Abstract
Context
Anthropogenic land use and cover changes impact biodiversity worldwide. However, ecological groups are differently affected by landscape composition and configuration. Understanding which groups are negatively affected and which thrive in human-modified landscapes is of paramount importance for conservation management, especially for species such as the frugivorous birds, which play an essential role in seed dispersal.
Objectives
We evaluated the relative importance of landscape composition and configuration, explaining taxonomic and functional diversity and their effects on frugivorous birds in the Brazilian Atlantic Forest.
Methods
We used a robust dataset encompassing 153 forest fragments in the Atlantic Forest hotspot. We classified species as frugivores based on the percentage of fruits in the diet, and used functional traits related to seed dispersal to measure functional diversity.
Results
Our results showed that landscape composition was more important than landscape configuration explaining taxonomic and functional diversity of frugivorous birds. In addition, the interaction between landscape composition and configuration explained the loss of functional traits.
Conclusions
We demonstrate a disproportional importance of landscape composition explaining taxonomic and functional diversity of frugivorous birds, whereas the traits related to seed dispersal were explained by both compositional and configurational variables. Thus, we highlighted the need to maintain high habitat amount to increase taxonomic and functional diversity of frugivorous birds. However, the interaction of landscape composition and configuration is of paramount importance to sustain functional traits of frugivores in tropical forest landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The increase in habitat conversion to anthropogenic land uses leads to severe changes in the structure and composition of once pristine landscapes (Dunning et al 1992; Newbold et al 2016). These changes have accelerated in recent decades, mainly due to increased growing human requirements for food, energy, agricultural commodities, timber, and other naturals resources (Laurance et al. 2014; Barlow et al. 2018). Alterations in human-modified landscapes include changes in landscape composition (the relative amount of different land use and cover types) and configuration (the spatial arrangement of habitat and non-habitat remnants, e.g. whether the habitat is more continuous or more fragmented) (Dunning et al. 1992). Both are predicted to affect species and population persistence.
Although landscape composition and configuration can affect population persistence (Galán-Acedo et al. 2019), there is an ongoing debate on which of the two is the most important and whether species richness increases or decreases with increasing fragmentation (Fahrig 2017; Fletcher et al. 2018). Landscape composition has been proposed as the main driver of species persistence (Fahrig 2013): the habitat amount hypothesis posits that species richness can be predicted based on the amount of habitat in the surrounding landscape—independent of either patch size or isolation (Fahrig 2013). Several studies found that forest cover, a proxy of habitat amount and the most used landscape composition variable, has positive effects on species richness of different taxonomic groups such as birds (Carrara et al. 2015; Kormann et al. 2018), bats (Arroyo-Rodríguez et al. 2016), macro-moth (Merckx et al. 2019), and primates (Galán-Acedo et al. 2019).
On the other hand, some authors reinforce that the spatial arrangement of habitat remnants is also important, suggesting that habitat fragmentation also affects biodiversity (Ewers and Didham 2006; Haddad et al. 2015). In fact, some studies showed that landscape configuration can positively (Arroyo-Rodríguez et al. 2016; Coelho et al. 2016) or negatively (Coelho et al. 2016) affect species richness. Additionally, some studies suggested that fragmentation effects only occur at low (Andrén 1994) or intermediate (Pardini et al. 2010) levels of forest cover. Thus, additional studies are necessary to understand which landscape attributes affect species richness and the ecosystem services provided by different species, as well as the direction of these effects.
Disentangling the effects of landscape composition and configuration on species richness is of paramount importance to guide conservation strategies. However, the relative importance of landscape composition and configuration might also depend on the taxonomic group evaluated (Klingbeil and Willig 2009). For instance, forest-dependent species are generally more impacted by forest loss than generalist species (Morante-Filho et al. 2015). Forest-dependent frugivorous birds are especially sensitive to anthopogenic disturbances (Coelho et al. 2016; Kupsch et al. 2019), declining in deforested landscapes, whereas habitat generalist frugivorous species increase (Morante-Filho et al. 2018b). Frugivorous birds play a key role in seed dispersal (Wenny et al. 2011), mainly in tropical forests where up to 90% of woody species depend on animal services for seed dispersal (Jordano 2014). Overall, these forest-dependent species are replaced by non-forest species in less forested landscapes in a sort of compensatory dynamics (Morante-Filho et al. 2018b, a).
The vast majority of studies have focused on species richness, but habitat loss and fragmentation could also affect other facets of biodiversity in contrasting ways (Bregman et al. 2016; Chapman et al. 2018; Hatfield et al. 2018). For instance, functional diversity, which is related to the functions performed by species in relation to ecosystem services (Petchey and Gaston 2006), was observed to increase with increasing forest cover, whereas the effects of landscape configuration were inconsistent (Hatfield et al. 2018). Here, we used a robust dataset on frugivorous birds (Hasui et al. 2018) encompassing 153 forest fragments in the Brazilian Atlantic Forest to: (1) evaluate the relative importance of landscape composition and configuration and (2) their effect size on taxonomic and functional diversity of frugivorous birds. We hypothesized that landscape composition (measured as the amount of forest cover at the landscape scale) is more important than landscape configuration (measured as edge density and mean nearest neighborhood distance) explaining both taxonomic and functional diversity of frugivorous birds. We expected positive effects of forest cover on frugivorous birds richness due to factors such as decreased isolation among patches, changes in plant community composition, increased supplementary resources (Dunning et al. 1992) and increased vegetation complexity (Morante-Filho et al. 2018a) which in turn results in more resource availability in highly forested areas (Dunning et al. 1992; Pessoa et al. 2017b). We also expected positive effects of forest cover on functional diversity of frugivorous birds (Hatfield et al. 2018) becausae anthropogenic disturbances affect species with extreme traits such as large body size (Vollstädt et al. 2017), thus decreasing the functional space filled by the species (Laliberté and Legendre 2010) present in these areas.
Methods
Study area
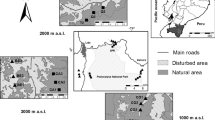
We used a published dataset on bird species (Hasui et al. 2018) encompassing the Brazilian Atlantic Forest (Fig. 1). Originally, this tropical forest covered an area of around 1.5 million square kilometers along the Brazilian coast (Joly et al. 2014). However, as a result of anthropogenic land use changes, less than 30% of its original forest cover still remain (Rezende et al. 2018). The Atlantic forest is one of the top five global biodiversity hotspots, presenting high species diversity, levels of endemism and number of threatened species (Myers et al. 2000; Joly et al. 2014). It harbors nearly 900 species of birds, 213 of which are endemic and 112 are threatened with extinction (Moreira-Lima 2014).
Map of the 153 study forest fragments in the Brazilian Atlantic Forest obtained from Hasui et al. (2018)
Bird assemblage selection
The database used here compiled bird studies for the whole Brazilian Atlantic Forest. This database cataloged 832 bird species in more than 4,000 sampling points using data from museum, on-line database, literature sources and unpublished records. Considering the three main quantitative sampling methods to inventory birds (mist net, point count and line transect) the dataset encompasses 576 communities (Hasui et al. 2018). We used only studies performed by Point counts (46% of studies out of 329) to standardize the methods and decrease the bias of different methodologies. In addition, point count is the most effective methodology for surveying birds in the Neotropics (Bibby et al 1992). We first used as inclusion criteria the precision of the geographic coordinate to select each assemblage. Thus, we only selected studies that had the coordinate of the central patch, coordinates of the four vertices of the patch, or the central coordinate of the mosaic of sampled habitat (see Hasui et al. 2018). We excluded studies carried out in anthropogenic habitats, restinga (coastal vegetation, including sand dunes and some types of coastal forest), or semideciduos urban forest. We included only fragments that had a minimum distance of two kilometers from one another to minimize spatial autocorrelation. When fragments were located within this distance, we chose the ones that had higher sampling effort. Our final dataset was composed of 153 forest fragments and 562 bird species. The year of sampling ranged from 1991 to 2014, and total effort ranged from 9 to 570 h (68.6 ± 88.2 mean ± SD).
Frugivore classification
From the 562 bird species recorded, we classified the frugivores following Kissling et al. (2009), which classify bird species based on nine categories of food items consumed (fruits, fish, nectar, vertebrates, terrestrial invertebrates, aquatic invertebrates, plant material, carrion, and seeds) and consider a species as frugivore if its diet is composed of more than 50% fruits. Thus, in our final dataset, only species that eat more than 50% fruits were included.
Functional traits
For all frugivorous species, we obtained four morphological traits that describe functions related to frugivory and seed dispersal by birds from the published literature (Table 1). Body mass reflects the amount of fruits that can be consumed by each species (Jordano and Schupp 2000), and was obtained from Wilman et al. (2014). Hand-wing index indicates the flight capability for long-distance dispersal (Weiss and Ray 2019) and was obtained from Bovo et al. (2018). Gape width represents the maximum fruit size that can be ingested (Wheelwright 1985). This trait was obtained from Bello et al. (2017) and Rodrigues et al. (2019). Foraging strata correspond to where the species forage and is an indicator of habitat use. For this trait we used Wilman et al. (2014) to determine the main forage strata used by each species. When a species used more than one stratum in equal proportion we classified it as “mixed” (Table 1).
Functional diversity
We calculated two functional diversity indexes that can be used for presence data (Laliberté and Legendre 2010) and are widely used to detect shifts in assembly processes related to disturbance (Mason et al. 2013). Functional richness (FRic) describes the niche space occupied by the community (Mason et al. 2005) and functional dispersion (FDis) depicts the mean distance of one species to the centroid of all species in the community (e.g. the spread of functional traits in the community) (Laliberté and Legendre 2010). To calculate the functional diversity indexes, we constructed a matrix of functional distance based on our matrix of species versus traits using Gower dissimilarity to account for categorical and continuous traits in our data (Podani and Schmera 2006). We used the package FD that tolerates missing data (Na’s) (Laliberté et al. 2014) to generate functional diversity index in R version 3.5.3 (R Core Team 2019). We excluded twelve sites that had less than four species because the “dbFD” function only calculates functional indexes if the number of species is equal to or higher than the number of traits (Laliberté et al. 2014).
We used null models to estimate the extent to which the functional diversity of bird communities is above or below that expected by chance (Gotelli and McCabe 2002). We standardized the effect size (SES) only for FRic because FDis is not affected by species richness (Laliberté and Legendre 2010). Standardized effect size for FRic (SESFRic) was calculated as (observed FRic – mean of expected FRic) / SD of expected FRic. Here, the expected FRic is the one expected under a null model based on the permutation of the presence/absence matrix (samples (rows) x species (columns)), with the trait matrix maintained as originally. We calculated these null models using the “independent swap” algorithm that randomizes the data matrix maintaining occurrence and sample richness (Gotelli 2000). The matrix was permuted 999 times according to the example from Plass-Johnson et al. (2016). Positive SES values indicate low functional redundancy, which can result from competitive exclusion, whereas negative values indicate high redundancy, which can be caused by environmental filtering (Mouchet et al. 2010).
Landscape descriptors
We calculated four metrics to describe landscape composition and configuration. We measured the percentage of forest cover as a descriptor of landscape composition and the number of fragments, mean nearest neighborhood distance, and edge density as metrics of landscape configuration. We used maps derived from 30 m resolution Landsat satellite images from MapBiomas (MapBiomas Project—Collection 3.1 of the Annual Series of Coverage and Land Use Maps in Brazil, accessed in [14/06/19]: [http://www.mapbiomas.org]). MapBiomas provides annual maps of land use and cover from 1985 to 2017. We downloaded maps considering the year each study started its sampling. We calculated all landscape metrics in QGIS (QGIS Development Team 2016) in the plugin LecoS (Jung 2016). All landscape metrics were calculated in a buffer radius of 1 km around the central point of each forest fragment. We choose this scale because each species respond differently to the landscape (Boscolo and Metzger 2009) and this scale represents the daily movement range of medium and small birds (Sekercioglu et al. 2007), which are the majority of species in our data. Our final dataset are representative of forest amount at the landscape scale in which 40 fragments (26%) have less than 30% of forest cover, 50 fragments (33%) have between 30 and 70% of forest cover and 63 fragments (41%) have more than 70% of forest cover (Supplementary material Fig. 1). See supplementary material for information on the mean, variance and range of each landscape predictor (Table S1).
Data analysis
We adjusted generalized linear mixed models using frugivorous bird richness as response variable for taxonomic diversity and linear mixed models for FDis and SESFRic for functional diversity. We used the study ID as random effect to account for studies that sampled multiples forest sites. We also used the coordinates of each forest fragment as covariate to account for spatial auto correlation in the form of spatial gradients (Beale et al. 2010). Because studies differ in sampling effort, we log transformed the sampling effort and included as offset in species richness models. We checked for collinearity among the explanatory variables with the variance inflation factor (VIF) (Dormann et al 2013), which resulted in less than four for all variables. However, we excluded the number of patches because its exclusion reduced VIF to 2.5 and because of its high correlation (r2 = 0.79) with mean nearest neighborhood distance. Thus, the explanatory variables for the final analysis were forest cover, mean nearest neighborhood distance and edge density. Because our variables have different scales, and to enable comparisons, we scaled and standardized all variables. For frugivore richness we used poisson distribution and for SESFRic and FDis we used normal distribution. We constructed models to test the effect of individual variables and of variable combinations as well as their interaction. Overall, we constructed 15 models (See script as supplementary resource). To account for the effect of landscape composition and configuration on functional traits (body mass, bill width and hand-wing index) we also constructed the same models using the mean value of each trait as response variable. We checked the spatial distribution of residuals (Supplementary Fig. 2) and semivariograms for all full models before the analysis.
To calculate variable importance, we used the function “AICctab” from package bbmle (Bolker 2020). We constructed a table with model weights and summed the Akaike weights (wi) of the models contained the variable in question (Burnham and Anderson 2002). We used the MuMIn package (Barton 2019) to perform model averaging (Burnham and Anderson 2002) to find the relative effect size of the explanatory variables using conditional coefficients. Model averaging calculates the average effect of each variable weighted by the Akaike weights of the models in which this variable occurs (Burnham and Anderson 2002). We performed all analysis in R version 3.5.3 (R Core Team 2019). The data and R codes used are available at https://github.com/CesarEco/Publications.
Results
From 562 bird species in our dataset, 96 were classified as frugivores (Appendix). Mean frugivore richness was 12.75 ± 7.03 SD (range 1—36). The most frequent species were Chiroxiphia caudata, Euphonia chlorotica, Patagioenas cayennensis, Psittacara leucophthalmus and Tangara sayaca, all present in more than 70 fragments. Eleven species occurred in a single fragment: Amazona farinosa, Aratinga solstitialis, Chlorophanes spiza, Chlorophonia cyanea, Crax fasciolata, Crypturellus strigulosus, Ortalis guttata, Pipraeidea bonariensis, Pteroglossus inscriptus, Tangara peruviana and Xipholena atropurpurea.
Landscape composition was the most important variable explaining taxonomic diversity of frugivorous birds (Fig. 2). Forest cover showed positive and significant effects on frugivore richness (Fig. 3, Table SII), whereas edge density and mean nearest neighborhood distance did not explain frugivore taxonomic diversity. In addition, the interactions between forest cover and edge density and forest cover and mean nearest neighborhood distance did not explain frugivorous richness (Fig. 3, Table SII).
Predictor variables and their importance for taxonomic and functional diversity and the traits of frugivorous birds in the Brazilian Atlantic Forest. The sum of Akaike weights (Σwi) shows the relative importance of each predictor variable for each response variable. FC = forest cover; ED = edge density; MNND = mean nearest neighborhood distance
Landscape effects on taxonomic and functional diversity and traits of frugivorous birds in the Brazilian Atlantic Forest. Significant results are those where the 95% confidence set do not overlap zero. Black dots are the estimate, blue lines are the adjusted standard error and the purple lines are the minimum and maximum confidence set. FC = forest cover; ED = edge density; MNND = mean nearest neighborhood distance; FC: ED = interaction between forest cover and edge density; FC: MNND = interaction between forest cover and mean nearest neighborhood distance; ED: MNND = interaction between edge density and mean nearest neighborhood distance
Considering functional diversity, forest cover was the most important variable (Fig. 2), negatively and significantly affecting both SESFRic and FDis (Fig. 3). Neither landscape configurational predictors nor interactions were significant for SESFRic or FDis.
Regarding the functional traits, the configurational predictor mean nearest neighborhood distance was important explaining hand-wing index, whereas for body mass and gape width forest cover, edge density and mean nearest neighborhood distance were equally important predictors (Fig. 2). However, no compositional or configurational metric alone were significant explaining functional traits (Fig. 3). Even so, for bill width, the interaction between forest cover and edge density was negative, meaning that increases in forest cover reduce the negative effects of edge density on bill width and vice-versa. In contrast, the interaction between edge density and mean nearest neighborhood distance were positive, showing that the bill sizes decreased with MNND, but this decrease is smaller when edge density is high (Fig. 4, Table SII).
Discussion
Our results encompassing the whole Brazilian Atlantic Forest show a disproportional importance of landscape composition than configuration for frugivorous birds. As predicted, forest loss has detrimental effects on frugivore diversity. However, we add important information showing that these effects are also pervasive on functional diversity with possible consequences for seed dispersal process. Interestingly, the combined effect of forest loss and fragmentation are affecting frugivore traits related to seed dispersal with a faster decline when fragmentation increase combined with high levels of habitat amount. These results emphasize that to sustain ecological processes performed by birds it is paramount to maintain high levels of forest amount as well as low levels of fragmentation.
We found that landscape composition affects the taxonomic diversity of frugivorous birds in the Atlantic Forest. As predicted, and consistent with several previous studies (Carrara et al. 2015; Arroyo-Rodríguez et al. 2016; Coelho et al. 2016; Godet et al. 2016; Galán-Acedo et al. 2019; Merckx et al. 2019), we demonstrated the importance of forest cover for species richness. On the other hand, edge density and mean nearest neighborhood distance were less important predictors of frugivore richness. But interestingly, forest cover did not interact with edge and isolation, explaining frugivore richness. It is worth noting that some studies pointed to an indirect effect of habitat configuration via habitat loss (Püttker et al. 2020) or even an intensified effect of landscape configuration at high levels of habitat loss (Villard and Metzger 2014). Our findings do not support these ideas, as we do not find interacting effects of habitat loss and fragmentation for species richness.
The positive effects of forest cover on frugivorous birds richness were expected. Our findings are consistent with previous studies that found increases in frugivorous birds diversity in forested landscapes (Morante-Filho et al. 2015; Coelho et al. 2016; Kupsch et al. 2019). Two main mechanisms might explain this pattern. First, local vegetation complexity increases in forested landscapes (Rocha-Santos et al. 2017), which results in an increase in niche width and resource exploitation (e.g. food, refuge, nesting sites) (MacArthur and MacArthur 1961). It has been shown that vegetation complexity directly affects forest-dependent frugivorous birds in the Brazilian Atlantic Forest (Morante-filho et al. 2018a). Second, landscape scale forest loss may reflect in the availability (Pessoa et al. 2017b) and quality (Pessoa et al. 2017a, b) of fruit resources, which in turn results in increased fruit consumption in forested landscapes (Menezes et al. 2016).
On the other hand, landscape configuration did not show significant effects on frugivorous richness. This result corroborates with those observed by Fahrig (2017), in which fragmentation per se (fragmentation independent of habitat amount) had in general positive or neutral effects on biodiversity. However, neutral effects were far more prevalent presented in more than 70% of the studies (Fahrig et al. 2019). However, other studies found negative effects of fragmentation and they argument that some species can be sensitive to habitat configuration, mainly when habitat amount is low (Martensen et al. 2012; Püttker et al. 2020), which for example, decreases functional connectivity and thus increases Allee effects (the fitness of individuals related to population density) (Villard and Metzger 2014) and decreases immigration rates of forest dependent species (Pardini et al. 2010; Martensen et al. 2012). Thus, our findings add important evidence that the fragmentation effects on richness of frugivorous birds may be absent.
Forest cover had negative effects on functional richness (either SESFRic or FDis), indicating lower functional redundancy in deforested landscapes. This result is in line with previous works (Prescott et al. 2016; Matuoka et al. 2020), which showed that bird communities in oil plantation and deforested landscapes had higher values of standardized functional diversity, respectively. In fact, forest loss can act as an environmental filter where forest-dependent species are negatively affected (Morante-Filho et al. 2018b). Yet, niche filtering excludes species with traits poorly adapted to ecosystem changes (Mouchet et al. 2010). On the other hand, positive SESFRic values can be found in deforested landscapes probably due to competitive exclusion, where the coexistence of dissimilar species is favored (Mouchet et al. 2010). In deforested landscapes a shift in bird species composition occurs (Morante Filho et al. 2015), where forest-dependent species are replaced by non-forest dependent ones, probably functionally distinct from one another, explaining the lower functional redundancy in deforested landscapes. Also, the negative effects of forest cover on functional dispersion means that in forested landscapes the traits of species are near to the centroid of all species in the community (Laliberté and Legendre 2010)), revealing that forested landscapes can buffer functional traits against the extinction.
We showed that functional traits were affected by both landscape composition and configuration. In fact, some studies point to an increased resource availability for frugivorous species, for example, comparing edge with the fragment interior (Saavedra et al. 2014) or increasing habitat amount (Pessoa et al. 2017b). In addition, among the traits related to frugivory, gape width was affected by the interactions between forest cover and edge density and mean nearest neighborhood distance and edge density. Our results showed that increasing edge amount led to a decrease in bill width, but at higher amounts of forest cover this decrease is faster. This result can also be explained by niche filtering, where more forested landscapes retain small species which ultimately has small gape. Yet the increase in edge amount favors plant species that in general are pioneer and have small fruits (Santos et al. 2012), attracting frugivorous with small bill width. For example, small-gaped species such as Chiroxiphia pareola and Manacus manacus can be attracted to the edge searching for small fruits, reducing the mean bill width in more forested landscapes. Conversely, reducing edge amount favors large-gaped frugivores. The increase in mean nearest neighborhood distance interacting with high edge density amount increases bill width (greater bill widths at landscapes with high isolation and edge density), whereas decreasing edge amount decreases this attribute. This finding could reflect that landscapes with high isolation and high edge amount can lead to species spillover into the matrix, favoring species that are able to cross the matrix and that use the fruits located at edges (Boesing et al. 2018). On the other hand, landscapes with high levels of isolation and with low edge amount would favor more generalist species, with smaller gape widths, due to the small fruit quality in deforested landscapes (Pessoa et al. 2017a). However, for hand-wing index and body mass, the lack of significant effects may indicate a compensation between large- and small-bodied species, as previously shown for forest and non-forest dependent frugivorous (Morante-filho et al. 2018b).
Conclusions
This work unveiled the patterns related to the importance and magnitude of the effects of landscape composition (namely forest cover) and configuration for frugivorous birds in the Brazilian Atlantic Forest. Our approach showed that landscape composition is the strongest determinant of taxonomic and functional diversity of frugivorous birds in the Brazilian Atlantic forest. However, landscape composition and configuration interact affecting frugivorous traits related to seed dispersal. We reinforce that conservation initiatives in this biodiversity hotspot should emphasize the maintenance of large habitat amounts at the landscape scale and low edge amount and isolation to hold species and functions performed by frugivorous birds. This group performs the important function of seed dispersal that is essential for regeneration and diversity of tropical plants (Jordano 2014). Here, we showed that habitat loss affects frugivorous birds and can have pervasive consequences for seed dispersal by them, such as a decrease in the number of fruits consumed and dispersal distance (McConkey et al. 2012). Additionally, we showed that forest cover is a strong environmental filter selecting species with redundant functions, which can prevent functional extinction. In addition, the combined effects of landscape composition and configuration affect functional traits of frugivorous birds. This is especially worrying because the Atlantic Forest suffered from intense deforestation creating isolated fragments. Also, more than 40% of remaining forest patches in the Brazilian Atlantic Forest are at a distance of 100 m or less from edges (Haddad et al. 2015), which could synergistically with habitat loss drive a functional debt related to seed dispersal. Thus, the maintenance of high habitat amount, in combination with low levels of fragmentation in the landscape, can favor a higher number of frugivorous birds and maintain the functions performed by them.
References
Andrén H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355–366
Arroyo-Rodríguez V, Rojas C, Saldaña-Vázquez RA, Stoner KE (2016) Landscape composition is more important than landscape configuration for phyllostomid bat assemblages in a fragmented biodiversity hotspot. Biol Conserv 198:84–92
Barlow J, França F, Gardner Hicks CC, Lennox GD, Berenguer E, Castello L, Economo EP, Ferreira J, Guénard B, Gontijo Leal C, Isaac V, Lees AC, Parr CL, Wilson SK, Young PJ, Graham NAJ (2018) The future of hyperdiverse tropical ecosystems. Nature 559:517–526
Barton K (2019) MuMIn: Multi-Model Inference. R package version 1.43.6. https://CRAN.R-project.org/package=MuMIn
Beale CM, Lennon JJ, Yearsley JM, Brewer MJ, Elston DA (2010) Regression analysis of spatial data. Ecol Lett 13:246–264
Bello C, Galetti M, Montan D, Pizo MA, Mariguela TC, Culot L, Bufalo F, Labecca F, Pedrosa F, Constantini R, Emer C, Silva WR, da Silva FR, Ovaskainen O, Jordano P (2017) Atlantic frugivory: a plant–frugivore interaction data set for the Atlantic Forest. Ecology 98:1729
Bibby CJ, Burgess ND, Hill DA (1992) Bird census techniques. Academic Press, New York
Boesing AL, Nichols E, Metzger JP (2018) Biodiversity extinction thresholds are modulated by matrix type. Ecography (cop) 41:1520–1533
Bolker B, R Development Core Team (2020). bbmle: Tools for General Maximum Likelihood Estimation. R package version 1.0.23.1. https://CRAN.R-project.org/package=bbmle
Boscolo D, Metzger JP (2009) Is bird incidence in Atlantic forest fragments influenced by landscape patterns at multiple scales? Landsc Ecol 24:907–918
Bovo AAA, Ferraz KMPMB, Magioli M, Alexandrino ER, Hasui É, Ribeiro MC, Tobias JA (2018) Habitat fragmentation narrows the distribution of avian functional traits associated with seed dispersal in tropical forest. Perspect Ecol Conserv 16:90–96
Bregman TP, Lees AC, MacGregor HEA, Darski B, de Moura NG, Aleixo A, Barlow J, Tobias JA (2016) Using avian functional traits to assess the impact of land-cover change on ecosystem processes linked to resilience in tropical forests. Proc R Soc B Biol Sci 283:20161289
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Carrara E, Arroyo-Rodríguez V, Vega-Rivera JH, Schondube JE, de Freitas SM, Fahrig L (2015) Impact of landscape composition and configuration on forest specialist and generalist bird species in the fragmented Lacandona rainforest, Mexico. Biol Conserv 184:117–126
Chapman PM, Tobias JA, Edwards DP, Davies RG (2018) Contrasting impacts of land-use change on phylogenetic and functional diversity of tropical forest birds. J Appl Ecol 55:1604–1614
Coelho MTP, Raniero M, Silva MI, Hasui É (2016) The effects of landscape structure on functional groups of Atlantic forest birds. Wilson J Ornithol 128:520–534
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, Mcclean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography (cop) 36:27–46
Dunning JB, Danielson BJ, Pulliam HR (1992) Ecological processes that affect populations in complex landscapes. Oikos 65:169–175
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
Fahrig L (2017) Ecological responses to habitat fragmentation per se. Annu Rev Ecol Evol Syst 48:1–23
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40:1649–1663
Fahrig L, Arroyo-Rodríguez V, Bennett JR, Boucher-Lalonde V, Cazetta E, Currie DJ, Eigenbrod F, Ford AT, Harrison SP, Jaeger JAG, Koper N, Martin AE, Martin JL, Metzger JP, Morrison P, Rhodes JR, Saunders DA, Simberloff D, Smith AC, Tischendorf L, Vellend M, Watling JI (2019) Is habitat fragmentation bad for biodiversity? Biol Conserv 230:179–186
Fletcher RJ, Didham RK, Banks-Leite C, Barlow J, Ewers RM, Rosindell J, Holt RD, Gonzalez A, Pardini R, Damschen EI, Melo FPL, Ries L, Prevedello JA, Tscharntke T, Laurance WF, Lovejoy T, Haddad NM (2018) Is habitat fragmentation good for biodiversity? Biol Conserv 226:9–15
Galán-Acedo C, Arroyo-Rodríguez V, Cudney-Valenzuela SJ, Fahrig L (2019) A global assessment of primate responses to landscape structure. Biol Rev 94:1605–1618
Godet L, Devictor V, Burel F, Robin J-G, Ménanteau L, Fournier J (2016) Extreme landscapes decrease taxonomic and functional bird diversity but promote the presence of rare species. Acta Ornithol 51:23–38
Gotelli NJ (2000) Null model analysis of species co-occurrence patterns. Ecology 81:2606–2621
Gotelli NJ, McCabe DJ (2002) Species co-occurrence: a meta-analysis of J. M. Diamond’s assembly rules model. Ecology 83:2091–2096
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song DX, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv 1:1–10
Hasui É, Metzger JP, Pimentel R, Silveira LF, Bovo AAdA, Martensen AC, Uezu A, Regolin AL, Bispo de Oliveira A, Gatto CAFR, Duca C, Andretti CB, Banks-Leite C, Luz D, Mariz D, Alexandrino ER, de Barros FM, Martello F, Pereira IMS, da Silva JN, Ferraz KMPMdB, Naka LN, dos Anjos L, Efe MA, Pizo MA, Pichorim M, Gonçalves MSS, Cordeiro PHC, Dias RA, Muylaert RdL, Rodrigues RC, da Costa TVV, Cavarzere V, Tonetti VR, Silva WR, Jenkins CN, Galetti M, Ribeiro MC (2018) Atlantic birds: a data set of bird species from the Brazilian Atlantic Forest. Ecology 99:497
Hatfield JH, Harrison MLK, Banks-Leite C (2018) Functional diversity metrics: how they are affected by landscape change and how they represent ecosystem functioning in the tropics. Curr Landsc Ecol Rep 3:35–42
Joly CA, Metzger JP, Tabarelli M (2014) Experiences from the Brazilian Atlantic Forest: ecological findings and conservation initiatives. New Phytol 204:459–473
Jordano P (2014) Fruits and frugivory. In: Gallagher RS (ed) Seeds: the ecology of regeneration in plant communities, 3rd edn. CAB International, London, pp 18–61
Jordano P, Schupp EW (2000) Seed disperser effectiveness: the quantity component and patterns of seed rain for Prunus mahaleb. Ecol Monogr 70:591–615
Jung M (2016) LecoS: a python plugin for automated landscape ecology analysis. Ecol Inform 31:18–21
Kissling WD, Böhning-Gaese K, Jetz W (2009) The global distribution of frugivory in birds. Glob Ecol Biogeogr 18:150–162
Klingbeil BT, Willig MR (2009) Guild-specific responses of bats to landscape composition and configuration in fragmented Amazonian rainforest. J Appl Ecol 46:203–213
Kormann UG, Hadley AS, Tscharntke T, Betts MG, Robinson WD, Scherber C (2018) Primary rainforest amount at the landscape scale mitigates bird biodiversity loss and biotic homogenization. J Appl Ecol 55:1288–1298
Kupsch D, Vendras E, Ocampo-Ariza C, Batáry P, Motombi FN, Bobo KS, Waltert M (2019) High critical forest habitat thresholds of native bird communities in Afrotropical agroforestry landscapes. Biol Conserv 230:20–28
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305
Laliberté E, Legendre P, Shipley B (2014) Package FD – measuring functional diversity (FD) from multiple traits, and other tools for functional ecology. Version1.0–12
Laurance WF, Sayer J, Cassman KG (2014) Agricultural expansion and its impacts on tropical nature. Trends Ecol Evol 29:107–116
MacArthur R, MacArthur J (1961) On bird species diversity. Ecology 42:594–598
MapBiomas – Coleção [3.1] da Série Anual de Mapas de Cobertura e Uso de Solo do Brasil. http://www.mapbiomas.org. Accessed 14 June 2019
Martensen AC, Ribeiro MC, Banks-Leite C, Prado PI, Metzger JP (2012) Associations of forest cover, fragment area, and connectivity with neotropical understory bird species richness and abundance. Conserv Biol 26:1100–1111
Mason NWH, De Bello F, Mouillot D, Pavoine S, Dray S (2013) A guide for using functional diversity indices to reveal changes in assembly processes along ecological gradients. J Veg Sci 24:794–806
Mason NWH, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111:112–118
Matuoka MA, Benchimol M, Morante-Filho JC (2020) Tropical forest loss drives divergent patterns in functional diversity of forest and non-forest birds. Biotropica 52:738–748
McConkey KR, Prasad S, Corlett RT, Campos-Arceiz A, Brodie JF, Rogers H, Santamaria L (2012) Seed dispersal in changing landscapes. Biol Conserv 146:1–13
Menezes I, Cazetta E, Morante-Filho JC, Faria D (2016) Forest cover and bird diversity: drivers of fruit consumption in forest interiors in the Atlantic Forest of Southern Bahia, Brazil. Trop Conserv Sci 9:549–562
Merckx T, Dantas de Miranda M, Pereira HM (2019) Habitat amount, not patch size and isolation, drives species richness of macro-moth communities in countryside landscapes. J Biogeogr 46:956–967
Morante-Filho JC, Arroyo-Rodríguez V, de Andrade ER, Santos BA, Cazetta E, Faria D (2018a) Compensatory dynamics maintain bird phylogenetic diversity in fragmented tropical landscapes. J Appl Ecol 55:256–266
Morante-Filho JC, Arroyo-Rodríguez V, Pessoa MS, Cazetta E, Faria D (2018b) Direct and cascading effects of landscape structure on tropical forest and non-forest frugivorous birds. Ecol Appl 28:2024–2032
Morante-Filho JC, Faria D, Mariano-Neto E, Rhodes J (2015) Birds in anthropogenic landscapes: the responses of ecological groups to forest loss in the Brazilian Atlantic forest. PLoS ONE 10:1–18
Moreira-Lima L (2014) Aves da Mata Atlântica: riqueza, composicão, status, endemismos e conservacão. Dissertation, Universidade de São Paulo
Mouchet MA, Villéger S, Mason NWH, Mouillot D (2010) Functional diversity measures: an overview of their redundancy and their ability to discriminate community assembly rules. Funct Ecol 24:867–876
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Newbold T, Hudson LN, Arnell AP, Contu S, De Palma A, Ferrier S, Hill SLL, Hoskins AJ, Lysenko I, Phillips HRP, Burton VJ, Chng CWT, Emerson S, Gao D, Hale GP, Hutton J, Jung M, Sanchez-Ortiz K, Simmons BI, Whitmee S, Zhang H, Scharlemann JPW, Purvis A (2016) Has land use pushed terrestrial biodiversity beyond the planetary boundary? A global assessment. Science 353:288–291
Pardini R, de Bueno AA, Gardner TA, Prado PI, Metzger JP (2010) Beyond the fragmentation threshold hypothesis: regime shifts in biodiversity across fragmented landscapes. PLoS ONE. https://doi.org/10.1371/journal.pone.0013666
Pessoa MS, Hambuckers A, Benchimol M, Rocha-Santos L, Bomfim JA, Faria D, Cazetta E (2017a) Deforestation drives functional diversity and fruit quality changes in a tropical tree assemblage. Perspect Plant Ecol Evol Syst 28:78–86
Pessoa MS, Rocha-Santos L, Talora DC, Faria D, Mariano-Neto E, Hambuckers A, Cazetta E (2017b) Fruit biomass availability along a forest cover gradient. Biotropica 49:45–55
Petchey OL, Gaston KJ (2006) Functional diversity: Back to basics and looking forward. Ecol Lett 9:741–758
Plass-Johnson JG, Taylor MH, Husain AAA, Teichberg MC, Ferse SCA (2016) Non-random variability in functional composition of coral reef fish communities along an environmental gradient. PLoS ONE 11:1–18
Podani J, Schmera D (2006) On dendrogram-based measures of functional diversity. Oikos 115:179–185
Prescott GW, Gilroy JJ, Haugaasen T, Medina Uribe CA, Foster WA, Edwards DP (2016) Reducing the impacts of Neotropical oil palm development on functional diversity. Biol Conserv 197:139–145
Püttker T, Crouzeilles R, Almeida-Gomes M, Schmoeller M, Maurenza D, Alves-Pinto H, Pardini R, Vieira MV, Banks-Leite C, Fonseca CR, Metzger JP, Accacio GM, Alexandrino ER, Barros CS, Bogoni JA, Boscolo D, Brancalion PHS, Bueno AA, Cambui ECB, Canale GR, Cerqueira R, Cesar RG, Colletta GD, Delciellos AC, Dixo M, Estavillo C, Esteves CF, Falcão F, Farah FT, Faria D, Ferraz KMPMB, Ferraz SFB, Ferreira PA, Graipel ME, Grelle CEV, Hernández MIM, Ivanauskas N, Laps RR, Leal IR, Lima MM, Lion MB, Magioli M, Magnago LFS, Mangueira JRAS, Marciano-Jr E, Mariano-Neto E, Marques MCM, Martins SV, Matos MA, Matos FAR, Miachir JI, Morante-Filho JM, Olifiers N, Oliveira-Santos LGR, Paciencia MLB, Paglia AP, Passamani M, Peres CA, Pinto Leite CM, Porto TJ, Querido LCA, Reis LC, Rezende AA, Rigueira DMG, Rocha PLB, Rocha-Santos L, Rodrigues RR, Santos RAS, Santos JS, Silveira MS, Simonelli M, Tabarelli M, Vasconcelos RN, Viana BF, Vieira Emerson M, Prevedello JA (2020) Indirect effects of habitat loss via habitat fragmentation: a cross-taxa analysis of forest-dependent species. Biol Conserv 241:108368
QGIS Development Team (2016) QGIS Geographic Information System. Version 3.4.13. Open Source Geospatial Foundation Project. Retrieved from https://www.qgis.org
R Development Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Rezende CL, Scarano FR, Assad ED, Joly CA, Metzger JP, Strassburg BBN, Tabarelli M, Fonseca GA, Mittermeier RA (2018) From hotspot to hopespot: an opportunity for the Brazilian Atlantic Forest. Perspect Ecol Conserv 16:208–214
Rocha-Santos L, Benchimol M, Mayfield MM, Faria D, Pessoa MS, Talora DC, Mariano-Neto E, Cazetta E (2017) Functional decay in tree community within tropical fragmented landscapes: Effects of landscape-scale forest cover. PLoS ONE 12:1–18
Rodrigues RC, Hasui É, Assis JC, Pena JCC, Muylaert RL, Tonetti VR, Martello F, Regolin AL, Costa TVV, Pichorim M, Carrano E, Lopes LE, Vasconcelos MF, Fontana CS, Roos AL, Gonçalves F, Banks-Leite C, Cavarzere V, Efe MA, Alves MAS, Uezu A, Metzger JP, de Antas PTZ, Ferraz KMPMB, Calsavara LC, Bispo AA, Araujo HFP, Duca C, Piratelli AJ, Naka LN, Dias RA, Gatto CAFR, Vallejos MAV, Menezes GR, Bugoni L, Rajão H, Zocche JJ, Willrich G, Silva ES, Manica LT, Guaraldo AC, Althmann G, Serafini PP, Francisco MR, Lugarini C, Machado CG, Marques-Santos F, Bobato R, Souza EA de, Donatelli RJ, Ferreira CD, Morante-Filho JC, Paes-Macarrão ND, Macarrão A, Lima MR, Jacoboski LI, Candia-Gallardo C, Alegre VB, Jahn AE, Barbosa KVC, Cestari C, Silva JN, Silveira NS, Crestani ACV, Petronetto AP, Bovo AAA, Viana AD, Araujo AC, Santos AH, Amaral ACA, Ferreira A, Vieira-Filho AH, Ribeiro BC, Missagia CCC, Bosenbecker C, Medolago CAB, Espínola CRR, Faxina C, Nunes CEC, Prates C, Luz DTA, Moreno DJ, Mariz D, Faria D, Meyer D, Doná EA, Alexandrino ER, Fischer E, Girardi F, Giese FB, Shibuya FLS, Faria FA, Farias FB, Favaro FL, Freitas FJF, Chaves FG, Las-Casas FMG, Rosa GLM, Torre GMD La, Bochio GM, Bonetti GE, Kohler G, Toledo-Lima GS, Plucenio GP, Menezes Í, Torres IMD, Provinciato ICC, Viana IR, Roper JJ, Persegona JE, Barcik JJ, Martins-Silva J, Just JPG, Tavares-Damasceno JP, Ferreira JR de A, Rosoni JRR, Falcon JET, Schaedler LM, Mathias LB, Deconto LR, Rodrigues LC, Meyer MAP, Repenning M, Melo MA, Carvalho MAS, Rodrigues M, Nunes MFC, Ogrzewalska MH, Gonçalves ML, Vecchi MB, Bettio M, Baptista MNM, Arantes MS, Ruiz NL, Andrade PGB, Ribeiro PHL, Junior PMG, Macario P, Fratoni RO, Meurer R, Saint-Clair RS, Romagna RS, Lacerda RCA, Cerboncini RAS, Lyra RB, Lau R, Rodrigues RC, Faria RR, Laps RR, Althoff SL, de Jesus S, Namba S, Braga TV, Molin T, Câmara TPF, Enedino TR, Wischhoff U, de Oliveira VC, Leandro-Silva V, Araújo-Lima V, Lunardi VO, Gusmão RF, Correia JMS, Gaspar LP, Fonseca RCB, Neto PAFP, Aquino ACMM, Camargo BB, Cezila BA, Costa LM, Paolino RM, Kanda CZ, Monteiro ECS, Oshima JEF, Alves-Eigenheer M, Pizo MA, Silveira LF, Galetti M, Ribeiro MC (2019) Atlantic bird traits: a data set of bird morphological traits from the Atlantic forests of South America. Ecology 100:1–2
Saavedra F, Hensen I, Beck SG, Böhning-Gaese K, Lippok D, Töpfer T, Schleuning M (2014) Functional importance of avian seed dispersers changes in response to human-induced forest edges in tropical seed-dispersal networks. Oecologia 176:837–848
Santos GGA, Santos BA, Nascimento HEM, Tabarelli M (2012) Contrasting demographic structure of short- and long-lived pioneer tree species on Amazonian Forest edges. Biotropica 44:771–778
Sekercioglu CH, Loarie SR, Brenes FO (2007) Persistence of forest birds in the Costa Rican agricultural countryside. Conserv Bio 21:482–494
Villard MA, Metzger JP (2014) Beyond the fragmentation debate: a conceptual model to predict when habitat configuration really matters. J Appl Ecol 51:309–318
Vollstädt MGR, Ferger SW, Hemp A, Howell KM, Töpfer T, Böhning-Gaese K, Schleuning M (2017) Direct and indirect effects of climate, human disturbance and plant traits on avian functional diversity. Glob Ecol Biogeogr 26:963–972
Weiss KCB, Ray CA (2019) Unifying functional trait approaches to understand the assemblage of ecological communities: synthesizing taxonomic divides. Ecography 42:1–9
Wenny DG, DeVault TL, Johnson MD, Kelly D, Sekercioglu CH, Tomback DF, Whelan CJ (2011) The need to quantify ecosystem services provided by birds. Auk 128:1–14
Wheelwright NT (1985) Fruit-size, gape width, and the diets of fruit-eating birds. Ecology 66:808–818
Wilman H, Belmaker J, Simpson J, de La Rosa C, Rivadeneira MM, Jetz W (2014) Elton Traits 1.0: Species-level foraging attributes of the world’s birds and mammals. Ecology 95:2027
Acknowledgements
We are thankful for Anderson Saldanha Bueno, Érica Hassui and Luis dos Anjos for helpful comments on an earlier version of this manuscript. We also thank Elaine Rios, Amanda Cerqueira and Maria Sales for helpful comments and ideas on the earliest version of the manuscript. We also are grateful to the editor and two anonymous reviewers for invaluable comments to improve the manuscript. This study was financed in part by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bonfim, F.C.G., Dodonov, P. & Cazetta, E. Landscape composition is the major driver of the taxonomic and functional diversity of tropical frugivorous birds. Landscape Ecol 36, 2535–2547 (2021). https://doi.org/10.1007/s10980-021-01266-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-021-01266-y