Abstract
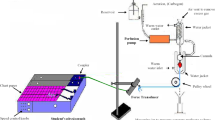
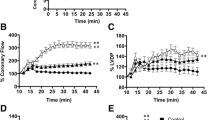
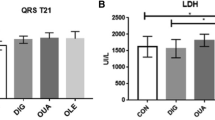
Previous results from our laboratory have shown that a novel carotenoid derivative (disodium disuccinate astaxanthin; CardaxTM) produced dose-related reductions in myocardial infarct size (IS) in Sprague–Dawley rats when it was administered at any of three doses (25, 50 and 75 mg/kg, iv) on four consecutive days, followed by the acute infarct size study on day 5. Maximum salvage occurred at the highest dose (75 mg/kg) tested, and was shown as a 56% reduction in IS. In the present follow-up study, we used a more relevant large animal model, the dog, and looked at the effect of administering CardaxTM iv either acutely 2 h prior to occlusion (N = 8) or for 4 days at 50 mg/kg iv as previously done in the rat model (N = 6). The results were compared to a saline vehicle-treated group (N = 10). In all groups, dogs were subjected to 60 min of left anterior descending (LAD) coronary artery occlusion and 3 h of reperfusion. IS was determined using a triphenyltetrazolium chloride (TTZ) histochemical stain and was expressed as a percent of the area at risk (IS/AAR). IS/AAR was 20.9 ± 1.6 % (mean ± S.E.M.) in controls and was reduced to 11.0± 1.7% (47.3% salvage; p < 0.01) in dogs treated only once iv at 2 h prior to occlusion, and 6.6± 2.8% (68.4% salvage; p < 0.001) in dogs treated for 4 days. In the chronic treatment group, two of the three dogs with plasma concentrations of non-esterified astaxanthin above 1 μM had 0% IS/AAR (100% cardioprotection). These results suggest that CardaxTM has marked cardioprotective properties in both rodents and canines. Thus, CardaxTM may be a novel and powerful new means to prevent myocardial injury and/or necrosis associated with elective and/or urgent cardiac surgical interventions such as coronary angioplasty and stenting, as well as coronary artery bypass surgery (CABG).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Britton G: Structure and properties of carotenoids in relation to function. FASEB J 9: 1551–1558, 1995
Britton G, Liaaen-Jensen S, Pfander H, Mercadante AZ, Egeland ES (eds). Carotenoids Handbook. Birkhäuser, Basel, 2004
Oliveros E, Braun AM, Aminian-Saghafi T, Sliwka HR: Quenching of singlet oxygen by carotenoid derivatives: Kinetic analysis by near-infrared luminescence. New J Chem 18: 535–539, 1994
Burton GW, Ingold KU: Beta-carotene: An unusual type of lipid antioxidant. Science 224: 569–573, 1984
Cardounel AJ, Dumitrescu C, Zweier JL, Lockwood SF: Direct superoxide anion scavenging by a disodium disuccinate astaxanthin derivative: Relative efficacy of individual stereoisomers versus the statistical mixture of stereoisomers by electron paramagnetic resonance imaging. Biochem Biophys Res Commun 307: 704–712, 2003
Jackson HL, Cardounel AJ, Zweier JL, Lockwood SF: Synthesis, characterization, and direct aqueous superoxide anion scavenging of a highly water-dispersible astaxanthin-amino acid conjugate. Bioorg Med Chem Lett 14: 3985–3991, 2004
Frey DA, Kataisto EW, Ekmanis JL, O’Malley S, Lockwood SF: The efficient synthesis of disodium disuccinate astaxanthin (Cardax). Org Process Res Dev 8: 796–801, 2004
Zsila F, Simonyi M, Lockwood SF: Interaction of the disodium disuccinate derivative of meso-astaxanthin with human serum albumin: From chiral complexation to self-assembly. Bioorg Med Chem Lett 13: 4093–4100, 2003
Gross GJ, Lockwood SF: Cardioprotection and myocardial salvage by a disodium disuccinate astaxanthin derivative (CardaxTM). Life Sci 75: 215–224, 2004
Østerlie M, Bjerkeng B, Liaaen-Jensen S: Plasma appearance and distribution of astaxanthin E/Z and R/S isomers in plasma lipoproteins of men after single dose administration of astaxanthin(1). J Nutr Biochem 11: 482–490, 2000
Showalter LA, Weinman SA, Østerlie M, Lockwood SF: Plasma appearance and tissue accumulation of non-esterified, free astaxanthin in c57bly6 mice after oral dosing of a disodium disuccinate diester of astaxanthin (HeptaxTM). Comp Biochem Physiol C Toxicol Pharmacol 137: 227–236, 2004
Mizumura T, Auchampach JA, Linden J, Bruns RF, Gross GJ: PD 81,723, an allosteric enhancer of the A1 adenosine receptor, lowers the threshold for ischemic preconditioning in dogs. Circ Res 79: 415–423, 1996
Mizumura T, Nithipatikom K, Gross GJ: Infarct size-reducing effect of nicorandil is mediated by the KATP channel but not by its nitrate-like properties in dogs. Cardiovasc Res 32: 274–285, 1996
O’Keefe JH Jr, Grines CL, DeWood MA, Bateman TM, Christian TF, Gibbons RJ: Factors influencing myocardial salvage with primary angioplasty. J Nucl Cardiol 2: 35–41, 1995
Kopecky SL, Aviles RJ, Bell MR, Lobl JK, Tipping D, Frommell G, Ramsey K, Holland AE, Midei M, Jain A, Kellett M, Gibbons RJ: AMP-579 delivery for myocardial infarction reduction study. Am Heart J 146: 146–152, 2003
Auchampach JA, Grover GJ, Gross GJ: Blockade of ischaemic preconditioning in dogs by the novel ATP dependent potassium channel antagonist sodium 5-hydroxydecanoate. Cardiovasc Res 26: 1054–1062, 1992
Auchampach JA, Maruyama M, Cavero I, Gross GJ: Pharmacological evidence. For a role of ATP-dependent potassium channels in myocardial stunning. Circulation 86: 311–319, 1992
Auchampach JA, Cavero I, Gross GJ: Nicorandil attenuates myocardial dysfunction associated with transient ischemia by opening ATP-dependent potassium channels. J Cardiovasc Pharmacol ([0-9]+): 765–771, 1992
Curry S, Brick P, Franks NP: Fatty acid binding to human serum albumin: New insights from crystallographic studies. Biochim Biophys Acta 1441: 131–140, 1999
Miki W: Biological functions and activities of animal carotenoids. Pure Appl Chem 63: 141–146, 1991
Hix LM, Lockwood SF, Bertram JS: Upregulation of connexin 43 protein expression and increased gap junctional communication by water soluble disodium disuccinate astaxanthin derivatives. Cancer Lett 211: 25–37, 2004
Kurihara H, Koda H, Asami S, Kiso Y, Tanaka T: Contribution of the antioxidative property of astaxanthin to its protective effect on the promotion of cancer metastasis in mice treated with restraint stress. Life Sci 70: 2509–2520, 2002
Foss BJ, Sliwka HR, Partali V, Cardounel AJ, Zweier JL, Lockwood SF: Direct superoxide anion scavenging by a highly water-dispersible carotenoid phospholipid evaluated by electron paramagnetic resonance (EPR) spectroscopy. Bioorg Med Chem Lett ([0-9]+): 2807–2812, 2004
Bertram JS: Carotenoids and gene regulation. Nutr Rev 57: 182–191, 1999
Hix LM, Lockwood SF, Bertram JS: Bioactive carotenoids: Potent antioxidants and regulators of gene expression. Redox Rep 9: 181–191, 2004
Lee SJ, Bai SK, Lee KS, Namkoong S, Na HJ, Ha KS, Han JA, Yim SV, Chang K, Kwon YG, Lee SK, Kim YM: Astaxanthin inhibits nitric oxide production and inflammatory gene expression by suppressing I(kappa)B kinase-dependent NF-kappab activation. Mol Cells 16: 97–105, 2003
Ohgami K, Shiratori K, Kotake S, Nishida T, Mizuki N, Yazawa K, Ohno S: Effects of astaxanthin on lipopolysaccharide-induced inflammation in vitro and in vivo. Invest Ophthalmol Vis Sci 44: 2694–2701, 2003
Li W, Hellsten A, Jacobsson LS, Blomqvist HM, Olsson AG, Yuan XM: Alpha-tocopherol and astaxanthin decrease macrophage infiltration, apoptosis and vulnerability in atheroma of hyperlipidaemic rabbits. J Mol Cell Cardiol 37: 919–948, 2004
Singh RB, Singh NK, Rastogi SS, Wander GS, Aslam M, Onouchi Z, Kummerow FA, Nangia S: Antioxidant effects of lovastatin and vitamin E on experimental atherosclerosis in rabbits. Cardiovasc Drugs Ther 11: 575–580, 1997
Aoi W, Naito Y, Sakuma K, Kuchide M, Tokuda H, Maoka T, Toyokuni S, Oka S,Yasuhara M, Yoshikawa T: Astaxanthin limits exercise-induced skeletal and cardiac muscle damage in mice. Antioxid Redox Signal 5: 139–144, 2003
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gross, G.J., Lockwood, S.F. Acute and chronic administration of disodium disuccinate astaxanthin (CardaxTM) produces marked cardioprotection in dog hearts. Mol Cell Biochem 272, 221–227 (2005). https://doi.org/10.1007/s11010-005-7555-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-7555-2