Abstract
Abscisic acid (ABA), the hormone associated with seed maturation and stress responses, activates a signal transduction pathway culminating in stomatal closure. Reflecting the central position of this hormone in many plant processes, many genes involved in ABA signaling have been identified. However, biotechnological approaches manipulating these genes in guard cells have rarely been applied to crops with an objective of engineering drought tolerance. We have used the Arabidopsis bZIP transcription factor, ABF4, under the control of guard-cell-specific KST1 promoter for transformation of tobacco and tomato. Expression was shown to be guard-cell-specific, and ABF4-mGFP fusion proteins accumulated in guard cell nuclei. All transgenic lines showed significant increase in drought tolerance when seedlings or mature plants were subjected to prolonged water deficit. These plants exhibited significantly lower water loss per unit leaf area compared to wild type, higher water potential, and relative water contents during water deficit. Importantly, the plants did not develop the severe growth retardation frequently encountered when stress-related transcription factors were expressed constitutively, demonstrating the advantage of tissue- or cell-specific gene manipulations as useful tools to improve plant drought tolerance and also other water-related traits under water-limiting environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During their life cycle, plants are subjected to multiple environmental stresses such as water deficit, low temperature, flooding, or high salinity. Therefore, the ability of plants to adjust to adverse conditions in a timely fashion is critical for maintenance of productivity as well as for survival in prolonged inadequate conditions. Among environmental stresses, drought is one of the most devastating constraints for agriculture. Due to drought, nearly 40% loss of maize yield is estimated in North America alone (Boyer 1982). Because drought is one of the major global environmental bottleneck for agriculture, enhancing drought tolerance has immense economic impact (Umezawa et al. 2006).
Drought triggers various plant responses including the accumulation of the phytohormone ABA. ABA is a pivotal player by reducing water loss by inducing stomatal closure through changing the expression of many stress-responsive genes including the family of ABRE-binding factors/ABRE-binding bZIP proteins, ABFs/AREBs. Many ABA responsive genes feature consensus cis-acting elements known as ABA-responsive elements (ABREs) in their regulatory regions (Guiltinan et al. 1990; Yamaguchi-Shinozaki and Shinozaki 1994), and the most abundant ABREs contain the C/TACGTGGC sequence (Kang et al. 2002). Further detailed promoter analyses of ABA-responsive genes revealed that multiple ABREs or the combination of ABRE and coupling element (CE) are required for the expression (Narusaka et al. 2003; Yoshida et al. 2010). These ABA-responsive genes are upregulated by the binding of transcription factors such as ABFs/AREBs (Choi et al. 2000; Kang et al. 2002; Uno et al. 2000). ABF2/AREB1 and ABF4/ABRE2 are upregulated by dehydration and exogenous ABA (Choi et al. 2000; Kang et al. 2002; Uno et al. 2000), ultimately activating downstream genes.
Transgenic plants overexpressing Arabidopsis ABF3 or ABF4 were significantly more tolerant to water deficit than wild type in Arabidopsis (Kang et al. 2002) or in rice (Oh et al. 2005), and their seeds were hypersensitive to ABA in germination assays (Kang et al. 2002). Both 35S:ABF3 and 35S:ABF4 transgenic Arabidopsis exhibited reduced stomatal apertures compared to wild type Arabidopsis (Kang et al. 2002), indicating that drought tolerance of these transgenic plants is likely due to reduced water loss through the stomata. In plant species other than Arabidopsis, several AREB-like proteins were identified and used successfully to increase abiotic stress tolerance such as drought or salt stress (Hsieh et al. 2010; Jin et al. 2009; Kerr et al. 2017; Orellana et al. 2010; Wang et al. 2016). In tomato, two AREB-like proteins, SIAREB1 and SIAREB2, were identified and overexpression of SIAREB1 increased drought and salt tolerance of Arabidopsis or tomato (Hsieh et al. 2010; Orellana et al. 2010). Also, constitutive expression of ABFs/AREBs homologs from other plant species such as wheat TaAREB3 (Wang et al. 2016) and cotton GhABF2D (Kerr et al. 2017) enhanced drought tolerance, suggesting that ABF/AREB is functionally well conserved across plant species.
Higher level of stress-related transcription factors such as ABF/AREBs commonly causes undesirable dwarf phenotype (Kang et al. 2002; Kerr et al. 2017). As exogenous ABA inhibits plant growth, the dwarf phenotype of 35S:ABF3 and 35S:ABF4 transgenic Arabidopsis plants may have been due to an ABA-hypersensitive response caused by the transgene (Kang et al. 2002), and/or the persistently lower stomatal aperture resulted in reduced carbon fixation causing dwarfing. A similar phenotype was also observed in plants constitutively expressing stress-related transcription factors such as LeCBF1, AtCBF1, and AtCBF3 (Gilmour et al. 2000; Hsieh et al. 2002; Knight et al. 2004; Zhang et al. 2004). However, dwarf phenotypes caused by strong CaMV 35S promoter were not observed in transgenic plants expressing stress-responsive genes such as AtABF4 or AtCBF3 under the control of the ubiquitin1 promoter (Oh et al. 2005) or ABA-/drought-responsive RD29A promoter (Kasuga et al. 2004), indicating promoter selection as probably the most crucial parameter to obtain desirable outcomes without deleterious side effects. Since most water loss in plant occurs through stomatal pores, alternative way to improve drought tolerance can be targeted drought-responsive gene expression in guard cells. It was suggested that targeted gene expression under the control of a guard-cell-specific and drought-responsive promoter could be a very effective way to improve drought tolerance while minimizing side effects (Na 2005; Na and Metzger 2014). Recently, a few studies reported that guard-cell-specific targeted gene expression enhanced plant drought tolerance (Chen et al. 2016; Kelly et al. 2013; Lu et al. 2013; Lugassi et al. 2015; Na 2005). In these studies, guard-cell-specific promoter such as KST1, GC1, KAT1, or RAB18 promoter was used to induce stress-responsive genes in guard cells. Among them, RAB18 promoter showed guard-cell specificity and responsiveness to exogenous ABA.
In this study, we investigated whether guard-cell-specific expression of an Arabidopsis ABRE-binding protein, ABF4 (Choi et al. 2000; Uno et al. 2000), could improve drought tolerance of tobacco and tomato plants while minimizing deleterious effects on growth. We found that the expression of ABF4 under the control of the guard-cell-specific KST1 promoter (Plesch et al. 2001) was sufficient to increase drought tolerance in both crop species by reducing transpiration while significantly minimizing the dwarf phenotype caused by transgene expression, as compared to Arabidopsis plants with 35S:ABF4 (Kang et al. 2002).
Materials and methods
Generation of transgenic plants with KST1 (35S):ABF4-mGFP
A 1.4 kb of a full-length cDNA for Arabidopsis ABF4 gene was amplified by gene-specific primer pairs (F: 5′- ACGCGTCGACATGGGAACTCACATCAATTTC-3′, R: 5′-ACAGTCATGAACCATGGTCCGGTTAATGTC-3′), and a PCR fragment was cloned into the Sal1/Nco1 site of the pCambia 1302 binary vector to generate the ABF4-mGFP fusion gene. Then, a 0.3-kb KST1 PCR fragment or the CaMV 35S promoter from a modified pUC18 vector (Sheen et al. 1995) was ligated into the HindIII/Sal1 sites to generate the KST1(35S):ABF4-mGFP construct. The 0.3-kb KST1 promoter having a guard-cell-specific element (Plesch et al. 2001) was amplified from potato genomic DNA using KST1-specific primer pairs (F: 5′-CGCGGATCCATCTGCGTACAGTCTACCT –3′, R: 5′-CATGCCATGGGTCGACTATTATATATTGCTGCTTCTTT-3′). The PCR fragment was first cloned at BamH 1/Nco1 sites of modified pUC18 vector, digested with Hind III/Nco1, and the digest having KST1 promoter was cloned into the pCambia 1302 vector. Both pCambia vectors harboring KST1(35S):ABF4-mGFP were transferred into Agrobacterium tumefaciens GV3101 by electroporation and used for transformation of tobacco and tomato by following methods described previously (Horsch et al. 1998; Park et al. 2003). The transgenic KST1(35S):ABF4-mGFP plants having resistance to hygromycin (30 mgL−1) were selected and then verified for single-locus T-DNA insertion by examining 3:1 ratio of antibiotic (hygromycin) resistance. Homozygous transgenic tobacco lines were obtained from transgenic tobacco lines with 3:1 ratio of antibiotic resistance and used for further analysis. As for tomato, however, T2-segregating tomato lines with 3:1 ratio of antibiotic resistance were used for phenotypic analysis. Total RNA was isolated from 3-week-old tobacco seedlings in MS medium using an RNeasy® Plant Mini Kit (Qiagen USA, Valencia, CA) or from 1-month-old tomato plants in the greenhouse. Transgene expression was examined by RT-PCR (Takara, Madison, WI) using the following primers: forward primer 5′-ATGGGAACTCACATCAATTTC-3′, reverse primer 5′-ACAGTCATGAACCATGGTCCGGTTAATGTC-3′. GFP and brightfield images were photographed under the PCM2000 confocal laser scanning microscope (Nikon).
Protein extraction and Western blot analysis
Total protein was extracted from 1-month-old leaf tissues (approximately 100 mg) of wild type or transgenic tobacco plants. To extract protein, leaf tissues were ground using a mortar and pestle after adding 500 μL of SDS extraction buffer (0.125-M tris-HCl, 4% SDS, 20% glycerol, 2% 2-mercaptoethanol, pH 6.8). The slurry was transferred into a 2-mL Eppendorf tube and centrifuged for 3 min at 10,000 rpm at 4 °C. The supernatant was transferred to a new tube and mixed with 1.5 mL of 100% acetone. After 30-min incubation at 4 °C, the samples were centrifuged for 3 min at 10,000 rpm at 4 °C and the pellet was dissolved in 2X SDS protein loading buffer. Then, an equal volume of each sample was separated on 8% SDS-PAGE gel. After electrophoresis, the gel was stained by Coomassie blue staining buffer, and protein levels were determined based on intensity. Protein was transferred to poly (vinylidene difluoride) membrane (Bio-Rad) by standard techniques (Sambrook et al. 1989). The membrane was blocked in 1X TBS (10-mM Tris HCl, pH 8.0/150-mM NaCl) containing 1% (wt/vol) non-fat dry milk at room temperature for 1 h, incubated with anti-GFP rabbit polyclonal antibody (Molecular Probes, Eugene, OR, USA), and developed using peroxidase-conjugated secondary antibody (dilution ratio of 1:5000) by enhanced chemiluminescence (Amersham Pharmacia).
Measurement of water potential and relative water contents
For tobacco, two KST1:ABF4-mGFP transgenic (NK3 and NK27) and wild-type plants were grown in 2-L pots in the greenhouse for 7 weeks. After that, three plants from each of the three genotypes were used to measure the water potential using a WP4-T dewpoint potentiometer (Decagon, Pullman, WA, USA). For tomato, the water potential of 2-month-old tomato plants in the greenhouse was measured using a Scholander-type pressure chamber (the Model 1000 pressure chamber, PMS, Albany, OR, USA). Three to five plants from T2 heterozygous KST1(35S):ABF4 and wild-type plants were used for measurement of water potential between 12:00 pm to 2:00 pm (non-stressed conditions) or 4:00 pm to 6:00 pm (water-stressed conditions). At the same time, leaf tissues of similar size were collected from each plant to measure the relative water content (RWC).
To determine RWC, leaf tissues were used to measure fresh weight (FW) and turgid weight (TW). For FW, leaf tissues were weighed immediately after harvest, and for TW, the tissue was fully hydrated by placing it in a 15-mL polyethylene cup with 3–5 mL of distilled water and placed in a refrigerator at 4 °C for 1 day, and the weight was re-measured. The tissue was then dried in an oven at 60 °C for 1 day and the dry weight (DW) was determined. RWC was calculated using the following equation: RWC = (FW − DW)/(TW − DW).
Stress tolerance
Two KST1:ABF4-mGFP transgenic and wild-type tobacco seeds were sown in 10-cm diameter pots filled with soilless media (Metro-Mix 360, The Scotts-Sierra Co., Marysville, OH, USA). Pots were placed in the greenhouse with 16-h light conditions with regular irrigation for the first month. To apply a water deficit, water was withheld for 20 days. After 20-day water deprivation, pots with plants were re-watered and pictures were taken after 5-day recovery. Early wilting was observed from 2-month-old tobacco and 1-month-old tomato plants by withholding water for 1 day.
To examine how transgenic tomato plants respond to periodical water deficit, 10 seeds of each wild type (WT), KST1:ABF4-mGFP (LK34) or 35S:ABF4-mGFP (LS41) T2-segregating tomato plants, were sown in 2-L pots and grown for a month in the greenhouse. In the following month, these plants were treated with a 3- to 5-day deprivation of water each week until they exhibited severe wilting symptom. Plants were photographed after 1-month recovery with regular watering from severe water deficit.
Measurement of plant water usage
Two-week-old transgenic and wild-type tobacco or tomato seedlings in MS (Sigma) media were individually transferred into 2-L pots filled with soilless media and grown in the greenhouse for 2 months. For tobacco, 6 plants from transgenic or wild-type plants were harvested prior to recording daily water loss as a reference for leaf area, height, leaf number, and dry and fresh weight. Daily evapotranspiration was determined by lysimetry. Pots containing plants were weighed using a top loading balance twice a day at 11:00 am and 12:30 pm for 10 days for tobacco and for 23 days for tomato. The plants were irrigated each day at 11:30 am. Daily evapotranspiration was determined by calculating the difference in the weights of the pot at 12:30 pm and the weight at 11:00 am the following day. Transpiration was deduced by subtracting the average amount of evaporation from three 2-L pots filled with soilless media without plants. Transpiration per leaf unit area was determined by dividing water loss per each 2 h by the total leaf area.
Results and discussion
Generation of transgenic tobacco and tomato plants with Arabidopsis ABF4
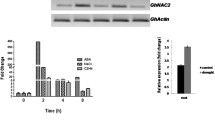
To study effects of the expression of the Arabidopsis ABF4 gene in guard cells, tobacco and tomato were transformed with pCambia-1302 binary vectors carrying an ABF4-mGFP fusion gene construct under the control of the guard-cell-specific 0.3-kb KST1 promoter (Muller-Rober et al. 1995; Plesch et al. 2001) or strong 35S CaMV promoter (Fig. 1a). For tobacco, 11 KST1:ABF4-mGFP transgenic lines were obtained from hygromycin selection and two T3 transgenic tobacco lines (designated NK3 and NK27) were used for further study. For tomato, two segregating T2 KST1:ABF4-mGFP lines (LK21 and LK34) and one segregating T2 35S:ABF4-mGFP line (LS41) were used for further analysis. The expression of transgene ABF4 was examined by RT-PCR and only detected in the transgenic tobacco or tomato plants (Fig. 1b, c). The expression of ABF4-mGFP was analyzed by Western blot analysis, and the expected 75-kD fusion protein was detected in transgenic tobacco plant using polyclonal GFP antibody (Fig. 1b).
Analysis of transgene expression in tobacco and tomato plants. a Construct used for plant transformation. Arabidopsis ABF4 gene was placed under the control of constitutive CaMV 35S or potato KST1 guard-cell-specific promoter. b RT-PCR analysis of ABF4 expression in wild type (WT) and two KST1:ABF4-mGFP homozygous transgenic tobacco lines (NK3, NK27). The expression of ABF4-mGFP fusion protein in NK3 transgenic plants was determined by Western blot with anti-GFP antiserum. c RT-PCR analysis of ABF4 expression in WT, KST1:ABF4-mGFP (LK21 and LK34) and 35S:ABF4-mGFP T2 transgenic tomato (LS41). Numbers (1 and 2) below LK21, LK34, and LS41 indicate two different transgenic lines used for RT-PCR analysis. d Localization of ABF4-mGFP fusion proteins in NK3 tobacco plant was observed under the PCM2000 confocal laser scanning microscope (Nikon). e Magnified and superimposed image of (d)
ABF4 accumulates strongly in nuclei of guard cells of transgenic tobacco plants
Transcription factor ABF4 activates dehydration-responsive genes such as RD29B upon binding on ABRE elements in their promoters, as was shown previously in in vitro or transient expression assays (Kang et al. 2002; Uno et al. 2000). To visualize the cellular localization of ABF4 in plants, ABF4 was fused to the mGFP reporter gene as shown in Fig. 1a. Heterologous ABF4-mGFP fusion protein accumulated in nuclei of guard cells in transgenic tobacco plants (Fig. 1d, e). The localization in guard cells is consistent with the previous demonstration that the KST1 promoter led to strong expression of GUS reporter gene in guard cells (Plesch et al. 2001). However, the ABF4-mGFP protein was also detected in the nuclei of epidermal cells, although it was weaker than in guard cells as indicated by an arrow (Fig. 1d, e). Nonetheless, the localization in nuclei is consistent with result from ABF4 transactivation assays in that ABF4 activates ABA-responsive genes upon binding to ABRE elements (Kang et al. 2002; Uno et al. 2000).
Guard-cell-specific ABF4 expression improves drought tolerance
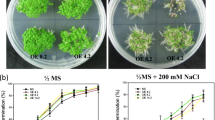
To examine whether ABF4 expression in guard cells improves drought tolerance as shown in Arabidopsis plants constitutively expressing ABF3 or ABF4 (Kang et al. 2002; Kim et al. 2004), KST1:ABF4-mGFP transgenic and wild-type plants were grown in pots in a greenhouse for 1 month with regular irrigation. For tobacco, transgenic plants, NK3 and NK27, exhibited minor wilting symptoms when water was withheld for 18 days, whereas wild-type plants were severely wilted. After 20 days, however, all lines had severely wilted (Fig. 2a). To test the ability to recover from the 20-day water deficit, the plants were irrigated and pictures taken 5 days later. Both transgenic lines, NK3 and NK27, recovered within 5 days after irrigation, whereas most wild-type plants were dead or did not fully recover (Fig. 2a).
KST1:ABF4-mGFP transgenic plants were more tolerant to water deficit than wild-type plants. a Water deficit and recovery of WT and transgenic tobacco plants, NK3 and NK27 (n = ± 100). b Two-month-old WT tobacco and c one-month-old WT tomato exhibited wilting symptom earlier than KST1/35S:ABF4-mGFP transgenic plants. d Pots containing 10 1-month-old plants of each WT tomato, T2-segregating KST1:ABF4-mGFP (LK34), or T2-segregating 35S:ABF4-mGFP (LS41) were periodically water-stressed by stopping irrigation for 1 month and followed by recovery for a month with regular watering. e Two transgenic and WT tomato plants were photographed to show the difference in growth between transgenic and WT from (d), and the red arrows indicate transgenic plants, LK34 and LS41. f The average height of transgenic or WT tomato plants from each pot from d was shown in graph. Number in parenthesis indicates number of WT or transgenic plants among 10 plants
We also examined whether ABF4 transgenic tobacco and tomato plants exhibited drought tolerance at an older stage of development during a typical sunny day when daily irrigation could not keep up with evapotranspiration. In 2-month-old wild-type tobacco plants, severe wilting was observed earlier than in KST1:ABF4-mGFP transgenic plants during the day (Fig. 2b). Also, as with tobacco, a similar result was observed in transgenic tomato plants. When wild-type tomato plants exhibited leaf bending as an early wilting symptom, KST1:ABF4-mGFP and 35S:ABF4-mGFP transgenic plants were not affected (Fig. 2c), indicating that the transgenic plants are more resistant to water deficit. These results are consistent with previous study, in which both Arabidopsis with 35S:ABF4 or 35S:ABF3 and rice plants with ubi1:ABF3 exhibited tolerance to water deficit (Kang et al. 2002; Oh et al. 2005).
Also, these results suggest that the expression of ABF4 under the control of the guard-cell-specific promoter KST1 improved drought tolerance in both species. However, the extent to which the low level of expression observed in epidermal cells of KST1:ABF4-mGFP plants contributes to the increase in drought tolerance is unknown (Fig. 1e, f). Recently, several studies reported that targeted gene expression in guard cells reduced water loss. Kelly et al. (2013) reported that hexokinase (HXK) expression in guard cell using KST1 promoter decreased water loss by transpiration in tomato plants. Also, phospholipase Da1 (PLDα1) expression under the control of guard-cell-specific KAT1 promoter reduced water loss while increasing seed production of canola, Brassica napus, under drought (Lu et al. 2013). Another study reported that Arabidopsis plants expressing a tobacco vacuolar invertase inhibitor, Nt-inhh, using an ABA-inducible and guard-cell-specific AtRab18 promoter, improved drought tolerance (Chen et al. 2016).
KST1:ABF4-mGFP transgenic plants exhibit mild pleiotropic growth effects
Although a stunted phenotype was not observed in early stages of development in either the transgenic tobacco or tomato plants (Fig. 2b, c), KST1:ABF4-mGFP tobacco (Tables 1 and 2) and KST1(35S):ABF4-mGFP tomato plants (Fig. 2b, c, d) exhibited mild growth reduction at later stages of development. The growth of the transgenic tobacco plants was reduced in leaf area by 8%, fresh weight by 9%, dry weight by 16%, and height by 18% compared to wild-type plants over a 3-month experimental period (Tables 1 and 2). Interestingly, these growth reductions were mainly attributed to those obtained during the last 10-day experimental period (Tables 1 and 2), suggesting that the stunting phenotype may be initiated depending on developmental progression after a certain amount of time and/or that the persistent presence of transgene may increase a sensitivity of the transgenic plants to water deficit.
In a previous study, Arabidopsis expressing 35S:ABF4 showed stunting, the severity of which correlated with the expression level of the ABF4 gene (Kang et al. 2002). Therefore, the mild stunting observed in KST1:ABF4-mGFP transgenic tobacco and tomato plants may be due to a relatively low expression level of the transgene ABF4. Clearly, transcript levels of ABF4 in 35S:ABF4-mGFP tomato plants were higher than the levels in the KST1:ABF4-mGFP lines, LK21 and LK34 (Fig. 1c), and the 35S:ABF4-mGFP lines exhibited a slightly more severe stunting than the KST1:ABF4-mGFP tomato plants, especially with water deficit (Fig. 2e), consistent with the observation that this phenotype in Arabidopsis was correlated with ABF4 expression driven by the 35S constitutive promoter (Kang et al. 2002). Another possible explanation is that the ABF4 transgene in either tobacco or tomato may require modification to exert its maximum effect to induce the expression of downstream-responsive genes. In Arabidopsis, ABF2 and ABF4 were highly activated by phosphorylation in an ABA-dependent manner (Choi et al. 2005; Furihata et al. 2006), suggesting that ABF4 transgenic tobacco or tomato plants may yet develop a more severe stunted phenotype with an increase in the endogenous ABA under water deficit.
Therefore, we had examined whether long-term water deficit would result in a stunting phenotype in the transgenic plants. For the test, water deficit was carried out with both 35S:ABF4-mGFP and KST1:ABF4:mGFP tomato plants by repeating a 3- to 5-day deprivation of water each week for a month. The KST1:ABF4-mGFP transgenic and wild-type tomato plants did not show any significant change in growth in the experiment. However, 35S:ABF4-mGFP tomato plants exhibited a very severe stunted phenotype and did not recover from the water deficit in the month following with regular irrigation (Fig. 2d, e; LS41). KST1:ABF4-mGFP tomato plants showed insignificantly lower growth during the prolonged water deficit compared to wild type, while the 35:ABF4-mGFP plants were severely reduced in height and biomass (Fig. 2f). This suggests that guard-cell-specific expression of ABF4 does not interfere with normal growth and probably that the mild stunting phenotype of KST1:ABF4-mGFP plants is attributable to leaky ABF4 expression in other cell types (Fig. 1d, e). Similarly, growth inhibition was compared by expressing HXK gene under the control of 35S or guard-cell-specific KST1 promoter (Kelly et al. 2013), in which 35S:HXK plants did show dwarfism while KST1:HXK plant grew normal like wild type, indicating that guard-cell-specific stress-responsive gene expression is useful tool to minimize dwarfism.
ABF4 transgenic plants conserve water by reducing transpiration
Since transgenic tobacco and tomato plants expressing ABF4-mGFP in guard cells exhibited drought tolerance, we examined whether the drought tolerance of these transgenic plants was due to altered stomatal activity. Daily water loss was measured by weighing pots with 2-month-old KST1(35S):ABF4-mGFP transgenic or wild-type plants. Daily water use patterns of transgenic tobacco (Fig. 3a) and tomato (Fig. 3b) plants were significantly lower than those of their wild-type counterparts. In addition, transgenic plants transpired less water per 100-cm2 unit leaf area than wild-type plants under normal conditions (Fig. 3c, d), and this result was coincident with the daily water use pattern of transgenic plants, indicating that lower daily water use of transgenic plants is likely due to reduced transpiration. The expression of ABF3 or ABF4 in Arabidopsis (Kang et al. 2002) and ABF3 in rice (Oh et al. 2005) conferred drought tolerance, and Arabidopsis with 35S:ABF3 or 35S:ABF4 showed reduced stomatal aperture (Kang et al. 2002), suggesting that both tomato and tobacco KST1:ABF4-mGFP plants may reduce transpiration via reduction in stomatal aperture.
KST1(35S):ABF4-mGFP transgenic plants conserve significantly more water than WT. Daily water loss of a NK3 and NK27 tobacco plants with KST1:ABF4-mGFP or b transgenic tomato plants LK21 and LK34 with KST1:ABF4-mGFP or LS41 with 35S:ABF4-mGFP was much less than that of WT (n = 12 for tobacco, n = 4 to 5 for tomato). Time-course of transpiration of c 3-month-old transgenic and WT tobacco plants or d 3-month-old tomato plants during the day (n = 4 for tobacco and n = 4 to 5 for tomato). Relative water content of e tobacco or f tomato plants. Water potential of g tobacco and h tomato plants. In f and g graphs, relative water content (RWC) and water potentials were measured at non-stressed conditions between 12:00 pm and 2:00 pm and water-stressed conditions between 4:00 pm and 6:00 pm. Vertical bars denote the mean ± SD (n ≥ 3 plants in triplicates for water potential, n = 6 plants in duplicate for relative water content)
Water use efficiency (WUE) of KST1:ABF4-mGFP tobacco plants was examined by dividing the gain of dry weight by the amount of water used during 10-day experimental period. Although WUE of transgenic tobacco plants was slightly higher than wild type, the difference was not significant (Table 2). It is unclear whether an extended experimental period can result in more difference. On the other hand, it can be speculated that ABF4 expression in guard cells does not affect WUE, but it may cause reduced stomatal aperture, resulting in reduced water loss. Consequently, the reduced stomatal aperture might lead lower CO2 assimilation due to reduced CO2 uptake, and in turn results in a mildly stunted phenotype of KST1:ABF4-mGFP plants without changing WUE.
Relative water content (RWC) and water potential of transgenic tobacco plants expressing KST1:ABF4-mGFP were measured only under well-watered conditions. RWC was significantly higher in transgenic tobacco than wild type (Fig. 3e), which was not consistent with KST1:ABF4-mGFP tomato plants that did not exhibit any difference in RWC from wild type without water deficit (Fig. 3f). However, water potential in transgenic tobacco versus wild type was not different (Fig. 3f), consistent with water potential of transgenic tomato plants under non-stressed conditions between 12:00 pm and 2:00 pm.
RWC of KST1:ABF4-mGFP transgenic tomato and wild type was similar under non-stress conditions during 12:00 to 2:00 pm in the early afternoon, but it was significantly higher in transgenic tomato than wild type under water-stressed conditions between 4:00 to 6:00 pm in the late afternoon (Fig. 3g) when wild type exhibited minor wilting. Consistent with RWC, water potential of both transgenic and wild-type tomatoes was similar under non-stressed conditions, but it was significantly higher in the transgenic tomato plants at the end of the day (water-stressed conditions during 4:00 pm to 6:00 pm) (Fig. 3h). These results make sense in that high transpiration of wild type can result in the depletion of water earlier than in KST1:ABF4-mGFP transgenic plants. As a result, transgenic plants can maintain higher water content as well as higher water potential longer than wild type, which can be critical for plant survival during periodic water deficit in field performance.
In summary, several studies have demonstrated that the guard-cell targeted manipulation of genes related to drought-adaptive mechanisms can be a useful tool to improve plant drought tolerance. Consistently with previous observations, we found that the expression of ABF4 in guard cells reduces transpiration and significantly improves drought tolerance of transgenic tobacco and tomato plants while minimizing undesirable side effects such as stunted growth.
References
Boyer JS (1982) Plant productivity and environment. Science 218(4571):443–448. https://doi.org/10.1126/science.218.4571.443
Chen SF, Liang K, Yin DM, Ni DA, Zhang ZG, Ruan YL (2016) Ectopic expression of a tobacco vacuolar invertase inhibitor in guard cells confers drought tolerance in Arabidopsis. Journal of Enzyme Inhibition and Medicinal Chemistry 31(6):1381–1385. https://doi.org/10.3109/14756366.2016.1142981
Choi H, Hong J, Ha J, Kang J, Kim SY (2000) ABFs, a family of ABA-responsive element binding factors. J Biol Chem 275(3):1723–1730. https://doi.org/10.1074/jbc.275.3.1723
Choi HI, Park HJ, Park JH, Kim S, Im MY, Seo HH, Kim YW, Hwang I, Kim SY (2005) Arabidopsis calcium-dependent protein kinase AtCPK32 interacts with ABF4, a transcriptional regulator of abscisic acid-responsive gene expression, and modulates its activity. Plant Physiol 139(4):1750–1761. https://doi.org/10.1104/pp.105.069757
Furihata T, Maruyama K, Fujita Y, Umezawa T, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K (2006) Abscisic acid-dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proc Natl Acad Sci U S A 103(6):1988–1993. https://doi.org/10.1073/pnas.0505667103
Gilmour SJ, Sebolt AM, Salazar MP, Everard JD, Thomashow MF (2000) Overexpression of the Arabidopsis CBF3 transcriptional activator mimics multiple biochemical changes associated with cold acclimation. Plant Physiol 124(4):1854–1865. https://doi.org/10.1104/pp.124.4.1854
Guiltinan MJ, Marcotte WR Jr, Quatrano RS (1990) A plant leucine zipper protein that recognizes an abscisic acid response element. Science 250(4978):267–271. https://doi.org/10.1126/science.2145628
Horsch RB, Fry J, Hoffmann N, Neidermeyer J, Rogers SG, Fraley RT (1998) Leaf disc transformation. In: Gelvin SB, Schilperoort RA, DPS V (eds) Plant molecular biology manual. Kluwer Academic Publishers, Dordrecht, pp A51–A59
Hsieh TH, Lee JT, Charng YY, Chan MT (2002) Tomato plants ectopically expressing Arabidopsis CBF1 show enhanced resistance to water deficit stress. Plant Physiol 130(2):618–626. https://doi.org/10.1104/pp.006783
Hsieh TH, Li CW, Su RC, Cheng CP, Sanjaya TYC, Chan MT (2010) A tomato bZIP transcription factor, SlAREB, is involved in water deficit and salt stress response. Planta 231(6):1459–1473. https://doi.org/10.1007/s00425-010-1147-4
Jin XF, Xiong AS, Peng RH, Liu JG, Gao F, Chen JM, Yao QH (2009) OsAREB1, an ABRE-binding protein responding to ABA and glucose, has multiple functions in Arabidopsis. BMB Rep 43:34–39
Kang JY, Choi HI, Im MY, Kim SY (2002) Arabidopsis basic leucine zipper proteins that mediate stress-responsive abscisic acid signaling. Plant Cell 14(2):343–357. https://doi.org/10.1105/tpc.010362
Kasuga M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2004) A combination of the Arabidopsis DREB1A gene and stress-inducible rd29A promoter improved drought- and low-temperature stress tolerance in tobacco by gene transfer. Plant & Cell Physiology 45(3):346–350. https://doi.org/10.1093/pcp/pch037
Kelly G, Moshelion M, David-Schwartz R, Halperin O, Wallach R, Attia Z, Belausov E, Granot D (2013) Hexokinase mediates stomatal closure. The Plant Journal : for cell and molecular biology 75(6):977–988. https://doi.org/10.1111/tpj.12258
Kerr TCC, Abdel-Mageed H, Aleman L, Lee J, Payton P, Cryer D, Allen RD (2017) Ectopic expression of two AREB/ABF orthologs increases drought tolerance in cotton (Gossypium hirsutum). Plant Cell Environ:n/a–n/a. https://doi.org/10.1111/pce.12906
Kim JB, Kang JY, Kim SY (2004) Over-expression of a transcription factor regulating ABA-responsive gene expression confers multiple stress tolerance. Plant Biotechnol J 2(5):459–466. https://doi.org/10.1111/j.1467-7652.2004.00090.x
Knight H, Zarka DG, Okamoto H, Thomashow MF, Knight MR (2004) Abscisic acid induces CBF gene transcription and subsequent induction of cold-regulated genes via the CRT promoter element. Plant Physiol 135(3):1710–1717. https://doi.org/10.1104/pp.104.043562
Lu S, Bahn SC, Qu G, Qin H, Hong Y, Xu Q, Zhou Y, Hong Y, Wang X (2013) Increased expression of phospholipase Dalpha1 in guard cells decreases water loss with improved seed production under drought in Brassica napus. Plant Biotechnol J 11(3):380–389. https://doi.org/10.1111/pbi.12028
Lugassi N, Kelly G, Fidel L, Yaniv Y, Attia Z, Levi A, Alchanatis V, Moshelion M, Raveh E, Carmi N, Granot D (2015) Expression of Arabidopsis hexokinase in citrus guard cells controls stomatal aperture and reduces transpiration. Front Plant Sci 6:1114. https://doi.org/10.3389/fpls.2015.01114
Muller-Rober B, Ellenberg J, Provart N, Willmitzer L, Busch H, Becker D, Dietrich P, Hoth S, Hedrich R (1995) Cloning and electrophysiological analysis of KST1, an inward rectifying K+ channel expressed in potato guard cells. EMBO J 14(11):2409–2416
Na JK (2005) Genetic approaches to improve drought tolerance of tomato and tobacco. Dissertation, The Ohio State University
Na JK, Metzger JD (2014) Chimeric promoter mediates guard cell-specific gene expression in tobacco under water deficit. Biotechnol Lett 36(9):1893–1899. https://doi.org/10.1007/s10529-014-1553-y
Narusaka Y, Nakashima K, Shinwari ZK, Sakuma Y, Furihata T, Abe H, Narusaka M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Interaction between two cis-acting elements, ABRE and DRE, in ABA-dependent expression of Arabidopsis rd29A gene in response to dehydration and high-salinity stresses. The Plant Journal: for cell and molecular biology 34(2):137–148. https://doi.org/10.1046/j.1365-313X.2003.01708.x
Oh SJ, Song SI, Kim YS, Jang HJ, Kim SY, Kim M, Kim YK, Nahm BH, Kim JK (2005) Arabidopsis CBF3/DREB1A and ABF3 in transgenic rice increased tolerance to abiotic stress without stunting growth. Plant Physiol 138(1):341–351. https://doi.org/10.1104/pp.104.059147
Orellana S, Yanez M, Espinoza A, Verdugo I, Gonzalez E, Ruiz-Lara S, Casaretto JA (2010) The transcription factor SlAREB1 confers drought, salt stress tolerance and regulates biotic and abiotic stress-related genes in tomato. Plant Cell Environ 33(12):2191–2208. https://doi.org/10.1111/j.1365-3040.2010.02220.x
Park SH, Morris JL, Park JE, Hirschi KD, Smith RH (2003) Efficient and genotype-independent Agrobacterium-mediated tomato transformation. J Plant Physiol 160(10):1253–1257. https://doi.org/10.1078/0176-1617-01103
Plesch G, Ehrhardt T, Mueller-Roeber B (2001) Involvement of TAAAG elements suggests a role for Dof transcription factors in guard cell-specific gene expression. The Plant Journal: for cell and molecular biology 28(4):455–464
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sheen J, Hwang S, Niwa Y, Kobayashi H, Galbraith DW (1995) Green-fluorescent protein as a new vital marker in plant cells. The Plant Journal: for cell and molecular biology 8(5):777–784. https://doi.org/10.1046/j.1365-313X.1995.08050777.x
Umezawa T, Fujita M, Fujita Y, Yamaguchi-Shinozaki K, Shinozaki K (2006) Engineering drought tolerance in plants: discovering and tailoring genes to unlock the future. Curr Opin Biotechnol 17(2):113–122. https://doi.org/10.1016/j.copbio.2006.02.002
Uno Y, Furihata T, Abe H, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K (2000) Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc Natl Acad Sci U S A 97(21):11632–11637. https://doi.org/10.1073/pnas.190309197
Wang J, Li Q, Mao X, Li A, Jing R (2016) Wheat transcription factor TaAREB3 participates in drought and freezing tolerances in Arabidopsis. Int J Biol Sci 12(2):257–269. https://doi.org/10.7150/ijbs.13538
Yamaguchi-Shinozaki K, Shinozaki K (1994) A novel cis-acting element in an Arabidopsis gene is involved in responsiveness to drought, low-temperature, or high-salt stress. Plant Cell 6(2):251–264. https://doi.org/10.1105/tpc.6.2.251
Yoshida T, Fujita Y, Sayama H, Kidokoro S, Maruyama K, Mizoi J, Shinozaki K, Yamaguchi-Shinozaki K (2010) AREB1, AREB2, and ABF3 are master transcription factors that cooperatively regulate ABRE-dependent ABA signaling involved in drought stress tolerance and require ABA for full activation. The Plant Journal: for cell and molecular biology 61(4):672–685. https://doi.org/10.1111/j.1365-313X.2009.04092.x
Zhang X, Fowler SG, Cheng H, Lou Y, Rhee SY, Stockinger EJ, Thomashow MF (2004) Freezing-sensitive tomato has a functional CBF cold response pathway, but a CBF regulon that differs from that of freezing-tolerant Arabidopsis. The Plant Journal: for cell and molecular biology 39(6):905–919. https://doi.org/10.1111/j.1365-313X.2004.02176.x
Acknowledgements
We are most grateful to Dr. JC Jang and Dr. Biao Ding for providing various materials and equipment. This research was supported by the DC Kiplinger Endowment for Floriculture and a SEEDS Graduate Research Grant from the OARDC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Na, J.K., Metzger, J.D. Guard-cell-specific expression of Arabidopsis ABF4 improves drought tolerance of tomato and tobacco. Mol Breeding 37, 154 (2017). https://doi.org/10.1007/s11032-017-0758-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-017-0758-x