Abstract
Kelps are economically valuable primary producers; therefore, many studies on breeding have attempted to increase kelp productivity and quality. However, most cultivation tests have been performed in the ocean, thereby limiting the development of new cultivars. To reduce the breeding duration period and confirm cultivar phenotypes, we developed a novel tank culture system, referred to as a circulation and floating culture system (CFCS), for cultivating macroalgae. In the CFCS, kelp can be cultivated under controlled environmental conditions. Water velocity in the CFCS can be regulated by changing the angle of a seawater inlet spout without changing the volume of seawater in the tank. Undaria pinnatifida and Saccharina japonica cultivated in the CFCS exhibited morphological features very similar to those of plants grown naturally in the ocean. The result suggests that the facility is useful for identifying water motion conditions suitable for increasing the production of any macroalgae species. Using this facility, both species were grown from juvenile sporophytes (20 mm) to maturity; for U. pinnatifida, the subsequent generation was successfully cultivated. Improved growth of U. pinnatifida was achieved in fast flows compared with slow flows, whereas S. japonica developed a wider shape and heavier biomass in slow flows compared with fast flows. We discuss the application and implication of the CFCS for breeding research and the physiological ecology of macroalgae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Kelps are economically valuable primary producers in coastal ecosystems (Mann 1972). Kelp productivity has been estimated to be 2.8 times greater than that of sugarcane, the most productive land plant under cultivation (Gao and McKinley 1993). Various species of kelp are used as edible foods and industrial materials (Jensen 1993), and the total global kelp production in 2008 was estimated to be 1.5 million tonnes (Food and Agricultural Organisation (FAO) 2010). Furthermore, brown macroalgae have become the focus of attention as ideal feedstocks for biofuel production because they do not require arable land, fertilisers or fresh water resources (Wargacki et al. 2012).
A relation between decreased kelp abundance and ocean warming has been reported for Macrocystis pyrifera off the east coast of Tasmania, Australia (Johnson et al. 2011); for Saccharina japonica and Costaria costata off northern Honshu, Japan (Kirihara et al. 2006) and for Laminaria hyperborea along the Iberian Peninsula (Müller et al. 2009). A remarkable decline in Saccharina was reported in Hokkaido, northern Japan, where the production of Saccharina ochotensis has decreased to 12% of its peak production achieved during the 1940s (Nabata et al. 2003). Given this instability in natural kelp resources, market demand is driving the development of new cultivation technologies (FAO 2010). Progress in the development of new breeding techniques is required to increase kelp productivity.
Previous studies on kelp breeding identified heterosis in Undaria pinnatifida (Hara and Akiyama 1985) and M. pyrifera (Westermeier et al. 2010). By breeding for five generations, Zhang et al. (2011) obtained a new variety of S. japonica, which was given the name ‘Rongfu’ and which attained yields of 24–27% higher than those of other varieties. To reduce the breeding duration and confirm cultivar phenotypes, a novel tank culture system in which kelp can be cultivated through its entire lifecycle under controlled environmental conditions is required. Previous studies showed that water motion impacts the morphological characteristics and productivity of kelp (Hurd et al. 1996; Nanba et al. 2011; Peteiro and Freire 2011); therefore, the ability to control water velocity was required in the new tank system. Tank culture systems that are suitable for the small perennial macroalgae Chondrus crispus (Neish et al. 1977) and Kappaphycus alvarezii (Zuldin et al. 2016), the small annual macroalgae Ulva prolifera (Hiraoka and Oka 2008) and Porphyra spp. (Israel et al. 2006) and the young sporophyte stage of the large perennial macroalgae M. pyrifera and Lessonia trabeculata (Westermeier et al. 2006) have previously been demonstrated. However, a tank system that can be used to cultivate large numbers of mature kelp sporophytes under controlled environmental conditions has yet to be put into practical use.
In the present study, we developed a novel tank system in which temperature, light intensity and water velocity can be controlled. The new system was designated as a circulation and floating culture system (CFCS) for macroalgae based on the characteristic water motion in the tank. We cultivated U. pinnatifida and S. japonica sporophytes in the CFCS, evaluated their morphological and growth responses to water velocity and compared these to published data on off-shore aquaculture lines. For U. pinnatifida, we verified whether the subsequent generation was successfully cultured in the CFCS.
Materials and methods
Development of a new tank system
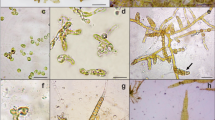
A polycarbonate circular tank (SPS-2000, Earth Co., Ltd., Tokyo, Japan) 90 cm tall and 180 cm in diameter with a capacity of 2000 L was used as the main component of the CFCS (Figs. 1 and 2a). A hole 30 mm in diameter was created in the centre of the bottom of the tank and connected to a circular base (CB-2000, Earth Co., Ltd) for drainage (Fig. 2b, c). A vertical poly vinyl chloride (PVC) pipe 100 cm in length was connected to the hole in the centre of the tank (Fig. 2d). A conical structure 85 cm in length was installed inside the tank and connected to the PVC pipe through the hole (Fig. 2e). Holes 2 mm in diameter at 20-mm intervals were created across the entire surface of the conical structure. To supply air to the tank, eight porous stones were placed around the narrow end of the conical structure at the bottom of the tank (Fig. 2f). The porous stones were connected to an air compressor by 5 mm in diameter silicon tubing. Four of these CFCS systems were installed at the Iwate Fisheries Technology Center, Iwate Prefecture, Japan. Seawater was filtered through a 1 μm microfibre filter (TCW-CS, Advantec MFS, USA) and sterilised using ultraviolet (UV) lamps (Flonlizer 4L, Chiyoda-Kouhan Co., Ltd., Japan). Seawater was continuously flowed through the CFCS.
Schematic view of a new tank system termed the circulation and floating culture system (CFCS), for the cultivation of macroalgae: a circular polycarbonate tank, b circular base, c drainage hole, d poly vinyl chloride (PVC) pipe for seawater drainage, e conical polystyrene structure, f aeration stone, g PVC inlet pipe, h spout of PVC inlet pipe, i variable of inlet pipe, j seawater flow from inlet pipe, k seawater flow to drainage, l filtration bag and m air flow
Light, seawater temperature and nutrient conditions in the CFCS
Three fluorescent lights were installed above the tank, and the level of light intensity at the surface of the seawater could be regulated from 0 to 200 μmol photons m−2 s−1 by adjusting the distance of the lights from the surface. A switch timer was used to regulate photoperiods. The seawater temperature was regulated to between 5 and 25 °C using a heater/cooler (BFL-80F, Mitsubishi, Japan). The concentrations of nutrients of the seawater in the CFCS during the cultivation trial were in the following ranges: NO3–N, 4.8–6.3 μM; NH4–N, 0.8–1.2 μM; PO4-P, 0.2–0.4 μM. Nutrients were measured using an autoanalyser (QuAAtro 2-HR, BLTEC, Osaka, Japan) once every 2 weeks (26 November 2012–10 March 2013, 24 June 2013–30 July 2013).
Measurement of water velocity in the CFCS
Water motion influences the morphological characteristics and productivity of kelp (Hurd 2000). The water velocity in the CFCS was measured for 10 min (n = 3) using an electromagnetic current meter (Infinity-Em, JFE Advantec Co., Ltd., Japan). Measurements of water velocity in the CFCS were taken at three positions: (1) an upper position 5 cm below the surface of the seawater, (2) a position at a depth equidistant of the seawater surface and the tank bottom and (3) a lower position 5 cm above the tank bottom. The water velocity was measured as the sum total of the value at the horizontal X-axis and Y-axis. The average water velocity at each position in the tank was calculated from 600 values recorded during a period of 10 min.
Cultivation of U. pinnatifida and S. japonica in the CFCS and morphological measurements
Parental sporophytes of U. pinnatifida and S. japonica were collected from wild populations growing along the coast near Hirota Bay (38°95′N, 141°67′E) in southern Iwate Prefecture on 25 July 2012 and in Shukunohe (40°37′N, 141°76′E) in northern Iwate Prefecture on 20 August 2012. Sections (3 × 3 cm) were excised from U. pinnatifida sporophylls and from S. japonica sori, cleaned in sterilised seawater and placed in paper towels at 4 °C for the induction of zoospores. After 12 h, the pieces were washed three times in sterilised seawater and placed in a 1-L beaker containing 800-mL sterilised seawater at 15 °C, followed by zoospore induction after approximately 15 min. The zoospores were collected using a micropipette and placed into plastic dishes containing 30 mL PESI medium (Tatewaki 1966). Approximately 2000 individual zoospores were counted in each dish using a haemocytometer. The dishes were incubated under a 12-h light/12-h dark cycle with light intensity of 20 μmol photons m−2 s−1 and temperature of 20 °C. After 80 days for U. pinnatifida and 50 days for S. japonica, one male and one female gametophyte were transferred to a 15-mL glass vial containing 10 mL sterilised seawater and homogenised at 5000 rpm for 2 min using a microblender (T 18 digital Ultra-Turrax, IKA, Japan). Gametophyte fragments of U. pinnatifida and S. japonica were incubated in separate dishes containing 30 mL PESI medium under a 10-h light/14-h dark cycle at 50 μmol photons m−2 s−1 and 20 °C for 2 weeks to promote maturation.
Germinated sporophytes of U. pinnatifida and S. japonica were cultivated in 3-L Erlenmeyer flasks containing 2 L of 1/4-strength PESI medium with aeration and a 12-h light/12-h dark cycle at 90 μmol photons m−2 s−1 and 15 °C. The start dates for cultivation were 27 October for U. pinnatifida and 23 October for S. japonica. The culture medium was changed at 3-day intervals. After the average total length of the sporophytes reached 20 mm, the 300 longest healthy juvenile sporophytes of both species without damage were selected. Sporophytes of U. pinnatifida and S. japonica were transferred to the separate CFCSs on 26 November and 7 December for U. pinnatifida and S. japonica, respectively. Sporophytes of both species were precultured in the separate CFCSs under 180 μmol photons m−2 s−1 at 10 °C, and the spout angle was set at 0° for 2 weeks, after which the longest 200 sporophytes were selected and divided equally into two groups. In the total four CFCSs, the angles of the inlet spouts of two of the CFCSs were set at 0°, whereas those of the remaining two were set at 90°. For each species, 100 sporophytes were cultivated in the separate CFCSs with the angle of the inlet spout set at 0° or 90°. The sporophytes were collected, and the longest 30 individuals were selected from each group after 15–30 days. The total lengths and total weights of the U. pinnatifida and S. japonica sporophytes were measured. In addition, the number of U. pinnatifida plants with sporophylls was counted. For S. japonica, the maximum blade width was measured.
Measurements of relative growth rates of U. pinnatifida cultivated in the CFCS
To evaluate the growth rate of U. pinnatifida cultivated in the CFCS under different starting weights, relative growth rates (RGRs) were calculated as according to Eq. 1 (Yong et al. 2013):
where W 0 is the initial fresh weight, W t is the final fresh weight at the end of the experiment and t is the number of days of cultivation.
Cultivation of U. pinnatifida germinated from a single strain in the CFCS
Juvenile sporophytes were induced from a male and female pairing of gametophytes and precultured according to the method described above. After the average total length of the sporophytes reached 20 mm (on 24 June 2013), all plants were transferred to the CFCS and cultivated at 10 °C under a 12-h photoperiod, a light intensity of 180 μmol photons m−2 s−1 and an inlet angle of 90°. After 36 days (30 July 2013), the 230 longest plants were selected, tagged for individual identification and cultivated in the CFCS for 20 days. Fresh weights of all sporophytes were measured after being blotted dry both before and at the end of the experiments.
Statistical analysis
The statistical significance of differences in water velocities in the CFCS among tanks and water inlet angles was analysed using one-way analysis of variance (ANOVA) followed by Gomes–Howell post hoc comparison. The statistical significance of differences in morphological characteristics of U. pinnatifida and S. japonica during the measurement periods was analysed using the Kruskal–Wallis test followed by a post hoc test using SPSS (ver. 23, IBM, USA) because not all data exhibited a normal distribution and homogeneous variance. The statistical significance of differences in morphological characteristics of the two species cultivated at various water velocities was analysed using the Mann–Whitney U test. Results were regarded as significant at P < 0.05.
Results
Water motion and water velocity in the CFCS
Sterilised seawater was added to the tank through an angled PVC pipe located at the edge of the tank (Fig. 2g). By changing the direction of the angled pipe at the edge of the tank (Fig. 2h, i), the water velocity in the tank could be regulated (Fig. 2j). The volume of the seawater introduced into the tank was controlled by a ball valve. Seawater was removed from the tank by flowing through holes in the conical structure through the top of the central vertical PVC pipe (Fig. 2k) and through a 1-μm mesh filter (CUNO filter bag, 10EA, 3 M, Tokyo, Japan) (Fig. 2l).
Water motion in the CFCS was circumferential along the inner wall of the circular tank. Compressed air ascended from air stones at the centre of the bottom of the tank to the surface of the seawater along the outer wall of the central conical structure with circumferential movement followed by movement of the air bubbles to the edge of the tank in a spiral motion (Figs. 1 and 2m). This movement of air was helpful in avoiding the concentration and entanglement of plants in the central lower part. To evaluate the effects of water velocity in the tank, the direction of the spout was set at angles of 0, 45 and 90° relative to the diameter of the tank (Fig. 2j). The flow rate of seawater into the tank was set at 1500 L h−1 using a ball valve. The water velocity significantly varied with the inlet angle at all measuring positions (P < 0.01, Supplemental Table 1), whereas no significant difference in the water velocity was detected among the three facilities (P > 0.05, Supplemental Table 2). A time series of water velocity for 10 min in the three CFCSs with the inlet angle set at all measuring positions is represented as average velocities per min in Fig. 3. The values ranged between 1.76–4.04, 4.14–8.29 and 12.47–14.70 cm s−1 at 0, 45 and 90°, respectively, in the CFCS.
Time series of water velocity for 10 min at the upper (a), middle (b) and lower (c) measurement sites in the circulation and floating culture system (CFCS). Inlet angles to introduce seawater into the circular tank were set at 0° (triangle), 45° (square) and 90° (circle) relative to the tank diameter. The measurements were conducted within three CFCSs (each time series described using blue, green and red), and each point represents the average water velocity over 60 s
Changes in morphological characteristics of U. pinnatifida and S. japonica cultivated under various water velocities in the CFCS
With water agitation, the movements of U. pinnatifida and S. japonica cultivated in the CFCS followed that of the bubbles, and the plants drifted in the circumferential current of the tank without becoming entangled (Fig. 1). The total lengths and individual weights of U. pinnatifida exhibited significant increases through the cultivation periods (Fig. 4a, b, P = 0.00, Supplemental Table 3). After day 68, the total length of U. pinnatifida grown in the faster flow treatment was 1.40–1.45 times greater than that grown in slow water flow (Fig. 4a, P = 0.00, Supplemental Table 4). Similarly, after day 68, the wet weight of U. pinnatifida grown in the fast water flow treatment was 1.20–1.85 times greater than that grown in slow water flow (Fig. 4b, P = 0.00, Supplemental Table 4). At day 68, 96% of U. pinnatifida grown in the fast flow experimental treatment possessed sporophylls, whereas 60% grown in the slow flow treatment had sporophylls (Fig. 4c). Spores released from the U. pinnatifida sporophylls cultivated in the CFCS germinated normally into gametophytes, following which they germinated into sporophytes. The sporophytes subsequently grew and formed sporophylls.
Effect of water velocity on morphological characteristics and maturation of U. pinnatifida cultivated in the circulation and floating culture system (CFCS). a Total length, b individual weight and c proportion of individual plants that formed sporophylls. Vertical bars indicate standard deviations. Different letters and asterisks indicate significant differences (P < 0.05) between cultivation periods and water velocities, respectively
The total lengths, maximum blade widths and individual weights of S. japonica exhibited significant increases throughout the cultivation period (Fig. 5a–c, P = 0.00, Supplemental Table 5). No effect of water velocity on the final lengths of S. japonica was evident (Fig. 6), the average of which was 227 cm (Fig. 5a, P = 0.73, Supplemental Table 6). Meanwhile, effects of water velocity on the final maximum blade widths were evident, and the average of the final blade widths of S. japonica grown in the slow flow treatment was 1.76 times than those grown in the fast flow (Fig. 5b, P = 0.00, Supplemental Table 6). Effects of water velocity on the individual weight was also evident, and there was a statistically significant difference between the weights of sporophytes grown in the slow flow and in the fast flow, with weights of sporophytes in the slow flow being 2.1 times heavier than those grown in the fast flow throughout the cultivation period, except for day 48 (Fig. 5c, P < 0.05, Supplemental Table 6).
Effect of water velocity on morphological characteristics of Saccharina japonica cultivated in the circulation and floating culture system (CFCS). a Total length, b maximum blade width and c individual weight. Vertical bars indicate standard deviations. Different letters and asterisks indicate significant differences (P < 0.05) between cultivation periods and water velocities, respectively
Variation in relative growth rates of individual U. pinnatifida plants derived from a single strain
The RGR of 229 plants, except for one eroded plant, ranged from a minimum value of 3.32% day−1 to a maximum value of 9.65% day−1 (Fig. 7), although all of the plants germinated from a single pair of gametophytes.
Discussion
Macroalgae cultivation in the CFCS
The CFCS facilitated the cultivation of kelp under a water velocity similar to that of typical cultivation sites. Water velocity with a spout angle of 90° was higher than that of a U. pinnatifida and S. japonica cultivation site at Okirai Bay, Iwate Prefecture (Nanba et al. 2011), which is one of the major U. pinnatifida cultivation sites in Japan. A previous typical tank culture system given the name ‘tumble culture’ was used for small algae (reviewed in Neish et al. 1977; Lobban and Harrison 1994; Hiraoka and Oka 2008), although water velocity was not considered. Furthermore, in this tank culture system, plants continued to move vertically at the same position; hence, this system is not suitable for kelp species of large and long shapes due to the entanglement of each individual. In contrast, the movement of kelp in the CFCS was maintained in horizontal, circular and vertical spirals without tangling, even if the U. pinnatifida and S. japonica used were over 1 m in total length (Figs. 4 and 5). Therefore, the CFCS system is a technological advance that overcomes the previous limitations of a tank culture system for large seaweeds.
Changes in morphological characteristics of U. pinnatifida and S. japonica cultivated under various water velocities in the CFCS
Hurd et al. (1996) demonstrated that a rapid wave motion provides increased water velocity and turbulence, thereby reducing the diffusion boundary layer along the algal surface and increasing the uptake of nutrients and carbon dioxide. In the present study, the enhanced growth of U. pinnatifida in the CFCS using the high water velocity was likely the result of the reduced diffusion boundary layer. The growth of U. pinnatifida is affected by water motion, with higher velocities leading to larger, thicker fronds (Nanba et al. 2011) and higher biomass (Peteiro and Freire 2011). Our results showed that the growth characteristics of U. pinnatifida sporophytes cultured in the CFCS exhibited the same dependency on water velocities (Fig. 4) as those shown in previous studies in the ocean (Nanba et al. 2011; Peteiro and Freire 2011). Therefore, cultivation of U. pinnatifida in the CFCS under significantly different water velocity conditions facilitated the evaluation of the effects of water velocity on the morphological characteristics and productivity of kelp.
Kelp species develop relatively smooth, flat blades and a narrow, thick morphology when exposed to wave action (Gerard and Mann 1979; Hurd et al. 1996; Hurd and Pilditch 2011). This morphology reduces drag forces that would otherwise damage or remove the blades and increases the blade strength (Gerard and Mann 1979). As predicted, S. japonica had a narrower shape when cultivated in the CFCS using the high velocity (Figs. 5 and 6), suggesting that the water velocities in the CFCS reproduced the growing conditions of S. japonica in the ocean. Similarly, the individual weight of U. pinnatifida was higher at the high velocity than the low velocity (Fig. 4b). However, the weight of S. japonica was unexpectedly lower at the high velocity compared to the low velocity (Figs. 5b and 6a, b). It is suggested that different species achieve maximal production at different water velocities. For example, fishermen in Japan have observed differences in the cultivation of S. japonica and U. pinnatifida at sheltered or exposed sites. Therefore, water velocity should be optimised for each species of cultivated kelp to increase productivity.
Implications for breeding of kelp and land-based cultivation of macroalgae
Efficient breeding requires analysis of the phenotypes of many individuals cultured under the same environmental conditions. In the CFCS, U. pinnatifida sporophytes could be cultured with the attachment of small plastic tags whereby the growth rates of all plants can be measured. Free-living male and female gametophyte cultures of U. pinnatifida were established from a single zoospore discharged from mature sporophytes, thereby enabling the production of genetically identical seedlings (Ishikawa 1991; Dan et al. 2015; Niwa 2015). Niwa (2015) demonstrated double cropping with U. pinnatifida in Pyropia farms by using a single pair of free-living male and female gametophyte cultures to enable the start of Undaria cultivation 2 months later. Niwa and Harada (2016) examined the forced cultivation of U. pinnatifida in the Seto Inland Sea in southern Japan and found that it was possible to harvest the mature sporophyte (approximately 1 m in length) from late December, and although the crop is usually harvested during February and March around the region, the harvest in their study was conducted six times from the same ropes until late February. From these industrial trials, the requirements for breeding of U. pinnatifida were highlighted (Dan et al. 2015; Niwa 2015; Niwa and Harada 2016). In the present study, the RGR of U. pinnatifida sporophytes could be measured under CFCS cultivation. The values varied, although all of the plants had germinated from a single pair of gametophytes. Given that this result shows that individuals of higher growth can be selected based on the weight data of individual kelp cultivated in the CFCS, this cultivation method could be useful for developing candidates with higher growth rates and productivity compared with ordinary strains.
Previously, kelp breeding studies were conducted in the ocean using a rope set horizontally at the surface of the seawater (Li et al. 2007; Zhang et al. 2007, 2011). These studies could be conducted only once per year, highlighting the need to reduce the length of time required for breeding studies. The results of our study suggest that the selective breeding of kelp can be conducted independently from the cultivation schedule in the ocean by using CFCS cultivation.
Gao et al. (2013) conducted transplantation experiments and demonstrated that juvenile sporophytes among geographically separated populations showed different levels of tolerance to high temperatures. The authors concluded that these characteristics were the result of the genetic differentiation of ecotypes rather than of phenotypic plasticity. Sato et al. (2016) demonstrated the phenotypic differentiation of morphology and nutrient uptake kinetics among U. pinnatifida cultivated at six sites in Japan and suggested the existence of ecotypes according to nutrient uptake kinetics. To further advance the selection breeding of U. pinnatifida, the determination of whether the difference in the phenotype is the result of genetic or environmental variation is required. It is also necessary to confirm the phenotype for candidates that would be suitable cultivars. The CFCS enables sporophytes to be cultivated under the same environmental conditions throughout the year and could be used to determine relevant genetic characteristics and to shorten the breeding period.
Land-based cultivation of macroalgae has recently attracted attention because this could be the most viable means for generating biomass suitable for high-value functional products (Hafting et al. 2012). Given that the water velocity can be regulated in the CFCS, the device is useful for identifying conditions of water motion suitable for increasing the production of any macroalgae species. The results also suggest that the CFCS is useful for research on the physiological ecology of macroalgae because large numbers of individuals can be cultivated in the tank under controlled environmental conditions.
References
Dan A, Ohno M, Matsuoka M (2015) Changes of the research and development on the resources of Undaria and Laminaria in the culture ground of Tokushima coasts. Bull Tokushima Pref Fish Res Inst 10:25–48 (in Japanese with English abstract)
FAO (2010) The state of world fisheries and aquaculture. FAO, Rome, p. 197
Gao K, McKinley K (1993) Use of macroalgae for marine biomass production and CO2 remediation: a review. J Appl Phycol 6:45–60
Gao X, Endo H, Taniguchi K, Agatsuma Y (2013) Genetic differentiation of high-temperature tolerance in the kelp Undaria pinnatifida sporophytes from geographically separated populations along the Pacific coast of Japan. J Appl Phycol 25:567–574
Gerard VA, Mann KH (1979) Growth and production of Laminaria longicruris populations exposed to different intensities of water movement. J Phycol 15:33–41
Hafting JT, Critchley AT, Cornish ML, Hubley SA, Archibald AF (2012) On-land cultivation of functional seaweed products for human usage. J Appl Phycol 24:385–392
Hara M, Akiyama K (1985) Heterosis in growth of Undaria pinnatifida (Harvey) Suringar. Bull Tohoku Reg Fish Res Lab 47:47–50
Hiraoka M, Oka N (2008) Tank cultivation of Ulva prolifera in deep seawater using a new “germling cluster” method. J Appl Phycol 20:97–102
Hurd CL (2000) Water motion, marine macroalgal physiology, and production. J Phycol 36:453–472
Hurd CL, Pilditch CA (2011) Flow-induced morphological variations affect diffusion boundary-layer thickness of Macrocystis pyrifera (Heterokontophyta, Laminariales). J Phycol 47:341–351
Hurd CL, Harrison PJ, Druehl LD (1996) Effect of seawater velocity on inorganic nitrogen uptake by morphologically distinct forms of Macrocystis integrifolia from wave-sheltered and exposed sites. Mar Biol 126:205–214
Ishikawa Y (1991) Analysis of quantitative traits in cultured Wakame for breeding. Fish Genet Breed Sci 16:19–24 (in Japanese)
Israel A, Levy I, Friedlander M (2006) Experimental tank cultivation of Porphyra in Israel. J Appl Phycol 18:235–240
Jensen A (1993) Present and future needs for algae and algal products. Hydrobiologia 260/261:15–23
Johnson CR, Banks SC, Barrett NS, Cazassus F, Dunstan PK, Edgar GJ, Frusher SD, Gardner C, Haddon M, Helidoniotis F, Hill KL, Holbrook NJ, Hosie GW, Last PR, Ling SD, Thomas JM, Miller K, Pecl GT, Richardson AJ, Ridgway KR, Rintoul SR, Ritz DA, Ross DJ, Sanderson JC, Shepherd SA, Slotwinski A, Swadling KM, Taw N (2011) Climate change cascades: shifts in oceanography, species’ ranges and subtidal marine community dynamics in eastern Tasmania. J Exp Mar Biol Ecol 400:17–32
Kirihara S, Nakamura T, Kon N, Fujita D, Notoya M (2006) Recent fluctuations in distribution and biomass of cold and warm temperature species of Laminarialean algae at Cape Ohma, northern Honshu, Japan. J Appl Phycol 18:521–527
Li XJ, Cong YZ, Yang GP, Qu SC, Li ZL, Wang GW, Zhang ZZ, Luo SJ, Dai HL, Xie JZ, Jiang JL, Wang TY (2007) Trait evaluation and trial cultivation of Dongfang no. 2, the hybrid of a male gametophyte clone of Laminaria longissima (Laminariales, Phaeophyta) and a female one of L. japonica. J Appl Phycol 19:139–151
Lobban CS, Harrison PJ (1994) Seaweed ecology and physiology. Cambridge University Press, Cambridge, pp. 294–297
Mann KH (1972) Ecological energetics of the seaweed zone in a marine bay on the Atlantic coast of Canada. 2. Productivity of the seaweeds. Mar Biol 12:1–10
Müller R, Laepple T, Bartsch I, Wiencke C (2009) Impact of oceanic warming on the distribution of seaweeds in polar and cold-temperate waters. Bot Mar 52:617–638
Nabata S, Takiya A, Tada M (2003) On the decreased production of natural kelp, Laminaria ochotensis in Rishiri Island, northern Hokkaido. Sci Rep Hokkaido Fish Exp Stn 64:127–136
Nanba N, Fujiwara T, Kuwano K, Ishikawa Y, Ogawa H, Kado R (2011) Effect of water flow velocity on growth and morphology of cultured Undaria pinnatifida sporophytes (Laminariales, Phaeophyceae) in Okirai Bay on the Sanriku coast, Northeast Japan. J Appl Phycol 23:1023–1030
Neish AC, Shacklock PE, Fox CH, Simpson EJ (1977) The cultivation of Chondrus crispus: factors affecting growth under greenhouse conditions. Can J Bot 55:2263–2271
Niwa K (2015) Experimental cultivation of Undaria pinnatifida for double cropping in Pyropia farms around Akashi Strait, Hyogo Prefecture. Jpn J Phycol (Sorui) 63:90–97 (in Japanese with English abstract)
Niwa K, Harada K (2016) Experiment on forcing cultivation of Undaria pinnatifida sporophytes in the Seto Inland Sea by using free-living gametophytes cultured in laboratory. Jpn J Phycol (Sorui) 64:10–18 (in Japanese with English abstract)
Peteiro C, Freire O (2011) Effect of water motion on the cultivation of the commercial seaweed Undaria pinnatifida in a coastal bay of Galicia, Northwest Spain. Aquaculture 314:269–276
Sato Y, Hirano T, Niwa K, Suzuki T, Fukunishi N, Abe T, Kawano S (2016) Phenotypic differentiation in the morphology and nutrient uptake kinetics among Undaria pinnatifida cultivated at six sites in Japan. J Appl Phycol. doi:10.1007/s10811-016-0857-9
Tatewaki M (1966) Formation of a crustose sporophyte with unilocular sporangia in Scytosiphon lomentaria. Phycologia 6:62–66
Wargacki AJ, Leonard E, Win MN, Regitsky DD, Santos CNS, Kim PB, Cooper SR, Raisner RM, Herman A, Sivitz AB, Lakshmanaswamy A, Kashiyama Y, Baker D, Yoshikuni Y (2012) An engineered microbial platform for direct biofuel production from brown macroalgae. Science 335:308–313
Westermeier R, Patino D, Piel MI, Maier I, Mueller DG (2006) A new approach to kelp mariculture in Chile: production of free-floating sporophyte seedlings from gametophyte cultures of Lessonia trabeculata and Macrocystis pyrifera. Aquac Res 37:164–171
Westermeier R, Patiño D, Müller H, Müller DG (2010) Towards domestication of giant kelp (Macrocystis pyrifera) in Chile: selection of haploid parent genotypes, outbreeding, and heterosis. J Appl Phycol 22:357–361
Yong YS, Yong WTL, Anton A (2013) Analysis of formulae for determination of seaweed growth rate. J Appl Phycol 25:1831–1834
Zhang QS, Tang XX, Cong YZ, Qu SC, Luo SJ, Yang GP (2007) Breeding of an elite Laminaria variety 90-1 through interspecific gametophyte crossing. J Appl Phycol 19:303–311
Zhang J, Liu Y, Yu D, Song H, Cui J, Liu T (2011) Study on high-temperature resistant and high-yield Laminaria variety “Rongfu”. J Appl Phycol 23:165–171
Zuldin WH, Yassir S, Shapawi R (2016) Growth and biochemical composition of Kappaphycus (Rhodophyta) in customized tank culture system. J Appl Phycol 28:2453–2458
Acknowledgements
We sincerely thank Mr. Inoguchi, the former president and director of the Iwate Fisheries Technology Center and Mr. Takahashi, the former director of the Iwate Fisheries Technology Center, for their guidance and suggestions. We also thank Dr. Nanba and Ms. Shinozuka for their kind advice and cooperation in the measurement of nutrients. We thank Mr. Hagiwara of Riken Food Co., Ltd. for his technical support of the tank culture system. This study was funded by the Formation of Tohoku Marine Science Center Project (Technical Development That Leads to the Creation of New Industries) of the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Supplemental Table 1
(PDF 43 kb)
Supplemental Table 2
(PDF 38 kb)
Supplemental Table 3
(PDF 46 kb)
Supplemental Table 4
(PDF 41 kb)
Supplemental Table 5
(PDF 39 kb)
Supplemental Table 6
(PDF 42 kb)
Rights and permissions
About this article
Cite this article
Sato, Y., Yamaguchi, M., Hirano, T. et al. Effect of water velocity on Undaria pinnatifida and Saccharina japonica growth in a novel tank system designed for macroalgae cultivation. J Appl Phycol 29, 1429–1436 (2017). https://doi.org/10.1007/s10811-016-1013-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-016-1013-2