Abstract
Cyanobacteria are photosynthetic organisms that have been considered for space applications, such as oxygen production in bioregenerative life support systems, and can be used as a model organism for understanding microbial survival in space. Akinetes are resting-state cells of cyanobacteria that are produced by certain genera of heterocystous cyanobacteria to survive extreme environmental conditions. Although they are similar in nature to endospores, there have been no investigations into the survival of akinetes in extraterrestrial environments. The aim of this work was to examine the survival of akinetes from Anabaena cylindrica in simulated extraterrestrial conditions and in Low Earth Orbit (LEO). Akinetes were dried onto limestone rocks and sent into LEO for 10 days on the ESA Biopan VI. In ground-based experiments, the rocks were exposed to periods of desiccation, vacuum (0.7 × 10−3 kPa), temperature extremes (−80 to 80°C), Mars conditions (−27°C, 0.8 kPa, CO2) and UV radiation (325–400 nm). A proportion of the akinete population was able to survive a period of 10 days in LEO and 28 days in Mars simulated conditions, when the rocks were not subjected to UV radiation. Furthermore, the akinetes were able to survive 28 days of exposure to desiccation and low temperature with high viability remaining. Yet long periods of vacuum and high temperature were lethal to the akinetes. This work shows that akinetes are extreme-tolerating states of cyanobacteria that have a practical use in space applications and yield new insight into the survival of microbial resting-state cells in space conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cyanobacteria are photosynthetic organisms that play a significant role in the production of organic matter and oxygen (Whitton and Potts 2000). They are widely distributed and have adapted to live in some of the most extreme environments on Earth, varying from the hyper-arid, cold Antarctic Dry Valleys to the hot, dry Atacama Desert (Friedmann 1980; Wierzchos et al. 2006). Furthermore, under certain environmental stresses various genera of heterocystous cyanobacteria differentiate morphologically and ultra-structurally into resting-state cells for survival (Seckbach 2007).
Akinetes are resting-state cells of cyanobacteria whose formation is induced by a number of environmental factors including low temperature, desiccation, elevated levels of salt and iron depletion (Adams and Duggan 1999). In nature, akinetes are believed to play an important role in perennating, allowing the producer strain to survive cold winters, and to withstand adverse environmental stresses (Sukenik et al. 2007). Experimental work demonstrated that in the winter months, the number of akinetes is significantly higher than in summer (McKnight et al. 2000). Under optimised conditions, the akinetes germinate and vegetative-state cells are produced. The cyanobacteria can remain as akinetes for long periods of time without losing viability. For example, the akinetes produced by Anabaena cylindrica were found to germinate after five years of desiccation (Yamamoto 1975).
Akinetes are specialised spore-like cells (Adams and Duggan 1999). Although they are not heat resistant and do not structurally resemble other spore-like cells, they are resistant to cold and desiccation, and germinate under ideal conditions similar to endospores of Gram-positive bacteria (Adams and Duggan 1999; Li and McClane 2006; Setlow 1995). Endospores, in particular, the endospores of Bacillus subtilis, have been extensively studied. Physiological experiments have demonstrated that they can survive a large array of extreme conditions. These include heat, desiccation, vacuum, UV radiation (325 and 400 nm), gamma radiation and chemical toxins (Nicholson et al. 2000). Furthermore, long term space experiments demonstrated that endospores of B. subtilis are able to survive the hostile environment of space for 2,107 days, provided that the spores are protected from UV radiation (>110 nm) (Horneck et al. 1994). Yet there has been no investigation into the survival of akinetes in extraterrestrial environments.
Cyanobacteria have been recommended for a number of space applications. These include oxygen production in bioregenerative life support systems, such as MELiSSA (Lehto et al. 2006; Hendrickx and Mergeay 2007); dust control, and soil conditioning on the moon and Mars (Liu et al. 2008). Whilst extremophilic cyanobacteria, such as Chroococcidiopsis, are potential candidates they are not ideal due to their slow growth rates (generation time of 16 days) (Billi and Caiola 1996). Instead, a faster growing cyanobacterium that generates sufficient oxygen to support a life support system and produces spore-like cells to survive storage and transportation would be an ideal candidate.
In this study, we investigated the survival of akinetes from A. cylindrica in simulated extraterrestrial conditions and in LEO. A. cylindrica was selected, as it is easily grown under laboratory conditions, has a generation time of 1 day and can produce akinetes without difficulty (Adams and Carr 1981; Olli et al. 2005; Yamamoto 1975). These are all important prerequisites for use in space applications. We hypothesised that like endospores, akinetes would be able to survive the adverse conditions associated with these extreme environments. This study raises the question of the potential survival and utilisation of akinetes in extraterrestrial environments.
Material and Methods
Growth Conditions
Akinetes were induced from cultures of A. cylindrica (PCC 6309). The organism was grown in a modified BG-11 medium that contained the following (per g l−1): 1.5 of NaNO3, 0.0305 of K2HPO4.3H2O, 0.036 of CaCl2.2H2O, 0.006 of citric acid, 0.001 of EDTA (disodium salt), 0.02 of Na2CO3, 0.075 of MgSO4.7H2O, 0.006 of ferric ammonium citrate and 1 ml l−1 of A5 trace metal mix and Co [(per g l−1): 2.86 of H3BO3, 1.81 of MnCl2, 0.222 of ZnS04.7H2O, 0.390 of Na2MoO4.2H2O, 0.079 of CuSO4.5H2O, 0.0494 of Co(NO3)2.6H2O)]. All of the reagents, except the MgSO4.7H2O and the ferric ammonium, were added to dd H2O, and the pH was adjusted to 7.4. After the medium was autoclaved (20 min at 121°C), filtered sterilised MgSO4.7H2O and ferric ammonium were added (this did not alter the pH). The cultures were grown at 25°C, under natural sunlight and day/night cycle. The akinetes were induced by transferring log phase cells into an iron limited (no iron) BG-11 medium, followed by three washes in the same medium. After 35 days of growth, the akinetes were harvested by allowing them to settle to the bottom of the flask. They were then washed and resuspended in dd H2O. The akinetes were stored in a refrigerator at 4°C until required (Fig. 1). For each experiment, we added 100 µl of akinetes (30 × 103 ml−1) to the surface of 1cm2 blocks of sandstone (from coastal limestone/sandstone cliffs in Beer, Devon, UK) and allowed them to dry to the surface under laboratory conditions (20°C). All chemicals were supplied by Sigma-Aldrich, Gillingham, UK, unless otherwise stated.
Low Earth Orbit Experiments
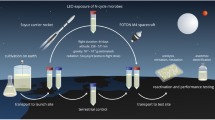
The study of the effects of LEO was carried out as part of the ESA funded Biopan VI mission (Demets et al. 2005). The samples were launched into LEO by a Soyuz launch vehicle (Russian Space Agency). The Biopan, which is a space exposure facility, was installed on the external surface of the Foton capsule and opened in orbit to expose the samples to the combined environmental conditions of LEO for 10 days, as shown in Fig. 2A. The rock samples were placed on two sample plates. The upper plate allowed for exposure to space conditions including, extraterrestrial solar UV radiation (>170 nm), and visible light. Cut-off filters were used to expose samples to selected wavelengths ranges (>110, >200, >290 and >400 nm).The lower layer enabled the samples to be exposed to space conditions without UV or visible radiation (Fig. 2B). An identical set of samples were kept for the same period in the laboratory at ambient conditions (21°C, atmospheric pressure) and in darkness to serve as ground-based controls.
A The inside lid of the Biopan VI module, which was opened in orbit to expose the biological samples to LEO conditions. The Biopan contained a temperature sensor AD590 and thermoluminescence detector Litho-Dose, to monitor the conditions. The Biopan contained 2 plates; a top (level-1) and a bottom (level-2) plate where the samples were located. B The cells of the top plate were covered by optical long-pass filters with the following characteristics: (1) MgF2, which is transparent for extraterrestrial solar UV radiation of λ > 110 nm; (2) SQ0 synthetic quartz transmitting solar UV of λ > 200 nm, thereby simulating the UV radiation climate on the surface of Mars; (3) long-pass filter for λ > 290 nm to simulate the terrestrial UV radiation climate (as a control) and (4) for λ > 400 nm thereby cutting off all solar UV radiation. C The Mars Simulated Chamber. The samples were placed inside the chamber, before tightly securing the lid and adding the CO2 gas slowly. The pressure was adjusted until 0.8 kPa ± 0.2 by means of an Edward rotary pump
After 10 days of exposure the samples were returned to Earth, transported to the laboratory at the Open University, and split into half under aseptic conditions to prevent laboratory contamination. The two halves were incubated separately in 15 ml sterilised glass bottles containing 5 ml of BG-11 medium. The samples were incubated in natural day/light cycles at 25°C. After 70 days, the surfaces of the rocks were scraped with a sterile blade and inoculated into liquid BG-11 media and spread onto 1 % agar (Bactereological Agar No. 1, Oxoid, Basingstoke) plates of the same composition. The samples were monitored for growth.
Identification
Identification was confirmed by microscopic and molecular biological methods. Colonies were examined under 1,000 times magnification (bright field microscopy) on a Leica DMRP microscope, and by 16S rDNA analysis. For 16S rDNA analysis a region of the gene was amplified with cyanobacteria specific primers: CYA106F (5′-CGG ACG GGT GAG TAA CGC GTG A-3′) and CYA781R [an equimolar mixture of CYA781R(a) (5′-GAC TAC TGG GGT ATC TAA TCC CAT T-3) and CYA781R(b) (5′-GAC TAC AGG GGT ATC TAA TCC CTT T-3)] (Nubel et al. 1997). The reaction mixture contained: a colony; 200 µM dNTP; 1 µM primers; 5 U of Taq DNA polymerase; 1× PCR Buffer (20 mM Tris–HCl (pH 8.4), 50 mM KCl) and 2.5 mM MgCl (Invitrogen, Paisley, UK). Amplification consisted of incubation at 94°C for 15 min: this was followed by 35 cycles of 1 min at 94°C, 1 min at 60°C and a 1 min extension at 72°C; with a final extension of 5 min at 72°C. Sequences were examined by BlastX using the GenBank database (http://www.ncbi.nlm.nih.gov/).
Ground-Based Experiments
Experimental Set-up
Experiments were conducted in the laboratory to simulate extraterrestrial conditions. Each of the ground-based experiments described below consisted of triplicate samples, with replicas stored under ambient conditions in darkness, as controls. Samples, including the controls, were collected aseptically after 1, 4, 7, 21 and 28 days (unless stated differently below). All of the samples were incubated in 15 ml sterile glass bottles containing 5 ml of BG-11 medium (unless stated differently below), and incubated under a natural day/light cycles at 25°C. The cultures were monitored, using a Leica DMRP microscope, and the presence of vegetative cells in the media was defined as the end of the lag phase. The cultures were incubated for a further 10 days before the filamentous cells were diluted by tenfold and counted using a 1,000 times magnification on a Leica DMRP microscope. The total number of vegetative-state cells, per ml, was counted for both the control and exposed samples. The percentage of vegetative-state cells in the exposed samples compared to the corresponding control was also calculated to determine the effect of exposure on the viability of the akinetes. In some cases, no growth was observed after a number of weeks. In these cases we scraped the surface of the rocks with a sterile scalpel, and spread the material onto 1% agar BG-11 plates, to determine if any survival had occurred, since agar plates can be used to determine more clearly whether single colonies would grow compared to liquid cultures.
Mars Simulation, Vacuum, and UV Radiation
To determine the effects of Mars conditions, we used a Mars Simulation Chamber (MSC) (Fig. 2C). The MSC is a stainless steel cylinder chamber measuring 15 cm in diameter and 30 cm in height. Environmental conditions within the MSC were all monitored and controlled independently. This included the pressure (0.8 kPa ± 0.2), the gas composition [99% pure CO2 (BOC, Manchester, UK)], and the temperature (−28 ± 1°C). Since the temperature on Mars can vary between 25°C and −143°C, depending on regional and seasonal variations, we carried out these experiments at −28°C. This was due to the Viking Landers data indicating that in the northern hemisphere the temperature rarely rises above −28°C (Hess et al. 1977; Thomas and Schimel 1991). The rocks were positioned inside the MSC within a sterilised Petri dish, and the lid was removed when the temperature dropped to −28°C. To ensure that the temperature was constant, the MSC was placed within a low temperature controlled environment, for the duration of the experiment.
UV radiation resistance experiments (UVA spectrum) were carried out in the MSC with a xenon-arc lamp (Muller 450W lamp, model XBO450W). The light was transferred into the chamber via a fibre-optical cable. The total flux (i.e. the total dose) from 325–400 nm was 54.6 W/m2. The experiment was carried out under Martian simulated conditions, as described above (the temperature of the samples was not affected by the lamp). The samples were exposed for 1, 5, 10, 30, 45 and 60 min. The rocks were then incubated in BG-11, as previously described.
We investigated the effect of vacuum by lowering the pressure inside the MSC to 0.7 × 10−3 kPa by means of an Edward rotary pump. The pressure was maintained at 0.7 × 10−3 ± 0.01 for the duration of the experiment. At the end of the experiment, the pressure was slowly returned to atmospheric pressure, over a 5 min period. The rocks were immediately placed in BG-11 media to rehydrate the akinetes.
Desiccation, Temperature, and pH
The effect of desiccation was measured at room temperature in a desiccator containing silica gel beads. The silica gel beads were heated overnight at 80°C to eliminate moisture, prior to use.
To determine the effect of temperature on the survival of the akinetes, rocks were incubated at −80°C in an ultra deep freeze (MDF-U73V, SANYO), −20°C in a freezer (ISU24, Lec), 4°C in a fridge (ISR45, Lec) and at 30°C, 40°C, 50°C, 60°C, 70°C and 80°C in a heated incubator (SI-300R, Fischer Scientific), for 1, 4, 7, 21 and 28 days.
The effect of short-term pH exposure was measured in sterilised potassium phosphate buffer, over a pH range of 4–11. Twenty milliliters of each of the buffers were poured into 50 ml sterilised glass bottles. The rocks were placed aseptically in to the bottles and exposed for 1, 4, 7, 21 and 28 days. After this time, the rocks were aseptically removed and incubated in 50 ml of BG-11 media. A large volume of media was used to ensure that the pH of the incubation media was maintained at near neutral.
Germination on Volcanic Rocks
Germination experiments were conducted with volcanic rocks to determine whether these materials would provide sufficient elements to allow akinetes to germinate. Volcanic rocks are found on both Mars and the moon. Two types of end member volcanic rocks were used—basalt and rhyolite. The basalt used in this experiment was collected from near the Torfajökull volcano in Iceland (64°5.47 N, 19°3.46 W) and has a typical basaltic composition (Table 1). Six basalt rocks were crushed and the powder was pooled. In contrast, the rhyolite is high in SiO2, Na2O and K2O, but is depleted in MgO, CaO compared to the basalt (Table 1). The rhyolite was collected from outcrops at 64°2.01 N, 19°7.75 W. Four rhyolite rocks were crushed and the powder was pooled. Whole-rock major element compositions for both the basalt and rhyolite were obtained using an ARL 8420+ dual goniometer wavelength-dispersive XRF spectrometer at the Open University, UK. XRF analysis was carried out on glass discs prepared by fusing one part of a powdered sample with five parts of FluXana flux (20% Lithium tetraborate/80% lithium metaborate mix). Prior to use, the grounded rocks were autoclaved at 121°C for 20 min. One gram of rock (basalt or rhyolite) was added to 5 ml of autoclaved dd H2O. One hundred µl of akinetes were added to each of the solutions, and incubated under natural day/light cycles at 25°C.
Results
Low Earth Orbit Experiments
The environmental parameters during the LEO experiment were as followed: vacuum 0.133 × 10−6 kPa; temperature −22 to +30°C; microgravity 10−4 g and solar radiation (>170 nm). The effect of LEO on the survival of the akinetes was evaluated by their ability to germinate on their return to Earth. The lag phases for the control samples varied between 25 and 32 days; however, no growth was detected on the rocks exposed LEO. Therefore, after 70 days of incubation, the rocks were examined using a dissection microscope, but no growth was observed. The surface of the rocks was scraped, and plated onto BG-11 agar. The results are shown in Table 2.
After 31 days, a green filamentous colony started to grow from a small fragment of rock that had been exposed to LEO conditions and no UV radiation (>200 nm position on the second platform), as shown in Fig. 3. After another 14 d, a green filamentous colony grew from the rock exposed to no UV radiation (>400 nm position on the second platform).
The morphology of the colonies exhibited long filamentous cells, which is characteristic of A. cylindrica. We confirmed the identity of the cyanobacterium by sequencing the PCR product with the cyanobacteria specific 16S rDNA. BlastX analysis of the sequence confirmed that the cyanobacterium showed 100% identity to that of A. cylindrica.
Ground-Based Experiments
Mars simulation, Vacuum, and UV Radiation
The conditions during the simulated Mars experiment stayed constant with the pressure at 0.8 kPa ± 0.2 and the temperature at –28 ± 1°C. Inoculation of the rocks in BG-11, after exposure demonstrated that the akinetes were able to survive the simulated Mars environment for the period examined (Table 3). After 28 days the percentage of vegetative-state cells was 35.3 ± 2.5% (the percentage with the standard deviation) and the lag phase for germination was 25.3 ± 1.5 days. Although a proportion of the akinetes were able to survive the 28 days of exposure, the viability of the akinetes was found to decrease as the exposure time increased. This was demonstrated by a reduction in percentage of vegetative-state cells and an increase in the lag phase.
Exposure to UV radiation of between 325 and 400 nm caused complete loss of viability. The akinetes were unable to germinate after 1 min of exposure, even after 100 days of incubation under favourable growth conditions.
Vacuum (0.7×10−3 kPa) had a lethal effect after 14 days of exposure. However, after 7 days of exposure some akinetes survived; the percentage of vegetative-state cells was only 22.3 ± 1.5 % and the lag phase for germination was 25 ± 1 days.
Desiccation, Temperature, and pH
Akinetes were able to germinate after 28 days of exposure to desiccation. As with the Mars simulated experiment, the exposure time influenced the akinetes survival. After 1 day of exposure the percentage of vegetative-state cells was 98.3 ± 5.7% compared to 29.3 ± 2.5% after 28 days. The lag phase for germination increased from 15 ± 2 to 20 ± 1 days respectively, as shown in Table 3.
The akinetes were placed in a potassium phosphate buffer, over a pH range of 4 to 11, to determine the effect of pH on viability. Exposure to both extreme acid and alkaline conditions had a detrimental effect on the akinetes, as shown in Table 4. They were unable to survive exposure to pH 4 and could only survive 1 day of exposure to pH 5 (lag phase was 23.0 ± 1.0 and percentage of vegetative-state cells was 56.0 ± 5%). In addition, the akinetes were unable to survive exposure to pH 11 and only survived 1 day of exposure to pH 10 (lag phase was 33.3 ± 2.0, percentage of vegetative-state cells was 25.0 ± 6.0%). The maximum viability was retained at pH 7 and 8, after 28 days the lag phase for growth was 11.6 ± 1.5 (for pH 7) and 11.0 ± 1.5 (for pH 8) and the percentage of vegetative-state cells was 96.4 ± 5.0% (for pH 7) and 77.0 ± 1.0 % (for pH 8). When BG-11 plates (pH range 4 to 11) were inoculated with akinetes, germination was only detected at pH 6, 7 and 8 (data not shown).
The effect of temperature on the akinetes was determined by exposing the rocks to temperatures of −80°C to 80°C. The akinetes were able to survive exposure to low temperatures with high viability; however, high temperatures had a damaging effect on growth. For example, at −80°C, after 28 days, the percentage of vegetative-state cells was 22.0 ± 2%, and the lag phase for germination was 27 ± 2.6 days. In contrast, exposing the akinetes to a temperature of 40°C, or above, affected the viability of the akinetes. The akinetes were unable to survive an exposure time of 14 days at 40°C. At 60 and 80°C, after 1 day, viability was reduced to 25.5% or less. At these temperatures no akinetes survived longer than 1day shown in Table 5.
Germination on Volcanic Rocks
In addition to the exposure experiments, we carried out germination experiments with basalt and rhyolite as growth substrates. The akinetes were able to use either types of rock as a substrate (they were unable to germinate in only dd H2O). Germination was observed by a green layer forming on top of the crushed rock, as shown in Fig. 4. This was morphologically identified as A. cylindrica. The lag phases for germination in the basalt and rhyolite solutions were 32 ± 3 and 41 ± 5 days respectively.
Discussion
In this communication we carried out the first investigation into akinetes survival in extreme conditions associated with extraterrestrial environments. Rocks were inoculated with akinetes from A. cylindrica prior to exposure to LEO as part of the ESA funded Biopan VI experiment. Viability was lowered, but some akinetes survived exposure to LEO, when protected from solar radiation (>170 nm); however, the lag period was significantly longer compared to the control samples. This may be due to the damage caused by the combined environmental conditions experienced in LEO, which include vacuum (0.133 × 10−6 kPa), temperature fluctuations, and elevated ionising radiation. Previous laboratory experiments have demonstrated that akinetes require a certain internal metabolic energy and/or nutrient level to be reached before germination can occur (Karlsson 1999).
We also investigated the effect of simulated Mars conditions on the survival of the akinetes. This was to determine whether akinetes could be used as a form of cyanobacteria storage under ambient Martian conditions. The MSC experiments demonstrated that the akinetes were able to survive exposure to 0.8 kPa and −28°C for 28 days, without a detrimental effect. We showed in further experiments that temperature plays a crucial role in akinete viability. When the exposure temperature was lowered to −80°C, the akinetes were still resistant, but they were susceptible to 80°C. Previous studies have indicated that akinetes are not heat resistant, unlike endospores of Bacillus, but are resistant to low temperatures (Sutherland et al. 1979). On Mars, the temperature rarely rises above 25°C, but can drop as low as −143°C, depending on seasonal and regional variation (de Pater and Lissauer 2001). Our experiments suggest that akinetes would be able to survive the temperature variations on Mars.
The surface of Mars is also an arid environment, similar to terrestrial deserts. On Earth, cyanobacteria dominate desiccated environments, such as intertidal marine mats and terrestrial crusts (Potts 1999). We investigated the effect of desiccation on the survival of the akinetes. After 28 days of desiccation, the akinetes were able to germinate. Although our experiments were limited to 28 days; previous experiments by Yamamoto et al. showed that desiccated akinetes of A. cylindrica retained their capacity to germinate for at least 5 years in the dark, compared to vegetative-state cells dried under the same conditions that failed to grow after 15 days (Yamamoto 1975).
Although akinetes are highly resistant to desiccation, they are susceptible to vacuum. The susceptibility may be due to a number of reasons, including severe loss of the water content, and the time it takes to dehydrate the cells. Vacuum exposure may cause complete desiccation, which would not be the case in the non-vacuum desiccation experiment. In addition, dehydration is reached quickly in the vacuum experiment. Experimental work with cells of E. coli has demonstrated that the drying time has an effect on viability. The longer the drying time the higher the viability that is retained (Sleesman and Leben 1976).
Although the akinetes were only exposed to wavelengths in the UVA spectrum and not in the UVB or UVC regions, which are also associated with the surface of Mars (Kuhn and Atreya 1979), the exposure was damaging. Most biological action spectra, which describe the UV damage at different wavelengths, peak sharply in the UVC (200–280 nm) and UVB (280–315 nm) regions. Therefore, the lack of resistance, when exposed to the UVA spectrum, suggests that the akinetes would not survive the UV radiation associated with the surface of Mars. However, it has been hypothesised that on Mars, windblown dust can attenuate solar UV radiation (>200 nm), resulting with partial or complete shielding of viable bacteria (Osman et al. 2008). For example, endospores of Bacillus subtilis are killed within minutes of exposure to UV radiation; however, the addition of a few microns of dust enables the endospores to survive longer periods (Mancinelli and Klovstad 2000). Experimental work by Cockell et al., has also suggested that under 1 mm of Mars soil, vegetative-state cells of Chroococcidiopsis-like organisms could cope with the intense UV radiation, so it would not be a limiting factor for life (Cockell et al. 2005). So although UV radiation would rapidly kill akinetes, any type of shielding would allow for their survival.
The ability of some akinetes to survive both in LEO and Mars simulated environments, when protected from UV radiation, make them ideal candidates for space applications. Fast growing cyanobacteria have been suggested for use in bioregenerative life support systems and soil conditioning on Mars (Liu et al. 2008; Hendrickx and Mergeay 2007), with desiccated vegetative-state cells as a method for transportation and storage. However, the findings in this paper suggest that akinetes are robust, and subsequently would be ideal for long-term storage of cyanobacteria. On Mars, akinetes could be stored in containers that are only covered by a UV shield. Whilst on the moon, the akinetes would have to be stored in a pressurised container, to protect them from the effects of vacuum. Our experiments show that when they are required in these systems they could be germinated on locally available volcanic rocks, such as basalts. This would eliminate the requirement for specialised storage facilities and complex growth media. The survival of akinetes in ambient Martian conditions shows that they could also be used as a means of storage and emergency supply of cyanobacteria in bioregenerative life systems. If there was a malfunction in the system resulting in decompression, the akinetes would survive and able to reinitiate growth, providing redundancy.
The detrimental conditions of vacuum and UV radiation on akinetes have implications for their long-term survival not just on the moon, but in interplanetary space. One question to which these results have relevance is lithopanspermia—the concept of the natural transfer of life from one planet to another in rocks ejected from planetary surfaces in impact events. In artificial meteorite atmospheric entry experiments Cockell et al. showed that active photosynthetic organisms, because of their requirement for sufficient light for photosynthesis, are likely to be killed during the ablation and heating of the surface layers of meteorites during atmospheric entry (Cockell et al. 2007). One means by which they suggested these organisms could survive is as metabolically inactive states, such as akinetes, deep within a rock. Our results show that although these resting-states can confer resistance to some environmental extremes, not all akinetes have vacuum tolerance that would be required for long-term survival in space.
In conclusion, akinetes are spore-like cells that are extreme-tolerant resting-state cells of cyanobacteria. Although they are structurally different to endospores of Bacillus, they are able to survive short periods in LEO, and exposure to solar and UV radiation has a lethal effect similar to that observed in endospores (Horneck et al. 1994). Unlike endospores, our experimental evidence suggests that akinetes are unable to survive long-term exposure to vacuum (Horneck 1993). However, they may be of practical use in space applications that require fast growing, non-extremophilic, photosynthetic microorganisms. Finally, this work furthers our understanding of the survival of resting-state microorganisms in space conditions.
References
Adams DG, Carr NG (1981) Heterocyst differentiation and cell division in the cyanobacterium Anabaena Cylindrica: effect of high light intensity. J Cell Sci 49:341–352
Adams DG, Duggan PS (1999) Heterocyst and akinete differentiation in cyanobacteria. New Phytologist 144:3–33
Billi D, Caiola MG (1996) Effects of nitrogen limitation and starvation on Chroococcidiopsis sp. (Chroococcales). New Phytologist 133:563–5715
Cockell CS, Schuerger AC, Billi D, Friedmann EI, Panitz C (2005) Effects of a simulated Martian UV flux on the cyanobacterium, Chroococcidiopsis sp. 029. Astrobiology 5:127–140
Cockell CS, Brack A, Wynn-Williams DD, Baglioni P, Brandstatter F, Demets R, Edwards HG, Gronstal AL, Kurat G, Lee P, Osinski GR, Pearce DA, Pillinger JM, Roten CA, Sancisi-Frey S (2007) Interplanetary transfer of photosynthesis: an experimental demonstration of a selective dispersal filter in planetary island biogeography. Astrobiology 7:1–9
De Pater I, Lissauer JJ (2001) Planetary sciences. Cambridge University Press, Cambridge
Demets R, Schulte W, Baglioni P (2005) The past, present and future of BIOPAN. Adv Space Res 36:311–316
Friedmann EI (1980) Endolithic microbial life in hot and cold deserts. Orig Life 10:223–235
Hendrickx L, Mergeay M (2007) From the deep sea to the stars: human life support through minimal communities. Curr Opin Microbiol 10:231–237
Hess SL, Henry RM, Leovy CB, Ryan JA, Tillman JE (1977) Meteorological results from surface of Mars—Viking 1 and 2. Trans Am Geophys Union 58:827–838
Horneck G (1993) Responses of Bacillus subtilis spores to space environment: results from experiments in space. Orig Life Evol Biosph 23:37–52
Horneck G, Bucker H, Reitz G (1994) Long-term survival of bacterial spores in space. Adv Space Res 14:41–45
Karlsson I (1999) On the germination of the akinete-forming cyanobacterium Gloeotrichia echinulata in Lake Erken, Sweden. Arch Hydrobiol Algolog Stud 94:175–180
Kuhn WR, Atreya SK (1979) Solar radiation incident on the Martian surface. J Mol Evol 14:57–64
Lehto KA, Lehto HJ, Kanervo EA (2006) Suitability of different photosynthetic organisms for an extraterrestrial biological life support system. Res Microbiol 157:69–76
Li JH, McClane BA (2006) Further comparison of temperature effects on growth and survival of Clostridium perfringens type A isolates carrying a chromosomal or plasmid-borne enterotoxin gene. Appl Environ Microbiol 72:4561–4568
Liu Y, Cockell CS, Wang G, Hu C, Chen L, De Philippis R (2008) Control of lunar and Martian dust-experimental insights from artificial and natural cyanobacterial and algal crusts in the desert of Inner Mongolia, China. Astrobiology 8:75–86
Mancinelli RL, Klovstad M (2000) Martian soil and UV radiation; microbial viability assessment on spacecraft surfaces. Planet Space Sci 48:1093–1097
McKnight DM, Howes BL, Taylor CD, Goehringer DD (2000) Phytoplankton dynamics in a stably stratified Antarctic lake during winter darkness. J Phycol 36:852–861
Nicholson WL, Munakata N, Horneck G, Melosh HJ, Setlow P (2000) Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol Mol Biol Rev 64:548–572
Nubel U, Garcia-Pichel F, Muyzer G (1997) PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl Environ Microbiol 63:3327–3332
Olli K, Kangro K, Kabel M (2005) Akinete production of Anabaena lemmermannii and A. Cylindrica (Cyanophyceae) in natural populations of N- and P-limited coastal mesocosms. J Phycol 41:1094–1098
Osman S, Peeters Z, La Duc MT, Mancinelli R, Ehrenfreund P, Venkateswaran K (2008) Effect of shadowing on survival of bacteria under conditions simulating the Martian atmosphere and UV radiation. Appl Environ Microbiol 74:959–970
Potts M (1999) Mechanisms of desiccation tolerance in cyanobacteria. Eur J Phycol 34:319–328
Seckbach J (2007) Algae and cyanobacteria in extreme environments. Springer, Berlin
Setlow P (1995) Mechanisms for the prevention of damage to DNA in spores of Bacillus species. Annu Rev Microbiol 49:29–54
Sleesman JP, Leben C (1976) Bacterial desiccation: effects of temperature, relative humidity, and culture age on survival. Phytopathology 66:1334–1338
Sukenik A, Beardall J, Hadas O (2007) Photosynthetic characterization of developing and mature akinetes of Aphanizomenon ovalisporum (Cyanoprokaryota). J Phycol 43:780–788
Sutherland JM, Herdman M, Stewart WDP (1979) Akinetes of the cyanobacterium Nostoc PCC7524: macromolecular composition, structure and control of differentiation. J Gen Microbiol 115:273–287
Thomas DJ, Schimel JP (1991) Mars after the Viking missions: is life still possible? Icarus 91:199–206
Whitton BA, Potts M (2000) The ecology of cyanobacteria—their diversity in time and space. Kluwer, Dordrecht
Wierzchos J, Ascaso C, McKay CP (2006) Endolithic cyanobacteria in halite rocks from the hyperarid core of the Atacama Desert. Astrobiology 6:415–422
Yamamoto Y (1975) Effect of desiccation on the germination of akinetes of Anabaena cylindrica. Planet Cell Physiol 16:749–752
Acknowledgments
This work was supported by the STFC grant (PP/E001408/1). We would like to thank the European Space Agency for the flight opportunity, and the Russian Space Agency for access to the launch vehicle and facilities. Finally we would like to thank Dr Manish Patel for his help with the xenon light, and CEPSAR for the use of the Mars Simulated Chamber.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Olsson-Francis, K., de la Torre, R., Towner, M.C. et al. Survival of Akinetes (Resting-State Cells of Cyanobacteria) in Low Earth Orbit and Simulated Extraterrestrial Conditions. Orig Life Evol Biosph 39, 565–579 (2009). https://doi.org/10.1007/s11084-009-9167-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11084-009-9167-4