Abstract
We tested differences in female pheromone production and male response in three species of the genus Adoxophyes in Korea. Females of all three species produced mixtures of (Z)-9-tetradecenyl acetate (Z9–14:OAc) and (Z)-11-tetradecenyl acetate (Z11–14:OAc) as major components but in quite different ratios. The ratio of Z9–14:OAc and Z11–14:OAc in pheromone gland extracts was estimated to be ca. 100:200 for Adoxophyes honmai, 100:25 for Adoxophyes orana, and 100:4,000 for Adoxophyes sp. Field tests showed that males of each species were preferentially attracted to the two-component blends of Z9–14:OAc and Z11–14:OAc mimicking the blends found in pheromone gland extracts of conspecific females. The effects of minor components identified in gland extracts on trap catches varied with species. Addition of 10-methyldodecyl acetate (10me-12:OAc) or (E)-11-tetradecenyl acetate (E11–14:OAc) to the binary blend of Z9–14:OAc and Z11–14:OAc significantly increased captures of A. honmai males, whereas E11–14:OAc exhibited a strongly antagonistic effect on catches of Adoxophyes sp. males. Moreover, (Z)-9-tetradecen-1-ol (Z9–14:OH) or (Z)-11-tetradecen-1-ol (Z11–14:OH) added to the binary blends increased attraction of male A. orana but not A. honmai and Adoxophyes sp. males, suggesting that these minor components, in addition to the relative ratios of the two major components, play an important role in reproductive isolation between Adoxophyes species in the southern and midwestern Korea where these species occur sympatrically.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several species in the genus Adoxophyes (Lepidoptera: Tortricidae) are economically important pests of fruit and tea trees in Asia and Europe (Tamaki et al. 1971a; Meijer et al. 1971; Kou et al. 1990). In the past, it was believed that Adoxophyes orana is the only species found in Korea (Shin et al. 1994), but field trapping revealed that there is considerable variability in male response to pheromone blends among different geographical populations of Adoxophyes spp. (Han 2002; Yang et al. 2005). These findings prompted us to study the taxonomic status of Korean Adoxophyes populations by using mitochondrial gene sequences, and we found three distinct species, Adoxophyes honmai, A. orana, and Adoxophyes sp. (Park et al. 2008). Larvae are difficult to differentiate, but adults of the different species are distinguishable by wing patterns and male genitalia (Han 2002; Byun, personal communication).
Adoxophyes species often share host plants (Han 2002; Yang et al. 2005) and have some seasonal overlap in Korea (Choi 2002; Yang 2002). Furthermore, the diel periodicity of pheromonal emission also overlaps, with females of all three species calling at the end of the scotophase and beginning of the photophase (Sato and Tamaki 1977; Den Otter and Klijnstra 1983; Han 2002). Therefore, differences in these parameters appear insufficient to maintain reproductive isolation among these species. Males of Adoxophyes species, however, are rarely attracted to heterospecific females in fields (Yang, unpublished), suggesting that species-specific blends of pheromone components may be responsible for reproductive isolation between sympatric species (Roelofs and Brown 1982; Löfstedt et al. 1991). Thus, the principal objectives of this study were to identify the sex pheromones of three Adoxophyes species and to determine the role of the pheromones as prezygotic reproductive isolating mechanisms between sympatric congeners in Korea.
Methods and Materials
Insects
Larvae of Adoxophyes spp. were collected from pear orchards in Naju, Cheonan, and Namyangju, Korea during June and July of 2006. The larvae were reared individually on artificial diet (Bio-serv, Frenchtown, NJ, USA) in transparent plastic Petri dishes (1.5 cm high and 5.5 cm diameter) and maintained at 23°C under a 16:8-h ratio of light to dark photoperiod. Pupae were separated by sex and kept individually in plastic bottles (7 cm high and 2.5 cm diameter). After eclosion, moths were identified based on forewing color patterns (Han 2002) and then provided with a cotton pad soaked with a 5% (w/v) sucrose solution as food.
Chemicals
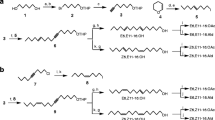
Synthetic dodecenyl acetate (12:OAc), tridecyl acetate (13:OAc), (Z)-9-dodecenyl acetate (Z9–12:OAc), 10-methyldodecyl acetate (10me-12:OAc), tetradecyl acetate (14:OAc), Z9–14:OAc, E11–14:OAc, Z11–14:OAc, (Z)-11-hexadecenyl acetate (Z11–16:OAc), (Z)-9-tetradecen-1-ol (Z9–14:OH), (Z)-11-tetradecen-1-ol (Z11–14:OH), and octadecanal (18:Ald) were purchased from Pherobank (Wageningen, The Netherlands). Isomeric purity of these compounds exceeded 99%.
Gland Extraction
Pheromone gland extracts were taken from 2- to 3-day-old females at the end of the scotophase, during their calling periods (Den Otter and Klijnstra 1983; Han 2002). The pheromone gland of each female was extruded by applying gentle pressure to the abdomen, excised with fine forceps, and individually extracted in 10 μl hexane containing 10 ng of 13:OAc as an internal standard in a 0.3-ml conical vial (Wheaton, Millville, NJ, USA) for 30 min at room temperature. The supernatant was transferred into another vial and stored at −80°C until analysis.
Chemical Analysis
Pheromone gland extracts were analyzed on an Agilent 6890N GC equipped with a split/splitless injector and a flame ionization detector. Samples were run on DB-5 and DB-Wax columns (30 m × 0.25 mm ID, 0.25 μm film thickness, J&W Scientific, Folsom, CA, USA) in the splitless mode. Injector and detector temperature were 250°C. Helium was used as carrier gas (1 ml/min). The gas chromatograph (GC) oven temperature was programmed from 80°C (1 min hold) to 220°C at 5°C/min and held for 10 min. Components in pheromone gland extracts were identified by comparison of retention times with those of authentic standards on two different columns. The quantity of each component was estimated by comparing its GC peak area with that of the internal standard.
Gas chromatography–mass spectrometry (MS) analyses of the crude extract of pheromone glands were performed on an Agilent 6890N GC interfaced to an Agilent 5973 mass-selective detector. Samples were analyzed on the DB-Wax column (30 m × 0.25 mm ID, 0.25 μm film thickness) with the temperature program described above. The ionization voltage was 70 eV. The ion source temperature was 230°C. Components in gland extracts were tentatively identified by comparison of their mass spectra with the mass spectra library (Wiley-NIST, Hoboken, NJ, USA), and identifications were confirmed by comparison of retention times and mass spectra with those of authentic standards.
Field Experiments
Field experiments were conducted in pear orchards at Naju, Cheonan, and Namyangju, respectively, during May–July of 2007. Sticky Delta traps (Green Agro Tech, Korea) baited with rubber septa (Aldrich Chemical Co., Milwaukee, WI, USA) impregnated with test chemicals in hexane were hung on branches 1–1.5 m above ground level. All field tests employed a complete randomized block design with five replicate blocks. The distance between traps within a block was at least 15 m. Twice a week, moths were counted, removed, and identified to species by using wing venation and then verified by examination of the genitalia.
Experiment 1 investigated the attraction of A. honmai, A. orana, and Adoxophyes sp. males to the two major components Z9–14:OAc and Z11–14:OAc singly and in binary blends. Experiment 2 was conducted to test the individual effects of the nine minor components identified in female gland extracts; 12:OAc, Z9–12:OAc, 10me-12:OAc, 14:OAc, E11–14:OAc, Z9–14:OH, Z11–14:OH, Z11–16:OAc, and 18:Ald as possible synergists or antagonists, by using standard baits of the two major components. The standard baits used for A. honmai, A. orana, and Adoxophyes sp. were 1 mg of 35:65, 80:20, and 3:97 mixtures of Z9–14:OAc and Z11–14:OAc, respectively. Trap catch data (x) were transformed to log (x + 1) and submitted to one-way analysis of variance. Means were compared by Tukey’s test at α = 0.05 (SAS Institute Inc. 2004).
Distribution of Adoxophyes Species
We sampled larvae of Adoxophyes spp. from pear orchards at eight different sites in Korea during June–August of 2007 and 2008 and reared them on artificial diet. After eclosion, female moths were assigned to three species according to their wing characters and subsequently verified by examination of the sex pheromones. Individuals were classified as A. honmai, A. orana, and Adoxophyes sp. when Z9–14:OAc and Z11–14:OAc in pheromone gland extracts were present in a ca. 100:200, 100:25, and 100:4,000 ratios, respectively. In all cases, the pheromone blend of individual glands supported the initial species identification based on adult morphological characters.
Results
Chemical Analysis
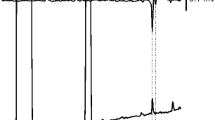
GC and GC–MS analyses of pheromone gland extracts showed that Z11–14:OAc is the major component of A. honmai and Adoxophyes sp., whereas Z9–14:OAc is the most abundant component in the pheromone blend of A. orana (Fig. 1). The ratios of Z9–14:OAc and Z11–14:OAc in pheromone blends were 100:209, 100:25, and 100:3,810 for A. honmai, A. orana, and Adoxophyes sp., respectively. In addition to these two major components, three acetate esters (12:OAc, 14:OAc, and E11–14:OAc), Z11–14:OH, and 18:Ald, as well as nonadecane, heneicosane, and tricosane, were detected in gland extracts of all species (Table 1). Small amounts of Z9–14:OH and Z11–16:OAc were detected in A. honmai and A. orana, while Z9–12:OAc and 10me-12:OAc were present only in A. honmai.
Comparison of gas chromatograms of pheromone gland extracts of Adoxophyes honmai (A), A. orana (B), and Adoxophyes sp. (C) collected in Korea. 1 Dodecyl acetate, 2 (Z)-9-dodecenyl acetate, 3 10-methyldodecyl acetate, 4 tetradecyl acetate, 5 (Z)-9-tetradecenyl acetate, 6 (E)-11-tetradecenyl acetate, 7 (Z)-11-tetradecenyl acetate, 8 (Z)-9-tetradecen-1-ol, 9 (Z)-11-tetradecen-1-ol, 10 (Z)-11-hexadecenyl acetate, 11 octadecanal, C19 nonadecane, C21 heneicosane, C23 tricosane
Field Experiments
Traps baited with Z9–14:OAc or Z11–14:OAc alone caught no Adoxophyes males. Responses of males to various blends of Z9–14:OAc and Z11–14:OAc differed markedly among species. Male A. honmai were equally attracted 30:70 to 80:20 ratios of Z9–14:OAc and Z11–14:OAc (Fig. 2a), while A. orana males were preferentially attracted to ratios from 50:50 to 90:10 (Fig. 2b). In contrast, Adoxophyes sp. males were most attracted to traps baited with Z11–14:OAc as a major component (Fig. 2c). Regression analysis indicated a positive linear relationship between trap catch of male A. orana and Z9–14:OAc concentration for binary blends (r 2 = 0.75, P < 0.05), while a negative relationship was observed with Adoxophyes sp. (r 2 = 0.86, P < 0.01).
Number of A. honmai (a; Naju, May 11–18, 2007), A. orana (b; Cheonan, May 17–24, 2007), and Adoxophyes sp. (c; Namyangju, May 18–25, 2007) males captured in traps baited with lures containing different ratios of Z9–14:OAc and Z11–14:OAc at pear orchards in Korea. Bars with the same letter are not significantly different (Tukey’s test: P > 0.05)
The effects of minor component of gland extracts on field trap catches varied significantly among species. Specifically 10me-12:OAc and E11–14:OAc showed a strong synergistic effect with the 35:65 mixture of Z9–14:OAc and Z11–14:OAc for A. honmai (Fig. 3a), while the capture of male A. orana increased when the two alcohols, Z9–14:OH and Z11–14:OH, were added to the 80:20 mixture of Z9–14:OAc and Z11–14:OAc (Fig. 3b). In the case of Adoxophyes sp., the only significant effect of minor components was the strong inhibitory effect of adding E11–14:OAc to a 3:97 ratio of the primary binary blend (Fig. 3c).
The effect of adding 5% of different minor components to standard baits with 1 mg/septum of Z9–14:OAc and Z11–14:OAc on captures of male A. honmai (a; Naju, June 22–29, 2007), A. orana (b; Cheonan, July 12–19, 2007), and Adoxophyes sp. (c; Namyangju, July 13–20, 2007) at pear orchards in Korea. Bars with the same letter are not significantly different (Tukey’s test: P > 0.05)
Distribution of Adoxophyes Species
It is evident from the pheromone gland analyses of females collected as larvae from different sites that there is considerable difference in the geographic distribution of the three species (Table 2).
Discussion
The analyses of glands extracts indicate that all three Adoxophyes species have Z9–14:OAc and Z11–14:OAc as major components, but in markedly different ratios. Males were preferentially attracted to the blend found in pheromone gland extracts of their conspecific females, but males of A. honmai and A. orana both responded to a broad range of ratios of Z9–14:OAc and Z11–14:OAc. These findings suggest that while difference in the relative composition of two major pheromone components is a component in the reproductive isolation of the sympatric Adoxophyes species, this is not sufficient to provide species-specific communication channels. However, our data suggest that two minor components, 10me-12:OAc and E11–14:OAc, play a role in isolating A. honmai and Adoxophyes sp. where they occur sympatrically in the southern part (Ulju, Jinju) of Korea.
A. orana also produces Z9–14:OH and Z11–14:OH. Their presence in the lure increases trap catch and our results support the idea that these two alcohols, as well as the relative ratios of the two major components, contribute to the maintenance of premating reproductive isolation between sympatric populations of A. orana and Adoxophyes sp. in the midwestern part (Ansung, Cheonan) of Korea. In our study, we found no evidence that A. honmai and A. orana occur sympatrically in Korea, but if they do, the differences in the ratios of major components and synergistic pheromone compounds, such as 10me-12:OAc and Z9–14:OH, may prevent cross-attraction between them.
Even though E11–14:OAc is present at low levels in the pheromone glands of A. orana and Adoxophyes sp., the addition of this compound to standard baits significantly decreased attraction for conspecific males. The antagonistic effect of minor compounds found in gland extracts of conspecific females has been reported for other moths (McElfresh and Millar 1999; Wu et al. 1999). However, it is not known whether E11–14:OAc is actually released from the female glands during calling. Further research is required to compare the attractiveness of pheromone blends actually emitted with those found in female gland extracts of these two species. Similarly, the roles of Z9–12:OAc and Z11–16:OAc, present in small amounts in gland extracts of some Adoxophyes species, need to be elucidated. Do they function as antagonists to sympatric moth species, or are they just by-products of the pheromone biosynthesis? Additionally, the influence of several components present simultaneously within blends on male trap catch also needs to be investigated.
Female pheromone glands from Japanese females of A. honmai contain Z9–14:OAc and Z11–14:OAc as major components at 67:33 (Tamaki et al. 1971a) or 73:27 (Noguchi et al. 1985) ratios, with two behaviorally important minor components, 10me-12:OAc and E11–14:OAc, at 4–5% and 2–3% of the two major components, respectively (Tamaki et al. 1979; Noguchi et al. 1985). These ratios are markedly different from those observed in this study (see Table 1), providing evidence of pheromonal variation in geographic populations of A. honmai. Interestingly, the observed ratio of the two major pheromone components in Korean A. honmai feeding on tea tree as well as on fruit trees (Han 2002) is similar to that reported from an Adoxophyes sp. from tea gardens in Taiwan (Kou et al. 1990), again supporting the idea of geographic variability in the pheromones within the genus.
The pheromone glands of A. orana females in Switzerland (Guerin et al. 1986) and females from different populations of A. orana fasciata in Japan (Tamaki et al. 1971b; Sugie et al. 1984; Noguchi et al. 1985) have similar ratios (77:23 to 88:12) of the two major components. Furthermore, pheromone traps baited with lures with similar ratios are effective in capturing males at all sites (Tamaki et al. 1971b; Meijer et al. 1971; Sugie et al. 1984; Guerin et al. 1986), thus supporting the idea that populations of A. orana and/or A. orana fasciata in Asia and Europe use similar blends of the major pheromone components. The addition of Z9–14:OH or Z11–14:OH to the binary blend of Z9–14:OAc and Z11–14:OAc significantly increased captures of A. orana males in Europe (Guerin et al. 1986), and a similar pattern was noted in our field trials in Korea (Fig. 3). However, while these minor components are present in pheromone gland extracts of A. orana fasciata (Noguchi et al. 1985), their addition to lures does not enhance trap efficiency in Japan (Sugie et al. 1984). Thus, the pheromone characteristics of Korean population of A. orana are identical to those of European population of A. orana, but different from those reported for Japanese A. orana fasciata.
Our field results show that Adoxophyes sp. is the dominant species of Adoxophyes in Korea. This species has a 3:97 blend of Z9–14:OAc and Z11–14:OAc, a ratio that has not been reported from any Adoxophyes species in Japan, Taiwan, or Europe. A 1:9 ratio of Z9–14:OAc and Z11–14:OAc captured Adoxophyes privatana males in Vietnam (Hai et al. 2002), so the Korean Adoxophyes sp. may be A. privatana, although nothing is known about the pheromone gland content of this species. Additional research that examines different Adoxophyes species around the world is necessary to clarify the composition of sex pheromones of different species within this genus. Parallel morphological and molecular studies should also be carried out to clarify the phylogenetic relationships and to understand the evolution of pheromone communication in this genus.
References
Choi, K. H. 2002. Ecology and management strategy of Adoxophyes orana (Lepidoptera: Tortricidae) on apple orchards in Korea. Ph.D. dissertation. Kyungpook National University, Taegu, Korea.
Den Otter, C. J., and Klijnstra, J. W. 1983. Behaviour of male summerfruit tortrix moths, Adoxophyes orana (Lepidoptera: Tortricidae), to synthetic and natural female sex pheromone. Entomol. Exp. Appl. 28:15–21.
Guerin, P. M., Arn, H., Buser, H. R., and Charmillot, P. J. 1986. Sex pheromone of Adoxophyes orana: Additional components and variability in ratio of (Z)-9- and (Z)-11-tetradecenyl acetate. J. Chem. Ecol. 12:763–772.
Hai, T. V., Vang, L. V., Son, P. K., Inomata, S., and Ando, T. 2002. Sex attractants for moths of Vietnam: Field attraction by synthetic lures baited with known lepidopteran pheromones. J. Chem. Ecol. 28:1473–1481.
Han, K. S. 2002. Sexual isolation of two Adoxophyes orana (Lepidoptera: Tortricidae) types in Korea. Ph.D. dissertation. Seoul National University, Seoul, Korea.
Kou, R., Tang, D. S., Chow, Y. S., and Tseng, H. K. 1990. Sex pheromone components of female smaller tea tortrix moth, Adoxophyes sp. (Lepidoptera: Tortricidae) in Taiwan. J. Chem. Ecol. 16:1409–1415.
Löfstedt, C., Herrebout, W. M., and Menken, S. B. J. 1991. Sex pheromones and their potential role in the evolution of reproductive isolation in small ermine moths (Yponomeutidae). Chemoecology 2:20–28.
Mcelfresh, J. S., and Millar, J. G. 1999. Sex pheromone of Nuttall's sheep moth, Hemileuca nuttalli, from the eastern Sierra Nevada Mountains of California. J. Chem. Ecol. 25:711–726.
Meijer, G. M., Ritter, F. J., Persoons, C. J., Minks, A. K., and Voerman, S. 1971. Sex pheromones of summer fruit tortrix moth, Adoxophyes orana: Two synergistic isomers. Science 175:1469–1470.
Noguchi, H., Sugie, H., Tamaki, Y., and Oomasa, Y. 1985. Sex-pheromone components and related compounds released by virgin females of Adoxophyes sp. and Adoxophyes orana fasciata (Lepidoptera: Tortricidae). Jap. J. Appl. Entomol. Zool. 29:278–283.
Park, H., Park, I. J., Lee, S. Y., Han, K. S., Yang, C. Y., Boo, K. S., Park, K. T., Lee, J. W., and Cho, S. 2008. Molecular identification of Adoxophyes orana complex (Lepidoptera: Tortricidae) in Korea and Japan. J. Asia Pacific Entomol. 11:49–52.
Roelofs, W. L., and Brown, R. L. 1982. Pheromones and evolutionary relationships of Tortricidae. Annu. Rev. Ecol. Evol. Syst. 13:395–422.
SAS Institute 2004. Statview. SAS Institute Inc., Cary, NC.
Sato, R., and Tamaki, Y. 1977. Isolating factors between the smaller tea tortrix and the summer-fruit tortrix (Lepidoptera: Tortricidae) VI. Role of the pheromonal components, (Z)-9- and (Z)-11-tetradecen-1-ol acetates. Appl. Entomol. Zool. 12:50–59.
Shin, Y. H., Park, K. T., and Ahn, S. B. 1994. Lepidoptera, p. 314, in H. S. Ryu (ed.). Check List of Insects from Korea. Konkuk University Press, Seoul, South Korea.
Sugie, H., Tamaki, Y., Shirasaki, S., and Kitamura, T. 1984. Further studies on the sex pheromone of the summer fruit tortrix moth, Adoxophyes orana fasciata. Jap. J. Appl. Entomol. Zool. 28:156–160.
Tamaki, Y., Noguchi, H., Yushima, T., and Hirano, C. 1971a. Two sex pheromones of the smaller tea tortrix: Isolation, identification, and synthesis. Appl. Entomol. Zool. 6:139–141.
Tamaki, Y., Noguchi, H., Yushima, T., Hirano, C., Honma, K., and Sugawara, H. 1971b. Sex pheromone of the summerfruit tortrix: Isolation and identification. Kontyu 39:338–340.
Tamaki, Y., Noguchi, H., Sugie, H., Sato, R., and Kariya, A. 1979. Minor components of the female sex-attractant pheromone of the smaller tea tortrix moth (Lepidoptera: Tortricidae): Isolation and identification. Appl. Entomol. Zool. 14:101–113.
Wu, W., Cottrell, C. B., Hansson, B. S., and Löfstedt C. 1999. Comparative study of pheromone production and response in Swedish and Zimbabwean populations of the turnip moth, Agrotis segetum. J. Chem. Ecol. 25:177–196.
Yang, C. Y. 2002. Monitoring and mating disruption of the oriental fruit moth, Grapholita molesta (Lepidoptera: Tortricidae) with sex pheromone at Korean pear orchards. Ph.D. dissertation. Seoul National University, Seoul, Korea.
Yang, C. Y., Jeon, H. Y., and Boo, K. S. 2005. Geographical variation in sex pheromone composition of Adoxophyes spp. (Lepidoptera: Tortricidae) in pear orchards. Korean J. Appl. Entomol. 44:31–36.
Acknowledgments
The authors thank Prof. Soowon Cho and Dr. Bong Kyu Byun for identification of Adoxophyes species and two anonymous reviewers and Jeremy McNeil for invaluable comments. We also acknowledge the assistance of Tae Hyun Kyung with field trials.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, C.Y., Han, K.S. & Boo, K.S. Sex Pheromones and Reproductive Isolation of Three Species in Genus Adoxophyes . J Chem Ecol 35, 342–348 (2009). https://doi.org/10.1007/s10886-009-9602-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-009-9602-z