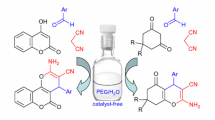
Abstract
An efficient, one-pot, three-component synthesis of various biologically important heterocyclic compounds is described via a tandem Knoevengel-cyclo condensation reaction of aromatic aldehydes, malono derivatives, and phenyl hydrazine derivatives in water and ethanol at room temperature.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrogen-linked heterocyclic compounds have received considerable attention in recent times due to their wide applications. The cyclization reaction of suitable linear compounds is one of the most common and popular methods for preparing these heterocyclic compounds [1–7]. Between these aza containing heterocyclic compounds, pyrazoles have a long history of application in agrochemical and pharmaceutical industries [8, 9]. These compounds are known to display anti-tumor [10, 11], anti-bacterial [12–14], anti-microbial [15–19], anti-fungal [20], anti-inflammatory [21, 22], analgesic [23, 24], anti-depressant [25], anti-convulsant [26], anti-pyretic activities [27]. anti-parasitic [28, 29], anti-malarial [30], anti-tumor [31], and anti-viral activities [32, 33]. It is well-known that the study of pyrazole derivatives is significant in pesticide chemistry, because of their herbicidal [34, 35], and insecticidal activities [36]. Moreover, applications of pyrazole containing compounds as ligands in coordination chemistry are well-documented in the literature [37, 38]. A previous investigation revealed that 5-amino-4-cyanopyrazole derivatives have anti-bacterial activity [39, 40]. The pyrazole motif makes the core structure of blockbuster drugs such as Celebrex(R) [41] and Viagra(R) [42] that act as PDE-5 inhibitors, Zoniporide [43] as sodium hydrogen ion exchanger inhibitors, 1-aryl-5-aminopyrazole as NPY5 antagonist [44], and PNU-32945 as HIV-reverse transcriptase inhibitors [45] (Fig. 1).
Many synthetic methods are available for the synthesis of pyrazole derivatives [46–50]. The most popular methods for the preparation of 1,3,4,5-tetrasubstituted pyrazoles are the reactions between 1,3-difunctional compounds with hydrazine derivatives (Scheme 1a) [51], 1,3-dipolar cycloadditions of diazo compounds onto triple bonds (Scheme 1b) [52, 53] and the oxidative N–N bond formation of enamines and nitriles (Scheme 1c) [54]. Another used approach is the functionalization of preformed trisubstituted pyrazoles by either nucleophilic substitution or transition metal catalyzed C-N bond formation (Scheme 1d) [55–60]. A survey of the literature shows that the majority of the strategies involve either multistep sequences, or expensive catalysts, inert atmosphere, anhydrous conditions, lengthy reaction times, and laborious workup.
It is well-known that nitriles are widely used as intermediates for a large number of heterocyclic compounds [61, 62]. In continuation of our research interest in the synthesis of biologically important heterocyclic compounds, we have synthesized a series of new pyrazole derivatives via a tandem Knoevengel cyclo-condensation reaction of aromatic aldehydes, malono derivatives, and phenyl hydrazinium chloride derivatives, in water and ethanol at room temperature (Scheme 2).
Results and discussion
To find the optimized reaction conditions, we initiated a catalyst screening exercise employing benzaldehyde (1 mmol), malononitrile (1 mmol), and phenylhydrazine (1 mmol) in the presence of various base catalysts such as \(\text{ Et }_{3}\)N, DABCO, DBU, NaOH, and \(\text{ K }_{2}\text{ CO }_{3}\) at room temperature. Screening of the reaction conditions established that the nature of the catalyst had no significant effect on the yield of pyrazole. Interestingly, in the absence of any base catalyst, this three-component coupling cyclization reaction proceeded smoothly to afford the desired 5-amino-4-cyano 1, 3 diphenyl pyrazole in excellent yield after 30 min at room temperature in the mixture of water and ethanol (1:1 v/v) as solvent. Therefore, the phenylhydrazine itself is acting as both a Brønsted base catalyst in this reaction and as a nucleophile. This is why the bases had no effect on the reaction yield. Hence, we monitored the effect of the amount of phenylhydrazine on the yield of reaction. With a higher amount of phenylhydrazine no increase in the yield of 5-amino-4-cyano 1, 3 diphenyl pyrazole is observed. However, diminishing the amount of phenylhydrazine resulted in incomplete conversion.
With these optimized conditions in hand, this three-component reaction can be readily diversified through a combination of a range of aryl aldehydes, malono derivatives, and phenyl hydrazine derivatives. Among the malono derivatives, malononitrile afforded excellent yields of products (Table 1, entries 1–17). Remarkably, low nucleophilic malono derivatives also gave products (Table 1, entries 21–24) in excellent yields. Similarly, dialdehydes were also successfully employed to give bis poly-substituted pyrazoles in excellent yields (Table 1, entries 18–20).
To explore the generality of the reaction, aldehydes with electron-withdrawing substituents on aromatic ring were also employed (Table 1, entries 5, 6, 7, 8, 23). It is worth mentioning that sterically bulky aldehydes were readily converted into the desired products (Table 1, entries 3, 7). To further expand the scope of the reaction, the use of heteroaryl aldehydes was investigated (Table 1, entries 11, 12).
Some aliphatic aldehydes were also screened to carry out the three-component coupling by this method and the results are listed in Table 1. However, no products were obtained when aliphatic aldehydes were involved in this one-pot room temperature catalyst-free reaction (Table 1, entries 24, 25). The trend observed due chiefly to the lower reactivity of alyphatic aldehydes toward nucleophilic addition in comparison with aromatic aldehydes.
The selectivity in the synthesis of 5-amino-1,3-aryl-1-1\(H\)-pyrazole-4-carbonitriles can be explained by the strict sequence of the reactions shown in Scheme 3. On the basis of this suggested mechanism, olefin (4) readily prepares in situ from Knoevenagel condensation of aromatic aldehyde (1) with a highly reactive malono derivative (2). Phenylhydrazine derivative (3), to be able to react easily with olefin (4) and to eventually give rise to the formation of intermediate (5), which then causes the inner molecular ring to be formed after a tautomeric proton shift to produce 2,3-dihydropyrazole derivative (7). Finally, atmospheric oxygen acts as an oxidant to convert 2,3-dihydropyrazole derivative to the corresponding pyrazole (8). In this study, we did not see any dependence of reaction time and yield to the order of addition of reactants to the reaction mixture. Therefore, another plausible mechanism can be possible for the formation of imine (6) from the reaction of aryl aldehyde (1) with phenylhydrazine derivative (3). Malono derivative (2), to be able to react with imine (6) to give rise to the formation of intermediate (5) that finally convert to pyrazole (8) (Scheme 3).
There are various reports in the literature that pyrazole to be formed through the oxidation of initially formed dihydropyrazole (pyrazoline), using atmospheric oxigen as oxidant (the reaction was run in air) [63] or nickel peroxide or manganese dioxide or DDQ in benzene [64–66] served as the oxidant for this transformation. However, because of the difficulties of the separation of pyrazoline intermediate even under an inert atmosphere, we selected isatin (an active ketone) as a model to clarify that this reaction proceed through the pyrazoline intermediate. The reaction of isatin (9) with malononitrile (2) and phenylhydrazine (3) yielded a spirosystem with 2-oxoindole and pyrazoline fragment (10). The first stage of the reaction potentially follows a classic mechanism of a nucleophilic attack of the malononitrile in position C3 on isatin throgh the formation of 2-(2-oxoindolin-3-ylidene)malononitrile followed by Michael addition of phenylhydrazine and then intramolecular cyclization with toutomerization to 5\(^\prime \)-amino-2-oxo-1\(^\prime \)- phenyl-1\(^\prime \), 2-dihydrospiro[indoline-3,3\(^\prime \)-pyrazole]-4-carbon-itrile (Scheme 4).
Thus, from a practical point of view, the newly developed protocol is a significant proof of the fact that nitrile is one of the most versatile functional groups as it can be readily transformed into various other functional groups. Significantly, the reaction occurred in a catalyst-free fashion with high selectivity and atom economy. To our knowledge, the use of catalyst-free reactions, namely Knoevenagel reaction, Michael-type reaction, ring closure, and subsequent aromatization, in one pot has not been previously reported.
Conclusions
In conclusion, we have disclosed a novel and convenient one-pot synthesis of polysubstituted amino pyrazole analogues via multi-component reactions. This catalyst-free reaction proceeded smoothly in good to excellent yields and offered several other advantages including short reaction time, simple experimental workup procedures, and no toxic by-products. The approach to pyrazole systems presented herein avoids the use of catalyst, toxic organic solvent, and anhydrous conditions. This protocol represents a promising green route for the synthesis of this class of compounds.
Experimental
Chemicals and apparatus
All chemicals were purchased from Merck or Fluka Chemical Companies and they were used as received. The \(^{1}\) H NMR (500 MHz, 400 MHz) and \(^{13}\) C NMR (125 MHz, 100 MHz) spectra were recorded on a Bruker Avance DPX-250, FT-NMR spectrometer (\(\delta \) in ppm). Tetramethylsailane (TMS) was used as internal standard. The abbreviations used for NMR signals are: s = singlet, d = doublet, t = triplet, q = quartet, and m = multiplet. Melting points were recorded on a Büchi B-545 apparatus in open capillary tubes and are uncorrected.
General procedure for the preparation of 5-Amino-1,3-diaryl-1 \(H\)-pyrazole-4-carbonitriles derivatives
Phenyl hydrazine derivative, (1 mmol) aromatic aldehyde (1 mmol), and malono derivative (1 mmol) were added in a 25 ml round-bottomed flask contained water and ethanol (50 % v/v) (6 mL), and the resulting mixture was stirred at room temperature. After completion of the reaction (as monitored by TLC), crystals of the product were formed, collected by filtration and then recrystallized from hot ethanol to obtain pure products.
Selected spectral data of the products
5-Amino-1,3-diphenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 1)
White powder (0.247 g, 95 %), M.P. = \(160-161\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3485, 3341, 3083, 2359, 1599, 1412, 1253, 1126, 1100, 1075 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.91 (t, \(J\) = 7.3 Hz, 1H), 7.16 (d, \(J\) = 7.6 Hz, 2H), 7.29–7.35 (m, 3H), 7.41 (t, \(J\) = 7.7 Hz, 2H), 7.64 (s, 1H), 7.70 (d, \(J\) = 7.2 Hz, 2H), 7.72 (s, 1H). \(^{13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 112.80, 113.26, 120.58, 126.65, 128.9, 129.05, 129.75, 135.74, 137.81, 145.09, 150.41, 156.50. MS (m/z): 260 (M+). Anal.Calcd for \(\text{ C }_{16}\text{ H }_{12}\text{ N }_{4}\): C, 73.83; H, 4.65; N, 21.52 %. Found: C, 73.48; H, 4.86; N, 21.72 %.
5-Amino-1-phenyl-3-p-tolyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 2)
Pink powder (0.241g, 88%), M.P. = 117–118\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3482, 3319, 3095, 2925, 2359, 1598, 1415, 1257, 1127, 1113, 1096 cm\(^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 2.41 (s, 3H), 6.91 (dd, \(J\) = 3.5 Hz and \(J\) = 7.3 Hz, 1H), 7.15 (d, \(J\) = 7.76 Hz, 2H), 7.22 (d, \(J \)= 7.9 Hz, 2H), 7.29–7.33 (m, 2H), 7.59 (d, \(J\) = 7.9 Hz, 2H), 7.70 (s, 2H). \(^{13}\) C NMR (\(\text{ CDCl }_{3,}\) 125 MHz): \(\delta \) (ppm) 21.90, 104.65, 113.22, 120.44, 126.62, 129.71, 129.77, 132.95, 138.14, 138.97, 145.20, 148.82, 153.20. Anal.Calcd for \(\text{ C }_{17}\text{ H }_{14}\text{ N }_{4}\): C, 74.43; H, 5.14; N, 20.42 %. Found: C, 74.88; H, 5.18; N, 20.12 %.
5-Amino-3-(2-hydroxyphenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 3)
Yellow powder (0.252g, 96 %), M.P. = 161–162\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3583, 3487, 3341, 3102, 2358, 2197, 1602, 1413, 1222, 1192, 1108, 1050 \(cm^{-1}\) .\(^{ 1}\)H NMR (DMSO–\(d_{6},\) 500 MHz): \(\delta \) (ppm) 6.76 (t, \(J\) = 7.3 Hz, 1H), 6.85–6.90 (m, 2H), 6.96 (d, \(J\) = 7.6 Hz, 2H), 7.14–7.18 (m, 1H), 7.24 (dd, \(J\) = 7.5 Hz and \(J\) = 8.3 Hz, 2H), 7.53 (dd, \(J\) = 1.5 Hz and \(J\) = 7.7 Hz, 1H), 8.14 (s, 1H), 10.38 (s, 1H), 10.52 (s, 1H).\(^{ 13}\) C NMR (DMSO–\(d_{6}\), 125 MHz): \(\delta \) (ppm) 112.60, 116.80, 119.83, 120.25, 121.35, 125.45, 128.17, 130.05, 130.15, 138.13, 145.62, 150.55, 152.30, 156.51. Anal.Calcd for \(\text{ C }_{16}\text{ H }_{12}\text{ N }_{4}\) O: C, 69.55; H, 4.38; N, 20.28 %. Found: C, 69.48; H, 4.46; N, 20.35 %.
5-Amino-3-(4-chlorophenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 4)
White powder (0.279g, 92 %), M.P. = 129–130\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3448, 3315, 3074, 2358, 1595, 1414, 1293, 1254, 1133, 1083 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.93 (t, \(J\) = 7.3 Hz, 1H), 7.15 (d, \(J\) = 7.7 Hz, 2H), 7.29–7.34 (m, 2H), 7.37 (d, \(J\) = 8.4 Hz, 2H), 7.62 (d, \(J\) = 8.4 Hz, 2H), 7.67 (s, 2H).\(^{ 13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 110.24, 113.29, 120.85, 127.71, 129.26, 129.77, 134.26, 134.44, 136.35, 144.78, 150.21, 155.45. Anal.Calcd for \(\text{ C }_{16}\text{ H }_{11}\text{ ClN }_{4}\): C, 65.20; H, 3.76; N, 19.01 %. Found: C, 65.37; H, 3.81; N, 18.95 %.
5-Amino-3-(4-nitrophenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 5)
Red powder (0.298 g, 98 %), M.P. = 164–165\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3467, 3350, 3102, 2359, 1600, 1415, 1457, 1344, 1256, 1123, 1107, 1095 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.98 (s, 1H) 7.18 (d, \(J\) = 7.5 Hz, 2H), 7.29–7.34 (m, 2H), 7.73–7.79 (m, 3H), 8.05 (s, 1H), 8.24 (d, \(J\) = 7.6 Hz, 2H). MS (m/z): 305 (M+). Anal.Calcd for \(\text{ C }_{16} \text{ H }_{11}\text{ N }_{5}\text{ O }_{2}\): C, 62.95; H, 3.63; N, 22.94 %. Found: C, 63.05; H, 3.58; N, 23.03 %.
5-Amino-3-(3-nitrophenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 6)
Orange powder (0.292 g, 96 %), M.P. = 129–130\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3452, 3324, 3103, 2357, 1594, 1478, 1447, 1344, 1338, 1263, 1147, 1100, 1096 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.97 (t, \(J\)=7.2 Hz, 1H), 7.18 (d, \(J\)=8.4 Hz, 2H), 7.35 (t, \(J\)=7.5 Hz, 2H), 7.56 (t, \(J\)=7.9 Hz, 1H), 7.74 (s, 1H), 7.89 (s, 1H), 8.01 (d, \(J\)=7.5 Hz, 1H), 8.14 (d, \(J\)=8.0 Hz, 1H), 8.48 (s, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3,}\) 125 MHz): \(\delta \) (ppm) 112.44, 113.42, 121.10, 121.38, 122.98, 129.85, 129.94, 131.80, 134.30, 137.72, 144.27,149.16, 156.41. Anal.Calcd for \(\text{ C }_{16}\text{ H }_{11}\text{ N }_{5}\text{ O }_{2}\): C, 62.95; H, 3.63; N, 22.94 %. Found: C, 62.78; H, 3.77; N, 22.90 %.
5-Amino-3-(2-nitrophenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 7)
Red powder (0.289 g, 95 %), M.P. = 160–161\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3439, 3296, 3024, 2358, 1600, 1497, 1469, 1342, 1333, 1298, 1254, 1163, 1134 cm\(^{-1}\). \(^{1}\)H NMR (DMSO–\(d_{6}\), 500 MHz): \(\delta \) (ppm) 6.81 (t, \(J\) = 7.3 Hz, 1H), 7.11 (d, \(J\) = 7.7 Hz, 2H), 7.25 (t, \(J\) = 7.6 Hz, 2H), 7.47 (t, \(J\) = 7.2 Hz, 1H), 7.69 (t, \(J\) = 7.5 Hz, 1H), 7.95 (d, \(J\) = 7.5 Hz, 1H), 8.15 (d, \(J\) = 7.8 Hz, 1H), 8.26 (s, 1H), 10.88 (s, 1H). Anal.Calcd for \(\text{ C }_{16}\text{ H }_{11}\text{ N }_{5}\text{ O }_{2}\): C, 62.95; H, 3.63; N, 22.94 %. Found: C, 63.12; H, 3.55; N, 22.85 %.
5-Amino-3-(4-cyanophenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 8)
Yellow powder (0.256 g, 90 %), M.P. = 159–160\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3438, 3306, 3279, 2359, 2224, 1593, 1478, 1262, 1159, 1107, 1093 \(cm^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.80 (t, \(J\) = 7.2 Hz, 1H), 7.12 (d, \(J\) = 7.6 Hz, 2H), 7.24 (t, \(J\) = 7.4 Hz, 2H), 7.78–7.81 (m, 4H), 7.87 (s, 1H), 10.73 (s, 1H). Anal.Calcd for \(\text{ C }_{17}\text{ H }_{11}\text{ N }_{5}\): C, 71.57; H, 3.89; N, 24.55 %. Found: C, 71.35; H, 3.70; N, 24.50 %.
5-Amino-3-(4-isopropylphenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 9)
Pink powder (0.235 g, 78 %), M.P. = 139–140\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3417, 3295, 3102, 2923, 2357, 1600, 1481, 1257, 1125, 1096, 1048 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 1.33 (d, \(J\) = 6.9 Hz, 6H), 2.94–3.00 (m, 1H), 6.91 (t, \(J\) = 7.2 Hz, 1H), 7.15 (d, \(J\) = 7.8 Hz, 2H), 7.27–7.33 (m, 4H), 7.63 (d, \(J\) = 7.965 Hz, 2H), 7.70 (s, 1H), 8.35 (s, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 24.32, 34.45, 111.28, 113.17, 120.38, 126.69, 127.14, 129.71, 133.39, 137.99, 145.27, 149.32, 149.93, 156.43. Anal.Calcd for \(\text{ C }_{19}\text{ H }_{18}\text{ N }_{4}\): C, 75.47; H, 6.00; N, 18.53 %. Found: C, 75.25; H, 5.78; N, 19.05 %.
5-Amino-3-(3,4-dimethoxyphenyl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 10)
White powder (0.233 g, 73 %), M.P. = 122–123\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3432, 3295, 3007, 2939, 2357, 1600, 1476, 1331, 1264, 1236, 1167, 1130, 1018 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 3.93 (s, 3H), 3.99 (s, 3H), 6.86–6.94 (m, 2H), 7.06 (d, \(J\) = 7.1 Hz, 1H), 7.13 (d, \(J\) = 8.2 Hz, 2H), 7.30 (t, \(J\) = 7.4 Hz, 2H), 7.40 (s, 1H), 7.63 (s, 1H), 9.90 (s, 1H). \(^{13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 56.20, 56.23, 113.62, 114.70, 118.87, 118.96, 119.56, 120.43, 131.71, 131.91, 132.85, 133.19, 138.59, 140.46, 150.73, 156.47. Anal.Calcd for \(\text{ C }_{18}\text{ H }_{16}\text{ N }_{4}\text{ O }_{2}\): C, 67.49; H, 5.03; N, 17.49 %. Found: C, 67.37; H, 5.10; N, 17.52 %.
5-Amino-1-phenyl-3-(thiophen-2-yl)-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 11)
Yellow powder (0.234 g, 88 %), M.P. = 140–141\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3412, 3325, 3092, 2357, 1600, 1478, 1298, 1264, 1138, 1069 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.92 (t, \(J\) = 7.3 Hz, 1H), 7.05 (dd, \(J\) = 3.6 Hz and \(J\) = 4.9 Hz, 1H), 7.12 (d, \(J\) = 7.4 Hz, 3H), 7.31 (dd, \(J\) = 5.0 Hz and \(J\) = 14.1 Hz, 3H), 7.56 (s, 1H), 7.84 (s, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3,}\) 125 MHz): \(\delta \) (ppm) 113.24, 114.21, 120.64, 122.44, 126.33, 126.89, 127.66, 129.76, 132.70, 140.91, 144.86, 155.22. Anal.Calcd for \(\text{ C }_{14}\text{ H }_{10}\text{ N }_{4}\)S: C, 63.14; H, 3.78; N, 21.04 %. Found: C, 63.33; H, 3.82; N, 20.88 %.
5-Amino-3-(5-methylthiophen-2-yl)-1-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 12)
Yellow powder (0.252 g, 90 %), M.P. = 131–132\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3427, 3303, 3102, 2919, 2357, 1600, 1469, 1257, 1230, 1126, 1100, 1070 cm\(^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 2.53 (s, 3H), 6.68–6.71 (m, 1H), 6.88–6.92 (m, 2H), 7.10 (d, \(J\) = 7.7 Hz, 2H), 7.28–7.32 (m, 2H), 7.46 (s, 1H), 7.76 (s, 1H). \(^{13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 16.08, 113.15, 114.13, 120.42, 125.86, 126.10, 127.16, 129.71, 133.18, 138.66, 141.38, 145.05, 156.73. Anal.Calcd for \(\text{ C }_{15}\text{ H }_{12}\text{ N }_{4}\) S: C, 64.26; H, 4.31; N, 19.98 %. Found: C, 64.37; H, 4.25; N, 20.05 %.
5-Amino-3-(naphthalene-6-yl)-1-phenyl -1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 13)
Yellow powder (0.264 g, 93 %), M.P. = 229–230\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3403, 3341, 3112, 2358, 1600, 1478, 1252, 1122, 1113, 1082 cm\(^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 6.87–6.98 (m, 1H), 7.20 (d, \(J\) = 7.4 Hz, 2H), 7.34 (t, \(J\) = 7.5 Hz, 2H), 7.50–7.52 (m, 2H), 7.70 (s, 1H), 7.79–7.94 (m, 5H), 8.07 (d, \(J\) = 8.5 Hz, 1H).\(^{ 13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 113.27, 114.81, 120.66, 123.43, 126.63, 126.83, 126.95, 127.63, 128.28, 128.44, 128.83, 129.77, 130.58, 132.22, 133.98, 137.87, 143.30, 155.97. Anal.Calcd for \(\text{ C }_{20}\text{ H }_{14}\text{ N }_{4}\): C, 77.40; H, 4.55; N, 18.05 %. Found: C, 77.18; H, 4.72; N, 18.13 %.
5-Amino-1-(4-chlorophenyl)-3-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 14)
Pink powder (0.261 g, 89 %), M.P. = 134–135\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3421, 3318, 3098, 2356, 1597, 1477, 1257, 1135, 1096, 1074 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 7.08 (d, \(J\) = 8.6 Hz, 2H), 7.25 (d, \(J\) = 8.5 Hz, 2H), 7.35–7.36 (m, 1H) 7.41 (t, \(J\) = 7.9 Hz, 2H), 7.55 (s, 1H), 7.68 (d, \(J\) = 7.7 Hz, 2H), 7.72 (s, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 112.10, 114.32, 125.10, 126.70, 129.08, 129.12, 129.62, 132.32, 135.43, 138.40, 143.70, 155.98. Anal.Calcd for \(\text{ C }_{16}\text{ H }_{11}\text{ ClN }_{4}\): C, 65.20; H, 3.76; N, 19.01 %. Found: C, 64.97; H, 3.87; N, 19.11 %.
5-Amino-1-(4-bromophenyl)-3-phenyl-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 15)
White powder (0.299 g, 89 %), M.P. = 130–131\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3425, 3317, 3114, 2357, 1597, 1480, 1253, 1137, 1094, 1068 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3,}\) 500 MHz): \(\delta \) (ppm) 7.03 (d, \(J\) = 8.8 Hz, 2H), 7.36 (d, \(J \)= 7.3 Hz, 1H), 7.39–7.43 (m, 4H), 7.59 (s, 1H), 7.68 (d, \(J\) = 5.1 Hz, 2H), 7.68 (s, 1H).\(^{ 13}\)C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 112.29, 114.78, 125.65, 126.73, 129.10, 129.16, 130.12, 132.51, 135.41, 138.50, 144.15, 151.78. Anal.Calcd for \(\text{ C }_{16}\text{ H }_{11}\text{ BrN }_{4}\): C, 56.66; H, 3.27; N, 16.52 %. Found: C, 56.97; H, 3.15; N, 16.43 %.
5-Amino-1-(4-bromophenyl)-3-(naphthalen-1-yl)-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 16)
Yellow powder (0.260 g, 90 %), M.P. = 139–140\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3470, 3306, 3082, 2358, 1591, 1480, 1250, 1137, 1096, 1068 cm\(^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 7.08 (d, \(J \)= 8.4 Hz, 2H), 7.43 (d, \(J \)= 8.5 Hz, 2H), 7.51–7.59 (m, 2H), 7.63 (t, \(J\) = 8.1 Hz, 1H), 7.72 (s, 1H), 7.87 (t, \(J \)= 6.2 Hz, 2H), 7.92 (d, \(J\) = 8.0 Hz, 1H), 8.32 (s, 1H), 8.76 (d, \(J \)= 8.4 Hz, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 112.37, 114.81, 119.02, 124.21, 124.63, 125.87, 126.42, 127.24, 128.12, 128.86, 129.25, 129.75, 130.79, 132.59, 134.44, 138.04, 144.10, 156.32. Anal.Calcd for \(\text{ C }_{20}\text{ H }_{13}\text{ BrN }_{4}\): C, 61.71; H, 3.37; N, 14.39 %. Found: C, 61.55; H, 3.44; N, 14.28 %.
5-Amino-3-(4-ethoxyphenyl)-1\(H\)-pyrazole-4-carbonitrile (Table 1, entry 17)
White powder (0.261 g, 86 %), M.p. = 123–124\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3416, 3329, 3102, 2921, 2358, 1602, 1481, 1252, 1126, 1118, 1096 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 500 MHz): \(\delta \) (ppm) 1.47(t, \(J\) = 6.9 Hz, 3H), 4.10 (q, \(J\) = 6.9 Hz, 2H), 6.88 (t, \(J\) = 8.0 Hz, 1H), 6.94 (d, \(J\) = 8.6 Hz, 2H), 7.14 (d, \(J\) = 8.0 Hz, 2H), 7.29–7.32 (m, 2H), 7.57 (s, 1H), 7.63 (d, \(J\) = 8.6 Hz, 2H), 7.70 (s, 1H). \(^{13}\) C NMR (\(\text{ CDCl }_{3}\), 125 MHz): \(\delta \) (ppm) 15.231, 63.98, 112.34, 113.27, 115.108, 120.38, 123.55, 128.16, 129.70, 138.01, 138.32, 138.52, 145.89, 159.99. Anal.Calcd for \(\text{ C }_{18}\text{ H }_{16}\text{ N }_{4}\) O: C, 71.04; H, 5.30; N, 18.41 %. Found: C, 71.22; H, 5.16; N, 18.52 %.
3,3\(^{\prime }\)-(1,4-phenylene)bis(5-amino-1-phenyl-1\(H\)-pyrazole-4-carbonitrile) (Table 1, entry 18)
Orange powder (0.420 g, 95 %), M.P \(>230\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3422, 3302, 3108, 2357, 1594, 1469, 1254, 1140, 1102, 1055 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (DMSO–\(d_{6}\), 500 MHz): \(\delta \) (ppm) 6.76 (t, \(J\) = 7.6 Hz, 2H) 7.09 (d, \(J\) = 7.7 Hz, 4H), 7.23 (t, \(J\) = 7.5 Hz, 4H), 7.65 (d, \(J\) = 4.2 Hz, 4H), 7.87 (s, 2H), 10.38 (s, 2H). \(^{13}\)C NMR (DMSO–\(d_{6}\), 125 MHz): \(\delta \) (ppm) 112.92, 113.86, 119.69, 124.30, 126.75, 130.00, 136.21, 137.06, 146.07, 156.76. MS (m/z): 442 (M+). Anal.Calcd for \(\text{ C }_{26}\text{ H }_{18}\text{ N }_{8}\): C, 70.58; H, 4.10; N, 25.32 %. Found: C, 70.82; H, 3.95; N, 25.21 %.
3,3\(^{\prime }\)-(1,3-phenylene)bis(5-amino-1-phenyl-1\(H\)-pyrazole-4-carbonitrile) (Table 1, entry 19)
Yellow powder (0.406 g, 92 %), M.P\(>230\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3482, 3305, 3076, 2357, 1592, 1480, 1252, 1133, 1101, 1072 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (DMSO–\(d_{6}\), 500 MHz): \(\delta \) (ppm) 6.77 (t, \(J\) = 7.2 Hz, 2H), 7.11 (d, \(J\) = 7.9 Hz, 4H), 7.24 (t, \(J\) = 7.6 Hz, 4H), 7.41 (t, \(J\) = 7.6 Hz, 1H), 7.60 (d, \(J\) = 7.6 Hz, 2H), 7.86 (s, 1H), 7.92 (s, 2H), 10.38 (s, 2H). \(^{13}\) C NMR (DMSO–\(d_{6}\), 125 MHz): \(\delta \) (ppm) 112.94, 113.70, 119.72, 121.80, 123.95, 125.81, 129.84, 130.02, 137.09, 137.13, 146.13, 156.44. MS (m/z): 442 (M+). Anal.Calcd for C\(_{26}\)H\(_{18}\)N\(_{8}\): C, 70.58; H, 4.10; N, 25.32 %. Found: C, 70.45; H, 4.17; N, 25.39 %.
Diethyl 3,3\(^{\prime }\)-(1,4-phenylene)bis(5-amino-1-phenyl-1\(H\)-pyrazole-4-carboxylate) (Table 1, entry 20)
Yellow powder (0.466 g, 87 %), M.P\(>230\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3486, 3348, 3108, 2996, 1722, 1598, 1475, 1251, 1130, 1121, 1079 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (DMSO–\(d_{6}\), 400 MHz): \(\delta \) (ppm) 1.43 (t, \(J\) = 7.2 Hz, 6H), 4.42 (q, \(J\) = 7.2 Hz, 4H), 6.96 (t, \(J\) = 7.2 Hz, 2H), 7.18 (d, \(J\) = 7.6 Hz, 4H), 7.34 (t, \(J\) = 8.0 Hz, 4H), 7.71 (s, 2H), 7.79 (d, \(J\) = 8.4 Hz, 2H), 8.04 (d, \(J\) = 8.4 Hz, 2H), 8.25 (s, 2H). MS (m/z): 536 (M+). Anal.Calcd for \(\text{ C }_{30}\text{ H }_{28}\text{ N }_{6}\text{ O }_{4}\): C, 67.15; H, 5.26; N, 15.66 %. Found: C, 67.23; H, 5.33; N, 15.54 %.
Methyl 5-amino-1,3-diphenyl-1\(H\)-pyrazole-4-carboxylate (Table 1, entry 21)
White powder (0.272 g, 93 %), M.p. = 161–162\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3463, 3312, 3104, 2985, 1719, 1600, 1481, 1254, 1134, 1118, 1083 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 400 MHz): \(\delta \) (ppm) 3.68 (s, 3H), 6.93 (t, \(J\) = 7.2 Hz, 1H), 7.16 (d, \(J\) = 8 Hz, 2H), 7.29–7.37 (m, 3H), 7.42 (t, \(J \)= 7.4 Hz, 2H), 7.61 (s, 1H), 7.68 (s, 1H), 7.70 (d, \(J\) = 7.6 Hz, 2H). MS (m/z): 293 (M+). Anal.Calcd for \(\text{ C }_{18}\text{ H }_{13}\text{ N }_{3}\text{ O }_{2}\): C, 71.28; H, 4.32; N, 13.85 %. Found: C, 70.94; H, 4.54; N, 13.78 %.
Ethyl 5-amino-1,3-diphenyl-1\(H\)-pyrazole-4-carboxylate (Table 1, entry 22)
White powder (0.292 g, 95 %), M.p. = 163–164\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3459, 3316, 3078, 2988, 1718, 1593, 1479, 1256, 1128, 1114, 1106 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 400 MHz): \(\delta \) (ppm) 1.36 (t, \(J \)= 7.6 Hz, 3H), 4.8 (q, \(J \)= 7.6 Hz, 2H), 6.94 (t, \(J\) = 7.2 Hz, 1H), 7.17 (d, \(J\) = 7.6 Hz, 2H), 7.32–7.37 (m, 3H), 7.42 (t, \(J\) = 7.4 Hz, 2H), 7.62 (s, 1H), 7.66 (s, 1H), 7.71 (d, \(J\) = 7.2 Hz, 2H). MS (m/z): 307 (M+). Anal.Calcd for \(\text{ C }_{19}\text{ H }_{15}\text{ N }_{3}\text{ O }_{2}\): C, 71.91; H, 4.76; N, 13.24 %. Found: C, 71.38; H, 4.89; N, 13.45 %.
Methyl 5-amino-3-(4-nitrophenyl)-1-phenyl-1\(H\)-pyrazole-4-carboxylate (Table 1, entry 23)
Red powder (0.321 g, 95 %), M.p. = 162–163\(\,^{\circ }\text{ C }, \nu _{\mathrm{max}}\) (KBr) 3452, 3339, 3110, 2989, 1723, 1600, 1482, 1497, 1348, 1254, 1133, 1120, 1081 \(\text{ cm }^{-1}\). \(^{1}\)H NMR (\(\text{ CDCl }_{3}\), 400 MHz): \(\delta \) (ppm) 3.80 (s, 3H), 6.98 (t, \(J\) = 7.4 Hz, 1H), 7.18 (d, \(J \)= 7.6 Hz, 2H), 7.36 (t, \(J\) = 4 Hz, 2H), 7.73 (s, 1H), 7.80 (d, \(J\) = 8.8 Hz, 2H), 8.02 (s, 1H), 8.25 (d, \(J\) = 8.8 Hz, 2H). MS (m/z): 338 (M+). Anal.Calcd for \(\text{ C }_{18}\text{ H }_{12}\text{ N }_{4}\text{ O }_{4}\): C, 62.07; H, 3.47; N, 16.09 %. Found: C, 62.50; H, 3.63; N, 15.79 %.
5\(^{\prime }\)-Amino-2-oxo-1\(^{\prime }\)-phenyl-1\(^{\prime }\),2\(^{\prime }\)-dihydrospiro[indoline-3,3\(^{\prime }\)-pyrazole]-4-carbonitrile (10, Scheme 4)
Yellow powder M.p. = 214–216 \(^{1}\)H NMR (\(\text{ CDCl }_{3,}\) 500 MHz) \(\delta \) (ppm): 88.92 9s, 1H), 6.96 (d, \(J\)=7.75 Hz, 1H), 7.09–7.127 (m, 1H), 7.15(d, \(J\)=7.56, 1H), 7.27 (dd, \(J\)=4.0 Hz, \(J\)=8.0 Hz, 1H), 7.39–7.42 (m, 4H), 7.68 (d, \(J\)=7.5 Hz, 1H), 8.26 (s, 1H), 12.74 (s, 1H). \(^{13}\)C NMR (\(\text{ CDCl }_{3,}\) 125 MHz) \(\delta \) (ppm): 103.84, 110.70, 114.88, 119.60, 122.54, 123.077, 123.81, 127.52, 128.48, 129.86, 138.55, 141.72, 142.96, 155.22, 164.09.
References
Hasaninejad A, Zare A, Shekouhy M (2011) Sulfuric acid-modified PEG-6000 (PEG-OSO3H): an efficient, bio-degradable and reusable polymeric catalyst for the solvent-free synthesis of poly-substituted quinolines under microwave irradiation. Green Chem 13:958–964. doi:10.1039/c0gc00953a
Hasaninejad A, Zare A, Shekouhy M (2010) Catalyst-free one-pot four component synthesis of polysubstituted imidazoles in neutral. Ionic liquid 1-butyl-3-methylimidazolium bromide. J Comb Chem 12:844–849. doi:10.1021/cc100097m
Hasaninejad A, Zare A, Mohammadizadeh MR, Shekouhy M (2010) Lithium bromide as an efficient, green, and inexpensive catalyst for the synthesis of quinoxaline derivatives at room temperature. Green Chem Lett Rev 3:143–148. doi:10.1080/17518251003619192
Hasaninejad A, Zare A, Shekouhy M (2010) Highly efficient synthesis of triazolo[1,2-\(a\)]indazole-triones and novel spiro triazolo[\(1,2-a\)]indazole-tetraones under solvent-free conditions. Tetrahedron 67:390–400. doi: 10.1016/j.tet.2010.11.029
Hasaninejad A, Zare A, Zolfigol MA, Shekouhy M (2009) Zirconium Tetrakis(dodecyl Sulfate) [Zr(DS)4] as an efficient Lewis acid-surfactant combined catalyst for the synthesis of quinoxaline derivatives in aqueous media. Synth Commun 39:569–579. doi:10.1080/00397910802406737
Hasaninejad A, Golzar N, Shekouhy M, Zare A (2011) Diversity-oriented synthesis of novel 2\(\prime \)-aminospiro[11\(H\)-indeno[1,2-\(b\)]quinoxaline-11,4\(\prime -[4H]\)pyran] derivatives via a one-pot four-component reaction. Helv Chim Acta 94:2289–2294. doi: 10.1002/hlca.201100201
Hasaninejad A, Shekouhy M, Zare A (2012) Silica nanoparticles efficiently catalyzed synthesis of quinolines andquinoxalines. Catal Sci Technol 2:201–214. doi:10.1039/c1cy00332a
Anzai K, Furuse M, Yoshida A, Matsuyama A, Moritake T, Tsuboi K, Ikota N (2004) In vivo radioprotection of mice by 3-methyl-1-phenyl-2-pyrazolin-5-one (Edaravone; Radicut), a clinical drug. J Radiat Res 45:319–323
Yet L (2008) in Comprehensive heterocyclic chemistry III. In: Katritzky AR, Ramsden CA, Scriven EFV, Taylor RJK (eds.). Elsevier, Oxford 4:1
Daidone G, Maggio B, Plescia S, Raffa D, Musiu C, Milia C, Perra G, Marongiu ME (1998) Antimicrobial and antineoplastic activities of new 4-diazopyrazole derivatives. Eur J Med Chem 33:375–382. doi:10.1016/S0223-5234(98)80004-4
Taylor EC, Patel H, Kumar H (1992) Synthesis of pyrazolo 3,4-dpyrimidine analogues of the potent agent N-4-2-2-amino-4 3H-oxo-7H-pyrrolo 2,3-dpyrimidin-5-yl ethylbenzoyl-L-glutamic acid (LY231514). Tetrahedron 48:8089–8100. doi:10.1016/S0040-4020(01)80479-8
Akbas E, Berber I, Sener A, Hasanov B (2005) Synthesis and antibacterial activity of 4-benzoyl-1-methyl-5-phenyl-1\(H\)-pyrazole-3-carboxylic acid and derivatives. IL Farmaco 60:23–26. doi: 10.1016/j.farmac.2004.09.003
Tanitame A, Oyamada Y, Ofuji K (2004) Design, synthesis and structure-activity relationship studies of novel indazole analogues as DNA gyrase inhibitors with Gram-positive antibacterial activity. Bioorg Med Chem Lett 14:2857–2862. doi:10.1016/j.bmcl.2004.03.044
Bekhita AA, Abdel-Aziem T (2004) Design, synthesis and biological evaluation of some pyrazole derivatives as anti-inflammatory-antimicrobial agents. Bioorg Med Chem 12:1935–1945. doi:10.1016/j.bmc.2004.01.037
Stahman MA, Huebner CF, Link KP (1941) Studies on the hemorrhagic sweet clover disease: v. identification and synthesis of hemorrhagic agent. J Biol Chem 138:513–527
Nauduri D, Reddy GB (1998) Antibacterials and antimycotics: part 1: synthesis and activity of 2-pyrazoline derivatives. Chem Pharm Bull 46:1254–1260
Foks H, Pancechowska-Ksepko D, Kedzia A, Zwolska Z, Janowiec M, Augustynowicz-Kopec E (2005) Synthesis and antibacterial activity of 1H-pyrazolo[3,4-b]pyrazine and -pyridine derivatives. IL Farmaco 60:513–517. doi:10.1016/j.farmac.2005.05.002
Dardari Z, Lemrani M, Sebban A, Bahloul A, Hassar M, Kitane S, Berrada M, Boudouma B (2006) Ethyl 5-amino-1-(4-chloro-2-nitro-phen-yl)-1\(H\)-pyrazole-4-carboxyl-ate. Arch Pharm (Weinheim Ger) 339:291–298. doi:10.1107/S1600536809000488
Gilbert AM, Failli A, Shumsky J, Yang Y (2006) Pyrazolidine-3,5-diones and 5-hydroxy-1\(H\)-pyrazol-3(2\(H)\)-ones, Inhibitors of UDP-\(N\)-acetylenolpyruvyl glucosamine reductase. J Med Chem 49:1202–1204. doi: 10.1021/jm060499t
Tanitame A, Oyamada Y, Ofugi K, Fujimoto M, Iwai N, Hiyama Y, Suzuki KM, Yamagishi J (2004) Synthesis and antibacterial activity of a novel series of potent DNA gyrase inhibitors. Pyrazole derivatives. J Med Chem 47:3693–3696. doi: 10.1021/jm030394f
Badawey AM, El-Ashmawey IM (1998) Nonsteroidal antiinflammatory agents - part 1: antiinflammatory, analgesic and antipyretic activity of some new 1-(pyrimidin-2-yl)-3-pyrazolin-5-ones and 2-(pyrimidin-2-yl)-1,2,4,5,6,7-hexahydro-3\(H\)-indazol-3-ones. Eur J Med Chem 33:349–361. doi: 10.1016/S0223-5234(98)80002-0
Tewari AK, Mishra A (2001) Synthesis and anti-inflammatory activities of \(\text{ N }^{4},\text{ N }^{5}\)-disubstituted-3-methyl-1H-pyrazolo[3,4-c]pyridazines\(.\) Bioorg Med Chem 9:715–718. doi:10.1016/S0968-0896(00)00285-6
Wise LD, Butler DE, Dewald HA, Lustgarten DM, Pattison IC, Schweiss DN, Coughenour LL, Downs DA, Heffner TG, Pugsley TA (1987) 1,3-Dialkyl-4-(iminoarylmethyl)-1H-pyrazol-5-ols. A series of novel potential antipsychotic agents. J Med Chem 30:1807–1812. doi:10.1021/jm00393a021
Gursoy SA, Demirayak G, Capan K (2000) Synthesis and preliminary evaluation of new 5-pyrazolinone derivatives as analgesic agents. Eur J Med Chem 35:359–364. doi:S0223523400001173/FLA
Bailey DM, Hansen PE, Hlavac AG, Baizman ER, Pearl J, Defelice AF, Feigenson ME (1985) 3,4-Diphenyl-1H-pyrazole-1-propanamine antidepressants. J Med Chem 28:256–260. doi:10.1021/jm00380a020
Özdemir Z, Kandilci HB, Gümüsel B, Calis Ü, Bilgin AA (2007) Synthesis and studies on antidepressant and anticonvulsant activities of some 3-(2-furyl)-pyrazoline derivatives. Eur J Med Chem 42:373–379. doi:10.1016/j.ejmech.2006.09.006
Menozzi G, Mosti L, Schenone P, D-Amico M, Filippelli A, Rossi F (1992) 4H-thieno[3,4-c]pyrazole derivatives with antiinflammatory, analgesic, antipyretic and platelet antiaggregating activities. Farmaco 47:1495–1511
Rathelot P, Azas N, El-Kashef H, Delmas F (2002) 1,3-Diphenylpyrazoles: synthesis and antiparasitic activities of azomethine derivatives. Eur J Med Chem 37:671–679. doi:10.1016/S0223-5234(02)01388-0
Bernardino AMR, Gomes AO, Charret KS, Freitas ACC (2006) Synthesis and leishmanicidal activities of 1-(4-X-phenyl)-N\(\prime \)-[(4-Y-phenyl)methylene]-1\(H\)-pyrazole-4-carbohydrazides. Eur J Med Chem 41:80–87. doi:10.1016/j.ejmech.2005.10.007
Katiyar SB, Srivastava K, Purib SK, Chauhana PMS (2005) Synthesis of 2-[3,5-substituted pyrazol-1-yl]-4,6-trisubstituted triazine derivatives as antimalarial agents. Bioorg Med Chem Lett 15: 4957–4960. doi:10.1016/j.bmcl.2005.08.023
Farag AM, Ali KA, Mayhoub AS, Abdalla TM, Amr AE, Abdel-Hafez N, Abdalla MM (2010) Design, synthesis and structure-activity relationship study of novel pyrazole-based heterocycles as potential antitumor agents. Eur J Med Chem 45:5887–5898. doi:10.1016/j.ejmech.2010.09.054
Moukha-Chafiq O, Taha ML, Lazrek HB, Vasseur JJ (2002) Synthesis and biological activity of some 4-substituted 1-[1-(2,3-dihydroxy-1-propoxy)methyl-1,2,3-triazol-(4 & 5)-ylmethyl]-1\(H\)-pyrazolo[3,4-\(d\)]pyrimidines. IL Farmaco 57:27–32. doi:10.1016/S0014-827X(01)01152-1
Allen SH, Johns BA, Gudmundsson KS, Freeman GA (2006) Synthesis of C-6 substituted pyrazolo[1,5-\(a\)]pyridines with potent activity against herpesviruses. Bioorg Med Chem 14:944–954. doi: 10.1016/j.bmc.2005.09.015
He FQ, Liu XH, Wang BL, Li ZM (2008) Synthesis and biological activities of novel bis-heterocyclic pyrrodiazole derivatives. Heteroatom Chemistry 19:21–27. doi:10.1002/hc.20369
Frinkelstein BL, Strok CJ (1997) Synthesis and insecticidal activity of novel pyrazole methanesulfonates. J Pestic Sci 50:324–328. doi:10.1002/(SICI)1096-9063(199708)
Parlow JJ (1998) Synthesis of pyrazolecarbonylaminopyridinecarboxamides as herbicides. J Heterocycl Chem 35:1493–1499. doi:10.1002/jhet.5570350646
Halcrow MA (2009) Pyrazoles and pyrazolides–flexible synthons in self-assembly. Dalton Trans 2059–2073: doi:10.1039/B815577A
Singer RA, Dore M, Sieser JE, Berliner MA (2006) Development of nonproprietary phosphine ligands for the Pd-catalyzed amination reaction. Tetrahedron Lett 47:3727–3731. doi:10.1016/j.tetlet.2006.03.132
Kane JL, Hirth BH, Laing O, Gourlie BB, Nahill S, Barsomiam G (2003) Ureas of 5-aminopyrazole and 2-aminothiazole inhibit growth of gram-positive bacteria. Bioorg Med Chem Lett 13: 4463–4466. doi:10.1016/j.bmcl.2003.09.013
Kumar V, Aggarwal R, Tyagi P, Singh SP (2005) Synthesis and antibacterial activity of some new 1-heteroaryl-5-amino-4-phenyl-3-trifluoromethylpyrazoles. Eur J Med Chem 40:922–927. doi:10.1016/j.ejmech.2005.03.021
Penning TD, Talley JJ, Bertenshaw SR, Carter JS, Collins PW, Doctor S, Graneto MJ, Lee LF, Malecha JW, Miyashiro JM, Rogers RS, Rogier DJ, Yu SS, Anderson GD, Burton EG, Cogburn JN, Gregory SA, Koboldt CM, Perkins WE, Seibert K, Veenhuizen AW, Zhang YY, Isakson PC (1997) Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors:? identification of 4-[5-(4-methylphenyl)-3- (trifluoromethyl)-1\(H\)-pyrazol-1-yl]benzenesulfonamide (SC-58635, celecoxib). J Med Chem 40:1347–1365. doi: 10.1021/jm960803q
Terrett NK, Bell AS, Brown D, Ellis P (1996) Sildenafil (\(\text{ VIAGRA }^{TM})\), a potent and selective inhibitor of type 5 cGMP phosphodiesterase with utility for the treatment of male erectile dysfunction. Bioorg Med Chem Lett 6:1819–1824. doi:10.1016/0960-894X(96)00323-X
Guzman-Perez A, Webster RT, Allen MC, Brown JA, Buchholz AR, Cook ER, Day WW, Hamanaka ES, Kennedy SP, Knight DR, Kowalczyk PJ, Marala RB, Mularski CJ, Novomisle WA, Ruggeri RB, Tracey WR, Hill RJ (2001) Discovery of zoniporide: A potent and selective sodium-hydrogen exchanger type 1 (NHE-1) inhibitor with high aqueous solubility. Bioorg Med Chem Lett 11:803–807. doi:10.1016/S0960-894X(01)00059-2
Kordik CP, Luo C, Zanoni BC, Lovenberg TW, Wilson SJ, Vaidya AH, Crooke JJ, Rosenthal DI, Reitz AB (2001) Pyrazolecarboxamide human neuropeptide Y5 receptor ligands with in vivo antifeedant activity. Bioorg Med Chem Lett 11:2287–2290. doi:10.1016/S0960-894X(01)00449-8
Genin MJ, Biles C, Keiser BJ, Poppe SM, Swaney SM, Tarpley WG, Yagi Y, Romero DL (2000) Novel 1,5-diphenylpyrazole nonnucleoside HIV-1 reverse transcriptase inhibitors with enhanced activity versus the delavirdine-resistant P236L mutant:? lead identification and SAR of 3- and 4-substituted derivatives. J Med Chem 43:904–909. doi:10.1021/jm990383f
Hanzlowsky A, Jelencic B, Recnik S, Svete J, Golobic A, Stanovnik B (2003) Regioselective synthesis of ethyl pyrazolecarboxylates from ethyl 3-[(dimethylamino)methylidene]pyruvate and diethyl 3-[(dimethylamino)methylidene]-2-oxosuccinate. Isolation of ethyl 4,5-dihydro-1-heteroaryl-5-hydroxy-1 H -pyrazole-5-carboxylates as stable intermediates in the pyrazole ring formation. J Heterocycl Chem 40:487–498. doi:10.1002/jhet.5570400313
Varvounis G, Fiamegos Y, Pilidis G (2004) Pyrazol-3-ones. Part II: reactions of the ring atoms. Adv Heterocycl Chem 87:141–272
Salaheldin AM, Oliveira-Campos AMF, Rodrigues LM (2007) N-Bromosuccinimide assisted oxidation of 5-aminopyrazoles: formation of bis diazenylderivatives. Tetrahedron Lett 48:8819–8822. doi:10.1016/j.tetlet.2007.10.079
Varvounis G, Fiamegos Y, Pilidis G (2007) Pyrazol-3-ones. Part III: reactivity of the ring substituents. Adv Heterocycl Chem 95:27–141
Fustero S, Simon-Fuentes A, Sanz-Cervera JF (2009) Recent advances in the synthesis of pyrazoles. Org Prep Proced Int 41: 253–290. doi:10.1080/00304940903077832
Heller ST, Natarajan SR (2006) 1,3-Diketones from acid chlorides and ketones: a rapid and general one-pot synthesis of pyrazoles. Org Lett 8:2675–2678. doi:10.1021/ol060570p
Aggarwal VK, Vicente J, Bonnert RV (2003) A novel one-pot method for the preparation of pyrazoles by 1,3-dipolar cycloadditions of diazo compounds generated in situ. J Org Chem 68:5381–5383. doi:10.1021/jo0268409
Martin R, Rivero MR, Buchwald SL (2006) Domino Cu-catalyzed C–N coupling/hydroamidation: a highly efficient synthesis of nitrogen heterocycles. Angew Chem Int Ed Engl 45:7079–7082. doi:10.1002/anie.200602917
Neumann JJ, Suri M, Glorius F (2010) Efficient synthesis of pyrazoles: oxidative C–C/N–N bond-formation cascade. Angew Chem Int Ed 49:903–907. doi:10.1002/anie.201002389
Antilla JC, Baskin JM, Barder TE, Buchwald SL (2004) Copper–diamine-catalyzed N-arylation of pyrroles, pyrazoles, indazoles, imidazoles, and triazoles. J Org Chem 69:5578–5587. doi:10.1021/jo049658b
Cristau HJ, Cellier PP, Spindler JF, Taillefer M (2004) Mild conditions for copper-catalysed N-arylation of pyrazoles. Eur J Org Chem 2004:695–709. doi:10.1021/jo049658b
Mukherjee A, Sarkar A (2004) Pyrazole-tethered arylphosphine ligands for Suzuki reactions of aryl chlorides: how important is chelation? Tetrahedron Lett 45:9525–9528. doi:10.1016/j.tetlet.2004.11.016
Zhu L, Guo P, Li G, Lan J, Xie R, You J (2007) Simple copper salt-catalyzed N-arylation of nitrogen-containing heterocycles with aryl and heteroaryl halides. J Org Chem 72:8535–8538. doi:10.1021/jo0712289
Xi Z, Liu F, Zhou Y, Chen W (2008) CuI/L (L=pyridine-functionalized 1,3-diketones) catalyzed C–N coupling reactions of aryl halides with NH-containing heterocycles. Tetrahedron 64:4254–4259. doi:10.1016/j.tet.2008.02.082
Goikhman R, Jacques TL, Sames D (2009) C–H bonds as ubiquitous functionality: a general approach to complex arylated pyrazoles via sequential regioselective \(C\)-arylation and \(N\)-alkylation enabled by SEM-group transposition. J Am Chem Soc 131:3042–3048. doi:10.1021/ja8096114
Hasaninejad A, Shekouhy M, Golzar N, Zare A, Doroodmand MM (2011) Silica bonded n -propyl-4-aza-1-azoniabicyclo[2.2.2]octane chloride (SB-DABCO): a highly efficient, reusable and new heterogeneous catalyst for the synthesis of 4 H -benzo[b] pyran derivatives. Appl Catal A: General 402:11–22. doi:10.1016/j.apcata.2011.04.012
Aziz SI, Kandeel ZE, Husein MM, El-Gohary S, El-Maged EIA (1995) Activated nitriles in heterocyclic synthesis: Novel syntheses of 3H–1,2,4-triazolo-[1,5-a]pyridine derivatives. Heteroat Chem 6:319–323. doi:10.1002/hc.520060408
Verma D, Mobin S, Namboothiri INN (2011) Highly selective synthesis of pyrazole and spiropyrazoline phosphonates via base-assisted reaction of the \(\text{ Bestmann }\_\text{ Ohira }\) reagent with Enones. J Org Chem 76:4764–4770. doi:10.1021/jo200582z
Balachandran KS, Bhatnagar I, George MV (1968) Oxidation by metal oxides. IV. Oxidation of organic compounds using nickel peroxide. J Org Chem 33:3891–3895. doi:10.1021/jo01274a043
Huang YR, Katzenellenbogen JA (2000) Regioselective synthesis of 1,3,5-triaryl-4-alkylpyrazoles: novel ligands for the estrogen receptor. Org Lett 2:2833–2836. doi:10.1021/ol0062650
Jung ME, Min SJ, Houk KN, Ess D (2004) Synthesis and relative stability of 3,5-diacyl-4,5-dihydro-1 \(H\)-pyrazoles prepared by dipolar cycloaddition of enones and \(\alpha \)-diazoketones. J Org Chem 69:9085–9089. doi: 10.1021/jo048741w
Acknowledgments
The authors thank Persian Gulf University Research Councils for the financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hasaninejad, A., Firoozi, S. Catalyst-free, one-pot, three-component synthesis of 5-amino-1,3-aryl-1\(H\)-pyrazole-4-carbonitriles in green media. Mol Divers 17, 459–469 (2013). https://doi.org/10.1007/s11030-013-9445-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-013-9445-y