Abstract
The microclimate of an improved hay meadow was studied using Tinytag dataloggers to record sward temperature after cutting. Temperatures in the sward were then compared to grasshopper abundances to see if mowing created an excessively hot microclimate unfavourable for sustained grasshopper activity in mid summer. The abundance of Chorthippus albomarginatus and Chorthippus parallelus was significantly reduced on the hay plots compared to the unmanaged control swards, which may have been due to high sward temperatures created by the absence of tall, shady vegetation in which grasshoppers may take refuge to avoid overheating. This study suggests that a combination of mortality caused by the physical process of mowing, and high sward temperatures created by removal of the standing crop by cutting may cause the low abundance of grasshoppers in improved grassland in eastern England. This research is particularly important when considering the orthopteran assemblages of Environmental Stewardship Scheme field margins where mowing for hay in July and August may seriously reduce grasshopper populations. If mowing of grassland has to occur during the grasshopper season, we suggest a later cut in September or a system of rotational mowing, leaving areas of uncut grassland as shelter.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Grasshoppers (Orthoptera: Acrididae) are known to suffer mortality during mechanised mowing of hay meadows during July in the UK (Gardiner and Hill 2006b). It is thought that the large size of the mature nymphs and adults and their location in the lower layers of the sward (<20 cm; Gardiner and Hill 2005b) may render them particularly susceptible to death or damage through contact with the cutting blades. Indeed, Chorthippus parallelus was in significantly reduced abundance 1 h after mowing on a hay treatment (abundance m−2 1 h before cutting: 0.30, 1 h after cutting: 0.11) when compared to an uncut control where numbers did not vary 1 h before or after cutting (abundance m−2 before cutting: 0.22, after cutting: 0.20), suggesting a substantial mortality rate for this grasshopper (Gardiner 2006).
Grasshoppers that survive the physical process of mowing may find the resultant short, homogenous sward (<10 cm in height; Gardiner 2006) unsuitable for sustained reproduction or feeding possibly due to the lack of tussocks of vegetation that provide shelter from inclement weather and avian predation (Hamilton 1975; Young 1979; Gardiner et al. 2002). Short grasslands may be unfavourable for grasshoppers due to high microclimatic temperatures (>44°C), which can lead to shade-seeking behaviour and vigorous escape responses in several grasshopper species (Uvarov 1966; Willott 1997). The optimum air temperature for development of grasshoppers in the UK is thought to be 35–40°C (Willott 1997). Responses to microclimatic temperatures may differ between species, for example, Myrmeleotettix maculatus is a short sward specialist and its small size may be an adaptation for the high temperatures it experiences (Willott 1992). Contrastingly, Omocestus viridulus, a long grass species in the UK, is a large insect which may overheat in short, hot grasslands and it therefore avoids those habitats (Willott 1997).
It is not known how high temperatures may rise in recently mown hay meadows and whether this is likely to affect grasshopper populations present in the sward after cutting. In microclimatically hostile environments such as cut and heavily grazed habitats, nymphal and adult Chorthippus parallelus may disperse to taller patches of grass that may provide the required conditions for sustained feeding and reproductive bouts (Gardiner and Hill 2004; Gardiner 2006), leading to a low abundance of this species in short grasslands with vegetation <10 cm in height (Gardiner et al. 2002). Grass field margins created under the Countryside Stewardship Scheme (CSS) have been found to have a low abundance of grasshoppers in eastern England (Gardiner and Hill 2005a), perhaps due to the cutting of the strips for hay in July and August which removed most of the grassland habitat in one event, leaving a very short sward not conducive to sustained development and reproduction. Grasshoppers such as C. parallelus are scarce in agricultural land in mid Essex (Gardiner et al. 2002; Gardiner and Hill 2003) and grass field margins may be important in ensuring its continued existence on farmland in the area. Large grasshopper populations may also be an important source of prey for a range of invertebrates such as the Wasp Spider Argiope bruennichi (Gardiner and Hill 2005a), and for declining farmland bird species such as Alauda arvensis and Emberiza cirlus (Evans et al. 1997) in the UK. Understanding the effects of farmland cutting on grasshoppers will allow management to be put in place that enhances orthopteran abundance, and therefore benefits their avian and invertebrate predators.
It is the aim of this paper to present the results of a small-scale study of the microclimate and grasshopper populations of an improved hay meadow at Writtle College, Essex, UK, and to discuss the likely implications for conservation management of grass field margins on farmland in the UK.
Methods
Microclimate, sward height and grasshopper abundance were measured after cutting in an improved hay meadow at Writtle College, Chelmsford, Essex, UK (O.S. grid reference: TL664067), in 2003 and 2004. The vegetation was dominated by Lolium perenne, interspersed with small patches of bare earth. The experiment was small-scale and composed of 2 replicates of a hay treatment, which was cut on 10 July in both years and the cuttings removed after a 4-day period to facilitate drying of the hay. Two replications of an uncut control treatment were also established to allow a comparison with the hay treatment. Replicates were 20 × 20 m and were situated at random throughout the Lolium sward. A rotary ride-on mower was used to cut the hay replicates with the cutting height set at 9 cm.
Microclimate
The microclimate of both treatments was characterised by taking temperature readings on an hourly basis for 40 days after mowing on 10 July in 2003 and 2004 (last recording day 19 August) using Tinytag dataloggers (Gemini Data Loggers, Chichester, West Sussex, UK). The dataloggers were yellow in colour and were situated in plastic bags (to protect them from the effects of precipitation; Willott (1997)) on wooden stakes at a height of 10 cm. To obtain the temperature readings, dataloggers (1 per replicate) were downloaded using GLM software (available from Gemini Data Loggers) on a standard PC after the 40-day recording period had finished. It is understood that the dataloggers may not be indicative of the basking temperature of grasshoppers but they do provide a comparative estimate of the microclimatic temperatures of both treatments and give an indication of the likely sward temperature. The exposed dataloggers recorded a combination of air temperature and solar radiation and it was believed that the probes did not need shade, as this would be very unrepresentative of the temperatures experienced in the sward. One datalogger was placed at random in each replicate and all dataloggers faced westwards (at right angles to the sun through the hottest part of the day) to avoid the effect of differing aspect on temperature readings.
Grasshopper populations
Open quadrats were used to sample grasshopper populations of the hay and control treatments. This method is frequently used for surveys of Orthoptera in grassland ecosystems (Richards and Waloff 1954; Gardiner et al. 2002; Gardiner and Hill 2006a) and relies mainly upon the active jumping behaviour of the insect. The size of quadrats used in this study was 2 × 2 m (4 m2). The quadrats used in both treatments were in fixed positions for the duration of the study to overcome any problems with contagiously dispersed populations (Gardiner et al. 2005), markers denoting their corners. Each treatment had a total of 32 quadrats (16 quadrats within the boundaries of each replicate). All quadrats were sampled for grasshoppers on 4 occasions after cutting in 2003 and this methodology was repeated in 2004 (dates of sampling in this year were as near as possible to 2003 dates). All quadrat surveys were undertaken in similar weather conditions (>17°C, sunny) and at the same time of day (between 09:00 and 16:00 h). Surveying criteria were modified from the Butterfly Monitoring Scheme outlined in Pollard and Yates (1993) and should be suitable for Orthoptera which prefer similar weather conditions (Marshall and Haes 1988).
Grasshoppers only jump when they are disturbed (often in response to predators) or to pass easily through the leaf canopy (Clarke 1948). The method used in this study was to move from one edge of the quadrat to the other (at a walking speed of 0.25 m/s) brushing the vegetation with a pole (diameter: 50 mm, length: 1 m) to cause any grasshoppers present in the quadrat to jump (Gardiner et al. 2002; Gardiner and Hill 2006a). The ‘flushing’ in this study was conducted in a similar way to sweeping as the observer moved from one edge of the quadrat to the other sweeping the vegetation in an 180° arc (Gardiner and Pye 2001). Only Orthoptera within the quadrat at the start of the sweep were recorded by Gardiner et al. (2002), with those leaping in from outside discounted. It was possible to accurately sort adult grasshoppers to species on sight (Richards and Waloff 1954).
This method for surveying grasshoppers is an accurate, quick and inexpensive form of sampling Orthoptera assemblages (Gardiner et al. 2005; Gardiner and Hill 2006a), which provides an index of abundance and activity in a sward. Grasshoppers may be more easily observed in short vegetation due to the absence of places where they may hide from the surveyor; therefore they may have been more easily sighted in the short grass of the mown hay meadow compared to the unmanaged control.
Sward height
The height of the vegetation was recorded using a drop disc method (Smith et al. 1993; Stewart et al. 2001). The disc was dropped from the top of a metre rule standing vertically and the height of the vegetation where the disc settled was measured in cm. The disc weighed 200 g and had a diameter of 300 mm and was constructed from plywood. Stewart et al. (2001) conclude that this method is objective and simple to use, although the data may be influenced by shoot density and the strength of the plant stems on which the disc rests (e.g. stems of some species are rigid and the disc may rest on the top giving an unrealistic assessment of vegetation height). Sward height was sampled on 6 August in 2003 and 20 August in 2004, with 64 individual height measurements taken at random from both treatments on each occasion.
Statistical analysis
For the purposes of statistical analysis, the 40-day period after cutting was subdivided into 4 blocks of 10-days each (e.g. 1–10 days after cut, 11–20 days after cut and so on). For each 10-day block in each year, mean daily maximum sward temperature, mean daily minimum sward temperature and mean daily sward temperature were calculated for each replicate of the hay and control treatments. All sward temperatures were averaged for both replicates for each treatment in all 10-day periods so that the data could be analysed using a 3-way ANOVA with treatment (hay, control), time interval after cutting (1–10 days, 11–20 days etc.) and year as the factors. All temperature data were square root transformed prior to analysis to correct for non-normality. A 3-way ANOVA was conducted on mean maximum, mean minimum and mean temperature to determine whether temperature was affected by the treatments or whether it varied at different time intervals after cutting, or between years, which may have consequences for grasshoppers present in the sward.
The grasshopper data for Chorthippus albomarginatus and C. parallelus adults for each treatment in each 10-day block in 2003 and 2004 (data averaged for both replicates in each treatment similar to temperature analysis) was square root transformed to correct for non-normality and subjected to a 3-way ANOVA to determine if cutting affected grasshopper density/activity or if abundance varied at differing time intervals after mowing or between years. SPSS was used to calculate the ANOVA statistics (SPSS 2006).
Results
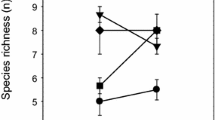
Chorthippus albomarginatus and C. parallelus were substantially more abundant on the control treatment in both 2003 and 2004 (Fig. 1). Indeed, treatment was found to significantly affect the abundance of both species (C. albomarginatus F = 17.21, d.f. 1, P < 0.01; C. parallelus F = 73.46, d.f. 1, P < 0.01). Year had no significant impact on the abundance of either grasshopper species (C. albomarginatus F = 1.65, d.f. 1, P = 0.23; C. parallelus F = 0.61, d.f. 1, P = 0.45) which had similar population densities in both 2003 and 2004 (Fig. 1). It seemed that there was a large rise in the abundance of C. parallelus on the control plots 11–20 days after cutting in 2004 (Fig. 1). However, there was no significant difference between abundances at varying time intervals after cutting for C. albomarginatus (F = 0.56, d.f. 3, P = 0.65) or C. parallelus (F = 1.67, d.f. 3, P = 0.24).
Cutting significantly affected mean sward temperature (F = 6.28, d.f. 1, P < 0.05) with higher temperatures being recorded on the hay plots (Fig. 2). However, cutting treatment had no significant effect on maximum (F = 0.72, d.f. 1, P = 0.42) or minimum (F = 0.65, d.f. 1, P = 0.44) sward temperature, which differed little between hay and control swards in both years of the study (Fig. 2). There was no significant effect of time interval after cutting for maximum (F = 2.21, d.f. 3, P = 0.15) or minimum temperature (F = 1.66, d.f. 3, P = 0.24), only mean temperature differed significantly after cutting (F = 3.94, d.f. 3, P < 0.05).
In 2004, it was noticeably cooler and the maximum sward temperature did not exceed 43°C on either treatment, in contrast to 2003 when maximum temperature frequently exceeded 44°C (Fig. 2). Indeed, there was a significant year effect detected for mean (F = 21.55, d.f. 1, P < 0.01), maximum (F = 7.52, d.f. 1, P < 0.05) and minimum (F = 15.12, d.f. 1, P < 0.01) sward temperature.
There were few tussocks of tall vegetation on the mown hay plots (11% of drop disc readings >10 cm in 2003, 33% >10 cm in 2004) in comparison to the uncut control where there was an abundance of shady, tall vegetation (90% of drop disc readings >10 cm in 2003, 98% >10 cm in 2004).
Discussion
The higher sward temperatures on the hay plots, particularly in 2003 (Fig. 2), may have posed a substantial problem for grasshoppers, which may become physiologically stressed at high temperatures (Uvarov 1966; Willott 1997). The hot microclimate of the hay treatment may have led to vigorous escape responses of grasshoppers away from the cut vegetation to taller grass where they could cool down in the shade provided by these conditions. However, it was difficult to detect any large-scale emigration of insects from inspection of our data due to the limited replication and wide variation in the numbers of grasshoppers between replicates, particularly for C. albomarginatus (Fig. 1). Despite data variability there was a suggestion of a large influx of individuals of C. parallelus on the control treatment 11–20 days after cutting in 2004 (Fig. 1), implying movement away from the adjacent cut swards to the uncut plots. This kind of behavioural thermoregulation at high temperatures has been stressed as particularly important in many reviews (e.g. Uvarov 1977; Chappell and Whitman 1990), and seems to be echoed by the research presented in this paper.
In this study there were few tussocks of tall vegetation on the mown hay plots in comparison to the uncut control where there was an abundance of shady, tall vegetation in which grasshoppers could take refuge to avoid overheating. In short-grassland habitats, grasshoppers may overheat and have a higher susceptibility to water loss and desiccation than in taller grassland where humidity may be higher and temperatures lower (Haskell 1958). Both C. albomarginatus and C. parallelus are fairly large grasshoppers with body lengths between 17–21 mm and 16–22 mm for adult females respectively (Marshall and Haes 1988). This large body size (and therefore surface area) could make it difficult for both species to cool down quickly in hot environments, making behavioural thermoregulation their only option for survival.
Definite responses to cutting were observed for C. albomarginatus and C. parallelus in both years of the study, the adult abundances of which were significantly lower on the cut hay treatment (Fig. 1). We partly attribute these differences to the excessively hot microclimate of the mown hay plots leading to dispersal of adult individuals, which have a high degree of mobility (Gardiner and Hill 2004), to surrounding tall grassland habitats (which may include the uncut control replicates) in search of shade, although such movements may have been small-scale over a period of several days in this study and were barely detectable using quadrat sampling (Fig. 1). Movements of C. parallelus from short to tall grassland habitats have been noted in other studies on the Writtle College Estate and movements can be highly directional and possibly instigated by grasshoppers ‘seeing’ favourable habitats over short distances (<10 m) and orienting and moving towards them (Gardiner and Hill 2004).
In conclusion, it would seem that the management of hay meadows may affect grasshoppers in two ways: through direct mortality caused by the cutting machinery (Wagner 2004; Gardiner and Hill 2006b) and by creating a thermally hostile environment with excessively high temperatures (often >44°C), which may necessitate the movement of grasshoppers to taller vegetation with a higher occurrence of shade habitat (e.g. tussocks) and lower sward temperatures which are nearer the ‘optimum’ temperature for growth and development (35–40°C). These responses to management may be the causative factors behind the low abundance of grasshoppers in short farmland swards <10 cm in height (Gardiner et al. 2002). However, it is possible to alter the timing of grassland cutting so that the main period of grasshopper activity is avoided; we would suggest cutting the sward in September to minimise the effect on grasshoppers. If cutting has to be undertaken in June or July, then a system of rotational mowing may be the best option for grasshoppers, making sure to leave areas uncut every year as shelter for insects. Environmental Stewardship (ES) Scheme 6 m grass field margins can be cut with greater flexibility than those established under the CSS, for example, the land manager is able to cut the 3 m next to the crop edge annually after mid July but can only cut the inner 3 m to control woody growth not more than once in 5 years (Rural Development Service 2005). This diverse approach to cutting field margins should be beneficial to Orthoptera (Gardiner 2006), although the abundance of grasshoppers may also depend on the location of the grass strips, those on the exposed, windward side of hedgerows may not be particularly favourable whatever the management regime adopted (Gardiner and Dover 2008).
It is acknowledged that this study was only small-scale (2 replicates for each treatment) and further research is needed into the effect of microclimate on grasshoppers in a variety of managed habitats with a larger number of replicates. Mark, release, recapture studies may also more accurately determine the movements of grasshoppers from short, cut vegetation to longer uncut grassland where the microclimate is cooler, as the quadrat sampling used in this study made it difficult to detect movements between plots.
References
Chappell MA, Whitman DW (1990) Grasshopper thermoregulation. In: Chapman RF (ed) Biology of Grasshoppers. Wiley Interscience, New York, pp 143–172
Clarke EJ (1948) Studies in the ecology of British grasshoppers. Trans R Entomol Soc Lond 99:173–222
Evans AD, Smith KW, Buckingham DL, Evans J (1997) Seasonal variation in breeding performance and nestling diet of Cirl Buntings Emberiza cirlus in England. Bird Study 44:66–79
Gardiner T (2006) The impact of grassland management on Orthoptera populations in the UK. Unpublished PhD thesis, University of Essex
Gardiner T, Dover J (2008) Is microclimate important for Orthoptera in open landscapes? J Insect Conserv. doi:10.1007/s10841-007-9104-7
Gardiner T, Hill J (2003) Are there any grasshoppers on farmland? Antenna 27:115–116
Gardiner T, Hill J (2004) Directional dispersal patterns of Chorthippus parallelus (Orthoptera: Acrididae) in patches of grazed pastures. J Orthoptera Res 13:135–141
Gardiner T, Hill J (2005a) A study of grasshopper populations in Countryside Stewardship Scheme field margins in Essex. Br J Entomol Nat Hist 18:73–80
Gardiner T, Hill J (2005b) Behavioural observations of Chorthippus parallelus (Orthoptera: Acrididae) adults in managed grassland. Br J Entomol Nat Hist 18:1–8
Gardiner T, Hill J (2006a) A comparison of three sampling techniques used to estimate the population density and assemblage diversity of Orthoptera. J Orthoptera Res 15:45–51
Gardiner T, Hill J (2006b) Mortality of Orthoptera caused by mechanised mowing of grassland. Br J Entomol Nat Hist 19:38–40
Gardiner T, Pye M (2001) Habitats of Orthoptera on the Writtle College Estate in Essex. Bull Amateur Entomol Soc 60:154–160
Gardiner T, Pye M, Field R, Hill J (2002) The influence of sward height and vegetation composition in determining the habitat preferences of three Chorthippus species (Orthoptera: Acrididae) in Chelmsford, Essex, UK. J Orthoptera Res 11:207–213
Gardiner T, Hill J, Chesmore D (2005) Review of the methods frequently used to estimate the abundance of Orthoptera in grassland ecosystems. J Insect Conserv 9:151–173
Hamilton WJ (1975) Coloration and its thermal consequences for diurnal desert insects. In: Hadley NF (ed) Environmental physiology of desert organisms. Dowden, Hutchinson and Ross, London, pp 67–89
Haskell PT (1958) The relation of stridulation behaviour to ecology in certain grasshoppers. Insectes Sociaux 5:287–298
Marshall JA, Haes ECM (1988) Grasshoppers and allied insects of Great Britain and Ireland. Harley Books, Colchester
Pollard E, Yates TJ (1993) Monitoring butterflies for ecology and conservation. Chapman and Hall, London
Richards OW, Waloff N (1954) Studies on the biology and population dynamics of British grasshoppers. Anti-Locust Bull 17:1–182
Rural Development Service (2005) Entry Level Stewardship handbook. DEFRA, London
Smith H, Feber RE, Johnson PJ, McCallum K, Plesner Jensen S, Younes M, Macdonald DW (1993) English nature science no. 18: the conservation management of arable field margins. English Nature, Peterborough
SPSS (2006) SPSS version 15. SPSS, Chicago
Stewart KEJ, Bourn NAD, Thomas JA (2001) An evaluation of three quick methods commonly used to assess sward height in ecology. J Appl Ecol 38:1148–1154
Uvarov BP (1966) Grasshoppers and locusts, vol 1. Cambridge University Press, Cambridge
Uvarov BP (1977) Grasshoppers and locusts, vol 2. Centre for Overseas Pest Research, London
Wagner C (2004) Passive dispersal of Metrioptera bicolor (Phillipi 1830) (Orthopteroidea: Ensifera: Tettigoniidae) by transfer of hay. J Insect Conserv 8:287–296
Willott SJ (1992) The thermal ecology and population dynamics of grasshoppers in relation to grazing on a Breckland grass heath. Unpublished PhD thesis, University of East Anglia
Willott SJ (1997) Thermoregulation in four species of British grasshoppers (Orthoptera: Acrididae). Funct Ecol 11:705–713
Young AJ (1979) Studies of the ecology and behaviour of the grasshopper Chorthippus brunneus (Thunb.). Unpublished PhD thesis, University of Wales
Acknowledgements
We wish to thank Writtle College for funding and supporting this project and to members of staff (especially James Stack and Martin Heywood) who helped with the management of the hay plots and collection of data. TG’s PhD supervisor, Dr. Julian Hill, was also extremely helpful in formulating experimental design and choice of statistics.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gardiner, T., Hassall, M. Does microclimate affect grasshopper populations after cutting of hay in improved grassland?. J Insect Conserv 13, 97–102 (2009). https://doi.org/10.1007/s10841-007-9129-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-007-9129-y