Abstract
Biglycan (BGN) is overexpressed in cancer stem cells of colon cancer and induces the activation of NF-κB pathway which contributes to the chemotherapy resistance of diverse cancer types. Therefore, we hypothesized that the overexpression of BGN also promoted the development of multiple drug resistance (MDR) in colon cancer via NF-κB pathway. The expression of BGN was bilaterally modulated in colon cancer cell lines HT-29 and SW-480 and the effect of treatments on the cell proliferation and resistance to 5-FU was assessed. Moreover, the role of NF-κB signaling in the BGN-mediated formation of MDR was further investigated by subjecting BGN-overexpressed SW-480 cells to the co-treatment of chemo-agents and NF-κB inhibitor, PDTC. The inhibition of BGN expression decreased the proliferation potential of HT-29 cells while the induction of BGN expression increased the potential of SW-480 cells. BGN knockdown increased HT-29 cells’ sensitivity to 5-FU, represented by the lower colony number and higher apoptotic rate. To the contrary, BGN overexpression promoted the resistance of SW-480 cells to 5-FU. The effect of BGN modulation on colon cancer cells was associated with the changes in apoptosis and NF-κB pathways: BGN inhibition increased the expressions of pro-apoptosis indicators and suppressed NF-κB pathway activity while BGN overexpression had the opposite effect. It was also found that the BGN-mediated formation of MDR was impaired when NF-κB pathway was blocked. Findings outlined in the current study showed that BGN contributed to the formation of chemotherapy resistance in colon cancer cells by activating NF-κB signaling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colon cancer is the third most frequently diagnosed cancer type worldwide [1,2,3], with more than 1.2 million new cases being confirmed and 600 thousand patients dead each year. The incidence of colon cancer increases with age and is reported to be higher in countries in Europe, North America, and Oceania [4]. Currently, surgery and chemotherapy are the two major treating strategies for the management of colon cancer. Compared with tumor resection, chemotherapies are more universal for colon cancer patients at different stages and are often employed as the adjuvant therapy after tumor removal [5]. However, more than 50% colon cancer patients will develop metastasis even after the initial treatment for localized tumors [6, 7], which limits the treating efficacy of chemical agents [8].
It is generally recognized that the chemotherapy failure in the treatment of cancers is attributed to the development of multidrug resistance (MDR). MDR of cancers is referred to the resistance to multiple structurally and functionally unrelated chemotherapies after the exposure to a single cytotoxic compound [9]. There is emerging evidence showing that the existence of cancer stem cells (CSCs) is responsible for the formation of MDR in colon cancer [10]. The CSC theory infers that only a subpopulation of cells within a tumor, termed as CSC, has the capability to regenerate, sustain the growth of tumors, and increase cancers’ resistance to drugs [11]. Regarding colon cancer, the CD133+ CSC population has been proved to be capable of inducing tumor formation [12] and is more resistant to 5-FU-based chemotherapies [10]. Furthermore, according to the study of Fang et al., except for expressing surface antigens such as CD133 and CD44, the colon cancer CSCs are also characterized by the high expression of biglycan (BGN) [13]. In our previous studies, the modulation of BGN influenced the growth, metastasis, and angiogenesis of colon cancer cells [14, 15]. Therefore, it is reasonable to investigate the role of BGN in the formation of chemotherapy resistance in colon cancer as well.
BGN is a classical type of extracellular matrix proteins that play important roles in the modulation of the morphology, growth, migration, and differentiation of epithelial cells [16]. The dys-expression of BGN has been reported in osteosarcoma and ovarian cancers, and has been proved to be associated with the chemotherapy-resistant phenotypes of these cancers [17, 18]. The overexpression of BGN induces the activation of NF-κB pathway via TLR2/4 signaling [19, 20]. The activation of NF-κB is associated with the aggressive growth of tumors and contributes to the resistance to chemotherapies during cancer treatments [21]. Regarding colon cancer, the key role of NF-κB signaling in promoting the cancer progression is also becoming clear [22, 23]. Taken the information together, the major purpose of the current study was to explore the role of BGN in the chemotherapy resistance of colon cancer. To underline the mechanism driving the function of BGN, the current study also focused on its interaction with NF-κB signaling. To fulfill the purpose, the expression of BGN was bilaterally modulated in colon cancer cell lines and the effect of different treatments on the cell characteristics, resistance to chemotherapeutic agents, and NF-κB pathway were detected.
Materials and methods
Cell cultures
Human cancer cell lines SW-480, HCT166, and HT-29 were purchased from Shanghai Institute of Cell Biology, Chinese Academy of Sciences (Shanghai, China) and maintained in dulbecco’s modified eagle medium (DMEM) (catalog. no. 12100-46, Gibco, USA) supplemented with 10% fetal bovine serum (FBS) (catalog. no. SH30084.03, Hyclone, USA) at 37 °C in an atmosphere consisting of 5% CO2 and 95% air. Cells from passage three to five were used for subsequent assays. The expression levels of BGN in the three cell lines were determined using reverse transcription real time PCR (RT2-PCR) and western blotting assay as described in the following.
Construction of expression and interference vectors for BGN
Specific shRNA targeting BGN gene (5′-GCUCAACUACCUGCGCAUC-3′) was ligated into pRNA-H1.1 plasmid (between the HindIII and BamHI sites) to form BGN knockdown vector pRNA-H1.1-shBGN (BGN-shRNA). A non-targeting shRNA was ligated into pRNA-H1.1 plasmid to serve as negative control (pRNA-H1.1-NC) for assays based on BGN knockdown. The coding sequence of human BGN was amplified based on the mRNA sequence (NM_001711.4) and ligated into pcDNA3.1 plasmid between EcoRI and XhoI sites to form BGN expression vector pcDNA3.1-BGN (BGN-OE). The empty pcDNA3.1 plasmid was served as NC for assays based on BGN overexpression.
Transfection
Transfections were performed using Liposome 2000 (11668-019, Invitrogen, USA) according to the manufacturers’ instruction. Based on the results of RT2-PCR and western blotting detections, the expression level of BGN was highest in HT-29 cells while lowest in SW-480 cells (Figure S1). Thus, the level of BGN was knocked down in HT-29 cells but induced in SW-480 cells using respective vectors. The stable transfected cells were screened using G418 (catalog. no. 11811023, Invitrogen, USA) and cultured in DMEM supplemented with 10% FBS.
RT2-PCR
Total RNA was extracted using RNA Purified Total RNA Extraction Kit (catalog. no. RP1201, BioTeke, China). cDNA templates were achieved using super M-MLV reverse transcriptase (catalog. no. PR6502, BioTeke, China) from total RNA. The reaction mixture of real time PCR contained 10 µl SYBR GREEN Master Mix (catalog. no. SY1020, Solarbio, China), 0.5 µl of each primer (BGN, forward: 5′-GGGTCTCCAGCACCTCTACGC-3′, backward: 5′-TGAACACTCCCTTGGGCACCT-3′; β-actin, forward: 5′-CTTAGTTGCGTTACACCCTTTCTTG-3′, backward: 5′-CTGTCACCTTCACCGTTCCAGTTT-3′), 1 µl cDNA template, and 8 µl ddH2O. Amplification was performed following thermal cycling parameters: a denaturation step at 94 °C for 10 min, followed by 40 cycles of amplification at 94 °C for 10 s, 60 °C for 20 s and 72 °C for 30 s. The signal was detected after the step at 72 °C for 30 s using Exicycler™ 96 (BIONEER, South Korea). The relative expression level of BGN was calculated according to the formula of 2−ΔΔct.
Western blotting
Cells in different groups were lysated using RIPA lysis buffer (catalog. no. P0013B, Beyotime Biotechnology, China) supplemented with 1% PMSF (catalog. no. ST506, Beyotime Biotechnology, China). Total protein in cells was collected by centrifugation at 10,000 × g for 4 min. The protein concentration was determined using BCA Protein Concentration Determination Kit (catalog. no. P0009, Beyotime Biotechnology, China) according to the manufacturers’ instruction. For western blotting assay, 40 µg protein from each sample were subjected to 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS–PAGE) at 80 V for 1.5 h. Then the protein was transferred onto polyvinylidene difluoride (PVDF) membranes and blocked with skimmed milk solution for 1 h. Afterwards, the membranes were incubated with the primary antibodies against BGN (1:1000) (catalog. no. D221985, Sangon Biotech, China), cleaved caspase 3 (1:1000) (catalog. no. ab2302, Abcam, UK), cleaved caspase 9 (1:1000) (catalog. no. #7237, CST, USA), cleaved PARP (1:1000) (catalog. no. ab32561, Abcam, UK), IκBα (1:500) (catalog. no. bs-1287R, Bioss, China), p-IκBα (1:500) (catalog. no. bs-2513R, Bioss, China), p-p65 (Ser536) (1:1000) (catalog. no. D155097, Sango Biotech, China), β-actin (1:500) (catalog. no. KGAA001-1, KeyGEN BioTECH, China), and Histone H3 (1:500) (catalog. no. Bioss, bs-17422R, China) at 4 °C overnight. After four washes with TTBS, secondary HRP-conjugated IgG antibodies (1:5000) (catalog. no. A1018&A0208&A0216, Beyotime Biotechnology, China) were added onto the membranes and incubated for 45 min at 37 °C. The protein bands were developed using the Beyo ECL Plus reagent (catalog. no. P0018, Beyotime, China) and the images were captured using Gel Imaging System. The relative expression levels of proteins were calculated using Gel-Pro Analyzer (Media Cybernetics, USA).
Chemo-agent administration
Colon cancer cells were firstly allowed to grow into 90% confluence before incubated with 5-fluorouracil (5-FU) (catalog. no. V900394, Sigma, USA) at different concentrations (0, 5, 10, 20, 30, and 50 µg/ml) at 37 °C for 48 h. For SW-480 cells transfected with BGN expression vector, cells were further incubated with cisplatin (catalog. no. 15663-27-1, Meilunbio, China) at different concentrations (0, 2.5, 5, 10, 20, 40 mg/l) at 37 °C for 48 h. The IC50 concentrations of different chemo-agents on cells underwent different administrations were determined using MTT assays as described following. For assays including colony formation, Hoechst staining, flow cytometry, and western blotting, the detections were all performed based on IC50 concentrations. To explore the role of NF-κB signaling, SW-480 cells overexpressing BGN were treated with 10 µM PDTC for 1 h (NF-κB inhibitor, catalog. no. S1809, Beyotime Biotechnology, China) 1 h prior to 5-FU or cisplatin treatment .
MTT assay
MTT detection was performed by incubating cells that underwent different treatments with 0.5 mg/ml MTT at 37 °C for 4 h. The reaction was stopped by DMSO and the cell growth was represented by OD value at 570 nm detected by a microplate reader (ELX-800, BIOTEK, USA).
Colony formation assay
Colon cancer cells in different groups were suspended in the culture media and then inoculated onto 35 mm plates at a density of 300 cells per plate. After culture at 37 °C for 2 h, the cells were treated with 5-FU at IC50 concentrations for 72 h. After another culture for 2 weeks, the colonies on the plates were fixed with 4% paraformaldehyde for 20 min and then stained with Wright–Giemsa stain for 5 min. The numbers of colonies (> 50 cells) on each plate were recorded using an inverted phase microscope (AE31, Motic, China). The colony formation rate is equal to “colony number/inoculated cell number × 100%.”
Flow cytometry
Apoptotic rates were determined with Cell Cycle Detection Kit (catalog. no. KGA106, KeyGen BioTECH, China) according to the manufacturers’ instruction using a FACS flow cytometer (Accuri C6, BD, USA): briefly, cells in different groups were re-suspended using 500 µl Binding buffer and then incubated with 5 µl Annexin V-FITC and 5 µl Propidium Iodide at room temperature in dark for 15 min. The total apoptotic rate was the sum of the late apoptotic rate (UR, upper right quadrant–advanced stage apoptosis) and the early apoptotic rate (LR, lower right quadrant–prophase apoptosis).
Hoechst staining
Colon cancer cells administrated with different agents for 48 h were cultured with fixing solution for 20 min at room temperature and then washed with PBS for two times. Hoechst 33,258 solution was added into the wells and incubated for 5 min. The morphological changes of cell nuclei were detected using a fluorescence microscope (BX53, Olympus, Japan) at 400 × magnification.
Statistical analysis
The data were expressed as mean ± standard deviation (SD). One-way ANOVA and post hoc test using LSD method were performed with a significant level of 0.05 (two-tailed P value). All the statistical analyses were conducted using Graphpad Prism version 6.0 (GraphPad Software, Inc., San Diego, CA).
Results
Effect of BGN modulation on the proliferation potential of colon cancer cells
Based on the detections of BGN expression status with RT2-PCR and western blotting, the level of BGN was highest in HT-29 cells and lowest in SW-480 cells (Figure S1). Therefore, HT-29 cells were subjected to BGN knockdown administration (Fig. 1a) and SW-480 cells were subjected to BGN induction administration (Fig. 1b). Afterwards, the effect of BGN modulation on the proliferation abilities of both cell lines was assessed with MTT assay in a 96-h time period. As shown in Fig. 1c, the proliferation of HT-29 cells was impaired by BGN knockdown and the OD570 values of BGN-shRNA Group were lower than those in NC Groups, and the differences were statistically significant since the 72nd hour (P < 0.05). In contrary to the effect of BGN knockdown, the effect of BGN overexpression promoted the proliferation of SW-480 cells, and the differences between BGN-OE Group and NC Group were also statistically significant since the 72nd hour (Fig. 1d) (P < 0.05).
Effect of BGN on the proliferation potential of colon cancer cells. The expression of BGN was knockdown in HT-29 cells and was induced in SW-480 cells, and the proliferation potential of the cells were detected by MTT assay. a RT2-PCR and western blotting detection results of the expression of BGN in HT-29 cells. b RT2-PCR and western blotting detection results of the expression of BGN in SW-480 cells. c MTT assay detection results of HT-29 cells. d MTT assay detection results of SW-480 cells. “*” P < 0.05 versus NC/Vector Group
Effect of BGN modulation on the 5-FU resistance in colon cancer cells
Colon cancer cells with different BGN expression statuses were further incubated with 5-FU at different concentrations for 48 h and the cell viabilities were determined with MTT assay. As shown in Fig. 2a, b, the inhibiting effect of 5-FU on HT-29 cells was strengthened by BGN knockdown while suppressed by BGN overexpression. Based on the results of MTT assays, the IC50 values were calculated and shown in Tables S1 and S2. Then cells in different groups were treated with 5-FU at respective IC50 concentrations for 48 h before subjecting to colony formation assay, Hoechst staining, flow cytometry, and western blotting. The administrations of 5-FU decreased the colony formation rates in both cell lines, representing the inhibition on anchorage-independent growth of colon cancer cells (Fig. 3a, b). After BGN knockdown, the colony formation rate of HT-29 cells was further decreased (Fig. 3a) while BGN overexpression restored the colony formation rate of SW-480 cells (Fig. 3b). Similar changing patterns were also observed for detections of cell apoptosis: BGN knockdown increased the apoptotic rate and Hoechst positive cell number in 5-FU-treated HT-29 cells (Fig. 3c, e) while BGN overexpression inhibited the apoptosis-inducing effect of 5-FU on SW-480 cells (Fig. 3d, f).
MTT assay detection of the IC50 values of 5-FU on colon cancer cells. Colon cancer cells in different groups were treated with 5-FU at different concentrations (from 0 to 50 µg/ml) for 48 h and the cell viability was measured with MTT assay. a MTT assay detection results of HT-29 cells. b MTT assay detection results of SW-480 cells. “*” P < 0.05 versus NC + 5-FU/Vector + 5-FU Group
Effect on BGN on the 5-FU resistance of colon cancer cells. Cells in different groups were treated with 5-FU at IC50 concentrations and subjected to colony formation, flow cytometry, and Hoechst staining to determine the resistance to 5-FU. a Representative images and quantitative analysis results of colony formation assay of HT-29 cells. b Representative images and quantitative analysis results of colony formation assay of SW-480 cells. c Representative images and quantitative analysis results of flow cytometry of HT-29 cells. d Representative images and quantitative analysis results of flow cytometry of SW-480 cells. e Representative images and quantitative analysis results of Hoechst staining of HT-29 cells. f Representative images and quantitative analysis results of Hoechst staining of SW-480 cells. “*” P < 0.05 versus HT-29/SW-480 Group. “#” P < 0.05 versus NC + 5-FU/Vector + 5-FU Group. Magnification, 400 ×
Effect of BGN modulation on apoptosis and NF-κB signal transduction in colon cancer cells treated with 5-FU
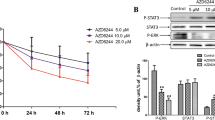
At molecular level, the knockdown of BGN induced the expressions of cleaved caspase 3, cleaved caspase 9, and cleaved PARP (Fig. 4a) while BGN overexpression inhibited the expressions (Fig. 4b), which further confirmed the pro-survival effect of BGN in the development of colon cancer. NF-κB signal transduction is a classical pathway promoting the chemotherapy resistance of cancer cells [21]. Therefore, the effect of BGN modulation on the activity of NF-κB pathway in colon cancer cells treated with 5-FU was also detected. In HT-29 cells, the knockdown of BGN decreased the levels of p-p65 and p-IκBα while increased the level of IκBα (Fig. 4c). The administration 5-FU activated NF-κB pathway in HT-29 cells by increasing the expressions of p-p65, p-IκBα and decreasing the expression of IκBα (Fig. 4c). The inhibiting effect of BGN knockdown on NF-κB pathway could also be observed under the treatment of 5-FU (Fig. 4c). Compared with HT-29 cells, the administration of 5-FU had little influence on the activity of NF-κB pathway in SW-480 cells (Fig. 4d). However, the levels of p-p65 and p-IκBα were dramatically higher in SW-480 cells co-treated with 5-FU and BGN expression vector than those in SW-480 cells solely treated with BGN expression vector (Fig. 4d).
Effect on BGN on apoptosis and NF-κB pathways in 5-FU-treated colon cancer cells. Cells in different groups were treated with 5-FU at IC50 concentrations and subjected to western blotting assay for determining the expressions of molecules involved in apoptosis and NF-κB pathways. a Representative images and quantitative analysis results of western blotting assay of apoptosis pathway in HT-29 cells. b Representative images and quantitative analysis results of western blotting assay of apoptosis pathway in SW-480 cells. c Representative images and quantitative analysis results of western blotting assay of NF-κB pathway in HT-29 cells. d Representative images and quantitative analysis results of western blotting assay of NF-κB pathway in SW-480 cells. “*” P < 0.05 versus HT-29/SW-480 Group. “#” P < 0.05 versus NC + 5-FU/Vector + 5-FU Group. “&” P < 0.05 versus BGN-shRNA/BGN-OE Group
Role of NF-κB pathway in mediating the function of BGN in the development of MDR of colon cancer cells
Our results have verified the activating effect of BGN on NF-κB pathway. However, whether the function of BGN in promoting the chemotherapy resistance of colon cancer cells indispensably depended on the activation of NF-κB pathway still needed to be explored. Therefore, SW-480 cells with BGN overexpression were subjected to the treatments of 5-FU and cisplatin, both of which were co-treated with NF-κB inhibitor PDTC. After 48 h, the apoptotic process was detected with flow cytometry and Hoechst staining. The results showed that once the activity of NF-κB pathway was inhibited by PDTC, the effect of BGN overexpression in inducing chemotherapy resistance was blocked. As shown in Fig. 5, compared with SW-480 cells with BGN overexpression and chemo-agent administration, higher apoptotic rates and Hoechst staining positive cell numbers were recorded in PDTC-treated groups, indicating that the activation of NF-κB pathway was necessary for the BGN-induced development of MDR in colon cancer cells. The central role of NF-κB pathway in the formation of MDR in colon cancer cells was further validated with SW-480 and SW-480 transfected with NC vector: after the inhibition of NF-κB pathway, the apoptotic process was further induced (Figure S2). Moreover, SW-480 in different groups had similar response to the co-administration of 5-FU and PDTC, indicating that the overexpression of BGN had no side effect on the biological behaviors of colon cancer cells.
Effect of NF-κB pathway on the BGN-mediated 5-FU resistance in SW-480 cells. SW-480 cells overexpressing BGN were pre-treated with NF-κB inhibitor PDTC (10 µM) for 1 h before being treated with 5-FU or cisplatin at IC50 concentration. Then the apoptotic rates were detected with flow cytometry and Hoechst staining. a Representative images and quantitative analysis results of flow cytometry of SW-480 cells treated with 5-FU. b Representative images and quantitative analysis results of flow cytometry of SW-480 cells treated with cisplatin. c Representative images and quantitative analysis results of Hoechst staining of SW-480 cells treated with 5-FU. d Representative images and quantitative analysis results of Hoechst staining of SW-480 cells treated with cisplatin. “*” P < 0.05 versus BGN-OE Group. “#” P < 0.05 versus BGN-OE + 5-FU/BGN-OE + cisplatin Group. Magnification, 400 ×
Discussion
Colon cancer cell lines with high and low expression levels of BGN were selected in the current study to detect the effect of BGN modulation on the development of MDR. Based on the current results, the induced expression of BGN contributed to the higher resistance of SW-480 cells to the administrations of 5-FU and cisplatin while the suppressed expression of BGN lowered the primary 5-FU resistance of HT-29 cells. By detecting the influence of BGN modulation on the activity of NF-κB pathway, our study also showed that the function of BGN in promoting the MDR feature of colon cancer cells was dependent on the activation of NF-κB signaling.
CSCs play a key role in driving the formation and spread of tumor, and in facilitating the drug resistance of colon cancers [24]. Based on the study of Fang et al., colon cancer CSCs are characterized by the high expressions of surface-associated proteins including CD133, CEACAM5, cadherin 17, CD29, and secreted protein BGN [13]. In our previous studies, we have proved that BGN promotes the growth, metastasis, and angiogenesis of colon cancer cells [14, 15] and is a critical modulator of the apoptotic process in colon cancer cells. In the current study, the role of BGN in the development of MDR of colon cancer cells were further studied. The expression of BGN was bilaterally modulated in two colon cancer cells. For HT-29 cells with high expression level of BGN, the cells were transfected with BGN knockdown vector. The suppression of BGN resulted in the decreased cell proliferation of HT-29 cells. Moreover, HT-29 cells with inhibited BGN expression were also more sensitive to the treatment of 5-FU. Compared with parental cells, BGN-suppressed cells exhibited lower growth potential and higher apoptotic rate when subjecting to 5-FU. Given that HT-29 cells is a colon cell with high chemotherapy resistance potential, the results inferred that the suppressed expression of BGN could block the development of MDR in colon cancer cells. To the contrary of HT-29 cells, colon cancer cell line SW-480 is cell type sensitive to the administration of 5-FU. Therefore, we induced the expression of BGN in SW-480 cells. It was found that the overexpression of BGN promoted the survival of SW-480 cells under the treatment of 5-FU, thus confirming that BGN was a promoting factor in the development of MDR in colon cancer.
The association of BGN with chemotherapy resistance has been previously reported in pediatric osteosarcoma [18]. But in most cases, BGN is conceived to be an autocrine angiogenic factor of tumor endothelial cells [25] and is expressed in inflammatory and fibrotic tissues [26,27,28]. Generally, BGN interacts with it receptors, TLR2/4, and subsequently influences the downstream pro-inflammation NF-κB pathway [19, 29]. Given that the dys-function of TLRs is associated with the oncogenesis of multiple tumor types [30, 31] and that NF-κB pathway is a contributor to the development of chemotherapy resistance in cancer cells [21, 22], the function of BGN to promote MDR in colon cancer might be associated with its regulation on NF-κB pathway. In both cell lines, the administration of 5-FU increased the expressions of p-p65 and p-IκBα and decreased the expression of IκBα, representing the development of chemotherapy resistance in the cells via the activation of NF-κB signaling. In HT-29 cells with BGN knockdown, the activity of NF-κB pathway was inhibited and in SW-480 cells with BGN overexpression, the activity of NF-κB pathway was induced. However, our results also showed the influence of BGN overexpression on NF-κB pathway in 5-FU-treated SW-480 cells was much stronger than that of BGN knockdown in 5-FU-treated HT-29 cells. We thought the difference in the changing pattern of NF-κB pathway between HT-29 and SW-480 cells was attributed to the original expression levels of NF-κB members which was already high in HT-29 cells. Following the verification of BGN’s influence on NF-κB pathway in colon cancer cells under 5-FU treatment, the role of NF-κB pathway in mediating the function of BGN was further explored. SW-480 cells overexpressing BGN were co-treated with 5-FU and PDTC or cisplatin and PDTC. The results showed that once the activity of NF-κB pathway was inhibited in colon cancer cells, the resistance against chemotherapies was also blocked even with the overexpression of BGN. The results evidently proved that the effect of BGN to promote the development of MDR in colon cancer was dependent on the activation of NF-κB pathway. With the activated NF-κB pathway, BGN can transduce its function to downstream factors such as COX-2, Cyclin D1, Bcl-2 family, Survivin, and epidermal growth factor receptor [21], influencing not only the chemotherapy resistance but also the normal behaviors of colon cancer cells.
Conclusively, the current study made a supplement to our serial studies focusing on the function of BGN in colon cancer. By activating NF-κB signaling, BGN promoted the formation of resistance to multiple chemo-agents. The findings also bridged the gap through which BGN influenced the apoptosis and angiogenesis in colon cancers. Taken together, BGN played multi-pronged roles in the development of colon cancer and regarding its function to facilitate the formation of chemotherapy resistance, the factor exerted its function by activating NF-κB pathway. Findings outlined in the current study provided a novel therapeutic target for the further development of anti-colon cancer strategies.
References
Gill S, Thomas RR, Goldberg RM (2003) Review article: colorectal cancer chemotherapy. Aliment Pharmacol Ther 18:683–692
Hu T, Fei Li L, Shen J, Zhang L, Hin Cho C (2015) Chronic inflammation and colorectal cancer: the role of vascular endothelial growth factor. Curr Pharm Des 21(21):2960–2967
Li LF, Chan RLY, Lu L, Shen J, Zhang L, Wu WKK, Wang L, Hu T, Li MX, Cho CH (2014) Cigarette smoking and gastrointestinal diseases: the causal relationship and underlying molecular mechanisms. Int J Mol Med 34(2):372–380
Center MM, Jemal A, Smith RA, Ward E (2009) Worldwide variations in colorectal cancer. CA 59(6):366–378
Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, Nordlinger B, Starling N (2010) Colorectal cancer. Lancet 375:1030–1047
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA 60(5):277–300
Van Cutsem EJ (2008) Advanced colorectal cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 19(2):21–22
Dallas NA, Xia L, Fan F, Gray MJ, Gaur P, Buren GV, Samuel S, Kim MP, Lim SJ, Ellis LM (2009) Chemoresistant colorectal cancer cells, the cancer stem cell phenotype, and increased sensitivity to insulin-like growth factor-I receptor inhibition. Cancer Res 69(5):1951
Thomas H, Coley HM (2003) Overcoming multidrug resistance in cancer: an update on the clinical strategy of inhibiting p-glycoprotein. Cancer Control 10(2):159
Ong CW, Kim LG, Kong HH, Low LY, Iacopetta B, Soong R, Salto-Tellez M (2010) CD133 expression predicts for non-response to chemotherapy in colorectal cancer. Mod Pathol 23(3):450–457
Ward RJ, Dirks PB (2007) Cancer stem cells: at the headwaters of tumor development. Annu Rev Pathol 2(2):175–189
Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R (2007) Identification and expansion of human colon-cancer-initiating cells. Nature 445(7123):111–115
Fang DD, Kim YJ, Lee CN, Aggarwal S, McKinnon K, Mesmer D, Norton J, Birse CE, He T, Ruben SM (2010) Expansion of CD133 + colon cancer cultures retaining stem cell properties to enable cancer stem cell target discovery. Br J Cancer 102(8):1265–1275
Xing X, Gu X, Ma T, Ye H (2015) Biglycan up-regulated vascular endothelial growth factor (VEGF) expression and promoted angiogenesis in colon cancer. Tumor Biol 36(3):1773–1780
Xing X, Gu X, Ma T (2015) Knockdown of biglycan expression by RNA interference inhibits the proliferation and invasion of, and induces apoptosis in, the HCT116 colon cancer cell line. Mol Med Rep 12(5):7538–7544
Frantz C, Stewart KM, Weaver VM (2010) The extracellular matrix at a glance. J Cell Sci 123(24):4195–4200
Pan S, Cheng L, White JT, Lu W, Utleg AG, Yan X, Urban ND, Drescher CW, Hood L, Lin B (2009) Quantitative proteomics analysis integrated with microarray data reveals that extracellular matrix proteins, catenins, and p53 binding protein 1 are important for chemotherapy response in ovarian cancers. OMICS 13(4):345–354
Mintz MB, Sowers R, Brown KM, Hilmer SC, Mazza B, Huvos AG, Meyers PA, LaFleur B, McDonough WS, Henry MM (2005) An expression signature classifies chemotherapy-resistant pediatric osteosarcoma. Cancer Res 65(5):1748–1754
Song R, Ao L, Zhao K-s, Zheng D, Venardos N, Fullerton DA, Meng X (2014) Soluble biglycan induces the production of ICAM-1 and MCP-1 in human aortic valve interstitial cells through TLR2/4 and the ERK1/2 pathway. Inflamm Res 63(9):703–710
Rajamannan NM (2006) Calcific aortic stenosis: a disease ready for prime time. Circulation 114(19):2007
Li F, Sethi G (2010) Targeting transcription factor NF-κB to overcome chemoresistance and radioresistance in cancer therapy. BBA-Rev Cancer 1805(2):167–180
Sakamoto K, Maeda S (2010) Targeting NF-κB for colorectal cancer. Expert Opin Ther Targets 14(6):593–601
Sui H, Zhou LH, Zhang YL, Huang JP, Liu X, Ji Q, Fu XL, Wen HT, Chen ZS, Deng WL (2016) Evodiamine suppresses ABCG2 mediated drug resistance by inhibiting p50/p65 NF-κB pathway in colorectal cancer. J Cell Biochem 117(6):1471–1481
Dick JE (2009) Looking ahead in cancer stem cell research. Nat Biotechnol 27(1):44–46
Yamamoto K, Ohga N, Hida Y, Maishi N, Kawamoto T, Kitayama K, Akiyama K, Osawa T, Kondoh M, Matsuda K (2012) Biglycan is a specific marker and an autocrine angiogenic factor of tumour endothelial cells. Br J Cancer 106(6):1214–1223
Westermann D, Mersmann J, Melchior A, Freudenberger T, Petrik C, Schaefer L, Lüllmann-Rauch R, Lettau O, Jacoby C, Schrader J (2008) Biglycan is required for adaptive remodeling after myocardial infarction. Circulation 117(10):1269–1276
Babelova A, Moreth K, Tsalastra-Greul W, Zeng-Brouwers J, Eickelberg O, Young MF, Bruckner P, Pfeilschifter J, Schaefer RM, Gröne H-J (2009) Biglycan, a danger signal that activates the NLRP3 inflammasome via toll-like and P2X receptors. J Biol Chem 284(36):24035–24048
Mohan H, Krumbholz M, Sharma R, Eisele S, Junker A, Sixt M, Newcombe J, Wekerle H, Hohlfeld R, Lassmann H (2010) Extracellular matrix in multiple sclerosis lesions: fibrillar collagens, biglycan and decorin are upregulated and associated with infiltrating immune cells. Brain Pathol 20(5):966–975
Song R, Zeng Q, Ao L, Jessica AY, Cleveland JC, Zhao K-s, Fullerton DA, Meng X (2012) Biglycan induces the expression of osteogenic factors in human aortic valve interstitial cells via Toll-like receptor-2. Arterioscler Thromb Vasc Biol 32(11):2711–2720
Shatz M, Menendez D, Resnick MA (2012) The human TLR innate immune gene family is differentially influenced by DNA stress and p53 status in cancer cells. Cancer Res 72(16):3948
Korneev KV, Atretkhany KN, Drutskaya MS, Grivennikov SI, Kuprash DV, Nedospasov SA (2017) TLR-signaling and proinflammatory cytokines as drivers of tumorigenesis. Cytokine 89:127
Acknowledgements
This study was supported by Grants from the Foundation of Department of Science and Technology, Liaoning Province (No. 2015020254) and the Wu Jieping Medical Foundation (No. 320.6750.17250).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose no conflict of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, B., Xu, T., Xu, X. et al. Biglycan promotes the chemotherapy resistance of colon cancer by activating NF-κB signal transduction. Mol Cell Biochem 449, 285–294 (2018). https://doi.org/10.1007/s11010-018-3365-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3365-1