Abstract
Ponds are among the world’s most endangered freshwater ecosystems. A comprehensive knowledge of pond biodiversity is urgently required to inform effective pond management and conservation actions. Most studies about pond biodiversity focus on aquatic taxa, while the terrestrial biodiversity, especially of birds, has been little studied. Moreover, the few studies existing on pond biodiversity do not account for different detection rates of species, thus yielding biased results. Here, we apply a hierarchical Bayesian modelling technique to data obtained from visual censuses to estimate bird species richness associated with small ponds in a semiarid region, considering the imperfect detection of species. The model incorporates specific responses to site characteristics (pond typology), landscape (environmental heterogeneity) and at regional scale (mean annual precipitation). The studied ponds were used by two thirds of the terrestrial breeding bird community of the study region. Our modelling approach increased by an average of 7.5 species the observed site-specific richness. Drinking troughs supported a greater richness than other pond types. Environmental heterogeneity was positively related with species richness, whereas no clear relation was observed between richness and precipitation. In addition to ecosystem services provided by ponds to human welfare, our results suggest these small isolated habitats may act as key landscape elements for terrestrial birds in semiarid regions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Freshwater habitats host almost 9.5% of the species described to date, even though they cover only 0.01% of the total water surface area of the Earth (Balian et al., 2008). However, inland waters are considered among the most endangered ecosystems in the world and their associated biodiversity is declining much faster than that of marine and terrestrial environments (Reid et al., 2019). Large aquatic ecosystems such as rivers, lakes and reservoirs have traditionally attracted the attention of freshwater conservationists and managers (Oertli et al., 2009). However, ponds and other small waterbodies can make collectively a non-negligible contribution to the total freshwater area of the world, even higher than rivers and large lakes (Downing et al., 2006; Downing, 2010), and making up 3–4% of some landscapes (Lehner & Döll, 2004). Despite their contribution, these small isolated waterbodies are often neglected by wildlife managers and policy makers (Céréghino et al., 2008).
Ponds are temporary or permanent water sources (both natural and man-made) with a size varying from 1 m2 to 2 ha and no more than 8 m in depth (Oertli et al., 2005). In the last two decades, several studies have highlighted the great potential of small ponds to support a high richness of aquatic species (Gómez-Rodríguez et al., 2009; Oertli et al., 2010; Akasaka & Takamura, 2012; Biggs et al., 2016), including rare and unique taxa (Lemmens et al., 2013; Ilg & Oertli, 2014; Fait et al., 2020). Moreover, the contribution of ponds to biodiversity conservation is particularly important at regional scale, with a high proportion of the regional species pool being present in ponds (Williams et al., 2004; Davies et al., 2008; Zamora-Marín et al., 2021a). The role of ponds to provide key services for biodiversity may be even more pronounced in arid and semiarid regions, where temporary or permanent waterbodies are in short supply (Abellán et al., 2006; Sebastián-González et al., 2010; Lisón & Calvo, 2014). However, just as other freshwater ecosystems, ponds are exposed to many pressures derived from human activities, such as land drainage, nutrient loading, the decline of traditional farming and cattle raising, as well as the introduction of invasive alien species (Declerck et al., 2006), most of these threats having increased in recent years. Among these pressures, land-use changes related to intensive farming are particularly affecting temporary ponds (Ferreira & Beja, 2013) and traditional drinking troughs (Buono et al., 2019), leading to infilling, groundwater overexploitation or pond abandonment. As a consequence, many, if not most, ponds have vanished from the farmlands of continental Europe, even the loss rate reaching as much as 90% in some countries (Oertli et al., 2005). This is why studies aimed at providing robust knowledge on the role of small ponds in biodiversity conservation are urgently required in order to establish pond management strategies.
To date, pond biodiversity research has mainly focused on strict or facultative aquatic taxa, such as macrophytes (Della Bella et al., 2008; Bubíková & Hrivnák, 2018), macroinvertebrates (Florencio et al., 2014; Hill et al., 2016; Fait et al., 2020) and amphibians (Arntzen et al., 2017). However, recent studies have pointed to the need to assess the importance of freshwater ecosystems for conserving not only aquatic organisms but also terrestrial species (Soininen et al., 2015; Smit et al., 2019), including pollinating insects (Walton et al., 2020), bats (Razgour et al., 2010), large mammals (Harper et al., 2019) and farm birds (Lewis-Phillips et al., 2019a). In the case of terrestrial fauna, these habitats provide essential cross-system services such as food (Davies et al., 2016) or drinking water (Abdu et al., 2018), both extremely important resources for wildlife, especially during the breeding season (Lewis-Phillips et al., 2020) or during hot periods (Lee et al., 2017). Until now, very few studies have analysed the role of small ponds in providing services to protect terrestrial biodiversity. In the case of birds, Lewis-Phillips et al., (2019a, b, 2020) found a greater bird abundance and richness in managed open-canopy ponds than in unmanaged overgrown ponds, due mainly to the higher abundance of emergent insects in the former. More generally, bird communities associated with small waterbodies have been described in some arid regions (Bock, 2015; Abdu et al., 2018; Smit et al., 2019), pointing to the moderate contribution of services made by ponds to terrestrial birds. However, these studies did not account for differences in species detectability, which strongly affects occupancy estimates in birds (Einoder et al., 2018), thus yielding biased results that may underestimate the true species richness associated with certain sampling sites (MacKenzie & Bailey, 2004).
Species richness is the most commonly used biological measurement in ecological studies and monitoring programmes (MacKenzie et al., 2006; Kéry et al., 2009). However, determining species richness with accuracy can be complicated because of variability in species-specific detection rates (Kéry & Schmidt, 2008). Previous approaches have traditionally ignored detectability by using raw species counts, leading to questionable inferences with respect to species richness patterns (Kéry & Royle, 2008). Rare species are frequently overlooked in community-level studies, mainly due to their limited detection rates, which are influenced by low abundances (MacKenzie et al., 2005), despite the fact that most of them are considered as species of conservation concern or even have poor conservation status. Nevertheless, management and conservation strategies should consider all species inhabiting a particular site, not just those species which provide enough data (Zipkin et al., 2010). Concern about the imperfect detection of species in ecological studies has increased over recent years, and the rapid development of hierarchical models has led to different approaches being used (Guillera-Arroita, 2017). Among the many advantages of hierarchical models in comparison with other previous approaches (see Kéry & Royle, 2008) -e.g. classical richness estimators- is the fact that they can incorporate habitat and sampling effects that influence occupancy and detection processes, thus enabling the extensive evaluation of several features that may modulate species richness and detectability (Maphisa et al., 2019). Moreover, richness estimation through hierarchical models allows to compute independently occupancy and detectability estimates for each species within a community, thus accounting for the different detection rates of species (Kéry & Royle, 2016). Furthermore, hierarchical occupancy models have rarely been applied to aquatic ecosystems and are vastly underrepesented in the literature (Devarajan et al., 2020). Indeed, very few studies apply hierarchical models to pond biodiversity data (Ferreira & Beja, 2013).
This study looks at the species richness of terrestrial breeding birds associated with 39 small ponds in the most arid region of Europe (southeast of the Iberian Peninsula), using hierarchical Bayesian multi-species occupancy modelling to account for differences in detectability across species. Our hierarchical modelling incorporates three spatial scale covariates, which were expected to affect bird species richness in the study ponds. Firstly, at pond site scale, we hypothesized that some pond attributes can promote the terrestrial bird richness associated with ponds (Davies et al., 2016). For this reason, we explore differences in species richness associated with three pond types that differ in their structural characteristics: artificial pools, cattle ponds and drinking troughs. Secondly, we evaluate a measure of environmental heterogeneity (hereafter, EH) as an environmental variable shaping bird richness associated with ponds at landscape scale. Lastly, mean annual precipitation was incorporated in the model as one of the main climatic factors shaping bird richness at regional scale. Based on previous studies (Qian & Kissling, 2010; Stein et al., 2014; Lorenzón et al., 2016), we hypothesized that both environmental factors (EH and precipitation) might be drivers of bird species richness associated with ponds. Therefore, our objectives were to estimate species richness of terrestrial birds associated with three different pond types and to explore its relationship with EH and precipitation.
Materials and methods
Study area
The study was conducted in the province of Murcia, in the Iberian southeast, which is the most arid region of continental Europe (Armas et al., 2011). The province of Murcia extends over an area of 11,317 km2 and is characterized by a dry warm Mediterranean climate, with a strong water deficit during spring and summer. Mean annual precipitation is 350 mm in most of the province and the average annual temperature ranges from 18.5°C in the driest coastal areas—characterized by scarce precipitation- to 12.0°C in the coldest inland zones (Machado et al., 2011). However, during dry hydrological years, mean annual precipitation usually does not exceed 200 mm, with rainfall being extremely unusual during late spring and summer (mean monthly precipitation 11.5 mm). Despite the semiarid conditions, the existence of a coast-inland climatic gradient promotes high ecosystem heterogeneity, which is even more pronounced as a result of multiple environmental factors, which include weather conditions, intense human pressure, topography and the availability of water resources. Three main different types of ecosystem can be identified in the study area: semidesert zones, Mediterranean shrublands and coniferous forests, mainly composed of Pinus halepensis Miller 1768 and Pinus pinaster Aiton 1789 (Esteve et al., 2015). In general, the further one moves inland from the coast, the greater the presence of mountainous areas, the more continental the climate (colder winters and higher mean annual precipitation) and the lower the pressure of human occupancy. Indeed, mean annual precipitation can usually reach 680 mm in some inland places, which is more than twice the 250 mm normally recorded in the most arid zones.
Regarding the principal land uses, rainfed and irrigated agriculture predominate in the lowlands and high plateaus, whereas steeper areas are occupied by Mediterranean shrubland and pine forests. In recent decades, intensive irrigated agriculture has expanded to almost half of the study area, which has involved the excessive overexploitation of groundwater and surface water resources (Rupérez-Moreno et al., 2017). In addition, traditional livestock grazing has experienced a sharp decline over the last decades, thus leading to the lack of maintenance of cattle ponds and drinking troughs. Moreover, some traditional waterbodies are also exposed to some unsuitable practices such as piping or channelling (López Bermúdez et al., 2016). Indeed, the loss rate of traditional ponds in the study area have been estimated in 36% for cattle ponds (Verdiell-Cubedo, 2012) and 23% for other types of small ponds in the last decades (Ballester-Sabater et al., 2003). This situation has dramatically decreased the availability of water resources (both groundwater fed or not) for wildlife (Valera et al., 2011), especially in periods with a marked water deficit, usually spring and summer. Thus, the availability of water resources for wildlife is considerably lower in natural or semi-natural zones of the study area, where the main land uses are rainfed agriculture and Mediterranean shrubland and forests, and no irrigation ponds appear in the landscape. Consequently, due to the marked scarcity of rivers and streams, artificial pools and traditional ponds (cattle ponds and drinking troughs) still present become the dominant waterbodies in these rural landscapes and they can play an essential role in supporting terrestrial biodiversity (Lisón & Calvo, 2014; Abdu et al., 2018).
Sampling sites: pond types and environmental variables
Study ponds were selected according to the following criteria: (1) easy pond access by birds, so that they could drink safely from the pond shoreline or border; (2) size (up to 450 m2), allowing the authors to visually record bird activity along the whole pond shoreline from the same position; (3) a degree of isolation from other water sources including wetlands, rivers, intermittent streams and other waterbodies (average distance to nearest water source was 935 ± 728 m), which ensured that birds from the surrounding area would use the particular pond; and (4) location in areas far from human settlements (urban centres) in order to avoid biases from human influence –other than pond use- on the bird community composition. As far as possible, ponds were selected along a coast-inland climatic gradient to ensure representative information on the bird communities associated with ponds in areas under different climatic conditions.
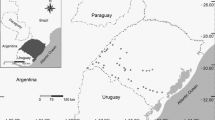
Overall, we selected 39 ponds spread over the study area (Fig. 1) and belonging to three different pond types according to their structural features: 14 artificial pools, 12 cattle ponds and 13 drinking troughs (see a representative picture for each pond type in Fig. 1). Artificial pools are permanent ponds with cemented bottoms and a round or square structure, mostly located in forest or agroforestry landscapes, where extensive agriculture and hunting are the main land uses. They are intermediate in size and depth compared with the other two pond types. Most of them are directly fed from small natural springs, while the rest are filled by farmers or rangers. Cattle ponds are temporal round waterbodies, which were originally dug into Mediterranean farmlands to collect runoff water and provide drinking water for cattle. They hold water at least during nine months per annual cycle. Cattle ponds are larger (270 ± 96 m) and deeper (100 ± 37 cm) than the other two pond types. Although cattle ponds were dug artificially, they appear natural because of their silt bottom and absence of artificial structures around them. They are typically located in plateaus dominated by extensive grassland and tree farming. Lastly, drinking troughs are lineal permanent artificial small waterbodies where cattle drink. In contrast to cattle ponds, drinking troughs are exclusively located in mountain areas dominated by Mediterranean mature forests with small scattered patches of extensive agriculture. Moreover, they are characterized by their small size and shallowness, which limits the development of aquatic vegetation. Although fed from small natural springs, all of them have been modified by lining with cement to ensure water permanence, thus adding to their artificial appearance. Both permanent and temporary ponds held surface water during the study period.
Map of the study area and the location of the surveyed ponds. Coordinates are indicated as UTM 30S (metres). Elevation data and outline maps were obtained from public national data sources (https://www.ign.es/web/cbg-area-cartografia). A representative picture of each pond type is provided below: a artificial pool; b cattle pond; and c drinking trough. Photo credits: José Manuel Zamora
We considered environmental heterogeneity (EH) and mean annual precipitation to be the main environmental variables affecting bird richness associated with study ponds at landscape and regional scale, respectively. Regarding EH, a 1-km radius around each study pond enabled the land cover proportion occupied by four main land-use types to be calculated: tree crop (almond trees and vineyards), herbaceous farming (cereal crops), Mediterranean shrubland and woodland (pine forests). This buffer size was selected because bird community composition associated with ponds is markedly influenced by nearby land uses (Sebastián-González & Green, 2014). To identify the land-use types, the land cover mapping data from the Spanish National Forestry Inventory (Dirección General de Desarrollo Rural y Política, 2012) were used, along with the free software QGIS (version 2.18.19). Following Bain & Stevenson (1999), standard deviation was calculated as a proxy of the land cover EH, using the cover proportion occupied by each land-use type. A high standard deviation meant the dominance of a single land-use type (e.g. 1-km radius fully occupied -100%- by woodland), thus indicating low habitat heterogeneity. Conversely, the lowest standard deviation meant the four selected land-use types (tree crop, herbaceous farming, shrubland and woodland) extend over a similar proportion of land cover (e.g. each land-use type covering 25% of land cover), thus indicating high habitat heterogeneity. Mean annual precipitation was extracted from the climate atlas of the province of Murcia (Garrido et al., 2013) with a 1-km2 grid size.
Bird surveys
We conducted three visits to 19 study ponds in 2017 and to 20 ponds in 2018. Visits were carried out in early-mid spring (April), late spring (May–June) and early summer (July) to cover the whole breeding season of the bird species in the study area. Intervals between visits to each pond were no longer than 40 days. As the ponds were far away from each other (average distance = 4.2 km), spatial autocorrelation in our bird observations at the study ponds was unlikely. Home-range size in passerine birds is often particularly reduced during the breeding season (Roldano, 2002), due to territoriality and brooding activities, and it rarely exceeds 2 ha. (Morganti et al., 2017). Indeed, in a previous study using mist netting to individually mark birds in some of the same study ponds (Zamora-Marín et al., 2021b), we reported no recapture cases between different near ponds, despite most birds were caught twice at the same pond. The order of the visits was constant throughout the study period to correct for the slightly earlier phenology of bird species in warmer coastal areas (unpublished data). Each visit consisted of a 3-h census conducted by direct observation from within a portable hide, which was deployed in a position (at least 10 m from the pond) that maximized the visibility of pond shoreline. As far as possible, the hide was set up close to surrounding vegetation to avoid affecting bird behaviour. In addition, conventional video cameras (Panasonic Handycam, HC-V180, Panasonic Corporation, Osaka, Japan) were set in 14 out of the 39 study ponds during the 3-h censuses with the aim of covering the entire water surface at the largest ponds. Surveys began at sunrise (7:00–8:30 h) in good weather conditions (rainless and windless days) and were always conducted by the same surveyors (JMZ-M and AZ-L). This time of the day has been reported as being the period with greatest bird activity, after which species detectability declines steeply (Lynch, 1989; Whitman et al., 1997). As far as was possible, surveys were conducted during rainless periods because birds visit ponds much less in rainy conditions in arid regions (Lynn et al., 2008; García-Castellanos et al., 2016). We recorded all birds seen or heard within a 10-m buffer from the study pond. All birds were identified at species level, except crested lark (Galerida cristata (L., 1758)) and Thekla´s lark (Galerida theklae (Brehm, 1858)), which were pooled at genus level (Galerida sp.) to avoid misidentification (Guillaumet et al., 2005).
Modelling framework
We used a Bayesian multi-species occupancy model (Dorazio & Royle, 2005; Dorazio et al., 2006) to evaluate the bird species richness associated with the study ponds, as well as to examine the influence of pond typology, EH and mean annual precipitation on bird richness. This model is an extension of the single species site occupancy model (MacKenzie et al., 2002), whereby the hierarchical structure combines community- and species-level attributes within a single analytical framework (Zipkin et al., 2010). The hierarchical model is composed of the ecological process (governed by occupancy probability) and the observational process (governed by detectability probability). Data are compiled as a 2 × 2 matrix (Y) with i rows by k columns, corresponding to sites and species, respectively. The number of temporal replicates or surveys j for each site i where the species k was observed is quantified in the matrix. The ecological process assumes that site-specific occupancy (i. e. ‘true’ presence/absence) for species k at site i is denoted z(i,k), where z(i,k) = 1 if species k occurs in site i and is zero otherwise. The model for occurrence is specified as z(i,k) ~ Bern(ψi,k) where ψi,k is the probability that species k occurs at site i. The true occurrence is imperfectly observed, and we define the detection model for species k at site i in survey j as Y(i,k) ~ Bern(pi,k·z(i,k)) where pi,k is the detection probability of species k for the jth temporal replicate at site i, given that species k is in fact present at site i (Zipkin et al., 2009). In the simplest specification of the model, the occurrence and detection probabilities are composed of species-specific effects and site-level effects (Dorazio et al., 2006; Kéry & Royle, 2016). Extensions of this basic model have explicitly incorporated landscape characteristics into the probability of occupancy (Zipkin et al., 2009; Jiménez-Franco et al., 2019; Maphisa et al., 2019). Following this approach, we modelled the occurrence probability for species k at site i by incorporating the three above-mentioned site-specific covariates: pond type (artificial pool, AP; cattle pond, CP; drinking trough, DT), environmental heterogeneity of the surrounding landscape (EH) and mean annual precipitation (PREC). Linear and quadratic effects of EH and precipitation were included, and both quantitative covariates were standardized. Therefore, the probability of occupancy was defined as follows:
The coefficients from \(\beta_{1,k}\) to \(\beta_{7,k}\) are the effects of pond type, EH of the landscape (linear and squared) and precipitation (linear and squared), for species k, respectively. We assumed that detection probabilities varied depending on the species but were not influenced by survey characteristics: \({\text{logit(}}p_{k} ) \sim {\text{Normal}}(\mu_{{{\text{lp,}}k}} ,\sigma_{{{\text{lp,}}k}}^{{2}} )\). The model was fitted using JAGS (Plummer, 2003) run in R version 3.6.3 (R Core Team, 2020) with the package jagsUI (Kellner, 2015), using uninformative priors, three chains, 15,000 iterations and a burn-in of 5 000 iterations and a thin rate of 2. Convergence was assessed by examining the R-hat values for each parameter estimate (Brooks & Gelman, 1998). We present posterior means and the 95% credible intervals (CRI), the Bayesian analogue to confidence intervals.
Results
We recorded a total of 80 bird species associated with the 39 surveyed small ponds of different types (Table S1, Supporting Information). The observed species corresponded to 34 different avian families, with flycatchers (Muscicapidae) and finches (Fringillidae) being the richest families (nine and eight species, respectively), whereas finches were also the dominant family in terms of abundance (64% of the total abundance). European serin (Serinus serinus L., 1766), common chaffinch (Fringilla coelebs L., 1758) and common linnet (Linaria cannabina (L., 1758)) were the most frequent species in general, occurring in 97.4%, 87.2% and 76.9% of the study ponds, whereas 16 species were occasional and were only recorded in a single pond site. Detection probability estimates ranged from 0.10 to 0.84 (median = 0.35) which illustrates the high variability in detectability among recorded species. Interestingly, our multi-species model revealed that eight or more species were undetected at 12 out of our 39 study ponds. Indeed, richness estimation increased by an average of 7.5 species over observed richness (Fig. 2).
Almost full occupancy estimates in artificial pools and drinking troughs were revealed for rock bunting (Emberiza cia L., 1766), red crossbill (Loxia curvirostra L., 1758), great tit (Parus major L., 1758), European serin, common chaffinch and Eurasian blackbird (Turdus merula L.,1758), indicating the widespread use of small ponds by these species (Fig. S1, Supporting Information). Common linnet, larks (Galerida sp.) and European serin were the most frequent species in cattle ponds.
The multi-species occupancy model revealed the important effect of pond type on associated bird richness (Fig. 3), with a higher visually estimated local richness in drinking troughs (median = 31.8, range = 25.9, 45.2) than in the other pond types. Cattle ponds supported the second highest estimated richness (median = 22.7, range = 15.4, 33.3), whereas artificial pools were the poorest pond type in terms of estimated richness (median = 21.0, range = 13.8, 33.1). Some 29 bird species were exclusively associated with a single pond type, with differences between the typologies, cattle ponds supporting more exclusive taxa (16 species) than drinking troughs and artificial pools (10 and 3 exclusive species, respectively). Considered as a whole, drinking troughs represented the pond type with the highest observed bird richness (61 species), followed closely by cattle ponds (55), whereas artificial pools were the species-poorest, with 41 taxa. On the other hand, EH was positively related to bird richness after controlling for differences in detectability across species (Fig. 4). However, no clear relation was observed in the case of mean annual precipitation (Fig. 5), even though the wettest sites seemed to support the highest bird richness.
Relationship between the estimated number of bird species and the environmental heterogeneity variable (EH) in 39 ponds in the province of Murcia, south-eastern Spain. Vertical lines represent 95% credible intervals. The curve represents a cubic smoothing spline fitted to the data to show the general trend. Note that low EH values indicate high environmental heterogeneity
Discussion
In addition to their widely reported contribution to freshwater biodiversity, small ponds can also play an essential role in providing ecological services for terrestrial wildlife (Lisón & Calvo, 2014; Sutherland et al., 2018; Lewis-Phillips et al., 2019a). Our study supports the importance of small ponds for maintaining terrestrial bird communities in semiarid regions, which is highlighted by the high terrestrial bird richness associated with the study ponds. Interestingly, two thirds of the species composing the entire community of terrestrial breeding birds in the study region, which comprises 120 species (Calvo et al., 2017), were observed visiting these small isolated aquatic ecosystems. This richness value is similar to reported from pond-uninfluenced line transects conducted through the province of Murcia (73 species, see Jiménez-Franco et al., 2019), thus suggesting study ponds were used by a large proportion of the bird assemblage breeding in terrestrial habitats of the study area. Indeed, 1-km line transects conducted in terrestrial habitats adjacent to study ponds revealed that 71% (± 14.5% SD) of the bird species composing the local breeding bird community were recorded making use of the study ponds (authors´ unpublished data). However, this proportion of species may have been even higher because of our multi-species occupancy model yielded a median detection probability of 0.35 among all species. In this sense, several factors have been shown to affect detectability in bird species (Rigby & Johnson, 2019), including the effects from survey design, species behaviour, environmental conditions and observer performance (Jarzyna & Jetz, 2016; Guillera-Arroita, 2017; Jiménez-Franco et al., 2019). In our case, the use of a non-remote survey method may have underestimated the occurrence of elusive and large species, such as raptors and crows, which have been reported to make use of waterbodies through remote cameras (Votto et al., 2020). Even though our sampling method was deemed suitable for the study aims and some large or medium-sized bird species were occasionally detected (e.g. Accipiter gentilis (L., 1758), Columba palumbus L., 1758 and Corvus corone L., 1758), this survey technique may trigger avoidance behaviour in some elusive species due to the hide’s presence. However, a methodological study recently conducted at the same ponds showed direct observation as the most efficient sampling method for detecting bird species associated with ponds, being even more effective than other traditional techniques such as mist netting (Zamora-Marín et al., 2021b). Indeed, direct observation (e.g. point counts and line transects) is a suitable survey method for community-level studies based on bird richness and abundance data, because of it allows to efficiently record different avian guilds (including gregarious and large birds), thus providing representative data on the whole community (Whitman et al., 1997; Faaborg et al., 2004). Furthermore, conversely to other survey methods, direct observation demands less human and economic resources (Poulin et al., 2000). In our case, it should be noted we only surveyed a representative subset of the total number of small waterbodies in the study area, meaning that the bird richness associated with small ponds in the study area could be even greater than reported here.
The studied ponds accounted for a greater proportion of the regional bird species pool (80 out of 120 terrestrial breeding bird species, 67%, in the study area) than reported for small waterbodies from arid regions, although comparisons must be considered with care due to the different sampling designs. For instance, less than 50% of the local bird communities were recorded as using artificial waterholes in the Kalahari desert (Abdu et al., 2018; Smit et al., 2019) and about 36% using natural ponds from Western Cape, South Africa (Lee et al., 2017). These differences in the use of ponds by terrestrial birds may be explained by the dissimilar physiological responses of bird species inhabiting different aridity scenarios. In arid regions, the availability of water resources is naturally scarcer (or even totally absent) compared with semiarid zones, meaning that avifauna from arid regions have been under greater selection pressures to become independent of drinking water (Smit et al., 2019), for example, by obtaining water from the diet (Smit, 2013). In semiarid study areas like ours, several natural springs and other small waterbodies (farm ponds, cattle ponds and drinking troughs, among others) have traditionally been present (López Bermúdez et al., 2016), providing water and other resources for wildlife. For this reason, most bird species from semiarid regions have not developed physiological responses to become independent of drinking water, since they have evolved in landscapes with certain availability of free-water resources. These standing water resources were originally represented by natural springs, rock pools and pools in intermittent or ephemeral streams (Sánchez‐Montoya et al., 2016), and more recently by artificial waterbodies such as cattle ponds and drinking troughs. Therefore, bearing in mind the scarcity of natural water resources in semiarid regions, small traditional ponds seem to have become essential for supporting terrestrial biodiversity, particularly bird communities.
Importantly, higher terrestrial bird richness was observed in drinking troughs than in the other pond types. In our opinion, two factors seem to be responsible for the patterns of species richness associated with our study pond types. On the one hand, vegetation cover is a local factor (i.e. pond attribute) that has been reported to negatively affect terrestrial bird richness associated with ponds (Lewis-Phillips et al., 2019b). Most of the artificial pools we studied were encroached upon or overgrown by emergent vegetation, making the water less visible and accessible for birds, while decreasing food availability as a result of the development and emergence of aquatic insects, consequently decreasing the associated bird richness (Lewis-Phillips et al., 2020). Moreover, habitats overgrown by emergent vegetation may also increase the predation risk perceived by small passerines (Whittingham & Evans, 2004), thus promoting an avoidance behaviour. In our study, drinking troughs showed a pond shoreline discontinuously covered by bush vegetation, thus allowing diversified bird use. Most of the observations for foliage gleaners in our study (e.g. Sylviidae and Phylloscopidae families) corresponded to birds accessing pond shoreline through adjacent bushes, as a behavioural response from their feeding habits. However, ground-foraging bird species (e.g. Fringillidae and Columbidae families) were often observed accessing pond shoreline from non-vegetated open areas. Thus, the occurrence of contrasting structural features within a given pond (i.e. high within-pond heterogeneity) seems to play an important role in promoting the use by a diversified bird community, as previously suggested (Davies et al., 2016). On the other hand, regional factors may directly influence the likelihood of species inhabiting a given pond site. These regional factors include large-scale environmental conditions that affect all systems in a region (e.g. land-use patterns), thus determining the regional species pool (De Meester et al., 2005). Previous studies have reported the strong influence of land uses on terrestrial bird richness (Waltert et al., 2004; Allen et al., 2019). In our case, it was expected that land uses around the ponds would directly affect the regional pool of bird species and consequently the number of bird species using small ponds. Drinking troughs were mainly situated in mature forests with small scattered patches of extensive tree crops. On a global scale, forest and agroforest areas have been shown to support more bird species than agricultural landscapes (Sekercioglu, 2012). This pattern is explained by the high food availability (both fleshy fruits and insects) and complex vegetation of mature forests (Waltert et al., 2004), factors which promote the occurrence of high diversity of insectivorous and frugivorous bird species. Moreover, mature forests and tree plantations provide suitable sites for nesting and roosting, as well as microclimatic refugia for several bird species (Sekercioglu, 2007). Therefore, within-pond heterogeneity and land-use patterns may act synergistically in favour of drinking troughs for promoting the use by high terrestrial bird diversity, but further studies are needed to assess this relationship. The lower bird richness recorded at cattle ponds may be explained by the fact that cereal farming was the dominant land-use type around these ponds, thus promoting the occurrence of a species-poor and highly specialized community dominated by granivorous birds such as larks or sparrows (Mahiga et al., 2019; Vaccaro et al., 2019). This pattern is attributed to the great amounts of weed seeds and other food-grain resources provided by cereal crops for granivorous species (Ndang’ang’a et al., 2013). Moreover, cattle ponds often show a non-vegetated open pond shoreline, thus precluding pond use by foliage gleaners, as highlighted by the fact that species from Sylviidae and Phylloscopidae families were poorly detected (or even undetected) at cattle ponds (Fig. S1, Supporting Information).
Understanding how species richness differs among pond types can provide useful information for improving the effectiveness of management strategies, for instance, by prioritizing conservation actions in those kinds of pond supporting greater species diversity or those considered to have a rarity value. Global conservation priorities are frequently based on bird endemisms or taxonomic uniqueness (among others) rather than species diversity, since richness is generally driven by common and widespread species (Brooks et al., 2006). In our case, drinking troughs were visited by a visibly higher number of bird species than the other pond types, pointing to their greater potential for maintaining more diverse bird communities. Interestingly, however, cattle ponds were visited by more exclusive species than the other two pond types and most observations corresponded with species of conservation concern or threatened taxa, such as calandra lark (Melanocorypha calandra L., 1766), greater short-toed lark (Calandrella brachydactyla Leisler, 1814), lesser kestrel (Falco naumanni Fleischer, 1818) and black-bellied sandgrouse (Pterocles orientalis (L., 1758)), all listed in the European Birds Directive (2009/14/CE), and the locally endangered lesser short-toed lark (Alaudala rufescens (Veillot, 1820)) (Robledano, 2006). This can be explained by the fact that cattle ponds are placed in steppe areas, which are considered among the most threatened habitats in Europe, and leading to the poor conservation status of steppe birds (Burfield, 2005; Traba & Morales, 2019). Therefore, pond conservation and management priorities must consider the importance of these small isolated habitats for supporting both bird richness and threatened species (Davies et al., 2016; Lewis-Phillips et al., 2019a), and ensure the long-term protection of most species-rich pond sites, but especially those that contribute to protecting threatened species. This conservation goal can only be attained through the promotion of different pond types at regional scale (Oertli, 2018; Zamora-Marín et al., 2021a), ensuring the continued presence of a wide variety of ponds with different features that provide complementary ecological services. In this context, the potential role of some management practices for game species (i.e. providing water points) should be considered because they may also benefit threatened bird species (Estrada et al., 2015). In addition to the contributions of ponds to biodiversity, their social and cultural values cannot be overlooked because of most traditional ponds in the study area appear in association with livestock trials, which promote also landscape connectivity for wildlife.
Here, EH was measured as the evenness of four types of land use in a 1-km radius around ponds. As expected, EH showed a positive relation with bird richness. EH has been shown to be among the main drivers of species richness for different biotic groups around the world (Qian & Kissling, 2010; Stein et al., 2014), with particularly positive effects in the case of bird diversity (Lorenzón et al., 2016; Hung-ming et al., 2020). This general pattern is related to the niche-based hypothesis, which states that different species are associated with different habitat types (Hutchings et al., 2000). In our study, some habitat specialist species were related to large habitat patches dominated by a single type of land use (e. g. Periparus ater (L. 1758) and Regulus ignicapilla (Temminck, 1820) in woodlands; Melanocorypha calandra and Alaudala rufescens in grasslands). Thus, a high EH around the study ponds indicates a balanced proportion of the four land-use types, which can promote the occurrence of high number of habitat specialist species when required habitat appears in a sufficiently large extension. However, other ecological processes may also contribute to the high species richness as a result of the selection of heterogeneous sites by individual species. For instance, some bird species show wide ecological requirements and often need to use different habitat types simultaneously in order to meet their daily requirements and tasks (i.e. roosting, feeding or nesting) (Camacho et al., 2014). This relationship stems from the fact that a combination of habitat types can provide more resources for wildlife (Tews et al., 2004) from different origins, thus complementing each other and allowing the coexistence of a wide variety of species.
Finally, no clear effects of mean annual precipitation were observed on bird species richness. This ambiguous relationship between precipitation and bird richness was also seem in an earlier study in the same region (Jiménez-Franco et al., 2019). However, precipitation has been widely shown to positively influence bird richness in many places in the world (Cueto & De Casenave, 1999; Qian & Kissling, 2010; Duclos et al., 2019), mainly through the increase in plant diversity, which ultimately implies more food resources for birds (Rompré et al., 2007). Climate predictors of species richness are often believed to be stronger at broader spatial scales (Field et al., 2009), so the relatively small size of our study area might be masking the true effects of precipitation on bird richness. Indeed, the above-mentioned studies supporting the precipitation-richness relationship were conducted in large regions (often at country or continental scale).
In this study, we have applied a hierarchical approach, which is considered particularly useful for diverse communities (Zipkin et al., 2010). This multi-species occupancy model integrates information from all the sites surveyed, while accounting for the imperfect detection of species. This yields species-specific occupancy probabilities, thus providing valuable information for monitoring programme design. Similarly, the model provides estimates of true species richness for each sampling site, enabling inferences about richness distribution to be made for different habitat types. Indeed, the accurate estimation of total species richness is considered to be extremely important for making conservation, management and policy decisions (Guillera-Arroita et al., 2019). As recently reported, the potential contribution of farm ponds to terrestrial wildlife has been greatly undervalued in conservation and management strategies (Lewis-Phillips et al., 2019b). Even though small waterbodies are believed to provide key ecological services to terrestrial biodiversity (Céréghino et al., 2014; Biggs et al., 2016), the contribution of these isolated freshwater habitats to several non-aquatic animal groups remains largely unexplored and these cross-system services have been rarely addressed in pond literature. According to our results, small ponds can provide important ecological services for terrestrial bird communities, in addition to their role in supporting freshwater biodiversity (Oertli, 2018). Two thirds of the breeding bird species inhabiting the study area were observed using ponds. Thus, since birds are involved in several key services like pest control, pollination and seed dispersal (Sekercioglu, 2006), small ponds can really act as critical habitats at landscape scale for bird conservation and ecosystem functioning. Indeed, the installation of artificial drinking troughs has been proven as a successful management tool for promoting the frugivore-mediated restoration of old fields (e.g. abandoned lands) by attracting terrestrial birds in semiarid regions (García-Castellanos et al., 2016; Martínez-López et al., 2019). The study ponds were visited by a representative set of the local breeding bird communities inhabiting the landscape where the ponds were sited, highlighting the potential of these small isolated freshwater habitats to provide services for terrestrial birds, mainly related to drinking water provision or bath-related plumage care. These findings should provide information on pond management strategies to effectively protect the ecological integrity of these small isolated freshwater habitats, especially bearing in mind the wide variety of ecological services they provide. Further studies are needed to explore the contribution of small ponds to the local bird species pool with a focus on assessing their importance at community level, as well as to quantify and assess the ecological services they provide for terrestrial birds.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abdu, S., A. T. K. Lee, & S. J. Cunningham, 2018. The presence of artificial water points structures an arid-zone avian community over small spatial scales. Ostrich 89: 339–346.
Abellán, P., D. Sanchéz-Fernández, A. Millán, F. Botella, J. A. Sánchez-Zapata, & A. Giménez, 2006. Irrigation pools as macroinvertebrate habitat in a semi-arid agricultural landscape (SE Spain). Journal of Arid Environments 67: 255–269.
Akasaka, M., & N. Takamura, 2012. Hydrologic connection between ponds positively affects macrophyte alpha and gamma diversity but negatively affects beta diversity. Ecology 93: 967–973.
Allen, D., H. Bateman, P. Warren, F. Suzart de Albuquerque, S. Arnett-Romero, & B. Harding, 2019. Long‐term effects of land‐use change on bird communities depend on spatial scale and land‐use type. Ecosphere 10: e02952.
Armas, C., J. D. Miranda, F. M. Padilla, & F. I. Pugnaire, 2011. Special issue: the Iberian Southeast. Journal of Arid Environments 75: 1241–1243.
Arntzen, J. W., C. Abrahams, W. R. M. Meilink, R. Iosif, & A. Zuiderwijk, 2017. Amphibian decline, pond loss and reduced population connectivity under agricultural intensification over a 38 year period. Biodiversity and Conservation 26: 1411–1430.
Bain, M. B., & N. J. Stevenson, 1999. Aquatic habitat assessment: common methods. American Fisheries Society, Bethesda, Maryland.
Balian, E. V., H. Segers, C. Lévêque, & K. Martens, 2008. The freshwater animal diversity assessment: an overview of the results. Hydrobiologia 595: 627–663.
Ballester-Sabater, R., M. Vidal-Abarca, M. Esteve-Selma, & M. Suárez-Alonso, 2003. Humedales y ramblas de la Región de Murcia. Comunidad Autónoma de la Región de Murcia, Murcia, Spain.
Biggs, J., S. von Fumetti, & M. Kelly-Quinn, 2016. The importance of small waterbodies for biodiversity and ecosystem services: implications for policy makers. Hydrobiologia 793: 3–39.
Bock, C. E., 2015. Abundance and variety of birds associated with point sources of water in southwestern New Mexico, U. S. A. Journal of Arid Environments 116: 53–56.
Brooks, S. P., & A. Gelman, 1998. General methods for monitoring convergence of iterative simulations. Journal of Computational and Graphical Statistics 7: 434–455.
Brooks, T. M., R. A. Mittermeier, G. A. B. Da Fonseca, J. Gerlach, M. Hoffmann, J. F. Lamoreux, C. G. Mittermeier, J. D. Pilgrim, & A. S. L. Rodrigues, 2006. Global biodiversity conservation priorities. Science 313: 58–61.
Bubíková, K., & R. Hrivnák, 2018. Artificial ponds in Central Europe do not fall behind the natural ponds in terms of macrophyte diversity. Knowledge and Management of Aquatic Ecosystems 419: 1–10.
Buono, V., A. M. Bissattini, & L. Vignoli, 2019. Can a cow save a newt? The role of cattle drinking troughs in amphibian conservation. Aquatic Conservation: Marine and Freshwater Ecosystems 29: 964–975.
Burfield, I. J., 2005. The conservation status of steppic birds in Europe. In Bota, G., M. B. Morales, S. Mañosa, & J. Camprodon (eds), Ecology and Conservation of Steppe-land Birds. Lynx Editcions: 119–140.
Calvo, J. F., A. J. Hernández-navarro, F. Robledano, M. Á. Esteve, G. Ballesteros, A. Fuentes, F. A. García-castellanos, C. González-revelles, Á. Guardiola, V. Hernández, R. Howard, J. E. Martínez, A. Zamora-López, & J. M. Zamora-Marín, 2017. Catálogo de las aves de la Región de Murcia (España). Anales de Biología 39: 7–33.
Camacho, C., S. Palacios, P. Sáez, S. Sánchez, & J. Potti, 2014. Human-induced changes in landscape configuration influence individual movement routines: lessons from a versatile, highly mobile species. PLoS ONE 9: 1–9.
Céréghino, R., J. Biggs, B. Oertli, & S. Declerck, 2008. The ecology of European ponds: defining the characteristics of a neglected freshwater habitat. Hydrobiologia 597: 1–6.
Céréghino, R., D. Boix, H. M. Cauchie, K. Martens, & B. Oertli, 2014. The ecological role of ponds in a changing world. Hydrobiologia 723: 1–6.
Cueto, V. R., & J. L. De Casenave, 1999. Determinants of bird species richness: role of climate and vegetation structure at a regional scale. Journal of Biogeography 26: 487–492.
Davies, B., J. Biggs, P. Williams, M. Whitfield, P. Nicolet, D. Sear, S. Bray, & S. Maund, 2008. Comparative biodiversity of aquatic habitats in the European agricultural landscape. Agriculture, Ecosystems and Environment 125: 1–8.
Davies, S. R., C. D. Sayer, H. Greaves, G. M. Siriwardena, & J. C. Axmacher, 2016. A new role for pond management in farmland bird conservation. Agriculture, Ecosystems and Environment 233: 179–191.
De Meester, L., S. Declerck, R. Stoks, G. Louette, F. Van De Meutter, T. De Bie, E. Michels, & L. Brendonck, 2005. Ponds and pools as model systems in conservation biology, ecology and evolutionary biology. Aquatic Conservation: Marine and Freshwater Ecosystems 15: 715–725.
Declerck, S., T. De Bie, D. Ercken, H. Hampel, S. Schrijvers, J. Van Wichelen, V. Gillard, R. Mandiki, B. Losson, D. Bauwens, S. Keijers, W. Vyverman, B. Goddeeris, L. De meester, L. Brendonck, & K. Martens, 2006. Ecological characteristics of small farmland ponds: associations with land use practices at multiple spatial scales. Biological Conservation 131: 523–532.
Della Bella, V., M. Bazzanti, M. G. Dowgiallo, & M. Iberite, 2008. Macrophyte diversity and physico-chemical characteristics of Tyrrhenian coast ponds in central Italy: implications for conservation. Hydrobiologia 597: 85–95.
Devarajan, K., T. L. Morelli, & S. Tenan, 2020. Multi‐species occupancy models: review, roadmap, and recommendations. Ecography 1–13.
Dorazio, R. M., & J. A. Royle, 2005. Estimating size and composition of biological communities by modeling the occurrence of species. Journal of the American Statistical Association 100: 389–398.
Dorazio, R. M., J. A. Royle, B. Söderström, & A. Glimskär, 2006. Estimating species richness and accumulation by modeling species occurrence and detectability. Ecology 87: 842–854.
Downing, J. A., 2010. Emerging global role of small lakes and ponds: little things mean a lot. Limnetica 29: 9–24.
Downing, J. A., Y. T. Prairie, J. J. Cole, C. M. Duarte, L. J. Tranvik, R. G. Striegl, W. H. McDowell, P. Kortelainen, N. F. Caraco, J. M. Melack, & J. J. Middelburg, 2006. Abundance and size distribution of lakes, ponds and impoundments. Limnol. Oceanogr 51: 2388–2397.
Duclos, T. R., W. V. DeLuca, & D. I. King, 2019. Direct and indirect effects of climate on bird abundance along elevation gradients in the Northern Appalachian mountains. Diversity and Distributions 25: 1670–1683.
Einoder, L. D., D. M. Southwell, J. J. Lahoz-Monfort, G. R. Gillespie, A. Fisher, & B. A. Wintle, 2018. Occupancy and detectability modelling of vertebrates in northern Australia using multiple sampling methods. PLoS ONE 13: 1–21.
Esteve, M. A., J. Martínez, I. Hernández, F. Robledano, M. A. Pérez, & L. F., 2015. Cambio climático y biodiversidad en el contexto de la Región de Murcia In Victoria, F. (ed), Cambio climático en la Región de Murcia. Evaluación basada en indicadores. Oficina de Impulso Socioeconómico del Medio, Murcia, Spain: 105–132.
Estrada, A., M. Delibes-Mateos, J. Caro, J. Viñuela, S. Díaz-Fernández, F. Casas, & B. Arroyo, 2015. Does small-game management benefit steppe birds of conservation concern? A field study in central Spain. Animal Conservation 18: 567–575.
Faaborg, J., W. J. Arendt, & K. M. Dugger, 2004. Bird population studies in Puerto Rico using mist nets: general patterns and comparisons with point counts. Studies in Avian Biology 29: 144–150.
Fait, P., E. Demierre, C. Ilg, & B. Oertli, 2020. Small mountain reservoirs in the Alps: new habitats for alpine freshwater biodiversity?. Aquatic Conservation: Marine and Freshwater Ecosystems 1–14.
Ferreira, M., & P. Beja, 2013. Mediterranean amphibians and the loss of temporary ponds: are there alternative breeding habitats?. Biological Conservation 165: 179–186.
Field, R., B. A. Hawkins, H. V. Cornell, D. J. Currie, J. A. F. Diniz-Filho, J.-F. Guégan, D. M. Kaufman, J. T. Kerr, G. G. Mittelbach, T. Oberdorff, E. M. O’Brien, & J. R. G. Turner, 2009. Spatial species-richness gradients across scales: a meta-analysis. Journal of Biogeography 36: 132–147.
Florencio, M., C. Díaz-Paniagua, C. Gómez-Rodríguez, & L. Serrano, 2014. Biodiversity patterns in a macroinvertebrate community of a temporary pond network. Insect Conservation and Diversity 7: 4–21.
García-Castellanos, F. A., F. Robledano-Aymerich, V. Zapata, V. Martínez-López, & G. González-Barberá, 2016. Bebederos de aves: dinámica de uso y función en la dispersión ornitócora. Revista de Anillamiento 35: 55–63.
Garrido, R., J. E. Palenzuela, & L. M. Bañón, 2013. Atlas Climático de la Región de Murcia. Agencia Estatal de Meteorología.
Dirección General de Desarrollo Rural y Política, C., 2012. Cuarto Inventario Nacional Forestal: Región de Murcia. Ministerio de Agriculgura, Alimentación y Medio Ambiente, Madrid.
Gómez-Rodríguez, C., C. Díaz-Paniagua, L. Serrano, M. Florencio, & A. Portheault, 2009. Mediterranean temporary ponds as amphibian breeding habitats: the importance of preserving pond networks. Aquatic Ecology 43: 1179–1191.
Guillaumet, A., P. A. Crochet, & B. Godelle, 2005. Phenotypic variation in Galerida larks in Morocco: the role of history and natural selection. Molecular Ecology 14: 3809–3821.
Guillera-Arroita, G., 2017. Modelling of species distributions, range dynamics and communities under imperfect detection: advances, challenges and opportunities. Ecography 40: 281–295.
Guillera‐Arroita, G., M. Kéry, & J. J. Lahoz‐Monfort, 2019. Inferring species richness using multispecies occupancy modeling: estimation performance and interpretation. Ecology and Evolution 9: 780–792.
Harper, L. R., L. Lawson Handley, A. I. Carpenter, M. Ghazali, C. Di Muri, C. J. Macgregor, T. W. Logan, A. Law, T. Breithaupt, D. S. Read, A. D. McDevitt, & B. Hänfling, 2019. Environmental DNA (eDNA) metabarcoding of pond water as a tool to survey conservation and management priority mammals. Biological Conservation 238: 108225.
Hill, M. J., D. B. Ryves, J. C. White, & P. J. Wood, 2016. Macroinvertebrate diversity in urban and rural ponds: implications for freshwater biodiversity conservation. Biological Conservation 201: 50–59.
Hung-ming, T., F. Meng-Wen, & K. Jerome Chie-Jen, 2020. Different habitat types affect bird richness and evenness. Scientific Reports 10: 1221.
Hutchings, M. J., E. A. John, & J. A. Stewart, 2000. The ecological consequences of environmental heterogeneity. Blackwell Science, Oxford, UK.
Ilg, C., & B. Oertli, 2014. How can we conserve cold stenotherm communities in warming Alpine ponds?. Hydrobiologia 723: 53–62.
Jarzyna, M. A., & W. Jetz, 2016. Detecting the multiple facets of biodiversity. Trends in Ecology and Evolution 31: 527–538.
Jiménez-Franco, M. V., M. Kéry, M. León-Ortega, F. Robledano, M. A. Esteve, & J. F. Calvo, 2019. Use of classical bird census transects as spatial replicates for hierarchical modeling of an avian community. Ecology and Evolution 9: 825–835.
Kellner, K., 2015. jagsUI: A Wrapper Around “rjags” to Streamline “JAGS” Analyses. R Package Version1.3.7. Retrieved from http://CRAN.Rproject.org/package=jagsUI.
Kéry, M., & J. A. Royle, 2008. Hierarchical Bayes estimation of species richness and occupancy in spatially replicated surveys. Journal of Applied Ecology 45: 589–598.
Kéry, M., & J. A. Royle, 2016. Applied Hierarchical Modeling in Ecology: Analysis of Distribution, Abundance and Species Richness in R and BUGS: Volume 1: Prelude and Static Models. Cambridge, MA: Academic Press.
Kéry, M., & B. Schmidt, 2008. Imperfect detection and its consequences for monitoring for conservation. Community Ecology 9: 207–216.
Kéry, M., J. A. Royle, M. Plattner, & R. M. Dorazio, 2009. Species richness and occupancy estimation in communities subject to temporary emigration. Ecology 90: 1279–1290.
Lee, A. T. K., D. Wright, & P. Barnard, 2017. Hot bird drinking patterns: drivers of water visitation in a fynbos bird community. African Journal of Ecology 55: 541–553.
Lehner, B., & P. Döll, 2004. Development and validation of a global database of lakes, reservoirs and wetlands. Journal of Hydrology 296: 1–22.
Lemmens, P., J. Mergeay, T. de Bie, J. Van Wichelen, L. de Meester, & S. A. J. Declerck, 2013. How to maximally support local and regional biodiversity in applied conservation? Insights from pond management. PLoS ONE 8: 1–13.
Lewis-Phillips, J., S. J. Brooks, C. D. Sayer, R. McCrea, G. Siriwardena, H. Robson, A. L. Harrison, & J. C. Axmacher, 2019a. Seasonal benefits of farmland pond management for birds. Bird Study 66: 342–352.
Lewis-Phillips, J., S. Brooks, C. D. Sayer, R. McCrea, G. Siriwardena, & J. C. Axmacher, 2019b. Pond management enhances the local abundance and species richness of farmland bird communities. Agriculture, Ecosystems and Environment 273: 130–140.
Lewis-Phillips, J., S. J. Brooks, C. D. Sayer, I. R. Patmore, G. M. Hilton, A. Harrison, H. Robson, & J. C. Axmacher, 2020. Ponds as insect chimneys: restoring overgrown farmland ponds benefits birds through elevated productivity of emerging aquatic insects. Biological Conservation 241: 108253.
Lisón, F., & J. F. Calvo, 2014. Bat activity over small ponds in dry Mediterranean forests: implications for conservation. Acta Chiropterologica 16: 95–101.
López Bermúdez, F., J. M. Quiñonero Rubio, R. García Marín, E. Martín de Valsameda Guijarro, C. Sánchez Fuster, C. Chocano Vañó, & F. Guerero García, 2016. Fuentes y manantiales de la cuenca del Segura: Región de Murcia. Fundación Instituto Euromediterráneo del Agua, Murcia (España).
Lorenzón, R. E., A. H. Beltzer, P. F. Olguin, & A. L. Ronchi-Virgolini, 2016. Habitat heterogeneity drives bird species richness, nestedness and habitat selection by individual species in fluvial wetlands of the Paraná River, Argentina. Austral Ecology 41: 829–841.
Lynch, J. F., 1989. Distribution of overwintering Nearctic migrants in the Yucatan Peninsula, I: general patterns of occurrence. The Condor 91: 515.
Lynn, J. C., S. S. Rosenstock, & C. L. Chambers, 2008. Avian use of desert wildlife water developments as determined by remote videography. Western North American Naturalist 68: 107–112.
Machado, M. J., G. Benito, M. Barriendos, & F. S. Rodrigo, 2011. 500 Years of rainfall variability and extreme hydrological events in southeastern Spain drylands. Journal of Arid Environments 75: 1244–1253.
MacKenzie, D. I., & L. L. Bailey, 2004. Assessing the fit of site-occupancy models. Journal of Agricultural, Biological, and Environmental Statistics 9: 300–318.
MacKenzie, D., J. Nichols, & G. Lachman, 2002. Estimating site occupancy rates when detection probabilities are less than one. Ecology 83: 2248–2255.
MacKenzie, D. I., J. D. Nichols, N. Sutton, K. Kawanishi, & L. L. Bailey, 2005. Improving inferences in population studies of rare species that are detected imperfectly. Ecology 86: 1101–1113.
MacKenzie, D. I., J. D. Nichols, J. A. Royle, K. H. Pollock, L. L. Bailey, & J. E. Hines, 2006. Occupancy estimation and modeling: inferring patterns and dynamycs of species occurence. Academic Press.
Mahiga, S. N., P. Webala, M. J. Mware, & P. K. Ndang’ang’a, 2019. Influence of land-use type on forest bird community composition in Mount Kenya Forest. International Journal of Ecology 2019: 1–8.
Maphisa, D. H., H. Smit-Robinson, & R. Altwegg, 2019. Dynamic multi-species occupancy models reveal individualistic habitat preferences in a high-altitude grassland bird community. PeerJ 7: e6276.
Martínez-López, V., V. Zapata, P. De la Rúa, & F. Robledano, 2019. Uncovering mechanisms of bird seed dispersal in semiarid environments to help to restore them. Ecosphere 10: 1–12.
Morganti, M., G. Assandri, J. I. Aguirre, Á. Ramirez, M. Caffi, & F. Pulido, 2017. How residents behave: home range flexibility and dominance over migrants in a Mediterranean passerine. Animal Behaviour 123: 293–304.
Ndang’ang’a, P. K., J. B. Njoroge, & M. Githiru, 2013. Vegetation composition and structure influences bird species community assemblages in the highland agricultural landscape of Nyandarua, Kenya. Ostrich 84: 171–179.
Oertli, B., 2018. Freshwater biodiversity conservation: the role of artificial ponds in the 21st century. Aquatic Conservation: Marine and Freshwater Ecosystems 28: 264–269.
Oertli, B., J. Biggs, R. Céréghino, P. Grillas, P. Joly, & J. B. Lachavanne, 2005. Conservation and monitoring of pond biodiversity: introduction. Aquatic Conservation: Marine and Freshwater Ecosystems 15: 535–540.
Oertli, B., R. Céréghino, A. Hull, & R. Miracle, 2009. Pond conservation: from science to practice. Hydrobiologia 634: 1–9.
Oertli, B., J. Biggs, R. Cereghino, S. Declerck, A. Hull, & M. R. Miracle, 2010. Pond Conservation in Europe. Springer, Dordrecht, the Netherlands.
Plummer, M., 2003. JAGS: A program for analysis of Bayesian graphical models using Gibbs sampling. In K. Hornik, F. Leisch, & A. Zeileis (eds), Proceedings of the 3rd International Workshop in Distributed Statistical Computing (DSC 2003) Vienna, Austria: 20–22.
Poulin, B., G. Lefebvre, & P. Pilard, 2000. Quantifying the breeding assemblage of reedbed passerines with mist-net and point-count surveys. Journal of Field Ornithology 71: 443–454.
Qian, H., & W. D. Kissling, 2010. Spatial scale and cross-taxon congruence of terrestrial vertebrate and vascular plant species richness in China. Ecology 91: 1172–1183.
R Core Team, 2016. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing. Retrieved from https://www.R-project.org/.
Razgour, O., C. Korine, & D. Saltz, 2010. Pond characteristics as determinants of species diversity and community composition in desert bats. Animal Conservation 13: 505–513.
Reid, A. J., A. K. Carlson, I. F. Creed, E. J. Eliason, P. A. Gell, P. T. J. Johnson, K. A. Kidd, T. J. MacCormack, J. D. Olden, S. J. Ormerod, J. P. Smol, W. W. Taylor, K. Tockner, J. C. Vermaire, D. Dudgeon, & S. J. Cooke, 2019. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biological Reviews 94: 849–873.
Rigby, E. A., & D. H. Johnson, 2019. Factors affecting detection probability, effective area surveyed, and species misidentification in grassland bird point counts. The Condor 121: 1–10.
Robledano, F., 2006. Libro rojo de los vertebrados de la Región de Murcia. Dirreción General del Medio Natural, Consejería de Industria y Medio Ambiente. Comunidad Autónoma de la Región de Murcia, Murcia, Spain.
Roldano, A., 2002. On the ecology of home range in birds. Revue de Écologie 57: 53–73.
Rompré, G., W. D. Robinson, A. Desrochers, & G. Angehr, 2007. Environmental correlates of avian diversity in lowland Panama rain forests. Journal of Biogeography 34: 802–815.
Rupérez-Moreno, C., J. Senent-Aparicio, D. Martinez-Vicente, J. L. García-Aróstegui, F. C. Calvo-Rubio, & J. Pérez-Sánchez, 2017. Sustainability of irrigated agriculture with overexploited aquifers: the case of Segura basin (SE, Spain). Agricultural Water Management 182: 67–76.
Sánchez‐Montoya, M. M., M. Moleón, J. A. Sánchez‐Zapata, & K. Tockner, 2016. Dry riverbeds: corridors for terrestrial vertebrates. Ecosphere. https://doi.org/10.1002/ecs2.1508.
Sebastián-González, E., & A. Green, 2014. Habitat use by waterbirds in relation to pond size, water depth and isolation: lessons from a restorarion in Southern Spain. Restoration Ecology 22: 311–318.
Sebastián-González, E., J. A. Sánchez-Zapata, & F. Botella, 2010. Agricultural ponds as alternative habitat for waterbirds: spatial and temporal patterns of abundance and management strategies. European Journal of Wildlife Research 56: 11–20.
Sekercioglu, C. H., 2006. Increasing awareness of avian ecological function. Trends in Ecology and Evolution 21: 464–471.
Sekercioglu, C. H., 2007. Conservation ecology: area trumps mobility in fragment bird extinctions. Current Biology 17: R283–R286.
Sekercioglu, C. H., 2012. Bird functional diversity and ecosystem services in tropical forests, agroforests and agricultural areas. Journal of Ornithology 153: 153–161.
Smit, B., 2013. Taking the heat: integrating behavioural and physiological variables to predict avian responses to climate change in the Kalahari Desert. University of Pretoria.
Smit, B., S. Woodborne, B. O. Wolf, & A. E. McKechnie, 2019. Differences in the use of surface water resources by desert birds are revealed using isotopic tracers. The Auk 136: 1–13.
Soininen, J., P. Bartels, J. Heino, M. Luoto, & H. Hillebrand, 2015. Towards more integrated ecosystem research in aquatic and terrestrial environments. BioScience 65: 174–182.
Stein, A., K. Gerstner, & H. Kreft, 2014. Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecology Letters 17: 866–880.
Sutherland, K., M. Ndlovu, & A. Pérez-Rodríguez, 2018. Use of artificial waterholes by animals in the southern region of the Kruger National Park, South Africa. African Journal of Wildlife Research 48: 1–15.
Tews, J., U. Brose, V. Grimm, K. Tielbörger, M. C. Wichmann, M. Schwager, & F. Jeltsch, 2004. Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. Journal of Biogeography 31: 79–92.
Traba, J., & M. B. Morales, 2019. The decline of farmland birds in Spain is strongly associated to the loss of fallowland. Scientific Reports 9: 9473.
Vaccaro, A. S., J. Filloy, & M. I. Bellocq, 2019. What land use better preserves taxonomic and functional diversity of birds in a grassland biome?. 14: 1.
Valera, F., C. Díaz-Paniagua, J. A. Garrido-García, J. Manrique, J. M. Pleguezuelos, & F. Suárez, 2011. History and adaptation stories of the vertebrate fauna of southern Spain semiarid habitats. Journal of Arid Environments 75: 1342–1351.
Verdiell-Cubedo, D., 2012. Inventario y estado de conservación de las charcas ganaderas en la Región de Murcia (SE Península Ibérica). Anales de Biología 34: 1–8.
Votto, S. E., F. J. Dyer, V. Caron, & J. A. Davis, 2020. Thermally-driven thresholds in terrestrial avifauna waterhole visitation indicate vulnerability to a warming climate. Journal of Arid Environments 181: 104217.
Waltert, M., A. Mardiastuti, & M. Mühlenberg, 2004. Effects of land use on bird species richness in Sulawesi, Indonesia. Conservation Biology 15: 1339–1346.
Walton, R. E., C. D. Sayer, H. Bennion, & J. C. Axmacher, 2020. Open-canopy ponds benefit diurnal pollinator communities in an agricultural landscape: implications for farmland pond management. Insect Conservation and Diversity.
Whitman, A. A., J. M. Hagan, & N. V. L. Brokaw, 1997. A comparison of two bird survey techniques used in a subtropical forest. The Condor 99: 955–965.
Whittingham, M. J., & K. L. Evans, 2004. The effects of habitat structure on predation risk of birds in agricultural landscapes. Ibis 146: 210–220.
Williams, P., M. Whitfield, J. Biggs, S. Bray, G. Fox, P. Nicolet, & D. Sear, 2004. Comparative biodiversity of rivers, streams, ditches and ponds in an agricultural landscape in Southern England. Biological Conservation 115: 329–341.
Zamora-Marín, J. M., C. Ilg, E. Demierre, N. Bonnet, A. Wezel, J. Robin, D. Vallod, J. F. Calvo, F. J. Oliva-Paterna, & B. Oertli, 2021a. Contribution of artificial waterbodies to biodiversity: a glass half empty or half full?. Science of the Total Environment 753: 141987.
Zamora-Marín, J. M., A. Zamora-López, J. F. Calvo, & F. J. Oliva-Paterna, 2021b. Comparing detectability patterns of bird species in small ponds using multi-method occupancy modelling. Scientific Reports 11: 22785.
Zipkin, E. F., A. Dewan, & J. A. Royle, 2009. Impacts of forest fragmentation on species richness: a hierarchical approach to community modelling. Journal of Applied Ecology 46: 815–822.
Zipkin, E. F., J. Andrew Royle, D. K. Dawson, & S. Bates, 2010. Multi-species occurrence models to evaluate the effects of conservation and management actions. Biological Conservation 143: 479–484.
Acknowledgements
We thank Tamara Díaz, Debora Forte, Sarah Díaz, Julián Castaño, Antonio Fernández-Caro and members of the Department of Zoology and Physical Anthropology of the University of Murcia for their help in the fieldwork, as well as Francisco Alberto García Castellanos for inspiring part of this study. We also thank Miguel Guillén and the Dirección General de Medio Ambiente of the Autonomous Community of Murcia for permission to access to private and public protected areas, respectively. J.M.Z.-M. was supported by a predoctoral grant from the University of Murcia. M.V.J.F. was supported by a postdoctoral grant co-funded by the Regional Valencian Government and the European Social Fund (APOSTD/2018/043) and a “Juan de la Cierva-Incorporación” research contract from the Spanish Ministry of Economy and Competitiveness (reference IJC2019-039145-I). This study was conducted in accordance with the relevant animal ethic guidelines.
Author information
Authors and Affiliations
Contributions
JMZ-M, JFC and FJO-P conceived and supervised the entire study. JMZ-M, AZ-L and JFC performed the fieldwork. MVJF and JFC conducted the statistical analysis. JMZ-M and MVJF wrote a first version of manuscript. All authors reviewed and contributed to the manuscript, as well as approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Handling editor: Dani Boix
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zamora-Marín, J.M., Zamora-López, A., Jiménez-Franco, M.V. et al. Small ponds support high terrestrial bird species richness in a Mediterranean semiarid region. Hydrobiologia 848, 1623–1638 (2021). https://doi.org/10.1007/s10750-021-04552-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-021-04552-7