Abstract
Purpose
In this study, we evaluated the feasibility, efficacy, and safety of radiofrequency ablation of paroxysmal atrial fibrillation (AF) with the use of an ablation catheter only (non-CMC group) by retrospectively comparing its procedural success and recurrence rates at 1 year to ablation performed with the help of a circular mapping catheter (CMC group).
Methods
We compared the success and recurrence rates between 226 patients and 251 patients who underwent index ablation with and without the use of CMC, respectively.
Results
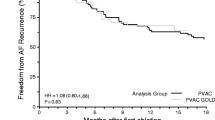
Freedom from recurrence was defined as a 1-year absence of AF/atrial tachycardia (AT) episodes > 30 s, beyond the 3-month blanking period. There was no significant difference between the number of pulmonary vein isolations, recurrence rate of AF/AT, and the use of antiarrhythmic drugs after 1 year of ablation. The procedure and fluoroscopy times were lower in the non-CMC group compared with the CMC group (106 ± 33 vs. 125 ± 32 min, p < 0.0001; 2.2 ± 1.9 vs. 2.7 ± 2.3 min, p = 0.0002, respectively).
Conclusions
Pulmonary vein isolation without the use of a CMC is feasible; moreover, the material costs, procedure time, and radiation exposure were reduced compared with the CMC group. Freedom of recurrence was similar between groups. Optimized use of 3D electro-anatomical mapping systems could reduce the radiation exposure for both the patient and physician.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Pulmonary vein isolation (PVI) is the cornerstone for paroxysmal atrial fibrillation (AF) ablation [1, 2]. This procedure can be performed using radiofrequency ablation combined with a three-dimensional (3D) navigation system in order to improve the outcomes and reduce radiation exposure to the patient [3]. The cost of this procedure remains high due to the use of a navigation ablation catheter with contact force sensor and a circular mapping catheter (CMC) to confirm the PVI [2].
Validation of the PVI could be performed by the ablation catheter. Moreover, the site of reconnection is easily located by simple mapping and/or pacing manoeuvres [4].
In this study, we aimed to evaluate the feasibility, procedural safety, clinical success, and recurrence rates of paroxysmal AF ablation with 3D navigation system and contact force-guided radiofrequency ablation catheter but without the use of CMC.
2 Methods
2.1 Patients characteristics
In this single-centre study, we evaluated the feasibility of PVI without the use of a CMC catheter. Moreover, we compared the procedural success and recurrence rates after index paroxysmal AF ablation with and without the use of CMC in two successive groups of patients with clinical follow-up. The first group (CMC group) included 226 patients who underwent ablation with the use of a CMC between November 2013 and March 2016. The second group (non-CMC group) included 251 patients who underwent ablation without the use of a CMC between March 2016 and July 2018.
The study protocol was approved by the local institutional ethics committee (internal local ethic committee, submission number 168/2019).
2.2 Ablation procedure
Pulmonary vein isolation (PVI) was performed by 2 operators with the patients under general anaesthesia.
A 3D electro-anatomical mapping system (CARTO®, Biosense Webster Inc., Diamond Bar, CA, USA), a contact force-guided radiofrequency ablation catheter (RF-CF) (ThermoCool® SmartTouch™ Catheter, Biosense Webster Inc.; electrode spacing, 1–6–2 mm), and a duodecapolar catheter (Ismus® Catheter, Biosense Webster Inc.; electrode spacing, 2–12–2 mm) were used in both groups of patients. Additionally, a decapolar CMC (Lasso® 2515 Variable Mapping Catheter, Biosense Webster Inc.; electrode spacing, 8 mm) was used in the CMC group.
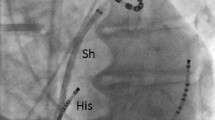
After right femoral vein puncture and placement of the duodecapolar catheter in the coronary sinus (CS), transseptal puncture under transesophageal echocardiography guidance (two punctures in the CMC group and one in the non-CMC group) was performed using 8.5 French sheath(s) (SWART, SL0, St. Jude Medical, Minnetonka, MN, USA) and a transseptal needle (HeartSpan®, 50°, Biosense Webster Inc.). Fluoroscopy was discontinued following the introduction of the ablation catheter into the left atrium. A 3D anatomical model of the left atrium was reconstructed using the ablation catheter in both groups. After that, in the CMC group, the CMC was placed into the left atrium using non-fluoroscopic visualization.
Precise delimitation of the ostia of the pulmonary veins was performed using the “swing-fall” technique. As we pulled the catheter back from the veins into the atrium, we inserted a “swing-fall” tag precisely where the catheter vector “swings” and/or the tip of the catheter abruptly “falls” into the atrium (Fig. 1).
The “Swing-Fall” technique. The catheter is pulled back from the PV into the atrium. A “Swing-fall” tag (white tag) is inserted at the precise location where the vector “swings” and the tip of the catheter “falls” into the left atrium to determine the venoatrial delimitation. In panel a (anterior part of the LSPV), the swing-fall tag is inserted on the fourth image; in the b (anterior septum) and c (posterior wall) panels, the swing-fall tag is inserted on the third image and deleted on the fourth image to demonstrate the “swing” of the vector. PV, pulmonary veins; LSPV, left superior pulmonary veins
Before ablation, pulmonary vein potential (PVP) was identified using a CMC in the first group and an ablation catheter in the second group. PVP was annotated with yellow tag points in the second group (Fig. 2).
Posterior view of the right pulmonary veins. Multiples PVP tags (yellow tags) are placed before the isolation of the veins as illustrated by the local electrogram in two of these tags. After anatomical circular ablation around the pulmonary veins (red tags), isolation is confirmed by the absence of residual PVP and the absence of capture of the atrium by venous stimulation or by dissociated potentials (heavy grey tags or scar tags). The white tags correspond to the “swing-fall” tags. PVI, pulmonary vein isolation; RSPV, right superior pulmonary vein; RIPV, right inferior pulmonary vein
PVI was performed with a point-by-point approach to anatomically enclose the pulmonary veins (Fig. 3) with an interlesion distance (ILD) < 4 mm (diameter of the lesion tag was 4 mm with a contiguous tag) and a force time integral index (FTI) > 200 g s on the posterior wall (25 W) and > 300 g s on the anterior wall (35 W).
Posterior view of the left atrium with the CT scan shell in light brown, the reconstructed shape using the ablation catheter is grey, the “swing-fall” tag is white, and the ablation tag is red. The merging of the CT-scan shell and the reconstructed shape appears to be perfect. The “swing-fall” tags correspond at the venoatrial delimitation of both the shell and shape. To avoid ablation into the veins, the ablations tags follow an imaginary line between successive “swing-fall” tags. This imaginary line is always stretched to the atrium to draw a natural curve between the “swing-fall” tags (as demonstrated in the bottom of the right inferior pulmonary veins). CT, computed tomography
In case of dislocation, a new RF application was applied with a target FTI based on the user’s clinical judgement according to the FTI of the previous application, changes in power, contact force, catheter stability, impedance drop, electrogram attenuation, and lesion contiguity. All patients enrolled underwent ablation with the VisiTag™ module.
PVI was validated by the absence of pulmonary vein potential (entrance block) and the absence of venoatrial capture or by dissociated potentials (exit block). In the first group of patients, the validation was performed using CMC; in the second group, the ablation catheter was used to validate the previously tagged PVP (Fig. 2).
In the absence of PVI after the first anatomical circular ablation, the gap was located based on the earliest PVP identified (Fig. 4) [4]. This manoeuvre was done using the sinus rhythm or by pacing on the duodecapolar catheter placed in CS; in case of multiples gaps, the manoeuvre had to be repeated until the PV was isolated. In the CMC group, we put the CMC into the veins and identified the gap as the earliest activation inside the circumferential ablation around the PV. In the non-CMC group, the ablation catheter was placed within the circumferential ablation line. The localization of the conduction gap was identified at the earliest recorded PVP. If necessary, the venoatrial conduction time, measured between the stimulation on the ablation catheter (placed within the circumferential ablation line) and the duodecapolar catheter, was also used to guide additional touch-up ablation until PVI.
Representation of the techniques used to determine the localization of a conduction gap in the CMC (a) and non-CMC group (b, c). (a) CMC is placed inside the ablation line; the gap is identified at the shorter atrio-PV activation time (during sinus rhythm or pacing on the Ismus® placed in the CS). (b) Localization of the shorter atrio-PV activation time is determined by moving the ablation catheter inside the ablation line (during SR or pacing on the Ismus® placed in the CS). (c) Pacing is performed from the ablation catheter placed inside the ablation line; the shortest time from the captured PV tissue to the CS reference signal denotes the localization of the conduction gap. The square symbol represents a pacing point; the star represents a recording point. CMC, circular mapping catheter; CS, coronary sinus; PV, pulmonary vein; SR, sinus rhythm
2.3 Follow-up
All the patients at our institution underwent follow-up at 3, 6, and 12 months; referred patients were encouraged to apply the same follow-up; additionally, all patients were asked to contact their cardiologist for any symptoms suggestive of AF/atrial tachycardia recurrence. At each visit, a 12-lead ECG and an assessment of current antiarrhythmic medication and symptoms of atrial fibrillation, such as palpitations and loss of exercise capacity, were performed. Depending on the patient’s cardiologist, Holter monitoring (24 h or 7 days) was performed either systematically or, at a minimum, in cases with any symptoms suggestive of symptomatic arrhythmia recurrence.
After a 3-month blanking period, anticoagulation was continued according to the stroke risk; antiarrhythmic medication was continued at the discretion of the treating physician. Freedom from recurrence was defined as a 1-year absence of AF/atrial tachycardia (AF/AT) episodes > 30 s, after the 3-month blanking period [2].
2.4 Statistical analysis
The statistical analysis results are expressed as mean ± standard deviation. The comparison between variables was performed using the Mann-Whitney test and the chi-squared test, as appropriate. A p value < 0.05 was considered statistically significant.
3 Results
3.1 Patient characteristics
All patients had paroxysmal AF, and none had previously undergone AF ablation. There were no differences in the baseline clinical characteristics between the two groups (Table 1).
3.2 Procedural characteristics
The procedural characteristics are summarized in Table 2. There was no significant difference in the number of PVI obtained. The procedure time (106 ± 33 vs. 125 ± 32 min, p < 0.0001), fluoroscopy time (132 ± 119 vs. 161 ± 137 s, p = 0.0002), and dose-area product (1531 ± 2481 vs. 1684 ± 2045mGy cm2, p = 0.03) were lower in the non-CMC group compared with the CMC group.
3.3 One-year follow-up
The results are summarized in Table 2. The percentage of follow-up and Holter monitoring were comparable between the groups during the first year of follow-up. The rate of freedom from AT/AF and the percentage of patients on ADT at 1 year were similar between groups.
3.4 Safety of PVI
In the CMC group, there was one case each of an arteriovenous fistula (conservative treatment), phrenic nerve palsy, and a pericardial tamponade that required surgical drainage. In the non-CMC group, there was one case each of an oesophageal tear with symptoms beginning on the day after ablation secondary to the transoesophageal echocardiography (treated by an endoscopic oesophageal prosthesis for 2 months and antibiotics), a femoral vein thrombosis secondary to hematoma, probable transitory ischemic attack, an arteriovenous fistula that was treated surgically, and a pseudoaneurysm that was treated by radiological embolization (Table 2).
4 Discussion
4.1 Main findings
This study evaluated the feasibility of PVI without the use of a CMC catheter and retrospectively compared its efficiency and safety to PVI with the use of a CMC. The acute success rate of PVI and the freedom from AF/AT were similar between the groups at 1 year. This success rate, which was close to 90%, was similar to that reported by several studies that used paroxysmal AF ablation with contact force sensing technology [5], FTI [6], and AI [7, 8]. Validating the PVI and/or localizing the gap without the use of the CMC was not time-consuming; moreover, there was a decrease in procedural and radiation durations in the non-CMC group compared with the CMC group, probably due to the absence of the second transseptal puncture and mobilization of the CMC (Table 2). Furthermore, these reductions were drastically lower than those previously reported (Table 3) [5, 6, 8]. This highlights the need for a 3D electro-anatomical mapping system protocol to drastically reduce (7–27-fold) the radiation exposure for both the patient and physician (2.2 ± 1.9 min per procedure and dose-area product of 1531 ± 2481 mGy cm2 in the non-CMC group; Table 3).
We tagged the ablation site with a contiguous 4-mm point, and avoided any visual gaps [9], to prevent PV reconnection. Our choice to use 4-mm point rather than 6-mm point was validated by subsequent studies, which highlighted the importance of avoiding high interlesion distances [7], especially those greater than 5 mm [10].
4.2 The “swing-fall” technique
The localization of these ablation points is crucial for the prevention of complications, such as pulmonary vein stenosis, and to maximize the effectiveness of the procedure [11]. Currently, three main approaches are used for PVI: ostial isolation, wide antral PVI (classically performed ≥ 1.5 cm away from PV ostium) [11], and nephroid-shaped towards the carina [12]. Each of these approaches relies on the delineation of ostium of the PV; however, to our knowledge, no precise technique has been described that defines the electro-anatomical location of the ostium using a 3D navigation system. The “swing-fall” technique allows for the precise delimitation of these venoatrial junctions (ostia) and could standardize localization in RF applications in future studies. In this study, the ablation site was on the ostium for the anterior part of the veins, and an area between the ostium and the wide antral circumferential ablation for the posterior wall (like the “CLOSE” protocol [12] but without targeting the carina).
4.3 Study limitations
This was a retrospective study and several limitations must be underlined. First, the ablations were all performed by the same two operators from 2013 to 2018 and the two groups were consecutive; therefore, the improved skills of the operators could also explain, at least partially, the reduction in the procedure and fluoroscopy times. Nevertheless, the same radiofrequency ablation parameters and technologies were applied in both groups.
Second, the recurrence of AF was evaluated by clinical follow-up (patient interviews, ECG, and Holter ECG), which can underestimate AF recurrence. However, the evaluations and the number of Holter monitoring were similar between groups. Despite methodological limitations, previous studies on feasibility used this methodology for a first evaluation of a new ablation protocol [13,14,15]. Third, with a one-catheter approach, the 3D anatomical estimated volume might be suboptimal and underestimated (Fig. 3). Fourth, due to the retrospective design of this study, a more in-depth analysis of the target value for radiofrequency delivery was not possible. The absence of AI at the time of the ablation did not allow for the evaluation of the ablation line contiguity index (ALCI) proposed by El Haddad et al. [7]. Fifth, the risk of stroke or bleeding might be expected to be lower by reducing the catheter dwell time in the LA, but it was not significantly reduced in this study. Nevertheless, stroke and bleeding events were very low, and the intraprocedural anticoagulation could not be estimated in this study (which depends on the patient’s anticoagulation with a vitamin K antagonist or a direct oral anticoagulant, on the continuation or interruption of this oral anticoagulation during the ablation procedure, and on the characteristics of the patient).
Prospective studies are required to evaluate the efficiency of our methods and address the study limitations.
Additionally, the “swing-fall” technique must be evaluated to validate the reproducibility of localizing the swing-fall tag by different operators.
5 Conclusion
PVI isolation without the use of a CMC is feasible, effective, and safe. There is a reduction in procedural and fluoroscopy times with a similar success rate at 1 year as that of PVI with the use of CMC. Depending of the reimbursement model of the country, the cost of the procedure can be reduced to various degrees. Furthermore, this method may drastically reduce patient and physician radiation exposure in routine clinical ablation procedures. Moreover, this study suggests that the use of the “swing-fall” technique could standardize the anatomical localization of the RF application used to encircle pulmonary veins.
References
Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893–962.
Calkins H, Hindricks G, Cappato R, Kim YH, Saad EB, Aguinaga L, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017;14(10):e275–444.
Marini M, Martin M, Ravanelli D, Del Greco M, Quintarelli S, Guarracini F, et al. Extensive use of 3D nonfluoroscopic mapping systems for reducing radiation exposure during catheter ablation procedures: an analysis of 10 years of activity. Biomed Res Int. 2019;2019:4217076.
Martins RP, Galand V, Behar N, Daubert JC, Mabo P, Leclercq C, et al. Localization of residual conduction gaps after wide antral circumferential ablation of pulmonary veins. JACC Clin Electrophysiol. 2019;5(7):753–65.
Marijon E, Fazaa S, Narayanan K, Guy-Moyat B, Bouzeman A, Providencia R, et al. Real-time contact force sensing for pulmonary vein isolation in the setting of paroxysmal atrial fibrillation: procedural and 1-year results. J Cardiovasc Electrophysiol. 2014;25(2):130–7.
Andrade JG, Monir G, Pollak SJ, Khairy P, Dubuc M, Roy D, et al. Pulmonary vein isolation using “contact force” ablation: the effect on dormant conduction and long-term freedom from recurrent atrial fibrillation—a prospective study. Heart Rhythm. 2014;11:1919–24.
El Haddad M, Taghji P, Phlips T, Wolf M, Demolder A, Choudhury R, et al. Determinants of acute and late pulmonary vein reconnection in contact force-guided pulmonary vein isolation: identifying the weakest link in the ablation chain. Circ Arrhythm Electrophysiol. 2017;10(4):e004867.
Taghji P, El Haddad M, Phlips T, Wolf M, Knecht S, Vandekerckhove Y, et al. Evaluation of a strategy aiming to enclose the pulmonary veins with contiguous and optimized radiofrequency lesions in paroxysmal atrial fibrillation: a pilot study. JACC Clin Electrophysiol. 2018;4(1):99–108.
Miller MA, D’avila A, Dukkipati SR, Koruth JS, Viles-Gonzalez J, Napolitano C, et al. Acute electrical isolation is a necessary but insufficient endpoint for achieving durable PV isolation: the importance of closing the visual gap. Europace. 2012;14(5):653–60.
Park CI, Lehrmann H, Keyl C, Weber R, Schiebeling J, Allgeier J, et al. Mechanisms of pulmonary vein reconnection after radiofrequency ablation of atrial fibrillation: the deterministic role of contact force and interlesion distance. J Cardiovasc Electrophysiol. 2014;25(7):701–8.
Proietti R, Santangeli P, Di Biase L, Joza J, Bernier ML, Wang Y, et al. Comparative effectiveness of wide antral versus ostial pulmonary vein isolation: a systematic review and meta-analysis. Circ Arrhythm Electrophysiol. 2014;7(1):39–45.
Duytschaever M, O’neill M, Martinek M. Increasing the single-procedure success rate of pulmonary vein isolation. Arrhythm Electrophysiol Rev. 2017;6(4):217–21.
Reichlin T, Lane C, Nagashima K, Nof E, Chopra N, Ng J, et al. Feasibility, efficacy, and safety of radiofrequency ablation of atrial fibrillation guided by monitoring of the initial impedance decrease as a surrogate of catheter contact. J Cardiovasc Electrophysiol. 2015;26(4):390–6.
Scaglione M, Caponi D, Anselmino M, Di Clemente F, Blandino A, Ferraris F, et al. Pulmonary vein isolation with a new multipolar irrigated radiofrequency ablation catheter (nMARQ™): feasibility, acute and short-term efficacy, safety, and impact on postablation silent cerebral ischemia. J Cardiovasc Electrophysiol. 2014;25(12):1299–305.
Swaans MJ, Post MC, Rensing BJ, Boersma LV. Ablation for atrial fibrillation in combination with left atrial appendage closure: first results of a feasibility study. J Am Heart Assoc. 2012;1(5):e002212.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethic approval
The study protocol was approved by the local institutional ethics committee (internal local ethic committee, submission number 168/2019).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xhaët, O., Deceuninck, O., Robaye, B. et al. A circular mapping catheter is not mandatory for isolating pulmonary veins during paroxysmal atrial fibrillation ablation with radiofrequency. J Interv Card Electrophysiol 62, 285–292 (2021). https://doi.org/10.1007/s10840-020-00895-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-020-00895-8