Abstract
The high diversity of fish species found in Brazilian streams has attracted scientific interest in recent years. However, it is not clear how studies have addressed biodiversity knowledge shortfalls. We conducted a scientometric analysis of stream fish studies in Brazil to identify trends and gaps in how objectives, spatial coverage, and biodiversity knowledge shortfalls have been studied. Our review covered 690 articles published between 1981 and 2019, and we found an increase in the number of publications, the spatial scale of studies, and the number of streams studied. We also found biases in the distribution of publications concerning regions, biomes, basins, and biodiversity knowledge shortfalls. The Southeast region, the Paraná River basin, the Atlantic Forest biome, and the Hutchinsonian shortfall contributed to a greater number of studies while the Northeast region, the Pantanal biome, Parnaíba basin, Western Northeast Atlantic basin, and the Prestonian shortfall were less represented. Therefore, to improve our knowledge about stream fish, we recommend new collection efforts in under-sampled regions and new studies focused on filling less-addressed biodiversity knowledge shortfalls. Moreover, we emphasize the importance of standardized sampling protocols, a complete sharing of data, and an increase in scientific collaboration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Our knowledge on biodiversity has increased in recent years for taxonomic groups at a rate of more than 18,000 newly described species per year since 2006 (Costello et al., 2013). Despite this increase, knowledge remains inadequate due to a myriad of reasons. For one thing, knowledge about biodiversity is not uniformly distributed in space (Hortal et al., 2015; Oliveira et al., 2016), focusing on specific regions, habitats, biomes, and watersheds (Lobo et al., 2007; Collen et al., 2008). Furthermore, certain taxonomic groups have received more attention from researchers than others. For example, terrestrial vertebrates and plants have been inventoried and described in more detail than almost all invertebrates or unicellular organisms (Mora et al., 2011; Costello 2020). The absence of information on biodiversity composition, distribution patterns, and life-history strategies is a limiting factor in producing knowledge, advances in ecological studies, and conservation of species (Hortal et al., 2015).
In general, the gaps in biodiversity knowledge may be classified into seven categories: (i) the Linnean shortfall, our lack of knowledge about the identify of species, many of which have not yet been described (Bini et al., 2006; Hortal et al., 2015); (ii) the Wallacean shortfall, our lack of knowledge about species distributions (Bini et al., 2006; Hortal et al., 2015); (iii) the Prestonian shortfall, our lack of knowledge about species abundance and population dynamics in space and time (Cardoso et al., 2011; Hortal et al., 2015); (iv) the Darwinian shortfall, our lack of knowledge about phylogeny and species traits and evolution (Diniz-Filho et al., 2013; Hortal et al., 2015); (v) the Raunkiæran shortfall, our lack of knowledge regarding the ecological characteristics and functions of species (Cardoso et al., 2011; Hortal et al., 2015); (vi) the Hutchinsonian shortfall, our lack of knowledge about species tolerance for environmental conditions (Hortal et al., 2015); and (vii) the Eltonian shortfall, our lack of knowledge about species interactions and the effects of such interactions on individual adaptation (Hortal et al., 2015). These knowledge gaps are still more evident in areas with high species diversity and regions threatened by the rapid conversion rate of natural ecosystems in human landscapes (Foley, 2005; Collen et al., 2008; Vörösmarty et al., 2010; Gibson et al., 2011). In other words, the knowledge is missing where it is most needed.
Neotropical streams are considered one of the world’s biodiversity hotspots (Dudgeon, 2019; Reid et al., 2019), and they continue to represent a major frontier for biodiversity knowledge. Compared to temperate streams, Neotropical streams are still poorly studied, and this bias has a strong effect on observed biodiversity knowledge gaps (Oliveira et al., 2016). Partially, the challenge to provide better knowledge of these systems is reinforced by the large biotic diversity they harbor, including a wide array of fish species (Meyer et al., 2007; Winemiller et al., 2008). Moreover, poor taxonomic knowledge of species is still a barrier in these systems even in our present information era (Freitas et al., 2020). Additionally, they represent important targets for biological and ecological studies. For instance, stream fish are excellent indicators of environmental conditions because they are sensitive to changes in a wide array of environmental stressors (Karr, 1981), as found in several Brazilian streams (Terra et al., 2013; Carvalho et al., 2017).
Brazilian streams have a rich fish diversity with more than 1900 stream fish species registered (Castro, 1999; Reis et al., 2016), including many species with restricted distribution or threatened by human activities (Nogueira et al., 2010; Reis et al., 2016). This high diversity and great number of threatened stream fish species, has attracted the researchers’ attention to these ecosystems (Dias et al., 2016; Junqueira et al., 2020; Tourinho et al., 2020). Although the number of studies about stream fish has increased in recent years (see Junqueira et al., 2020), this knowledge is not uniformly distributed (neither temporally, spatially, or between research fields). This bias affects our understanding of this ecosystem and impedes implementing effective conservation measures on a broader spatial scale (Hortal et al., 2015). Therefore, to develop measures aimed at the conservation of stream fish, it is necessary to acquire detailed information on the ecosystems (Dudgeon et al., 2006; Barletta et al., 2010).
Scientometric analysis has become an important and low-cost tool for assessing the trends and knowledge gaps in taxonomic groups or research areas (Nabout et al., 2012). The synthesis of existing knowledge provides valuable insights for guiding future scientific research and subsidies that decision-makers can use in the implementation of effective measures towards biodiversity conservation (Pullin & Stewart, 2006; Nesshöver et al., 2016; Pullin et al., 2016). Recent studies provided a scientometric review about stream fishes and showed (i) an increase in the number of studies in the last years, (ii) most published studies are descriptive, and (iii) most studies are conducted mainly at small spatial and temporal scales (Dias et al., 2016; Junqueira et al., 2020). Using articles published until 2012, Dias et al. (2016) showed an increased number of studies with stream fish assemblage and a predominance of studies with small spatial scales. In turn, Junqueira (2020), using published articles until 2016, corroborated these results and showed that sampling effort is still low considering the number of streams sampled in all river basins studied. However, filling biodiversity knowledge gaps on stream fish remains a major challenge.
Here, we followed the similar path of the studies mentioned above, using a dataset with published articles until 2020, and explored mainly questions about biodiversity knowledge gaps in stream fish studies, encompassing all knowledge fields. Thus, our objective was to analyze stream fish studies in Brazil to answer the following questions: (i) Concerning the published studies on stream fish, are there increases in the number of published studies, the number of sampled streams, and the study area’s size? (ii) In which regions, biomes, and basins were these studies focused? (iii) What questions did the researchers attempt to answer? (vi) What were the major research fields addressed? (v) What were the biodiversity knowledge gaps addressed?
Material and methods
Literature review
We conducted a literature search on stream-dwelling fish in two databases, Web of Science (WoS) and Scopus, to increase our research coverage. We performed searches by topic (article title, abstract, and keywords) for the following strings ((fish* OR ichthyofauna OR pisces) AND (stream* OR headwaters) AND (Brazil)) in both databases. Our searches were conducted initially in January 2018, with an update on May 20, 2020. In our analysis, we included only articles with publication dates through 2019.
We constructed our database by searching the scientific literature on stream fish, published in journals indexed in the WoS and Scopus platforms. While we recognize that grey literature also provides some information about stream fish studies, we chose to restrict our survey to articles published in indexed journals, since the peer review process guarantees a minimum level of data reliability and transparency. After that, we refined the database by: (i) consolidating the search results from both platforms into a single database to remove duplicates; (ii) using the abstract_screener function from the metagear R package (Lajeunesse, 2016) to read the titles and abstracts to restrict our database to articles with study areas in Brazil and exclude items about other animals associated with stream environments (e.g., insects, amphibians, mammals, and birds), as well as articles exclusively focusing on fish occurring in other aquatic environments (e.g., rivers, lakes, and reservoirs); and (iii) conducting full-text screening on all articles selected at the previous stage. We included in our database articles about other aquatic environments (e.g., rivers and lakes) that included streams in their sampling sites when it was possible to extract the stream information separately. Through the above steps, we identified 690 relevant studies (Online Resource 1, Fig. S1).
Data collection
From each of the 690 studies we identified, we extracted the following information: (i) the year of publication; (ii) the number of studied streams (streams sampled in multiple stretches were counted only once); (iii) the geographical coordinates of sampling sites, either as stated in the study or as inferred from adequately georeferenced maps using WebPlotDigitizer (https://automeris.io/WebPlotDigitizer/) when site coordinates were not available in the text; (iv) the Brazilian biome; (vi) the river basin, according to the National Water Agency (ANA, 2015) and (viii) the objectives addressed.
While most of the defined objectives were self-explanatory, some needed to be clarified. To achieve this, we read the full texts of all the objectives, isolated the most pertinent information and identified sets of texts with similar aims. For example, studies with objectives such as “description of the occurrence of intersexuality in populations,” “reproductive biology,” “reproductive behavior,” and “reproductive strategies” were all included in a single category called “reproductive aspects.” The list of studies utilized in our review and the information extracted for each study is shown in the Supplementary Material (Online Resource 2).
We also categorized each article according to the following research fields: behavior, conservation, ecology, evolution, genetics, histology, morphology, parasitology, physiology, systematics, and toxicology (Online Resource 1; Table S1). Some articles involved more than one research field. To check which biodiversity knowledge gaps the studies addressed, we read and classified each article according to the seven shortfalls proposed by Hortal et al. (2015), and here again, some articles belonged to more than one category. (For more details, see Online Resource 1; Table S2).
To assess the spatial scale of each study, we considered the longest linear distance (km) calculated from the pairwise comparison between geographic coordinates of sampling sites. For articles that provided only a figure of the study area with a scale bar, we measured the distance in centimeters between the farthest points on the maps and converted it to kilometers using the scale bar provided (see Dias et al., 2016).
Data analysis
We fitted two models that represented competing hypotheses to evaluate the growth in the number of publications over time: a non-saturating function (exponential) that modeled a continuous growth, and a saturating function (logistic) that modeled a dynamic of growth and stabilization. We fitted the models using non-linear least squares regression (Pinheiro & Bates, 2000), and their performance was compared using Akaike’s Information Criteria (AIC) (Burnham & Anderson, 2002). To ensure that the temporal trends in publication on stream-dwelling fish were not a result of the overall increase in the scientific literature, we performed data standardization by dividing the number of articles published each year by the number of articles present in the WoS database that year (Nabout et al., 2012). We used the annual number of publications from WoS because that information was only available in that database, and WoS is widely recognized as a major database of research publications and citations (Li et al., 2018).
To check for an increase in the number of studied streams and the spatial scale over the period studied, we used quantile regressions (Koenker & Bassett, 1978; Cade & Noon, 2003). The quantile regression approach allows the relationship between the response variable and the predictor to be estimated for any proportion of data distribution rather than just the mean response (Cade & Noon, 2003), and it is less sensitive to the presence of outliers and heterogeneity in data distribution (Koenker & Bassett, 1978; Cade & Noon, 2003). To identify potential changes over time in the magnitude of the extreme values of the variables mentioned above, we used the median (50th percentile), two measures of the upper extreme values (75th and 95th percentiles), and a measure of the lower extreme values (25th percentile) (τ = 0.25, 0.5, 0.75, and 0.95). We used the confidence intervals constructed by the kernel estimation from the covariance matrix to test the significance of each quantile (Koenker & Machado, 1999). We tested the analyses’ assumptions observing the distribution of the residual plots, and we standardized the data using \(\mathrm{log}10(x+1)\) when needed. Finally, we analyzed the number of articles published by region, biome, river basins, objective categories, research fields, and type of biodiversity knowledge gaps addressed by counting their respective frequencies.
To test whether there was a temporal trend in the diversity of the objectives of the articles, we used Pearson correlation (r) to test for association between the year of publication and the Shannon diversity index. We used the Shannon diversity index, a tool widely used in scientometric studies (Rousseau, 1998; Carvalho et al., 2005; Carneiro et al., 2008), to measure the existent diversity in some scientometric variables (e.g., the diversity of journals, the diversity of objectives, and the diversity of keywords in the published articles). First, we built an objective frequency matrix using objective categories as columns and years as rows (sites). Then, we calculated the diversity of objectives for each year using the Shannon index. We performed all the analyses using R version 3.4.2 (R Core Team, 2017), with a significance level of 5% (α = 0.05). For quantile regression calculations, we used the quantreg package (Koenker, 2018), and for drawing the figures, we used the ggplopt2 package (Wickham, 2009).
Results
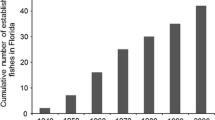
Our searches returned 1899 articles, of which 1259 came from the WoS database and 640 from Scopus. After filtering and removing the duplicates, our final database was composed of 690 articles. We found a more rapid increase in the number of articles on the topic between 1999 and 2009, with an asymptotic trend where the number of articles published annually corresponds to ~ 0.00092% of global publication (Fig. 1a).
Temporal trends in articles on stream fish in Brazil between 1982 and 2019. a Relationship between annual proportion of publications on stream-dwelling fish and the year of publication; b quantile regression for the relationship between the number of streams sampled by studies; and c increase in the extent of study areas of the articles and the year of publication. The barplot in figure a represents the total number of studies on stream-dwelling fish published by year. In figures b, c, the dotted line represents the linear regression model (OLS), while the turquoise, black, blue, and yellow lines represent quantile regressions at 0.25, 0.50, 0.75, and 0.95 percentiles, respectively
We found an increase in the number of streams per study over the years for the percentiles above the median, the 75th (regression coefficient = 0.033 ± 0.004 SE; t = 8.930; P < 0.001) and 95th percentile (regression coefficient = 0.038 ± 0.008 SE; t = 4.406; P < 0.001) (Fig. 1b), while for the 25th percentile, we found no significant relationship (regression coefficient = 0.000 ± 0.003 SE, t = 0.000, P = 1.000, Fig. 1b).
We found no evidence of increase in the spatial scale of studies for the upper limit (95th percentile) of the data distribution (regression coefficient = 0.012 ± 0.009 SE, t = 1.288; P = 0.198; Fig. 1c). In contrast, we observed a positive relationship between spatial scale and time for the other percentiles analyzed (75th percentile: coefficient = 0.048 ± 0.008 SE; t = 5.48; P < 0.001; 25th percentile: coefficient = 0.038 ± 0.010 SE; t = 3.67 P < 0.001; Fig. 1c).
The number of streams by study varied from studies with only one stream to study with 659 streams (mean = 12.03, sd = 35.60). Around 35.65% of the studies (n = 246) covered only one stream, and approximately 11.88% (n = 82) of the articles covered more than 25 streams (Fig. 2a). The size of the articles’ study area was, on average, equal to 111.27 km (sd = 275.92). However, most of the studies assessed a relatively small study area; 24.49% (n = 169) with a study area of less than 1 km and 14.05% (n = 97) with a study area of 1–10 km (Fig. 2b).
As for the number of articles published by the Brazilian regions, the Southeastern was the region with the highest number of studies (n = 284; 38.6%), followed by the Southern (n = 161; 21.9%) and the Midwest (n = 144, 19,6%). The North (n = 101; 13.7%) and Northeastern regions (n = 45; 6.1%) had the lowest number of studies. Considering the production of articles by Brazilian biome, the Atlantic Forest had more than half of the published studies (n = 417), followed by the Cerrado, and Amazon Forest, with 168 and 117 studies, respectively (Fig. 3). The Brazilian watershed with the highest number of studies was the Paraná River basin, with 295 studies, followed by the Southeast Atlantic and Amazon basin with 128 and 104 studies, respectively (Fig. 3).
We found many objective categories (n = 88). The most frequent categories were: “trophic ecology” (n = 110; 9.39%), “describing the structure of the fish community” (n = 86; 7.34%), “genetic diversity” (n = 84; 7.17%), “species description” (n = 83; 7.08%) “spatial distribution” (n = 80; 6.83%), and “environmental influence variables” (n = 73, 6.23%). Together, these categories represented about 44.04% of the objectives (Fig. 4). We also found a temporal increase in the diversity of objectives for studies of stream fish over the studied period (Online resource 1; Fig. S2; r = 0.915; P < 0.001). Regarding the number of stream fish studies per research field, “ecology” (n = 361, 58.4%), “genetics” (n = 75, 12.1%), and “systematics” (n = 69; 11.2%) produced the highest number of studies. Although objective categories for some research fields are shared, this association is generally weak (Online resource 1; Fig. S3).
We found that most of the articles addressed the Hutchinsonian (n = 300, 43.47%), Wallacean (n = 214, 31%), and Raunkiæran shortfalls (n = 160, 23.18%) (Fig. 5). We also found some articles dealing with the Linnean (n = 84, 12.17%), the Eltonian (n = 36, 5.21%), and the Prestonian shortfalls (n = 30, 4.34%).
Discussion
Historical perspectives
The number of studies about stream fish in Brazil has increased over the years, with a tipping point around the year 2001, followed by a trend of stabilization over the last years (Fig. 1a). This stabilization trend was not reported by previous studies (Dias et al., 2016; Junqueira et al. 2020), showing the importance of continuously conducting scientometric analysis for assessing the trends and knowledge gaps in taxonomic groups or research areas (Nabout et al., 2012). The increasing number of publications has been reported in several areas of knowledge or taxonomic groups (Carneiro et al., 2008; Lima et al., 2015; Guerra et al., 2018). More investments in research, the establishment of new graduate programs in areas distant from previously existing research centers, and the rise of a new generation of researchers (Azevedo et al., 2010) may explain such growth. Brazil underwent an expansion of public universities from 2003 to 2014 (Brasil, 2017) with an increase in the number of jobs for teacher-researchers, which directly influenced the number of articles published (Leta et al., 1998; Borges, 2008). We suppose that such stabilization did not occur due to knowledge about fish streams reached a very high level. Stabilization is likely associated with financial support limitations. It is essential to highlight that Brazilian researchers have faced problems related to budget reductions that have affected grants and scholarships in recent years (Martelli-Júnior et al., 2019; Escobar, 2019). Besides, much of the research on stream fish is carried out by postgraduate students funded by public institutions (Junqueira et al., 2020). Therefore, if the reduction in funding continues, it is probable that we will see stagnation or even a decline in the annual number of stream fish studies published in Brazil.
Bias in addressed objectives
Corroborating previous studies, the most frequent categories of objectives reflect interest in species composition, genetic diversity, distribution patterns, land use effects, and natural history (Peres-Neto, 2004; Dias et al., 2016). The prominence of descriptive objectives in most of the studies may explain the predominance of articles in journals with a low impact factor (Junqueira et al., 2020). In general, the predominance of studies with descriptive objectives and conducted at small spatial scales is not attractive to the international scientific community (Dias et al., 2016). However, studies with descriptive purposes are necessary because Brazil holds the greatest fish diversity in the world. We know little about the basic biological aspects of the species, and many of them need to be formally described. Studies at local scales (for example, descriptions of population structures and behavioral aspects) help fill essential knowledge gaps such as the Raunkiæran shortfall or planning conservation measures. Finally, the knowledge about the natural history of species is helpful to understand global (large-scale) threats such as climate change (Vörösmarty et al., 2010) and the establishment of non-native species (Ruaro et al., 2018; Erős et al., 2020). Despite the importance of descriptive studies, we recommend that Brazilian ichthyologists expand their research objectives to include other experimental approaches. Combining observational and experimental studies may provide helpful methods for testing more complex questions, and this approach warrants the attention of the international scientific community (Espírito-Santo et al., 2011, Ilha et al. 2018).
Although there was an increase in the number of objective categories over time, surveys on stream fish still face some barriers. For example, in the ecology field of study, most studies seek to understand local patterns (Aquino et al., 2009; Araújo & Tejerina-Garro, 2009; Dias et al., 2016). There are still few studies that attempt to address regional issues, such as macroecological patterns (Frederico et al., 2014; Vieira et al., 2018), the structure of metacommunities (Almeida & Cetra, 2016; Dala-Corte et al., 2017), climate change effects (Taniwaki et al., 2017), and both functional and phylogenetic diversity (Peres-Neto, 2004; Teresa & Casatti, 2017; Leitão et al., 2018). One approach absent from the Brazilian stream fish studies is the study of indigenous or traditional ecological knowledge. Because of their dependence on fisheries resources, indigenous or traditional peoples often have detailed information about species biology that is not yet recorded in the scientific literature (Silvano et al., 2008). The integration between research fields (for example, parasitology and ecology, or behavior and ecology) is also lacking; although there are shared objectives among research fields, this association remains weak (Online resource 1; Fig. S3). Interactions between study areas and researchers are essential for the emergence of new ideas. All the topics cited above are critical research avenues that merit further exploration.
Bias in spatial scale
Corroborating previous studies, there is a predominance of articles with relatively small study areas (less than 10 km; Fig. 3b) (see Dias et al., 2016; Junqueira et al., 2020). However, our findings indicated a trend of increasing spatial extent. The predominance of studies on small spatial scales is not exclusive to the scientific literature on freshwater aquatic ecosystems in Brazil. The scale issues emerged during the 80's, and gradually influenced biodiversity research (Wiens, 1989; Levin, 1992). Questions, patterns, and processes are scale-dependent (Wiens, 1989), and we need to pay attention to this fact in the research design process. Studies with a large spatial scale are required to improve the understanding of macroecological issues. However, in a megadiversity country like Brazil, studies requiring small spatial scales (e.g., natural history) are essential in filling some biodiversity knowledge gaps that need improvement (see next section). To solve the bias in spatial scale, we highlight that standardization of sampling protocols (Junqueira et al., 2020), collaboration among researchers, availability of the databases of biological collections (i.e., museums) through a digital repository, and complete sharing of data are also essential measures to fill these gaps. We recognize the importance of studies requiring small spatial scales, but we reinforce the necessity to encourage large-scale studies to improve our knowledge on these ecosystems.
Bias in the spatial distribution
Corroborating previous studies, our results showed an asymmetry in the published articles among regions (Dias et al., 2016; Junqueira et al., 2020), Brazilian biomes, and Brazilian basins distribution. The best-studied basins and biomes are located in the Southern and Southeastern regions. One of the possible explanations for this asymmetry is the major economic development of these regions and the concomitantly greater resources of their research agencies, research centers, and universities, as well as a higher number of graduate programs and students located in these regions (Azevedo et al., 2010; Junqueira et al., 2020). Another relevant factor is the ease of access to study areas by roads or trails (Oliveira et al., 2016), which are scarcer in other regions (e.g., remote Amazon regions).
On the other hand, the knowledge is most lacking in biomes that have historically been neglected by conservation policies, such as Caatinga, Cerrado, Pampa, and Pantanal (Overbeck et al., 2015). Although our findings showed a recent increase in studies in some basins, mainly in the Amazon and Tocantins-Araguaia basins (Fig. 3b), they occurred near large centers (e.g., Manaus and Belém) or near hydropower projects (Lees et al., 2016; Winemiller et al., 2016), because of the need of systematic fauna surveys to build such dams. In particular, in the Northeast region are located five of six less-studied Brazilian basins (Fig. 3a). The scarcity of ichthyological studies in these basins is probably because these basins are partially or totally inserted in a region of semi-arid climates (Caatinga). In this region, most streams are intermittent or seasonal, making ichthyological studies more difficult to develop (Rosa et al., 2003; Lima et al., 2017). We recommend new sampling efforts in under-served regions to best understand stream fish biodiversity to solve this bias concerning the spatial distribution of studies. With this, future research will reduce both the Linnean and Wallacean shortfalls (Bini et al., 2006; Hortal et al., 2015).
Bias in filling knowledge shortfalls
Our findings indicated a bias in filling knowledge gaps in stream fish studies from Brazil (see Fig. 5). In general, all shortfalls are interrelated, and linked to the Linnean shortfall (Hortal et al., 2015). Nevertheless, filling the Linnean shortfall remains a challenge (Hortal et al., 2015).
The Linnean shortfalls can have two distinct origins; (i) species yet to be sampled and described, (ii) species collected but not still described (Hortal et al., 2015). Although the rate of description of new fish species in freshwater ecosystems in Brazil has increased in recent years (Reis et al., 2016), progress is still slow. In this context, reducing the Linnean shortfall requires (i) improving sampling protocol and targeting undersampled regions (Mora et al., 2008; Hortal et al., 2015). (ii) To use species collected but not still described as a source to taxonomic review studies. Taxonomic reviews are responsible for re-identifying material deposited in the collections, updating the database (Meier & Dikow, 2004; Simkins et al., 2020). For example, Ferrer and Malabarba (2020) described six new Neotropical catfish genus Scleronema from Pampa grasslands using the taxonomic review approach. Thus, increased investment in the training of taxonomists and surveys of under-sampled regions is fundamental to reduce the Linnean shortfall.
Our results showed an increase in the number of studies addressed to fill the Wallacean shortfall (Fig. 5). However, the number of the studies along Brazilian river basins is asymmetrical, considering that intensely sampled basins also have poorly-sampled sites (Fig. 3). Reducing the Wallacean shortfall is directly related to the capacity of ichthyologists in improving the Linnean shortfall. The formal description of species allows assigning at least one location to the taxon, thus beginning to fill in the Wallacean shortfall (Freitas et al., 2020). Improving the basic knowledge about species distribution, reinforcing the importance of descriptive studies, is needed to solve the bias in biodiversity knowledge shortfalls.
Though also our results showed an increase in the number of studies addressed to fill Raunkiæran shortfall (Fig. 5). However, due the high diversity of fish species in Neotropical streams, this gap still needs a better understanding (Vitule et al., 2017). Functional traits can be an essential tool because they can predict how species, populations, and functionally-similar communities respond to environmental conditions (Laughlin & Laughlin, 2013). However, the functional approach is sensitive to the lack of detailed data on a representative subset of populations, as knowledge of within-population trait variability is essential to determine functional effects and responses (Hortal et al., 2015; Vitule et al., 2017). Besides, the Raunkiæran shortfall can be influenced by the Wallacean shortfall (Hortal et al., 2015). For example, the within-species variation of feeding habits, body shape, or behavior aspects can be affected by where fishes are caught. Manna et al. (2012) reported the variation in Astyanax taeniatus (Jenyns, 1842) feeding habits within the same stream, with herbivory or insectivory tendencies depending on environmental conditions. Thus, to reduce the Raunkiæran shortfall, more studies about life history (e.g., feeding habits, descriptions of population structures, behavioral aspects, and reproductive aspects such as fecundity, oocyte size) of fishes are still needed.
The knowledge about the patterns of interaction between species and population dynamics is essential to target conservation measures. However, we found no increase in trends towards filling gaps related to species interactions (the Eltonian shortfall) or species population dynamics (the Prestonian shortfall). The improvement of this knowledge shortfall in streams fish demands a herculean effort, mainly in a megadiverse country, like Brazil. In particular, the Prestonian shortfall results mainly from difficulties related to data gathering (Hortal et al., 2015). For example, some studies showed that most studies on stream fish assemblages have been short-term (Dias et al., 2016; Junqueira et al., 2020), and this pattern is explained mainly due most projects are funded for short periods (< 3 years) (Dias et al., 2016). Thus, we highlight the importance of long-term projects such as Long-Term Ecological Programs to reduce the Prestonian shortfall in stream fish species.
Conclusions
Our scientometric review found bias related to addressed objectives, spatial scale, spatial distribution in stream fish studies. These findings were also observed by other review studies that addressed Brazilian stream fish (Dias et al., 2016; Junqueira et al., 2020). Besides the previously mentioned bias, our work innovates in showing (i) the stabilization trend in the number of studies on stream fish published annually and (ii) unequal efforts to address biodiversity knowledge shortfalls. Although studies should be planned according to their objectives, grants, logistics, and previous knowledge, future collection efforts should be directed to under-sampled regions to address the most significant shortfalls. Reducing these gaps will require a concerted and joint effort of taxonomists, ecologists, and biogeographers. Finally, our study about knowledge shortfalls on the stream fish provides a guide to future studies to fill distinct biodiversity gaps in these ecosystems.
References
Almeida, R. S., & M. Cetra, 2016. Longitudinal gradient effects on the stream fish metacommunity. Natureza & Conservação 14: 112–119.
ANA, 2015. Conjuntura dos recursos hídricos no Brasil: regiões hidrográficas brasileiras. http://www.snirh.gov.br/portal/snirh/centrais-de-conteudos/conjuntura-dos-recursos-hidricos/regioeshidrograficas2014.pdf.
Aquino, P. P. U., M. Schneider, M. J. M. Silva, C. P. Fonseca, H. B. Arakawa, & D. R. Cavalcanti, 2009. Ictiofauna dos córregos do Parque Nacional de Brasília, bacia do Alto Rio Paraná, Distrito Federal, Brasil Central. Biota Neotropica 9: 217–230.
Araújo, N. B., & F. L. Tejerina-Garro, 2009. Influence of environmental variables and anthropogenic perturbations on stream fish assemblages, Upper Paraná River, Central Brazil. Neotropical Ichthyology 7: 31–38.
Azevedo, P. G., F. O. Mesquita, & R. J. Young, 2010. Fishing for gaps in science: a bibliographic analysis of Brazilian freshwater ichthyology from 1986 to 2005. Journal of Fish Biology 76: 2177–2193.
Barletta, M., A. J. Jaureguizar, C. Baigun, N. F. Fontoura, A. A. Agostinho, V. M. F. Almeida-Val, A. L. Val, R. A. Torres, L. F. Jimenes-Segura, T. Giarrizzo, N. N. Fabré, V. S. Batista, C. Lasso, D. C. Taphorn, M. F. Costa, P. T. Chaves, J. P. Vieira, & M. F. M. Corrêa, 2010. Fish and aquatic habitat conservation in South America: a continental overview with emphasis on Neotropical systems. Journal of fish biology 76: 2118–2176.
Bini, L. M., J. A. F. Diniz-Filho, T. F. L. V. B. Rangel, R. P. Bastos, & M. P. Pinto, 2006. Challenging Wallacean and Linnean shortfalls: knowledge gradients and conservation planning in a biodiversity hotspot. Diversity and Distributions 12: 475–482.
Borges, S. H., 2008. A importância do ensino de pós-graduação na formação de recursos humanos para o estudo da biodiversidade no Brasil: um estudo de caso na ornitologia. Biota Neotropica 8: 21–27.
Brasil, 2017. Ministério da Educação. Análise sobre a expansão das universidades federais 2003 a 2012. http://portal.mec.gov.br/.
Burnham, K. P., & D. R. Anderson, 2002. Model selection and multimodel inference: a practical information-theoretic approach. Springer New York, New York.
Cade, B. S., & B. R. Noon, 2003. A gentle introduction to quantile regression for ecologists. Frontiers in Ecology and the Environment 1: 412–420.
Cardoso, P., T. L. Erwin, P. A. V. Borges, & T. R. New, 2011. The seven impediments in invertebrate conservation and how to overcome them. Biological Conservation 144: 2647–2655.
Carneiro, F. M., J. C. Nabout, & L. M. Bini, 2008. Trends in the scientific literature on phytoplankton. Limnology 9: 153–158.
Carvalho, P., J. A. F. Diniz-Filho, & L. M. Bini, 2005. The impact of Felsenstein’s “phylogenies and the comparative method” on evolutionary biology. Scientometrics 62: 53–66.
Carvalho, D. R., C. G. Leal, N. T. Junqueira, M. A. Castro, D. C. Fagundes, C. B. M. Alves, R. M. Hughes, P. S. Pompeu, M. A. de Castro, D. C. Fagundes, C. B. M. Alves, R. M. Hughes, & P. S. Pompeu, 2017. A fish-based multimetric index for Brazilian savanna streams. Ecological Indicators 77: 386–396.
Castro, R. M. C., 1999. Evolução da ictiofauna de riachos sul-americanos: padrões gerais e possíveis processos causais. In Caramaschi, E. P., R. Mazzoni, & P. R. Peres-Neto (eds), Ecologia de peixes de riachos. Oecologia Brasiliensis, Rio de Janeiro, Brasil: 139–155.
Collen, B., M. Ram, T. Zamin, & L. McRae, 2008. The tropical biodiversity data gap: addressing disparity in global monitoring. Tropical Conservation Science 1: 75–88.
Costello, M. J., 2020. Taxonomy as the key to life. Megataxa 1: 105–113.
Costello, M. J., R. M. May, & N. E. Stork, 2013. Can we name earth’s species before they go extinct? Science 339: 413–416.
Dala-Corte, R. B., F. G. Becker, & A. S. Melo, 2017. The importance of metacommunity processes for long-term turnover of riffle-dwelling fish assemblages depends on spatial position within a dendritic network. Canadian Journal of Fisheries and Aquatic Sciences 74: 101–115.
Dias, M. S., J. Zuanon, T. B. A. Couto, M. Carvalho, L. N. Carvalho, H. M. V. Espírito-Santo, R. Frederico, R. P. Leitão, A. F. Mortati, T. H. S. Pires, G. Torrente-Vilara, J. do Vale, M. B. dos Anjos, F. P. Mendonça, & P. A. Tedesco, 2016. Trends in studies of Brazilian stream fish assemblages. Natureza & Conservação 14: 106–111.
Diniz-Filho, J. A. F., R. D. Loyola, P. Raia, A. O. Mooers, & L. M. Bini, 2013. Darwinian shortfalls in biodiversity conservation. Trends in Ecology & Evolution 28: 689–695.
Dudgeon, D., 2019. Multiple threats imperil freshwater biodiversity in the Anthropocene. Current Biology 29: R960–R967.
Dudgeon, D., A. H. Arthington, M. O. Gessner, Z.-I. Kawabata, D. J. Knowler, C. Lévêque, R. J. Naiman, A.-H. Prieur-Richard, D. Soto, M. L. J. Stiassny, & C. A. Sullivan, 2006. Freshwater biodiversity: importance, threats, status and conservation challenges. Biological reviews 81: 163–182.
Erős, T., L. Comte, A. F. Filipe, A. Ruhi, P. A. Tedesco, U. Brose, M. Fortin, X. Giam, K. Irving, C. Jacquet, S. Larsen, S. Sharma, & J. D. Olden, 2020. Effects of nonnative species on the stability of riverine fish communities. Ecography 43: 1156–1166.
Escobar, H., 2019. Brazilian scientists lament ‘freeze’ on research budget. Science 364: 111.
Espírito-Santo, H. M. V., W. E. Magnusson, J. Zuanon, & T. Emilio, 2011. Short-term Impacts of Fish Removal from Small Amazonian Forest Streams. Biotropica 43: 529–532.
Ferrer, J., & Malabarba, L. R. (2020). Systematic revision of the Neotropical catfish genus Scleronema (Siluriformes: Trichomycteridae), with descriptions of six new species from pampa grasslands. Neotropical Ichthyology, 18(2), 1–81.
Foley, J. A., 2005. Global consequences of land use. Science 309: 570–574.
Frederico, R. G., P. De Marco, & J. Zuanon, 2014. Evaluating the use of macroscale variables as proxies for local aquatic variables and to model stream fish distributions. Freshwater Biology 59: 2303–2314.
Freitas, T. M. S., L. F. A. Montag, P. De Marco, & J. Hortal, 2020. How reliable are species identifications in biodiversity big data? Evaluating the records of a Neotropical fish family in online repositories. Systematics and Biodiversity 18: 181–191.
Gibson, L., T. M. Lee, L. P. Koh, B. W. Brook, T. A. Gardner, J. Barlow, C. A. Peres, C. J. A. Bradshaw, W. F. Laurance, T. E. Lovejoy, & N. S. Sodhi, 2011. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478: 378–381.
Guerra, V., D. Llusia, P. G. Gambale, A. R. Morais, R. Márquez, & R. P. Bastos, 2018. The advertisement calls of Brazilian anurans: historical review, current knowledge and future directions. Plos One 13: e0191691.
Hortal, J., F. de Bello, J. A. F. Diniz-Filho, T. M. Lewinsohn, J. M. Lobo, & R. J. Ladle, 2015. Seven shortfalls that beset large-scale knowledge of biodiversity. Annual Review of Ecology, Evolution, and Systematics 46: 523–549.
Ilha, P., L. Schiesari, F. I. Yanagawa, K. J. Jankowski, & C. A. Navas, 2018. Deforestation and stream warming affect body size of Amazonian fishes. Plos One 13: 1–20.
Junqueira, N. T., L. F. Magnago, & P. S. Pompeu, 2020. Assessing fish sampling effort in studies of Brazilian streams. Scientometrics 123: 841–860.
Karr, J. R., 1981. Assessment of biotic integrity using fish communities. Fisheries 6: 21–27.
Koenker, R., 2018. quantreg: quantile regression., https://cran.r-project.org/package=quantreg.
Koenker, R., & G. Bassett, 1978. Regression quantiles. Econometrica 46: 33–50.
Koenker, R., & J. A. F. Machado, 1999. Goodness of fit and related inference processes for quantile regression. Journal of the American Statistical Association 94: 1296.
Lajeunesse, M. J., 2016. Facilitating systematic reviews, data extraction and meta‐analysis with the metagear package for r. Methods in Ecology and Evolution 7: 323–330.
Laughlin, D. C., & D. E. Laughlin, 2013. Advances in modeling trait-based plant community assembly. Trends in Plant Science 18: 584–593.
Lees, A. C., C. A. Peres, P. M. Fearnside, M. Schneider, & J. A. S. Zuanon, 2016. Hydropower and the future of Amazonian biodiversity. Biodiversity and Conservation 25: 451–466.
Leitão, R. P., J. Zuanon, D. Mouillot, C. G. Leal, R. M. Hughes, P. R. Kaufmann, S. Villéger, P. S. Pompeu, D. Kasper, F. R. de Paula, S. F. B. Ferraz, & T. A. Gardner, 2018. Disentangling the pathways of land use impacts on the functional structure of fish assemblages in Amazon streams. Ecography 41: 219–232.
Leta, J., D. Lannes, & L. De Meis, 1998. Human resources and scientific productivity in Brazil. Scientometrics 41: 313–324.
Levin, S. A., 1992. The problem of pattern and scale in ecology. Ecology 73: 1943–1967.
Li, K., J. Rollins, & E. Yan, 2018. Web of Science use in published research and review papers 1997–2017: a selective, dynamic, cross-domain, content-based analysis. Scientometrics 115: 1–20.
Lima, R. A. F., D. P. Mori, G. Pitta, M. O. Melito, C. Bello, L. F. Magnago, V. P. Zwiener, D. D. Saraiva, M. C. M. Marques, A. A. de Oliveira, & P. I. Prado, 2015. How much do we know about the endangered Atlantic Forest? Reviewing nearly 70 years of information on tree community surveys. Biodiversity and Conservation 24: 2135–2148.
Lima, S. M. Q., T. P. A. Ramos, M. J. Silva, & R. Souza Rosa, 2017. Diversity, distribution, and conservation of the Caatinga fishes: advances and challenges. In Silva, J. M. C., I. R. Leal, & M. Tabarelli (eds), Caatinga. Springer International Publishing, Cham: 97–131.
Lobo, J. M., A. Baselga, J. Hortal, A. Jiménez-Valverde, & J. F. Gómez, 2007. How does the knowledge about the spatial distribution of Iberian dung beetle species accumulate over time? Diversity and Distributions 13: 772–780.
Manna, L., C. Rezende, & R. Mazzoni, 2012. Plasticity in the diet of Astyanax taeniatus in a coastal stream from southeast Brazil. Brazilian Journal of Biology 72: 919–928.
Martelli-Júnior, H., D. R. Martelli, A. C. S. e Silva, M. C. L. Oliveira, & E. A. Oliveira, 2019. Brazil’s endangered postgraduate system. Science 363: 240.1–240.
Meier, R., & T. Dikow, 2004. Significance of specimen databases from taxonomic revisions for estimating and mapping the global species diversity of invertebrates and repatriating reliable specimen data. Conservation Biology 18: 478–488.
Meyer, J. L., D. L. Strayer, J. B. Wallace, S. L. Eggert, G. S. Helfman, & N. E. Leonard, 2007. The contribution of headwater streams to biodiversity in river networks. Journal of the American Water Resources Association 43: 86–103.
Mora, C., D. P. Tittensor, & R. A. Myers, 2008. The completeness of taxonomic inventories for describing the global diversity and distribution of marine fishes. Proceedings of the Royal Society B: Biological Sciences 275: 149–155.
Mora, C., D. P. Tittensor, S. Adl, A. G. B. Simpson, & B. Worm, 2011. How many species are there on earth and in the ocean? PLoS Biology 9: e1001127.
Nabout, J. C., P. Carvalho, M. U. Prado, P. P. Borges, K. B. Machado, K. B. Haddad, T. S. Michelan, H. F. Cunha, & T. N. Soares, 2012. Trends and biases in global climate change literature. Natureza & Conservação 10: 45–51.
Nesshöver, C., B. Livoreil, S. Schindler, & M. Vandewalle, 2016. Challenges and solutions for networking knowledge holders and better informing decision-making on biodiversity and ecosystem services. Biodiversity and Conservation 25: 1207–1214.
Nogueira, C., P. A. Buckup, N. A. Menezes, O. T. Oyakawa, T. P. Kasecker, M. B. Ramos Neto, & J. M. C. da Silva, 2010. Restricted-range fishes and the conservation of Brazilian freshwaters. Plos One 5: e11390.
Oliveira, U., A. P. Paglia, A. D. Brescovit, C. J. B. de Carvalho, D. P. Silva, D. T. Rezende, F. S. F. Leite, J. A. N. Batista, J. P. P. P. Barbosa, J. R. Stehmann, J. S. Ascher, M. F. de Vasconcelos, P. De Marco, P. Löwenberg-Neto, P. G. Dias, V. G. Ferro, & A. J. Santos, 2016. The strong influence of collection bias on biodiversity knowledge shortfalls of Brazilian terrestrial biodiversity. Diversity and Distributions 22: 1232–1244.
Overbeck, G. E., E. Vélez-Martin, F. R. Scarano, T. M. Lewinsohn, C. R. Fonseca, S. T. Meyer, S. C. Müller, P. Ceotto, L. Dadalt, G. Durigan, G. Ganade, M. M. Gossner, D. L. Guadagnin, K. Lorenzen, C. M. Jacobi, W. W. Weisser, & V. D. Pillar, 2015. Conservation in Brazil needs to include non-forest ecosystems. Diversity and Distributions 21: 1455–1460.
Peres-Neto, P. R., 2004. Patterns in the co-occurrence of fish species in streams: the role of site suitability, morphology and phylogeny versus species interactions. Oecologia 140: 352–360.
Pinheiro, J. C., & D. M. Bates, 2000. Mixed-Effects Models in S and S-PLUS. Springer-Verlag, New York.
Pullin, A. S., & G. B. Stewart, 2006. Guidelines for systematic review in conservation and environmental management. Conservation Biology 20: 1647–1656.
Pullin, A., G. Frampton, R. Jongman, C. Kohl, B. Livoreil, A. Lux, G. Pataki, G. Petrokofsky, A. Podhora, H. Saarikoski, L. Santamaria, S. Schindler, I. Sousa-Pinto, M. Vandewalle, & H. Wittmer, 2016. Selecting appropriate methods of knowledge synthesis to inform biodiversity policy. Biodiversity and Conservation 25: 1285–1300.
R Core Team, 2017. R: a Language and environment for statistical computing. Vienna, Austria, https://www.r-project.org/.
Reid, A. J., A. K. Carlson, I. F. Creed, E. J. Eliason, P. A. Gell, P. T. J. Johnson, K. A. Kidd, T. J. MacCormack, J. D. Olden, S. J. Ormerod, J. P. Smol, W. W. Taylor, K. Tockner, J. C. Vermaire, D. Dudgeon, & S. J. Cooke, 2019. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biological Reviews 94: 849–873.
Reis, R. E., J. S. Albert, F. Di Dario, M. M. Mincarone, P. Petry, & L. A. Rocha, 2016. Fish biodiversity and conservation in South America. Journal of Fish Biology 89: 12–47.
Rosa, R. S., N. a Menezes, H. a Britski, W. J. E. M. Costa, & F. Groth, 2003. Diversidade, padrões de distribuição e conservação dos peixes da caatinga. In Leal, I. R., M. Tabarelli, & J. M. C. Silva (eds), Ecologia e conservação da Caatinga. Editora Universitária UFPE, Recife, Brasil: 135–181.
Rousseau, R., 1998. Evenness as a descriptive parameter for department or faculty evaluation studies. In Smet, E. (ed), Werkgemeenschap Informatiewetenschap. Informatiewetenschap, Antwerp, Belgium: 135–145.
Ruaro, R., R. P. Mormul, É. A. Gubiani, P. A. Piana, A. M. Cunico, & W. J. da Graça, 2018. Non-native fish species are related to the loss of ecological integrity in Neotropical streams: a multimetric approach. Hydrobiologia 817: 413–430.
Silvano, R. A. M., A. L. Silva, M. Ceroni, & A. Begossi, 2008. Contributions of ethnobiology to the conservation of tropical rivers and streams. Aquatic Conservation: Marine and Freshwater Ecosystems 18: 241–260.
Simkins, A. T., G. M. Buchanan, R. G. Davies, & P. F. Donald, 2020. The implications for conservation of a major taxonomic revision of the world’s birds. Animal Conservation 23: 345–352.
Taniwaki, R. H., J. J. Piggott, S. F. B. Ferraz, & C. D. Matthaei, 2017. Climate change and multiple stressors in small tropical streams. Hydrobiologia 793: 41–53.
Teresa, F. B., & L. Casatti, 2017. Trait-based metrics as bioindicators: responses of stream fish assemblages to a gradient of environmental degradation. Ecological Indicators 75: 249–258.
Terra, B. D. F., R. M. Hughes, M. R. Francelino, & F. G. Araújo, 2013. Assessment of biotic condition of Atlantic Rain Forest streams: a fish-based multimetric approach. Ecological Indicators 34: 136–148.
Tourinho, M. P., A. P. T. Costa, K. P. Martins, M. G. S. Bandeira, & F. G. Barbosa, 2020. Scientific knowledge on threatened species of the Brazilian Red List: freshwater fish as a case study. Environmental Biology of Fishes Environmental Biology of Fishes 103: 719–731.
Vieira, T. B., C. S. Pavanelli, L. Casatti, W. S. Smith, E. Benedito, R. Mazzoni, J. I. Sánchez-Botero, D. S. Garcez, S. M. Q. Lima, P. S. Pompeu, C. S. Agostinho, L. F. de A. Montag, J. Zuanon, P. D. P. U. de Aquino, M. Cetra, F. L. Tejerina-Garro, L. F. Duboc, R. C. Corrêa, M. A. Pérez-Mayorga, G. L. Brejão, N. T. B. Mateussi, M. A. de Castro, R. P. Leitão, F. P. de Mendonça, L. R. P. da Silva, R. Frederico, & P. De Marco, 2018. A multiple hypothesis approach to explain species richness patterns in Neotropical stream-dweller fish communities. Plos One 13: e0204114.
Vitule, J. R. S., A. A. Agostinho, V. M. Azevedo-Santos, V. S. Daga, W. R. T. Darwall, D. B. Fitzgerald, F. A. Frehse, D. J. Hoeinghaus, D. P. Lima-Junior, A. L. B. Magalhães, M. L. Orsi, A. A. Padial, F. M. Pelicice, M. Petrere, P. S. Pompeu, & K. O. Winemiller, 2017. We need better understanding about functional diversity and vulnerability of tropical freshwater fishes. Biodiversity and Conservation 26: 757–762.
Vörösmarty, C. J., P. B. McIntyre, M. O. Gessner, D. Dudgeon, A. Prusevich, P. Green, S. Glidden, S. E. Bunn, C. A. Sullivan, C. R. Liermann, & P. M. Davies, 2010. Global threats to human water security and river biodiversity. Nature 467: 555–561.
Wickham, H., 2009. ggplot2: elegant graphics for data analysis. Springer New York, New York.
Wiens, J. A., 1989. Spatial scaling in ecology. Functional Ecology 3: 385–397.
Winemiller, K. O., A. A. Agostinho, & É. P. Caramaschi, 2008. Fish ecology in tropical streams. In Dudgeon, D. (ed), Tropical Stream Ecology. Elsevier, San Diego: 107–146
Winemiller, K. O., P. B. McIntyre, L. Castello, E. Fluet-Chouinard, T. Giarrizzo, S. Nam, I. G. Baird, W. Darwall, N. K. Lujan, I. Harrison, M. L. J. Stiassny, R. A. M. Silvano, D. B. Fitzgerald, F. M. Pelicice, A. A. Agostinho, L. C. Gomes, J. S. Albert, E. Baran, M. Petrere, C. Zarfl, M. Mulligan, J. P. Sullivan, C. C. Arantes, L. M. Sousa, A. A. Koning, D. J. Hoeinghaus, M. Sabaj, J. G. Lundberg, J. Armbruster, M. L. Thieme, P. Petry, J. Zuanon, G. T. Vilara, J. Snoeks, C. Ou, W. Rainboth, C. S. Pavanelli, A. Akama, A. v. Soesbergen, & L. Saenz, 2016. Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 351: 128–129.
Acknowledgements
We would like to thank Fundação de Amparo à Pesquisa do Estado de Mato Grosso (FAPEMAT) for granting a Ph.D. Scholarship to the first author (FAPEMAT Notice 002/2015, Process 155509/2015). We are also grateful to Fabrícius Domingos, Vanesa Guimarães, Henrique Giacomini, Eddie Lenza, Edson Fontes, Fabiano Corrêa, and Jean Ortega for valuable suggestions that contributed to the improvement of the manuscript. L. B. Lima was partially financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) Finance Code 001. P. De Marco Júnior and D. P. Lima-Junior are supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (308694/2015-5 and 305923/2020-0 grants, respectively). We acknowledge two anonymous reviewers and associate editor Fernando Mayer Pelicice for constructive criticisms to improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Fernando M. Pelicice.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lima, L.B., De Marco Júnior, P. & Lima-Junior, D.P. Trends and gaps in studies of stream-dwelling fish in Brazil. Hydrobiologia 848, 3955–3968 (2021). https://doi.org/10.1007/s10750-021-04616-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-021-04616-8