Abstract
Recent researches indicate that PGRN is closely related to diabetes and is regarded as a novel adipokine associated with obesity development, affecting adipocyte biology. In the present study, we investigated the effects and mechanisms of PGRN on porcine preadipocytes differentiation. Porcine preadipocytes were induced to differentiation with the addition of lentivirius-expressed PGRN shRNA at the early or late stage of induction period, and in the presence or absence of recombinant PGRN protein. The effects of PGRN on adipogenic genes expression and ERK activation were investigated. At the early stage of induction, knockdown of PGRN promoted differentiation, evidenced by enhanced lipid accumulation, upregulation of adipocyte markers, as well as master adipogenic transcription factors, PPARγ and C/EBPα. While, decreasing PGRN expression at the late stage of induction (day 3) had no effect on differentiation. These results suggested that PGRN functions in the early adipogenic events. Conversely, porcine preadipocytes differentiation was impaired by MDI and recombinant PGRN protein induction, the expressions of adipocyte markers were decreased. Further studies revealed that PGRN can specifically facilitate ERK1,2 activation, and this activation can be abolished by U0126. Moreover, PPARγ phosphorylation at serine 112 site was increased by PGRN treatment, which could reduce the transcriptional activity of PPARγ. We conclude that PGRN inhibits adipogenesis in porcine preadipocytes partially through ERK activation mediated PPARγ phosphorylation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity characterized by excess fat accumulation, associated with adipocytes hypertrophy and hyperplasia [1], is a major risk factor for the development of a broad range of diseases, including type II diabetes, cardiovascular diseases and cancer [2, 3]. Recent years, researches indicate that white adipose tissue (WAT) plays an important role in the generation of secreted adipokines rather than store excess energy [4]. These adipokines contain leptin, adiponectin, resistin, vaspin, tumor necrosis factor (TNF)-α, interleukins (ILs), which play important roles in regulating systemic processes, such as food intake and nutrient metabolism, insulin sensitivity, stress responses, reproduction, inflammation and so on [5–7].
Progranulin (PGRN), also known as proepithelin, granulin/epithelin precursor (GEP), acrogranin or PC cell-derived growth factor (PCDGF) [8]. Human PGRN is a 68.5 kDa secreted protein with a signal sequence and seven and one-half granulin domains connected by short linkers [9], these granulin domains are composed of highly conserved tandem repeats of a unique 12-cysteine motif [10]. PGRN is found in most eukaryotes, from yeast to arthropods, chordata and plants [11], but it is absent in drosophila [12]. It is widely expressed in tissues, abundantly expressed in rapidly cycling epithelial cells, immune system cells, and neurons [13]. PGRN has been shown to be a pluripotent growth factor that plays a role in many important biological processes, such as cell-cycle progression, wound healing and chondrogenesis [8]. PGRN is also implicated in various disease states, including breast and ovary cancers [14], neurodegenerative diseases [15], and rheumatoid arthritis [16].
In addition to this, recent researches indicate that PGRN is closely related to obesity development, involving in insulin resistance (IR), adipose tissue inflammation and food intake regulation. It was reported that PGRN is highly expressed in omental adipose tissue [17] and elevated serum PGRN is associated with visceral obesity, elevated plasma glucose and dyslipidemia [18]. Moreover, it’s found that inhibition of hypothalamic PGRN expression or action increased food intake and promoted weight gain, suggesting that endogenous PGRN functions as an appetite suppressor in the hypothalamus [19]. These data revealed that PGRN functions as a key cytokine participating in regulation of whole body metabolic balance.
However, to the best of our knowledge, the pathophysiological significance of elevated PGRN level in these obesity-related diseases remains unknown; the function of PGRN in the development of obesity has not been clarified. Therefore, investigating the function of PGRN in preadipocytes growth and differentiation may provide pivotal information for understanding the mechanisms underlying the association of PGRN and obesity. In this study, we used pig preadipocytes as experimental model and aimed to find the effects and mechanisms of PGRN in the process of differentiation.
Materials and methods
Reagents
Dulbecco’s Modified Eagle Medium (DMEM)/F12, fetal bovine serum (FBS), and type I collagenase were purchased from Gibco (Carlsbad, America). Oil red O was purchased from Sigma (Saint Louis, MO, America). Restriction enzyme BamHI and XhoI, Trizol, PrimeScript® RT reagent Kit (Perfect Real Time), and SYBR® Premix Ex Taq™ II were purchased from TaKaRa Biotechnology (Dalian, China). Lipofectamine 2000 was purchased from Invitrogen (CA, America). Mouse monoclonal anti-C/EBPβ and β-actin, rabbit polyclonal anti-C/EBPα, PPARγ, Pref-1, FAS, SREBP-1c, ERK1,2, phospho-ERK1,2 (Thr202/Tyr204), SAPK/JNK, phospho-SAPK/JNK (Thr183/Tyr185), p38 MAPK, and phospho-p38 MAPK (Thr180/Tyr182) antibodies were purchased from Cell Signaling Technology (Danvers, America). Goat polyclonal anti-aP2 and HRP-conjugated donkey anti-goat, goat anti-rabbit and goat anti-mouse secondary antibodies were purchased from Santa Cruz Biotechnology (CA, America).
Collection of animal tissues
Five healthy male crossbred (Duroc × Yorkshire × Landrace) pigs (180-day-old) were purchased from the experimental farm of Northwest A&F University (Yangling, China). All pigs were handled in accordance with the guidelines of Northwest A&F University Animal Care Committee. Heart, liver, spleen, lung, kidney, pancreas, skeletal muscle, subcutaneous fat adipose were isolated, collected, quickly frozen in liquid nitrogen, and stored at −80 °C for later use.
Porcine primary preadipocytes culture and adipogenic differentiation
Preadipocytes were isolated under sterile conditions by the culture method with mild modification [20]. Briefly, subcutaneous adipose tissue was separated from the neck and back of piglets, and rinsed three times in phosphate-buffered saline (PBS) with 100 U/ml Penicillin and 100 μg/ml Streptomycin (P/S). The tissue was minced into approximate 1 mm3 sections and digested with 1 mg/ml type I collagenase at 37 °C for 1 h in a shaking water bath, followed by adding DMEM/F12 medium containing 10 % FBS to stop digestion, then filtrated through 200 μm nylon mesh. The preadipocytes were collected by centrifugation at 1,000×g for 5 min and resuspended with PBS for three times, and then treated with red cell lysis buffer (154 mM NH4Cl, 10 mM KHCO3, 0.1 mM EDTA) for 5 min at room temperature. The preadipocytes were collected and seeded in culture plates at a density of 5 × 104 cells/cm2 and cultured at 37 °C in humidified atmosphere with 5 % CO2. Culture medium was changed every 2 days. To induce differentiation, 2 days after confluence, cells were supplied with differentiation medium [DMEM/F12 containing 10 % FBS plus 0.5 mM 3-isobutyl-1-methylxanthine, 1 μM DEX, 5 μg/ml insulin (MDI)] for 2 days, and then cultured in maintain medium [DMEM/F12 containing 10 % FBS plus 5 μg/ml insulin] for another 6 days, and medium was replaced every 2 days.
Lentivirus production and infection
PGRN shRNA oligonucleotides, corresponding to 208-228 (shRNA1), 686–706 (shRNA2), 1625–1643 (shRNA3) of porcine PGRN mRNA (Accession Number: NM_001044578), were annealed and cloned into plenti-H1 plasmid (between BamHI and XhoI sites) respectively. A scrambled oligonucleotides expressing vector was constructed, serving as control. Plasmids (10 μg plenti-PGRN shRNAs, 6 μg ∆8.9 and 9 μg VSVG) were co-transfected into 90 % confluent 293T cells in 100 mm plate with Lipofectamine 2000. 36 h and 72 h later, supernatant containing viral particles were collected, passed through 0.45 μm filters. For infection, cells were infected with lentivirus for 48 h, then the medium was replaced with MDI or maintain medium.
Real-time quantitative PCR analysis
Total RNA was extracted using Trizol reagent by standard procedure. 500 ng of total RNA was processed into single strand cDNA using reverse transcription kits with random primers. Table 1 lists the primers used for each gene studied. Real-time quantitative PCR reactions were performed in triplicate using the SYBR green kit with a Bio-Rad iQ™5 system. PCRs were performed with a 3 min pre-incubation at 95 °C, followed by 40 cycles of 15 s at 95 °C and 30 s at 60 °C. PCR products were verified by melting curve analysis. The 2−∆∆Ct method was used to analyze the relative expression level. Briefly, equivalent amount of cDNA template was used for transcripts amplification. Average Ct values were calculated and the ∆Ct values were calculated by subtracting the β-actin average Ct value for each sample. Subsequently, ∆∆Ct values were calculated by subtracting the average ∆Ct values of the control group. The final fold differences were calculated as 2−∆∆Ct for each gene among treatments. These measurements were repeated 3 times.
Western blot analysis
The cellular protein was extracted with RIPA buffer (Applygen, China) supplemented with protease inhibitor (Roche, USA). The protein content was measured by BCA method. The extracted proteins were electrophoresed in 12 % SDS–polyacrylamide gel under reducing conditions, and electrotransfered to nitrocellulose membranes (Millipore, USA). After blocking in 5 % skim milk, the membranes were incubated with primary antibodies overnight at 4 °C. After washing three times with TBST (Tris buffered saline plus 0.1 % Tween 20) at room temperature, the membranes were hybridized with secondary antibody for 1 h at room temperature, and then washed three times with TBST. The targeted proteins were detected using the Gel Doc XR System as per the instructions of the manufacturer.
Oil red O staining and extraction analysis
Differentiated porcine adipocytes were washed three times with PBS and fixed with 10 % paraformaldehyde for 30 min at room temperature. After washing three times with PBS, cells were stained with 1 % filtered Oil red O for 30 min at room temperature. Then cells were visualized by phase-contrast microscopy. To quantify intracellular lipid accumulation, the stained lipid droplets were dissolved with 100 % isopropanol for 10 min. The optical density was measured at 520 nm by spectrophotometer.
Statistical analysis
All the data were obtained from one independent experiment carried out in triplicate. The main and interactive effects were analyzed by one-way ANOVA using SPSS13.0 software. When justified by one-way ANOVA, mean differences between individual groups were analyzed by one sample Student’s t test. Differences were considered statistically significant if P < 0.05 and P < 0.01.
Results
PGRN was expressed in adipose and was developmentally regulated
To determine the tissue specificity of PGRN in normal porcine tissues, we extracted mRNA from tissues of 180-day-old pigs and subjected to real-time quantitative PCR analysis. As shown in Fig. 1a, PGRN is highly expressed in a number of tissues, including kidney, heart and spleen. Moreover, PGRN is moderately expressed in subcutaneous fat. In addition, we compared PGRN expression levels in preadipocytes and mature adipocytes, and we found preadipocytes are the dominant site of PGRN mRNA expression within the fat pad (Fig. 1b).
PGRN was expressed in adipose tissue in vivo and in vitro. a PGRN mRNA expression levels in different tissues of 180-day-old pigs were determined by real-time quantitative PCR. Liver serves as the control organ. b Subcutaneous fat tissue was further separated into preadipocytes and mature adipocytes. RNAs were extracted to perform real-time quantitative PCR for PGRN expression analysis. c The primary pig preadipocytes were induced to differentiation with MDI medium. RNAs were collected at the indicated time points (0, 1, 2, 4, 6, 8, 10 day) and were subjected to real-time quantitative PCR. Values represent the mean ± SEM. *P < 0.05, **P < 0.01
Next, primary porcine preadipocytes were used to assess PGRN expression over the course of adipogenesis. As differentiation proceeds, PGRN mRNA slightly declined on day 1, and then gradually increased to the maximum (about 4 fold of day 0’s level) on day 6, and after that it decreased by approximately 25 % on day 10, about 2.5 fold over day 0’s level (Fig. 1c).
Interference of PGRN promoted porcine preadipocytes differentiation and key adipogenic transcription factors expression
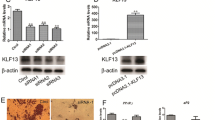
The developmental pattern of PGRN expression suggested that this gene might act as a regulator in adipogenesis. To test this hypothesis, three lentivirus-mediated shRNAs interfering of PGRN expression were constructed, and reduction of PGRN expression was performed during adipogenesis. As shown in Fig. 2A, shRNA1-3 can reduce PGRN mRNA expression, and the shRNA2 was the most effective one that can reduce 75 % of PGRN expression, then shRNA2 lentivirus was chosen to knockdown PGRN expression in the following experiments. Preadipocytes were infected with scrambled and shRNA2 lentivirus for 48 h, and then were induced to differentiation with MDI medium. Adipocyte formation, lipid accumulation, and adipocyte genes expression were evaluated on day 8. As shown in Fig. 2B, the shRNA2 treatment significantly promoted porcine preadipocytes differentiation. Lipid accumulation was also significantly enhanced in the shRNA2-treated cells (Fig. 2C). Moreover, the mRNA expression levels of adipocyte marker genes were robustly increased in shRNA2-treated cells (Fig. 2D), including PPARγ and aP2, increased about 7-fold and 7.3-fold respectively, SREBP-1c and LPL were also increased about 4.5-fold and 2.2-fold respectively. These results were also confirmed by Western blot (Fig. 2E). The protein expression levels of FAS, SREBP-1c, PPARγ and C/EBPα were enhanced in shRNA2 treated group, meanwhile, the expression of Pref-1 was reduced.
Interference of PGRN expression at the early stage of MDI induction promoted the preadipocytes differentiation. a Interference efficiency of PGRN shRNAs was determined by real-time quantitative PCR. Preadipocytes were not infected or infected with scrambled, shRNA1, shRNA2, shRNA3 lentivirus, RNAs was collected for real-time quantitative PCR 48 h later. b The primary preadipocytes were not infected (a) or infected with scrambled (b), shRNA2 (c) lentivirus for 48 h, and then were induced to differentiation. Oil red O staining was performed on day 8. c Lipid content was quantified by spectrophotometer at the absorbance of OD 520 nm. d mRNA expression levels of adipocyte genes PPARγ, SREBP-1c, aP2 and LPL were analyzed by real-time quantitative PCR. e Protein expression levels of FAS, SREBP-1c, PPARγ, C/EBPα, Pref-1 and aP2 were detected by Western blot. No infected cells serve as Control. Values represent the mean ± SEM. **P < 0.01
Interference of PGRN had no effect on late stage differentiation in porcine adipocytes
To determine whether PGRN had an effect on mature adipocyte differentiation, shRNA2 lentivirus was added to differentiating cells on day 3, and mature adipocyte formation, lipid accumulation and expression levels of key adipogenic factors were evaluated on day 8. The results showed that PGRN shRNA2 lentivirus did not affect mature adipocyte formation and lipid accumulation (Fig. 3A, B). In addition, the real-time quantitative PCR results indicated that shRNA2 lentivirus infection during the late stage of adipocyte differentiation did not alter the expression level of adipocyte markers, including PPARγ, SREBP-1c, aP2 and LPL (Fig. 3C). Consistent with this, western blot results showed that no significant differences were detected between three groups (Fig. 3D). These results suggested that interference of PGRN expression had no effect on porcine adipocytes terminal differentiation.
Reduction of the PGRN expression had no effect on late-stage differentiation in pig primary preadipocytes. A The differentiated preadipocytes were not infected (a) or infected with scrambled (b) and shRNA2 (c) lentivirus on day 3 for 48 h, and then were kept in maintain medium to day 8 till Oil red O staining was performed. B Lipid content was quantified by spectrophotometer at the absorbance of OD 520 nm. C mRNA levels of adipocyte genes PPARγ, SREBP-1c, aP2 and LPL were analyzed by real-time quantitative PCR. D Protein expression levels of FAS, SREBP-1c, PPARγ, C/EBPα, Pref-1 and aP2. Values represent the mean ± SEM
Recombinant PGRN protein inhibited porcine preadipocytes differentiation, attenuated adipogenic transcription factors expression
To further confirm the effect of PGRN on porcine preadipocytes differentiation, bioactive recombinant PGRN protein was used in the experiments. Postconfluent porcine preadipocytes were exposed to differentiation medium in the presence or absence of PGRN. Adipocyte formation, lipid accumulation, and adipocyte genes expression were evaluated on day 8. The Oil red O staining results showed that preadipocytes differentiation was significantly promoted by MDI (Fig. 4A-c) and was remarkably inhibited by addition of PGRN protein (Fig. 4A-d), it seems that preadipocytes differentiation was also impaired by PGRN treatment alone, but didn’t display significant effects (Fig. 4A-a, b). Compared with MDI induction, lipid accumulation was significantly reduced in the MDI+PGRN treated adipocytes (Fig. 4B). The real-time quantitative PCR results revealed that the mRNA expression levels of PPARγ, SREBP-1c, aP2 and LPL were decreased by MDI+PGRN treatment, comparing with MDI treatment alone (Fig. 4C). Western blot results showed that C/EBPα, Pref-1 and aP2 were decreased significantly in MDI+PGRN treated cells. (Fig. 4D).
PGRN treatment attenuated pig preadipocytes differentiation. Postconfluent preadipocytes were treated with PGRN (100 ng/ml) during differentiation. A Intracellular lipid was stained by Oil red O on day 8. (a) nondifferentiated control cells; (b) nondifferentiated cells treated with PGRN; (c) MDI treated cells; (d) MDI and PGRN treated cells. B Lipid content was quantified by spectrophotometer at the absorbance of OD 520 nm. C mRNA levels of adipocyte genes PPARγ, SREBP-1c, aP2 and LPL were analyzed by real-time quantitative PCR on day 8 after induction. D Protein expression levels of FAS, SREBP-1c, PPARγ, C/EBPα, Pref-1 and aP2. Values represent the mean ± SEM. *P < 0.05, **P < 0.01
PGRN specifically activated the MDI-induced phosphorylation of ERK1,2
It’s reported that PGRN can activate ERK signaling pathway in different cell types, including tumor cells and epithelial cells [11]. However, its potential effect on adipocyte has not been substantiated. Therefore we tested whether PGRN could activate ERK in the porcine preadipocytes. The porcine preadipocytes were treated with MDI medium plus PGRN, and the extent of phosphorylation of ERK1,2 (Thr202/Tyr204) was examined. The results showed that ERK1,2 phosphorylation was increased in a time-dependent manner in the cells which were incubated with 100 ng/ml PGRN, whereas the abundance of ERK1,2 was not affected (Fig. 5a). In control group, the ERK1,2 phosphorylation was peaked at 30 min, and then decreased to basal level at 60 min. Whereas in PGRN-treated group, the earliest increase in ERK1,2 phosphorylation was peaked at 15 min, and then maintained at a stable level. Meanwhile, phosphorylation of p38 MAPK and JNK was not affected by PGRN treatment.
Effects of PGRN on phosphorylation of ERK, p38 and JNK during early differentiation of porcine preadipocytes. a Postconfluent primary porcine preadipocytes were treated with MDI medium plus 100 ng/ml PGRN. Cell lyses were harvested at the indicated times and then subjected to western blot analysis detecting p-ERK1,2, total ERK1,2, p-p38 MAPK, total p38 MAPK, p-JNK, and total JNK expression levels. b, c Phosphorylated ERK1,2 levels were determined in preadipocytes cultured in MDI medium and supplemented with PGRN. When indicated, 10 μM U0126 was provided 1 h prior to MDI and PGRN treatment. b Western blot analysis of phosphorylated and total ERK1,2 under the experimental conditions. c The relative abundance of p-ERK1,2/ERK1,2 ratio. The p-ERK1,2/ERK1,2 ratio was normalized with respect to the adipogenic conditions (control). Values represent the mean ± SEM. **P < 0.01
To further confirm ERK1,2 activation in response to PGRN, MEK specific inhibitor U0126 was explored. Pre-exposure of preadipocytes to the U0126 prevented the PGRN-dependent increase in p-ERK1,2 (Fig. 5b). Relative abundance results showed that the ratio of p-ERK1,2/ERK1,2 was 2.397 ± 0.18 in the cells which were maintained in PGRN containing medium Whereas, in the presence of U0126, the value decreased, was 0.22 ± 0.011, meanwhile, in PGRN and U0126 incubated cells, the ratio was decreased to a similar level as U0126-treated cells.
PGRN increased ERK-mediated phosphorylation of PPARγ
PPARγ, standing out as a master regulator in the development of adipocytes, can be phosphorylated by ERK1,2 at serine 112 site, and this phosphorylation leads to the reduction of transcriptional activity [21–25]. As shown in the previous results, PGRN can specifically activate ERK1,2, we want to know whether PGRN have effects on PPARγ expression as well as phosphorylation. Comparing with MDI treatment, MDI and PGRN treatment did not cause a significant change in the relative abundance of PPARγ (Fig. 6a). Whereas, phosphorylation of PPARγ at serine 112 site was increased in the cells which were exposed to PGRN throughout the differentiation (Fig. 6a). Meanwhile, we also measured the phosphorylation of PPARγ in the PGRN knockdown cells. In shRNA2-treated cells, the phosphorylation of PPARγ was decreased (Fig. 6b). These results suggested that PGRN can increase PPARγ phosphorylation at the serine 112 site.
PGRN activated PPARγ phosphorylation at serine 112 site. a Postconfluent preadipocytes were treated with MDI, MDI plus 100 ng/ml PGRN for 8 days. The p-PPARγ and PPARγ were measured by western blot. b The primary preadipocytes were infected with scrambled, shRNA2 lentivirus for 48 h, and then were induced to differentiation. Western blot was performed on day 8 for p-PPARγ and PPARγ
Discussion
In the present study, we demonstrated for the first time that PGRN, a newly indentified adipokine, inhibits adipogenesis in porcine preadipocytes. Furthermore, we show that one mechanism whereby PGRN inhibits adipogenesis is by activating ERK1,2 signaling and subsequently inducing PPARγ phosphorylation. Interference of PGRN expression at the early stage of adipogenesis in porcine preadipocytes promoted differentiation, whereas knockdown of PGRN at the late stage has no effect. The inhibitory effect of PGRN on adipogenesis was also confirmed by recombinant human PGRN protein treatment study. We found that the adipogenesis of porcine preadipocytes was significantly inhibited by 100 ng/ml PGRN treatment. These results collectively indicate that PGRN is a potent inhibitor on adipogenesis in porcine preadipocytes.
PGRN was first purified as a growth factor from condition media, which functions as a mitogen for 3T3 cells as well as an autocrine growth factor for PC cells [26]. PGRN is ubiquitously expressed in various cell types and exhibits diverse functions [13]. In adult rat, PGRN is markedly expressed in spleen, lung, kidney and heart [27]. Consistent with the rat expression pattern, we observed that PGRN was highly expressed in kidney, heart and spleen in 180-day-old pigs. PGRN was also found to be predominantly expressed in omental adipose tissue of obese patients [17]. Our previous study showed that PGRN was highly expressed in subcutaneous adipose tissue of obesity-type and old pigs [28]. PGRN was predominantly detected in macrophages, as the development of obesity, macrophages infiltrate into adipose tissue, participating in inflammatory response and adipocyte biology regulation [29, 30]. In our study, mature adipocytes and preadipocytes were separated by digestion and low speed centrifugation, the macrophages were departed from adipocytes, but can mixed together with preadipocytes [31]. So under this circumstance, high expression of PGRN can be found in preadipocytes, which consists with the results obtained from mice [29]. It may suggest that macrophages is a source of PGRN. The serum concentration of PGRN is significantly higher in the type 2 diabetes patients, and is associated with omental obesity, elevated plasma glucose and dyslipidemia, and PGRN is regarded as a novel marker of chronic inflammation [18]. In addition to this, it’s found that alteration of PGRN production or action in hypothalamic may be linked to appetite dysregulation in obesity [19]. Recently, Matsubara demonstrated that PGRN is a key adipokine that mediates high fat diet induced IR and obesity [29]. These results collectively indicate that PGRN was involved in the regulation of energy homeostasis, and may play a key role in the development of obesity, IR and type 2 diabetes, but the expression and function of PGRN in adipocytes development has not been investigated. In current study, we found that PGRN was differentially expressed during the course of adipogenesis. It’s highly expressed at the late stage maybe due to residual macrophages and dexamethasone induction, a potential explanation could be that PGRN may involve in IR in the mature adipocytes. This was proved by Matsubara [29]. Through knockdown and recombinant protein treatment studies, we found that PGRN inhibits porcine preadipocytes differentiation, suggesting a direct inhibitory effect of PGRN on adipocytes differentiation. Moreover, PGRN abolished preadipocytes differentiation at the early stage of MDI induction.
The activation of ERK1,2 in different stages plays different role in adipogenesis. Activation of ERK1,2 at the early stage promotes adipogenesis by enhancing PPARγ and C/EBPα expression [32], while sustained activation of ERK1,2 signaling inhibits adipogenesis [33]. In the present study, we found that ERK1,2 phosphorylation was activated and maintained by PGRN treatment. As reported before, sustained activation of ERK1,2 impaired adipogenesis. Here, we found preadipocytes differentiation was attenuated by PGRN treatment. We also found that phosphorylation of PPARγ at serine 112 site was increased in PGRN-treated cells. This phosphorylation would decrease ligand-binding affinity of PPARγ, reduce the transcriptional activity, and eventually impair adipogenesis [21–25]. By the knockdown experiments, only interference of PGRN at the early stage can promote preadipocytes differentiation. This suggests that there may be an underlying pathway involving in accelerating adipogenesis by decreasing PGRN expression. Based on the existing results, we hypothesize that knockdown of PGRN may decrease the ERK1,2 phosphorylation after the initial adipogenic activation, and then maintain PPARγ at a lower phosphorylation level, finally enhance adipogenic differentiation. Thus, we can say that MAP kinase, a central regulator of cell growth, modifies PPARγ in a way that significantly reduces whose transcriptional activity, and impairs adipogenesis. In the previous reports, PGRN activates MAPK/ERK signaling pathway which has been widely illustrated [8, 18, 34]. However, the mechanism by which PGRN promotes ERK1,2 activation remains unresolved, which depends to a great extent on the biological receptors have not been exactly identified. Gonzalez et al. [35] found that PGRN interacted with the extracellular six EGF-like repeats in Dlk1. Besides, Hu et al. [36], found GRN peptide can bind to SORT1 which mediated endocytosis determines level It is also demonstrated that PGRN binds to TNFR and disturbs the TNFα–TNFR interaction [16]. All of these reports indicated that PGRN involves in adipocyte biology. But till to the recent time, no research demonstrates which molecule responses to PGRN’s action involving in the regulation of adipocyte biology. Therefore, elucidating the potential interactions between PGRN and individual components in the adipogenic pathway may provide insight into the mechanism of PGRN in adipogenesis.
In summary, our studies provided evidence for inhibition effect of PGRN on porcine primary preadipocytes differentiation, and suggested that PGRN may function as a negative regulator in adipogenesis by regulating ERK-mediated PPARγ phosphorylation. These results provide molecular information for further investigation of the mechanisms by which PGRN regulates adipocyte biology. Furthermore, these results could be important in devising mechanism-based and targeted therapeutic strategies for obesity.
References
Zaragosi L-E, Wdziekonski B, Villageois P, Keophiphath M, Maumus M, Tchkonia T, Bourlier V, Mohsen-Kanson T, Ladoux A, Elabd C, Scheideler M, Trajanoski Z, Takashima Y, Amri E-Z, Lacasa D, Sengenes C, Ailhaud G, Clément K, Bouloumie A, Kirkland JL, Dani C (2010) Activin A plays a critical role in proliferation and differentiation of human adipose progenitors. Diabetes 59(10):2513–2521
Arner P, Bernard S, Salehpour M, Possnert G, Liebl J, Steier P, Buchholz BA, Eriksson M, Arner E, Hauner H, Skurk T, Ryden M, Frayn KN, Spalding KL (2011) Dynamics of human adipose lipid turnover in health and metabolic disease. Nature 478(7367):110–113
Schwartz MW, Porte D (2005) Diabetes, obesity, and the brain. Science 307(5708):375–379
Kershaw EE, Flier JS (2004) Adipose tissue as an endocrine organ. J Clin Endocrinol Metab 89(6):2548–2556
Maury E, Brichard SM (2010) Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol Cell Endocrinol 314(1):1–16
MacDougald OA, Burant CF (2007) The rapidly expanding family of adipokines. Cell Metab 6(3):159–161
Harwood HJ Jr (2012) The adipocyte as an endocrine organ in the regulation of metabolic homeostasis. Neuropharmacology 63(1):57–75
Feng JQ, Guo F-J, Jiang B-C, Zhang Y, Frenkel S, Wang D-W, Tang W, Xie Y, Liu C-J (2010) Granulin epithelin precursor: a bone morphogenic protein 2-inducible growth factor that activates Erk1/2 signaling and JunB transcription factor in chondrogenesis. FASEB J 24(6):1879–1892
He Z, Bateman A (2003) Progranulin (granulin–epithelin precursor, PC-cell-derived growth factor, acrogranin) mediates tissue repair and tumorigenesis. J Mol Med (Berl) 81(10):600–612
Shoyab M, McDonald VL, Byles C, Todaro GJ, Plowman GD (1990) Epithelins 1 and 2: isolation and characterization of two cysteine-rich growth-modulating proteins. Proc Natl Acad Sci USA 87(20):7912–7916
De Muynck L, Van Damme P (2011) Cellular effects of progranulin in health and disease. J Mol Neurosci 45(3):549–560
Kamrava M, Simpkins F, Alejandro E, Michener C, Meltzer E, Kohn EC (2005) Lysophosphatidic acid and endothelin-induced proliferation of ovarian cancer cell lines is mitigated by neutralization of granulin–epithelin precursor (GEP), a prosurvival factor for ovarian cancer. Oncogene 24(47):7084–7093
Daniel R, He Z, Carmichael KP, Halper J, Bateman A (2000) Cellular localization of gene expression for progranulin. J Histochem Cytochem 48(7):999–1009
Lu R, Serrero G (2000) Inhibition of PC cell-derived growth factor (PCDGF, epithelin/granulin precursor) expression by antisense PCDGF cDNA transfection inhibits tumorigenicity of the human breast carcinoma cell line MDA-MB-468. Proc Natl Acad Sci USA 97(8):3993–3998
Cruts M, Van Broeckhoven C (2008) Loss of progranulin function in frontotemporal lobar degeneration. Trends Genet 24(4):186–194
Tang W, Lu Y, Tian QY, Zhang Y, Guo FJ, Liu GY, Syed NM, Lai Y, Lin EA, Kong L, Su J, Yin F, Ding AH, Zanin-Zhorov A, Dustin ML, Tao J, Craft J, Yin Z, Feng JQ, Abramson SB, Yu XP, Liu CJ (2011) The growth factor progranulin binds to TNF receptors and is therapeutic against inflammatory arthritis in mice. Science 332(6028):478–484
Qiu J, Ni Y-h, Gong H-x, Fei L, Pan X-q, Guo M, Chen R-h, Guo X-r (2007) Identification of differentially expressed genes in omental adipose tissues of obese patients by suppression subtractive hybridization. Biochem Biophys Res Commun 352(2):469–478
Youn B-S, Bang S-I, Klöting N, Park JW, Lee N, Oh J-E, Pi K-B, Lee TH, Ruschke K, Fasshauer M, Stumvoll M, Blüher M (2009) Serum progranulin concentrations may be associated with macrophage infiltration into omental adipose tissue. Diabetes 58(3):627–636
Kim H-K, Shin M-S, Youn B-S, Namkoong C, Gil SY, Kang GM, Yu JH, Kim M-S (2011) Involvement of progranulin in hypothalamic glucose sensing and feeding regulation. Endocrinology 152(12):4672–4682
Li Y, Lu RH, Luo GF, Pang WJ, Yang GS (2006) Effects of different cryoprotectants on the viability and biological characteristics of porcine preadipocyte. Cryobiology 53(2):240–247
Diradourian C, Girard J, Pégorier J-P (2005) Phosphorylation of PPARs: from molecular characterization to physiological relevance. Biochimie 87(1):33–38
Hauser S, Adelmant G, Sarraf P, Wright HM, Mueller E, Spiegelman BM (2000) Degradation of the peroxisome proliferator-activated receptor γ is linked to ligand-dependent activation. J Biol Chem 275(24):18527–18533
Hosooka T, Noguchi T, Kotani K, Nakamura T, Sakaue H, Inoue H, Ogawa W, Tobimatsu K, Takazawa K, Sakai M, Matsuki Y, Hiramatsu R, Yasuda T, Lazar MA, Yamanashi Y, Kasuga M (2008) Dok1 mediates high-fat diet-induced adipocyte hypertrophy and obesity through modulation of PPAR-[gamma] phosphorylation. Nat Med 14(2):188–193
Hu E, Kim JB, Sarraf P, Spiegelman BM (1996) Inhibition of adipogenesis through MAP kinase-mediated phosphorylation of PPARγ. Science 274(5295):2100–2103
Kim K-A, Kim J-H, Wang Y, Sul HS (2007) Pref-1 (preadipocyte factor 1) activates the MEK/extracellular signal-regulated kinase pathway to inhibit adipocyte differentiation. Mol Cell Biol 27(6):2294–2308
Zhou J, Gao G, Crabb JW, Serrero G (1993) Purification of an autocrine growth factor homologous with mouse epithelin precursor from a highly tumorigenic cell line. J Biol Chem 268(15):10863–10869
Bateman A, Bennett H (1998) Granulins: the structure and function of an emerging family of growth factors. J Endocrinol 158(2):145–151
Li XJ, Yang H, Li GX, Zhang GH, Cheng J, Guan H, Yang GS (2012) Transcriptome profile analysis of porcine adipose tissue by high-throughput sequencing. Anim Genet 43(2):144–152
Matsubara T, Mita A, Minami K, Hosooka T, Kitazawa S, Takahashi K, Tamori Y, Yokoi N, Watanabe M, Matsuo E-i, Nishimura O, Seino S (2012) PGRN is a key adipokine mediating high fat diet-induced insulin resistance and obesity through IL-6 in adipose tissue. Cell Metab 15(1):38–50
Xu H, Barnes GT, Yang Q, Tan G, Yang D, Chou CJ, Sole J, Nichols A, Ross JS, Tartaglia LA, Chen H (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest 112(12):1821–1830
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr (2003) Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest 112(12):1796–1808
Prusty D, Park BH, Davis KE, Farmer SR (2002) Activation of MEK/ERK signaling promotes adipogenesis by enhancing peroxisome proliferator-activated receptor gamma (PPARgamma) and C/EBPalpha gene expression during the differentiation of 3T3-L1 preadipocytes. J Biol Chem 277(48):46226–46232
MacDougald OA, Mandrup S (2002) Adipogenesis: forces that tip the scales. Trends Endocrinol Metab 13(1):5–11
Zanocco-Marani T, Bateman A, Romano G, Valentinis B, He Z-H, Baserga R (1999) Biological activities and signaling pathways of the granulin/epithelin precursor. Cancer Res 59(20):5331–5340
Gonzalez EM, Mongiat M, Slater SJ, Baffa R, Iozzo RV (2003) A novel interaction between perlecan protein core and progranulin: potential effects on tumor growth. J Biol Chem 278(40):38113–38116
Hu F, Padukkavidana T, Vægter CB, Brady OA, Zheng Y, Mackenzie IR, Feldman HH, Nykjaer A, Strittmatter SM (2010) Sortilin-mediated endocytosis determines levels of the frontotemporal dementia protein, progranulin. Neuron 68(4):654–667
Acknowledgments
This work was funded by National Natural Science Foundation of China (U1201213), National Basic Research Program of China (2012CB124705), Major Projects for Genetically Modified Organisms Breeding (2013ZX08006-005) and The Reproduction Technology of National Pork Industry Technology Systems (CARS-36). We are grateful to members of our laboratory for critical reading of the manuscript and helpful discussion.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, H., Cheng, J., Song, Z. et al. The anti-adipogenic effect of PGRN on porcine preadipocytes involves ERK1,2 mediated PPARγ phosphorylation. Mol Biol Rep 40, 6863–6872 (2013). https://doi.org/10.1007/s11033-013-2804-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2804-z