Abstract
Osteoarthritis (OA), in which inflammation plays a crucial role, is the most common joint disease characterized by cartilage degradation. Neferine (Nef), a dibenzyl isoquinoline alkaloid, has shown its anti-inflammatory effects on other inflammatory diseases. Therefore, we hypothesized that Nef might also have an anti-inflammatory effect on OA and explored its effect on IL-1β-treated rat chondrocytes. Sprague Dawley (SD) rat chondrocytes were stimulated with IL-1β (10 ng/ml) and Nef (1, 5, and 10 μM) or IL-1β (10 ng/ml) alone for 24 h. Expression of inducible nitric oxide synthase (iNOS), cyclooxygenase 2 (COX-2), matrix metalloproteinases (MMPs), and thrombospondin motifs-5 (ADAMTS5) was determined by quantitative real-time PCR and Western blotting. Expression of collagen II and aggrecan was examined by Western blotting, immunofluorescence, and safranin O staining. In addition, activation of MAPK and NF-κB signaling pathway was examined by Western blotting, and p65 nuclear translocation was evaluated by immunofluorescence. Nef reduced expression of inflammatory regulators (iNOS and COX-2) in IL-1β-treated chondrocytes. Expression of IL-1β-induced major catabolic enzymes (MMP3, MMP13, and ADAMTS5) was inhibited by Nef. Meanwhile, downregulation of collagen II and aggrecan expression was also ameliorated. Furthermore, Nef dampened abnormal activation of MAPK and NF-κB signaling pathway triggered by IL-1β. Overall, the results above showed that Nef inhibited IL-1β-induced excess production of inflammatory and catabolic factors in rat chondrocytes via inhibiting the MAPK and NF-κB pathways, suggesting a promising pharmacotherapy for OA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Osteoarthritis, the most common systemic musculoskeletal joint disease worldwide, has been investigated for many years, and cartilage degeneration is an important pathological feature of this condition [1, 2]. Obesity and age are two common contributions to the increasing incidence of OA [3]. Furthermore, OA not only affects the physical health of individuals, but also negatively impacts their mental health [4, 5]. However, we currently lack effective disease-modifying medical therapy for OA.
Inflammation is an essential component participating in the progression of cartilage and bone destruction in osteoarthritis [6]. Among numerous pro-inflammatory cytokines, IL-1β is regarded as an essential factor in osteoarthritis [7], and this cytokine significantly accelerates catabolism of articular cartilage [8, 9]. In addition, IL-1β has been reported to be significantly higher in synovial fluid and serum of OA patients than normal individuals [10]. IL-1β increases the secretion of inflammatory mediators such as iNOS and COX-2 [7], and then induces excessive release of proteolytic enzymes, such as ADAMTS and MMPs, which degrade the extracellular matrix (ECM) in the cartilage [11]. Among these proteinases, MMP3, MMP13, and ADAMTS5 have been extensively investigated [12].
Nef is a dibenzyl isoquinoline alkaloid isolated from lotus green seed embryo [13]. Increasing studies have shown that this compound has anti-inflammatory [14], anti-oxidative [15, 16], anti-tumor [17], autophagy-inducing [18, 19], and anti-apoptotic effects [20]. Moreover, Nef also has effects related to osteoporosis, such as inhibiting bone resorption and promoting bone formation [21]. Currently, non-steroidal anti-inflammatory drugs are commonly used for the symptomatic relief of OA. However, some side effects associated with long-term use, such as gastrointestinal and cardiovascular toxicity, have been reported, and these effects are of concerns especially in elderly individuals [22, 23]. Therefore, safe and efficient drugs are essential for long-term therapy of OA. Based on these findings, we explored the anti-inflammatory and chondrocyte-protective effects of Nef in IL-1β-stimulated rat chondrocyte and the underlying chondroprotective mechanism of this alkaloid.
MATERIALS AND METHODS
Chemicals and Reagents
Neferine (HY-N0441) was purchased from MedChemExpress (MCE). Sigma-Aldrich (St. Louis, MO, USA) provided toluidine blue and R&D Systems (501-RL-010, USA) provided rat IL-1β cytokine. Solarbio provided safranin O solution (Beijing, China). Primary antibodies against iNOS, COX-2, MMP-13, ADAMTS5, Aggrecan, and Collagen II were obtained from Abcam (Shanghai, China), components (P-P38, P38, P-JNK, JNK, P-ERK, and ERK) of the MAPK pathway and components (P-P65) of NF-κB pathway were acquired from CST (Beverly, MA, USA). In addition, Proteintech Group (Wuhan, Hubei, China) provided the corresponding primary antibody for MMP-3, GAPDH, and components (P65) of NF-κB pathway. Secondary antibodies, Cy3 and FITC conjugate secondary antibody, collagenase type II, tyrisin, and phosphate buffer saline (PBS) buffer solution were purchased from Boster Biological Technology (Wuhan, Hubei, China).
Chondrocyte Isolation And Culture
The Experimental Animal Center of Huazhong University of Science and Technology provided the SD rats. The knee joint cartilage of 5-day-old SD rats was utilized to obtain chondrocytes as described previously [24]. Briefly, the cartilage was minced into small pieces that were approximately 1 mm in diameter. Then, the samples were digested with 0.25% trypsin for 30 min and re-digested with 0.2% collagenase II for 8 h at 37 °C. Then, suspended chondrocytes were centrifuged at 1700 r/min for 5 min. Subsequently, the cell pellets were resuspended and blended in complete medium (DMEM/Ham’s F-12 (1:1) culture medium, 10% FBS, and 1% penicillin/streptomycin). Finally, the cells were cultured in 25-cm2 flasks at 37 °C with 5% CO2. After cells grew to approximately 90%, passaging and intervention were carried out. To reduce the sampling frequency and maintain stability of the cell phenotype, the second- and third-generation cells were routinely selected for subsequent cell experiments. In addition, rats were euthanized by dislocation of the neck to minimize their pain. In addition, the Ethics and Animal Research Committee of Huazhong University of Science and Technology approved all animal experiments.
Cell Viability Assay
The CCK8 kit was utilized to assess the cytotoxicity of Nef on rat articular chondrocytes. Cells were seeded into 96-well plates (5000/well) overnight. After stimulated with various concentrations (1, 5, 10 μM) of Nef in the presence or absence of IL-1β (10 ng/ml) for 24 h, 10 μl CCK-8 solution was added to each well. Four hours later, a microplate reader (Bio-Rad, Richmond, CA, USA) at 450 nm was utilized to assess the cell viability of chondrocytes.
Toluidine Blue Staining
Toluidine blue staining was used to observe the morphology of chondrocytes in previous reports [25, 26]. Briefly, chondrocytes were seeded in 35 mm plastic dishes (105 cells/well). At 80% confluency, cells were treated with 10 μM Nef for 24 h. Then, the chondrocytes were fixed with 4% paraformaldehyde for 15 min at room temperature after washing with PBS. Subsequently, the cells were gently washed with tri-distilled water and stained with toluidine blue for 2 h. Finally, excess dye was removed by washing with tri-distilled water three times for 5 min each. Then, the morphological characteristics of rat cartilage cells were observed using a microscope (Evos Fl Auto, Life Technologies, USA).
Cellular Safranin O Staining
Safranin O was utilized to evaluate relative content of proteoglycan in chondrocytes according to red staining intensities [26, 27]. Briefly, chondrocytes were seeded in 24 well plate (10,000 cells/well) overnight. Then, cells were stimulated with IL-1β (10 ng/ml) and Nef (10 μM) or IL-1β (10 ng/ml) alone. The medium containing both IL-1β and Nef or IL-1β alone was replaced every 3 days. Removed the medium on the seventh day, which the significant loss of proteoglycan could be observed under inflammatory situation [27, 28]. After fixed in 4% paraformaldehyde for 15 min at room temperature and washed by PBS, the cells were incubated with the safranin O solution (Solarbio, Beijing, China). After a 30-min incubation at room temperature, removed excess dye by washing with PBS three times for 5 min each. The 24 wells were scanned to analyze the macroscopic color change of each well, and photographs of different red staining levels in the cells were captured by a microscope (Evos Fl Auto, Life Technologies, USA).
Immunofluorescence Staining
Immunofluorescence staining was carried out as previously mentioned [29]. In brief, chondrocytes were cultured in 24 well plates (20,000/well) overnight, then stimulated by IL-1β (10 ng/ml) and Nef (10 μM) or IL-1β (10 ng/ml) alone. Time of stimulation for examining P65 and collagen II was 15 min and 24 h, respectively. After rinsing in PBS, the cells were fixed in 4% paraformaldehyde for 15 min, permeated in 0.2% Triton X-100 for 10 min, and subsequently blocked with 1% BSA at room temperature for 1 h, followed by incubation with primary antibodies against P65 and Collagen II (1:200) for 14 h at 4 °C. After washing with PBS three times for 5 min each, the chondrocytes were then incubated with Goat Anti-Rabbit IgG (H + L) Cy3 and FITC Conjugated Secondary Antibody (Boster, Wuhan, China, 1:100 dilution) for 1 h at room temperature and then labeled with DAPI for 10 min to stain the nuclei. Finally, fluorescence degree of collagen II and nuclear import of P65 was observed under a microscope (Evos Fl Auto, Life Technologies, USA).
Quantitative Real-time PCR
Quantitative real-time PCR was utilized to evaluate changes in inflammatory and catabolic indicators at the gene level. Total RNA of the chondrocytes was collected with a RNA isolation kit obtained from Omega (Feiyang, Guangzhou, China) according to the protocol. The concentration of RNA samples was detected by a microplate reader at 260 nm (Epoch, Winooski, USA), and the A260/A280 ratio was calculated to verify the purity of total RNA. Two micrograms of total RNA and a cDNA synthesis kit (TOYOBO, Osaka, Japan) were used to synthesize complementary DNA (cDNA). SYBR® Green Real-time PCR Master Mix (Vasyme, Nanjing, China) was utilized to amplify the cDNA. According to the dissociation curve, we selected primers with high specificity (Table 1) to amplify the cDNA. Real-time PCR reactions were conducted on a CFX Connect Real-time system for 45 cycles at 95 °C (15 s), 59 °C (10 s), and 72 °C (40 s). Relative expression (glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as internal control) levels of each gene was calculated by the comparative 2 -ΔΔCt method.
Western Blotting Analysis
After treatment with IL-1β (10 ng/ml) and Nef (1, 5, and 10 μM) or IL-1β (10 ng/ml) alone for 24 h, cells were lysed with RIPA Lysis Buffer (Boster, Wuhan, China) including 1 % of protease and phosphatase inhibitors for 30 min on ice. Followed by centrifugation at 12000 r/min for 30 min at 4 °C. The protein concentration was measured at 562 nm by a BCA kit (Boster, China) using microplate reader (Bio-Rad, Richmond, CA, USA). Protein samples and markers were separated by 10% SDS-PAGE and transferred to PVDF membranes (Millipore, USA), which were activated by methanol in advance. After blocking with 5% BSA for 1 h, the target proteins in the membrane were incubated with the correspondent primary antibodies for 14 h at 4 °C. Then, the membranes were washed with Tris Buffered Saline Tween (TBST) three times (3 × 10 min). Then, the cells were incubated with goat-anti-rat or goat-anti-rabbit secondory antibody antibodies (1:10000) for 1 h at room temperature, followed by another three washes with TBST (3 × 10 min). The target protein was detected using Supersensitive ECL chemiluminescent substrates (Boster, Wuhan, China) and an imager (Bio-Rad, USA), and the exposure intensity of each protein band was analyzed using the Image J software. Western blotting results were repeated with three independent samples.
Statistical Analysis
All experiments were repeated three times, and chondrocytes were from three different batches of rats. The date was analyzed by GraphPad Prism v.7.01 software (GraphPad Inc., La Jolla, CA, USA). The results of three independent experiments are expressed as the mean ± standard deviation (SD). In addition, the ratio of the target (gene or protein) to the related internal reference in the IL-1β-treated group was defined as unit “1.” One-way ANOVA followed by Tukey’s test was applied to analyze significant differences among groups and a P value less than 0.05 was set to be statistically significant.
RESULTS
Rat Chondrocyte Identification
Collagen II immunofluorescence staining and toluidine blue staining were utilized to identify primary rat chondrocytes we isolated. As shown in Fig. 1a, b, the collagen II was stained with green fluorescence in the cytoplasm, and the proteoglycans were stained with bluish-purple by toluidine blue. In addition, morphology of the normal rat chondrocyte was mainly characterized by polygon (Fig. 1b), which was alike with the previous study [31].
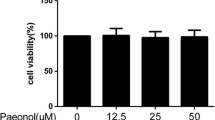
Identification of rat chondrocytes and the cytotoxicity of Nef on rat chondrocytes. a Collagen II immunofluorescence staining of normal rat chondrocytes (scale bar 50 μm). b Toluidine blue staining of normal rat chondrocytes. c Chemical structure of Nef. d Cell viability of rat chondrocytes after Nef treatment alone or both IL-1β and Nef treatment for 24 h (scale bar 400 μm). e Toluidine blue staining of chondrocytes treated with Nef for 24 h (scale bar 400 μm). Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). Significant differences among different groups are defined as #P < 0.05 versus (vs) control group.
Nef Has No Significant Cytotoxicity to Rat Articular Chondrocytes
CCK8 test in Fig. 1d showed that Nef (1, 5, 10 μM) had no significant impact on the vitality of chondrocytes. In addition, there was no significant change in the morphology of chondrocytes treated with Nef (10 μM) according to toluidine blue staining, and the polygon chondrocytes accounted for the vast majority in Fig. 1e. Therefore, concentrations below 10 μM were used in subsequent experiments.
Nef Inhibits Excess COX-2 and iNOS Expression in IL-1β-Treated Rat Chondrocytes
COX-2 and iNOS are two major pro-inflammatory mediators in the pathogenesis of OA. Therefore, the effects of Nef on IL-1β-induced COX-2 and iNOS expression were investigated. Results in Fig. 2a, b showed that chondrocytes treated with IL-1β (10 ng/ml) produced more iNOS and COX-2 than control group at the gene levels, and western blotting results in Fig. 2 (c-d) verified pro-inflammatory effect of IL-1β at the protein level. However, Nef could significantly moderate the inflammation in a dose-dependent way.
Nef inhibits excess expression of COX-2 and iNOS in IL-1β-treated rat chondrocytes. Rat chondrocytes were stimulated with 10 ng/ml IL-1β and Nef (1, 5, and 10 μM) or 10 ng/ml IL-1β alone for 24 h. a qRT-PCR results of COX-2 and iNOS. b Quantification analysis of qRT-PCR results. c Western blotting results of iNOS and COX-2. d Quantification analysis of western blotting results. Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). In addition, GAPDH was regarded as an internal reference. Significant differences among different groups are defined as #P < 0.05 vs control group; *P < 0.05, **P < 0.01, ***P < 0.001 vs IL-1β group.
Nef Weakens IL-1β-Induced Excess Productions of MMP3, MMP13 and ADAMTS5
Among the major catabolic enzymes of the chondrocyte matrix, MMPs and ADAMTS5 are key enzymes. Thus, we assessed the effects of Nef on MMP3, MMP13, and ADAMTS5 expression. The results in Fig. 3a–d showed that IL-1β significantly enhanced expression of MMP3, MMP13, and ADAMTS5 at the gene and protein level. However, Nef exhibited significant inhibiting effect on upregulation of these catabolic enzymes under inflammatory condition, which is in accordance with decrease of pro-inflammatory mediators in Fig. 2.
Nef weakens excess expression of MMP3, MMP13, and ADAMTS5 induced by IL-1β. Rat chondrocytes were treated with 10 ng/ml IL-1β and Nef (1, 5, and 10 μM) or 10 ng/ml IL-1β alone for 24 h. a qRT-PCR results of MMP3, MMP13, and ADAMTS5. b Quantification analysis of qRT-PCR results. c Western blotting results of MMP3, MMP13, and ADAMTS5. d Quantification analysis of western blotting results. Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). Besides, GAPDH was regarded as an internal reference. Significant differences among different groups are defined as #P < 0.05 vs control group; *P < 0.05, **P < 0.01, ***P < 0.001 vs IL-1β group.
Nef Ameliorates the Downregulation of Collagen II and Aggrecan Expression in IL-1β-Treated Chondrocytes
Collagen II and aggrecan are two major components of chondrocyte matrix, and contribute to preserving cell phenotype. Thus, we investigated effect of Nef on abnormal degradation of collagen II and aggrecan in chondrocyte induced by IL-1β. The results in Fig. 4a, b showed that IL-1β significantly decreased the expression of collagen II and aggrecan at protein level, but Nef (1, 5, and 10 μM) could inhibit this process at the protein level. In addition, immunofluorescence staining (Fig. 4c) and safranin O staining (Fig. 4d) of Nef-treated group showed increased fluorescence density and redness than IL-1β-treated group, which were consistence with western blotting results in Fig. 4a, b.
Nef ameliorates the downregulation of collagen II and aggrecan expression in IL-1β-treated chondrocytes. a Western blotting results of collagen II and aggrecan. b Quantification analysis of western blotting results. c Collagen expression was determined by immunofluorescence staining (scale bar 200 μm). d Safranin O staining for proteoglycans deposition in each group after a 7-days incubation (scale bar 200 μm). Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). In addition, GAPDH was regarded as an internal control. Significant differences are defined as #P < 0.05 vs control group; *P < 0.05, **P < 0.01, ***P < 0.001 vs IL-1β group.
Both Nef Pre- and Co-Administration to IL-1β-Stimulated Chondrocytes Can Inhibit Expression of Inflammatory Mediators and Matrix Degrading Enzymes
To verify effect of Nef on IL-1β-treated rat chondrocytes, we chose the 10 μM which showed good inhibitory effect, to explore whether Nef pretreatment was consistent with co- stimulation. At 80% cell confluency, we pretreated chondrocytes with Nef for 2 h, subsequently treated cells with fresh medium contained IL-1β (10 ng/ml) alone for another 24 h. As shown in Fig. 5a, IL-1β significantly increased expression of COX-2, MMP3, ADAMTS5, and decreased expression of collagen II. However, Nef pretreatment could inhibit upregulation of COX-2, MMP3, ADAMTS5, and ameliorates the downregulation of collagen II (Fig. 5b, c), which was consistent with the effect of Nef co-administration (Figs. 2, 3, and 4).
Both Nef pre- and co-administration to IL-1β-stimulated chondrocytes can inhibit expression of inflammatory mediators and matrix degrading enzymes. Chondrocytes were pretreated with Nef for 2 h, subsequently treated cells with fresh medium contained IL-1β (10 ng/ml) alone for another 24 h. a Western blotting results of COX-2, MMP3, ADAMTS5, and collagen II. b Quantification analysis of COX-2. c Quantification analysis of MMP3, ADAMTS5, and collagen II. Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). Besides, GAPDH was regarded as an internal reference. Significant differences among different groups are defined as #P < 0.05 vs control group; *P < 0.05, **P < 0.01, ***P < 0.001 vs IL-1β group.
Nef Inhibits Activation of the MAPK Signaling Pathway Induced by IL-1β
During OA progression, activation of MAPK pathways after treated with IL-1β is regarded as an earlier event in the IL-1β signal transduction [32]. Thus, we first selected several time points (0, 15, 30, 60, and 90 min) to verify the activation process after treatment with IL-1β (10 ng/ml). The results in Fig. 6a, b showed that activation of the MAPK (P38, ERK, JNK) signaling pathway induced by IL-1β was time-dependent, with the strongest activation after 15 min, followed by a gradual weakening. Therefore, we chose the activation peak after 15 min to explore the regulation of Nef on MAPK signaling pathway. As shown in Fig. 6c, d, IL-1β could significantly promote activation of JNK, P38, and ERK, However, Nef could partly moderate this impact by inhibiting the phosphorylation of P38 and ERK but not JNK.
Nef inhibits activation of the MAPK signaling pathway induced by IL-1β. a Western blotting results of P38, P-P38, ERK, P-ERK, JNK, and P-JNK from IL-1β-treated rat chondrocytes at different time points (0, 15, 30, 60, 90 min). b Quantification analysis of western blotting results. c Western blotting results of P38, P-P38, ERK, P-ERK, JNK, and P-JNK from rat chondrocytes after treated with 10 ng/ml IL-1β and Nef (1, 5, 10 μM) for 15 min. d Quantification analysis of Western blotting results. Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). Besides, the non-phosphorylated MAPK pathway (P38, ERK, JNK) protein was regarded as an internal control. Significant differences in Fig. 6b are defined as #P < 0.05 vs control group; *P < 0.05 vs control group; Significant differences in Fig. 6d are defined as #P < 0.05 vs control group, *P < 0.05*, *P < 0.01, ***P < 0.001 vs IL-1β group.
Nef Inhibits IL-1β Mediated Activation of NF-κB Pathway
The activation and inhibition of the NF-κB pathway were determined by immunofluorescence and western blotting. The results in Fig. 7a, b demonstrated that IL-1β significantly enhanced the P-P65 expression, especially after 15 min. However, when the chondrocytes were treated with Nef (5, 10 μM), the phosphorylation level of P65 was significantly decreased compared to IL-1β treated group (Fig. 7c, d). In addition, immunofluorescence staining of P65 further verified the inhibitory effect on signal transduction of NF-κB pathway. As showed in Fig. 7e, P65 was confined to cytoplasm area in unstimulated chondrocytes. After stimulation with IL-1β (10 ng/ml) for 15 min, we observed accumulation of P65 in chondrocyte nuclei compared to control group. However, Nef could partly inhibit this process, which showed decreased entry of P65 into the nucleus.
Nef inhibited IL-1β mediated activation of NF-κB pathway. a Western blotting results of P38, P-P38, ERK, P-ERK, JNK, and P-JNK from IL-1β-treated rat chondrocytes at different points (0, 15, 30, 60, 90 min). b Quantification analysis western blotting results. c Western blotting results of P38, P-P38, ERK, P-ERK, JNK, and P-JNK rom rat chondrocytes after stimulated with 10 ng/ml IL-1β and Nef (1, 5, 10 μM) for 15 min. d Quantification analysis of Western blotting results. e Nuclear translocation of P65/NF-κB was detected by immunofluorescence (scale bar 100 μm). Results from three independent experiments were analyzed by one-way ANOVA followed by Tukey’s test and are presented as means ± SD (n = 3). In addition, the non-phosphorylated NF-κB pathway (P65) protein was regarded as an internal control. Significant differences in Fig. 7b are defined as #P < 0.05 vs control group, *P < 0.05 vs control group; Significant differences in Fig. 7d are defined as #P < 0.05 vs control group, *P < 0.05*, *P < 0.01, ***P < 0.001 vs IL-1β group.
DISCUSSION
Increasing evidence has revealed that the iNOS and COX-2 expression was commonly upregulated by IL-1β during OA [33, 34], and IL-1β separately induced accumulation of NO and Prostaglandin E2 to trigger inflammation. Nevertheless, selective inhibition of iNOS could reduce the local tissue levels of IL-1β, MMPs, iNOS, and COX-2 [35]. Thus, treatments that inhibit iNOS and COX-2 could be a useful intervention for OA. Our data showed that the expression of iNOS and COX-2 in chondrocytes was upregulated after stimulation with IL-1β. However, Nef could significantly reduce this negative effect in IL-1β-treated chondrocytes. These results are consistent with a recent study suggesting that Nef reduces inflammatory response induced by LPS in RAW264.7 macrophages [14]. Thus, the anti-inflammatory effect of Nef may be mediated through inhibiting iNOS and COX-2.
As described above, hyper-catabolism in articular cartilage leads to tissue degeneration, and IL-1β plays a vital role in this pathological process [12]. Aggrecan and collagen II are the two main components of chondrocyte matrix and extracellular matrix, which can be degraded by related proteinases [36]. In healthy or osteoarthritic cartilage, MMPs degrade collagen II and aggrecan, and ADAMTS specifically degrade aggrecan [37,38,39]. Besides, MMP13 and ADAMTS5 knockout mice have been shown to be resistant to cartilage erosion compared with wild-type animals in a surgical osteoarthritis model [37, 40]. In our study, Nef inhibited IL-1β-induced excess expression of MMP3, MMP13, and ADAMTS-5, and decreased expression of collagen II and aggrecan was ameliorated at protein level, consequently maintaining homeostasis of chondrocyte matrix.
Increasing evidence shows that MAPK and NF-κB pathway are considered as key factors in the progression of OA [41,42,43,44]. IL-1β binds its cell receptor in chondrocytes and activates the phosphorylation of MAPK (ERK, p38, JNK) and NF-κB (P65) pathways, subsequently inducing upregulation of pro-inflammatory and pro-catabolic cytokines expression, eventually contributing to disruption of cartilage extracellular matrix [45]. Thus, we explored the relationship between the anti-osteoarthritic effects and the inhibition of MAPK and NF-κB pathway. Our study showed that Nef partly inhibited the IL-1β-induced activation of P38, ERK, and P65 and further suppressed IL-1β-induced P65 transfer from the cytoplasm into the nucleus. Collectively, these results showed that the inhibition of inflammatory mediators and matrix degrading enzymes expression by Nef might be associated with MAPK and NF-κB signaling pathways, and Nef might further inhibit IL-1β-induced signal transduction of NF-κB pathway via inhibiting the nuclear import of P65.
There are some limitations in our study. First, we did not ascertain these effects in vivo and further research is required to verify its anti-osteoarthritic effects in experimental animals. In addition, the current data simply showed that the inhibition of Nef on the inflammatory mediator expression was possibly by attenuating activation of the P38/ERK/MAPK and P65/NF-κB signaling pathway, but we failed to carry out more experiments to provide additional details about the OA-protective effects of Nef. In addition, we did not ascertain the effect of Nef on human chondrocytes due to shortage of normal cartilage. In future research, we plan to turn to other hospitals for human chondrocytes or replace human chondrocytes with the SW1353 cell, which is a cell line with only a very limited potential to mimic primary human chondrocytes [46, 47], to verify anti-inflammatory and anti-catabolism effect of Nef. Therefore, further research is needed to reveal the underlying mechanism and provide applicable results.
Our study showed inhibitory effect of Nef on expression of inflammatory mediators and matrix degrading enzymes in IL-1β-treated rat chondrocytes, and the underlying mechanism was most likely through regulation of MAPK and NF-κB signaling pathway. It indicated that Nef might be a promising agent for inhibiting inflammation and treating OA.
Change history
19 October 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10753-022-01760-8
References
Mandl, L.A. 2019. Osteoarthritis year in review 2018: Clinical. Osteoarthritis and Cartilage 27 (3): 359–364. https://doi.org/10.1016/j.joca.2018.11.001.
Chen, Di, Jie Shen, Weiwei Zhao, Tingyu Wang, Han Lin, John L. Hamilton, and Hee-Jeong Im. 2017. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res 5: 16044. https://doi.org/10.1038/boneres.2016.44.
Vina, E.R., and C.K. Kwoh. 2018. Epidemiology of osteoarthritis: Literature update. Current Opinion in Rheumatology 30 (2): 160–167. https://doi.org/10.1097/BOR.0000000000000479.
Kye, Su-Yeon, and Keeho Park. 2017. Suicidal ideation and suicidal attempts among adults with chronic diseases: A cross-sectional study. Comprehensive Psychiatry 73: 160–167. https://doi.org/10.1016/j.comppsych.2016.12.001.
Veronese, Nicola, Brendon Stubbs, Marco Solmi, Toby O. Smith, Marianna Noale, Cyrus Cooper, and Stefania Maggi. 2017. Association between lower limb osteoarthritis and incidence of depressive symptoms: Data from the osteoarthritis initiative. Age and Ageing 46 (3): 470–476. https://doi.org/10.1093/ageing/afw216.
Pelletier, J.P., J. Martel-Pelletier, and S.B. Abramson. 2001. Osteoarthritis, an inflammatory disease: Potential implication for the selection of new therapeutic targets. Arthritis and Rheumatism 44 (6): 1237–1247. https://doi.org/10.1002/1529-0131(200106)44:6<1237::Aid-art214>3.0.Co;2-f.
Kapoor, M., J. Martel-Pelletier, D. Lajeunesse, J.P. Pelletier, and H. Fahmi. 2011. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nature Reviews Rheumatology 7 (1): 33–42. https://doi.org/10.1038/nrrheum.2010.196.
Appleton, C.T. 2018. Osteoarthritis year in review 2017: Biology. Osteoarthritis and Cartilage 26 (3): 296–303. https://doi.org/10.1016/j.joca.2017.10.008.
Malemud, C.J. 2015. Biologic basis of osteoarthritis: State of the evidence. Current Opinion in Rheumatology 27 (3): 289–294. https://doi.org/10.1097/bor.0000000000000162.
Panina, Svetlana B., Igor V. Krolevets, Natalia P. Milyutina, Alexander B. Sagakyants, Igor V. Kornienko, Anzhelika A. Ananyan, Mikhail A. Zabrodin, Andrey A. Plotnikov, and Valeriy V. Vnukov. 2017. Circulating levels of proinflammatory mediators as potential biomarkers of post-traumatic knee osteoarthritis development. Journal of Orthopaedics and Traumatology 18 (4): 349–357. https://doi.org/10.1007/s10195-017-0473-8.
Loeser, R.F. 2006. Molecular mechanisms of cartilage destruction: Mechanics, inflammatory mediators, and aging collide. Arthritis and Rheumatism 54 (5): 1357–1360. https://doi.org/10.1002/art.21813.
Mueller, M.B., and R.S. Tuan. 2011. Anabolic/catabolic balance in pathogenesis of osteoarthritis: Identifying molecular targets. PM & R : The Journal of Injury, Function, and Rehabilitation 3 (6 Suppl 1): S3–S11. https://doi.org/10.1016/j.pmrj.2011.05.009.
Furukawa, H. 1965. On the alkaloids of Nelumbo Nucifera Gaertn. IX. Alkaloids of loti embryo. (2). Structure of neferine, a new biscoclaurine alkaloid. Yakugaku Zasshi 85: 335–338.
Wu, X., Y. Guo, X. Min, L. Pei, and X. Chen. 2018. Neferine, a bisbenzylisoquinoline alkaloid, ameliorates dextran sulfate sodium-induced ulcerative colitis. The American Journal of Chinese Medicine 46 (6): 1263–1279. https://doi.org/10.1142/S0192415X18500660.
Wu, C., J. Chen, R. Yang, F. Duan, S. Li, and X. Chen. 2019. Mitochondrial protective effect of neferine through the modulation of nuclear factor erythroid 2-related factor 2 signalling in ischaemic stroke. British Journal of Pharmacology 176 (3): 400–415. https://doi.org/10.1111/bph.14537.
Tang, Y.S., Y.H. Zhao, Y. Zhong, X.Z. Li, J.X. Pu, Y.C. Luo, and Q.L. Zhou. 2019. Neferine inhibits LPS-ATP-induced endothelial cell pyroptosis via regulation of ROS/NLRP3/Caspase-1 signaling pathway. Inflammation Research 68: 727–738. https://doi.org/10.1007/s00011-019-01256-6.
Kadioglu, O., B.Y.K. Law, S.W.F. Mok, S.W. Xu, T. Efferth, and V.K.W. Wong. 2017. Mode of action analyses of neferine, a bisbenzylisoquinoline alkaloid of Lotus (Nelumbo nucifera) against multidrug-resistant tumor cells. Frontiers in Pharmacology 8: 238. https://doi.org/10.3389/fphar.2017.00238.
Wong, V.K., A.G. Wu, J.R. Wang, L. Liu, and B.Y. Law. 2015. Neferine attenuates the protein level and toxicity of mutant huntingtin in PC-12 cells via induction of autophagy. Molecules 20 (3): 3496–3514. https://doi.org/10.3390/molecules20033496.
Pham, D.C., Y.C. Chang, S.R. Lin, Y.M. Fuh, M.J. Tsai, and C.F. Weng. 2018. FAK and S6K1 inhibitor, neferine, dually induces autophagy and apoptosis in human neuroblastoma cells. Molecules 23 (12). https://doi.org/10.3390/molecules23123110.
Priya, L.B., R. Baskaran, C.Y. Huang, and V.V. Padma. 2017. Neferine ameliorates cardiomyoblast apoptosis induced by doxorubicin: Possible role in modulating NADPH oxidase/ROS-mediated NFkappaB redox signaling cascade. Scientific Reports 7 (1): 12283. https://doi.org/10.1038/s41598-017-12060-9.
Chen, S., B. Chu, Y. Chen, X. Cheng, D. Guo, L. Chen, J. Wang, Z. Li, Z. Hong, and D. Hong. 2019. Neferine suppresses osteoclast differentiation through suppressing NF-kappaB signal pathway but not MAPKs and promote osteogenesis. Journal of Cellular Physiology. https://doi.org/10.1002/jcp.28857.
Dubreuil, Maureen, Qiong Louie-Gao, Christine E. Peloquin, Hyon K. Choi, Yuqing Zhang, and Tuhina Neogi. 2018. Risk of myocardial infarction with use of selected non-steroidal anti-inflammatory drugs in patients with spondyloarthritis and osteoarthritis. Annals of the Rheumatic Diseases 77 (8): 1137–1143. https://doi.org/10.1136/annrheumdis-2018-213089.
Cooper, Cyrus, Roland Chapurlat, Nasser Al-Daghri, Gabriel Herrero-Beaumont, Olivier Bruyere, Francois Rannou, Roland Roth, Daniel Uebelhart, and Jean-Yves Reginster. 2019. Safety of oral non-selective non-steroidal anti-inflammatory drugs in osteoarthritis: What does the literature say? Drugs & Aging 36: 15–24. https://doi.org/10.1007/s40266-019-00660-1.
Lu, W., J. Shi, J. Zhang, Z. Lv, F. Guo, H. Huang, W. Zhu, and A. Chen. 2016. CXCL12/CXCR4 axis regulates aggrecanase activation and cartilage degradation in a post-traumatic osteoarthritis rat model. International Journal of Molecular Sciences 17 (10). https://doi.org/10.3390/ijms17101522.
Shepard, N., and N. Mitchell. 1976. Simultaneous localization of proteoglycan by light and electron microscopy using toluidine blue OA study of epiphyseal cartilage. Journal of Histochemistry & Cytochemistry 24 (5):621–629. https://doi.org/10.1177/24.5.132503.
Schmitz, N., S. Laverty, V.B. Kraus, and T. Aigner. 2010. Basic methods in histopathology of joint tissues. Osteoarthritis and Cartilage 18 (Suppl 3): S113–S116. https://doi.org/10.1016/j.joca.2010.05.026.
Huang, W., C. Cheng, W.S. Shan, Z.F. Ding, F.E. Liu, W. Lu, W. He, J.G. Xu, and Z.S. Yin. 2019. Knockdown of SGK1 alleviates the IL-1beta-induced chondrocyte anabolic and catabolic imbalance by activating FoxO1-mediated autophagy in human chondrocytes. The FEBS Journal. https://doi.org/10.1111/febs.15009.
Bos, P.K., G.J. van Osch, D.A. Frenz, J.A. Verhaar, and H.L. Verwoerd-Verhoef. 2001. Growth factor expression in cartilage wound healing: Temporal and spatial immunolocalization in a rabbit auricular cartilage wound model. Osteoarthritis and Cartilage 9 (4): 382–389. https://doi.org/10.1053/joca.2000.0399.
Huang, X., Y. Xi, Q. Pan, Z. Mao, R. Zhang, X. Ma, and H. You. 2018. Caffeic acid protects against IL-1beta-induced inflammatory responses and cartilage degradation in articular chondrocytes. Biomedicine & Pharmacotherapy 107: 433–439. https://doi.org/10.1016/j.biopha.2018.07.161.
Mao, Z., P. Wang, Q. Pan, X. Huang, R. Zhang, X. Shang, X. Ma, and H. You. 2018. Pravastatin alleviates interleukin 1beta-induced cartilage degradation by restoring impaired autophagy associated with MAPK pathway inhibition. International Immunopharmacology 64: 308–318. https://doi.org/10.1016/j.intimp.2018.09.018.
Oda, Akinori, Ryota Takamiya, Rin Kaneko, Haruna Yoshida, Yuta Yanagita, Hatsumi Sekiguchi, Yoshihito Nobe, and Kazuaki Muramatsu. 2019. Utility of a rotation/revolution-type agitator for chondrocyte isolation during preparation of engineered cartilage. Journal of Bioscience and Bioengineering 128 (1): 117–122. https://doi.org/10.1016/j.jbiosc.2018.12.010.
Liacini, A., J. Sylvester, W.Q. Li, and M. Zafarullah. 2002. Inhibition of interleukin-1-stimulated MAP kinases, activating protein-1 (AP-1) and nuclear factor kappa B (NF-kappa B) transcription factors down-regulates matrix metalloproteinase gene expression in articular chondrocytes. Matrix Biology 21 (3): 251–262. https://doi.org/10.1016/s0945-053x(02)00007-0.
Pettipher, E.R., G.A. Higgs, and B. Henderson. 1986. Interleukin-1 induces leukocyte infiltration and cartilage proteoglycan degradation in the synovial joint. Proceedings of the National Academy of Sciences of the United States of America 83 (22): 8749–8753. https://doi.org/10.1073/pnas.83.22.8749.
Needleman, P., and P.T. Manning. 1999. Interactions between the inducible cyclooxygenase (COX-2) and nitric oxide synthase (iNOS) pathways: Implications for therapeutic intervention in osteoarthritis. Osteoarthritis and Cartilage 7 (4): 367–370. https://doi.org/10.1053/joca.1998.0237.
Pelletier, J.P., J.C. Fernandes, G. Tardif, V. Lascau-Coman, M. Dupuis, and J. Martel-Pelletier. 1998. Direct in vivo transfer of the IL-1Ra gene in osteoarthritic rabbit knee joints: Prevention of the disease progression. Arthritis and Rheumatism 41 (9): S342–S342.
Simkin, Peter A. 2009. Rethinking the physiology of articular cartilage. Journal of clinical Rheumatology 15 (5): 260–263. https://doi.org/10.1097/RHU.0b013e3181b1d873.
Stanton, H., F.M. Rogerson, C.J. East, S.B. Golub, K.E. Lawlor, C.T. Meeker, C.B. Little, K. Last, P.J. Farmer, I.K. Campbell, A.M. Fourie, and A.J. Fosang. 2005. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434 (7033): 648–652. https://doi.org/10.1038/nature03417.
Song, Ruo-Hua, Micky D. Tortorella, Anne-Marie Malfait, James T. Alston, Zhiyong Yang, Elizabeth C. Arner, and David W. Griggs. 2007. Aggrecan degradation in human articular cartilage explants is mediated by both ADAMTS-4 and ADAMTS-5. Arthritis and Rheumatism 56 (2): 575–585. https://doi.org/10.1002/art.22334.
Neuhold, L.A., L. Killar, W.G. Zhao, M.L.A. Sung, L. Warner, J. Kulik, J. Turner, et al. 2001. Postnatal expression in hyaline cartilage of constitutively active human collagenase-3 (MMP-13) induces osteoarthritis in mice. Journal of Clinical Investigation 107 (1): 35–44. https://doi.org/10.1172/jci10564.
Little, C.B., A. Barai, D. Burkhardt, S.M. Smith, A.J. Fosang, Z. Werb, M. Shah, and E.W. Thompson. 2009. Matrix metalloproteinase 13-deficient mice are resistant to osteoarthritic cartilage erosion but not chondrocyte hypertrophy or osteophyte development. Arthritis and Rheumatism 60 (12): 3723–3733. https://doi.org/10.1002/art.25002.
Fan, Z.Y., B. Bau, H.Q. Yang, and T. Aigner. 2004. IL-1 beta induction of IL-6 and LIF in normal articular human chondrocytes involves the ERK, p38 and NF kappa B signaling pathways. Cytokine 28 (1): 17–24. https://doi.org/10.1016/j.cyto.2004.06.003.
Saklatvala, J. 2007. Inflammatory signaling in cartilage: MAPK and NF-kappa B pathways in chondrocytes and the use of inhibitors for research into pathogenesis and therapy of osteoarthritis. Current Drug Targets 8 (2): 305–313. https://doi.org/10.2174/138945007779940115.
Herrero-Beaumont, Gabriel, Sandra Perez-Baos, Olga Sanchez-Pernaute, Jorge A. Roman-Blas, Ana Lamuedra, and Raquel Largo. 2019. Targeting chronic innate inflammatory pathways, the main road to prevention of osteoarthritis progression. Biochemical Pharmacology 165: 24–32. https://doi.org/10.1016/j.bcp.2019.02.030.
Yang, Da-Wei, Gui-Bin Qian, Ming-Jiu Jiang, Peng Wang, and Kun-Zheng Wang. 2019. Inhibition of microRNA-495 suppresses chondrocyte apoptosis through activation of the NF-kappaB signaling pathway by regulating CCL4 in osteoarthritis. Gene Therapy 26 (6): 217–229. https://doi.org/10.1038/s41434-019-0068-5.
Abramson, S.B. 2008. Osteoarthritis and nitric oxide. Osteoarthritis and Cartilage 16 (Suppl 2): S15–S20. https://doi.org/10.1016/s1063-4584(08)60008-4.
Schaefer, J.F., M.L. Millham, B. de Crombrugghe, and L. Buckbinder. 2003. FGF signaling antagonizes cytokine-mediated repression of Sox9 in SW1353 chondrosarcoma cells. Osteoarthritis and Cartilage 11 (4): 233–241.
Gebauer, M., J. Saas, F. Sohler, J. Haag, S. Soder, M. Pieper, E. Bartnik, J. Beninga, R. Zimmer, and T. Aigner. 2005. Comparison of the chondrosarcoma cell line SW1353 with primary human adult articular chondrocytes with regard to their gene expression profile and reactivity to IL-1beta. Osteoarthritis and Cartilage 13 (8): 697–708. https://doi.org/10.1016/j.joca.2005.04.004.
Funding
National Natural Science Foundation of China (Grant No. 81772390) and the Fundamental Research Funds for the Central Universities (Grant No. 2017KFYXJJ104) funded this research study.
Author information
Authors and Affiliations
Contributions
HY and BN conceived the experiments and wrote the manuscript. BN, XH, and YX carried out the cell experiments. ZM and XC purchased animals and isolated primary rat chondrocytes. RZ and XM analyzed the data and made charts and graphs. HY financed the study.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ni, B., Huang, X., Xi, Y. et al. Neferine Inhibits Expression of Inflammatory Mediators and Matrix Degrading Enzymes in IL-1β-Treated Rat Chondrocytes via Suppressing MAPK and NF-κB Signaling Pathways. Inflammation 43, 1209–1221 (2020). https://doi.org/10.1007/s10753-019-01143-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-019-01143-6