Curcumin (diferuloylmethane) is an orange-yellow component of turmeric (Curcuma longa), a spice often found in curry powder. Traditionally known for its an antiinflammatory effects, curcumin has been shown in the last two decades to be a potent immunomodulatory agent that can modulate the activation of T cells, B cells, macrophages, neutrophils, natural killer cells, and dendritic cells. Curcumin can also downregulate the expression of various proinflammatory cytokines including TNF, IL-1, IL-2, IL-6, IL-8, IL-12, and chemokines, most likely through inactivation of the transcription factor NF-κB. Interestingly, however, curcumin at low doses can also enhance antibody responses. This suggests that curcumin’s reported beneficial effects in arthritis, allergy, asthma, atherosclerosis, heart disease, Alzheimer’s disease, diabetes, and cancer might be due in part to its ability to modulate the immune system. Together, these findings warrant further consideration of curcumin as a therapy for immune disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Turmeric (called Haldi in Hindi language) and named by British as curry spice, is the dried rhizome powder of Curcuma longa, a perennial herb of the Zingiberaceae (ginger) family, which is 3–5 ft tall bearing oblong, pointed, short-stemmed leaves and funnel-shaped yellow flowers. The rhizome of turmeric is a valuable cash crop, which is widely cultivated in Asia, India, China, and other tropical countries (1). Turmeric, is commonly used as a spice in curries, food additive and also, as a dietary pigment. It has been used to treat various illnesses in the Indian subcontinent from the ancient times. Turmeric finds its use in one form or the other in the textile and pharmaceutical industries (2). It is used in Hindu religious ceremonies and Hindus also apply a mixture of turmeric and sandalwood powder on their foreheads. Turmeric has been used as a nontoxic drug in Ayurveda for centuries to treat a wide variety of disorders including rheumatism, bodyache, skin diseases, intestinal worms, diarrhea, intermittent, fevers, hepatic disorders, biliousness, urinary discharges, dyspepsia, inflammations, constipation, leukoderma, amenorrhea, and colic (3). Turmeric has been considered as an emmenagogue, diuretic, and carminative when taken orally, whereas topical application is commonly used to treat bruises, pains, sprains, boils, swellings, sinusitis, and various skin disorders (4). Turmeric is used to treat angina pectoris, stomachache, postpartum abdominal pain, and gallstones in the Chinese system of medicine (5). It seems to promote the qi flow, “stimulates menstrual discharge,” and relieves menstrual pain (6). The poultices prepared from turmeric are topically applied to relieve pain and inflammation (7). A mixture of turmeric powder and slaked lime is applied topically as a household remedy to cure injury-related sprains and swelling. Turmeric is also an effective household remedy for sore throat, cough, and common cold, where it is taken orally with tea or hot milk.
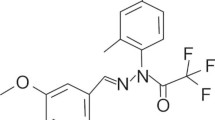
The major chemical principles of turmeric are curcuminoids, which impart characteristic yellow color to it. The curcuminoids can be separated from turmeric by ethanol extraction and it usually contains 0.3–5.4% curcumin (one of the major curcuminoids) depending on the season of its harvest (7). Vogel and Pellatier (8) first reported molecular formula of curcumin as C21H20O6, which was later identified as diferuloylmethane (8). The IUPAC name of curcumin is (1,7-bis (4-hydroxy-3-methoxy-phenyl) hepta-1, 6-diene-3, 5-dione) and its chemical structure (9) is depicted in Fig. 1. Curcumin is an orange-yellow, crystalline powder and does not dissolve in water; however, it readily goes into solution in ethanol and dimethylsulfoxide.
Curcumin as such does not possess any nutritive value however; it has been in constant use by humans as turmeric powder since Vedic times or even earlier and could be considered as pharmacologically safe. Human consumption of curcumin as a dietary spice ranges up to 100 mg/day (10) and recent phase I clinical trials indicate that humans can tolerate a dose of curcumin as high as 12 g/day, without any toxic side effects (11). The latest report has indicated safe dose of curcumin up to 12 g/day in humans (12).
The degradation kinetics of curcumin have been worked out under various pH conditions (13). Ninety percent of curcumin gets decomposed within 30 min in 0.1 M phosphate buffer and serum-free medium (pH 7.2 at 37°C). The decomposition of curcumin is pH-dependent (pH 3–10) and the rate of decomposition is higher under neutral-basic conditions. Curcumin is comparatively more stable in cell culture media containing 10% fetal calf serum and in human blood. Less than 20% of curcumin gets degraded after 1 h and approximately 50% decomposes after 8 h. The trans-6-(4′-hydroxy-3′-methoxyphenyl)-2,4-dioxo-5-hexenal has been reported as a major degradation product of curcumin, whereas vanillin, ferulic acid, and feruloyl methane were found to be the minor degradation products. Among all the major degradation products reported, the quantity of vanillin increases with time (13). Levels as low as 0.017 ng/mL of curcumin can be detected in aqueous solution in vitro by flurometric methods using mixed micelle of sodium dodecyl benzene sulfonate (SDBS) and cetyltrimethylammonium bromide (CTAB) surfactants (14).
Curcumin reportedly possesses several pharmacological properties including antiinflammatory, antimicrobial, antiviral, antifungal, antioxidant, chemosensitizing, radiosensitizing, and wound healing activities (2, 15–33). Curcumin can suppress tumor initiation, promotion, and metastasis in experimental models (34–44). It can also act as an antiproliferative agent by interrupting the cell cycle, disrupting mitotic spindle structures, and inducing apoptosis and micronucleation (45–48). Apparently, curcumin is a pluripotent pharmacological agent that utilizes multiple molecular pathways to leave its imprint on biological systems (47). This review is mainly focused on curcumin’s immunomodulatory activities.
EFFECT OF CURCUMIN ON IMMUNE CELLS
Curcumin has been found to modulate the growth and cellular response of various cell types of the immune system (Fig. 2). How this agent affects T cells, B cells, macrophages, neutrophils, NK cells, and dendritic cells is discussed in the following text.
Effect of Curcumin on T Cells
Numerous lines of evidence suggest that curcumin can modulate both the proliferation and the activation of T cells. Curcumin inhibited the proliferation induced by concanavalin A (Con A), phytohemagglutinin (PHA), and phorbol-12-myristate-13-acetate (PMA) of lymphocytes derived from fresh human spleen ((49); Table I). During these studies, curcumin also suppressed IL-2 synthesis; and IL-2 induced proliferation of lymphocytes. This correlated with suppression of NF-κB activation. Thus, these results suggest that curcumin exhibits immunosuppressive effects mediated through regulation of IL-2. In another study, the same group reported that curcumin inhibits the proliferation induced by PMA and anti-CD28 antibody or that induced by PHA of T lymphocytes isolated from healthy donors (50). In comparison, cyclosporine A was found to suppress PHA-induced T-cell proliferation but not that induced by PMA and anti-CD28 antibody. Thus, curcumin can overcome the resistance of PMA and CD28 pathway to cyclosporine A. These results suggest that curcumin exhibits immunosuppressive properties that are superior than cyclosporine A. Yadav and his group also reported that curcumin can suppress the PHA-induced proliferation of human peripheral blood mononuclear cells (PBMCs) and inhibit IL-2 expression and NF-κB (51). In still another report, curcumin inhibited the activation of human V γδT cells induced by phosphoantigens (52).
A study by Sikora et al., reported that curcumin treatment completely abolished the proliferation of Con A stimulated rat thymocytes and it also suppressed the dexamethasone-induced apoptosis in stimulated as well as nonstimulated rat thymocytes. This inhibition of apoptosis is accompanied by partial or complete oppression of AP-1 activity in nonstimulated or Con-A-stimulated thymocytes, respectively. A similar effect was also observable in rat thymocytes treated with dexamethasone; however, curcumin per se did not have any adverse effect on AP-1 activity (53). The immunomodulatory role of curcumin has been studied in HTLV-1-infected T-cells and primary ATL cells, where curcumin treatment preferentially inhibited the growth of HTLV-1-infected T-cells and primary ATL cells, but spared the normal PMBCs. This antiproliferative effect of curcumin on HTLV-1-infected T-cells and primary ATL cells was directly correlated to its ability to induce cell cycle arrest by downregulating the expression of cyclin D1, Cdk1, and Cdc25C and induce apoptosis by reducing the expression of XIAP and survivin. In addition, it also suppressed the constitutive AP-1 DNA-binding and transcriptional activity in these cells (54, 55).
Another study on mouse lymphocytes has reported that a low-dose curcumin increased the proliferation of splenic lymphocytes, whereas high-dose curcumin depressed it indicating its ability to differentially regulate the proliferation of splenic lymphocytes (56). In yet another study, curcumin treatment increased the proliferation of intestinal mucosal CD3+ T cells due to change in CD4+ T subsets in C57BL/6J-Min/+ (Min/+) mice bearing a germline mutation in Apc tumor suppressor gene (57). These studies further demonstrate the ability of curcumin to modulate immune functions in T cells.
The studies by Gerstch et al. on PMA-stimulated PBMCs have revealed that the low concentrations of curcumin significantly downregulated the expression of PMA-induced granulocyte macrophage colony stimulating factor (GM-CSF) mRNAs, whereas high concentrations upregulated interferon gamma (IFN-γ) mRNAs. These effects of curcumin were linked with the suppression of PMA-induced activation of NF-κB and downregulation of PMA-induced cyclin D1 mRNA expression in PMBCs (58). The experiments on rat splenic lymphocytes showed that curcumin treatment enhances the immune response of the lymphocytes by increasing IgG production (59).
The studies of ion transport enzyme activity in stimulated T cells revealed a marked regulatory activity of turmeric and its active principles, turmerin or curcumin. Treatment of Con A-stimulated and control human blood mononuclear T cells with different concentrations of turmeric, curcumin, and turmerin reduced ATPase levels on 3 and 5 days after treatment than the control. On the contrary, a three and twofold elevation in Ca2+ATPase and Na/K+ ATPase activities were observed on day 7, respectively (60). This could be one of the mechanisms of immunomodulation by curcumin in T cells.
Curcumin not only plays an important role in the immunomodulation of normal but also transformed T cells, where it adversely affects the cell proliferation of these cells by suppression of IL-2 gene expression and by inhibiting the activation of NF-κB (58). These results indicate that the antiproliferative activity of curcumin against T cells may be relevant for T-cell leukemia.
Effect of Curcumin on B Cells
In addition to affecting T cells, curcumin can also influence the proliferation of B cells and B lymphocyte-mediated immune function. Curcumin has been reported to block Epstein-bar virus (EBV) induced immortalization of human B cells. This effect of curcumin appears to be mediated through downregulation of oxidative stress induced by cyclosporine and hydrogen peroxide. Thus, posttransplant lymphoproliferative disorder (PLTD) associated with the use of cyclosporine during organ transplantation, can be reversed by curcumin (61). Churchill et al., have reported that curcumin treatment stimulates proliferation of B cells in the mucosa of intestine of C57BL/6J-Min/+ (Min/+) mice indicating its immunostimulatory activity (57).
Apart from affecting normal B-cells, curcumin has been found to differentially reduce the proliferation of immature B-cell lymphoma (BKS-2) cells, but not of normal cells, by inducing apoptosis and this is associated with downregulation of egr-1, c-myc, bcl-XL, and the tumor suppressor gene p53, and almost complete inhibition of NF-κB activity (62). These studies indicate that curcumin differentially regulates the immune function in normal as well as tumor cells, which could confer advantage in a therapeutic setting.
Effect of Curcumin on Macrophages
Many studies have shown curcumin’s ability to modulate the activation of macrophages. For example, curcumin seems to regulate the immune function of mice in a dose-dependent fashion as curcumin treatment enhanced the phagocytosis of peritoneal macrophages and differentially regulates the proliferation of splenocytes (56). Apart from cell proliferation, a daily diet of curcumin (30 mg/kg body weight/day) for 2 weeks in rats reportedly attenuated the ability of macrophages to generate ROS, (63) and secrete lysosomal enzymes collagenase, elastase, and hyaluronidase (64). The ability of curcumin to downregulate Th1 and NO production has been directly correlated to its ability to differentially activate the host macrophages (65).
Effect of Curcumin on Natural Killer Cells
Curcumin can also apparently modulate the activation of natural killer (NK) cells. Studies by South and his colleagues, in rats showed that curcumin at a dose of 1 and 20 mg/kg body weight could not enhance the IgG levels in the NK cells, whereas a higher dose (40 mg/kg) did elevate IgG levels significantly. More importantly, none of the three doses of curcumin significantly enhanced either delayed-type hypersensitivity or NK cell activity (66). The extended studies by these authors on YAC-1 and EL4 tumor cells and normal splenocytes in vitro showed that curcumin treatment exerted differential effects on cell viability and proliferation. Treatment with low-dose curcumin (1.25 μg/mL) enhanced the proliferation of YAC-1 cells but not that of either splenocytes or EL4 cells (66). A similar differential effect has been reported on NK cells by curcumin and it was linked to its ability to upregulate Th1 and NO production (65). In yet another study, curcumin treatment retarded the proliferation of splenic lymphocytes, cytotoxic T lymphocytes (CTLs), lymphokine-activated killer (LAK) cells, and macrophages (67). In one of the investigations, curcumin has been found to augment NK-cell cytotoxicity (51). All these studies indicate that curcumin acts like a good immunomodulatory agent.
In addition to its immunomodulation of normal NK cells, curcumin could also increase cell death of refractory natural killer/T-cell lymphoma (NKTL) cell lines (i.e., NKL, NK-92, and HANK1), which are resistant to other therapies. This was directly linked to the suppression of the NF-κB activation including the constitutively expressed NF-κB and also blockage of Bcl-xL, cyclin D1, XIAP, and c-FLIP expression and the subsequent cleavage and activation of caspase-8 and poly (ADP-ribose) polymerase (68). These observations indicate its potential as an antiproliferative agent that could play an important and decisive role in cancer chemotherapy.
Effect of Curcumin on Dendritic Cells
Dendritic cells are professional antigen-presenting cells that play a key role as immune sentinels in the initiation of T-cell responses to microbial pathogens, tumors, and inflammation (69, 70). Peripheral DCs are generally immature both phenotypically and functionally (71). They nevertheless have clinical potential as cellular adjuvants in the treatment of chronic infectious diseases and tumors (72). There is only one report to date on immune modulation of murine DCs using curcumin by Kim et al., who found that curcumin significantly depressed the expression of CD80, CD86, and MHC class II antigens in GM-CSF/IL-4 stimulated DCs without affecting MHC class I antigens. They also found that curcumin efficiently blocked the LPS-induced expression of IL-12 and inflammatory cytokines including IL-1β, IL-6, and TNF-α. Curcumin treatment enhanced the Ag capturing ability of DCs via mannose receptor-mediated endocytosis. However, their Th1 and normal cell-mediated immune response was very poor. Further studies showed that treatment of DCs with curcumin before LPS stimulation completely suppressed the LPS-induced phosphorylation of MAPK and NF-κB nuclear translocation (73). The direct suppression of these activities by curcumin in DCs may lead to the attenuated T-cell-mediated immune responses by interfering with handling and presentation of antigens by DCs.
Effect of Curcumin on Other Immune Cells
Apart from affecting T cells, B cells, macrophages, and NK cells, curcumin has also been reported to affect other immune cells such as neutrophils, etc. In one of the studies, curcumin inhibited the FMLP (a chemotactic peptide) and zymosan-activated plasma (ZAP)-induced aggregation of monkey neutrophils. However, such an action was absent in serum-treated zymosan (STZ) and arachidonic acid (AA) treated neutrophils. Curcumin also blocked the production of O2 − radicals, and myeloperoxidase, in AA-, STZ-, and fmlp-stimulated cells, except lysozymes, which were mildly affected (74). The studies on Balb/c mice spleen immunized with sheep red blood cells have shown several immunostimulatory actions of curcumin including increase in total white blood cell count, circulating antibody titer, and plaque-forming cells (75). In addition, curcumin also raised bone marrow cellularity, α-esterase-positive cells, and phagocytic activity of macrophages (75).
Moreover, curcumin has been found to induce reaginic antibody in β-lactoglobulin-challenged brown Norway rats maintained on diets comprising 10% coconut oil (CO), high oleic safflower oil, safflower oil (SO), or fish oil. Curcumin also reduced the secretion of rat chymase II (RChyII) in rats fed with SO and 0.5% curcumin, indicating variable effect on the synthesis of immunoglobulin E and the degranulation of intestinal mast cells (76). Curcumin treatment has been reported to inhibit 5-hydroxyeicosatetraenoic acid in human neutrophils in one of the studies (77).
In yet another study, curcumin caused cell death by apoptosis in both normal and transformed human (HL 60) and rodent cells despite the lack of oligonucleosomal DNA fragmentation (DNA “ladder”). However, curcumin blocked HL-60 in sub-G1 and increased caspase-3 activity (78). These results indicate that curcumin exerts its immunomodulatory action on other immune cells described earlier.
EFFECT OF CURCUMIN ON IMMUNE CYTOKINES
Effect of Curcumin on Expression and Action of TNF/TRAIL and Their Receptors
Cytokines are autocrine, paracrine, and acrine cell signaling molecules that play a crucial role in acquired as well as innate immunity. TNF-α is one of the most versatile pleiotropic cytokine that induces growth stimulation as well as inhibition by self-regulatory mechanisms of its own and plays a crucial role as an immunostimulant and mediator of host resistance to many infectious agents. Curcumin exerts its profound effects on various cytokines of the TNF superfamily. Curcumin can modulate the expression of both TNF and TNF-induced signaling and can also inhibit LPS-induced expression of TNF-α (79–81). It has also been reported to inhibit LPS or PMA-induced TNF-α in dendtritic cells, macrophages, monocytes, alveolar macrophages, and endothelial and bone marrow cells (73, 82, 83). An almost identical observation has been made in rats, where curcumin treatment attenuated TNF-α in sodium taurocholate-induced acute pancreatitis (84).
In another study, curcumin treatment blocked the expression of TNF-α mRNA in the rat model of hemorrhage and resuscitation (85). Studies by Siddiqui et al., on septic rats revealed that curcumin treatment both before and after the onset of sepsis could reduce tissue injury, mortality, and decrease TNF-α expression (86). The analysis of molecular pathways revealed that curcumin restores PPAR-γ expression in the liver of septic rats within 20 h. Similar results were obtained in endotoxin-treated cultured RAW 264.7 cells, where curcumin suppressed endotoxin-indued TNF-α expression and markedly elevated PPAR-γ expression (86).
Experiments on HT29 intestinal epithelial cells (IECs) stimulated with TNF-α and IL-1β, showed that curcumin can block the binding of Shiga-like toxins (Stx) to IECs by inhibiting Gb3 synthase (GalT6) mRNA expression (83). In another set of experiments, three major active principles namely, 1,7-bis (4-hydroxyphenyl)-1,4,6-heptatrien-3-one, procurcumenol, and epiprocurcumenol isolated from the crude methanol extract of the rhizomes of Curcuma zedoaria were reported to suppress the production of TNF-α in LPS-stimulated macrophages (87). These studies suggest that antiinflammatory activity of curcumin could well be correlated to its ability to inhibit inflammatory cytokines at protein as well as mRNA levels.
TNF-related apoptosis-inducing ligand (TRAIL) is another member of TNF superfamily that has been found to be markedly influenced by curcumin treatment in various investigations. Experiments conducted on the androgen-sensitive human prostate cancer cell line LNCaP have shown that curcumin increases cell death-promoting activity of TRAIL by inducing DNA fragmentation even though neither agent alone is significantly cytotoxic to LNCaP cells at low concentrations (10 μM curcumin and 20 ng/mL TRAIL). Further analysis of molecular mechanisms showed that combination treatment resulted in cleavage of procaspase-3, procaspase-8, and procaspase-9; truncation of Bid, and release of cytochrome c from the mitochondria (88, 89) and could be responsible for the observed increase in cytotoxicity.
Another study has reported the effect of curcumin on death receptor DR5 (DR5/TRAIL-R2) in Caki, HCT 116, HT 29, HepG2, and Hep 3B cells. This study clearly demonstrated that curcumin and TRAIL treatment synergistically increased the death of TRAIL-resistant Caki cells in a curcumin concentration-dependent manner, which could be directly correlated to the upregulated expression of DR5 and proapoptotic gene, C/EBP homologous protein (CHOP), at both the mRNA and protein levels in HCT 116, HT29, and HepG2 cell lines after curcumin treatment. This upregulation of DR5 and CHOP and cytotoxic effect of curcumin were due to its ability to generate ROS (90, 91). The combination studies on curcumin and TRAIL point that curcumin enhances the effect of TRAIL and could make TRAIL-resistant cells amenable to TRAIL therapy.
Effect of Curcumin on Interleukins
Interleukins are a group of cytokines that are secreted by leukocytes and act as communication channels between them. Curcumin can also alter the expression and activity of a variety of interleukins, especially IL-1, IL-2, IL-6, IL-8, IL-10, and IL-12 and thus can influence functions of different cells in a variety of ways (Table II). For example, treatment of PMBCs with curcumin inhibited LPS-induced IL-1β, IL-6, and TNF-α. Similarly, in rabbit experiments, curcumin reduced LPS-induced fever by attenuating the expression of IL-1β, IL-6, and TNF-α in the serum (81). This action of curcumin was mediated by suppression of NF-κB activation and downstream events that blocked these cytokines (81). Curcumin reportedly reduced PMA- or LPS-stimulated production of IL-1 and IL-8 in human peripheral blood monocytes and alveolar macrophages in a concentration- and time-dependent manner (79). A similar effect was observed for IL-2 production in PHA-stimulated human PMBCs (51).
In endothelial cell-based experiments, curcumin significantly retarded the transcriptional upregulation of IL-1α and TNF-α-induced HO-1 (an inducible form of hemeoxygenase that is upregulated in oxidant and inflammatory settings) mRNA (92). Curcumin can also reportedly block the activity of interleukin-1 (IL-1) receptor-associated kinase (IRAK) thiols in murine EL4 thymoma cells. It also abrogates the recruitment of IRAKs to the IL-1RI followed by the phosphorylation of IRAK and IL-1RI-associated proteins (93). In other studies, the production of IL-8 was abolished by curcumin in a dose and time-dependent fashion (93, 94). In experiments with SV40-transformed embryonic (WI-38 VA13) cells, dendritic cells and sodium taurocholate-induced pancreatitic rats, curcumin was able to arrest the expression of IL-6 (73, 84, 95). The studies conducted on IEC-6, HT-29, and Caco-2 cells showed that curcumin treatment represses the IL-1β-mediated ICAM-1 and IL-8 gene expression (96) and this action of curcumin was a result of suppression of NF-κB activation, RelA nuclear translocation, IκBα degradation, IκB serine 32 phosphorylation, and IκB kinase activity (96).
In still another study, curcumin depressed IL-2 synthesis in Con A-, PHA-, and PMA-stimulated human splenocytes in a concentration-dependent manner by blocking NF-κB activation (49). Similarly, curcumin successfully blocked the IL-1β-stimulated IL-8 gene expression in human bone marrow stromal cells (97). A study on a rat model of hemorrhage and resuscitation reported that curcumin treatment suppresses the production of multiple mRNA transcripts of IL-1α, IL-β, IL-2, IL-6, and IL-10 at 2 and 24 h after hemorrhage/resuscitation and this action is mediated through the inhibition of NF-κB activation and AP-1 (85). In another study, curcumin was found to exert a repressive effect on IL-12 p40 promoter activation in RAW264.7 monocytic cells transfected with p40 promoter/reporter constructs by blocking the activation of NF-κB (98). Similarly, curcumin pretreatment significantly suppressed IL-12 production and Th1 cytokine profile (i.e., decreased IFN-γ and increased IL-4 production) in CD4+ T cells stimulated with either LPS or heat-killed Listeria monocytogenes and ability of macrophages to induce IFN-γ (99). These studies show that one of the most important mechanisms of immunoregulation by curcumin is suppression of activation of NF-κB.
Effect of Curcumin on Toll-Like Receptors
Toll-like receptors (TLRs) are, type I transmembrane proteins that are key regulators of innate and adaptive immune responses in mammals that can recognize distinct pathogen-associated molecular signatures (100). A few studies have reported the influence of curcumin on TLRs. In one such study with Ba/F3 cells, curcumin abated LPS-induced IRF3 activation and LPS-induced TLR4 signaling by arresting both myeloid differentiation factor 88 (MyD88)- and the TIR domain containing adaptor inducing IFN-β (TRIF)-dependent pathways. However, curcumin could not abrogate the IRF3 activation in 293T cells caused by increased expression of TRIF, indicating that curcumin also targets the TLR4 receptor complex in addition to IKKβ (101). In studies using peritoneal mesothelial cells from C3H/HeN mice, curcumin has been shown to suppress lipid A-induced NF-κB, MCP-1, and MIP-2 mRNA, implying its role in TLR4 signaling (102). In still another experiment, treatment of C3H/HeN mouse splenic macrophages with curcumin was found to abrogate LPS-induced mRNA expression of TLR2 and block NF-κB activation (103). Studies on TLRs indicate that immunomodulatory activity of curcumin may also be due to its ability to target TLRs.
Effect of Curcumin on Chemokines
Chemokines are small, chemotactic cytokines, which direct leukocyte migration, activate inflammatory responses, and help regulate tumor growth. A number of studies in various study systems have confirmed curcumin’s potential to suppress various chemokines. In experiments with human pancreatic carcinoma cell lines, curcumin abated the constitutive production of IL-8 while raising the expression of IL-8 receptors CXCR1 and CXCR2 (93). Similarly, in human colorectal cancer cells, curcumin blocked in a time- and dose-dependent manner the IL-1β-stimulated and neurotensin receptor-induced expression of IL-8 (94). In other studies, curcumin blocked the expression of IL-6 in WI-38 VA13 and dendritic cells and also in sodium taurocholate-induced acute pancreatitis in rats (73, 84, 95, 96). The studies by Mendez-Samperio et al., on Mycobacterium bovis Calmette-Guerin (BCG)-stimulated human monocytes reported that curcumin abated BCG-induced IL-8 secretion by blocking nuclear translocation of NF-κB (104).
Curcumin reportedly arrests the expression of the chemokines MCP-1 (105) and interferon-inducible protein-10 kDa (IP-10) in mouse bone marrow stromal cells. This effect is apparently mediated by curcumin’s ability to prevent TNF, IL-1, and LPS-induced expression of MCP-1 and IP-10 mRNA, and it is completely reversible within 24 h after removing curcumin from the cell culture medium. The inhibition of AP-1 and NF-κB activation are responsible for this activity of curcumin (106, 107).
Studies on human gingival fibroblasts have shown that curcumin impedes the chemotactic activity of monocytes isolated from the culture supernatant of Porphyromonas gingivalis LPS (P-LPS)-treated cells (108). This effect of curcumin seems to be mediated by blocking the P-LPS-induced expression of MCP-1 gene and AP-1 and NF-κB activation in human gingival fibroblasts (108). Experiments by Kobayashi et al. have shown that curcumin arrests in a concentration-dependent manner the proliferation of lymphocytes from common house dust mite (Dermatophagoides jhrinea) atopic asthmatics and also their ability to secrete IL-2, IL-5, GM-CSF, and IL-4 (109). The immunomodulatory activity of curcumin may also be due to its ability to alter chemokine expression as indicated earlier.
Effect of Curcumin on Adhesion molecules
Experiments on human umbilical vein endothelial cells demonstrated that curcumin blocks the steady-state transcription of ICAM-1, VCAM-1, and E-selectin both temporally and reversibly (110).
EFFECT OF CURCUMIN ON INFLAMMATORY ENZYMES
Curcumin can markedly influence the activities of enzymes that are hallmark of inflammation and subsequently various disease states in humans (Table III). Curcumin can differentially block inflammatory enzymes involved in inflammation and extracellular matrix degradation at both the mRNA and protein levels (111–127). In several murine studies, curcumin has been shown to abate TPA-induced epidermal inflammation, and inhibit epidermal lipooxygenase and cyclooxygenase (COX) activities in dose-dependent fashion by downregulating TPA-induced NF-κB activation (111–113). Another study has also reported suppression of TPA-induced COX-2, prostaglandin E2 (PGE2), and MMP-9 expression. This was in direct correlation with the inhibition of ERK1/2 phosphorylation and NF-κB activation (114). A similar effect has been reported in Colo 205 colon carcinoma cells, where curcumin reduced COX-2, PGE2, matrix metalloproteinase (MMP)-2, COX-1, and MMP-9 levels, but had no effect on MMP-7 levels (115). Other studies have reported that curcumin blocked the expression of COX-2 mRNA (116, 117) but had no effect on COX-1 mRNAs (116). Plummer et al. observed that curcumin suppressed the protein levels of LPS-induced COX-2 and PGE2 in human leucocytes in a dose-dependent manner (118). A similar effect was reported in A549 human lung epithelial cells, where curcumin inhibited IL-1β or IFN-α-induced prostaglandin E2 and cyclooxygenase-2 at both the protein and the mRNA levels (119, 120). In rats with TNBS-induced colitis, curcumin blocked the expression of COX-2 and inflammatory cytokines while increasing PGE2 levels (121). Various studies on human lung epithelial cells exposed to cigarette smoke have shown that curcumin inhibited NNK-induced activation of NF-κB and COX-2 expression (122, 123). Similarly, in HaCaT cells, curcumin abolished UVB-induced COX-2 expression by suppressing p38 MAPK and JNK activity (124).
In human colon epithelial cells, curcumin arrested the TNF-α, and fecapentaene-12-induced COX-2 by blocking NF-κB activation and IKK activity (125). A similar effect has been observed in HT-29 colon cancer cells, where curcumin arrested the mRNA and protein expression of COX-2 but not of COX-1 (126). Studies on LPS-stimulated RAW-264.7 cells indicate that curcumin reduces COX-2 expression, PGE2 formation, and the catalytic activities of 5-LOX (127).
The studies on mouse peritoneal exudates have revealed that low-dose curcumin reduces LPS- and IFNγ-induced NO production, whereas higher doses do not (128). In another investigation, curcumin reduced the production of iNOS mRNA in cultured BALB/c mouse peritoneal macrophages ex vivo in a concentration-dependent manner and also iNOS mRNA expression in the livers of mice receiving two oral doses of 0.5 mL of a 10-μM curcumin (92 ng/g of body weight) and LPS (129). Similarly, in studies using activated macrophages, low-dose curcumin inhibited NO production at 24 h (IC50 of 6 μM) and 10 μM curcumin also reduced NOS activity than noncurcumin-treated activated macrophages (130). In another study, curcumin has been reported to reduce LPS-induced NO production in mouse macrophages (51). These studies affirm that curcumin acts as a strong antiinflammatory agent.
EFFECT ON CURCUMIN ON TRANSCRIPTION FACTOR NF-κB
The nuclear factor NF-κB is a ubiquitous transcription factor important for its pleiotropic effects, inducible expression patterns, unique regulatory mechanisms, and involvement in a large number of signaling and gene expression pathways (Table III). The activation of NF-κB is crucial to innate and adaptive immunity and it plays an important role in inflammation, autoimmune diseases, and cancer. As shown in a seminal study performed in our laboratory, curcumin abrogates NF-κB activation induced by TNF-α, PMA, or H2O2, by blocking the phosphorylation of IKKα (131). Moreover, our studies on tobacco smoke-induced NF-κB activation in myeloblastic and mantle cell lymphoma cells revealed that curcumin blocks NF-κB activation by inhibiting the phosphorylation and degradation of IκBα (132). Curcumin also reportedly abrogates LPS-induced MAPK activation and the translocation of NF-κB p65 in DCs (73). In several other experiments, curcumin has been reported to downregulate IL1 or TNF-α or LPS-induced NF-κB activation (51, 99, 133, 134). A study with A549 cells has reported that the ability of curcumin to arrest NF-κB binding activity is reversible within 30 min after IFN-α administration (134). In studies on LNCaP cancer cells, curcumin mediated TRAIL-induced apoptosis by blocking IκBα phosphorylation and degradation and subsequently abrogated NF-κB activation (88, 89). The studies with NCTC 2544 keratinocytes have shown that curcumin can inhibit UVB-induced TNF-α, IL-6, and IL-8 by impeding NF-κB activation (135).
The studies with WI-38 VA13 cells have revealed that curcumin can also inhibit the degradation of IκBα upstream and subsequent NF-κB DNA-binding activity and NF-κB-dependent expression of IL-6 downstream (95). Curcumin treatment also repressed NF-κB and Ap-1 activation induced by hemorrhage/resuscitation injury in rats (85). Similar to this, curcumin has also been found to arrest the TPA-induced NF-κB activation by attenuating the degradation of IκBα and the subsequent translocation of the p65 subunit in cultured HL-60 cells. Alternatively, curcumin also repressed the TPA-induced activation of NF-κB through direct interruption of the binding of NF-κB to its consensus DNA sequences (136). The experiments on EC-6, HT-29, and Caco-2 cells revealed that curcumin blocks cytokine-induced NF-κB DNA binding activity, RelA nuclear translocation, IκBα degradation, IκB serine 32 phosphorylation, and IKK activity (96).
Furthermore, as already mentioned earlier, curcumin can prevent and treat wasting and histopathologic symptoms associated with TNBS-induced intestinal inflammation by simultaneously blocking NF-κB activation, degradation of cytoplasmic IκBα protein, and cytokine mRNA expression (137). The experiments on mouse splenic macrophages have shown that high-dose curcumin can abrogate LPS-mediated TLR2 mRNA induction by inhibiting NF-κB activation (102, 103). The studies by Watanabe et al. have shown that curcumin can also arrest BCG-induced IL-8 production in human monocytes and gingival fibroblasts by inhibiting NF-κB activation (108). Finally, curcumin abolished constitutive activation of NF-κB in HTLV-1 infected T-cell lines and primary ATL cells, by inhibiting phosphorylation of IκBα and Tax-induced NF-κB transcriptional activity (55). The various studies outlined earlier indicate that suppression of NF-κB activity may be one of the most important properties of curcumin that could be responsible for its various immune functions.
EFFECT OF CURCUMIN ON IMMUNE DISEASES
Because of its ability to modulate immune cells and immune cell cytokines, curcumin has been shown to affect several autoimmune diseases (Fig. 3). Inflammation is a critical feature of most autoimmune diseases. Thus, the role of curcumin in the therapy of such disorders is expected.
Alzheimer’s Disease
Several reports suggest that curcumin has potential against Alzheimer’s disease (138–141), a disease characterized by the amyloid-induced inflammation in the brain. The effect of curcumin in Alzheimer’s disease is mediated through the downmodulation of cytokine (i.e., TNF-α and IL-1β) and chemokine (i.e., MIP-1b, MCP-1, and IL-8) activity in peripheral blood monocytes and reduces amyloid-β plaque formation (138–141).
Multiple Sclerosis
There are reports that curcumin may have potential against multiple sclerosis, another autoimmune disease. In animal model of this disease, curcumin was found to inhibit IL-12-induced tyrosine phosphorylation of Janus kinase 2, tyrosine kinase 2, and STAT3 and STAT4 transcription factors (142).
Cardiovascular Diseases
Curcumin has established antioxidant and antiinflammatory activities that offer promise in the treatment of cardiovascular diseases. For example, it can inhibit lipid peroxidation; reduce creatinine kinase and lactate dehydrogenase levels; and restore reduced glutathione, glutathione peroxidase, and superoxide dismutase to normal levels. Curcumin can also downregulate the expression of myocardial TNF-α and MMP-2 and upregulate the expression of eNOS mRNA (143–147).
Diabetes
In diabetes, curcumin can suppress blood glucose levels, increase the antioxidant status of pancreatic β-cells, and enhance the activation of PPAR-γ (148–153).
Allergy
As shown in experiments in vivo (in guinea pigs) and in vitro (rat basophilic leukemia cells), curcumin can help clear constricted airways and increase antioxidant levels (154, 155).
Asthma
That curcumin can relieve symptoms of asthma, has been reported. These effects are linked with reduction of the lymphocytic production of IL-2, IL-5, GM-CSF, and IL-4 that is associated with bronchial asthma (110, 154).
Inflammatory Bowel Disease
As shown in vivo in humans and rats, curcumin can ameliorate inflammatory bowel disease by reducing inflammatory cytokine levels, blunting NO and O2 production, and suppressing NF-κB activation in colon epithelium (156, 157).
Rheumatoid Arthritis
In rheumatoid arthritis, curcumin exerts beneficial effects by inhibiting the expression of collagenase and stromelysin and the proliferation of synoviocytes (158, 159).
Renal Ischemia
In renal ischemia, curcumin can exert beneficial effects that include reducing creatine levels; upregulating MnSOD levels; and inhibiting the expression of RANTES, MCP-1, and allograft inflammatory factor (160, 161).
Psoriasis
Clinical evaluation of topical application of 1% curcumin gel in psoriatic areas reduced the density of CD8+ T cells when compared to untreated areas, where density of CD8+ T cells showed an elevation (162). This and other studies suggest that curcumin treatment could be an effective paradigm in the treatment of psoriasis as it could also reduce the activity of phosphorylase kinase (163).
Scleroderma
Because scleroderma is a disease that involves excessive collagen deposition and hyperproliferation of fibroblasts, curcumin may be able to provide a therapeutic benefit through its ability to suppress the proliferation of lung fibroblasts in a process involving the inhibition of protein kinase Cε (164).
Acquired Immunodeficiency Disease (AIDS)
There are several reports indicating that curcumin may have potential against AIDS. These effects of curcumin are mediated through suppression of replication of human immunodeficiency virus (HIV) by inhibition of HIV long terminal repeat (165, 166) and HIV protease (167), inhibits HIV-1 integrase (168, 169), inhibits p300/CREB-binding protein-specific acetyltransferase, and represses the acetylation of histone/nonhistone proteins and histone acetyltransferase-dependent chromatin transcription (170). Thus, curcumin has a great potential also against AIDS.
CONCLUSIONS
The curcumin, an orange-yellow polyphenol present in curry spice, Curcuma longa has a long history of therapeutic use in the Ayurvedic and Chinese systems of medicine. The wisdom and scientific credentials of this approach have been corroborated by numerous studies conducted over the past 30 years. Indeed, curcumin has been found to possess antioxidant, antiinflammatory, anticancer, and several other activities listed in this review. Mechanistic studies have not only confirmed beyond doubt that curcumin employs multiple pathways to leave its imprint on biological systems, but also warrants its potential use as a modern nontoxic chemotherapy for numerous disorders. Curcumin primarily exerts its therapeutic effects by inhibiting the degradation of IκBα and subsequent inactivation of NF-κB, thus initiating a cascade of downstream inflammatory and immunogenic events. Curcumin’s inhibition of NF-κB activation, in turn, leads directly to the inhibition of expression of a number of proinflammatory cytokines (e.g., TNF, IL-1, IL-2, IL-6, IL-8, and IL-12) and downregulation of the mRNA expression of several proinflammatory enzymes (e.g., COX, LOX, MMPs, and NOS). In addition, curcumin’s immunogenic response is further enhanced by its ability to inhibit TLRs, Finally, curcumin exerts proimmune activity in several autoimmune disorders including Alzheimer’s disease, multiple sclerosis, cardiovascular disease, diabetes, allergy, asthma, inflammatory bowel disease, rheumatoid arthritis, renal ischemia, psoriasis, and scleroderma. Overall, these findings suggest that curcumin warrants further consideration as a potential immunoregulatory treatment in various immune disorders.
REFERENCES
Dobelis Hamper IN (ed): Magic and Medicine of Plants. Pleasantville, NY, Reader’s Digest Association, 1986
Srimal RC, Dhawan BN: Pharmacology of diferuloyl methane (curcumin), a non-steroidal anti-inflammatory agent. J Pharm Pharmacol 25(6):447–452, 1973
Jain SK, DeFilipps RA: Medicinal Plants of India. Algonac, MI, Reference, 1991, p 120
Nadkarni AK: Indian Materia Medica, Vol 1. Bombay, India, Popular Book Depot, 1954
Chang HM, But BPH: Pharmacology and Applications of Chinese Materia Medica, Vol 2. Philadelphia, PA, World Scientific, 1986, pp 936–939
Tu G, Fang Q, Guo J, Yuan S, Chen C, Chen J, Chen Z, Cheng S, Jin R, Li M, et al.: Pharmacopoeia of the People’s Republic of China. Guangzhou, P.R. China, Guangdong Science and Technology Press, 1992, pp 202–203
Leung A: Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics. New York, Wiley, 1980, pp 313–314
Lampe V, Milobedeska J, Kostanecki V: Ber Dtsch Chem Ges 43:2163, 1910
Lampe V, Milobedeska J: Ber Dtsch Chem Ges 46:2235, 1913
Ammon HP, Wahl MA: Pharmacology of Curcuma longa. Planta Med 57(1):1–7, 1991
Cheng AL, Hsu CH, Lin JK, Hsu MM, Ho YF, Shen TS, Ko JY, Lin JT, Lin BR, Ming-Shiang W, Yu HS, Jee SH, Chen GS, Chen TM, Chen CA, Lai MK, Pu YS, Pan MH, Wang YJ, Tsai CC, Hsieh CY: Phase I clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res 21(4B):2895–2900, 2001
Lao CD, Ruffin MT 4th, Normolle D, Heath DD, Murray SI, Bailey JM, Boggs ME, Crowell J, Rock CL, Brenner DE: Dose escalation of a curcuminoid formulation. BMC Complement Altern Med 6:10, 2006
Wang YJ, Pan MH, Cheng AL, Lin LI, Ho YS, Hsieh CY, Lin JK: Stability of curcumin in buffer solutions and characterization of its degradation products. J Pharm Biomed Anal 15(12):1867–1876, 1997
Wang F, Wu X, Wang F, Liu S, Jia Z, Yang J: The sensitive method for the determination of curcumin using the enhancement of mixed micelle. J Fluoresc 16(1):53–59, 2006
Arora R, Kapoor V, Basu N, Jain AP: Anti-inflammatory studies on Curcuma longa (turmeric). Ind J Med Res 59:1289–1295, 1971
Mukhopadhyay A, Basu N, Ghatak N: Anti-inflammatory and irritant activities of curcumin analogues rats. Agents Actions 12:508–515, 1982
Srivastava R: Inhibition of neutrophil response by curcumin. Agents Actions 28:298–303, 1989
Jobin C, Bradham CA, Russo MP, Juma, B, Narula AS, Brenner DA, Sartor RB: Curcumin blocks cytokine-mediated Nfkappa B activation and proinflammatory gene expression by inhibiting inhibitory factor I-κB kinase activity. J Immunol 163:3474–3483, 1999
Negi PS, Jayaprakasha GK, Jagan Mohan Rao L, Sakariah KK: Antibacterial activity of turmeric oil: A byproduct from curcumin manufacture. J Agric Food Chem 47:4297–4300, 1999
Mazumder A, Raghavan K, Weinstein J, Kohn KW, Pommier Y: Inhibition of human immunodeficiency virus type-1 integrase by curcumin. Biochem Pharmcol 49:1165–1170, 1995
Bourne KZ, Bourne N, Reising SF, Stanberry LR: Plant products as topical microbicide candidates: Assessment of in vitro and in vivo activity against herpes simplex virus type 2. Antivir Res 42:219–226, 1999
Apisariyakul A, Vanittanakom N, Buddhasukh D: Antifungal activity of turmeric oil extracted from Curcuma longa (Zingiberaceae). J Ethnopharmacol 49:163–169, 1995
Reddy AC, Lokesh BR: Effect of curcumin and eugenol on iron-induced hepatic toxicity in rats. Toxicology 107(1):39–45, 1996
Ramsewak RS, DeWitt DL, Nair MG: Cytotoxicity, antioxidant and anti-inflammatory activities of curcumins I-III from Curcuma longa. Phytomedicine 7:303–308, 2000
Ruby AJ, Kuttan G, Babu KD, Rajasekharan KN, Kuttan R: Anti-tumour and antioxidant activity of natural curcuminoids. Cancer Lett 94:79–83, 1995
Balasubramanyam M, Koteswari AA, Kumar RS, Monickaraj SF, Maheswari JU, Mohan V: Curcumin-induced inhibition of cellular reactive oxygen species generation: Novel therapeutic implications. J Biosci 28(6):715–721, 2003
Garg AK, Buchholz TA, Aggarwal BB: Chemosensitization and radiosensitization of tumors by plant polyphenols. Antioxid Redox Signal 7(11–12):1630–1647, 2005
Abraham SK, Sarma L, Kesavan PC: Protective effects of chlorogenic acid, curcumin and beta-carotene against gamma-radiation-induced in vivo chromosomal damage. Mutat Res 303(3):109–112, 1993
Rezvani M, Ross GA: Modification of radiation-induced acute oral mucositis in the rat. Int J Radiat Biol 80(2):177–182, 2004
Chendil D, Ranga RS, Meigooni D, Sathishkumar S, Ahmed MM: Curcumin confers radiosensitizing effect in prostate cancer cell line PC-3. Oncogene 23:1599–1607, 2004
Khafif A, Hurst R, Kyker K, Fliss DM, Gil Z, Medina JE: Curcumin: A new radiosensitizer of squamous cell carcinoma cells. Otolaryngol Head Neck Surg 132:317–321, 2005
Sidhu GS, Singh AK, Thaloor D, Banaudha KK, Patnaik GK, Srimal RC, Maheshwari RK: Enhancement of wound healing by curcumin in animals. Wound Repair Regen 6:167–177, 1998
Sidhu GS, Mani H, Gaddipati JP, Singh AK, Seth P, Banaudha KK, Patnaik GK, Maheshwari RK: Curcumin enhances wound healing in streptozotocin induced diabetic rats and genetically diabetic mice. Wound Repair Regen 7:362–374, 1999
Jiang MC, Yang-Yen HF, Yen JJ, Lin JK: Curcumin induces apoptosis in immortalized NIH 3T3 and malignant cancer cell lines. Nutr Cancer 26:111–120, 1996
Huang M-T, Lou RY, Ma W, Newmark HL, Reuhl KR, Conney AH: Inhibitory effects of dietary curcumin on forestomach duodenal and colon carcinogenesis in mice. Cancer Res 54:5841–5847, 1994
Huang M-T, Smart RC, Wong CQ, Conney AH: Inhibitory effect of curcumin, chlorogenic acid, caffeic acid and ferulic acid on tumor promotion in mouse skin by 12-O-tetradecanoyl phorbol-13-acetate. Cancer Res 48:5941–5946, 1988
Wang ZY, Georgiadis CA, Laskin JD, Conney AH: Inhibitory effects of curcumin on tumor initiation by benzo(a)pyrene and 7,12-dimethylbenz(a)anthracene. Carcinogenesis 54:5841–5847, 1992
Rao CV, Rivenson A, Simi B, Reddy BS: Chemoprevention of colon carcinogenesis by dietary curcumin, a naturally occurring plant phenolic compound. Cancer Res 55:259–266, 1995
Pereira MA, Grubbs CJ, Barnes LH, Li H, Olson GR, Eto I, Juliana M, Whitaker LM, Kelloff GJ, Steele VE, Lubet RA: Effects of the phytochemicals, curcumin and quercetin upon azoxymethane induced colon cancer and 7,12 dimethylbenz(a)anthracene induced mammary cancer in rats. Carcinogenesis 17:1305–1311, 1996
Bhide SV, Azuine MA, Lahiri M, Telang NT: Chemoprevention of mammary tumor virus-induced and chemical carcinogen-induced rodent mammary tumors by natural plant products. Breast Cancer Res Treat 30:233–242, 1994
Singletary K, MacDonald C, Wallig M, Fischer C: Inhibition of 7,12-dimethylbenz(a) anthracene (DMBA) induced mammary tumorigenesis and DMBA-DNA adduct formation by curcumin. Cancer Lett 103:137–141, 1996
Mohan R, Sivak J, Ashton P, Russo LA, Pham BQ, Kasahara N, Raizman MB, Fini ME: Curcuminoids inhibit the angiogenic response stimulated by fibroblast growth factor-2, including expression of matrix metalloproteinase gelatinase B. J Biol Chem 275:10405–10412, 2000
Perkins S, Verschoyle RD, Hill K, Parveen I, Threadgill MD, Sharma RA, Williams ML, Steward WP, Gescher AJ: Chemopreventive efficacy and pharmacokinetics of curcumin in the min/ +mouse, a model of familial adenomatous polyposis. Cancer Epidemiol Biomarkers Prev 11(6):535–540, 2002
Siwak DR, Shishodia S, Aggarwal BB, Kurzrock R: Curcumin-induced antiproliferative and proapoptotic effects in melanoma cells are associated with suppression of IkappaB kinase and nuclear factor kappaB activity and are independent of the B-Raf/mitogen-activated/extracellular signal-regulated protein kinase pathway and the Akt pathway. Cancer 104(4):879–890, 2005
Holy JM: Curcumin disrupts mitotic spindle structure and induces micronucleation in MCF-7 breast cancer cells. Mutat Res 518:71–84, 2002
Choudhuri T, Pal S, Agwarwal ML, Das T, Sa G: Curcumin induces apoptosis in human breast cancer cells through p53-dependent Bax induction. FEBS Lett 512:334–340, 2002
Aggarwal BB, Kumar A, Bharti AC: Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23(1A):363–398, 2003
LoTempio MM, Veena MS, Steele HL, Ramamurthy B, Ramalingam TS, Cohen AN, Chakrabarti R, Srivatsan ES, Wang MB: Curcumin suppresses growth of head and neck squamous cell carcinoma. Clin Cancer Res 11(19 Pt 1):6994–7002, 2005
Ranjan D, Chen C, Johnston TD, Jeon H, Nagabhushan M: Curcumin inhibits mitogen stimulated lymphocyte proliferation, NF-κB activation, and IL-2 signaling. J Surg Res 121(2):171–177, 2004
Ranjan D, Johnston TD, Wu G, Elliott L, Bondada S, Nagabhushan M: Curcumin blocks cyclosporine A-resistant CD28 costimulatory pathway of human T-cell proliferation. J Surg Res 77(2):174–178, 1998
Yadav VS, Mishra KP, Singh DP, Mehrotra S, Singh VK: Immunomodulatory effects of curcumin. Immunopharmacol Immunotoxicol 27:485–497, 2005
Cipriani B, Borsellino G, Knowles H, Tramonti D, Cavaliere F, Bernardi G, Battistini L, Brosnan CF: Curcumin inhibits activation of Vγ9Vδ2 T cells by phosphoantigens and induces apoptosis involving apoptosis-inducing factor and large scale DNA fragmentation. J Immunol 167(6):3454–3462, 2001
Sikora E, Bielak-Zmijewska A, Piwocka K, Skierski J, Radziszewska E: Inhibition of proliferation and apoptosis of human and rat T lymphocytes by curcumin, a curry pigment. Biochem Pharmacol 54(8):899–907, 1997
Tomita M, Kawakami H, Uchihara JN, Okudaira T, Masuda M, Takasu N, Matsuda T, Ohta T, Tanaka Y, Mori N: Curcumin suppresses constitutive activation of AP-1 by downregulation of JunD protein in HTLV-1-infected T-cell lines. Leuk Res 30(3):313–321, 2006
Tomita M, Kawakami H, Uchihara JN, Okudaira T, Masuda M, Takasu N, Matsuda T, Ohta T, Tanaka Y, Ohshiro K, Mori N: Curcumin (diferuloylmethane) inhibits constitutive active NF-kappaB, leading to suppression of cell growth of human T-cell leukemia virus type I-infected T-cell lines and primary adult T-cell leukemia cells. Int J Cancer 118:765–772, 2006
Li X, Liu X: Effect of curcumin on immune function of mice. J Huazhong Univ Sci Technol Med Sci 25(2):137–140, 2005
Churchill M, Chadburn A, Bilinski RT, Bertagnolli MM: Inhibition of intestinal tumors by curcumin is associated with changes in the intestinal immune cell profile. J Surg Res 89:169–175, 2000
Gertsch J, Guttinger M, Heilmann J, Sticher O: Curcumin differentially modulates mRNA profiles in Jurkat T and human peripheral blood mononuclear cells. Bioorg Med Chem 11(6):1057–1063, 2003
Kuramoto Y, Yamada K, Tsuruta O, Sugano M: Effect of natural food colorings on immunoglobulin production in vitro by rat spleen lymphocytes. Biosci Biotechnol Biochem 60(10):1712–1713, 1996
Cohly HHP, Rao MR, Kanji VK, Patlolla B, Taylor A, Wilson MT, Angel MF, Das SK: Effect of turmeric, turmerin, and curcumin on Ca2+, Na/K+ ATPases in concanavalin A-stimulated human blood mononuclear cells. Int J Mol Sci 4:34–44, 2003
Ranjan D, Siquijor A, Johnston TD, Wu G, Nagabhuskahn M: The effect of curcumin on human B-cell immortalization by Epstein-Barr virus. Am Surg 64(1):47–51, 1998
Han SS, Chung ST, Robertson DA, Ranjan D, Bondada S: Curcumin causes the growth arrest and apoptosis of B cell lymphoma by downregulation of egr-1, c-myc, bcl-XL, NF-kappa B, and p53. Clin Immunol 93:152–161, 1999
Joe B, Lokesh BR: Role of capsaicin, curcumin and dietary n − 3 fatty acids in lowering the generation of reactive oxygen species in rat peritoneal macrophages. Biochim Biophys Acta 1224(2):255–263, 1994
Joe B, Lokesh BR: Dietary n − 3 fatty acids, curcumin and capsaicin lower the release of lysosomal enzymes and eicosanoids in rat peritoneal macrophages. Mol Cell Biochem 203(1–2):153–161, 2000
Bhaumik S, Jyothi MD, Khar A: Differential modulation of nitric oxide production by curcumin in host macrophages and NK cells. FEBS Lett 483:78–82, 2000
South EH, Exon JH, Hendrix K: Dietary curcumin enhances antibody response in rats. Immunopharmacol Immunotoxicol 19(1):105–119, 1997
Gao X, Kuo J, Jiang H, Deeb D, Liu Y, Divine G, Chapman RA, Dulchavsky SA, Gautam SC: Immunomodulatory activity of curcumin: suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production in vitro. Biochem Pharmacol 68:51–61, 2004
Kim K, Ryu K, Ko Y, Park C: Effects of nuclear factor-kappaB inhibitors and its implication on natural killer T-cell lymphoma cells. Br J Haematol 131(1):59–66, 2005
Crowley M, Inaba K, Steinman RM: Dendritic cells are the principal cells in mouse spleen bearing immunogenic fragments of foreign proteins. J Exp Med 172:383–386, 1990
Steinman RM: The dendritic cell system and its role in immunogenicity. Annu Rev Immunol 9:271–296, 1991
Cella M, Sallusto F, Lanzavecchia A: Origin, maturation and antigen-presenting function of dendritic cells. Curr Opin Immunol 9:10–16, 1997
Banchereau J, Steinman RM: Dendritic cells and the control of immunity. Nature 392:245–252, 1998
Kim GY, Kim KH, Lee SH, Yoon MS, Lee HJ, Moon DO, Lee CM, Ahn SC, Park YC, Park YM: Curcumin inhibits immunostimulatory function of dendritic cells: MAPKs and translocation of NF-kappa B as potential targets. J Immunol 174(12):8116–8124, 2005
Srivastava R: Inhibition of neutrophil response by curcumin. Agents Actions 28(3–4):298–303, 1989
Antony S, Kuttan R, Kuttan G: Immunomodulatory activity of curcumin. Immunol Invest 28(5–6):291–303, 1999
Ju HR, Wu HY, Nishizono S, Sakono M, Ikeda I, Sugano M, Imaizumi K: Effects of dietary fats and curcumin on IgE-mediated degranulation of intestinal mast cells in brown Norway rats. Biosci Biotechnol Biochem 60(11):1856–1860, 1996
Flynn DL, Rafferty MF, Boctor AM: Inhibition of 5-hydroxy-eicosatetraenoic acid (5-HETE) formation in intact human neutrophils by naturally-occurring diarylheptanoids: Inhibitory activities of curcuminoids and yakuchinones. Prostaglandins Leukot Med 22(3):357–360, 1986
Bielak-Zmijewska A, Koronkiewicz M, Skierski J, Piwocka K, Radziszewska E, Sikora E: Effect of curcumin on the apoptosis of rodent and human nonproliferating and proliferating lymphoid cells. Nutr Cancer 38(1):131–138, 2000
Chan MM: Inhibition of tumor necrosis factor by curcumin, a phytochemical. Biochem Pharmacol 49(11):1551–1556, 1995
Lantz RC, Chena GJ, Solyomb AM, Jolad SD, Timmermann BN: The effect of turmeric extracts on inflammatory mediator production. Phytomedicine 12:445–452, 2005
Lee JJ, Huang WT, Shao DZ, Liao JF, Lin MT: Blocking NF-kappaB activation may be an effective strategy in the fever therapy. Jpn J Physiol 53(5):367–375, 2003
Abe Y, Hashimoto S, Horie T: Curcumin inhibition of inflammatory cytokine production by human peripheral blood monocytes and alveolar macrophages. Pharmacol Res 39(1):41–47, 1999
Moon DO, Jin CY, Lee JD, Choi YH, Ahn SC, Lee CM, Jeong SC, Park YM, Kim GY: Curcumin decreases binding of Shiga-like toxin-1B on human intestinal epithelial cell line HT29 stimulated with TNF-alpha and IL-1beta: Suppression of p38, JNK and NF-kappaB p65 as potential targets. Biol Pharm Bull 29(7):1470–1475, 2006
Gulcubuk A, Altunatmaz K, Sonmez K, Haktanir-Yatkin D, Uzun H, Gurel A, Aydin S: Effects of curcumin on tumour necrosis factor-α and interleukin-6 in the late phase of experimental acute pancreatitis. J Vet Med A Physiol Pathol Clin Med 53:49–54, 2006
Gaddipati JP, Sundar SV, Calemine J, Seth P, Sidhu GS, Maheshwari RK: Differential regulation of cytokines and transcription factors in liver by curcumin following hemorrhage/resuscitation. Shock 19(2):150–156, 2003
Siddiqui AM, Cui X, Wu R, Dong W, Zhou M, Hu M, Simms HH, Wang P: The anti-inflammatory effect of curcumin in an experimental model of sepsis is mediated by up-regulation of peroxisome proliferator-activated receptor-γ. Crit Care Med 34(7):1874–1882, 2006
Jang MK, Sohn DH, Ryu JH: A curcuminoid and sesquiterpenes as inhibitors of macrophage TNF-alpha release from Curcuma zedoaria. Planta Med 67(6):550–552, 2001
Deeb D, Xu YX, Jiang H, Gao X, Janakiraman N, Chapman RA, Gautam SC: Curcumin (diferuloyl-methane) enhances tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in LNCaP prostate cancer cells. Mol Cancer Ther 2:95–103, 2003
Deeb D, Jiang H, Gao X, Hafner MS, Wong H, Divine G, Chapman RA, Dulchavsky SA, Gautam SC: Curcumin sensitizes prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L by inhibiting nuclear factor-{kappa}B through suppression of I{kappa}B{alpha} phosphorylation. Mol Cancer Ther 3(7):803–812, 2004
Jung E, Park MJ, Choi KS, Park J-W, Lee HI, Lee KS, Kwon TK: Curcumin sensitizes tumor necrosis factor-related apoptosis- inducing ligand (TRAIL)-mediated apoptosis through CHOP-independent DR5 upregulation. Carcinogenesis 27(10):2008–2017, 2006
Jung EM, Lim JH, Lee TJ, Park J-W, Choi KS, Kwon TK: Curcumin sensitizes tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis through reactive oxygen species-mediated upregulation of death receptor 5 (DR5). Carcinogenesis 26:1905–1913, 2005
Terry CM, Clikeman JA, Hoidal JR, Callahan KS: Effect of tumor necrosis factor-α and interleukin-1α on heme oxygenase-1 expression in human endothelial cells. Am J Physiol Heart Circ Physiol 274(43):H883–H891, 1998
Hidaka H, Ishiko T, Furuhashi T, Kamohara H, Suzuki S, Miyazaki M, Ikeda O, Mita S, Setoguchi T, Ogawa M: Curcumin inhibits interleukin 8 production and enhances interleukin 8 receptor expression on the cell surface: impact on human pancreatic carcinoma cell growth by autocrine regulation. Cancer 95:1206–1214, 2002
Wang X, Wang Q, Ives KL, Ever BM: Curcumin inhibits neurotensin-mediated interleukin-8 production and migration of HCT116 human colon cancer cells. Clin Cancer Res 12(18):5356–5355, 2006
Renard P, Delaive E, Van Steenbrugge M, Remacle J, Raes M: Is the effect of interleukin-1 on glutathione oxidation in cultured human fibroblasts involved in nuclear factor-κB activation? Antioxid Redox Signal 3:329–340, 2001
Jobin C, Bradham CA, Russo MP, Juma B, Narula AS, Brenner DA, Sartor RB: Curcumin blocks cytokine-mediated NF-κB activation and proinflammatory gene expression by inhibiting inhibitory factor I-κB kinase activity. J Immunol 163(6):3474–3483, 1999
Chaudhary LR, Avioli LV: Regulation of interleukin-8 gene expression by interleukin-1beta, osteotropic hormones, and protein kinase inhibitors in normal human bone marrow stromal cells. J Biol Chem 271:16591–16596, 1996
Kang BY, Chung SW, Chung W, Im S, Hwang SY, Kim TS: Inhibition of interleukin-12 production in lipopolysaccharide-activated macrophages by curcumin. Eur J Pharmacol 384(2–3):191–195, 1999
Kang BY, Song YJ, Kim KM, Choe YK, Hwang SY, Kim TS: Curcumin inhibits Th1 cytokine profile in CD4+ T cells by suppressing interleukin-12 production in macrophages. Br J Pharmacol 128(2):380–384, 1999
Takeda K, Kaisho T, Akira S: Toll-like receptors. Annu Rev Immunol 21:335–376, 2003
Youn HS, Saitoh SI, Miyake K, Hwang DH: Inhibition of homodimerization of Toll-like receptor 4 by curcumin. Biochem Pharmacol 72(1):62–69, 2006
Kato S, Yuzawa Y, Tsuboi N, Maruyama S, Morita Y, Matsuguchi T, Matsuo S: Endotoxin-induced chemokine expression in murine peritoneal mesothelial cells: the role of toll-like receptor 4. J Am Soc Nephrol 15:1289–1299, 2004
Matsuguchi T, Musikacharoen T, Ogawa T, Yoshikai Y: Gene expressions of Toll-like receptor 2, but not Toll-like receptor 4, is induced by LPS and inflammatory cytokines in mouse macrophages. J Immunol 165(10):5767–5772, 2000
Mendez-Samperio P, Palma J, Vazquez A: Roles of intracellular calcium and NF-κB in the Bacillus Calmette-Guerin-induced secretion of interleukin-8 from human monocytes. Cell Immunol 211(2):113–122, 2001
Nakayama K, Furusu A, Xu Q, Konta T, Kitamura M: Unexpected transcriptional induction of monocytes chemoattractant protein 1 by proteasome inhibition: involvement of the c-Jun N-terminal kinase-activator protein 1 pathway. J Immunol 167(3):1145–1150, 2001
Xu YX, Pindolia KR, Janakiraman N, Noth CJ, Chapman RA, Gautam SC: Curcumin, a compound with anti-inflammatory and anti-oxidant properties, downregulates chemokine expression in bone marrow stromal cells. Exp Hematol 25(5):413–422, 1997
Xu YX, Pindolia KR, Janakiraman N, Chapman RA, Gautam SC: Curcumin inhibits IL1 alpha and TNF-alpha induction of AP-1 and NF-kB DNA-binding activity in bone marrow stromal cells. Hematopathol Mol Hematol 11:49–62, 1998
Watanabe A, Takeshita A, Kitano S, Hanazawa S: CD14-mediated signal pathway of Porphyromonas gingivalis lipopolysaccharide in human gingival fibroblasts. Infect Immun 64(11):4488–4494, 1996
Kobayashi T, Hashimoto S, Horie T: Curcumin inhibition of Dermatophagoides farinea-induced interleukin-5 (IL-5) and granulocyte macrophage-colony stimulating factor (GM-CSF) production by lymphocytes from bronchial asthmatics. Biochem Pharmacol 54(7):819–824, 1997
Gupta B, Ghosh B: Curcuma longa inhibits TNF-alpha induced expression of adhesion molecules on human umbilical vein endothelial cells. Int J Immunopharmacol 21(11):745–757, 1999
Huang MT, Lysz T, Ferraro T, Abidi TF, Laskin JD, Conney AH: Inhibitory effects of curcumin on in vitro lipooxygenase and cyclooxygenase activities in mouse epidermis. Cancer Res 51(3):813–819, 1991
Tunstall RG, Sharma RA, Perkins S, Sale S, Singh R, Farmer PB, Steward WP, Gescher AJ: Cyclooxygenase-2 expression and oxidative DNA adducts in murine intestinal adenomas: modification by dietary curcumin and implications for clinical trials. Eur J Cancer 42(3):415–421, 2006
Chun KS, Keum YS, Han SS, Song YS, Kim SH, Surh YJ: Curcumin inhibits phorbol ester-induced expression of cyclooxygenase-2 in mouse skin through suppression of extracellular signal-regulated kinase activity and NF-κB activation. Carcinogenesis 24:1515–1524, 2003
Lee KW, Kim JH, Lee HJ, Surh YJ: Curcumin inhibits phorbol ester-induced up-regulation of cyclooxygenase-2 and matrix metalloproteinase-9 by blocking ERK1/2 phosphorylation and NF-kappaB transcriptional activity in MCF10A human breast epithelial cells. Antioxid Redox Signal 7(11–12):1612–1620, 2005
Su CC, Chen GW, Lin JG, Wu LT, Chung JG: Curcumin inhibits cell migration of human colon cancer colo 205 cells through the inhibition of nuclear factor kappa B/p65 and down-regulates cyclooxygenase-2 and matrix metalloproteinase-2 expressions. Anticancer Res 26(2A):1281–1288, 2006
Park C, Kim GY, Kim GD, Choi BT, Park YM, Choi YH: Induction of G2/M arrest and inhibition of cyclooxygenase-2 activity by curcumin in human bladder cancer T24 cells. Oncol Rep 15(5):1225–1231, 2006
Atsumi T, Murakami Y, Shibuya K, Tonosaki K, Fujisawa S: Induction of cytotoxicity and apoptosis and inhibition of cyclooxygenase-2 gene expression, by curcumin and its analog, alpha-diisoeugenol. Anticancer Res 25(6B):4029–4036, 2005
Plummer SM, Hill KA, Festing MF, Steward WP, Gescher AJ, Sharma RA: Clinical development of leukocyte cyclooxygenase 2 activity as a systemic biomarker for cancer chemopreventive agents. Cancer Epidemiol Biomarkers Prev 10(12):1295–1299, 2001
Moon Y, Glasgow WC, Eling TE: Curcumin suppresses interleukin 1β-mediated microtonal prostaglandin E syntheses 1 by altering early growth response gene 1 and other signaling pathways. J Pharmacol Exp Ther 315(2):788–795, 2005
Lee J, Im YH, Jung HH, Kim JH, Park JO, Kim K, Kim WS, Ahn JS, Jung CW, Park YS, Kang WK, Park K: Curcumin inhibits interferon-alpha induced NF-kappaB and COX-2 in human A549 non-small cell lung cancer cells. Biochem Biophys Res Commun 334(2):313–318, 2005
Jiang H, Deng CS, Zhang M, Xia J: Curcumin-attenuated trinitrobenzene sulphonic acid induces chronic colitis by inhibiting expression of cyclooxygenase-2. World J Gastroenterol 12(24):3848–3853, 2006
Shishodia S, Potdar P, Gairola CG, Aggarwal BB: Curcumin (diferuloylmethane) down-regulates cigarette smoke-induced NF-kappaB activation through inhibition of IkappaB alpha kinase in human lung epithelial cells: correlation with suppression of COX-2, MMP-9 and cyclin D1. Carcinogenesis 24(7):1269–1279, 2003
Sharma C, Kaur J, Shishodia S, Aggarwal BB, Ralhan R: Curcumin down regulates smokeless tobacco-induced NF-kappaB activation and COX-2 expression in human oral premalignant and cancer cells. Toxicology 228(1):1–15, 2006
Cho JW, Park K, Kweon GR, Jang BC, Baek WK, Suh MH, Kim CW, Lee KS, Suh SI: Curcumin inhibits the expression of COX-2 in UVB-irradiated human keratinocytes (HaCaT) by inhibiting activation of AP-1: p38 MAP kinase and JNK as potential upstream targets. Exp Mol Med 37(3):186–912, 2005
Plummer SM, Holloway KA, Manson MM, Munks RJ, Kaptein A, Farrow S, Howells L: Inhibition of cyclo-oxygenase 2 expression in colon cells by the chemopreventive agent curcumin involves inhibition of NF-kappaB activation via the NIK/IKK signalling complex. Oncogene 18(44):6013–6020, 1999
Goel A, Boland CR, Chauhan DP: Specific inhibition of cyclooxygenase-2 (COX-2) expression by dietary curcumin in HT-29 human colon cancer cells. Cancer Lett 172(2):111–118, 2001
Hong J, Bose M, Ju J, Ryu JH, Chen X, Sang S, Lee MJ, Yang CS: Modulation of arachidonic acid metabolism by curcumin and related beta-diketone derivatives: Effects on cytosolic phospholipase A(2), cyclooxygenases and 5-lipoxygenase. Carcinogenesis 25(9):1671–1679, 2004
Chan MM, Ho CT, Huang HI: Effects of three dietary phytochemicals from tea, rosemary and turmeric on inflammation-induced nitrite production. Cancer Lett 96(1):23–29, 1995
Chan MM, Huang HI, Fenton MR, Fong D: In vivo inhibition of nitric oxide synthase gene expression by curcumin, a cancer preventive natural product with anti-inflammatory properties. Biochem Pharmacol 55(12):1955–1962, 1998
Brouet I, Ohshima H: Curcumin, an anti-tumour promoter and anti-inflammatory agent, inhibits induction of nitric oxide synthase in activated macrophages. Biochem Biophys Res Commun 206(2):533–540, 1995
Singh S, Aggarwal BB: Activation of transcription factor NF-κB is suppressed by curcumin (diferulolylmethane). J Biol Chem 270(42):24995–25000, 1995
Shishodia S, Amin HM, Lai, R, Aggarwal BB: Curcumin (diferuloylmethane) inhibits constitutive NF-kB activation, induces G1/S arrest, suppresses proliferation, and induces apoptosis in mantle cell lymphoma. Biochem Pharmacol 70:700–771, 2005
Brennan P, O’Neill LA: Inhibition of nuclear factor kappaB by direct modification in whole cells—mechanism of action of nordihydroguaiaritic acid, curcumin and thiol modifiers. Biochem Pharmacol 55(7):965–973, 1998
Lee J, Im YH, Jung HH, Kim JH, Park JO, Kim K, Kim WS, Ahn JS, Jung CW, Park YS, Kang WK, Park K: Curcumin inhibits interferon-alpha induced NF-kappaB and COX-2 in human A549 non-small cell lung cancer cells. Biochem Biophys Res Commun 334(2):313–318, 2005
Grandjean-Laquerriere A, Gangloff SC, Le Naour R, Trentesaux C, Hornebeck W, Guenounou M: Relative contribution of NF-kB and Ap-1 in the modulation by curcumin and pyrrolidine dithiocarbamate of the UVB-induced cytokine expression by keratinocytes. Cytokine 18(3):68–77, 2002
Han SS, Keum YS, Seo HJ, Surh YJ: Curcumin suppresses activation of NF-κB and AP-1 induced by phorbol ester in cultured human promyelocytic leukemia cells. J Biochem Mol Biol 35(3):337–342, 2002
Jian YT, Mai GF, Wang JD, Zhang YL, Luo RC, Fang YX: Preventive and therapeutic effects of NF-κB inhibitor curcumin in rats colitis induced by trinitrobenzene sulfonic acid. World J Gastroenterol 11(12):1747–1752, 2005
Lim GP, Chu T, Yang F, Beech W, Frautschy SA, Cole GM: The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci 21(21):8370–8377, 2001
Giri RK, Rajagopal V, Kalra VK: Curcumin, the active constituent of turmeric, inhibits amyloid peptide-induced cytochemokine gene expression and CCR5-mediated chemotaxis of THP-1 monocytes by modulating early growth response-1 transcription factor. J Neurochem 91(5):1199–1210, 2004
Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS, Chen PP, Kayed R, Glabe CG, Frautschy SA, Cole GM: Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem 280(7):5892–5901, 2005
Zhang L, Fiala M, Cashman J, Sayre J, Espinosa A, Mahanian M, Zaghi J, Badmaev V, Graves MC, Bernard G, Rosenthal M: Curcuminoids enhance amyloid-β uptake by macrophages of Alzheimer’s disease patients. J Alzheimers Dis 10:1–7, 2006
Natarajan C, Bright JJ: Curcumin inhibits experimental allergic encephalomyelitis by blocking IL-12 signaling through Janus kinase-STAT pathway in T lymphocytes. J Immunol 168(12):6506–6513, 2002
Nirmala C, Puvanakrishnan R: Protective role of curcumin against isoproterenol induced myocardial infarction in rats. Mol Cell Biochem 159(2):85–93, 1996
Dikshit M, Rastogi L, Shukla R, Srimal RC: Prevention of ischaemia-induced biochemical changes by curcumin & quinidine in the cat heart. Indian J Med Res 101:131–135, 1995
Cheng H, Liu W, Ai X: Protective effect of curcumin on myocardial ischemia reperfusion injury in rats. Zhong Yao Cai 28(10):920–922, 2005
Yao QH, Wang DQ, Cui CC, Yuan ZY, Chen SB, Yao XW, Wang JK, Lian JF: Curcumin ameliorates left ventricular function in rabbits with pressure overload: inhibition of the remodeling of the left ventricular collagen network associated with suppression of myocardial tumor necrosis factor-alpha and matrix metalloproteinase-2 expression. Biol Pharm Bull 27(2):198–202, 2004
Chai H, Yan S, Lin P, Lumsden AB, Yao Q, Chen C: Curcumin blocks HIV protease inhibitor ritonavir-induced vascular dysfunction in porcine coronary arteries. J Am Coll Surg 200(6):8208–8230, 2005
Arun N, Nalini N: Efficacy of turmeric on blood sugar and polyol pathway in diabetic albino rats. Plant Foods Hum Nutr 57(1):41–52, 2002
Srivivasan A, Menon VP, Periaswamy V, Rajasekaran KN: Protection of pancreatic beta-cell by the potential antioxidant bis-o-hydroxycinnamoyl methane, analogue of natural curcuminoid in experimental diabetes. J Pharm Pharm Sci 6(3):327–333, 2003
Babu PS, Srinivasan K: Influence of dietary curcumin and cholesterol on the progression of experimentally induced diabetes in albino rat. Mol Cell Biochem 152(1):13–21, 1995
Babu PS, Srinivasan K: Hypolipidemic action of curcumin, the active principle of turmeric (Curcuma longa) in streptozotocin induced diabetic rats. Mol Cell Biochem 166(1–2):169–175, 1997
Kuroda M, Mimaki Y, Nishiyama T, Mae T, Kishida H, Tsukagawa M, Takahashi K, Kawada T, Nakagawa K, Kitahara M: Hypoglycemic effects of turmeric (Curcuma longa L. rhizomes) on genetically diabetic KK-Ay mice. Biol Pharm Bull 28(5):937–939, 2005
Murugan P, Pari L: Antioxidant effect of tetrahydrocurcumin in streptozotocin-nicotinamide induced diabetic rats. Life Sci 79(18):1720–1728, 2006
Ram A, Das M, Ghosh B: Curcumin attenuates allergen-induced airway hyperresponsiveness in sensitized guinea pigs. Biol Pharm Bull 26(7):1021–1024, 2003
Suzuki M, Nakamura T, Iyoki S, Fujiwara A, Watanabe Y, Mohri K, Isobe K, Ono K, Yano S: Elucidation of anti-allergic activities of curcumin-related compounds with a special reference to their anti-oxidative activities. Biol Pharm Bull 28(8):1438–1443, 2005
Salh B, Assi K, Templeman V, Parhar K, Owen D, Gomez-Munoz A, Jacobson K: Curcumin attenuates DNB-induced murine colitis. Am J Physiol Gastrointest Liver Physiol 285(1):G235–G243, 2003
Holt PR, Katz S, Kirshoff R: Curcumin therapy in inflammatory bowel disease: a pilot study. Dig Dis Sci 50(11):2191–2193, 2005
Funk JL, Oyarzo JN, Frye JB, Chen G, Lantz RC, Jolad SD, Solyom AM, Timmermann BN: Turmeric extracts containing curcuminoids prevent experimental rheumatoid arthritis. J Nat Prod 69:351–355, 2006
Jackson JK, Higo T, Hunter WL, Burt HM: The antioxidants curcumin and quercetin inhibit inflammatory processes associated with arthritis. Inflamm Res 55:168–175, 2006
Shoskes D: Effect of bioflavonoids quercetin and curcumin on ischemic renal injury: a new class of renoprotective agents. Transplantation 66:147–152, 1998
Shahed AR, Jones E, Shoskes D: Quercetin and curcumin up-regulate antioxidant gene expression in rat kidney after ureteral obstruction or ischemia/reperfusion injury. Transplant Proc 33:2988, 2001
Heng MC, Song MK, Harker J, Heng MK: Drug-induced suppression of phosphorylase kinase activity correlates with resolution of psoriasis as assessed by clinical, histological and immunohistochemical parameters. Br J Dermatol 143:937–949, 2000
Bosman B: Testing of lipooxygenase inhibitors, cyclooxigenase inhibitors, drugs with immunomodulating properties and some reference antipsoriatic drugs in the modified mouse tail test, an animal model of psoriasis. Skin Pharmacol 7, 324–334, 1994
Tourkina E, Gooz P, Oates JC, Ludwicka-Bradley A, Silver RM, Hoffman S: Curcumin-induced apoptosis in scleroderma lung fibroblasts: role of protein kinase Cε. Respir Cell Mol Biol 31(1):28–35, 2004
Li CJ, Zhang LJ, Dezube BJ, Crumpacker CS, Pardee AB. Three inhibitors of type 1 human immunodeficiency virus long terminal repeat-directed gene expression and virus replication. Proc Natl Acad Sci USA 90(5):1839–1842, 1993
Barthelemy S, Vergnes L, Moynier M, Guyot D, Labidalle S, Bahraoui E: Curcumin and curcumin derivatives inhibit Tat-mediated transactivation of type 1 human immunodeficiency virus long terminal repeat. Res Virol 149(1):43–52, 1998
Sui Z, Salto R, Li J, Craik C, Ortiz de Montellano PR: Inhibition of the HIV-1 and HIV-2 proteases by curcumin and curcumin boron complexes. Bioorg Med Chem 1(6):415–422, 1993
Mazumder A, Raghavan K, Weinstein J, Kohn KW, Pommier Y: Inhibition of human immunodeficiency virus type-1 integrase by curcumin. Biochem Pharmacol 49(8):1165–1170, 1995
Vajragupta O, Boonchoong P, Morris GM, Olson AJ: Active site binding modes of curcumin in HIV-1 protease and integrase. Bioorg Med Chem Lett 15(14):3364–3368, 2005
Balasubramanyam K, Varier RA, Altaf M, Swaminathan V, Siddappa NB, Ranga U, Kundu TK: Curcumin, a novel p300/CREB-binding protein-specific inhibitor of acetyltransferase, represses the acetylation of histone/nonhistone proteins and histone acetyltransferase-dependent chromatin transcription. J Biol Chem 279(49):51163–51171, 2004
ACKNOWLEDGMENTS
We would like to thank Walter Pagel for carefully proofreading the manuscript and providing valuable comments. Dr. Aggarwal is a Ransom Horne, Jr., Professor of Cancer Research. This work was supported by a grant from the Clayton Foundation for Research (to B. B. A.), National Institutes of Health PO1 grant CA91844 on lung chemoprevention (to B. B. A.), National Institutes of Health P50 Head and Neck SPORE grant P50CA97007 (to B. B. A); and a core grant (CA16672).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jagetia, G.C., Aggarwal, B.B. “Spicing Up” of the Immune System by Curcumin. J Clin Immunol 27, 19–35 (2007). https://doi.org/10.1007/s10875-006-9066-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-006-9066-7