Abstract
Anthropogenic pollution from urban and agricultural activities continually affects freshwater ecosystems in Africa, adversely affecting water quality and biodiversity. In this study, we explored the responses of macroinvertebrate functional feeding groups (FFGs) to an urban-agricultural disturbance in the Niger Delta Region, aiming to examine their utility for long-term monitoring of urban-agricultural disturbance in the region. We sampled 17 sites in 11 rivers between 2008 and 2012. We classified the sites into urban, agricultural, and reference (control) sites based on the watershed land-uses and reach-scale anthropogenic disturbance. Permutational multivariate analysis of variance showed that physical and chemical variables differed significantly between the site groups (P < 0.05) across the land-use types in the study area. Multivariate analysis of similarity (ANOSIM) indicated that urban land-use had more effect on FFGs than agricultural activities, differing significantly from the forested sites (P < 0.05). Macroinvertebrate FFGs such as collectors-filter, collector-gatherers, and scrapers/grazers were identified as sensitive FFGs of urban-agricultural pollution. These FFGs showed strong negative associations with physical and chemical indicators increasing urban-agricultural pollution (e.g. conductivity, nutrients, and turbidity) and favoured by DO at the reference sites. Conversely, shredders and predators were tolerant, indicating significant positive associations with increasing urban-agricultural pollution. They were associated with variables such as temperature, BOD and nitrates. Overall, this study revealed that urban and agricultural pollution differentially affected macroinvertebrate functional feeding groups, and indicator FFGs were identified. This provided further insights on urban-agricultural land-use effects on Afrotropical riverine systems and using FFGs as reliable indicators of ecosystem integrity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Global biodiversity is changing at an alarming rate in response to human land-uses (Foley et al., 2005; da Conceição et al., 2020). In particular, urban-agricultural activities in the surrounding landscape of freshwater ecosystems have been implicated as the leading anthropic activities altering abiotic and biotic characteristics of freshwater ecosystems (Uriarte et al., 2011; Dalu et al., 2020). Agricultural activities, for example, affect aquatic ecosystems through habitat degradation, water abstraction for irrigation, and the removal of natural forests (Horak et al., 2020; Akamagwuna et al., 2021, 2022). Similarly, the rapid development of society, economy, and industrialisation in urban cities affect aquatic ecosystem integrity through the so-called "urban river syndrome" (Li et al., 2021; Oyedotun & Ally, 2021; Yadav et al., 2021). Urban-agricultural land-use causes the input of inorganic sediment, nutrients, and other pollutants such as metals and pesticides to water bodies and alters hydrological regimes, consequently reducing the water quality of freshwater ecosystems, biodiversity extinction, and ecosystem functioning (Davis et al., 2018; Ali & Chidambaram, 2020). For example, urban activities can cause temperature and acidity regimes and significantly modify basal resources supporting food webs and functional feeding groups (FFGs) structure in streams (Dalu et al., 2020; Sitati et al., 2021). These effects on water resources and ecosystem functioning are expected to worsen in Africa with the rapid population growth, projected to double by 2050 (Dao, 2013; World Bank Group, 2020).
Although significant efforts have been put in most developed countries to develop appropriate biomonitoring strategies to assess the impacts of urban-agricultural pollution (Chen et al., 2019; Wang et al., 2019; Tampo et al., 2020), a significant knowledge gap remains in the tropical region. For example, developing biomonitoring tools in the area primarily rely on macroinvertebrate structural assemblages without recourse to their functional ecology. To make things worse, taxonomic keys from the temperate region still form the basis for identification as well as their response to environmental pressures (Mangadze et al., 2019a; Lubanga et al., 2021). These challenges have limited our understanding of aquatic community response to environmental stress and identifying reliable indicators of anthropogenic pressure required to develop effective mitigation strategies for protecting and managing aquatic ecosystems in the region. A few authors have explored a functional trait-based approach in the last decade to monitor water quality deterioration in the region (e.g. Akamagwuna et al., 2019; 2022; Odume, 2020; Edegbene et al., 2021a, b; Edegbene et al., 2022; Matomela et al., 2021). These efforts have been constrained by sparse taxonomic expertise and the availability of trait information (Akamagwuna et al., 2021; Edegbene et al., 2021b; Edegbene et al., 2022). Furthermore, using multiple traits presents enormous challenges as multiple trait interactions have been demonstrated to influence responses of a given trait to anthropogenic stressors (seeVerberk et al., 2013; Pilière et al., 2016; Odume, 2020). Globally, macroinvertebrate FFGs have gained traction in assessing the ecological integrity of freshwater ecosystems, providing excellent insights into pollution studies (Varadinova & Kerakova, 2018). However, we lack explicit understanding regarding macroinvertebrate FFGs responses to urban-agricultural disturbance in tropical climates in the African continent, with the few studies on this subject producing ambiguous outcomes (Akamagwuna & Odume, 2020; Sitati et al., 2021).
Functional feeding groups define macroinvertebrates' behavioural and physiological mechanisms for food acquisition and habitat use in different environmental conditions (Cummins & Klug, 1979; Wallace & Merritt, 1980). The varied ecological and food requirements of macroinvertebrate FFGs make them respond differentially to environmental stressors, making them valuable indicators of aquatic ecosystem health and ecosystem functioning (Vannote et al., 1980; Wallace & Merritt, 1980). For example, studies have found a shift in FFGs structure with the elimination of some FFGs such as filter-feeders in response to urban-agricultural disturbance (Mangadze et al., 2019a). Miserendino & Masi (2010) revealed substantial differences in the biomass of shredders and collector-gatherers in Patagonian streams affected by various land-uses (agriculture and urbanisation). Similarly, Fu et al. (2016) observed that shredder and predator abundances significantly declined in urban streams of Dongjiang River in China. Akamagwuna & Odume (2020) observed differential responses of Afrotropical Ephemeroptera, Plecoptera, and Plecoptera FFGs to fine sediment from agricultural landscapes in the Tsitsa River, South Africa. However, our knowledge of FFGs responses to urban-agricultural pollution is limited to South America (Couceiro et al., 2011; Van Echelpoel et al., 2018) and Asia (Fu et al., 2016).
Anthropogenic pressures such as urban and agricultural land-use practices unique to different regions affect aquatic communities in various ways (Edegbene et al., 2020a; Akamagwuna, 2021). Hence, using classification keys from the temperate region for awarding FFGs to macroinvertebrates in a bid to use them for long-term biomonitoring can sometimes lead to ambiguous outcomes and results (Masese et al., 2014; Mangadze et al., 2019b; Sitati et al., 2021). These challenges have limited our ability to develop an appropriate FFG biomonitoring tool unique to the tropics for long-term monitoring of anthropogenic pressures. Developing a regional FFG bioassessment tool can allow us monitor ecological conditions in the region, avoiding the need to use whole macroinvertebrate taxa when resources and taxonomic expertise are scarce. Therefore, understanding macroinvertebrate FFGs' relationships with ecological and environmental descriptors (e.g. urban-agricultural land-use) are urgently needed to develop reliable FFG classification unique to the tropics.
In this study, we explored the response patterns of macroinvertebrate FFGs to urban-agricultural land-use induced water quality changes in the Niger Delta Region. We examined the utility of macroinvertebrate FFGs as reliable indicators of urban-agricultural disturbance in the area. The Niger Delta Region is influenced by different land-use practices, most notably agricultural and urban land-uses, which have been demonstrated to significantly affect the river systems' ecological integrity (Matemilola et al., 2018). However, an explicit understanding of how these activities affect the structure and function of the river systems is limited, hindering efforts to develop a reliable management strategy for water quality monitoring in the area. More importantly, the continued degradation of the river ecosystems occasioned by human activities in the region has been noted to significantly hamper the sustainable delivery of critical ecosystems services to humans and animals in the areas, drawing us far from achieving the global goals of wellbeing and livelihood for the communities around the rivers. Our hypothesis was that due to environmental filtering, macroinvertebrate FFGs such as predators, shredders and filter-feeders that are more susceptible to reduced litter input, clogging effects, and visual impairment will decrease with changes in water quality resulting from urban-agricultural-induced disturbances such as habitat fragmentation and deforestation, increased sedimentation and turbidity. Second, we hypothesised that agricultural and urban-induced disturbance would differentially influence FFGs, with urban disturbance having more harmful effects on FFGs due to intensive pressure from urban activities.
Materials and methods
Study area and samplings sites
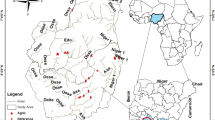
This study was undertaken in 11 rivers in the Niger Delta Region, with a watershed area of 35,340 km2. The rivers (Anwai, Ogba, Orogodo, Ossiomo, Umaluku, Edor, Eriora, Ethiope, Obosh, Owan, and Umu) are situated in the Delta and Edo administrative boundaries of the Niger Delta Region of Nigeria (Fig. 1). The rivers are tributaries to the Niger River, emptying into the Atlantic Ocean at the Gulf of Guinea (Fig. 1). These rivers flow through urban and agricultural land, where water is abstracted for irrigation (Edegbene et al., 2020a). Urban land-use in the area mainly includes urban development and industrialisation related to oil exploration (Ukhurebor et al., 2021; Umar et al., 2021). Urban expansion in the region is accelerating with increasing rural–urban migration (Edegbene et al., 2021b). Agriculture consists mainly of subsistence (crop cultivation and fisheries) and commercial (maize, yam, rice, cassava, sugarcane, and pineapples production; Ekanem & Nwachukwu, 2015).
Study area map showing the location of sampling points within the Niger Delta Region of Nigeria. Site 1 = Orogodo River, Site 2 = Umaluku River 1, Site 3 = Umaluku River 2, Site 4 = Anwai River, Site 5 = Edor River 1, Site 6 = Edor River 2, Site 7 = Ethiope River 1, Site 8 = Ethiope River 2, Site 9 = Ogba River 1, Site 10 = Ogba River 2, Site 11 = Eriora River, Site 12 = Ossiomo River 1, Site 13 = Ossiomo River 2, Site 14 = Obosh River 1, Site 15 = Obosh River 2, Site 16 = Umu River, Site 17 = Owan River
Average annual rainfall in the Nigeria Delta Region ranges from 1250 mm in the evergreen forests of the central region to over 2900 mm in the moist tropical forests (Igweze et al., 2014). The area is characterised by two distinct seasons (wet and dry; Edegbene et al., 2021b; Edegbene et al., 2022). The wet season starts from April to September, and the dry season is between October and March (Edegbene et al., 2021b). The annual temperature is about 28 °C (Tonkin et al., 2016). The underlying geology is shales, clay, loam, and sandy soil (Onyena et al., 2021).
We collected both physical and chemical variables and macroinvertebrate samples once a month from 17 sites (Sites 1–17) in the 11 rivers using objective criteria and best professional judgement to select the least impacted sites as control (reference) sites (Hughes et al., 1986; Stoddard et al., 2006; Whittier et al., 2007). Samples were collected between 2008 and 2012. We collected physical and chemical variables from the 17 sites from 2008 to 2012. For each year, sampling was done monthly covering three months in the dry season and three months in the wet season. We collected physical and chemical variables and macroinvertebrate samples once every month, covering three months both dry and wet seasons from 2008 to 2012 from 17 sites (Sites 1–17) in the 11 rivers. In 2008, sampling was done between March and August (6 months). In 2009, sampling was done between February and July (6 months). For 2010, sampling was done from June to December (7 months), while in 2011 we sampled between January and May (5 months). Finally, in 2012, we sampled from March to September (7 months). Overall sampling was carried out for 31 months in the entire study period. The sites were carefully selected to represent an increasing gradient of urban-agricultural disturbance and further confirmed by the water physical and chemical variables collected from the sites (Fig. 2). Control (reference) sites (Sites 14, 15, 16 and 17) were identified as those with no industrial and agricultural activities with > 80% forest land in their catchments (see Sitati et al., 2021). The four sites identified as reference sites, include Obosh River 1, Obosh River 2, Owan River and Umu River. The remaining 13 sites that did not meet reference criteria were classified as either urban (Sites 1–8) or agricultural (Sites 9–13) disturbed sites. Urban sites (Sites 1–8) were classified as those covered by > 70% human settlements and forest < 30% while agricultural sites had > 70% agricultural lands. We determined the main land-uses and cover in the rivers of each site through manual interpretation of high-resolution images from Google Earth (Google, 2019), and combined with knowledge of the field observation data and experience of the catchments.
Physical and chemical variables sampling
We collected physical and chemical variables from the 17 sites once every month, covering both dry and wet seasons from 2008 to 2012. Dissolved oxygen (DO) was taken using YSI 55 DO meter, whereas conductivity (EC), pH, and temperature were taken using the portable multi-meter analyser HANNA HI 9913001/1. We measured water depth (meters) using a calibrated rod and followed Gordon et al. (1994) approach to measure water flow velocity. Five-day biochemical oxygen demand (BOD5), nutrient variables, including nitrate and phosphate were measured by collecting separate water on each sampling occasion in a sterile bottle of 500 ml and analysed in the laboratory within 24 h of collection using a standard method by American Public Health Association (APHA, 1995).
Macroinvertebrate sampling
Concurrently with physical and chemical sampling, we collected macroinvertebrates covering both dry and wet seasons from 2008 to 2012 following the South African Scoring System version 5 protocol (SASS5; Dickens and Graham, 2002). These samples were collected quantitatively by kick-sampling method using a 1.5 m long using kick-net (dimension 30 × 30 cm, 500 μm). Four replicates were collected from all biotopes recognised by Dickens and Graham (2002); we spent 4 minutes sampling each biotope. Biotopes are the microhabitats inhabited by macroinvertebrates, including stone, vegetation, and sediments. Stone biotope is defined as pebbles and cobbles (2–25 cm), and boulders (> 25 cm) situated in riffles (stones in currents) and pools (stones out of current). Vegetation biotopes comprised marginal vegetation growing on the river edge and fringing into the river, and aquatic vegetation submerged in the main river channel. Sediment biotope was gravels (small stones usually less than 2 cm in diameter), sand and mud that are less than 2 mm and 0.06 mm, respectively. We stored the collected samples in 70% ethanol; transported them to the laboratory for taxonomic sorting, identification, and abundance counting. Macroinvertebrates samples were identified to families using taxonomic keys by Day and de Moor (2002), De Moor et al. (2003) and Merrit et al. (2008). Based on studies, each macroinvertebrate taxon was placed into one of six FFG classes, including filter-feeders, collector-feeders, scrapers, gatherers, predators, and shredders (Merritt & Cummins, 1996; Cummins et al., 2005; Merritt et al., 2008).
Data analysis
Based on Euclidean distance, the canonical analysis of principal coordinates (CAP; Anderson et al., 2008) was used to assess the effects of sites on physical and chemical variables. This analysis confirmed the site classification into urban, and agricultural sites based on land-use cover. We tested the differences in physical and chemical variables between the site categories (urban, and agricultural site categories) using the permutational multivariate analysis of variance (PERMANOVA; Anderson et al., 2008). We tested the assumption of analysis of variance using Levene's and Kolmogorov's tests before PERMANOVA. Univariate indices indicated changes in each FFG community composition between urban and agricultural site groups; therefore, the multivariate analyses of similarity (ANOSIM) were performed to check if this translated into shifts in the whole FFG community. Canonical correspondence analysis (CCA) was used to examine the correlations between macroinvertebrate FFGs and physical and chemical variables across the three sites groups (reference (control), agriculture and urban). The detrended canonical analysis was used to examine the gradient of FFGs community data before CCA. Monte-Carlo permutation test was further used to confirm the level of significance of the FFGs between the land-uses. Except for PERMANOVA that was conducted in Primer version 6, we performed all analyses in R (R Core Team, 2020).
Results
Water quality
The mean, standard deviation, and range values of the physical and chemical variables analysed for the land-use during the study are presented in Supporting information, Table S1. The canonical analysis of principal coordinates (CAP) revealed that the physical and chemical variables differed among urban, agricultural land-uses and the reference (control) sites (Fig. 2). The analysis revealed that 88% of the sites were correctly classified into their parent groups, supporting the prior site classification into land-use types (Table 1). The agriculturally impacted sites were clearly separated from other sites, whereas the urban sites clustered closely with the control (reference) sites (Fig. 2). PERMANOVA showed that the differences in physical and chemical variables between land-use types were highly significant (F = 2.2; P = 0.04; Table 2).
Macroinvertebrate functional structure in the Niger Delta ecoregion
A total of 55 macroinvertebrate families, represented by 11 orders, were identified across the sampling site categories during the study period (see Edegbene et al., 2020b). Generally, relative abundances of FFGs decreased from the Reference (Control) sites to Urban sites, with predators as the most predominant in the study area compared to other feeding groups, representing 34%, 58%, and 53% of FFGs recorded in the Urban, Agricultural, and Reference (Control), respectively (Fig. 3). Shredders (< 3%) were the least occurring FFGs in the Niger Delta Region; this was followed by scrapers that occurred less than 17% at all sites (Fig. 3). ANOSIM indicated that functional organisation of macroinvertebrates varied significantly between the land-uses (Global R-statistic = 0.25, P = 0.021; Table 3), and pairwise ANOSIM revealed a significant difference between Urban and Reference (Control) sites only (R-statistics = 0.49; P = 0.016). These findings suggest a stronger effect of urban disturbance on the functional structure of macroinvertebrates than agricultural disturbance.
Macroinvertebrate FFG responses to urban-agricultural pollution
The CCA results indicated distinct spatial distribution patterns of macroinvertebrate functional composition associated with physical and chemical variables (Fig. 4). The first two axes of the CCA model with eigenvalues of 0.04 and 0.03, respectively, explained 74% of the total relationships between physical and chemical variables and macroinvertebrate FFGs (Table 4). The Monte-Carlo test showed that this explained relationship was statistically significant (Table 4), indicating a good ordination model. The CCA plot showed that collector-filters, collector-gatherers, and scrappers were positively associated with the reference (control) sites (Sites 14, 15 and 17), favoured by increasing DO, and negatively associated with nutrients (e.g. nutrients, phosphate and nitrates; Fig. 4). On the other hand, FFGs such as shredders, grazers, and predators indicated strong negative associations with conductivity at the urban sites. Nevertheless, these FFGs (shredders, grazers and predators) were positively associated with the agriculturally impacted sites, and temperature, depth, BOD and nitrates, flow velocity (Fig. 4). These findings further suggest urban-induced stress affected FFGs in this study than agricultural disturbance.
Canonical correspondence analysis (CCA) of the relationships between physical and chemical variables and macroinvertebrate functional feeding groups (FFGs) in the Niger Delta Region. Temp temperature, DO dissolved oxygen, BOD biochemical oxygen demand. Site 1 = Orogodo River, Site 2 = Umaluku River 1, Site 3 = Umaluku River 2, Site 4 = Anwai River, Site 5 = Edor River 1, Site 6 = Edor River 2, Site 7 = Ethiope River 1, Site 8 = Ethiope River 2, Site 9 = Ogba River 1, Site 10 = Ogba River 2, Site 11 = Eriora River, Site 12 = Ossiomo River 1, Site 13 = Ossiomo River 2, Site 14 = Obosh River 1, Site 15 = Obosh River 2, Site 16 = Umu River, Site 17 = Owan River
Discussion
In this study, we explored the response patterns of macroinvertebrate FFGs to an urban and agricultural disturbance in 17 Sites from 11 rivers in the Niger Delta region, Nigeria. The study revealed that macroinvertebrates FFGs exhibited varying responses to physical and chemical indicators of urban- agricultural disturbance in the area, with urban disturbance having more substantial impacts on macroinvertebrates' FFGs. These results are congruent with studies that have reported significant effects of urban and agricultural disturbance on the structure and functioning of river ecosystems in the tropical region (Akamagwuna et al., 2019; Odume, 2020; Sitati et al., 2021) and elsewhere (Fierro et al., 2019; Kumari & Maiti, 2020). For example, Edegbene et al. (2020a, b) observed a significant influence of urban activities on macroinvertebrate assemblages in Nigeria's Niger Delta Region. Similarly, a recent study by Onyena et al. (2021) to examine the water quality and the assemblages of macroinvertebrates in the Chanomi Creek, Niger Delta, observed differential distribution of macroinvertebrates, with reduced abundance of sensitive filter-feeding taxa from organic pollution in the area.
Macroinvertebrate FFGs structure decreased in control (reference) sites compared to the urban and agricultural sites in the Niger Delta Region, with urban sites having stronger effects on FFGs compared to agricultural sites. The stronger effects of urban disturbance on macroinvertebrate FFGs than agricultural activities suggest that urban practices (e.g. dredging, industrial and waste-water discharges, and flow alteration) may have severe consequences on the functioning of the river systems. Urban rivers are prone to run-off and leakages from sewage facilities and waste disposal from agro-industrial activities (Aura et al., 2010), leading to anoxic and acidic conditions in-stream habitat through increased nutrient and other harmful contaminants. The more substantial effects of urban activities than agricultural activities may be attributed to the combined pollution from riparian habitat degradation and point source pollution from waste-water treatment plants and other industrial activities in urban cities (Kuzmanovic et al., 2017). The adverse effects of urban activities in Afrotropical rivers and their aquatic communities, including FFGs, have been observed in previous studies (Masese et al., 2014). Sitati et al. (2021) recently examined shifts in taxon richness, abundance and biomass of macroinvertebrate functional feeding groups (FFGs) in Kenyan rivers affected by urban and agricultural land-uses. Moreover, they found urban land-use to significantly influence macroinvertebrate FFGs. Macroinvertebrate FFGs are known to be highly sensitive to habitat degradation (i.e. reduced canopy), sedimentation (i.e. organic particles), and water quality (Fu et al., 2016; Gebrehiwot et al., 2017; Fierro et al., 2019; Mangadze et al., 2019b), suggesting why they decreased significantly in the urban sites compared to the agricultural and reference sites (forested sites).
The differential response of macroinvertebrate FFGs to an urban-agricultural disturbance on macroinvertebrate FFGs was evident in the CCA ordination results, with macroinvertebrates displaying FFGs-specific responses to physical and chemical indicators of urban-agricultural pollution (Fig. 4). We identified collector-filters and collector-gatherers as sensitive indicators of urban-agricultural pollution, indicating strong negative associations with increasing urban-agricultural pollution at urban impacted sites (Site 10 and 16; Fig. 4). Collector and filter-feeders dominate areas impacted by high human disturbance level, which reflect the relatively elevated accumulation of fine particulate organic matter in bed sediment and water column of such habitats (Miserendino & Masi, 2010). However, at elevated levels of fine organic sediments in perturbed areas, macroinvertebrate FFGs such as filter-feeders and collector-gatherers are negatively impacted by clogging and abrasion of sensitive parts (Jones et al., 2012; Akamagwuna et al., 2019). Fine sediment accumulation in the impacted sites was likely beyond the acceptable background level (Bilotta & Brazier, 2008; Jones et al., 2012), thereby affecting collectors in the impacted sites in the Niger Delta Region. Furthermore, this finding may be partly because filterers in our study are primarily caddisflies (e.g. Hydropsychidae) that require the stable coarse substrate net and case attachment (Statzner & Béche, 2011).
Concerning the grazers/scrapers, it was surprising that they proved sensitive to urban-agricultural pollution as increased nutrients and organic pollution stimulate algal and macrophyte growth, primary food sources for the grazers/scrapers (Odume, 2020). Scrapers consist of taxa exhibiting diverse feeding modes, and inhabit the trophic roles of herbivores, detritivores and generalist feeders (Mangadze et al., 2019a). The different environmental conditions in streams can cause macroinvertebrates to change their feeding mode (Rawer-Jost et al., 2000; Kelly et al., 2002). Moreover, taxa that are taxonomically linked can have different diets in different geographical regions (Tomanova et al., 2006). As such, researchers have alluded to the fact that some functional categorisation of macroinvertebrates is not well established (Tomanova et al., 2006). Thus, grouping taxa into a single trophic category based on FFG assignment may not be relevant (Hawkins & MacMahon, 1989). This may have affected the results of scrapers observed in the Niger Delta Region. This result emphasises a clear urgent need to improve and standardise FFG grouping and to develop FFG classifications unique to the tropical region. Nevertheless, grazers and scarpers' negative response to urban-agricultural pollution has been observed in studies in the tropics (Odume, 2020) and elsewhere (Van Echelpoel et al., 2018). The studies attributed the sensitivity of grazers and scrapers to increased unstable habitat in impacted sites.
On the other hand, shredders, predators and grazers significantly increased in control (reference) sites compared to the urban and agriculturally impacted sites. Shredder abundance is known to dominate in forested sites compared to sites in the other land-uses (e.g. urban and agriculture), as they are sensitive to reduced quality and quantity of detritus through deforestation and clearance of indigenous riparian vegetation, water pollution and habitat disturbance caused by farming activities and urbanisation in the region (Sitati et al., 2021). However, macroinvertebrate shredders such as Potamonautidae are known to have wide habitat distribution and a high degree of tolerance, especially to pollutants and habitat modification (Cumberlidge et al., 2008; Cumberlidge et al., 2009). Furthermore, shredders were the least dominant FFG recorded, and predatory taxa such Tipulidae were among the few shredders observed in the Niger Delta Region. The majority of the predatory shredders use specialise respiratory organs like the trachea and spiracles for intake of atmospheric oxygen, conferring them resilience to depleted DO, and hence their significant association with the urban-agricultural impacted sites and nutrients in the Niger Delta. In a study to investigate the influence of land-use practices on physical and chemical characteristics and macroinvertebrate FFGs in the Dongjiang River, southeast China, Fu et al. (2016) found predator abundance significantly increased in disturbed sites. Similarly, Odume (2020) observed increased proportions of predatory taxa in polluted urban sites in the Swartkops River, South Africa, and Compin and Céréghino (2007) detected significant increases in the proportions of predators in an agricultural landscape in the Adour-Garonne stream system in France. The combination of results from these studies, taken together with our results from the Niger Delta Region, supports the conclusion that the abundance of predatory taxa can indicate deteriorating water quality.
Conclusions
In the present study, we examined the functional composition responses of macroinvertebrates to an urban-agricultural disturbance in the Niger Delta region to identify FFG indicators of urban-agricultural disturbance. We observed differential effects of urban-agricultural pollution on macroinvertebrate FFGs, and these influences were stronger for urban disturbance compared to agricultural disturbance. Macroinvertebrate FFGs such as collector-filters and collector-gatherers were sensitive to urban-agricultural disturbance, showing strong positive associations to the reference sites and DO. Conversely, shredders, predators, and grazers/scrapers were tolerant FFGs of urban-agricultural disturbance. These FFGs were positively associated with physical and chemical indicators of urban-agricultural pollution such as EC, nutrients and BOD5. This finding indicated that urban-agricultural development in the Niger Delta has deteriorating effects on the biodiversity and functioning of natural ecosystems in this region. Overall, our study provides further insights and evidence of the potential use of macroinvertebrate FFGs as indicators of water quality impacted by urban-agricultural pollution.
Data availability
Dataset used in this study will be made available upon request by the corresponding without hesitation.
References
Akamagwuna, F. C., 2021. Application of Macroinvertebrate-Based Biomonitoring and Stable Isotopes for Assessing the Effects of Agricultural Land-Use on River Ecosystem Structure and Function in the Kat River, Eastern Cape, Rhodes University, Rhodes:, 4.
Akamagwuna, F. C. & O. N. Odume, 2020. Ephemeroptera, Plecoptera and Trichoptera (EPT) functional feeding group responses to fine grain sediment stress in a river in the Eastern Cape, South Africa. Environmental Monitoring and Assessment 2: 1–11.
Akamagwuna, F. C., P. K. Mensah, C. F. Nnadozie & O. N. Odume, 2019. Trait- based responses of Ephemeroptera, Plecoptera, and Trichoptera to sediment stress in the Tsitsa River and its tributaries, Eastern Cape, South Africa. River Research and Applications 4: 1–14.
Akamagwuna, F. C., P. Ntloko, A. O. Edegbene & O. N. Odume, 2021. Are Ephemeroptera, Plecoptera and Trichoptera traits reliable indicators of semi - urban pollution in the Tsitsa River, Eastern Cape Province of South Africa ? Environmental Monitoring and Assessment. https://doi.org/10.1007/s10661-021-09093-z.
Akamagwuna, F. C., O. N. Odume & N. B. Richoux, 2022. Exploring the community structure of Afrotropical macroinvertebrate traits and ecological preferences along an agricultural pollution gradient in the Kat River, Eastern Cape, South Africa. Ecological Indicators 135: 108570.
Ali, A. & S. Chidambaram, 2020. Assessment of trace inorganic contaminates in water and sediment to address its impact on common fish varieties along Kuwait Bay. Environmental Geochemistry and Health. https://doi.org/10.1007/s10653-020-00559-6.
Anderson, M., R. N. Gorley & K. R. Clarke, 2008. PERMANOVA + for PRIMER User Manual. PRIMER-E Ltd1, 1, 1:218 pp.
APHA, 1995. Standard Methods for the Examination of Water and Wastewater, American Public Health Association, Washington, DC:
Aura, M. C., P. O. Raburu & J. Herrmann, 2010. A preliminary macroinvertebrate index of biotic integrity for bioassessment of the Kipkaren and Sosiani Rivers, Nzoia River Basin, Kenya. Lakes & Reservoirs: Science, Policy and Management for Sustainable Use 15: 119–128.
Bilotta, G. S. & R. E. Brazier, 2008. Understanding the influence of suspended solids on water quality and aquatic biota. Water Research 42(12): 2849–2861.
Chen, K., A. R. Rajper, R. M. Hughes, J. R. Olson, H. Wei & B. Wang, 2019. Incorporating functional traits to enhance multimetric index performance and assess land use gradients. Science of the Total Environment 691: 1005–1015.
Compin, A. & R. Céréghino, 2007. Spatial patterns of macroinvertebrate functional feeding groups in streams in relation to physical variables and land-cover in Southwestern France. Landscape Ecology 22(8): 1215–1225.
Couceiro, S. R. M., N. Hamada, B. R. Forsberg & C. Padovesi-Fonseca, 2011. Trophic structure of macroinvertebrates in Amazonian streams impacted by anthropogenic siltation. Austrian Ecology 36: 628–637.
Cumberlidge, N., R. V. Sternberg & S. R. Daniels, 2008. A revision of the higher taxonomy of the Afrotropical freshwater crabs (Decapoda: Brachyura) with a discussion of their biogeography. Biological Journal of the Linnean Society 93: 399–413.
Cumberlidge, N., P. K. L. Ng, D. C. J. Yeo, C. Magalhaes, M. R. Campos, F. Alvarez, T. Naruse, S. R. Daniels, L. J. Esser, F. Y. K. Attipoe, F.-L. Clotilde-Ba, W. Darwall, A. McIvor, J. E. M. Baillie, B. Collen & M. Ram, 2009. Freshwatercrabs and the biodiversity crisis: Importance, threats, status, and conservation challenges. Biological Conservation 142(8): 1665–1673.
Cummins, K. W. & J. M. Klug, 1979. Feeding ecology of stream invertebrates. Annual Review of Ecology and Systematics 10: 147–172.
Cummins, K. W., R. W. Merritt & P. C. N. Andrade, 2005. The use of invertebrate functional groups to characterize ecosystem attributes in selected streams and rivers in south Brazil. Studies on Neotropical Fauna and Environment 40: 69–89.
da Conceição, A. A., E. F. Albertoni, S. V. Milesi & L. U. Hepp, 2020. Influence of anthropic impacts on the functional structure of aquatic invertebrates in subtropical wetlands. Wetlands. https://doi.org/10.1007/s13157-020-01317-1.
Dalu, T., R. N. Cuthbert, J. C. Taylor, M. L. Magoro, O. L. F. Weyl, P. W. Froneman & R. J. Wasserman, 2020. Benthic diatom-based indices and isotopic biomonitoring of nitrogen pollution in a warm temperate Austral river system. Science of the Total Environment 748: 142452.
Dao, M., 2013. Population and economic growth in Africa - Popolazione e crescita economica in Africa. Economia Internazionale/international Economics 66(2): 215–230.
Davis, S. J., D. Óhuallacháin, P. E. Mellander, A. M. Kelly, C. D. Matthaei, J. J. Piggott & M. Kelly-Quinn, 2018. Multiple-stressor effects of sediment, phosphorus and nitrogen on stream macroinvertebrate communities. Science of the Total Environment 637–638: 577–587.
Day, J. A. & I. J. de Moor, 2002. Guides to the freshwater invertebrates of southern Africa. Volume 6: Arachnida and Mollusca (Araneae, Water Mites and Mollusca). Water Research Commision, 6. WRC Report No. TT 182/02.
De Moor, I. J., J. A. Day & F. C. De Moor, 2003. Guides to the freshwater invertebrates of southern Africa. Volume 8: In- secta II: Hemiptera, Megaloptera, Neuroptera, Trichoptera and Lepidoptera. Water Research Commision, 8. WRCReport No. TT 214/03.
Dickens, C. W. & P. M. Graham, 2002. The South African scoring system (SASS) version 5 rapid bioassessment method for Rivers. African Journal of Aquatic Science 27(1): 1–10.
Edegbene, A. O., F. C. Akamagwuna, O. N. Odume, F. O. Arimoro, T. T. Edegbene Ovie, E. C. Akumabor, E. Ogidiaka, E. A. Kaine & K. H. Nwaka, 2022. A macroinvertebrate-based multimetric index for assessing ecological condition of forested stream sitesd raining Nigerian urbanizing landscapes. Sustainability 14: 11289. https://doi.org/10.3390/su141811289.
Edegbene, A. O., F. O. Arimoro & O. N. Odume, 2020a. Exploring the distribution patterns of macroinvertebrate signature traits and ecological preferences and their responses to urban and agricultural pollution in selected rivers in the Niger Delta ecoregion, Nigeria. Aquatic Ecology 54(2): 553–573. https://doi.org/10.1007/s10452-020-09759-9.
Edegbene, A. O., F. O. Arimoro & O. N. Odume, 2020b. How does urban pollution influence macroinvertebrate traits in forested riverine systems? Water 1: 2–17. https://doi.org/10.3390/w12113111
Edegbene, A. O., F. O. Arimoro, O. N. Odume, E. Ogidiaka & U. N. Keke, 2021a. Can macroinvertebrate traits be explored and applied in biomonitoring riverine systems draining forested catchments? Frontiers in Water 3: 607556.
Edegbene, A. O., O. N. Odume, F. O. Arimoro & U. N. Keke, 2021b. Identifying and classifying macroinvertebrate indicator signature traits and ecological preferences along urban pollution gradient in the Niger Delta. Environmental Pollution 281: 117076.
Ekanem, J. & I. Nwachukwu, 2015. Sustainable agricultural production in degraded oil producing and conflict prone communities of Niger Delta, Nigeria. Journal of Agriculture and Sustainability 8(1): 14–28.
Fierro, P., C. Valdovinos, I. Arismendi, G. Díaz, A. Jara-Flores, E. Habit & L. Vargas-Chacoff, 2019. Examining the influence of human stressors on benthic algae, macroinvertebrate, and fish assemblages in Mediterranean streams of Chile. Science of the Total Environment 686: 26–37.
Foley, J. A., R. DeFries, G. P. Asner, C. Barford, G. Bonan, S. R. Carpenter, F. S. Chapin, M. T. Coe, G. C. Daily, H. K. Gibbs, J. H. Helkowski, T. Holloway, E. A. Howard, C. J. Kucharik, C. Monfreda, J. A. Patz, I. C. Prentice, N. Ramankutty & P. K. Snyder, 2005. Global consequences of land use. Science 309(5734): 570–574.
Fu, L., Y. Jiang, J. Ding, Q. Liu, Q. Z. Peng & M. Y. Kang, 2016. Impacts of land use and environmental factors on macroinvertebrate functional feeding groups in the Dongjiang River basin, southeast China. Journal of Freshwater Ecology 31(1): 21–35.
Gebrehiwot, M., A. Awoke, A. Beyene, D. Kifle & L. Triest, 2017. Macroinvertebrate community structure and feeding interactions along a pollution gradient in Gilgel Gibe watershed, Ethiopia: Implications for biomonitoring. Limnologica 62: 68–76.
Google Earth version 6.1, 2019. Sattelite imagery of rivers in the Niger Delta region of Nigeria available. [available on internet at https://earth.google.com/intl/earth].
Gordon, N.D., T.A. McMahon & B.L. Finlayson, 1994. Stream Hydrology, an Introduction for Ecologists. John Wiley & Sons Ltd, New York.
Hawkins, C. P. & J. A. MacMahon, 1989. Guilds: the multiple meanings of a concept. Annual Review of Entomology 34: 423–451.
Horak, C. N., Y. A. Assef, M. G. Grech & M. L. Miserendino, 2020. Agricultural practices alter function and structure of macroinvertebrate communities in Patagonian piedmont streams. Hydrobiologia 847(17): 3659–3676.
Hughes, R. M., D. P. Larsen & J. M. Omernik, 1986. Regional reference sites: a method for assessing stream potentials. Environmental Management 10: 629–635.
Igweze, A., M. Amagoh & A. Ashinze, 2014. Analysis of rainfall variations in the Niger Delta region of Nigeria. IOSR Journal of Environmental Science, Toxicology and Food Technology 8(1): 25–30.
Jones, J. I., J. F. Murphy, A. L. Collins, D. A. Sear, P. S. Naden & P. D. Armitage, 2012. The impact of fine sediment on macroinvertebrates. River Research and Applications 28(8): 1055–1071.
Kelly, D. W., J. T. A. Dick & W. I. Montgomery, 2002. Predation on mayfly nymph, Baetis rhodani, by native and introduced Gammarus: direct effects and the facilitation of predation by salmonids. Freshwater Biology 47: 1257–1268.
Kumari, P. & S. K. Maiti, 2020. Bioassessment in the aquatic ecosystems of highly urbanized agglomeration in India: an application of physical and chemical and macroinvertebrate-based indices. Ecological Indicators 111(12): 106053.
Kuzmanovic, M., S. Dolédec, N. de Castro-Catala, A. Ginebreda, S. Sabater, I. Muñoz & D. Barceló, 2017. Environmental stressors as a driver of the trait composition of benthic macroinvertebrate assemblages in polluted Iberian rivers. Environmental Research 156: 485–493.
Li, Y., H. Chen, L. Song, J. Wu, W. Sun & Y. Teng, 2021. Effects on microbiomes and resistomes and the source-specific ecological risks of heavy metals in the sediments of an urban river. Journal of Hazardous Materials 409: 124472.
Lubanga, H. L., J. O. Manyala, A. Sitati, M. J. Yegon & F. O. Masese, 2021. Spatial variability in water quality and macroinvertebrate assemblages across a disturbance gradient in the Mara River. Ecohydrology & Hydrobiology. https://doi.org/10.1016/j.ecohyd.2021.03.001.
Mangadze, T., T. Dalu & P. William Froneman, 2019a. Biological monitoring in southern Africa: a review of the current status, challenges and future prospects. Science of the Total Environment 648: 1492–1499.
Mangadze, T., R. Wasserman, W. Froneman & T. Dalu, 2019b. Macroinvertebrate functional feeding group alterations in response to habitat degradation of headwater Austral streams. Science of the Total Environment 695: 133910.
Masese, F. O., N. Kitaka, J. Kipkemboi, G. M. Gettel, K. Irvine & M. E. McClain, 2014. Macroinvertebrate functional feeding groups in Kenyan highland streams: evidence for a diverse shredder guild. Freshwater Science 33(2): 435–450.
Matemilola, S., O. H. Adedeji & E. C. Enoguanbhor, 2018. Land use/land cover change in petroleum-producing regions of Nigeria. In Ndimele, P. E. (ed), The Political Ecology of Oil and Gas Activities in the Nigerian Aquatic Ecosystem. Elsevier Inc, Amsterdam.
Matomela, N. H., A. Chakona & W. T. Kadye, 2021. Comparative assessment of macroinvertebrate communities within three Afromontane headwater streams influenced by different land use patterns. Ecological Indicators 129: 107972.
Merritt, R. W. & K. W. Cummins, 1996. An Introduction to the Aquatic Insects of North America, Kendall/Hunt, Dubuque:
Merrit, R. W., K. W. Cummins & M. B. Berg, 2008. An Introduction to the Aquatic Insects of North America, 4th ed. Kendall Hunt Publishing Company. Kendall Hunt Publishing Company, Dubuque:
Miserendino, M. L. & C. I. Masi, 2010. The effects of land use on environmental features and functional organization of macroinvertebrate communities in Patagonian low order streams. Ecological Indicators 10(2): 311–319.
Odume, O. N., 2020. Searching for urban pollution signature and sensitive macroinvertebrate traits and ecological preferences in a river in the Eastern Cape of South Africa. Ecological Indicators 108: 105759.
Onyena, A. P., J. A. Nkwoji & L. O. Chukwu, 2021. Evaluation of hydrochemistry and benthic macroinvertebrates in Chanomi Creek, Niger Delta Nigeria. Regional Studies in Marine Science 46: 101907.
Oyedotun, T. D. T. & N. Ally, 2021. Environmental issues and challenges confronting surface waters in South America: a review. Environmental Challenges 3: 100049.
Pilière, A. F., W. C. Verberk, M. Gräwe, A. M. Breure, S. D. Dyer, L. Posthuma, D. De Zwart, M. A. Huijbregts & A. M. Schipper, 2016. On the importance of trait interrelationships for understanding environmental responses of stream macroinvertebrates. Freshwater Biology 61: 181–194.
R Core Team, E., 2020. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Rawer-Jost, C., J. Bohmer, J. Blank & H. Rahmann, 2000. Macroinvertebrate functional feeding group methods in ecological assessment. Hydrobiologia 422: 225–232.
Sitati, A., P. O. Raburu, M. J. Yegon & F. O. Masese, 2021. Land-use influence on the functional organization of Afrotropical macroinvertebrate assemblages. Limnologica 88: 125875.
Statzner, B. & L. A. Béche, 2011. Can biological invertebrate traits resolve effects of multiple stressors on running water ecosystems? Freshwater Biology 55: 80–119.
Stoddard, J. L., D. P. Larsen, C. P. Hawkins, R. K. Johnson & R. H. Norris, 2006. Setting expectations for the ecological condition of streams: the concept of reference condition. Ecological Applications 16: 1267–1276.
Tampo, L., I. M. Lazar, I. Kaboré, A. Oueda, K. V. Akpataku, G. Djaneye-Boundjou, L. M. Bawa, G. Lazar, W. Guenda, P. Fierro, I. Arismendi, R. M. Hughes, C. Valdovinos, A. Jara-Flores, C. Ntislidou, M. Lazaridou, V. Tsiaoussi, D. C. Bobori, Y. Zhang & M. I. Report, 2020. An improved macroinvertebrate multimetric index for the assessment of wadeable streams in the neotropical savanna. Ecological Indicators 9(1): 105953.
Tomanova, S., E. Goitia & J. H. Helesˇic, 2006. Trophic levels and functional feeding groups of macroinvertebrates in neotropical streams. Hydrobiologia 556(3): 251–264.
Tonkin, J. D., S. Stoll, S. C. Jähnig & P. Haase, 2016. Elements of metacommunity structure of river and riparian assemblages: communities, taxonomic groups and deconstructed trait groups. Ecological Complexity 25: 35–43.
Ukhurebor, K. E., H. Athar, C. O. Adetunji, U. O. Aigbe, R. B. Onyancha & O. Abifarin, 2021. Environmental implications of petroleum spillages in the Niger Delta region of Nigeria: a review. Journal of Environmental Management 293: 112872.
Umar, H. A., M. F. Abdul Khanan, C. Ogbonnaya, M. S. Shiru, A. Ahmad & A. I. Baba, 2021. Environmental and socioeconomic impacts of pipeline transport interdiction in Niger Delta, Nigeria. Heliyon 7(5): e06999.
Uriarte, M., C. B. Yackulic, Y. Lim & J. A. Arce-Nazario, 2011. Influence of land use on water quality in a tropical landscape: a multi-scale analysis. Landscape Ecology 26(8): 1151–1164.
Van Echelpoel, W., M. A. E. Forio, J. Van Butsel, K. Lock, J. A. D. Utreras, L. E. Dominguez-Granda & P. L. M. Goethals, 2018. Macroinvertebrate functional feeding group structure along an impacted tropical river: the Portoviejo River (Ecuador). Limnologica 73: 12–19.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell, C. E. Cushing & G. W. Iinshall, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Varadinova, E. D. & M. Y. Kerakova, 2018. Trophic structure of the riverine macroinvertebrates in type-specific reference conditions. Acta Zoologica Bulgarica 70: 101–108.
Verberk, W. C. E. P., C. G. E. van Noordwijk & A. G. Hildrew, 2013. Delivering on a promise: integrating species traits to transform descriptive community ecology into a predictive science. Freshwater Science 32(2): 531–547.
Wallace, J. B. & R. W. Merritt, 1980. Filter-feeding ecology of aquatic insects. Annual Review of Entomology 25(1): 103–132.
Wang, L., Y. Gao, B. P. Han, H. Fan & H. Yang, 2019. The impacts of agriculture on macroinvertebrate communities: from structural changes to functional changes in Asia’s cold region streams. Science of the Total Environment 676: 155–164.
Whittier, T. R., J. L. Stoddard, D. P. Larsen & A. T. Herlihy, 2007. Selecting reference sites for stream biological assessments: best professional judgment or objective criteria. Journal of North America Benthological Society 26: 349–360.
World Bank Group, 2020. e-Conomy Africa 2020 Africa’s $180 billion Internet economy future. E-Conomy Africa.
Yadav, R., V. Rajput & M. Dharne, 2021. Functional metagenomic landscape of polluted river reveals potential genes involved in degradation of xenobiotic pollutants. Environmental Research 192: 110332.
Acknowledgements
This project was supported by National Research Foundation of South Africa (NRF) and The World Academy of Sciences (TWAS) with grant number: 110894. Special thanks to Edegbene Ovie Tega Treasure (Mrs.) for her technical and office assistance.
Funding
This work was supported by the National Research Foundation of South Africa (NRF) and The World Academy of Science (TWAS) [grant number 110894].
Author information
Authors and Affiliations
Contributions
AOE and FCA: Project conceptualisation; AOE: Funding acquisition; AOE and FCA: Data analysis; AOE FCA, FOA, ECA and EAK: Methodology; AOE and FCA: Writing—original draft; AOE, FCA, FOA, ECA and EAK: Writing -review and editing; AOE: Manuscript finalization; AOE: Project supervision.
Corresponding author
Ethics declarations
Conflict of interest
We declare that there are no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Handling editor: Sally A. Entrekin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Edegbene, A.O., Akamagwuna, F.C., Arimoro, F.O. et al. Effects of urban-agricultural land-use on Afrotropical macroinvertebrate functional feeding groups in selected rivers in the Niger Delta Region, Nigeria. Hydrobiologia 849, 4857–4869 (2022). https://doi.org/10.1007/s10750-022-05034-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-05034-0