Abstract
Sarcothalia lanceata is a broad-bladed, New Zealand red alga. The tetrasporic life stage contains a lambda-carrageenan that has a strong potential for commercial utilisation. The male and female gametophytic life stages contain kappa-II carrageenan that also has commercial potential. However, fundamental information on the growth and variation in carrageenan content of this species in the wild is necessary to underpin possible aquaculture of this species. Therefore, growth in blade length and width was assessed for male and female gametophytes, and tetrasporophytes in two S. lanceata populations in New Zealand’s South Island, and monthly seawater nutrient and tissue nutrients, and carrageenan constituent sugar levels were analysed. Blade length did not vary significantly over time or between life history phases; however, blade width varied significantly for both factors with most temporal variation occurring in male blades. Analysis of seawater and tissue nutrient levels suggested that these populations are nutrient limited year-round. Constituent sugars varied temporally and between life history phases in most cases. Relative 3,6-anhydrogalactitol peracetate (for the males and females) and galactitol peracetate levels (all life stages) were higher in winter than in summer, while glucitol peracetate and xylitol peracetate levels were generally lower in winter than summer. The distinctive morphologies of the male and female plants of this species mean there is potential to cultivate and then separately harvest male and female plants in addition to tetrasporophytic plants. Male blades contain more 3,6-anhydrogalactitol peracetate and galactitol peracetate than female blades, and their morphology may make them less prone to breakage in an aquaculture situation. Experimentation on spore release and growth and the potential effects of nutrient manipulation on early stages are suggested as the direction of future research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carrageenans are complex polysaccharides that are widely utilised as gelling, stabilising and thickening agents in various industries, particularly the dairy food industry (Stanley 1990; Heertje 1993). They are also of interest as drug delivery agents (Li et al. 2014) and for their anti-viral properties (Ahmadi et al. 2015). The lambda-type carrageenan isolated from tetrasporophytes of New Zealand red seaweed Sarcothalia atropurpurea (as Gigartina atropurpurea) had the best anti-viral activity of the four species tested by Harden et al. (2009). Red seaweeds in the family Gigartinaceae are a rich source of these sulfated galactans and, globally, large quantities of red seaweeds are either cultivated or harvested from the wild as raw materials for carrageenan production because carrageenans are too complex to synthesise. Market demand for carrageenan-producing seaweeds has increased in recent years (Bixler 1996; Santelices 1996; Avila et al. 1999; Bixler and Porse 2011; Campbell and Hotchkiss 2017), which has resulted in overexploitation and, in some cases, collapse of natural supplies (e.g. Norambuena 1996; Rebours et al. 2014). Cultivation of Gigartinacean seaweeds is increasingly recognised as an important alternative source of commercial supply. Considerable research has been undertaken into aquaculture of key species, such as Gigartina skottsbergii and Sarcothalia crispata, over the last two decades (e.g. Avila et al. 1999; Romo et al. 2001; Buschmann et al. 2004; Romo et al. 2006; Hernández-González et al. 2007) to establish sustainable supplies of these species to meet market demand.
Sarcothalia lanceata (J. Agardh) Hommersand (Gigartinaceae, Rhodophyta) is one of several Gigartinacean seaweeds endemic to New Zealand. The tetrasporophytic life phase of this species contains lambda-type carrageenans while the male and female gametophytic life stages produce complex hybrid structures known as kappa-II type carrageenans. These hybrid molecules contain sections of kappa- and iota-carrageenans along with their biochemical pre-cursors mu- and nu-carrageenans. Details of the yields of carrageenan and the proportions of kappa-, mu-carrageenans extracted from gametophytic plants and lambda-carrageenan extracted from tetrasporophytic plants of this species were first reported by Pickmere et al. (1973, 1975) and Parsons et al. (1977). Significant advances in analytical techniques since then have enabled more detailed analyses of the carrageenans present in this species. Thus, a polysaccharide extract from purely tetrasporophytic S. lanceata has been characterised in detail both chemically (Falshaw and Furneaux 1998) and physically (Running et al. 2012). Its chemical structure corresponds very closely to the idealised structure of lambda-carrageenan (Falshaw and Furneaux 1998) in that it contains galactosyl units but no 3,6-anhydrogalactosyl units. Lambda-type carrageenans have traditionally been considered to produce viscous aqueous solutions that do not gel. However, the lambda-carrageenan from the tetrasporophytic phase of S. lanceata gels in the presence of iron (III) ions (Running et al. 2012). This novel finding creates added potential for commercial production and utilisation of S. lanceata. Aquaculture of Gigartinacean species has the added benefit of providing separate sources of gametophytic and tetrasporophytic material, and therefore separate sources of kappa-II type and lambda-type carrageenans. Yield or biomass is an important factor to consider when evaluating the potential of growing a seaweed by aquaculture. S. lanceata plants have broad-blades, which is an agronomically beneficial trait (Santelices 1999) since large amounts of biomass can be generated from a fewer plants than for smaller or filamentous species. Unusually for a gigartinacean seaweed, male and female gametophytes of this species can easily be distinguished in the field. Both are generally broader than tetrasporophytic blades; however, the female blades are thick and often heavily papillate but with a smooth margin, while the male blades are wider, thinner, and smooth, and carry few, or in most cases, no papillae (Nelson 2013; illustrated in Neill et al. 2016).
Sarcothalia lanceata inhabits the lower intertidal and upper subtidal of wave-exposed rocky shores between 41.5° and 51° S, specifically on the South Island of New Zealand, the Chatham Islands, Stewart Island, and several sub-Antarctic islands (Adams 1994, as Gigartina lanceata). Harvest of S. lanceata from the wild is limited by a lack of knowledge of the size of standing stocks and a moratorium on the issue of permits for collecting Gigartinaceae commercially. Consequently, aquaculture is considered to be the only practical method of obtaining large quantities of this species. McNeill et al. (2003) demonstrated successful cultivation of mature plants of both female gametophytic and tetrasporophytic plants of a related New Zealand endemic species, Sarcothalia atropurpurea, in open water so aquaculture of S. lanceata could be possible. More recently, experimental production of mature gametophytic S. atropurpurea plants has been achieved by releasing and cultivating spores (McNeill and Falshaw 2017).
The success of establishing the cultivation of a new aquaculture species relies on fundamental knowledge of its biology, including seasonal patterns of growth and carrageenan composition (Santelices 1999; Westermeier et al. 1999; Charrier et al. 2017) but for many species, this information is lacking (Buschmann et al. 2001). For growth, nitrogen availability is one of the most important regulators in seaweeds (Hurd et al. 2014). However, timing of growth is often uncoupled from that of nutrient supply due to the ability of many species to accumulate nutrients in their tissues (Atkinson and Smith 1983), and this phenomenon makes it difficult to directly correlate growth with ambient seawater nutrients (Rosenberg and Ramus 1982). Consequently, algal tissue analysis is a necessary part of any investigation into the seasonality of growth rates. The ratio of tissue carbon to tissue nitrogen (C:N ratio) is the most commonly used method for investigating the relationship between seaweeds and nutrient limitation (Hurd et al. 2014). Generally, greater seasonal variability occurs in tissue nitrogen content than in tissue carbon content (McLachlan and Bird 1986).
Seasonal patterns of carrageenan quantity vary between species, with some studies reporting significant temporal variation in carrageenan yield and properties (e.g., Fuller and Mathieson 1972; Friedlander and Lipkin 1982; Cosson et al. 1990; Breden and Bird 1994; Amimi et al. 2007; García Tasende et al. 2012; Véliz et al. 2017) while others report no seasonal differences (Pickmere et al. 1975; Piriz and Cerezo 1991; Chopin and Floc’h 1992). Seasonal variation in the composition of carrageenan present has been reported in some species. For example, 3,6-anhydrogalactose levels were shown to be higher in summer than winter in Eucheuma spp. (Dawes et al. 1974) and Hypnea cornuta (Friedlander and Zelikovitch 1984), but higher in winter than in summer for Gigartina pistillata (Amimi et al. 2007), Hypnea musciformis (Friedlander and Zelikovitch 1984), and Eucheuma uncinatum (Zertuche-González et al. 1993). In contrast, no seasonal variation was found for Neoagardhiella baileyi, Gigartina skottsbergii and Chondrus crispus (Asare 1980; Piriz and Cerezo 1991; Chopin et al. 1999).
The availability of essential nutrients (nitrogen and phosphorous) in seawater is thought to have a significant influence on carrageenan yields (Fuller and Mathieson 1972) and in a recent review, Véliz et al. (2017) found that nitrate and phosphate were two of the top four abiotic factors affecting carrageenan contents, along with light and temperature. An inverse relationship between carrageenan yield and nitrogen level was first established by Neish et al. (1977). This relationship became known as the ‘Neish effect’, whereby higher carrageenan yields in C. crispus occurred in plants grown in nitrogen deficient seawater. A similar relationship was later established between phosphorous and carrageenans, known as the ‘Chopin effect’ (e.g. Chopin et al. 1995). These relationships between nutrients and carageenans have also been assessed in other studies of Chondrus crispus (Chopin and Wagey 1999; Chopin et al. 1995; García Tasende et al. 2012) and Gigartina teedii (Zinoun et al. 1993).
The population size and structure of Sarcothalia lanceata at two sites in New Zealand’s South Island were assessed with a view to potential wild harvest (Neill et al. 2016). Here, we investigate growth and carrageenan production of S. lanceata to provide critical baseline information for the future aquaculture of this species. We address the following questions: (1) what is the temporal variation in growth and carrageenan content? and (2) are there relationships between nutrients and carrageenans, i.e. evidence of the Neish and Chopin effects?
Materials and methods
Two Sarcothalia lanceata populations on the Otago coastline were selected for this study: Murdering Beach (Whareakeake, 45.759978° S, 170.674067° E) and Purakaunui (45.739555° S, 170.629692° E), (Fig. 1). Both sites were situated in small crescent-shaped bays on rocky headlands at the eastern ends of the sandy surf beaches, and were subject to periods of sand inundation during this study (Neill et al. 2016). Sea surface temperatures at the study sites ranged between 9.5 and 17.0 °C over the study period.
Growth
In situ growth of selected S. lanceata plants was quantified by measuring changes in blade length and width over time. Plants were selected as follows: random paired co-ordinates were generated using a restricted randomisation method and used to select points within each of the pre-defined study areas. At each point, the nearest female, male and tetrasporophytic clumps (multiple blades arising from a basal crust) were located and tagged with uniquely colour-coded electrical ties. Initially six points (i.e. 18 clumps, three for each life history phase) were selected at each site, but early in the study tagged clumps were lost from Purakaunui and so the number of points there were increased to eight, giving a total of 24 tagged clumps in this study area. During monthly sampling, any lost tagged clumps or blades were replaced to maintain the required numbers of replicates.
Each month, the length and width of the three longest blades in each clump were measured to the nearest millimetre and these values were averaged to give a mean maximum blade length and width for each clump (n = 3). These means were then averaged across clumps to give a mean monthly value (n = 6–8, ± SE) for maximum blade length and width for each life history phase at each site. Blade length and width measurements were undertaken monthly between December 1999 and October 2000 at Murdering Beach (12 sampling dates), and March and October 2000 (9 sampling dates) at Purakaunui. Two sampling events fell within August 2000, and these events are referred to as August1 and August2, or A1 and A2.
In August 2000, the three largest blades in each clump were also individually tagged with uniquely colour-coded electrical ties. Between August and October 2000, the length of these three blades was recorded and relative elongation rates (expressed as percentage increase per day and used here to refer to both length and width increases) were calculated as follows:
where L1 is the length of a blade at Time 1 (T1), and L2 is the length of a blade at Time 2 (T2), approximately 1 month later.
Seawater and tissue nutrients
Each month, one S. lanceata blade was collected from each of three female, male and tetrasporophytic clumps at each field site. Blades were oven dried at 55 °C before being stored until analysis in re-sealable plastic bags containing silica gel. Blades were divided in half lengthways and one half of each blade was ground to a fine powder using a mortar and pestle. Ground samples were analysed for carbon and nitrogen content using a CHNS-O EA1108 Elemental Analyzer (Carlo Erba instruments). Percentage tissue carbon and percentage tissue nitrogen were then converted to a C:N ratio (atom:atom). Only samples from every second month were analysed.
Seawater samples (3 × 10 mL) were collected from each site each month. Samples were filtered through Whatman GF/C 47-mm filter paper and frozen for up to 3 months before being thawed, and analysed for nitrate (NO3−), phosphate (PO42+) and ammonium (NH4+) concentrations (μM) using a Lachat Quikchem 8000 Series Continuum Automated Ion Analyzer and Omnion FIA Software (Lachat Instruments, USA).
Carrageenan content
The composition of algal extracts can vary between different parts of the same plant (Dring 1982; Carrington et al. 2001). However, when carrageenans are extracted commercially, this within-plant variation is homogenised in the final product. On this basis, each blade in this study was dried and ground to produce a homogenous sample and no tests were carried out for within-blade variations in constituent sugar composition. The constituent sugar analysis method was used to break down all carrageenans and starch within the seaweed thalli into monosaccharide components, which enable detection as alditol acetate derivatives. The lambda-carrageenan from tetrasporic S. lanceata contains 3-linked-d-galactopyranosyl 2-sulfate units alternating with 4-linked-d-galactopyranosyl 2,6-disulfate units, which break down to two units of galactitol peracetate (Gal). Kappa-II type carrageenans (from gametophytic plants) contain hybrids of kappa-, iota-, mu- and nu-carrageenans. Kappa- and iota-carrageenan break down to produce one unit of 3,6-anhydrogalactitol peracetate (AnGal) and one unit of galactitol peracetate (Gal), whereas in mu- and nu-carrageenans produce two units of Gal. Both life phases also produce xylitol peracetate (Xyl), and glucitol peracetate (Glc) from floridean starch (Falshaw and Furneaux 1998; Stevenson and Furneaux 1991).
Three blades per life history phase, per site, per month (13 months for Murdering Beach and 11 months for Purakaunui) were collected for analysis, giving a total of 216 samples. Each blade was dried and ground using a domestic coffee grinder to produce a homogenous sample of fine powder. From each ground S. lanceata blade, a sample (~ 3 mg) was placed into an individual 13 × 100 mm Kimax tube with a Teflon-lined screw cap lid. The samples tended to spread up the sides of the tubes so were subjected to a crude in situ extraction before the reductive hydrolysis procedure was implemented. A portion of distilled water (0.6 mL) was added to each tube, and the tubes were heated in an aluminium heating block for 1 h at 85 °C. The tubes were cooled and then frozen before being freeze-dried in an Eyela FD-1 Freeze Drier (Tokyo Rikakikai Co. Ltd.). Constituent sugar composition of the extracts (still in the same tubes) was determined using a modified version of the reductive hydrolysis process described by Stevenson and Furneaux (1991).
Following Jol et al. (1999), tubes were heated for 30 min at 80 °C during the prehydrolysis stage of the procedure. Following total hydrolysis, residual water was removed from the solution by adding two separate 0.4 mL portions of acetonitrile (CH3CN) and evaporating twice, rather than a single 0.8 mL addition of CH3CN followed by evaporation. Myo-inositol was added as an internal standard to monitor the consistency of sample preparation (see Reani et al. 1998). Final extracts were analysed using gas liquid chromatography on a Hewlett Packard 5890 Series II Chromatograph with a Supelco 2330 15 m × 0.25 mm column, with isocratic programme set at 220 °C. A purified kappa-carrageenan was used as a control (~ 1 mg, Sigma Chemical Co.) and 18–22 algal samples were processed in each batch. Constituent sugars were identified by comparison of the retention times of extracts with the retention times of authentic standards, and are reported as normalised mole percentages.
The relative values of each constituent sugar in a sample were converted to absolute values. This was achieved by calculating the amount (weight in mg) of each constituent sugar in comparison with the known weight of the internal standard (myo-inositol) that was added to each sample. Results are reported as a percentage of the original weight of each powdered algal sample.
Statistical analysis
Statistical analyses were carried out using the SigmaStat 4.0 statistical software package. Variations in maximum blade length and blade width were assessed using two-way ANOVAs and the factors time and life history phase for each site. For each of the each of the four carrageenan derivatives, two-way ANOVAs were used to analyse variations in life history phase (female and male) and time (November 1999 through to October 2000). Levels of the four derivatives in tetrasporophytic blades were not statistically compared to levels in either female or male blades because the composition of the carrageenan in tetrasporophytic samples of S. lanceata is known to be different that of males and females and the levels of the four derivatives are proportional to each other. Instead, one-way ANOVAs were used to analyse the tetrasporophytic data.
Results
Growth
Blades of all life history phases were generally longer at Murdering Beach than at Purakaunui (Fig. 2a–c). Fluctuations in mean blade length over time were not statistically significant at Murdering Beach or Purakaunui (two-way ANOVAs, p = 0.941 and 0.138, respectively) (Fig. 2). Variations in blade length between life phase were not significantly different at Murdering Beach (p = 0.071) but were at Purakaunui (p < 0.001) with pairwise comparisons indicating that female blade lengths differed significantly from both male and tetrasporophytic blade lengths at this site (Fig. 2a, b).
Temporal variation in Sarcothalia lanceata mean blade length (a–c) and mean blade width (d–f). Two sampling events fell within August and are labelled A1 and A2. Data points represent the averages of the mean maximum blade length and mean maximum blade width for each clump (see text for details) ± SE, n = 1–6 at Murdering Beach (circles) and 1–8 at Purakaunui (triangles)
Blade widths (Fig. 2d–f) varied significantly over time at both sites (two-way ANOVAs, p = 0.039 for Murdering Beach and p = 0.040 for Purakaunui) and between life phases (p < 0.001 for both sites). For Murdering Beach, there was also a significant interaction between time and life phase (p < 0.001) indicating that the effects of time were not consistent between life history phases. Pairwise comparisons indicate that, at this site, temporal variation was only significant in males, and that male blade widths were significantly greater in September and October than in other months. Pairwise comparisons also show that female and male blade widths at Murdering Beach were significantly different from tetrasporophytic blade widths in most months. In blades from Purakaunui, male blade widths were significantly wider in October than March (p = 0.006), and female and male widths were significantly different from tetrasporophytic widths (p < 0.001 for both) in most months, and from each other (p = 0.034).
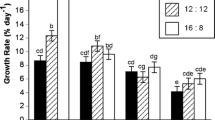
There was no easily discernible temporal pattern in the relative elongation rate (RER) of blade length at either site during the 4-month period of testing. In several months, the RER was near zero or showed negative values (Fig. 3a–c). Maximum positive RER in blade lengths at Murdering Beach were 0.04, 0.44 and 0.17% day−1 for females, males and tetrasporophytic blades, respectively. At Purakaunui, maximum positive RER values were 0.09, 0.54 and 0.74% day−1 for females, males and tetrasporophytic blades, respectively. Maximum negative RER values were −0.38, −0.48 and −0.04% day−1 for females, males and tetrasporophytic blades, respectively, for Murdering Beach. At Purakaunui, maximum negative RER was −0.30, −0.35 and −0.22% day−1 for females, males and tetrasporophytic blades, respectively (Fig. 3a–c).
Relative elongation rate in blade width was generally positive with maximum increases in width of 0.26, 0.77 and 0.24% day−1 for females, males and tetrasporophytic blades, respectively, at Murdering Beach and 0.13, 0.43 and 0.20% d−1 for females, males and tetrasporophytic blades, respectively, at Purakaunui (Fig. 3d–f). Negative RER in blade widths was limited to −0.03 in females, and −0.11% day−1 in male blades at Murdering Beach, and −0.01 in female blades and −0.10% day−1 in tetrasporophytic blades at Purakaunui.
Seawater and tissue nutrients
Levels of tissue carbon in S. lanceata blades showed little evidence of temporal variation in all three life history phases at either site (Fig. 4b, e). Values ranged between 23 and 30% in blades of all life phases from both Murdering Beach and Purakaunui. At both sites, female blades tended to have slightly higher levels of tissue carbon than male blades, which in turn had slightly higher levels of tissue carbon than tetrasporophytic blades. Tissue nitrogen levels exhibited a strong seasonal pattern of summer minima and winter maxima (Fig. 4c, f). Values for all three life history phases ranged between 0.8 and 1.8% at both sites. Carbon to nitrogen ratios of S. lanceata blades followed an inverse trend to that of tissue nitrogen, with summer maxima and winter minima (Fig. 4a, d).
Nitrate concentrations in seawater collected from Murdering Beach fluctuated between 0 and 4.7 μM, with maximum values occurring during winter (Fig. 5a). At Purakaunui, seawater nitrate concentrations were undetectable in summer (January and February), and peaked at 3.85 μM in winter (August) (Fig. 5a). For most of the study period, ammonium concentrations in seawater from Murdering Beach ranged between 0.18 and 2.00 μM, except in December 1999 when values peaked at 5.34 μM (Fig. 5b). Seawater collected from Purakaunui contained ammonium at concentrations between 0.45 and 2.77 μM, with this latter maximum value occurring in January (Fig. 5b). Phosphate levels in seawater collected from Murdering Beach and Purakaunui were similar and ranged between 0.08 and 0.34 μM. The exception was at Murdering Beach in December 1999 when levels rose to 0.65 μM (Fig. 5c).
Carrageenan content
The constituent sugar composition of Sarcothalia lanceata blades from Murdering Beach and Purakaunui is shown in Fig. 6. Total carrageenan content (AnGal and Gal combined) ranged between 75 and 97%. In tetrasporophytic blades, the total (i.e. lambda) carrageenan is equivalent to Gal.
Carrageenans: temporal, site and life history phase variation in the relative proportions of the constituent sugars of female (closed circles), male (open circles) and tetrasporophytic (triangles) Sarcothalia lanceata blades from Murdering Beach (left, a-d) and Purakaunui (right, e-h). AnGal 3,6-anhydrogalactitol peracetate, Gal galactitol peracetate, Glc glucitol peracetate, Xyl xylitol peracetate. Points are means ± SE, n = 3
Proportions of AnGal were significantly higher (Holms-Sidak test, p < 0.001) in male blades (31.3 to 35.1%) than female blades (23.5 to 30.5%) throughout the year, and varied significantly between life phase (two-way ANOVA, p < 0.001) and over time (p < 0.001) (Fig. 6a). There was also a significant interaction term between life phase and time (p = 0.005), indicating that the effects of time were not consistent between/among life phases. Pairwise comparisons confirmed significant differences between female and male AnGal in all months except August1 (A1). In female blades, many significant differences between monthly AnGal levels occurred, with a pattern of significance between summer/autumn and winter/spring months. Monthly levels of AnGal in male blades did not vary significantly.
The same pattern occurred at Purakaunui (Fig. 6e) (two-way ANOVA, life phase and time both p < 0.001) where male AnGal levels (29.5 to 34.3%) were significantly higher than female levels (24.6 and 30.5) (Holms-Sidak test, p < 0.001). The effects of time were not consistent between the life phases (interaction term, p = 0.008) with female blades varying between summer/autumn and winter/spring months, but male blades only varying significantly between June and August1. Pairwise comparisons confirmed significant differences in AnGal between female and male thalli in all months except July and August1.
Gal levels from gametophytic blades from both sites varied significantly between life phase (female and male) and over time (two-way ANOVAs, p < 0.001 for both sites). Significant differences in monthly Gal levels occurred between January/February/March and July/August1/September at Murdering Beach (Holms-Sidak tests), and multiple months at Purakanui with a pattern of summer months vs winter months being evident. In tetrasporophytic blades, Gal levels from both sites varied significantly over time (p < 0.001 for Murdering Beach and p = 0.001 for Purakaunui) with multiple significant pairwise comparisons indicating differences between summer and winter levels for both sites.
Proportions of Glc varied significantly between life phase and over time in blades from both sites (two-way ANOVAs, p < 0.001 for all) (Fig. 6c, g). For both sites, there was a significant interaction term between life phase and time (p = 0.006 for Murdering Beach and p = 0.023 for Purakaunui). Glc levels in blades from Murdering Beach were significantly different between female and male blades in all months except July and August1, while for Purakaunui female and male blades differed significantly in all months except August1 and October. For both sites, and both life phases, many months varied significantly from each other, again with a general pattern of differences between summer/autumn and winter/spring months. Glc in tetrasporophytic blades also varied significantly over time (p < 0.001 for both sites) and multiple significant pairwise interactions followed the same pattern of summer/autumn vs winter/spring levels.
Levels of Xyl in Murdering Beach female and male plants ranged between 0.95 and 2.63% and varied significantly over time (p < 0.001) but not between life phases (two-way ANOVA, p = 0.337) and multiple significant pairwise comparisons indicated differences between summer/autumn vs winter/spring levels. There was a significant interaction term (p = 0.008) between time and life phase, with fewer significant monthly differences occurring in males than females, and female and male levels of Xyl not significantly differing from each other in the months of November, January, August1, August2 (A2) September and October. Xyl levels in female and male blades from Purakaunui ranged between 0.73 and 2.15% and varied significantly over time (p < 0.001) and between life phase (p = 0.002) with a significant interaction term occurring between the two (p = 0.007). There were fewer significant monthly differences in females than males, and female Xyl levels differed from male levels in January, February, July and September. In tetrasporophytic blades, Xyl levels in blades from Murdering Beach varied significantly over time (p < 0.001, multiple significant pairwise interactions, summer vs winter), but those from Purakaunui did not (p = 0.051).
Discussion
The assessment of carrageenans from gametophytic plants of the Gigartinaceae is usually based on female plants (e.g. Zinoun et al. 1993; Pereira and Mesquita 2004; Véliz et al. 2017) or from mixed female and male plants as they cannot be easily distinguished, e.g. García Tasende et al. (2012). In this study, we report separately on the carrageenans of male and female gametophytes, which is novel. In S. lanceata, there is the potential to cultivate and then separately harvest male and female plants because of their distinctive morphologies.
Although the composition and physical properties of the carrageenans from tetrasporophytic blades of this species make this phase the most valuable target of aquaculture, gametophytic blades could also be grown to produce kappa-II type carrageenans. Male blades contain significantly more 3,6-anhydrogalactose and galactitol peracetate than female blades, and the level of these compounds did not vary significantly over time. Although male blades are lighter than both female and tetrasporophytic blades (Neill et al. 2016), this may also mean they are less prone to blade breakage or blade loss than the heavily papillate female blades. In Mastocarpus papillatus, the presence of papillae on female plants has been shown to increase their drag factor (Carrington 1990), which may contribute to increased blade loss.
Based on prior work in other Gigartinaceae, an inverse relationship might be expected between seawater nutrients and carrageenan (i.e. the Neish and Chopin effects). However, the 3,6-anhydrogalactose content in female S. lanceata blades followed the same pattern as ambient seawater nitrate concentrations and tissue nitrogen (e.g. summer minima, winter maxima). In contrast, the 3,6-anhydrogalactose in male blades did not follow this pattern. In addition, the Chopin effect was not evident as seawater phosphate levels varied very little.
In a field study of a Chilean species (Chondracanthus chamissoi), Véliz et al. (2017) found no seasonal variation in carrageenan composition, but significant seasonal variation in carrageenan yields, and higher yields in female plants from a nutrient enriched upwelling site than those from a site 40 km away. In this field study, we did not assess carrageenan yields, so it is not clear how nutrient availability would affect yields in S. lanceata; however, in culture, manipulation of nutrient levels has the potential to alter carrageenan yields. In experiments using female Chondrus crispus (Gigartinaceae), Chopin and Wagey (1999) investigated the effects of phosphorus (P) and nitrogen (N) enrichments on carrageenan production and found that optimal production was related to P and N levels, but not N:P ratios. Conversely, Zinoun et al. (1993) investigated female Sarcothalia teedii (as Gigartina teedii) and found that nitrate enrichments had no significant influence on 3,6-anhydrogalactose levels. González-Gómez et al. (1992) found that adding nutrients to tank cultures of Chondracanthus canaliculatus (as Gigartina canaliculata) once every second week gave higher carrageenan yields but lower gel strengths than adding nutrients once, twice or three times a week. Although no experimental work has been carried out with S. lanceata, the temporal patterns reported here indicate that, in this species, the components of carrageenen (determined as 3,6-anhydrogalactose and galactitol peracetate) do not respond to nitrogen limitation in the same way as carrageenan yield.
In this study, a single parameter was used initially to assess seasonal patterns of growth (blade length and width) then a second parameter was added later in the study (relative growth rate). Significant temporal variation in male blade widths was found at both sites as well as significant variation among all three life history phases. Also, the relative elongation rates of blade widths were positive in most months assessed, indicating growth. However, there was no significant temporal variation in maximum blade length at either site, significant variation in blade lengths among life history phases only occurred at one of the two sites (Purakaunui), and blade relative elongation rates were frequently negative (particularly in female blades). Blade length is a net result between the processes of growth and erosion (e.g. Santos 1994) and these results suggest that blades were being eroded apically resulting in the negative growth rates observed. However, by measuring blade width, it was possible to detect some seasonal patterns in growth in male blades. Assessment of growth using width as the parameter measured is not common in seaweeds, but may be particularly suitable for members of the Gigartinaceae. The reason for this is that the apical meristem may extend along the margin or sometimes be dispersed over the surface of the plant (Hommersand et al. 1993). Reports of growth assessment by width measurements in this family are relatively rare; however, Romo et al. (2006) used blade width to assess the growth of Gigartina skottsbergii in experimental aquaculture in Chile.
In many seaweed species, growth is initiated in winter (Lüning 1993) and peaks in spring or early summer, with growth in temperate waters commonly nitrogen-limited in summer (Kregting et al. 2008). The limited temporal growth patterns detected in the current study are consistent with this pattern, as they are evident in early spring. However, a lack of seasonality in growth has rarely been reported (Pybus 1977) so the results of this study raise two possibilities: firstly, there is little to no temporal variation in the rate of growth in this species, or secondly, temporal variations in growth were not detected by the methods employed in this study.
The C:N ratios of S. lanceata blades were above 15 throughout the study period and only fell below 20 over the winter months, while tissue nitrogen was below 2% throughout the study. These levels indicate that S. lanceata may be nutrient limited year-round. Nitrogen limitation of seaweed growth is generally thought to occur where C:N ratios are > 10–15 (Hanisak 1983) or in some red seaweeds at ratios > 20 (e.g. Greenwell et al. 1984; Rico and Fernández 1996 and authors therein). In various red seaweeds, a critical tissue nitrogen level of around 2% is proposed (Hanisak 1983), and below this level nitrogen limitation may occur (Rico and Fernández 1996 and authors therein). However, if year-round nutrient limitation of growth occurs in S. lanceata, why was there some evidence of a temporal change in the growth of male blades? The specific differences between male and female gametophytes observed here are virtually unknown in the Gigartinaceae although it is thought that sporophytic and gametophytic thalli have different investment in reproductive structures and differences in growth (Thornber 2006). Alternatively, temporal variation may occur in male blades and not female blades due to differences in reproductive costs—the cost of reproduction in male blades may be lower than female blades due to the simpler structures involved in male reproduction.
It is possible that patterns of growth that were not evident in the large and reproductively mature blades used in this study may have been detected if vegetative blades had been assessed. The onset of reproduction has been reported to result in a slowing or cessation of growth (Pybus 1977; Kain and Norton 1990; Guillemin et al. 2014). If reproduction in S. lanceata occurs year-round, this species may be putting few resources into growth, particularly in the case of the largest reproductive blades (May 1986). Large blades may also be more prone to fragmentation than smaller blades (Pybus 1977; Bhattacharya 1985; Santos 1994) and in some red seaweeds, large blades exhibit slower growth rates than small blades (e.g. Kain 1987).
This research was carried out to provide baseline biological data about S. lanceata against which to assess its potential as a source for carrageenan extraction. Wild harvest of S. lanceata does not occur at present, and would be limited by its lower intertidal and upper subtidal distribution on exposed rocky shores. Aquaculture is currently considered a more viable option for obtaining sufficient raw material for carrageenan extraction and is supported by recent success with the experimental culture of Sarcothalia atropurpurea in New Zealand (McNeill and Falshaw 2017). The lambda carrageenans of the tetrasporophytic blades would be the primary target of aquaculture; however, there is also potential value in growing the gametophytic stages of this species, especially as the male gametophyte is easily recognisable, and contains higher levels of 3,6-anhydrogalactose and galactose than female blades, which do not appear to vary significantly over time. Experimentation on spore release and growth and the potential effects of nutrient manipulation on early growth stages are suggested here as the directions of future research. In addition, investigation into the relationship between carrageenan yields and carrageenan composition is also needed.
References
Adams NM (1994) Seaweeds of New Zealand. An illustrated guide. Canterbury University Press, Christchurch
Ahmadi A, Moghadamtousi SZ, Abubakar S, Zandi K (2015) Antiviral potential of algae polysaccharides isolated from marine sources: a review. Biomed Res Int 2015:1–10
Amimi A, Mouradi A, Bennasser L, Givernaud T (2007) Seasonal variations in thalli and carrageenan composition of Gigartina pistillata (Gmelin) Stackhouse (Rhodophyta, Gigartinales) harvested along the Atlantic coast of Morocco. Phycol Res 55:143–149
Asare SO (1980) Seasonal changes in sulfate and 3,6-anhydrogalactose content of phycocolloids from two red algae. Bot Mar 23:595–598
Atkinson MJ, Smith SV (1983) C:N:P ratios of benthic marine plants. Limnol Oceanogr 28:568–574
Avila M, Ask E, Rudolph B, Nunez M, Norambuena R (1999) Economic feasibility of Sarcothalia (Gigartinales, Rhodophyta) cultivation. Hydrobiologia 398:435–442
Bhattacharya D (1985) The demography of fronds of Chrondrus crispus Stackhouse. J Exp Mar Biol Ecol 91:217–231
Bixler HJ (1996) Recent developments in manufacturing and marketing carrageenan. Hydrobiologia 327:35–57
Bixler HJ, Porse H (2011) A decade of change in the seaweed hydrocolloids industry. J Appl Phycol 23:321–335
Breden PC, Bird KT (1994) Effects of environmental factors on carrageenan from Gymnogongrus griffithsiae (Gigartinales, Rhodophyta). J Appl Phycol 6:371–380
Buschmann AH, Correa JA, Westermeier R, Hernandez-González MD, Norambuena R (2001) Red algal farming in Chile: a review. Aquaculture 194:203–220
Buschmann AH, Varela D, Cifuentes M, Hernandez-González MD, Henriquez L, Westermeier R, Correa JA (2004) Experimental indoor cultivation of the carrageenophytic red alga Gigartina skottsbergii. Aquaculture 241:357–370
Campbell R, Hotchkiss S (2017) Carrageenan industry market overview. In: Hurtado AQ, Critchley AT, Neish IC (eds) Tropical seaweed farming trends, problems and opportunities. Springer, Cham, pp 193–205
Carrington E (1990) Drag and dislodgment of an intertidal macroalga—consequences of morphological variation in Mastocarpus papillatus Kützing. J Exp Mar Biol Ecol 139:185–200
Carrington E, Grace SP, Chopin T (2001) Life history phases and the biomechanical properties of the red alga Chondrus crispus (Rhodophyta). J Phycol 37:699–704
Charrier B, Abreu MH, Araujo R, Bruhn A, Coates JC, De Clerck O, Katsaros C, Robaina R, Wichard T (2017) Furthering knowledge of seaweed growth and development to facilitate sustainable aquaculture. New Phytol 216:967–975
Chopin T and Floc’h JY (1992) Eco-physiological and biochemical study of two of the most contrasting forms of Chondrus crispus (Rhodophyta, Gigartinales). Mar Ecol Prog Ser 81:185–195
Chopin T, Gallant T, Davison I (1995) Phosphorus and nitrogen nutrition in Chondrus crispus (Rhodophyta): effects on total phosphorus and nitrogen content, carrageenan production, and photosynthetic pigments and metabolism. J Phycol 31:283–293
Chopin T, Sharp G, Belyea E, Semple R, Jones D (1999) Open-water aquaculture of the red alga Chondrus crispus in Prince Edward Island, Canada. Hydrobiologia 398:417–425
Chopin T, Wagey BT (1999) Factorial study of the effects of phosphorus and nitrogen enrichments on nutrient and carrageenan content in Chondrus crispus (Rhodophyceae) and on residual nutrient concentration in seawater. Bot Mar 42:23–31
Cosson J, Deslandes E, Braud JP (1990) Preliminary approach to the characterization and seasonal variation of carrageenans from four Rhodophyceae on the Normandy Coast (France). Hydrobiologia 204:539–544
Dawes CJ, Lawrence JM, Cheney DP, Mathieson AC (1974) Ecological studies of Floridian Eucheuma (Rhodophyta, Gigartinales) .3. Seasonal variation of carrageenan, total carbohydrate, protein, and lipid. Bull Mar Sci 24:286–299
Dring M (1982) The biology of marine plants. Edward Arnold Limited, London
Falshaw R, Furneaux RH (1998) Structural analysis of carrageenans from the tetrasporic stages of the red algae, Gigartina lanceata and Gigartina chapmanii (Gigartinaceae, Rhodophyta). Carbohydr Res 307:325–331
Friedlander M, Lipkin Y (1982) Rearing of agarophytes and carrageenophytes under field conditions in the Eastern Mediterranean. Bot Mar 25:101–105
Friedlander M, Zelikovitch N (1984) Growth rates, phycocolloid yield and quality of the red seaweeds, Gracilaria sp., Pterocladia capillacea, Hypnea musciformis, and Hypnea cornuta, in field studies in Israel. Aquaculture 40:57–66
Fuller SW, Mathieson AC (1972) Ecological studies of economic red algae. IV. Variations of carrageenan concentration and properties in Chondrus crispus Stackhouse. J Exp Mar Biol Ecol 10:49–58
García Tasende M, Cid M, Fraga MI (2012) Spatial and temporal variations of Chondrus crispus (Gigartinaceae, Rhodophyta) carrageenan content in natural populations from Galicia (NW Spain). J Appl Phycol 24:941–951
González-Gómez MA, Zertuche-González JA, Pacheco-Ruíz I (1992) Nitrogen effect on growth and carageenan content in Gigartina canaliculata Harv. (Rhodophyta, Gigartinales), in outdoor tank cultures. Cienc Mar 18:75–83
Greenwell M, Bird CJ, McLachlan J (1984) Depth-related variation in gross chemical composition of several seaweeds. Aquat Bot 20:297–305
Guillemin ML, Valenzuela P, Gaitán-Espitia JD, Destombe C (2014) Evidence of reproductive cost in the triphasic life history of the red alga Gracilaria chilensis (Gracilariales, Rhodophyta). J Appl Phycol 26:569–575
Hanisak MD (1983) The nitrogen relationships of marine macroalgae. In: Carpenter EJ, Capone DG (eds) Nitrogen in the marine environment. Academic Press, Inc., New York, pp 699–730
Harden EA, Falshaw R, Carnachan SM, Kern ER, Prichard MN (2009) Virucidal activity of polysaccharide extracts from four algal species against herpes simplex virus. Antivir Res 83:282–289
Heertje I (1993) Structure and function of food-products—a review. Food Struct 12:343–364
Hernández-González MC, Buschmann AH, Cifuentes M, Correa JA, Westermeier R (2007) Vegetative propagation of the carrageenophytic red alga Gigartina skottsbergii Setchell et Gardner: indoor and field experiments. Aquaculture 262:120–128
Hommersand MH, Guiry MD, Fredericq S, Leister GL (1993) New perspectives in the taxonomy of the Gigartinaceae (Gigartinales, Rhodophyta). Hydrobiologia 260:105–120
Hurd CL, Harrison PJ, Bischof K, Lobban CS (2014) Seaweed ecology and physiology. Cambridge University Press, Cambridge
Jol CN, Neiss TG, Penninkhof B, Rudolph B, De Ruiter GA (1999) A novel high-performance anion-exchange chromatographic method for the analysis of carrageenans and agars containing 3,6-anhydrogalactose. Anal Biochem 268:213–222
Kain JM (1987) Seasonal growth and photoinhibition in Plocamium cartilagineum (Rhodophyta) off the Isle of Man. Phycologia 26:88–99
Kain JM, Norton TA (1990) Marine ecology. In: Cole KM, Sheath RG (eds) Biology of the red algae. Cambridge University Press, Cambridge, pp 377–422
Kregting LT, Hepburn CD, Hurd CL, Pilditch CA (2008) Seasonal patterns of growth and nutrient status of the macroalga Adamsiella chauvinii (Rhodophyta) in soft sediment environments. J Exp Mar Biol Ecol 360:94–102
Li L, Ni R, Shao Y, Mao S (2014) Carrageenan and its applications in drug delivery. Carbohyd Polym 103:1–11
Lüning K (1993) Environmental and internal control of seasonal growth in seaweeds. Hydrobiologia 261:1–14
May G (1986) Life history variations in a predominantly gametophytic population of Iridaea cordata (Gigartinaceae, Rhodophyta). J Phycol 22:448–455
McLachlan J, Bird CJ (1986) Gracilaria (Gigartinales, Rhodophyta) and productivity. Aquat Bot 26:27–49
McNeill SE, Falshaw R (2017) Aquaculture of Sarcothalia atropurpurea tetraspores in New Zealand for carrageenan production. N Z J Mar Freshw Res 51:528–542
McNeill SE, Page M, Falshaw R (2003) Field trials to optimise timing and frequency of pruning for cultivation of a New Zealand carrageenophyte, Gigartina atropurpurea. J Appl Phycol 15:391–405
Neill KF, Nelson WA, Falshaw R, Hurd CL (2016) Clump structure, population structure and non-destructive biomass estimation of the New Zealand carrageenophyte Sarcothalia lanceata (Gigartinaceae, Rhodophyta). Bot Mar 59:373–385
Neish AC, Shacklock PF, Fox CH, Simpson FJ (1977) Cultivation of Chondrus crispus—factors affecting growth under greenhouse conditions. Can J Bot 55:2263–2271
Nelson WA (2013) New Zealand seaweeds. An illustrated guide. Te Papa Press, Wellington
Norambuena R (1996) Recent trends of seaweed production in Chile. Hydrobiologia 327:371–379
Parsons MJ, Pickmere SE, Bailey RW (1977) Carrageenan composition in New Zealand species of Gigartina (Rhodophyta): geographic variation and interspecific differences. N Z J Bot 15:589–595
Pereira L, Mesquita JF (2004) Population studies and carrageenan properties of Chondracanthus teedei var. lusitanicus (Gigartinaceae, Rhodophyta). J Appl Phycol 16:369–383
Pickmere SE, Parsons MJ, Bailey RW (1973) Composition of Gigartina carrageenan in relation to sporophyte and gametophyte stages in the life cycle. Phytochemistry 12:2441–2444
Pickmere SE, Parsons MJ, Bailey RW (1975) Variations in carrageenan levels and composition in three New Zealand species of Gigartina. N Z J Sci 18:585–590
Piriz ML, Cerezo AS (1991) Seasonal variation of carrageenans in tetrasporic, cystocarpic and “sterile” stages of Gigartina skottsbergii S. et G. (Rhodophyta, Gigartinales). Hydrobiologia 226:65–69
Pybus C (1977) The ecology of Chondrus crispus and Gigartina stellata (Rhodophyta) in Galway Bay. J Mar Biol Assoc UK 57:609–628
Reani A, Cosson J, Parker A (1998) Seasonal variation of growth, carrageenan content and rheological properties of Cystoclonium purpureum (Huds.) batters (Rhodophyta, Cystocloniaceae) from the Calvados Coast (France). Bot Mar 41:383–387
Rebours C, Marinho-Soriano E, Zertuche-González JA, Hayashi L, Vásquez JA, Kradolfer P, Soriano G, Ugarte R, Abreu MH, Bay-Larsen I, Hovelsrud G, Rødven R, Robledo D (2014) Seaweeds: an opportunity for wealth and sustainable livelihood for coastal communities. J Appl Phycol 26:1939–1951
Rico JM, Fernandez C (1996) Seasonal nitrogen metabolism in an intertidal population of Gelidium latifolium (Gelidiaceae, Rhodophyta). Eur J Phycol 31:149–155
Romo H, Alveal K, Werlinger C (2001) Growth of the commercial carrageenophyte Sarcothalia crispata (Rhodophyta, Gigartinales) on suspended culture in central Chile. J Appl Phycol 13:229–234
Romo H, Avila M, Nunez M, Perez R, Candia A, Aroca G (2006) Culture of Gigartina skottsbergii (Rhodophyta) in southern Chile. A pilot scale approach. J Appl Phycol 18:307–314
Rosenberg G, Ramus J (1982) Ecological growth strategies in the seaweeds Gracilaria foliifera (Rhodophyceae) and Ulva sp (Chlorophyceae)—soluble nitrogen and reserve carbohydrates. Mar Biol 66:251–259
Running CA, Falshaw R, Janaswamy S (2012) Trivalent iron induced gelation in lambda-carrageenan. Carbohyd Polym 87:2735–2739
Santelices B (1996) Seaweed research and utilization in Chile: moving into a new phase. Hydrobiologia 327:1–14
Santelices B (1999) A conceptual framework for marine agronomy. Hydrobiologia 399:15–23
Santos R (1994) Frond dynamics of the commercial seaweed Gelidium sesquipedale: effects of size and of frond history. Mar Ecol Prog Ser 107:295–305
Stanley NF (1990) Carrageenans. In: Harris P (ed) Food Gels. Springer Netherlands, Dordrecht, pp 79–119
Stevenson TT, Furneaux RH (1991) Chemical methods for the analysis of sulfated galactans from red algae. Carbohydr Res 210:277–298
Thornber CS (2006) Functional properties of the isomorphic biphasic algal life cycle. Integr Comp Biol 46:605–614
Véliz K, Chandía N, Rivadeneira M, Thiel M (2017) Seasonal variation of carrageenans from Chondracanthus chamissoi with a review of variation in the carrageenan contents produced by Gigartinales. J Appl Phycol 29:3139–3150
Westermeier R, Aguilar A, Sigel J, Quintanilla J, Morales J (1999) Biological basis for the management of Gigartina skottsbergii (Gigartinales, Rhodophyta) in southern Chile. Hydrobiologia 398:137–147
Zertuche-González JA, Pacheco-Ruiz I, Soria-Mercado IE (1993) Carrageenan yield and properties of Eucheuma uncinatum (Seth. and Gard.) Daw. cultured under natural conditions. Hydrobiologia 260/261:601–605
Zinoun M, Cosson J, Deslandes E (1993) Influence of culture conditions on growth and physicochemical properties of carrageenans in Gigartina teedii (Rhodophyceae, Gigartinales). Bot Mar 36:131–136
Acknowledgments
At the time this research was carried out KN and CH were affiliated with the University of Otago (OU), RF with Industrial Research Ltd (IRL) and WN with Te Papa Tongarewa Museum of New Zealand (TP). This work was funded by a OU Postgraduate Scholarship to KN, an OU Research Grant to CH, and grants from IRL and TP. Additional funds were received from the National Institute of Water & Atmospheric Research under Coasts and Oceans Programme 2 (COBR1701 2016/17 and 2017/18). Thanks to the staff at IRL for their assistance with the carrageenan work and to all our fieldwork helpers. The authors are grateful to Judy Sutherland and the anonymous reviewers for their helpful comments on this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Neill, K., Nelson, W., Hurd, C. et al. Growth and carrageenan composition of two populations of the New Zealand carrageenophyte Sarcothalia lanceata (Gigartinaceae, Rhodophyta). J Appl Phycol 30, 2485–2497 (2018). https://doi.org/10.1007/s10811-018-1416-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1416-3