Abstract
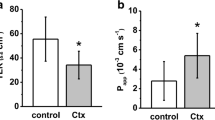
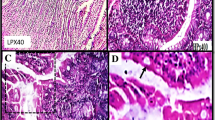
The activities of lactase, sucrase and alkaline phosphatase (AP) were studied in intestinal brush border membranes of control and toxin-treated rabbits. Purified Shiga toxin (Stx) exposure to ileal mucosa inhibited activities of brush border enzymes by 50%. Kinetic analysis revealed that the observed decrease in BBM enzyme activities was due to reduced Vmax with no change in the affinity constants of the systems. The observed changes in enzyme activities were corroborated by Western Blot analysis of lactase, sucrase and AP. The mRNA levels encoding sucrase and lactase proteins in control and Shiga toxin-treated rabbit ileum did not show any change in the rabbit ileum. Histopathological analysis showed short, blunt villi with increased number of inflammatory cells in the lamina propria and extrusion of cells in to the lumen of Stx-treated rabbit ileum. The present findings suggest that Shiga toxin act by inhibiting protein synthesis of these brush border functional proteins beyond their transcriptional level and by the direct damage to intestinal epithelium, which could be implicated in the pathogenesis of diarrhea.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sears CL, Kaper JB: Enteric bacterial toxins: Mechanisms of action and linkage to intestinal secretion. Microbiol Rev 60: 167–215, 1996
Donowitz M, Keusch GT, Binder HJ: Effect of Shigella enterotoxin on electrolyte transport in rabbit ileum. Gastroenterology 69: 1230–1237, 1975
Kaur T, Singh S, Ganguly NK: Role of enteric nervous system in Shigella dysenteriae1 toxin-induced fluid secretion in rabbit ileum. J Diarrhoeal Dis Res 13: 139–145, 1995
Binder HJ, Whiting DS: Inhibition of small intestinal sugar and amino acid transport by enterotoxin of Shigella dysenteriae1. Infect Immun 16: 510–512, 1977
De SN, Chatterjee DN: An experimental study on the mechanism of action of Vibrio cholera on the intestinal mucus membrane. J Pathol Bacteriol 66: 559–562, 1953
Schmitz J, Preiser H, Maestracci D, Ghosh BK, Cerdo JJ, Crane RK: Purification of the human intestinal brush border membrane. Biochim Biophys Acta 323: 98, 1973
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72: 248–254, 1976
Dahlqvist A: Method for the assay of intestinal disaccharidase. Anal Biochem 7: 18–25, 1964
Bergmeyer MVC: Method of enzymatic analysis, Vol. 40. Academic, New York, 1963, pp. 783–785
Towbin H, Stoehelin, T, Gorden J: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets, procedure and some applications. Proc Natl Acad Sci USA 76: 4350–4354, 1979
Chomezynski P, Sacchi N: Single-step method of RNA isolation by acidguanidinium thiocynate phenol chloroform extraction. Anal Biochem 162: 156–159, 1986
Lehrach H, Diamond D, Wozney JM, Bedtkar H: RNA molecular weight determinations of gel elecrophoresis under denaturing conditions, a critical examination. Biochemistry 16: 4743–4745, 1977
Reed KC, Mann DA: Rapid transfer of RNA from agarose gels to nylon membranes. Nucleic Acids Res 13: 7207, 1958
Mantei N, Villa M, Enzler T, Walker H, Bell W, Jame P, Hunziker W, Semenza G: Complete primary structure of human and rabbit lactase phlorizine hydolase. EMBO J 7: 2705–2713, 1988
Hanziker W, Spiess M, Semenza G, Lodish HF: Sucrose-isomaltase complex primary structure membrane orientation and evaluation of stalked intrinsic brush border protein. Cell 46: 227–234, 1986
Jacewicz M, Keusch GT: Pathogenesis of Shigella diarrhoea VIII. Evidence for translocation step in the cytotoxic action of Shiga toxin. J Infect Dis 148: 844–854, 1983
Obrig TG, Moran TP, Colinas RJ: Ribonuclease activity associated with the 60S ribosome-inactivating proteins ricin A, phytolaccin and Shiga toxin. Biochem Biophys Res Commun 130: 879–884, 1985
Endo Y, Mitsui K, Tsurugi K: The mechanism of action of ricin and related toxic lectins on eucaryotic ribosomes. J Biol Chem 262: 5908–5912, 1987
Chan YL, Endo Y, Wool IG: The sequence of the nucleotides at the α-sarcin cleavage site in rat 28S ribosomal ribonucleic acid. J Biol Chem 258: 12768–12770, 1983
Kandel G, Donohue-Rolfe A, Donowitz M, Keush GT: Pathogenesis of Shigella diarrhea. XVI. Selective targeting of Shiga toxin to villus cells of rabbit jejunum explains the effect of the toxin on intestinal electrolyte transport. J Clin Invest 84: 1509–1517, 1989
Broitmen SA: Gut lactase activity: decrease during Salmonella typhimurium infection in mice. Proc Soc Exp Biol Med 136: 1097–1099, 1971
Chitra, Suri P, Mahmood S: Effect of Salmonella typhimurium toxin on the expression of rabbit intestinal functions. Indian J Med Res 116: 186–191, 2002
Keush GT, Grady GF, Takeuchi A, Sprinz H: The pathogenesis of Shigella diarrhoea. III. Enterotoxin-induced acute enteritis in rabbit ileum. J Infect Dis 126: 92–95, 1972
Steinberg SE, Banwell JG, Yardley JH, et al. Comparison of secretory and histological effects of Shigella and cholera enterotoxins in rabbit jejunum. Gastroenterology 68: 309–317, 1975
Keenan KP, Sharpnack DD, Collins H, Formal SB, O'Brien: Morphologic evaluation of the effects of Shiga toxin and E. coli Shiga-like toxin on the rabbit intestine. Am J Pathol 125: 69–80, 1986
Buret A, O'Loughlin EV, Curtis GH, Gall DG: Effect of acute Yersinia enterocolitica infection on small intestinal ultrastructure. Gastroenterology 98: 1401–1407, 1990
O'Loughlin EV, Humphreys G, Dunn I, Kelly J, Lian CJ, Pai C, Gall DG: Clinical, morphological and biochemical alterations in acute intestinal Yersinosis. Pediatr Res 20: 602–608, 1986
Obrig TG, del Vecchio PJ, Brown JE, Moran TP, Powland BM, Judge TK, Rothman SW. Direct cytotoxic action of Shiga toxin on human vascular endothelial cells. Infect Immun 56: 2373–2378, 1988
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chopra, P., Verma, D., Khullar, M. et al. Shiga toxin exposure modulates intestinal brush border membrane functional proteins in rabbit ileum. Mol Cell Biochem 283, 85–92 (2006). https://doi.org/10.1007/s11010-006-2347-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-2347-x