Abstract
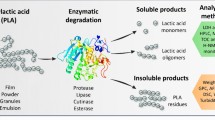
Poly(lactic acid) (PLA) is one of the most important polyester bioplastics, produced from agricultural renewable resources and due to its excellent properties already has found applications in several industrial sectors, including packaging. Its amount, relative to PET, in the waste stream is continuously increasing. Although waste bioplastics are biodegradable, the process sometimes needs long degradation times. Therefore, sufficient recycling techniques should be developed in terms of sustainable chemistry. Hydrolysis of PLA under microwave irradiation in an alkaline solution was investigated aiming in the chemical recycling of this biodegradable polymer and the recovery of the monomer, lactic acid. Several process parameters were examined, including the presence or not of an alkali solution, its amount and concentration, the presence or not of methanol in the reaction medium, together with reaction temperature and time. Moreover, several phase transfer catalysts were employed at various relative amounts to PLA. Reaction temperature, pressure and microwave power did not change significantly during degradation. Optimum experimental conditions, leading to PLA degradation more than 90%, were achieved using hexadecyltrimethylammonium bromide as phase transfer catalyst in a 10% w/v NaOH medium at 100 °C for 10 min irradiation time. Using such low temperature and degradation times results in great environmental benefit since it does not consume significant amount of energy compared to other similar techniques proposed in literature and thus leads to the sustainability of the process. Therefore, it seems to be a very efficient method to be used in the recycling of large amounts of this polymer.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nowadays the large amount of plastics coming from packaging, waste electric and electronic equipment or automobile industry, and ending in the final recipients, together with their low biodegradability has created a serious environmental problem. Therefore, the trend to replace in large scale production of consumables, the traditional petrochemical-based polymers with biodegradables having similar properties, has gained considerable interest over the past few years [1]. Poly(lactic acid) (PLA) is one of the most important polyester bioplastics, produced from agricultural renewable resources and already found applications in several industrial sectors, including packaging [1]. Besides its biocompatibility, due to its good mechanical properties it will soon replace several commodity polymers in several plastics applications [1, 2]. Already the amount of PLA relative to PET in the waste stream is estimated at 7% and is continuously increasing [2]. Although the waste bioplastics including PLA are biodegradable and the degradation products usually are non toxic, the process sometimes needs long degradation times. Therefore, sufficient recycling techniques should be developed in terms of sustainable chemistry [3].
At present, mechanical recycling of PLA, as in most other polymers, is one of the most widespread methods due to its easy implementation [4]. However, it results in the deterioration of polymer’s physicochemical and mechanical properties [5]. Chemical recycling, as an alternative method, has drawn much attention, recently since secondary value-added materials can be produced [6−7] including in the case of PLA, the monomer lactic acid. Thus, chain scission to functional macromolecules has been proposed [8−9], thermal degradation to lactides [10] and hydrolysis to lactic acid [11−14]. The later seems to be a promising technique and several alternative experimental conditions have been explored. Piemonte and Gironi [11] proposed hydrolysis at 160 to 180 °C for 120 min to result in nearly 95% degradation of PLA to lactic acid. Iniguez-Franco et al. [13] performed experiments in much lower temperature; though the required degradation time at 90 °C was as much as 20 h. Complete degradation of PLA was achieved by Annesini et al. [15] at low reaction times, 35 to 90 min, though using increased reaction temperatures at 170 to 200 °C. For the degradation of water soluble PLA oligomers, a temperature ranging from 40 to 120 °C was employed by Codari et al. [14] at times ranging from 3 to 50 h. The hydrolytic degradation of low molecular weight PLA was also investigated by Proikakis et al. [16]. The group of Fusheng Liu has also published a number of papers on the alcoholysis of PLA to obtain lactate esters or hydrolysis to calcium lactate using halide or ionic liquids as catalysts [17,18,19,−20].
Most of previous studies on PLA degradation through hydrolysis usually require large quantities of energy by using either high temperatures or prolonged degradation times. Considering also that some of the catalysts used may be corrosive, there is a great need to find an environmentally friendly solution for the chemical recycling of this important polymer. To this direction, following the approach applied from our group in the chemical recycling of other polyesters, such as PET or PC [6, 21,22,23], hydrolysis of PLA in an alkaline solution was employed using an appropriate phase transfer catalyst. Furthermore, the main innovative point of this research was to conduct the reaction under microwave irradiation. This way of providing energy to a reacting system has been shown to result in much lower degradation times or temperatures in several other polymer types [21]. Thus, the monomer lactic acid can be produced from the degradation of PLA using low amounts of energy either in the form of low degradation temperature or short time. Therefore, this method shows great potential for the chemical recycling of PLA in large amounts showing both economic and environmental benefits.
Experimental Section
Materials
The PLA used was the Igneo Biopolymer 3052D (from NatureWorks) with MFR 14 g/10 min at 210 °C and 2.16 kg. NaOH was purchased from Penta (purity > 98%). The phase transfer catalysts used were, (2-dimethylaminoethyl) triphenylphosphonium bromide ((2DME)-TPPB) (Lancaster), hexadecyltrimethylammonium bromide (HTMAB) obtained from Fluka Chemicals and aqueous solution of Hexadecyltrimethylammonium chloride (HTMAC), 25 wt%, from Aldrich. The chemical structure of these catalysts is shown in Scheme 1. All other chemicals used were reagent grade.
Hydrolytic Depolymerization
The PLA decomposition reaction was conducted in a microwave reactor (model Discover from CEM corporation), equipped with a digital temperature control system and pressure sensors inserted directly into the 10 mL polytetrafluoroethylene (PTFE) reaction tube. Pellets of sodium hydroxide (10 g) were dissolved in 100 mL of distilled water and the resultant NaOH solution (2.5 M, 10% w/v) was used for the experiments. Several other concentrations were also used. PLA flakes of almost 2 mm (0.5 g) together with 4 mL of NaOH solution were added to the reactor together with a magnetic agitator. The tubes were filed with nitrogen to retain inert atmosphere, sealed and the heat-up period to the desired set-point started. In some experiments 0.05 g of HTMAB (1:10 w/w PLA) or other phase transfer catalyst (PTC) were also fed into the reactor. When the set temperature was achieved the reaction time begun and PLA decomposition was followed for a specified time period.
After that time period, the reaction vessels were automatically cooled and the reaction mixture was filtered to remove the unreacted PLA residues. The mixture was washed with distilled water followed by washing with methanol to remove excess of NaOH. The final unreacted PLA was measured upon filtration of the final mixture through a G3 glass filter, washing with water, drying in a vacuum oven at 40 °C and weighing. The filtrate, which includes the degradation products (monomer and oligomers), excess of NaOH and the PTC was cooled at 4 °C for some days in a sealed tube, where a solid powder was formed and precipitated. The solid was filtered and washed with cold distilled water and then dried in a vacuum oven at 40 °C and weighed. No further acidification or other treatment was conducted.
The percent PLA degradation was estimated form the following equation
where WPLA,0 and WPLA,f refer to the initial and final weight of PLA, respectively.
Measurements
The chemical structure of the separated PLA and of the oligomers (possible lactic acid) was confirmed by recording its IR spectra. The instrument used was an FT-IR spectrophotometer from Perkin-Elmer, Spectrum One. The technique used was that of Attenuated Total Reflectance (ATR-FTIR) since solid materials were used. The material in the reflectance plate was ΖnSe and the angle was set at 45ο. The resolution of the instrument was 4 cm−1. The recorded wavenumber range was from 700 to 4000 cm−1 and 32 spectra were averaged to reduce the noise. A commercial software Spectrum v5.3.1 (Perkin Elmer LLC 1500F2429) was used to process and calculate all the data from the spectra.
Results and Discussion
Reaction Medium and Catalyst Screening
Initially, some screening experiments were performed in order to select the optimum reaction medium and catalyst system. Thus, initially the reaction medium was pure distilled water without any added catalyst and the reaction temperature and time were kept constant at 150 °C and 15 min, respectively. Results appear in Table 1. The amount of PLA depolymerized was very low, nearly 1%. Therefore, a catalyst usually used in such experiments was employed, i.e. Zinc acetate. However, the depolymerization degree was still low. After the addition of the phase transfer catalyst HTMAB the degree of depolymerization slightly increased, but the amount measured was again lower than 2%.
Furthermore, in order to improve depolymerization degrees the reaction medium was changed to either alcoholic or alkaline. In the first case methanol was used in the presence of HTMAB, whereas in the second NaOH solution was employed at two concentrations 5 and 10%. Results appear in Table 2. From the results illustrated in Table 2 it was clear that use of methanol results in a rather low PLA depolymerization degree (almost 12%), whereas the alkaline solution was proved to be a better medium compared to methanol. Therefore, a NaOH solution was further used in all experiments. Moreover, compared to Zinc acetate, it seems that the phase transfer catalyst, HTMAB, results in much higher comparative depolymerization degrees. Hence, (CH3COO)2Zn was not used further.
The type of the phase transfer catalyst was further investigated and results appear in Table 3 for three different compounds, namely (2-dimethylaminoethyl) triphenylphosphonium bromide, (2DME)-TPPB, Hexadecyltrimethyl ammonium bromide (HTMAB) and Hexadecyltrimethylammonium chloride (HTMAC).
Initially, it was clear that changing the anion from bromide to chloride did not result into any significant change in the depolymerization degree. In this investigation we preferred to use HTMAB instead of HTMAC since this catalyst was also used in the depolymerization of PET under similar experimental conditions [6] and therefore we could have compare results. Furthermore, the phase transfer catalyst used, HTMAB, fulfilled the requirements of having enough organic character in order to be lipophilic, while small enough in order to avoid steric hindrance [6]. In contrast, it seems that (2DME)-TPPB has also a lipophilic character but the bulky groups lead to steric hindrance phenomena resulting in slower reaction rates compared to HTMAB. Therefore, additional experiments were carried out using always HTMAB as a PTC.
From the above analysis it was clear that better results are obtained when using an alkaline solution with the addition of a phase transfer catalyst and particularly HTMAB. Specific additional reaction parameters were further investigated and appear in next section.
Effect of Alkaline Solution Concentration and Amount
The effect of alkaline concentration on the amount of PLA degradation is illustrated in Fig. 1a. It can be seen that PLA degradation presents a maximum at 10% w/v reaching almost 90% at 100 °C and 10 min of degradation time. Degradation significantly increases from 5 to 10% w/v, whereas afterwards it decreases to 63% at 20% w/v. Probably, the increased alkalinity of the solution does not permit the dissolution of the degradation products and thus it hinders depolymerization of PLA. Moreover, increased alkaline solution results in a more expensive investment. Therefore, it seems that a 10% alkaline solution is the best solution. In addition, the alkaline concentration was kept constant at 10% w/v ant the relative amount of alkali to PLA was changed in order to study its effect on the degradation rate. It seems that the amount of alkali significantly affects the degradation rate starting from very low values when no alkali is added and reaching a plateau after the addition of 0.8 g NaOH/g PLA (Fig. 1b). Therefore this relative amount was considered to be the optimum.
Effect of the Amount of the Phase Transfer Catalyst
Increasing the amount of HTMAB results in increased degradation values until approximately 10% whereas afterwards a plateau is achieved (Fig. 2). When no catalyst is added the degradation rate is very low. Thus the role of the phase transfer catalyst is confirmed. The cationic part of the PTC carries the hydroxide ion into the surface of the solid-organic phase resulting in an easier attack of the macromolecular chains leading to depolymerization. The anion returns to the aqueous phase and forms the monomer lactic acid or sodium lactate. The reaction proceeds to complete depolymerisation of PLA, while the catalyst remains in the aqueous phase. Therefore an amount of 10% (i.e. 1/10 HTMAB/PLA) was considered adequate.
Effect of Temperature
The effect of temperature on the degradation degree of PLA is shown in Fig. 3. Degradation is low at low temperatures and increases to nearly 90% at 100 °C. Afterwards the increase is milder and reaches to 95% after the increase of temperature by 50 °C. Therefore the optimum temperature is considered to be that of 100 °C.
Effect of Reaction Time
Finally, the depolymerization of PLA as a function of degradation time was measured at two temperatures (i.e. 100 and 150 °C) and results are illustrated in Fig. 4. It can be seen that degradation starts from a high value, nearly 83% even after 2 min of degradation at 100 °C and 87% after 5 min and 150 °C. These values increase to 92 and 99% after 20 min of degradation at 100 and 150 °C, respectively. The high degradation values measured at very small times is a direct consequence of the mode of providing energy, i.e. using microwave irradiation. It should be note here that similar trends were observed in previous experiments from our lab using the same microwave reactor for the degradation of PET using the same technique of alkaline hydrolysis using a phase transfer catalyst [6]. Again it was observed there that at the same temperature of 150 °C degradation of PET was nearly 60% after 10 min of irradiation and required another 50 min to increase to 80%. Since microwave irradiation provides energy directly to the molecules of the reacting species (the alkaline solution in this case) and not to the surrounding vessel or the environment, degradation is very high from the beginning of the reaction, whereas afterwards it increases at a much slower rate.

Data taken from reference [6]
Variation of the degree of PLA degradation with time during alkaline hydrolysis of PLA with NaOH at concentration 10 wt% using HTMAB as a phase transfer catalyst at 0.1 g/g of PLA, under microwave irradiation at two temperatures. Similar degradation data from the microwave assisted alkaline hydrolysis of PET at 120 and 150 °C.
Process Parameters
Indicative results on the variation of reaction temperature, pressure and microwave power with depolymerization time for the experiment carried out under constant temperature at 100 °C, for 20 or 30 min are illustrative in Fig. 5. It can be seen that the reaction temperature set point was achieved and remained constant soon after some initial small-scale oscillations. The time needed to achieve the constant value, decreased with temperature and at 100 °C it was less than 2 min. In order to maintain the reaction mixture at the desired temperature the microwave power undergoes some small-scale oscillations varying in between 2 and 10 W. These values were rather low since the required temperature was also relatively low. The system total pressure increased from 30 psi (almost 2 bar) to a constant value near 45 psi (3 bar) or 50 psi (3.4 bar).
Characterization of Unreacted PLA and the Product Obtained
The IR spectra of the unreacted PLA was recorded and compared to the initial PLA in Fig. 6. Typical peaks were observed with representative those at 1596 cm−1 due to the carbonyl stretching of the ester bond (C=O) and at 2920 cm−1 due to the sp3 bond of carbon with hydrogen (C–H). IR spectra of unreacted PLA are similar to those of fresh PLA, but they present two differentiation at nearly 1200 and 3500 cm−1. The peak at 3500 cm−1 is more pronounced in the polymer obtained after degradation at 50 °C. These peaks are attributed to hydroxyl groups and they are an indication that oligomers are present in the residue. PLA macromolecules have only a few terminal hydroxyl groups and thus a very small peak appears in this region for fresh PLA. As degradation starts at relatively low temperatures (i.e. 50 °C) macromolecular chains are cleaved into oligomers having shorter chain length. Thus, the number of hydroxyl terminal groups increases, since the total number of chains is increased. As degradation proceeds further at relatively higher temperatures (i.e. 100 °C), some of the oligomeric chains that have been formed are ultimately degrade to the monomer lactic acid which was removed during the purification step. Thus, fewer chains with terminal hydroxyl groups are present resulting in less intense band attributed to hydroxyl groups in the IR spectrum. This observation verifies that less degradation occurs at the lower temperatures.
The product obtained after depolymerization was characterized by using FT-IR, as given in Fig. 7. In this spectrum the following absorption peaks were identified: A broad peak ranging from 3100 to 3600 cm−1 and peaking at 3414 cm−1, which denotes the existence of hydroxyl groups from the alcohol present in the molecule; a small but wide peak at 2990 cm−1 is characteristic of –OH absorption but from a carboxylic group; the strong and narrow peak at 1726 cm−1 is characteristic of the existence of a COO stretching band, present in carboxyl units (i.e. COO–H), whereas that in 1596 cm−1 denotes the existence of carbonyl group C=O, present in ester (lactate) units (i.e. COO–Na); the peak at 1281 cm−1 shows the existence of a C–O bond present in a carboxylic acid; the peak at 1584 cm−1 refers to the C–H bond, whereas those at 1448 and 1141 cm−1 to the C–C bond. Therefore, it can be concluded that the product obtained from the depolymerization contains mainly the monomer lactic acid but also some sodium lactate. In order to quantify the amount of lactic acid produced extra measurements (such as NMR) are needed. Thus it seems that in order to get pure lactic acid for the depolymerization, extra acidification of the product obtained with some strong acid (e.g. H2SO4) is needed.
.
Possible Reaction Mechanism
A possible reaction mechanism for the alkaline hydrolysis of PLA is shown in Scheme 2. The PLA pellets were initially swelled or dissolved in the reaction medium. Hydrolysis of PLA in the presence of NaOH results in the breaking down of the ester linkages between the constituent units of PLA to yield two kinds of oligomers. These oligomers form salts with sodium cations, Na+ present in the reaction medium. The oligomers further reacted with water and give new oligomers with carboxyl ending units. The above step was repeated until lactic acid or sodium lactate was formed and the catalyst (alkylammonium salt) is regenerated.
Conclusions
Alkaline hydrolysis of PLA was investigated here using a phase transfer catalyst under microwave irradiation. Several process parameters were examined including the alkaline (NaOH) concentration, the presence or not of methanol in the reaction medium, the type and the amount of the phase transfer catalyst, reaction temperature and time. All degradation reactions carried out in a lab scale microwave reactor. The optimum experimental conditions, to obtain high PLA degradation, i.e. greater than 90%, were found to be at 10% w/v NaOH concentration using hexadecyltrimethylammonium bromide as phase transfer catalyst and degradation taking place at 100 °C for 10 min. The microwave power needed to reach these conditions was varying in between 2 and 10 W. A possible mechanism of PLA alkaline hydrolysis, using the specific reagents, was also proposed. The main innovative part of this research was to present a specific method for the degradation of PLA using low temperature and time resulting thus in a great energy saving compared to other similar techniques proposed in literature. Thus, this process seems to be very efficient to be scaled up in the recycling of large amounts of this polymer. Detailed investigation of the degradation kinetics of PLA using this specific system is going to be our next task.
References
Bastioli C (2005) Handbook of Biodegradable Polymers. Rapra Technology Limited, Mumbai
Alaerts L, Augustinus M, Van Acker K (2018) Impact of bio-based plastics on current recycling of plastics. Sustainability 10:1487
Nampoothiri KM, Nair NR, John RP (2010) An overview of the recent developments in polylactide (PLA) research. Bioresour Technol 101:8493–8501
Beltrán FR, Infante C, Orden MU, Urreaga JM (2019) Mechanical recycling of poly(lactic acid): evaluation of a chain extender and a peroxide as additives for upgrading the recycled plastic. J Clean Prod 219:46–56
Hamad K, Kaseem M, Deri F (2013) Recycling of waste from polymer materials: an overview of the recent works. Polym Degrad Stabil 98:2801–2812
Siddiqui MN, Achilias DS, Redhwi HH, Bikiaris DN, Katsogiannis KAG, Karayannidis GP (2010) Hydrolytic depolymerization of PET in a microwave reactor. Macromol Mater Eng 295:575–584
Siddiqui MN, Redhwi HH, Achilias DS (2012) Recycling of poly(ethylene terephthalate) waste through methanolic pyrolysis in a microwave reactor. J Anal Appl Pyrol 98:214–220
Plichta A, Lisowska P, Kundys A, Zychewicz A, Dębowski M, Florjańczyk Z (2014) Chemical recycling of poly(lactic acid) via controlled degradation with protic (macro)molecules. Polym Degrad Stabil 108:288–296
Nederberg F, Connor EF, Glausser T, Hedrick JL (2001) Organocatalytic chain scission of poly(lactides): a general route to controlled molecular weight, functionality and macromolecular architecture. Chem Commun 20:2066–2067
Fan Y, Nishida H, Mori T, Shirai Y, Endo T (2004) Thermal degradation of poly(L-lactide): effect of alkali earth metal oxides for selective L,L-lactide formation. Polymer 45:1197–1205
Piemonte V, Gironi F (2013) Lactic acid production by hydrolysis of poly(lactic acid) in aqueous solutions: an experimental and kinetic study. J Polym Environ 21:275–279
Tsuji H, Daimon H, Fujie K (2003) A new strategy for recycling and preparation of poly(L-lactic acid): hydrolysis in the melt. Biomacromolcules 4:835–840
Iñiguez-Franco F, Auras R, Dolan K, Selke S, Holmes D, Rubino M, Soto-Valdez H (2018) Chemical recycling of poly(lactic acid) by water-ethanol solutions. Polym Degrad Stabil 149:28–38
Codari F, Lazzari S, Soos M, Storti G, Morbidelli M, Moscatelli D (2012) Kinetics of the hydrolytic degradation of poly(lactic acid). Polym Degrad Stabil 97:2460–2466
Annesini MC, Frattari S, Gironi F, Piemonte V, Sabia R, Villani C (2018) Degradation of post-consumer PLA: hydrolysis of polymeric matrix and oligomers stabilization in aqueous phase. J Polym Environ 26:4396–4404
Proikakis CS, Mamouzelos NJ, Tarantili P, Andreopoulos AG (2006) Swelling and hydrolytic degradation of poly(d, l-lactic acid) in aqueous solutions. Polym Degrad Stabil 91(3):614–619
Liu HQ, Song XY, Liu FS, Liu SW, Yu ST (2015) Ferric chloride as an efficient and reusable catalyst for methanolysis of poly(lactic acid) waste. J Polym Res 22:135–142
Song XY, Zhang XJ, Wang H, Liu FS, Yu ST, Liu SW (2013) Methanolysis of poly(lactic acid) (PLA) catalyzed by ionic liquids. Polym Degrad Stabil 98:2760–2764
Liu HQ, Zhao RY, Song XY, Liu FS, Yu ST, Liu SW, Ge XP (2017) Lewis acidic ionic liquid [Bmim]FeCl4 as a high efficient catalyst for methanolysis of poly (lactic acid). Catal Lett 147:2298–2305
Song XY, Wang H, Yang X, Liu FS, Yu ST, Liu SW (2014) Hydrolysis of poly (lactic acid) into calcium lactate using ionic liquid [Bmim]OAc for chemical recycling. Polym Degrad Stabil 110:65–70
Achilias DS (2016) Polymer Degradation under Microwave Irradiation. Adv Polym Sci 274:309–346
Tsintzou GP, Antonakou EV, Achilias DS (2012) Environmentally friendly chemical recycling of poly(bisphenol-A carbonate) through phase transfer-catalysed alkaline hydrolysis under microwave irradiation. J Hazard Mater 241–242:137–145
Kosmidis VA, Achilias DS, Karayannidis GP (2001) Poly(ethylene terephthalate) recycling and recovery of pure terephthalic acid. Kinetics of a phase transfer catalyzed alkaline hydrolysis. Macromol Mater Eng 286:640–647
Acknowledgements
The authors would like to acknowledge the support provided by the Deanship of Scientific Research (DSR) at King Fahd University of Petroleum & Minerals (KFUPM), Dhahran, Saudi Arabia, for funding this work through Project Number IN161007. Laboratory of Polymer Chemistry and Technology, Department of Chemistry, Aristotle University of Thessaloniki, Greece is sincerely appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Siddiqui, M.N., Kolokotsiou, L., Vouvoudi, E. et al. Depolymerization of PLA by Phase Transfer Catalysed Alkaline Hydrolysis in a Microwave Reactor. J Polym Environ 28, 1664–1672 (2020). https://doi.org/10.1007/s10924-020-01716-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-020-01716-9