosphorylation, but CML24 is unlikely to be a protein kinase; so a screen aimed at identifying which proteins interact with CML24 may provide a handle on the relationship between CML24 activity, [Ca2+]cyt regulation of ion channel activity, and pollen germination and tube growth.
References
Azimzadeh J, Nacry P, Christodoulidou A, Drevensek S, Camilleri C, Amiour N, Parcy F, Pastuglia M, Bouchez D (2008) Arabidopsis TONNEAU1 proteins are essential for preprophase band formation and interact with centrin. Plant Cell 20(8):2146–2159
Bashirullah A, Cooperstock RL, Lipshitz HD (2001) Spatial and temporal control of RNA stability. Proc Natl Acad Sci USA 98:7025–7028
Batistic O, Kudla J (2012) Analysis of calcium signaling pathways in plants. Biochim Biophys Acta 1820(8):1283–1293
Boavida LC, McCormick S (2007) TECHNICAL ADVANCE: Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J 52(3):570–582
Cardenas L, Lovy-Wheeler A, Kunkel JG, Hepler PK (2008) Pollen tube growth oscillations and intracellular calcium levels are reversibly modulated by actin polymerization. Plant Physiol 146(4):1611–1621
Chebli Y, Kroeger J, Geitmann A (2013) Transport logistics in pollen tubes. Mol Plant 6(4):1037–1052
Chen N, Qu X, Wu Y, Huang S (2009) Regulation of actin dynamics in pollen tubes: control of actin polymer level. J Integr Plant Biol 51(8):740–750
Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59:547–572
Cheung AY, Duan QH, Costa SS, de Graaf BH, Di Stilio VS, Feijo J, Wu HM (2008) The dynamic pollen tube cytoskeleton: live cell studies using actin-binding and microtubule-binding reporter proteins. Mol Plant 1(4):686–702
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743
DeFalco TA, Bender KW, Snedden WA (2010) Breaking the code: Ca2+ sensors in plant signalling. Biochem J 425(1):27–40
Delk NA, Johnson KA, Chowdhury NI, Braam J (2005) CML24, regulated in expression by diverse stimuli, encodes a potential Ca2+ sensor that functions in responses to abscisic acid, daylength, and ion stress. Plant Physiol 139(1):240–253
Dong H, Pei W, Haiyun R (2012) Actin fringe is correlated with tip growth velocity of pollen tubes. Mol Plant 5(5):1160–1162
Fan LM, Wang YF, Wang H, Wu WH (2001) In vitro Arabidopsis pollen germination and characterization of the inward potassium currents in Arabidopsis pollen grain protoplasts. J Exp Bot 52(361):1603–1614
Franklin-Tong N (2010) Plant fertilization: bursting pollen tubes! Curr Biol 20(16):R681–R683
Fu Y, Wu G, Yang Z (2001) Rop GTPase-dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J Cell Biol 152(5):1019–1032
Fujiki Y, Yoshimoto K, Ohsumi Y (2007) An Arabidopsis homolog of yeast ATG6/VPS30 is essential for pollen germination. Plant Physiol 143(3):1132–1139
Gibbon BC, Kovar DR, Staiger CJ (1999) Latrunculin B has different effects on pollen germination and tube growth. Plant Cell 11(12):2349–2363
Gu Y, Fu Y, Dowd P, Li S, Vernoud V, Gilroy S, Yang Z (2005) A Rho family GTPase controls actin dynamics and tip growth via two counteracting downstream pathways in pollen tubes. J Cell Biol 169(1):127–138
Harrison-Lowe NJ, Olsen LJ (2008) Autophagy protein 6 (ATG6) is required for pollen germination in Arabidopsis thaliana. Autophagy 4(3):339
Hepler PK, Vidali L, Cheung AY (2001) Polarized cell growth in higher plants. Annu Rev Cell Dev Biol 17:159–187
Holdaway-Clarke TL, Feijo JA, Hackett GR, Kunkel JG, Hepler PK (1997) Pollen tube growth and the intracellular cytosolic calcium gradient oscillate in phase while extracellular calcium influx is delayed. Plant Cell 9(11):1999–2010
Hwang JU, Gu Y, Lee YJ, Yang Z (2005) Oscillatory ROP GTPase activation leads the oscillatory polarized growth of pollen tubes. Mol Biol Cell 16(11):5385–5399
Ishiguro S, Kawai-Oda A, Ueda J, Nishida I, Okada K (2001) The DEFECTIVE IN ANTHER DEHISCIENCE gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13(10):2191–2209
Iwano M, Shiba H, Miwa T, Che FS, Takayama S, Nagai T, Miyawaki A, Isogai A (2004) Ca2+ dynamics in a pollen grain and papilla cell during pollination of Arabidopsis. Plant Physiol 136(3):3562–3571
Jaffe LA, Weisenseel MH, Jaffe LF (1975) Calcium accumulations within the growing tips of pollen tubes. J Cell Biol 67(2PT.1):488–492
Konrad KR, Wudick MM, Feijó JA (2011) Calcium regulation of tip growth: new genes for old mechanisms. Curr Opin Plant Biol 14(6):721–730
Landoni M, De Francesco A, Galbiati M, Tonelli C (2010) A loss-of-function mutation in calmodulin2 gene affects pollen germination in Arabidopsis thaliana. Plant Mol Biol 74(3):235–247
Lazzaro MD, Cardenas L, Bhatt AP, Justus CD, Phillips MS, Holdaway-Clarke TL, Hepler PK (2005) Calcium gradients in conifer pollen tubes; dynamic properties differ from those seen in angiosperms. J Exp Bot 56(420):2619–2628
Liu Y, Bassham DC (2012) Autophagy: pathways for self-eating in plant cells. Annu Rev Plant Biol 63:215–237
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408
Lovy-Wheeler A, Wilsen KL, Baskin TI, Hepler PK (2005) Enhanced fixation reveals the apical cortical fringe of actin filaments as a consistent feature of the pollen tube. Planta 221(1):95–104
Ma W, Smigel A, Tsai YC, Braam J, Berkowitz GA (2008) Innate immunity signaling: cytosolic Ca2+ elevation is linked to downstream nitric oxide generation through the action of calmodulin or a calmodulin-like protein. Plant Physiol 148(2):818–828
McCormack E, Braam J (2003) Calmodulins and related potential calcium sensors of Arabidopsis. New Phytol 159(3):585–598
Mouline K, Very AA, Gaymard F, Boucherez J, Pilot G, Devic M, Bouchez D, Thibaud JB, Sentenac H (2002) Pollen tube development and competitive ability are impaired by disruption of a Shaker K(+) channel in Arabidopsis. Genes Dev 16(3):339–350
Obermeyer G, Weisenseel MH (1991) Calcium channel blocker and calmodulin antagonists affect the gradient of free calcium ions in lily pollen tubes. Eur J Cell Biol 56(2):319–327
Pandey S, Zhang W, Assmann SM (2007) Roles of ion channels and transporters in guard cell signal transduction. FEBS Lett 581(12):2325–2336
Picton JM, Steer MW (1983) Membrane recycling and the control of secretory activity in pollen tubes. J Cell Sci 63:303–310
Pierson ES, Miller DD, Callaham DA, van Aken J, Hackett G, Hepler PK (1996) Tip-localized calcium entry fluctuates during pollen tube growth. Dev Biol 174(1):160–173
Qu H, Jiang X, Shi Z, Liu L, Zhang S (2012) Fast loading ester fluorescent Ca2+ and pH indicators into pollen of Pyrus pyrifolia. J Plant Res 125(1):185–195
Ren H, Xiang Y (2007) The function of actin-binding proteins in pollen tube growth. Protoplasma 230(3–4):171–182
Schiefelbein J, Galway M, Masucci J, Ford S (1993) Pollen tube and root-hair tip growth is disrupted in a mutant of Arabidopsis thaliana. Plant Physiol 103(3):979–985
Staiger CJ, Poulter NS, Henty JL, Franklin-Tong VE, Blanchoin L (2010) Regulation of actin dynamics by actin-binding proteins in pollen. J Exp Bot 61(7):1969–1986
Taylor LP, Hepler PK (1997) Pollen germination and tube growth. Annu Rev Plant Physiol Plant Mol Biol 48:461–491
Tsai YC, Delk NA, Chowdhury NI, Braam J (2007) Arabidopsis potential calcium sensors regulate nitric oxide levels and the transition to flowering. Plant Signal Behav 2(6):446–454
Tsai YC, Koo Y, Delk NA, Gehl B, Braam J (2013) Calmodulin-related CML24 interacts with ATG4b and affects autophagy progression in Arabidopsis. Plant J 73(2):325–335
Vidali L, Rounds CM, Hepler PK, Bezanilla M (2009) Lifeact-mEGFP reveals a dynamic apical F-actin network in tip growing plant cells. PLoS One 4(5):e5744
Wang YF, Fan LM, Zhang WZ, Zhang W, Wu WH (2004) Ca2+-permeable channels in the plasma membrane of Arabidopsis pollen are regulated by actin microfilaments. Plant Physiol 136(4):3892–3904
Winter J, Roepcke S, Krause S, Müller E-C, Otto A, Vingron M, Schweiger S (2008) Comparative 3′UTR analysis allows identification of regulatory clusters that drive Eph/ephrin expression in cancer cell lines. PLoS One 3(7):e2780
Wu Y, Yan J, Zhang R, Qu X, Ren S, Chen N, Huang S (2010) Arabidopsis FIMBRIN5, an actin bundling factor, is required for pollen germination and pollen tube growth. Plant Cell 22(11):3745–3763
Xiang Y, Huang X, Wang T, Zhang Y, Liu Q, Hussey PJ, Ren H (2007) ACTIN BINDING PROTEIN 29 from Lilium pollen plays an important role in dynamic actin remodeling. Plant Cell 19(6):1930–1946
Zhang W, Fan LM, Wu WH (2007) Osmo-sensitive and stretch-activated calcium-permeable channels in Vicia faba guard cells are regulated by actin dynamics. Plant Physiol 143(3):1140–1151
Zhang H, Qu X, Bao C, Khurana P, Wang Q, Xie Y, Zheng Y, Chen N, Blanchoin L, Staiger CJ, Huang S (2010) Arabidopsis VILLIN5, an actin filament bundling and severing protein, is necessary for normal pollen tube growth. Plant Cell 22(8):2749–2767
Zhao LN, Shen LK, Zhang WZ, Zhang W, Wang Y, Wu WH (2013) Ca2+-dependent protein kinase11 and 24 modulate the activity of the inward rectifying K+ channels in Arabidopsis pollen tubes. Plant Cell 25:649–661
Acknowledgments
We thank Janet Braam (Biochemistry and Cell Biology, Rice University, Texas, USA) for providing the cml24-4 mutants used in our experiments, and ABRC for supplying the cml24-T1 T-DNA insertion line. We are grateful to Timothy E. Gookin for critical reading of the manuscript. This work was supported by the Natural Science Foundation of China (31170236 and 31271506 to W.Z.) and open fund of State Key Laboratory of Plant Physiology and Biochemistry (SKLPPBKF11003 to W.Z.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, X., Wang, SS., Wang, M. et al. Arabidopsis thaliana calmodulin-like protein CML24 regulates pollen tube growth by modulating the actin cytoskeleton and controlling the cytosolic Ca2+ concentration. Plant Mol Biol 86, 225–236 (2014). https://doi.org/10.1007/s11103-014-0220-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-014-0220-y