Abstract
A diverse assemblage of Late Quaternary continental ostracodes (Class Crustacea) belonging to at least 15 genera and 40 species (including 11 new species and one new genus, Plicocandona, described here) was found in sediments of former groundwater discharge deposits located in valleys of southern Nevada, U.S.A. The fossiliferous deposits are located in Las Vegas, Coyote Springs, Indian Springs, Sandy, and Pahrump Valleys. The ostracodes include species that were probably living in wetmeadow, seep, flowing-spring, stream, and wetland environments, and include aquifer species. Those environments indicate a higher level of effective moisture than occurs today during several episodes of the Late Pleistocene to Early Holocene, from ~140 to ~9 ka. The ostracode assemblages are significant to our understanding of regional paleohydrology and also contribute to the growing knowledge of aquifer taxa diversity and biogeography.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Growing interest and concern in the regional history of water on the landscape, or paleohydrology, have led to discoveries of complex modern hypogean ecosystems and fossil remnants of past hypogean activity (Quade et al., 1998; Hancock et al., 2005; Forester et al., 2005a; Humphreys, 2009; Pigati et al., 2014; Springer et al., 2015). Regional paleohydrology is accessible through geologic and paleontologic records, especially by the study of the microfossils of paleohydrologic deposits (Forester, 1991; Quade et al., 2003; Jayko et al., 2008; Pigati et al., 2011). Microfossils in these deposits provide ecological, biogeographical, and geochemical data which are useful in reconstructing past aquatic environments, and thus provide important information about past water supplies. Here, we examine the records of Late Quaternary fossil ostracodes in the paleowetland and stream deposits of the southern Nevada, U.S.A., tracers of the rise and fall of basin water tables during the Late Pleistocene and Early Holocene time. Freshwater ostracodes can be divided into epigean (surface dwelling) and hypogean (groundwater dwelling) taxa. We focus primarily on the hypogean species in these assemblages, and provide the first taxonomic study of the aquifer ostracode species assemblages from the southwestern United States.
Ostracodes are microscopic crustaceans with a calcareous, bivalved carapace. Ostracodes are useful in studies of modern and past hydrology, as well as paleoclimate, because of their sensitivity to environmental changes in aquatic environments including springs, streams, lakes, wetlands, and aquifers (Smith & Horne, 2002; Smith & Palmer, 2012). Within each of those settings, physical and chemical parameters, such as water temperature, major dissolved ion (solute) composition, and concentration (salinity as total dissolved solids, TDS), can further limit species distributions (Forester et al., 2005b). Since different environments commonly have characteristic species assemblages, fossil ostracodes are indicators of past environmental conditions and hydrologic habitats (Smith & Horne, 2002; Curry et al., 2012).
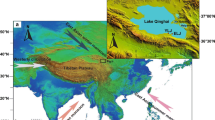
Fossil ostracodes are common in Late Quaternary deposits in southern Nevada, an area of fault block mountain ranges separated by valleys rimmed with alluvial fans (Fig. 1). Present climate is hot and dry, with an annual average temperature of 19.6°C, and average annual rainfall of 106 mm (1937–2012; www.wrcc.dri.edu/cgi-bin/cliMAIN) for Las Vegas, Nevada, U.S.A. However, in the past, wetter and cooler climates supported vigorous groundwater discharge from elevated water tables throughout the region. Many of the region’s valley bottoms are blanketed by fine-grained sediments that initially were interpreted as lake beds, but now are known to be groundwater discharge deposits (Quade, 1986; Quade & Pratt, 1989; Quade et al., 1995, 1998, 2003). While studying these deposits, Quade collected hundreds of mollusk samples, which typically contain ostracodes, to aid in interpretation of the paleoenvironments. In addition to ostracodes, terrestrial and aquatic gastropods, and bivalves, these deposits also contain the fossil remains of mammals/amphibians (Mawby, 1967; Springer et al., 2011), leaf impressions, diatoms (Quade et al., 1995), and pollen (Mehringer, 1967).
The age and paleoenvironmental setting for these deposits are fairly well established by the previous studies. A general stratigraphic nomenclature for the groundwater discharge deposits was established by Haynes (1967) for the Tule Springs area, which was subsequently refined and extended by Quade et al. (1995, 1998, 2003), Bell et al. (1998), and Pigati et al. (2011) to other valleys across the southern Nevada and southeast California. The general age range of the samples included in this study extends from about 140 to 9 ka, and includes Unit B (~140 ka? ~Penultimate Glacial Stage, ~MIS-Stage 6); Unit D (25–19 ka ~Last Glacial Maximum); Unit E1 (18–15 ka ~Post-Glacial); and Unit E2 (13–9 ka ~Late Glacial – early Holocene) (Haynes, 1967; Bell et al., 1998, 2009; Quade et al., 1998, 2003; Springer et al., 2015). We systematically examined ostracodes in over thirty samples. The locations, stratigraphic context, and paleoenvironmental setting of these samples are given in Tables 1 and 2.
The connectivity of the aquifers in this region was studied by Winograd & Thordarsen (1975). The hydrogeology of the region is controlled by the deep carbonate aquifer, a Paleozoic limestone unit with numerous fractures, faults, and small openings, and the overlying clay aquitards and Cenozoic alluvial aquifers. They determined that the Cenozoic alluvial aquifers and the deep carbonate aquifer are hydrologically connected by intrabasin flow, and that the water moves and circulates to depths of up to 1280 m (Winograd & Thordarsen, 1975). Considering the subsurface areal extent of this region, they note that “an area of at least 4500 square miles is hydraulically integrated into one groundwater basin” (Winograd & Thordarsen, 1975, p. C1).
The Quaternary paleohydrologic records from aquatic settings of the southwestern North America are characterized by a history of rising lake levels during glacial stages (pluvial lakes) and evidence of rising and falling water tables in response to large changes in precipitation amounts and patterns (Reheis et al., 2014). The Great Basin and surrounding areas were wetter and cooler during the times of deposition of groundwater discharge deposits of Units B, D, and E. In the northern and central Great Basin, lakes covered valley bottoms during these times. In southern, lower-elevation Nevada, however, warmer conditions prevented deep lake development. Extensive groundwater recharge from high-standing mountains in the region such as the Spring and Sheep Ranges led to a widespread spring discharge in valley bottoms (Paces et al., 1996). These springs in turn supported wet meadows, wetlands, and even perennial creeks which once connected to some now isolated valley segments. These environments then were the settings for the ostracodes as described in this paper (Tables 1, 2 and 3). This mosaic of aquatic habitats supported many freshwater epigean species of ostracodes that can be found throughout the northern hemisphere in similar environments, as well as epigean forms specific to the region. These widely dispersed epigean species are tolerant of hydroperiodic variability, and thus can survive in a range of aquatic conditions (Forester et al., 2005b). Along with these epigean taxa, however, are small, interstitial species typical of hypogean habitats that were discharged into these surface settings when the water table was higher and springs were active. Hypogean species, by comparison, live in more stable environments with little range in temperature, salinity, and seasonal change. Thus, these fossil assemblages contain both epigean and hypogean taxa, with epigean taxa being far more prevalent.
Our samples were also obtained to maximize geographic coverage and time range. Deposits from Corn Creek, Tule Springs, and Pahrump contain the widest range of ages (Units B through D) and paleoenvironments, whereas those from Cactus Springs, Sandy, and Coyote Springs Valleys (Fig. 1) are younger (Unit E, minor Unit D) and less paleoenvironmentally variable.
Methods and materials
Initially, the samples were collected to recover mollusks by dry sieving over an 80-mesh (180 μm) screen. Subsamples ranging in size from 0.05 to 41.8 g (but mostly 25 to 33 g) were later processed for ostracodes. Each subsample came from the dry-seived >80-mesh residue, which included sediment aggregates composed of particles <180 μm in size from which small valves were recovered. As the adult valves of most ostracodes are larger than 180 μm processing dried sediments presented no problem. However, the adults of many species that live in aquifers are often smaller than 180 μm and so dry sieving likely resulted in the loss of many if not most of those species. None the less, we recovered at least eight distinct aquifer species from these samples, many presumably from the sediment aggregate.
The ostracode preparation process (Forester, 1988) consisted of pouring about 600 ml of hot (90°C) deionized water over a weighed sediment sample in a steel beaker. About a teaspoon of commercial baking soda was added to the water and when it cooled to room temperature, a half teaspoon of commercial Calgon (sodium carbonate, sodium hexametaphosphate, soap) was added and the solution was stirred. The samples were frozen solid after standing for about 12 h, then thawed and allowed to stand for at least another 12 h. They were wet sieved over a 100-mesh (150 μm) screen, and the residue was air dried. The dried residue was sieved and the valves were picked onto micropaleontological slides. Of the 295 samples examined, 233 contained adult ostracode valves. These samples are stored in the Paleolimnology Laboratory of the Geology Department at Kent State University.
Results
The ostracodes
Of the 40 species of ostracodes found in the southern Nevada spring deposits (Table 3), only 12 were recovered from all five valleys sampled (Fig. 1): Candona aff. C. rawsoni Tressler, Candona cerca n. sp., Candona sp. E, Candona sp. R, Candona aff. C. stagnalis Sars, Cypridopsis vidua (Müller), Cypridopsis okeechobei Furtos, Heterocypris incongruens (Ramdohr), Limnocythere aff. L. paraornata Delorme, Scottia aff. S. tumida (Jones), Cavernocypris wardi Marmonier, Meisch & Danielopol, and Darwinula spp. (Table 3). Six more were found in all of the other valleys, but were missing from either the North Coyote Springs Valley or South Coyote Springs Valley: Candona acuminata Fischer, Candona caudata Kaufmann, Candona distincta Furtos, Candona averta n. sp., Ilyocypris bradyi Sars, and Eucypris meadensis Gutentag & Benson. All but three of the aquifer species are restricted to a single valley: Candona sp. N to the Pahrump Valley, Candona sp. K to the Coyote Valley, and Plicocandona sp. B, P. cassicula n. sp., P. macraina n. sp., and Candona sp. L to the Corn Creek area. Several species were found only in the Pahrump Valley: Strandesia sp. A., Candocyprinotus ovatus Delorme, and Candona sp. N. The most frequently found and most widespread species were Eucypris meadensis, Cypridopsis vidua, and Limnocythere aff. L. paraornata. C. okeechobei occurs in nearly half of the fossiliferous collections, and Candona aff. C. rawsoni, Candona cerca n. sp., and Scottia aff. S. tumida occur in a third of the fossiliferous collections. Heterocypris incongruens is also fairly widespread, but in most collections it is represented by fewer than two or three adult valves. Potamocypris aff. P. similis Müller is common in the Corn Creek area but rare elsewhere.
In general, ostracodes and oxygen isotope evidence (Forester, 1991; Quade et al., 2003) from our the southern Nevada assemblages indicate that surface waters were cold (<13°C), had low TDS, below ~1000 mg/l, and never evolved to saline solute compositions. Modest, localized discharge and lack of basin closure prevented the development of lakes and attendant higher water salinities. Solutes were dominated by calcium bicarbonate, reflecting interaction with limestone and dolostone bedrock in surrounding mountain ranges.
In most samples from the southern Nevada basins, wetland, spring taxa, and aquifer taxa are all present (Quade et al., 2003). Aquifer taxa refer to small, often highly ornate taxa with subtrapezoidal or subtriangular carapace shapes that evidently lived in sediment intergrain spaces (interstitially) in the saturated zone. Aquifer ostracodes are typically the least abundant group on a valve/gram basis (Quade et al., 2003). It is important to note that the fossil ostracode fauna in these valley bottom sediments represents a dynamic and inter-related mosaic of aquatic habitats, and the absolute abundance of aquifer taxa in one area vs another is not necessarily meaningful. Rather, their presence is significant of aquifer discharge.
Systematic paleontology
The terminology for carapace surface features used herein comes from Sylvester-Bradley and Benson (1971), whereas that for carapace morphology it comes from Van Morkhoven (1962). The systematic classification scheme is from Hartmann and Puri (1974) updated following Maddocks (1982) and Meisch (2000), the latter of which contains full references to the literature of the higher taxonomic ranks. The taxonomic descriptions presented here are entirely based on carapace features and shell morphology: no soft parts were preserved. Therefore, recently determined taxonomic distinctions for some species which are based upon soft part features could not be determined in these samples. In such cases, the older taxonomic identifications based on carapace features were used, as for example in the case of Candona species that may be recognized in modern specimens as Fabaeformiscandona species. Kempf (1997a, b, c, d) was consulted in the process of choosing names for these new species. Holotypes are housed in the collections of the Department of Paleobiology, National Museum of Natural History (USNM), Smithsonian Institution.
Class OSTRACODA Latreille, 1806
Subclass PODOCOPA Müller, 1894
Order PODOCOPIDA Sars, 1866
Family CANDONIDAE Kaufmann, 1900
Subfamily CANDONINAE Kaufmann, 1900
Genus Candona Baird, 1845
Type species: Cypris candida Müller, 1776
Candona averta n. sp.
Diagnosis Carapace subquadrate in side view; rounded anterior margin merging smoothly into nearly straight anterodorsal margin, which joins the nearly straight dorsum in gentle obtuse angle. Highest point of dorsum posterior of center smoothly joining curved, truncated (blunt) posterior. Venter concave, joining posterior at rounded, approximately 90° angle. In dorsal view, sides convex, posterior of right valve with narrow flange that fits inside posterior of left valve. Inner lamella relatively narrow anteriorly, even narrower ventrally and posteriorly; posteroventral portion of inner lamella of right valve bowed outward away from valve (outer lamella). Posterior portion of male carapace inflated, venter more deeply concave, with anteroventral projection. Shell surface smooth Fig. 2e–h.
Candona mojavensis n.sp. (a, b), Sandy Valley, SVF-87-11, Unite E, wetland. a Right valve exterior, b Right valve interior. Candona coyote springensis (c, d), Coyote Springs Valley, NCYSC-9, Unit E, spring. c Left valve exterior, d Left valve interior. Candona averta n.sp. (e–h) Corn Creek Flat, CSF87-33, Unit D, wetland. e Left valve exterior, female, f Left valve interior, female, g Right valve exterior, female, h Right valve exterior, female
Remarks This form is distinguished by the posterior flange and bowed-out posteroventral inner lamella on the right valve. Candona averta is an epigean species, similar in habitat preference and shell outline to Candona rawsoni Tressler; however, there is no posteroventral tab on the female right valve. The male is not common in the samples, and is similar in outline to the males of Candona rawsoni Tressler, showing distinct sexual dimorphism, with a well developed anteroventral projection.
Etymology From the Latin averta for “saddlebag”.
Holotype USNM 629800 Left valve; Paratype 629801 Right valve; Paratype 629802 Left valve.
Dimensions of measured specimens n = 4: Female length 1.20–1.27, height 0.64–0.67 mm, Male length 1.34, height 0.7 mm.
Candona cerca n. sp.
Diagnosis Carapace subtriangular in side view, with rounded anterior and acutely pointed posterior; sides convex in dorsal view, with greatest width posterior of center. Dorsum arched, with highest point posterior of center; posterdorsal margin slightly convex, anterodorsal margin straight to slightly concave. Venter concave with sinuous to straight posteroventral margin. Shell surface smooth. Left valve with narrow posterior flange that extends beyond and overlaps right valve. In lateral view of exterior, this flange creates a slight posteroventral notch, as if a fragment of shell is missing. Male carapace with more rounded posterior outline than female and slight anteroventral projection. Inner lamella very broad anteriorly, with semicircular inner margin; conspicuously narrower ventrally and posteriorly, with inner margin following shape of acutely pointed posterior outline in females, more rounded in males Fig. 3a–g.
Candona cerca n.sp., (a–g), Female, CSF-87-33, Corn Creek Flat, Unit D, wetland. a Left valve exterior, b Left valve interior, c Left valve posteroventral hook-like tab, d Right valve interior, e Left valve exterior, f Right valve exterior, g Left valve venter, small arrow points to posteroventral angle
Remarks This is an epigean species. The amount of sinuosity of the female posteroventral margin and the width of the posterodorsal flange are variable, but the humpbacked, triangular outline of this form, combined with its pointed posterior and the distinctive left valve flange on the posterodorsum near the venter, serves to distinguish it from other species of Candona. We note a similar form known from the northern Holarctic areas, Candona acutula Delorme, 1967, a possible junior synonym of Candona levanderi Hirschmann, 1912. In Candona acutula, the flange is a rounded semicircular tab on the right valve of the female, as compared to a vertical fold on the left valve observed in Candona cerca.
Etymology From the Greek cerca for “tail”.
Holotype USNM 629803 Left valve; Paratype 629804 Right valve; Paratype 629805 Left valve; Paratype 629806 Right valve; Paratype 629807 Left valve.
Dimensions of measured specimens: n = 5. Female Length 1.13–1.20 mm, Height 0.63–0.67 mm. Male Length 1.32 mm, Height 0.76 mm.
Candona corncreekensis n.sp.
Diagnosis Carapace extremely small, oblong in side view; anterior and posterior margins rounded, dorsum straight, venter concave. In dorsal view, sides subparallel in central portion, compressed anteriorly. Valves nearly equal; in end view, dorsal outline flattened rather than acutely pointed. Anterior inner lamella slightly broader than posterior inner lamella; inner margins smoothly semicircular anteriorly and posteriorly. Adductor muscle scar pattern of the Candona type. Shell surface reticulate with pore conuli; reticulum consists of relatively thin, well-defined muri and more-or-less regularly hexagonal, relatively large fossae. Pore conuli distinctive and also occur radially along antero- and postero-margins. No males observed Fig. 4a–d.
Candona corncreekensis n.sp., (a–d), Corn Creek Flat, CSF-87-15,Unit D-E, channel complex. a Left valve exterior, b Right valve exterior, c Posterolateral exterior view of Right valve, showing reticulation with hexagonal fossae, d Anterodorsal exterior view of right valve, showing pore conuli. Candona pahrumpensis n.sp., (e–h), Pahrump Valley, Pah.86 Mol.56, Unit B, wetland. e Left valve exterior, f Left valve interior, g Right valve exterior, h Right valve interior
Remarks This hypogean species is distinguished by its tiny size, presence of pore conuli, and heavily ornamented shell surface. It is very rare in our collections.
Etymology corncreekensis, named for the type locality of Corn Creek Flats, Nevada
Holotype USNM 629808 Left valve; Paratype 629809 Right valve.
Dimensions of measured specimens, n = 4: Length, 0.44–0.49 mm, Height, 0.20–0.23 mm.
Candona coyotespringensis n. sp.
Diagnosis Carapace elongate trapezoidal in side view; anterior margin broadly rounded, posterior margin narrowly rounded. Dorsum straight or slightly concave, meeting nearly straight antero- and posterodorsal margins at obtuse angles. Venter of right valve slightly concave anterior of center; ventral outline of left valve approximately straight, with ventral ridge extending below concave ventral margin. In dorsal view, sides very shallowly convex, subparallel in central portion. Valves equal in height. Anterior and posterior inner lamellae nearly equal in breadth; inner margins deeply concave. Adductor muscle scar pattern faintly visible, of Candona type. Shell surface smooth, with scattered punctae mainly along anterior and ventral peripheries. No males observed. Fig. 2c, d.
Remarks This hypogean species is distinguished by its elongate trapezoidal outline and the ventral ridge on the left valve, which is not developed in juvenile carapaces. It is very rare in our collections, possibly because of the thin carapace.
Etymology coyotespringensis, named for the type locality of Coyote Springs, Nevada
Holotype USNM 629814 Left valve; Paratype 629815 Right valve; Paratype 629816 Carapace; Paratype 629817 Carapace; Paratype 629818 Left valve.
Dimensions of measured specimens.: n = 3. Length 0.67–0.62 mm, Height 0.24 mm.
Candona mojavensis n. sp.
Diagnosis Carapace elongate in side view, with acutely pointed posterior margin and narrowly rounded anterior margin. Dorsum with highest point posterior of center, where it curves into sloping, elongate, nearly straight posterodorsal margin; slight change in curvature anterior of nearly straight central dorsal margin. Venter concave; posteroventral margin gently convex. In dorsal view, sides convex with widest point at center, anterior compressed to slightly concave; long, narrow flanges on posterodorsal margins of both valves. Inner lamella much broader anteriorly than ventrally and posteriorly. Shell surface smooth. Males not observed. Fig. 2a, b.
Remarks his is an epigean species associated with wetland taxa. This species is distinguished by its elongated, acutely pointed posterior and differs from the similar species C. caudata and C. acuminata in its convex posteroventral margin.
Etymology Named for its geographic distribution in sites within the Mojave Desert, Nevada.
Holotype USNM 629812 Left valve; Paratype 629813 Right valve.
Dimensions of measured specimens, n = 3. Length, 1.10–1.24 mm, Height, 0.56–0.6 mm.
Candona pahrumpensis n.sp.
Diagnosis Carapace elongate oblong in side view; anterior and posterior margins smoothly rounded. Dorsum straight, subparallel to venter; venter straight, with slight central concavity. In dorsal view, sides very shallowly convex, anteriorly compressed into slight concavity. Valves nearly equal. Inner lamella relatively broad both anteriorly and posteriorly; anterior inner margin more nearly vertical than posterior inner margin. Adductor muscle scar pattern faintly visible, of Candona type. Shell surface smooth. No males observed. Fig. 4e–h.
Remarks This hypogean species is distinguished by its narrow, elongate outline and equally rounded extremities, made more prominent by the broad inner lamellae. It is very rare in our collections, possibly because of its delicate and very thin carapace.
Etymology pahrumpensis, named for its type locality of Pahrump Valley, Nevada.
Holotype USNM 629810 Left valve; Paratype 629811 Right valve.
Dimensions of measured specimens: n = 3. Length 0.64–0.68 mm, Height, 0.21–0.23 mm.
Genus Plicocandona n. gen. Type species: Plicocandona valentis n. sp.
Diagnosis Carapace trapezoidal to subtrapezoidal or triangular in outline with dorsum generally straight and relatively elongate. Left valve higher than right, with dorsal portion “folded” over so that dorsal margin is ventral of dorsal outline. Adductor muscle scar pattern like Candona with additional small circular scar anterior of capping scar (Fig. 5).
Remarks This genus appears to be restricted to an aquifer environment. It resembles Caribecandona Broodbakker in its preferred environment, small size, and general appearance (trapezoidal or triangular outline and what Broodbakker (1983) called a “dorsal protuberance or comb”), and also shares similar carapace features with species of Danielocandona Broodbakker and Schellencandona Meisch. These carapace features include shell outline, dorsal hinge, and valve size difference, with the left valve higher than the right valve. However, Caribecandona, Danielocandona, and Schellencandona have a standard candonid adductor muscle scar, different from that observed in these specimens. The most distinguishing taxonomic characteristic of this new genus is the adductor muscle scar pattern which displays an additional and circular scar anterior of the capping scar. In addition to this difference, the Nevada genus has, in general, longer and straighter dorsal margins than the other genera mentioned here. Broodbakker’s genus Caribecandona is the most similar, but his detailed drawings show the presence of a classic candonid adductor muscle scar, not the scar observed in these valves.
Etymology From the Latin plico, for fold.
Plicocandona valentis n. sp.
Diagnosis Carapace subtrapezoidal in side view. Dorsum arched, with slight concavity between two convexities at highest level; anterodorsal margin slopes into broadly rounded anterior margin, and posterodorsal margin slopes more steeply to pointed posterior extremity. Venter slightly concave. In dorsal view, sides convex with anterior compressed, widest at center or slightly anterior of center. Left valve higher than right, with dorsal portion “folded” over so that dorsal margin is ventral of dorsal outline; dorsal margin of right valve fits into hinge groove in dorsal margin of left valve. Adductor muscle scar pattern like Candona with additional small circular scar anterior of capping scar. Inner lamellae relatively broad are both anteriorly and posteriorly. Shell surface finely punctate. No males observed Fig. 6a–g.
Remarks This hypogean species is distinguished by its trapezoidal outline, “folded” dorsal margin, and finely punctate shell surface.
Etymology From the Latin valentis, for “strong, powerful”.
Holotype USNM 629819 Left valve; Paratype 629820 Right valve; Paratype 629821 Left valve; Paratype 629822 Right valve; Paratype 629823 Left valve.
Dimensions of measured specimens, n = 6. Length 0.79–0.82 mm, Height 0.42–0.49 mm.
Plicocandona cassicula n.sp.
Diagnosis Carapace subrectangular in side view; dorsum straight, subparallel to venter. Anterior extremity broadly rounded, posterior extremity truncated, with shallowly convex outline. In dorsal view, sides shallowly convex, with central portions subparallel or with slight central concavity, slightly compressed anteriorly. Left valve larger and higher than right, with dorsum folded into acute plication; posterodorsal outline sinuous and ventral outline concave with slight anteroventral sinuosity. Right valve with anterodorsal marginal sinuosity; anteroventral marginal sinuosity more pronounced than that of left valve. Relatively broad anterior inner lamella, slightly narrower posteriorly. Adductor muscle scar pattern like Candona with additional small circular scar anterior of capping scar. Shell surface markedly reticulate; fossae range from single pits to paired or triple pits to elongate slits. No males observed Fig. 7a–d, f.
Plicocandona cassicula n. sp. (a–f), Corn Creek Flat, CSF87-98, Units D-E, wetland. a Left valve exterior, b Left valve exterior, c Right valve exterior, d Right valve exterior, e Left valve exterior, showing reticulate surface, fossae, and adductor muscle scar. Plicocandona macraina n. sp., (e, g, h), Corn Creek Flat, CSF87-99, Units D-E, channel complex. f Left valve exterior, g Right valve, exterior, h Left valve interior
Remarks This hypogean species is distinguished by its long, straight dorsal plication, subrectangular outline, and reticulate surface ornamentation.
Etymology From the Latin, cassicula, “fine net”.
Holotype USNM 629824 Left valve; Paratype 629825 Right valve.
Dimensions of measured specimens, n = 4. Length 0.56–0.57 mm, Height, 0.27–0.32 mm.
Plicocandona macraina n. sp.
Diagnosis Carapace elongate in side view, with straight dorsum, rounded anterior and posterior extremities, centrally concave venter. In dorsal view, sides very shallowly convex, slightly compressed anteriorly. Left valve slightly larger than right; dorsal fold inconspicuous. Inner lamellae relatively broad, with posterior inner margin slanting toward central ventral margin of valve. Adductor muscle scar pattern like Candona with additional small circular scar anterior of capping scar. Surface very finely punctate. No males observed. Fig. 7e, g, h.
Remarks This hypogean species is distinguished by its relatively low, elongate outline. A rare species here, possibly because of thin, fragile carapace.
Etymology: From Greek makros for “long”.
Holotype: USNM 629826 Left valve; Paratype 629827 Right valve.
Dimensions of measured specimens, n = 5. Length 0.56–0.71 mm, Height 0.24–0.31 mm.
Plicocandona sagena n. sp.
Diagnosis Carapace subtrapezoidal in side view, with straight dorsum joining antero-dorsal and posterodorsal outline in rounded obtuse angles. Anterodorsal outline straight to very slightly concave, smoothly joining broadly rounded anterior margin. Posterodorsal outline straight, sloping to bluntly pointed posterior margin. Venter straight on either side of central concavity. In dorsal view, sides shallowly convex with straight or slightly concave central portion; slightly compressed anteriorly. Juveniles with more pronounced central, central anterior, and posteroventral concavities. Left valve slightly higher than right. Inner lamellae relatively broad. Adductor muscle scar pattern like Candona with additional small circular scar anterior of capping scar. Shell surface reticulate, with very subdued, papillate muri. No males observed Fig. 8a–d.
Plicocandona sagena n. sp., (a–d), Cactus Springs, CacSprMol33, Unit E wetland. a Left valve exterior, b Left valve exterior, c Right valve exterior, d Right valve exterior. Plicocandona striola n.sp., (e–h), Pahrump Valley, PVMol59, Unit B, wetland. e Left valve exterior, f Left valve exterior, g Right valve exterior, h Right valve exterior
Remarks This hypogean species is distinguished by its straight dorsum, reticulate surface ornamentation, and lack of a flange at the ventral concavity.
Etymology From the Latin, sagena, for “fishnet”.
Holotype USNM 629828 Left valve; Paratype 629829 Right valve; Paratype 629830 Left valve.
Dimensions of measured specimens, n = 4. Length 0.70–0.74, Height 0.35–0.36 mm.
Plicocandona striola n. sp.
Diagnosis Carapace subtrapezoidal in side view, with slightly concave dorsum joining sloping anterodorsal and posterodorsal outlines in rounded obtuse angles. Anterior margin broadly rounded, posterior margin pointed, venter straight. In dorsal view, sides subparallel in central portion, conspicuously concave anteriorly; also concave posteroventrally. Left valve slightly higher than right, with dorsum slightly concave between anterodorsal and posterodorsal convexities; dorsal fold not as pronounced as in other species of Plicocandona. Anterodorsal outline of left valve slightly concave. Right valve with straight dorsum, very slight concavity in center venter. Inner lamella relatively broad and nearly equal anteriorly and posteriorly; inner margin of anterior inner lamella nearly straight and subvertical, inner margin of posterior inner lamella sloping toward center venter. Plicocandonid adductor muscle scar pattern. Shell surface with irregular, subparallel, wrinkle-like carinae except on anterocentral and posterocentral areas. No males observed Fig. 8e–h.
Remarks This hypogean species is distinguished by its conspicuous anterior and posteroventral concavities and its distinct surface ornamentation of striations everywhere except on smooth anterocentral and posterocentral areas.
Etymology From Latin striola for “little furrow”.
Holotype USNM 629831 Left valve; Paratype 629832 Right valve; Paratype 629833 Left valve; Paratype 629834 Right valve.
Dimensions of measured specimens, n = 5. Length 0.67–0.70 mm, Height 0.32–0.36 mm.
Discussion
Hypogean ostracodes and paleohydrology
The assemblages present in these samples are composed of epigean (surface dwelling) and hypogean (groundwater dwelling) taxa. Most of the epigean species have Nearctic to Holarctic distributions as reported in the nonmarine ostracode databases NANODe (North American Nonmarine Ostracode Database, www.kent.edu/NANODe (Forester et al., 2005b)) and NODE (Nonmarine Ostracod Distribution in Europe (Horne et al., 1998)), and as listed in Martens & Savatenalinton (2011). They are typical of freshwater wetlands dominated by groundwater discharge. It is the hypogean forms that are of special interest here because their presence in the assemblages indicates a higher regional water table and vigorous groundwater discharge into valleys that do not experience such hydrologic activity today. Until recently, the working model to explain how large amounts of precipitation arrived in the Great Basin during these Late Pleistocene times involved the jet stream deflecting south of the ice sheet and bringing storm tracks to the region (COHMAP Members, 1988). However, this model has been challenged by a new hypothesis in which precipitation changes originate in the Gulf of Mexico and moves northward across the Great Basin (Lyle et al., 2012). Whether the precipitation was sourced in the westerly storm tracks (COHMAP Members, 1988) or from the Gulf of Mexico (Lyle et al., 2012), the fossil hypogean ostracodes occurring in these samples suggest that the delivery system for precipitation was markedly different compared to today, leading to a hydrologically active landscape at about 140 ka (MIS-Stage 6), 25–19 ka ~(Last Glacial Maximum); 18–15 ka ~(Post-Glacial) and 13–9 ka (~Late Glacial—early Holocene).
Hypogean ecosystems are not well understood and only partly explored. It has been about 35 years since Winograd & Robertson (1982, p. 1228) reported that carbonate aquifers in North America had dissolved oxygen levels of 2–8 mg/l at great depth and far from recharge points, including the Nevadan deep carbonate aquifer as discussed in this paper. In such alkaline environments, with fracture flow, dissolved oxygen, and chemolithotrophic bacteria, groundwater fauna including ostracodes can be found. Although a great deal of work since that time has been done generally on the crustacean fauna of these North American aquifers, little has been done on the ostracodes. There are several geographically extensive oxygenated carbonate aquifers in North America that have produced ostracode taxa in pump tests or high-discharge springs, with probably the best known case studies coming from the Edwards Aquifer in Texas (Longley, 1981, 1992). Studies of alluvial aquifer ostracode fauna from the Flathead River, Montana (Ward et al., 1994) and the Platte River, Colorado (Ward & Voelz, 1994) show the presence of a well established hyporheic fauna. However, few North American studies of hypogean ostracoda exist, in comparison with studies around the world, and particularly in Europe (Danielopol, 1980; Danielopol et al., 2000; Marmonier & des Chatelliers, 1992; Karanovic et al., 2015). Australia (Reeves et al., 2007) and the Caribbean (Broodbakker, 1983). Since ostracodes produce calcite bivalved shells, the hypogean species have potential as biomonitors of isotopic, geochemical, and biogeochemical conditions in aquifers. However, to make use of such information, fundamental knowledge of species identification and biogeography is required. Such basic information is also needed to aid in phylogenetic studies to better understand the evolutionary history of hypogean taxa.
Origins of hypogean ostracodes
The aquifer species described in these samples represent only a low percentage of the fauna, and share the general features of small size and trapezoidal to triangular shape. They are of the genus Candona or a closely related genus, here introduced as Plicocandona. They appear to be endemic to the region, that is, to our knowledge that these species are not known to occur elsewhere. These general characteristics of size and shape as well as their endemism are features shared by hypogean taxa reported from other regions around the world. Many researchers (see Danielopol, 1980; Broodbakker, 1983; Gibert et al., 1994 for overviews) over the years have raised questions about the origin(s) of the groundwater ostracode fauna: when did they invade the aquifers, and under what conditions? Two characteristics of these Late Quaternary fossil aquifer species tend to support conclusions presented by Danielopol et al. (2000) and subsequent authors (Reeves et al., 2007), which are that the candonids appear to be pre-disposed to a hypogean lifestyle, and that multiple invasions are likely to have occurred with a convergent evolutionary pressure guiding the small size and trapezoidal to triangular shapes that best accommodate an interstitial habitat. Although a definitive answer awaits genetic analysis, it seems likely that the presence of subpopulations identifiable by distinct muscle scar patterns supports the concept of multiple invasions. The timing of the invasions(s) by groundwater ostracode taxa into aquifers remains unknown.
As noted earlier, Winograd & Thordarson (1975) reported that the Cenozoic alluvial aquifers and the Paleozoic deep carbonate aquifer in the study region of southern Nevada are hydraulically connected, and that vertical flow occurs throughout the groundwater basin. Reeves et al. (2007) noted the association of ostracode shell characteristics with aquifer features in a study of the groundwater ostracodes of the Pilbara region of Australia. Smooth forms were associated with alluvial aquifers, whereas ornate forms were associated with elevated Mg2+ and lower pH, possibly from deeper aquifers. In the deposits from the southern Nevada, both smooth and ornate forms are found together in the assemblages, consistent with Winograd & Thordarson’s (1975) conclusion that the deep carbonate aquifer and the alluvial aquifers are hydraulically connected. Both forms were discharged into the springs and wetlands of the valley bottoms when the water table was high. However, more sampling and study is needed in order to test a relationship between shell morphology and aquifer type, and a future study should involve obtaining live ostracodes from wells and springs in the region for genetic analysis.
Conclusion
Ostracode assemblages can be used to identify past hydrologic changes on the landscape, including the rising and falling regional water table induced by hydroclimatic events. During the episodes of enhanced precipitation in the Late Pleistocene, the water table in the region rose, resulting in the discharge of aquifer species into wetlands and springs forming in the valley bottoms. Due to their calcite shells, ostracodes provide a way to identify not only the presence of groundwater discharge, but they also have the potential to be geochemical signals of precipitation source. The hypogean species recovered in these samples are unlike the species from the few groundwater sampling localities examined in other studies, and tend to support the hypothesis that multiple invasions of the groundwater have occurred through time, and that candonid ostracodes can easily adapt to the subterranean, interstitial environment. Further research is required to begin to understand the biogeographic significance for ostracodes of the extensive groundwater basin in southern Nevada.
References
Bell, J. W., A. R. Ramelli & S. J. Caskey, 1998. Geologic Map of the Tule Springs Park Quadrangle. Nevada Bureau of Mines and Geology, Reno. MAP 113, scale 1:24,000.
Bell, J. W., J. Caskey & P. K. House, 2009. Geologic Map of the Lahonton Mountains Quadrangle, Churchill County, Nevada. Nevada Bureau of Mines and Geology, Reno. Map 168, scale 1:24,000.
Broodbakker, N. W., 1983. The subfamily Candoninae (Crustacea, Ostracoda) in the West Indies: Amsterdam expeditions to the West Indian Islands, Report 34. Bijdragen tot de Dierkunde 53: 287–326.
COHMAP Members, 1988. Climate changes of the last 18,000 years: observations and model simulations. Science 241: 1043–1052.
Curry, B. B., L. D. Delorme, A. J. Smith, D. F. Palmer & B. J. Stiff, 2012. The biogeography and physiochemical characteristics of aquatic habitats of freshwater ostracodes in Canada and the United States. In Horne, D., J. Holmes, J. Rodrigez-Lasaro & F. Viehberg (eds) Ostracoda as Proxies for Quaternary Climate Change, Vol. 17., Developments in Quaternary Science series Elsevier Science Publishing, London: 85–115.
Danielopol, D. L., 1980. An essay to assess the age of the freshwater interstitial ostracods of Europe. Bijdragen tot de Dierkunde 50: 243–291.
Danielopol, D. L., P. Pospisil & R. Rouch, 2000. Biodiversity in groundwater: a large scale view. Trends in Ecology and Evolution 15: 223–224.
Forester, R. M., 1988. Nonmarine calcareous microfossil sample preparation and data acquisition procedures. U.S. Geological Survey Technical Procedure HP-78, R1:1–9.
Forester, R. M., 1991. Ostracode assemblages from springs in the western United States: implications for paleohydrology. In D. D. Williams & H. V. Danks (eds) Arthropods of Springs, with Particular Reference to Canada: Memoirs of the Entomological Society of Canada 155: 181–201.
Forester, R. M., T. K. Lowenstein & R. J. Spencer, 2005a. An ostracode based paleolimnologic and paleohydrologic history of Death Valley, 200 to 0 ka. Geological Society of America Bulletin 17: 1379–1386.
Forester, R. M., A. J. Smith, D. F. Palmer & B. B. Curry, 2005b. North American Ostracode Database “NANODe” Version 1, http://www.kent.edu/NANODe, Kent State University, Kent.
Gibert, J., J. A. Stanford, M.-J. Dole-Olivier & J. V. Ward, 1994. Basic attributes of groundwater ecosystems and prospects for research. In Gibert, J., D. L. Danielopol & J. A. Stanford (eds) Groundwater Ecology. Academic Press, San Diego: 7–40.
Hancock, P. J., A. J. Boulton & W. F. Humphreys, 2005. Towards an ecological understanding of groundwater. Hydrogeology Journal 13: 98–111.
Hartmann, G. & H. S. Puri, 1974. Summary of neontological and paleontological classification of Ostracoda. Hamburger Zoologisches Museum und Institut, Mitteilungen 70: 7–73.
Haynes Jr., C. V., 1967. Quaternary geology of the Tule Springs area, Clark County, Nevada. In Wormington, H. M. & D. Ellis (eds) Pleistocene Studies in Southern Nevada. Nevada State Museum of Anthropology, Carson City: 1–10.
Horne, D. J., A. Baltanas & G. Paris, 1998. Geographical distribution of reproductive modes in living non-marine ostracods. In Martens, K. (ed.), Sex and Parthenogenesis: Evolutionary Ecology of Reproductive Modes in Non-marine Ostracods. Backhuys Publishers, Kerkwerve: 77–100.
Humphreys, W. F., 2009. Hydrogeology and groundwater ecology: does each inform the other? Hydrogeology Journal 17: 5–21.
Jayko, A. S., R. M. Forester, D. S. Kaufman, F. M. Phillips, J. C. Yount, J. McGeehin, & S. A. Mahan, 2008. Late Pleistocene lakes and wetlands, Panamint Valley, Inyo County, California. In Reheis, M. C., R. Herchler & D. M. Miller (eds), Late Cenozoic Drainage History of the Southwestern Great Basin and Lower Colorado River Region: Geologic and Biotic Perspectives, Geological Society of America Special Papers 439: 151–184.
Karanovic, I., D. Sidorov & P. Marmonier, 2015. Zoogeography of the ostracod genus Nannocandona with a description of two new species from Europe, Annales de Limnologie. International Journal of Limnology 51: 297–313.
Kempf, E. K., 1997a. Index and Bibliography of non-marine ostracods. 6. Index A, Supplement 1. Sonderveröffentlichungen des Geologischen Institut der Universität zu Köln 109, 142 pp.
Kempf, E. K., 1997b. Index and Bibliography of non-marine ostracods. 7. Index B, Supplement 1. Sonderveröffentlichungendes Geologischen Institut der Universität zu Köln 110, 134 pp.
Kempf, E. K., 1997c. Index and Bibliography of non-marine ostracods. 8. Index C, Supplement 1. Sonderveröffentlichungen des Geologischen Institut der Universität zu Köln 111, 152 pp.
Kempf, E. K., 1997d. Index and Bibliography of non-marine ostracods. Bibliography C. Sonderveröffentlichungen des Geologischen Institut der Universität zu Köln 112, 150 pp.
Longley, G., 1981. The Edwards aquifer–Earth’s most diverse groundwater ecosystem? International Journal of Speleology 11: 123–128.
Longley, G., 1992. The subterranean aquatic ecosystem of the Balcones Fault Zone Edwards Aquifer in Texas - threats from over pumping. In Groundwater Ecology, First International Conference, Tampa, Florida, April 1992, Proceedings: American Water Resources Association, Bethesda, MD: 291–300.
Lyle, M., L. Heusser, C. Ravelo, M. Yamamoto, J. Barron, N. Diffenbaugh, T. Herbert & D. Andreasen, 2012. Out of the tropics: the Pacific, Great Basin Lakes, and Late Pleistocene water cycle in the western United States. Science 337: 1629–1633.
Maddocks, R., 1982. Ostracoda. In Bliss, D. E. (ed.), The Biology of Crustacea, Vol 1: Systematics, the Fossil Record and Biogeography. Academic Press, New York: 221–239.
Marmonier, P. & M. Creuze des Chatelliers, 1992. Biogeography of the benthic and interstitial living Ostracods (Crustacea) of the Rhone River (France). Journal of Biogeography 19: 693–704.
Martens, K. & S. Savatenalinton, 2011. A subjective checklist of the Recent, free-living, non-marine Ostracoda (Crustacea). Zootaxa 2855: 1–79.
Mawby Jr., J., 1967. Fossil vertebrates of the Tule Springs site, Nevada. In Wormington, H. M. & D. Ellis (eds) Pleistocene Studies in Southern Nevada. Nevada State Museum of Anthropology, Carson City: 105–128.
Meisch, C., 2000. Freshwater Ostracoda of Western and Central Europe. Spektrum Akademischer Verlag, Heidelberg: 522 pp.
Mehringer Jr., P., 1967. Pollen analysis of the Tule Springs area, Nevada. In Wormington, H. M. & D. Ellis (eds) Pleistocene Studies in Southern Nevada. Nevada State Museum of Anthropology, Carson City: 129–200.
Paces, J. B., R. M. Forester, J. F. Whelan, S. A. Mahan, J. P. Bradbury, J. Quade, L. A. Neymark & L. M. Kwak, 1996. Synthesis of ground-water discharge deposits near Yucca Mountain,, U.S. Department of Energy and U.S. Geological Survey, Yucca Mountain Project Branch, 1996 Milestone Report 3GQH671M, 75 pp.
Pigati, J., D. Miller, J. Bright, S. Mahan, J. Nekola & J. Paces, 2011. Chronology, sedimentology and microfauna of groundwater discharge deposits in the central Mojave Desert, Valley Wells, California. Geological Society of America Bulletin 123: 2224–2239.
Pigati, J. S., J. A. Rech, J. Quade & J. A. Bright, 2014. Desert wetlands in the geologic record. Annual Reviews in Earth Sciences 132: 67–81.
Quade, J., 1986. Late Quaternary environmental changes in the upper Las Vegas Valley, Nevada. Quaternary Research 26: 340–357.
Quade, J. & W. L. Pratt, 1989. Late Wisconsin groundwater discharge environments of the southwestern Indian Springs Valley, southern Nevada. Quaternary Research 31: 351–370.
Quade, J., M. D. Mifflin, W. L. Pratt, W. McCoy & W. L. Burckle, 1995. Fossil spring deposits in the southern Great Basin and their implications for changes in water-table levels near Yucca Mountain, Nevada, during Quaternary time. Geological Society of America Bulletin 107: 213–230.
Quade, J., R. M. Forester, W. L. Pratt & C. Carter, 1998. Black mats, spring-fed streams, and late-glacial-age recharge in the southern Great Basin. Quaternary Research 49: 129–148.
Quade, J., R. M. Forester & J. Whelan, 2003. Late Quaternary paleohydrologic and paleotemperature change in southern Nevada. In Enzel, Y., S. Wells, & N. Lancaster (eds) Paleoenvironments and paleohydrology of the Mohave Desert and southern Great Basin deserts, Geological Society of America Special Paper 368: 165–188.
Reeves, J., P. De Deckker & S. A. Halse, 2007. Groundwater ostracods from the arid Pilbara region of northwestern Australia: distribution and water chemistry. Hydrobiologia 585: 99–118.
Reheis, M. C., K. D. Adams, C. G. Oviatt & S. Bacon, 2014. Pluvial lakes in the Great Basin of the western United States, the view from the outcrop. Quaternary Science Reviews 97: 33–57.
Smith, A. J. & D. J. Horne, 2002. Ecology of marine, marginal marine, and nonmarine ostracods. In Chivas, A. R. & J. A. Holmes (eds) The Ostracoda: Applications in Quaternary Research. Geophysical Monograph Series, American Geophysical Union, Washington, DC: 37–64.
Smith, A. J. & D. F. Palmer, 2012. The versatility of Quaternary ostracods as palaeoclimate proxies: comparative testing of geochemical and ecological / biogeographical approaches. In Horne, D., J. Holmes, J. Rodrigez-Lasaro & F. Viehberg (eds) Ostracoda as Proxies for Quaternary Climate Change, Vol. 17., Developments in Quaternary Science series Elsevier Science Publishing, London: 183–203.
Springer, K. B., E. Scott, C. R. Manker & S. Rowland, 2011. Vertebrate paleontology of Pleistocene lakes and groundwater discharge deposits of the Mojave Desert and southern Great Basin. Nevada State Museum Paleontological Papers 1: 168–243.
Springer, K. B., C. R. Manker & J. S. Pigati, 2015. Dynamic response of desert wetlands to abrupt climate change. Proceeedings of the National Academy of Sciences 112: 14522–14526.
Sylvester-Bradley, P. C. & R. H. Benson, 1971. Terminology for surface features in ornate ostracodes. Lethaia 4: 249–286.
Van Morkhoven, F. P. C. M., 1962. Post-Palaeozoic Ostracoda, Vol. 1. Elsevier, London, General: 204.
Ward, J. & N. Voelz, 1994. Fauna of the South Platte River, Colorado. In Gibert, J., D. Danielopol & J. Stanford (eds) Groundwater Ecology. Academic Press, San Diego: 391–423.
Ward, J., J. Stanford & N. Voelz, 1994. Spatial distribution patterns of Crustacea in the floodplain aquifer of an alluvial river. Hydrobiologia 287: 11–17.
Winograd, I. J. & F. N. Robertson, 1982. Deep oxygenated ground water: anomaly or common occurrence? Science 216: 1227–1230.
Winograd, I. J. & W. Thordarsen, 1975. Hydrogeologic and hydrochemical framework, south-central Great Basin, Nevada-California, with special reference to the Nevada Test Site, United States Geological Survey Professional Paper 712-C, 125p.
Acknowledgements
We thank Koen Martens, Patrick De Deckker, and two anonymous reviewers for their help in improving this paper. AJS thanks Elizabeth Dirska for her assistance with Photoshop editing of the eSEM images.
This paper has a history. It began as one of the several incomplete manuscripts that Rick Forester was working on at the time of his death. Long after he and his coauthor Claire Carter had retired from the USGS, he continued to think about and discuss with colleagues the paleohydrologic significance of groundwater ostracode species, especially those from the southwestern U.S. The subject of hypogean ostracodes, their evolutionary ecology and environmental significance, was of great interest to him. Sadly, the original set of slides he planned to use as holotypes has vanished. However, using his original SEM photos and descriptions, reviewing his field maps, and repicking specimens from the original samples, we were able to account for most of the aquifer species originally described by him, and we prepared new holotype slides and new plates. A few species were so rare that we were not able to locate new material from the original samples. The paleowetlands of southern Nevada await further study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Richard M. Forester—deceased.
Claire Carter—retired.
Guest editors: Alison J. Smith, Emi Ito, B. Brandon Curry & Patrick De Deckker / Multidisciplinary aspects of aquatic science: the legacy of Rick Forester
Rights and permissions
About this article
Cite this article
Forester, R.M., Carter, C., Quade, J. et al. Aquifer and surface-water ostracodes in Quaternary paleowetland deposits of southern Nevada, USA. Hydrobiologia 786, 41–57 (2017). https://doi.org/10.1007/s10750-016-2966-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2966-5