Abstract
Purpose.
To investigate the possibility of intracellular delivery of Bak BH3 peptide using sonoporation effect by microbubble-enhanced ultrasound.
Methods.
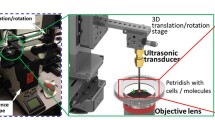
HeLa and BJAB cells were exposed to 1.696-Mhz focused ultrasound with 2% microbubble contrast agents (OPTISON®). Cell-impermeable calcein was used as an indicator for successful sonoporation, and propidium iodide staining was used for cell viability assessment. Peptides were also exposed to ultrasound with OPTISON® and analyzed with mass spectrometry for evaluation of stability under ultrasound exposure. The effect of transduced Bak BH3 peptide was evaluated by the cell viability of successfully sonoporated cells.
Results.
Bak BH3 peptides did not undergo mechanical degradation with microbubble-enhanced ultrasound exposure. With the increase of acoustic energy exposure, the sonoporation efficiency saturated both in BJAB and HeLa cells, while direct cell death rate by ultrasound exposure tended to increase. When BJAB cells were treated with 100 ϜM Bak BH3 peptides, and ultrasound exposure with ultrasound contrast agents (OPTISON®), an increased 35% cell death was confirmed. On the other hand, although HeLa cells had a similar trend, they failed to exhibit statistical significance.
Conclusions.
Our results suggest that microbubble-enhanced focused ultrasound peptide transduction is possible. Further optimization of ultrasound exposure conditions may be necessary.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ATCC:
-
American Type Culture Collection
- BH3/4:
-
Bcl-2 homology 3/4
- CW:
-
continuous wave
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- DMSO:
-
dimethyl sulfoxide
- FBS:
-
fetal bovine serum
- HIV:
-
human immunodeficiency virus
- HPLC:
-
high-pressure liquid chromatography
- LHRH:
-
luteinizing hormone releasing hormone
- MALDI-TOF:
-
matrix-assisted laser desorption ionization-time-of-flight
- PI:
-
propidium iodide
- PTD:
-
protein-transduction domain
- PW:
-
pulse wave
- RF signals:
-
radiofrequency signals
References
1. M. Narita, S. Shimizu, T. Ito, T. Chittenden, R. J. Lutz, H. Matsuda, and Y. Tsujimoto. Bax interacts with the permeability transition pore to induce permeability transition and cytochrome c release in isolated mitochondria. Proc. Natl. Acad. Sci. USA 95:14681–14686 (1998).
2. E. P. Holinger, T. Chittenden, and R. J. Lutz. Bak BH3 peptides antagonize Bcl-xL function and induce apoptosis through cytochrome c-independent activation of caspases. J. Biol. Chem. 274:13298–13304 (1999).
3. C. Moreau, P. F. Cartron, A. Hunt, K. Meflah, D. R. Green, G. Evan, F. M. Vallette, and P. Juin. Minimal BH3 peptides promote cell death by antagonizing anti-apoptotic proteins. J. Biol. Chem. 278:19426–19435 (2003).
4. A. D. Schimmer, D. W. Hedley, S. Chow, N. A. Pham, A. Chakrabartty, D. Bouchard, T. W. Mak, M. R. Trus, and M. D. Minden. The BH3 domain of BAD fused to the Antennapedia peptide induces apoptosis via its alpha helical structure and independent of Bcl-2. Cell Death Differ. 8:725–733 (2001).
5. H. L. Vieira, P. Boya, I. Cohen, C. El Hamel, D. Haouzi, S. Druillenec, A. S. Belzacq, C. Brenner, B. Roques, and G. Kroemer. Cell permeable BH3-peptides overcome the cytoprotective effect of Bcl-2 and Bcl-X(L). Oncogene 21:1963–1977 (2002).
6. S. S. Dharap and T. Minko. Targeted proapoptotic LHRH-BH3 peptide. Pharm. Res. 20:889–896 (2003).
7. S. S. Dharap, B. Qiu, G. C. Williams, P. Sinko, S. Stein, and T. Minko. Molecular targeting of drug delivery systems to ovarian cancer by BH3 and LHRH peptides. J. Control. Rel. 91:61–73 (2003).
8. Y. Taniyama, K. Tachibana, K. Hiraoka, T. Namba, K. Yamasaki, N. Hashiya, M. Aoki, T. Ogihara, K. Yasufumi, and R. Morishita. Local delivery of plasmid DNA into rat carotid artery using ultrasound. Circulation 105:1233–1239 (2002).
9. P. E. Huber, M. J. Mann, L. G. Melo, A. Ehsan, D. Kong, L. Zhang, M. Rezvani, P. Peschke, F. Jolesz, V. J. Dzau, and K. Hynynen. Focused ultrasound (HIFU) induces localized enhancement of reporter gene expression in rabbit carotid artery. Gene Ther. 10:1600–1607 (2003).
10. M. Shimamura, N. Sato, Y. Taniyama, S. Yamamoto, M. Endoh, H. Kurinami, M. Aoki, T. Ogihara, Y. Kaneda, and R. Morishita. Development of efficient plasmid DNA transfer into adult rat central nervous system using microbubble-enhanced ultrasound. Gene Ther. 11:1532–1539 (2004).
11. S. Shimizu, A. Konishi, T. Kodama, and Y. Tsujimoto. BH4 domain of antiapoptotic Bcl-2 family members closes voltage-dependent anion channel and inhibits apoptotic mitochondrial changes and cell death. Proc. Natl. Acad. Sci. USA 97:3100–3105 (2000).
12. R. Sugioka, S. Shimizu, T. Funatsu, H. Tamagawa, Y. Sawa, T. Kawakami, and Y. Tsujimoto. BH4-domain peptide from Bcl-xL exerts anti-apoptotic activity in vivo. Oncogene 22:8432–8440 (2003).
13. D. Klein, M. M. Ribeiro, V. Mendoza, S. Jayaraman, N. S. Kenyon, A. Pileggi, R. D. Molano, L. Inverardi, C. Ricordi, and R. L. Pastori. Delivery of Bcl-XL or its BH4 domain by protein transduction inhibits apoptosis in human islets. Biochem. Biophys. Res. Commun. 323:473–478 (2004).
14. M. Becker-Hapak, S. S. McAllister, and S. F. Dowdy. TAT-mediated protein transduction into mammalian cells. Methods 24:247–256 (2001).
15. D. Derossi, A. H. Joliot, G. Chassaing, and A. Prochiantz. The third helix of the Antennapedia homeodomain translocates through biological membranes. J. Biol. Chem. 269:10444–10450 (1994).
16. L. D. Walensky, A. L. Kung, I. Escher, T. J. Malia, S. Barbuto, R. D. Wright, G. Wagner, G. L. Verdine, and S. J. Korsmeyer. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 305:1466–1470 (2004).
17. C. M. Tempany, E. A. Stewart, N. McDannold, B. J. Quade, F. A. Jolesz, and K. Hynynen. MR imaging-guided focused ultrasound surgery of uterine leiomyomas: a feasibility study. Radiology 226:897–905 (2003).
18. K. Hynynen, G. T. Clement, N. McDannold, N. Vykhodtseva, R. King, P. J. White, S. Vitek, and F. A. Jolesz. 500-element ultrasound phased array system for noninvasive focal surgery of the brain: a preliminary rabbit study with ex vivo human skulls. Magn. Reson. Med. 52:100–107 (2004).
19. K. Hynynen, N. McDannold, N. Vykhodtseva, and F. A. Jolesz. Noninvasive MR imaging-guided focal opening of the blood-brain barrier in rabbits. Radiology 220:640–646 (2001).
20. M. Ward, J. Wu, and J. F. Chiu. Experimental study of the effects of Optison concentration on sonoporation in vitro. Ultrasound Med. Biol. 26:1169–1175 (2000).
21. H. R. Guzman, A. J. McNamara, D. X. Nguyen, and M. R. Prausnitz. Bioeffects caused by changes in acoustic cavitation bubble density and cell concentration: a unified explanation based on cell-to-bubble ratio and blast radius. Ultrasound Med. Biol. 29:1211–1222 (2003).
22. C. X. Deng, F. Sieling, H. Pan, and J. Cui. Ultrasound-induced cell membrane porosity. Ultrasound Med. Biol. 30:519–526 (2004).
23. H. R. Guzman, D. X. Nguyen, A. J. McNamara, and M. R. Prausnitz. Equilibrium loading of cells with macromolecules by ultrasound: effects of molecular size and acoustic energy. J. Pharm. Sci. 91:1693–1701 (2002).
24. H. R. Guzman, D. X. Nguyen, S. Khan, and M. R. Prausnitz. Ultrasound-mediated disruption of cell membranes. II. Heterogeneous effects on cells. J. Acoust. Soc. Am. 110:597–606 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kinoshita, M., Hynynen, K. Intracellular Delivery of Bak BH3 Peptide by Microbubble-Enhanced Ultrasound. Pharm Res 22, 716–720 (2005). https://doi.org/10.1007/s11095-005-2586-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-2586-7