Abstract
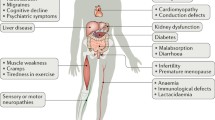
Mitochondria play a pivotal role in mammalian cell metabolism, hosting a number of important biochemical pathways including oxidative phosphorylation. As might be expected from this fundamental contribution to cell function, abnormalities of mitochondrial metabolism are a common cause of human disease. Primary mutations of mitochondrial DNA result in a diverse group of disorders often collectively referred to as the mitochondrial encephalomyopathies. Perhaps more importantly in numerical terms are those neurodegenerative diseases caused by mutations of nuclear genes encoding mitochondrial proteins. Finally there are mitochondrial abnormalities induced by secondary events e.g. oxidative stress that may contribute to senescence, and environmental toxins that may cause disease either alone or in combination with a genetic predisposition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the last 20 years there has been an increasing recognition of the role played by the mitochondrion in causing human diseases [1–3]. These are mediated through mutations of mitochondrial DNA (mtDNA), nuclear DNA genes encoding mitochondrial proteins and endogenous and exogenous toxins that target mitochondrial metabolism. These diseases can affect any organ although muscle and nerve are most frequently involved. This review will focus upon mitochondrial dysfunction in selected neurodegenerative diseases.
Chronic Progressive External Ophthalmoplegia and Kearns-Sayre Syndrome
Chronic progressive external ophthalmoplegia (CPEO) is predominantly a myopathic disorder but there have been reports of dementia, seizures, myoclonus and stroke like episodes [4, 5]. In Kearns Sayre syndrome (KSS), CPEO is accompanied by pigmentary retinopathy, and one or more of complete heart block, a cerebrospinal fluid (CSF) protein level of above 1 g/l, and ataxia [6]. The diagnostic criteria quote an age of onset below 20 years, but improved identification of cases has revealed later onset in some patients.
Mitochondrial DNA from CPEO/KSS patients shows large single deletions detectable in DNA extracted from muscle samples, but blood mtDNA analysis usually is normal. These deletions are found in 80% of those with KSS, and 70% of those with CPEO [7–9]. Histological examination of muscle reveals ragged red fibres (RRFs) and a mosaic pattern of cytochrome oxidase (COX) negative fibres. Deletions probably arise de novo during oogenesis in the mother and patients with mtDNA deletions therefore present as sporadic cases [10]. The risk of transmission of deletions is approximately 4% and it is rare for more than one sibling to be affected.
Encephalomyopathies
The common mitochondrial encephalopathies include MELAS (myopathy, encephalopathy, lactic acidosis and stroke-like episodes) and MERRF (myoclonic epilepsy and ragged red fibres). MELAS can include psychomotor retardation, ataxia, cognitive impairment, deafness, diabetes mellitus and limb weakness. Of all cases meeting the clinical criteria for MELAS, 80% are positive for an A to G transition at base pair 3243 within the tRNA Leu UUR gene [11–13].
The core clinical features of MERRF are myoclonus, ataxia, and seizures. Myoclonus is often the presenting symptom, and may be induced by action, noise, or photic stimulation. Seizure types are variable, but include drop attacks, focal seizures, and photo-sensitive tonic-clonic seizures [14, 15]. Myopathy is usually either mild, subclinical, or absent. RRFs are seen on muscle biopsy in the majority of patients, but not in all cases. Additional features include ophthalmoplegia, ptosis, deafness, peripheral neuropathy, headache, foot deformity, optic atrophy and cervical lipomas. The most commonly detected mutation, found in approximately 80% of cases fulfilling the clinical criteria for MERRF, is at position 8344 within the tRNA Lys [11, 16, 17].
Neurogenic Weakness Ataxia and Retinal Pigmentation (NARP)
The key features of NARP are peripheral neuropathy, ataxia, retinitis pigmentosa, seizures and dementia [18]. A spectrum of neurological findings have been described in NARP including migraine and mental retardation [18–24]. Clinical features are variable as is the age of onset which in one series varied between 1 and 32 years [25, 26]. Inheritance is maternal.
The commonest mutation is a T to G transversion at nucleotide position 8993. This causes a change from the highly conserved leucine to arginine within subunit 6 of the mitochondrial F0F1 ATP synthase. Patients with NARP usually have above 80% mutant mtDNA levels. With mutant mtDNA levels below 75% patients usually suffer from pigmentary retinopathy alone, or suffer migraines, or are asymptomatic [18–23]. Ragged-red fibers and other morphological mitochondrial hallmarks are lacking in muscle biopsies from patients with NARP [18].
Leigh Syndrome (LS)
Leigh syndrome (LS) is a subacute necrotising encephalomyelopathy characterised by bilateral symmetrical focal necrotic lesions within the thalamus, extending into the pons, inferior olives and spinal cord. The clinical features of LS are of psychomotor retardation, hypotonia, failure to thrive, respiratory abnormalities, oculomotor disturbances, ataxia, optic atrophy, seizures and lactic acidosis. Biochemical abnormalities include defects of oxidative phosphorylation [27] (in particular complex I [28] or complex IV [29, 30], and deficiency of the pyruvate dehydrogenase complex [31] and biotinidase deficiency [32]. The majority of LS cases are believed to result from nuclear gene defects [33, 34]. This has been confirmed for cases of LS with PDH complex deficiency [33], complex I [35], and II [36]. Complex IV deficient LS results from mutations of the Surf or other assembly genes [37–39]. Up to 20% of LS patients have the T to G, or T to C, mtDNA mutation at position 8993 within the ATPase 6 gene of complex V [40–44]. Mutant loads are above 90%, and lower levels of this mutation are associated with the NARP syndrome. Other mtDNA mutations have been described including base substitutions, deletions and depletion of mtDNA levels [45–48].
Friedreich’s Ataxia
Friedreich’s ataxia (FRDA) is an autosomal recessive disease characterised clinically by a progressive gait and limb ataxia, absence of deep tendon reflexes and loss of position and vibration sense in the lower limbs. Skeletal abnormalities including kyphosis, hypertrophic cardiomyopathy and less commonly, diabetes and optic atrophy are also present in FRDA patients.
Over 95% of FRDA patients have a homozygous expansion of a GAA triplet repeat (6–34 GAA repeats in controls expanded to between 67 and 1700 in patients) in intron 1 of the frataxin gene on chromosome 9 [49]. Most of the remaining patients are compound heterozygotes with the GAA expansion in one allele and point mutations in the other [50]. No patient has yet been described with a point mutation in each allele.
Defects of the mitochondrial respiratory chain have been demonstrated in yeast YFH1 mutants [51, 52], conditional frataxin knockout transgenic mice [53] and post mortem heart and skeletal muscle from FRDA patients [54, 55]. The pattern of respiratory chain dysfunction (complexes I, II and III) is similar to that caused by oxidative stress caused and knockout of manganese superoxide dismutase [56], or secondary to excitotoxicity in Huntington’s disease [57]. However, all the enzyme activities decreased in FRDA contain Fe–S clusters which led to the proposal that an abnormality of Fe–S cluster synthesis may be responsible for these defects.
31Phosphorous magnetic resonance spectroscopy (31P MRS) is an in vivo technique that can measure high energy phosphorous compounds (phosphocreatine and ATP) in heart and skeletal muscle. In skeletal muscle phosphocreatine (PCr) levels fall during exercise, and the analysis of the rate at which PCr levels recover following exercise (Vmax) is a measure of the efficiency of oxidative phosphorylation [58]. The PCr/ATP ratio is used as a good measure of energy availability in heart. 31P MRS analysis of FRDA patients revealed markedly decreased oxidative phosphorylation in the heart [59] and skeletal muscle, with the latter correlating with the size of the smallest GAA repeat [60]. These data underline the role of mitochondrial dysfunction in FRDA and suggest it is playing a primary role in disease pathogenesis [61].
Vitamin E is a naturally occurring lipid soluble antioxidant distributed throughout cellular membranes and is particularly abundant in mitochondrial membranes. The treatment of FRDA patients with vitamin E has only been reported in conjunction with coenzyme Q10. We have assessed the efficacy of long term treatment of 10 patients with FRDA with high doses of vitamin E (2100 IU/day) and coenzyme Q10 (400 mg/day). After 6 months 31P MRS data indicated that heart and skeletal muscle energetics were significantly improved [62]. Four year follow up data from this study showed the enhanced energy levels were maintained, clinical parameters were stabilised or improved in 7 out of 10 patients and heart fraction shortening had improved [63]. This effect of vitamin E is supported by the consequences of vitamin E deficiency on mitochondrial function [64].
Idebenone is a short chain analogue of coenzyme Q10, is well tolerated by humans, crosses the blood brain barrier, has been reported to be a relatively good antioxidant [65], and has been used in a variety of diseases with some benefits [66, 67]. The effect of idebenone upon cardiac hypertrophy in FRDA patients was assessed using echocardiography, but other clinical improvements were not reported. After 6 months treatment cardiac hypertrophy was decreased in up to half the patients tested, although this was not always associated with improved fraction shortening [68].
Parkinson’s Disease
A mitochondrial defect in PD was first identified in 1989 in substantia nigra from patients with PD [69–73]. These study has been expanded over the years and results to date show that there is about a 35% complex I deficiency in PD nigra [74]. This defect in complex I activity does not affect any other part of the respiratory chain an unusual feature of mitochondrial disease [75]. In addition, no defect in mitochondrial activity has been identifiable in any other part of PD brain, including caudate putamen, globus pallidus, tegmentum, cortex, cerebellum or substantia innominata [76].
The discovery of complex I deficiency in PD raised issues regarding its role in terms of whether it played a primary or secondary part in pathogenesis. The PD brains examined were from patients with longstanding disease who had also been treated with a variety of drugs e.g. levodopa. However, there was no deficiency of complex I activity in PD striatum which one might expect from the rat model of levodopa toxicity [77]. Furthermore, patients with multiple system atrophy who have taken L-dopa in quantities and for duration comparable to patients with PD have no defect of mitochondrial activity in their substantia nigra [78]. Other drugs such as dopamine agonists and monoamine oxidase B inhibitors are not known to influence complex I activity.
Following the report of complex I deficiency in PD substantia nigra, respiratory chain abnormalities were described in skeletal muscle mitochondria from PD patients. This particular area has proved very contentious, with several groups either describing similar defects, or no abnormality [78, 79, 80]. Finally, mitochondrial complex I deficiency was also identified in platelet mitochondria of PD patients [81, 82].
Several studies have investigated the structure of mtDNA in tissues from PD patients to determine whether the complex I defect is associated with an underlying mtDNA mutation [83]. No reproducible abnormality of mtDNA has been identified in PD. However, the level of mtDNA deletions in single substantia nigral neurons appears to increase substantially over the age of 65 years [84]. One study found a high proportion of deleted mtDNA (43 ± 9.3%) in nigral neurons of controls and a higher proportion in parkinsonian nigra (52.3 ± 9.3%). These results confirm that the human substantia nigra is a site of free radical mediated damage to mtDNA and that this is enhanced in neurodegenerative diseases.
Two studies have used genetic transplantation to investigate the potential for PD mtDNA to determine the complex I defect. In one, unselected PD platelets were fused and grown in mixed culture [85]. In another, PD patients were selected on the basis of demonstrating a peripheral complex I deficiency. These patients’ cells were then fused with rho-zero cells and grown both in mixed and clonal culture [86]. In both, mtDNA transferred from the PD patients induced a complex I defect in the recipient cybrid cells. These results indicate that the mtDNA in these patients caused the complex I deficiency through either inherited or somatic mutations. Further experiments suggested that the recipient cells also developed abnormal calcium handling and a lower mitochondrial membrane potential.
MtDNA polymerase gamma (POLG) mutations have been demonstrated in patients with progressive external ophthalmoplegia (PEO) and parkinsonism [87].
The human POLG gene includes a trinucleotide microsatellite CAG repeat that encodes a polyglutamine tract in the amino-terminal region of the POLG protein. Although expansion of CAG repeats are a cause of several neurodegenerative diseases no changes were seen in the CAG repeat of PD patients that might have contributed to modifications of mtDNA [88].
Recessive mutations in PINK1 were found to be responsible for a familial form of early-onset parkinsonism, previously mapped to chromosome 1p36 (the PARK6 locus) [89]. PINK1 mutations are relatively rare. The PINK1 gene is ubiquitously transcribed and encodes a mitochondrial kinase [90]. Preliminary data have suggested that PINK1 may play a role in protecting cells against stress conditions that affect mitochondrial membrane potential, but the downstream targets through which PINK1 mediates its protection have not been identified. Parkin mutations are an uncommon cause of autosomal recessive PD. There is some evidence to implicate mitochondrial dysfunction in the pathogenesis of parkin mutations. One report has shown that parkin may be localised within mitochondria [91]. Parkin was present within the mitochondrial matrix in proliferating cells and entered the cytosol when the permeability pore was open. This extra-mitochondrial localisation was also seen in differentiated cells. Up-regulation of parkin resulted in an increase in mtDNA transcription and translation via an interaction with mitochondrial transcription factor A, while RNA silencing of parkin lead to a reduction of levels. It is notable that parkin over-expression can compensate for PINK1 mutations in Drosophila [92, 93]. The implication is that parkin functions downstream of PINK1 and intervenes in the pathogenetic pathway of PINK1 mutations.
Mutations in DJ1 are another rare cause of early onset autosomal recessive PD [94]. DJ1 is a widely expressed cytoplasmic 23 kDa protein that is distributed in several subcellular compartments including the mitochondrial matrix and inter-membranous space [95]. DJ1 is thought to function as an anti-oxidant protein. There is some evidence that the distribution of DJ1 may be modified by mutations and that this leads to decreased nuclear and increased mitochondrial localisation which in turn results in impaired transcriptional co-activator function rendering cells more sensitive to pyrimidine binding tract protein-associated splicing factor (PSF) induced apoptosis [96].
Mutations in the Omi/HTRA2 gene have been identified in four PD patients with an additional mutation thought to represent a risk factor for PD [97]. The Omi/HTRA2 protein is a mitochondrial protein located within the inter-membranous space and functions as a procaspase, released as part of the apoptotic cascade. The G399S mutation results in reduced serine-protease activity. Omi/HTRA2 knockout mice display a parkinsonian phenotype that includes rigidity but additional features that include ataxia muscle wasting and premature death [98].
The benefits of an improved understanding of the mitochondrial involvement in the pathogenesis of PD has already translated into potential therapeutic interventions to slow progression of the disease [99].
Mutations have likewise been found in nuclear genes encoding mitochondrial proteins in neurodegenerative diseases such as chromosome 16-linked hereditary spastic paraplegia (paraplegin), autosomal dominant optic atrophy (OPA1) and Charcot Marie Tooth disease (mitofucin) [100]. Severe defects of complexes II and III of the respiratory chain have also been described in Huntington’s disease brain [101, 102]. The relevance of the latter has been highlighted by the recent observation that mutant huntingtin reduces the expression of the peroxisome proliferator-activated receptor-γ co-activator 1α (PGC-1α) [103]. This protein controls many metabolic processes including mitochondrial biogenesis and oxidative phosphorylation by the regulation of expression of proteins of the respiratory chain.
Conclusions
Mitochondria undoubtedly play an important role in human pathology and there is little doubt that numerous other disorders will be attributed to mitochondrial dysfunction. The difficulty is in finding appropriate treatments. Targeting the mitochondrion with suitable drugs may become an important focus for future therapeutic approaches.
References
Leonard JV, Schapira AH (2000) Mitochondrial respiratory chain disorders I: mitochondrial DNA defects. Lancet 355:299–304. doi:10.1016/S0140-6736(99)05225-3
Leonard JV, Schapira AH (2000) Mitochondrial respiratory chain disorders II: neurodegenerative disorders and nuclear gene defects. Lancet 355:389–394. doi:10.1016/S0140-6736(99)05226-5
Schapira AH (2006) Mitochondrial disease. Lancet 368:70–82. doi:10.1016/S0140-6736(06)68970-8
Morgan-Hughes JA, Sweeney MG, Cooper JM, Hammans SR, Brockington M, Schapira AH, Harding AE, Clark JB (1995) Mitochondrial DNA (mtDNA) diseases: correlation of genotype to phenotype. Biochim Biophys Acta 1271:135–140
Morgan-Hughes JA, Schapira AH, Cooper JM, Clark JB (1988) Molecular defects of NADH-ubiquinone oxidoreductase (complex I) in mitochondrial diseases. J Bioenerg Biomembr 20:365–382. doi:10.1007/BF00769638
Berenberg RA, Pellock JM, DiMauro S et al (1977) Lumping or splitting? “Ophthalmoplegia-plus” or Kearns-Sayre syndrome? Ann Neurol 1:37–54. doi:10.1002/ana.410010104
Holt IJ, Harding AE, Morgan-Hughes JA (1988) Deletions of muscle mitochondrial DNA in patients with mitochondrial myopathies. Nature 331:717–719. doi:10.1038/331717a0
Holt IJ, Harding AE, Cooper JM et al (1989) Mitochondrial myopathies: clinical and biochemical features of 30 patients with major deletions of muscle mitochondrial DNA. Ann Neurol 26:699–708. doi:10.1002/ana.410260603
Moraes CT, DiMauro S, Zeviani M et al (1989) Mitochondrial DNA deletions in progressive external ophthalmoplegia and Kearns-Sayre syndrome. N Engl J Med 320:1293–1299
Zeviani M, Moraes CT, DiMauro S et al (1998) Deletions of mitochondrial DNA in Kearns-Sayre syndrome. Neurology 51:1525
Hammans SR, Sweeney MG, Brockington M et al (1991) Mitochondrial encephalopathies: molecular genetic diagnosis from blood samples. Lancet 337:1311–1313. doi:10.1016/0140-6736(91)92981-7
De Volder A, Ghilain S, de Barsy T et al (1988) Brain metabolism in mitochondrial encephalomyopathy: a PET study. J Comput Assist Tomogr 12:854–857. doi:10.1097/00004728-198809010-00024
Kobayashi Y, Momoi MY, Tominaga K et al (1990) A point mutation in the mitochondrial tRNA(Leu)(UUR) gene in MELAS (mitochondrial myopathy, encephalopathy, lactic acidosis and stroke-like episodes). Biochem Biophys Res Commun 173:816–822. doi:10.1016/S0006-291X(05)80860-5
Hammans SR, Sweeney MG, Brockington M et al (1993) The mitochondrial DNA transfer RNA(Lys)A–>G(8344) mutation and the syndrome of myoclonic epilepsy with ragged red fibres (MERRF). Relationship of clinical phenotype to proportion of mutant mitochondrial DNA. Brain 116(Pt 3):617–632. doi:10.1093/brain/116.3.617
Berkovic SF, Carpenter S, Evans A et al (1989) Myoclonus epilepsy and ragged-red fibres (MERRF). 1. A clinical, pathological, biochemical, magnetic resonance spectrographic and positron emission tomographic study. Brain 112(Pt 5):1231–1260. doi:10.1093/brain/112.5.1231
Shoffner JM, Lott MT, Lezza AM, Seibel P et al (1990) Myoclonic epilepsy and ragged-red fiber disease (MERRF) is associated with a mitochondrial DNA tRNA(Lys) mutation. Cell 61:931–937. doi:10.1016/0092-8674(90)90059-N
Zeviani M, Amati P, Bresolin N et al (1991) Rapid detection of the A—G(8344) mutation of mtDNA in Italian families with myoclonus epilepsy and ragged-red fibers (MERRF). Am J Hum Genet 48:203–211
Holt IJ, Harding AE, Petty RK et al (1990) A new mitochondrial disease associated with mitochondrial DNA heteroplasmy. Am J Hum Genet 46:428–433
Tsairis P, Engel W, Kark P (1973) Familial Myoclonid epilepsy syndrome associated with skeletal muscle mitochondrial abnormalities. Neurology 23:408
Takeda S, Wakabayashi K, Ohama E et al (1988) Neuropathology of myoclonus epilepsy associated with ragged-red fibers (Fukuhara’s disease). Acta Neuropathol 75:433–440. doi:10.1007/BF00687129
Ortiz RG, Newman NJ, Shoffner JM, Kaufman AE, Koontz DA, Wallace DC (1993) Variable retinal and neurologic manifestations in patients harboring the mitochondrial DNA 8993 mutation. Arch Ophthalmol 111:1525–1530
Fryer A, Appleton R, Sweeney MG et al (1994) Mitochondrial DNA 8993 (NARP) mutation presenting with a heterogeneous phenotype including ‘cerebral palsy’. Arch Dis Child 71:419–422
Makela-Bengs P, Suomalainen A, Majander A et al (1995) Correlation between the clinical symptoms and the proportion of mitochondrial DNA carrying the 8993 point mutation in the NARP syndrome. Pediatr Res 37:634–639. doi:10.1203/00006450-199505000-00014
Puddu P, Barboni P, Mantovani V et al (1993) Retinitis pigmentosa, ataxia, and mental retardation associated with mitochondrial DNA mutation in an Italian family. Br J Ophthalmol 77:84–88. doi:10.1136/bjo.77.2.84
Uziel G, Moroni I, Lamantea E et al (1997) Mitochondrial disease associated with the T8993G mutation of the mitochondrial ATPase 6 gene: a clinical, biochemical, and molecular study in six families. J Neurol Neurosurg Psychiatry 63:16–22
Chowers I, Lerman-Sagie T, Elpeleg ON et al (1999) Cone and rod dysfunction in the NARP syndrome. Br J Ophthalmol 83:190–193
DiMauro S, De Vivo DC (1996) Genetic heterogeneity in Leigh syndrome. Ann Neurol 40:5–7. doi:10.1002/ana.410400104
van Erven PM, Gabreels FJ, Ruitenbeek W, Renier WO, Fischer JC (1987) Mitochondrial encephalomyopathy. Association with an NADH dehydrogenase deficiency. Arch Neurol 44:775–778
Willems JL, Monnens LA, Trijbels JM, Veerkamp JH, Meyer AE, van Dam K, van Haelst U (1977) Leigh’s encephalomyelopathy in a patient with cytochrome c oxidase deficiency in muscle tissue. Pediatrics 60:850–857
Van Coster R, Lombres A, De Vivo DC, Chi TL, Dodson WE, Rothman S, Orrechio EJ, Grover W, Berry GT, Schwartz JF (1991) Cytochrome c oxidase-associated Leigh syndrome: phenotypic features and pathogenetic speculations. J Neurol Sci 104:97–111. doi:10.1016/0022-510X(91)90222-S
Kretzschmar HA, DeArmond SJ, Koch TK, Patel MS, Newth CJ, Schmidt KA, Packman S (1987) Pyruvate dehydrogenase complex deficiency as a cause of subacute necrotizing encephalopathy (Leigh disease). Pediatrics 79:370–373
Baumgartner ER, Suormala TM, Wick H, Probst A, Blauenstein U, Bachmann C, Vest M (1989) Biotinidase deficiency: a cause of subacute necrotizing encephalomyelopathy (Leigh syndrome). Report of a case with lethal outcome. Pediatr Res 26:260–266. doi:10.1203/00006450-198909000-00021
Matthews PM, Marchington DR, Squier M, Land J, Brown RM, Brown GK (1993) Molecular genetic characterization of an X-linked form of Leigh’s syndrome. Ann Neurol 33:652–655. doi:10.1002/ana.410330616
Vazquez-Memije ME, Shanske S, Santorelli FM, Kranz-Eble P, Davidson E, DeVivo DC, DiMauro S (1996) Comparative biochemical studies in fibroblasts from patients with different forms of Leigh syndrome. J Inherit Metab Dis 19:43–50. doi:10.1007/BF01799347
Loeffen J, Smeitink J, Triepels R, Smeets R, Schuelke M, Sengers R, Trijbels F, Hamel B, Mullaart R, van den HL (1998) The first nuclear-encoded complex I mutation in a patient with Leigh syndrome. Am J Hum Genet 63:1598–1608. doi:10.1086/302154
Bourgeron T, Rustin P, Chretien D, Birch-Machin M, Bourgeois M, Viegas-Pequignot E, Munnich A, Rotig A (1995) Mutation of a nuclear succinate dehydrogenase gene results in mitochondrial respiratory chain deficiency. Nat Genet 11:144–149. doi:10.1038/ng1095-144
Tiranti V, Hoertnagel K, Carrozzo R, Galimberti C, Munaro M, Granatiero M, Zelante L, Gasparini P, Marzella R, Rocchi M, Bayona-Bafaluy MP, Enriquez JA, Uziel G, Bertini E, Dionisi-Vici C, Franco B, Meitinger T, Zeviani M (1998) Mutations of SURF-1 in Leigh disease associated with cytochrome c oxidase deficiency. Am J Hum Genet 63:1609–1621. doi:10.1086/302150
Zhu Z, Yao J, Johns T, Fu K, De Bie I, Macmillan C, Cuthbert AP, Newbold RF, Wang J, Chevrette M, Brown GK, Brown RM, Shoubridge EA (1998) SURF1, encoding a factor involved in the biogenesis of cytochrome c oxidase, is mutated in Leigh syndrome. Nat Genet 20:337–343. doi:10.1038/3804
Sue CM, Karadimas C, Checcarelli N, Tanji K, Papadopoulou LC, Pallotti F, Guo FL, Shanske S, Hirano M, De Vivo DC, Van Coster R, Kaplan P, Bonilla E, DiMauro S (2000) Differential features of patients with mutations in two COX assembly genes, SURF-1 and SCO2. Ann Neurol 47:589–595. doi:10.1002/1531-8249(200005)47:5<589::AID-ANA6>3.0.CO;2-D
Ciafaloni E, Santorelli FM, Shanske S, Deonna T, Roulet E, Janzer C, Pescia G, DiMauro S (1993) Maternally inherited Leigh syndrome. J Pediatr 122:419–422. doi:10.1016/S0022-3476(05)83431-6
Santorelli FM, Shanske S, Macaya A, DeVivo DC, DiMauro S (1993) The mutation at nt 8993 of mitochondrial DNA is a common cause of Leigh’s syndrome. Ann Neurol 34:827–834. doi:10.1002/ana.410340612
de Vries DD, van Engelen BG, Gabreels FJ, Ruitenbeek W, van Oost BA (1993) A second missense mutation in the mitochondrial ATPase 6 gene in Leigh’s syndrome. Ann Neurol 34:410–412. doi:10.1002/ana.410340319
Santorelli FM, Shanske S, Jain KD, Tick D, Schon EA, DiMauro S (1994) A T-->C mutation at nt 8993 of mitochondrial DNA in a child with Leigh syndrome. Neurology 44:972–974
Cox GB, Fimmel AL, Gibson F, Hatch L (1986) The mechanism of ATP synthase: a reassessment of the functions of the b and a subunits. Biochim Biophys Acta 849:62–69. doi:10.1016/0005-2728(86)90096-4
Rahman S, Blok RB, Dahl HH, Danks DM, Kirby DM, Chow CW, Christodoulou J, Thorburn DR (1996) Leigh syndrome: clinical features and biochemical and DNA abnormalities. Ann Neurol 39:343–351. doi:10.1002/ana.410390311
Yamamoto M, Clemens PR, Engel AG (1991) Mitochondrial DNA deletions in mitochondrial cytopathies: observations in 19 patients. Neurology 41:1822–1828
Yamadori I, Kurose A, Kobayashi S, Ohmori M, Imai T (1992) Brain lesions of the Leigh-type distribution associated with a mitochondriopathy of Pearson’s syndrome: light and electron microscopic study. Acta Neuropathol 84:337–341. doi:10.1007/BF00227830
Morris AA, Taanman JW, Blake J, Cooper JM, Lake BD, Malone M, Love S, Clayton PT, Leonard JV, Schapira AH (1998) Liver failure associated with mitochondrial DNA depletion. J Hepatol 28:556–563. doi:10.1016/S0168-8278(98)80278-X
Campuzano V, Montermini L, Molto MD, Pianese L, Cossee M, Cavalcanti F, Monros E, Rodius F, Duclos F, Monticelli A, Zara F, Canizares J, Koutnikova H, Bidichandani SI, Gellera C, Brice A, Trouillas P, De Michele G, Filla A, De Frutos R, Palau F, Patel PI, Di Donato S, Mandel JL, Cocozza S, Koenig M, Pandolfo M (1996) Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 271:1423–1427. doi:10.1126/science.271.5254.1423
Pook MA, Al Mahdawi SA, Thomas NH, Appleton R, Norman A, Mountford R, Chamberlain S (2000) Identification of three novel frameshift mutations in patients with Friedreich’s ataxia. J Med Genet 37:E38. doi:10.1136/jmg.37.11.e38
Foury F, Cazzalini O (1997) Deletion of the yeast homologue of the human gene associated with Friedreich’s ataxia elicits iron accumulation in mitochondria. FEBS Lett 411:373–377. doi:10.1016/S0014-5793(97)00734-5
Koutnikova H, Campuzano V, Foury F, Dolle P, Cazzalini O, Koenig M (1997) Studies of human, mouse and yeast homologues indicate a mitochondrial function for frataxin. Nat Genet 16:345–351. doi:10.1038/ng0897-345
Puccio H, Simon D, Cossee M, Criqui-Filipe P, Tiziano F, Melki J, Hindelang C, Matyas R, Rustin P, Koenig M (2001) Mouse models for Friedreich ataxia exhibit cardiomyopathy, sensory nerve defect and Fe–S enzyme deficiency followed by intramitochondrial iron deposits. Nat Genet 27:181–186. doi:10.1038/84818
Bradley JL, Blake JC, Chamberlain S, Thomas PK, Cooper JM, Schapira AH (2000) Clinical, biochemical and molecular genetic correlations in Friedreich’s ataxia. Hum Mol Genet 9:275–282. doi:10.1093/hmg/9.2.275
Rotig A, de Lonlay P, Chretien D, Foury F, Koenig M, Sidi D, Munnich A, Rustin P (1997) Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet 17:215–217. doi:10.1038/ng1097-215
Melov S, Coskun P, Patel M, Tuinstra R, Cottrell B, Jun AS, Zastawny TH, Dizdaroglu M, Goodman SI, Huang TT, Miziorko H, Epstein CJ, Wallace DC (1999) Mitochondrial disease in superoxide dismutase 2 mutant mice. Proc Natl Acad Sci USA 96:846–851. doi:10.1073/pnas.96.3.846
Tabrizi SJ, Workman J, Hart PE, Mangiarini L, Mahal A, Bates G, Cooper JM, Schapira AH (2000) Mitochondrial dysfunction and free radical damage in the Huntington R6/2 transgenic mouse. Ann Neurol 47:80–86. doi:10.1002/1531-8249(200001)47:1<80::AID-ANA13>3.0.CO;2-K
Kemp GJ, Taylor DJ, Thompson CH, Hands LJ, Rajagopalan B, Styles P, Radda GK (1993) Quantitative analysis by 31P magnetic resonance spectroscopy of abnormal mitochondrial oxidation in skeletal muscle during recovery from exercise. NMR Biomed 6:302–310. doi:10.1002/nbm.1940060504
Lodi R, Rajagopalan B, Blamire AM, Cooper JM, Davies CH, Bradley JL, Styles P, Schapira AH (2001) Cardiac energetics are abnormal in Friedreich ataxia patients in the absence of cardiac dysfunction and hypertrophy: an in vivo 31P magnetic resonance spectroscopy study. Cardiovasc Res 52:111–119. doi:10.1016/S0008-6363(01)00357-1
Lodi R, Cooper JM, Bradley JL, Manners D, Styles P, Taylor DJ, Schapira AH (1999) Deficit of in vivo mitochondrial ATP production in patients with Friedreich ataxia. Proc Natl Acad Sci USA 96:11492–11495. doi:10.1073/pnas.96.20.11492
Cooper JM, Schapira AH (2007) Friedreich’s ataxia: coenzyme Q10 and vitamin E therapy. Mitochondrion 7(Suppl):S127–S135. doi:10.1016/j.mito.2007.04.001
Lodi R, Hart PE, Rajagopalan B, Taylor DJ, Crilley JG, Bradley JL, Blamire AM, Manners D, Styles P, Schapira AH, Cooper JM (2001) Antioxidant treatment improves in vivo cardiac and skeletal muscle bioenergetics in patients with Friedreich’s ataxia. Ann Neurol 49:590–596. doi:10.1002/ana.1001
Hart PE, Lodi R, Rajagopalan B, Bradley JL, Crilley JG, Turner C, Blamire AM, Manners D, Styles P, Schapira AH, Cooper JM (2005) Antioxidant treatment of patients with Friedreich ataxia: four-year follow-up. Arch Neurol 62:621–626. doi:10.1001/archneur.62.4.621
Thomas PK, Cooper JM, King RH, Workman JM, Schapira AH, Goss-Sampson MA, Muller DP (1993) Myopathy in vitamin E deficient rats: muscle fibre necrosis associated with disturbances of mitochondrial function. J Anat 183(Pt 3):451–461
Mordente A, Martorana GE, Minotti G, Giardina B (1998) Antioxidant properties of 2, 3-dimethoxy-5-methyl-6-(10-hydroxydecyl)-1, 4-benzoquinone (idebenone). Chem Res Toxicol 11:54–63. doi:10.1021/tx970136j
Gutzmann H, Hadler D (1998) Sustained efficacy and safety of idebenone in the treatment of Alzheimer’s disease: update on a 2-year double-blind multicentre study. J Neural Transm Suppl 54:301–310
Ranen NG, Peyser CE, Coyle JT, Bylsma FW, Sherr M, Day L, Folstein MF, Brandt J, Ross CA, Folstein SE (1996) A controlled trial of idebenone in Huntington’s disease. Mov Disord 11:549–554. doi:10.1002/mds.870110510
Hausse AO, Aggoun Y, Bonnet D, Sidi D, Munnich A, Rotig A, Rustin P (2002) Idebenone and reduced cardiac hypertrophy in Friedreich’s ataxia. Heart 87:346–349. doi:10.1136/heart.87.4.346
Schapira AH, Cooper JM, Dexter D, Jenner P, Clark JB, Marsden CD (1989) Mitochondrial complex I deficiency in Parkinson’s disease. Lancet 1:1269. doi:10.1016/S0140-6736(89)92366-0
Schapira AH, Cooper JM, Dexter D, Clark JB, Jenner P, Marsden CD (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54:823–827. doi:10.1111/j.1471-4159.1990.tb02325.x
Schapira AH (2007) Mitochondrial dysfunction in Parkinson’s disease. Cell Death Differ 14:1261–1266. doi:10.1038/sj.cdd.4402160
Schapira AH (2008) Mitochondria in the aetiology and pathogenesis of Parkinson’s disease. Lancet Neurol 7:97–109. doi:10.1016/S1474-4422(07)70327-7
Orth M, Schapira AH (2002) Mitochondrial involvement in Parkinson’s disease. Neurochem Int 40:533–541. doi:10.1016/S0197-0186(01)00124-3
Mann VM, Cooper JM, Daniel SE, Srai K, Jenner P, Marsden CD, Schapira AH (1994) Complex I, iron, and ferritin in Parkinson’s disease substantia nigra. Ann Neurol 36:876–881. doi:10.1002/ana.410360612
Schapira AH, Cooper JM, Morgan-Hughes JA, Patel SD, Cleeter MJ, Ragan CI, Clark JB (1988) Molecular basis of mitochondrial myopathies: polypeptide analysis in complex-I deficiency. Lancet 1:500–503. doi:10.1016/S0140-6736(88)91296-2
Schapira AH, Mann VM, Cooper JM, Dexter D, Daniel SE, Jenner P, Clark JB, Marsden CD (1990) Anatomic and disease specificity of NADH CoQ1 reductase (complex I) deficiency in Parkinson’s disease. J Neurochem 55:2142–2145. doi:10.1111/j.1471-4159.1990.tb05809.x
Cooper JM, Daniel SE, Marsden CD, Schapira AH (1995) L-dihydroxyphenylalanine and complex I deficiency in Parkinson’s disease brain. Mov Disord 10:295–297. doi:10.1002/mds.870100311
Gu M, Gash MT, Cooper JM, Wenning GK, Daniel SE, Quinn NP, Marsden CD, Schapira AH (1997) Mitochondrial respiratory chain function in multiple system atrophy. Mov Disord 12:418–422. doi:10.1002/mds.870120323
Schapira AH (1994) Evidence for mitochondrial dysfunction in Parkinson’s disease—a critical appraisal. Mov Disord 9:125–138. doi:10.1002/mds.870090202
Taylor DJ, Krige D, Barnes PR, Kemp GJ, Carroll MT, Mann VM, Cooper JM, Marsden CD, Schapira AH (1994) A 31P magnetic resonance spectroscopy study of mitochondrial function in skeletal muscle of patients with Parkinson’s disease. J Neurol Sci 125:77–81. doi:10.1016/0022-510X(94)90245-3
Parker WD Jr, Boyson SJ, Parks JK (1989) Abnormalities of the electron transport chain in idiopathic Parkinson’s disease. Ann Neurol 26:719–723. doi:10.1002/ana.410260606
Krige D, Carroll MT, Cooper JM, Marsden CD, Schapira AH (1992) Platelet mitochondrial function in Parkinson’s disease. The Royal Kings and Queens Parkinson Disease Research Group. Ann Neurol 32:782–788. doi:10.1002/ana.410320612
Mann VM, Cooper JM, Schapira AH (1992) Quantitation of a mitochondrial DNA deletion in Parkinson’s disease. FEBS Lett 299:218–222. doi:10.1016/0014-5793(92)80118-Z
Kraytsberg Y, Kudryavtseva E, McKee AC, Geula C, Kowall NW, Khrapko K (2006) Mitochondrial DNA deletions are abundant and cause functional impairment in aged human substantia nigra neurons. Nat Genet 38:518–520. doi:10.1038/ng1778
Swerdlow RH, Parks JK, Miller SW, Tuttle JB, Trimmer PA, Sheehan JP, Bennett JP Jr, Davis RE, Parker WD Jr (1996) Origin and functional consequences of the complex I defect in Parkinson’s disease. Ann Neurol 40:663–671. doi:10.1002/ana.410400417
Gu M, Cooper JM, Taanman JW, Schapira AH (1998) Mitochondrial DNA transmission of the mitochondrial defect in Parkinson’s disease. Ann Neurol 44:177–186. doi:10.1002/ana.410440207
Luoma P, Melberg A, Rinne JO, Kaukonen JA, Nupponen NN, Chalmers RM, Oldfors A, Rautakorpi I, Peltonen L, Majamaa K, Somer H, Suomalainen A (2004) Parkinsonism, premature menopause, and mitochondrial DNA polymerase gamma mutations: clinical and molecular genetic study. Lancet 364:875–882. doi:10.1016/S0140-6736(04)16983-3
Taanman JW, Schapira AH (2005) Analysis of the trinucleotide CAG repeat from the DNA polymerase gamma gene (POLG) in patients with Parkinson’s disease. Neurosci Lett 376:56–59. doi:10.1016/j.neulet.2004.11.023
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MM, Harvey K, Gispert S, Ali Z, Del Turco D, Bentivoglio AR, Healy DG, Albanese A, Nussbaum R, Gonzalez-Maldonado R, Deller T, Salvi S, Cortelli P, Gilks WP, Latchman DS, Harvey RJ, Dallapiccola B, Auburger G, Wood NW (2004) Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 304:1158–1160. doi:10.1126/science.1096284
Unoki M, Nakamura Y (2001) Growth-suppressive effects of BPOZ and EGR2, two genes involved in the PTEN signaling pathway. Oncogene 20:4457–4465. doi:10.1038/sj.onc.1204608
Kuroda Y, Mitsui T, Kunishige M, Shono M, Akaike M, Azuma H, Matsumoto T (2006) Parkin enhances mitochondrial biogenesis in proliferating cells. Hum Mol Genet 15:883–895. doi:10.1093/hmg/ddl006
Park J, Lee SB, Lee S, Kim Y, Song S, Kim S, Bae E, Kim J, Shong M, Kim JM, Chung J (2006) Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature 441:1157–1161. doi:10.1038/nature04788
Clark IE, Dodson MW, Jiang C, Cao JH, Huh JR, Seol JH, Yoo SJ, Hay BA, Guo M (2006) Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature 441:1162–1166. doi:10.1038/nature04779
Bonifati V, Rizzu P, van Baren MJ, Schaap O, Breedveld GJ, Krieger E, Dekker MC, Squitieri F, Ibanez P, Joosse M, van Dongen JW, Vanacore N, van Swieten JC, Brice A, Meco G, van Duijn CM, Oostra BA, Heutink P (2003) Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science 299:256–259. doi:10.1126/science.1077209
Zhang L, Shimoji M, Thomas B, Moore DJ, Yu SW, Marupudi NI, Torp R, Torgner IA, Ottersen OP, Dawson TM, Dawson VL (2005) Mitochondrial localization of the Parkinson’s disease related protein DJ-1: implications for pathogenesis. Hum Mol Genet 14:2063–2073. doi:10.1093/hmg/ddi211
Xu J, Zhong N, Wang H, Elias JE, Kim CY, Woldman I, Pifl C, Gygi SP, Geula C, Yankner BA (2005) The Parkinson’s disease-associated DJ-1 protein is a transcriptional co-activator that protects against neuronal apoptosis. Hum Mol Genet 14:1231–1241. doi:10.1093/hmg/ddi134
Strauss KM, Martins LM, Plun-Favreau H, Marx FP, Kautzmann S, Berg D, Gasser T, Wszolek Z, Muller T, Bornemann A, Wolburg H, Downward J, Riess O, Schulz JB, Kruger R (2005) Loss of function mutations in the gene encoding Omi/HtrA2 in Parkinson’s disease. Hum Mol Genet 14:2099–2111. doi:10.1093/hmg/ddi215
Martins LM, Morrison A, Klupsch K, Fedele V, Moisoi N, Teismann P, Abuin A, Grau E, Geppert M, Livi GP, Creasy CL, Martin A, Hargreaves I, Heales SJ, Okada H, Brandner S, Schulz JB, Mak T, Downward J (2004) Neuroprotective role of the Reaper-related serine protease HtrA2/Omi revealed by targeted deletion in mice. Mol Cell Biol 24:9848–9862. doi:10.1128/MCB.24.22.9848-9862.2004
Schapira AH, Bezard E, Brotchie J, Calon F, Collingridge GL, Ferger B, Hengerer B, Hirsch E, Jenner P, Le Novere N, Obeso JA, Schwarzschild MA, Spampinato U, Davidai G (2006) Novel pharmacological targets for the treatment of Parkinson’s disease. Nat Rev Drug Discov 5:845–854. doi:10.1038/nrd2087
Schapira AH (2002) The “new” mitochondrial disorders. J Neurol Neurosurg Psychiatry 72:144–149. doi:10.1136/jnnp.72.2.144
Tabrizi SJ, Cleeter MW, Xuereb J, Taanman JW, Cooper JM, Schapira AH (1999) Biochemical abnormalities and excitotoxicity in Huntington’s disease brain. Ann Neurol 45:25–32. doi:10.1002/1531-8249(199901)45:1<25::AID-ART6>3.0.CO;2-E
Mann VM, Cooper JM, Javoy-Agid F, Agid Y, Jenner P, Schapira AH (1990) Mitochondrial function and parental sex effect in Huntington’s disease. Lancet 336:749. doi:10.1016/0140-6736(90)92242-A
Cui L, Jeong H, Borovecki F, Parkhurst CN, Tanese N, Krainc D (2006) Transcriptional repression of PGC-1alpha by mutant huntingtin leads to mitochondrial dysfunction and neurodegeneration. Cell 127:59–69. doi:10.1016/j.cell.2006.09.015
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Dr. Anna Maria Giuffrida-Stella.
Rights and permissions
About this article
Cite this article
Schapira, A.H.V. Mitochondrial Dysfunction in Neurodegenerative Diseases. Neurochem Res 33, 2502–2509 (2008). https://doi.org/10.1007/s11064-008-9855-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9855-x