Abstract
The effects of the El Niño Southern Oscillation (ENSO)-mediated environmental changes to marine resources were long recognized; however, species-specific responses were also reported to vary, possibly more so, under the emerging event-to-event diversity of the ENSO conditions. Hence, the objective of this study is to characterize the potential impacts of the ENSO-regulated environmental variability to squid habitat in the central North Pacific under the different ENSO flavors, using habitat models developed from at least a decade of fishery-dependent dataset and environmental parameters. Our findings revealed that the potential squid habitats were largely influenced by ENSO-forced environmental changes during the squid’s spawning and nursery periods, resulting in substantial reduction/enhancement of available habitats in the succeeding summers of Central Pacific El Niño/La Niña, where the latter led to an expansion of favorable spawning and nursery grounds. However, the autumn–winter periods of weaker and short-lived Eastern Pacific El Niño showed elevated potential habitats due to minimal sea surface temperature drop and close proximity of spawning and nursery grounds to optimal feeding environments. The quick, albeit variable, squids’ responses to ENSO flavors accentuate their promising potential as ecological beacons under climate changes, aiding the development of adaptive management strategies for commercially exploited fisheries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cephalopods are short-lived species that quickly respond to the drastic changes in the environment (Rodhouse, 2001; Coll et al., 2013). Both the short- and long-term fluctuations in their abundance and distribution were attributed to the availability of the optimal conditions for feeding (Martins & Perez, 2007; Robinson et al., 2013) and spawning (Waluda et al., 2001; Waluda & Rodhouse, 2006). Optimal feeding and spawning conditions for the squid and other marine fisheries are, in turn, largely influenced by environmental variability driven by climate changes occurring across different spatial and temporal scales (Sakurai et al., 2000; Tian et al., 2013).
The neon flying squid, Ommastrephes bartramii (Lesueur, 1821), is a colossal oceanic cephalopod that thrives in the subtropical and temperate waters of the global ocean (Bower & Ichii, 2005). In the North Pacific, O. bartramii population is made up of two seasonal cohorts—winter–spring and autumn spawning groups, which are linked with the basin-wide oceanic circulation and with life history stages very responsive to the changes in oceanographic regimes (Ichii et al., 2009, 2011). Both squid cohorts undergo seasonal migration between their spawning (subtropical region) and foraging grounds (transition and subarctic), and like most cephalopods, neon flying squids are opportunistic species that prey on zooplankton, other squids, and myctophid fishes (Watanabe et al., 2004). O. bartramii is also an important prey of apex predators such as blue sharks (Seki, 1993) and marine mammals (Mori et al., 2001).
One of the extensively studied climatic events in the literature is the El Niño Southern Oscillation (ENSO), which represents the strongest signal in the inter-annual variation of ocean–atmosphere system (Wang et al., 1999; Latif & Keenlyside, 2009), with its cycle comprised of the warm (El Niño) and cold (La Niña) climate phases occurring at periods of roughly 3–4 years (McPhaden, 1999). While ENSO is primarily a tropical event, it has far-reaching global impacts on the terrestrial and aquatic biota (Holmgren et al., 2001; Hoegh-Guldberg & Bruno, 2010; Doney et al., 2012). Earlier studies on cephalopods further revealed that El Niño and La Niña-mediated environmental variations largely affected the populations of juvenile (Anderson & Rodhouse, 2001) and adult jumbo squids (Ichii et al., 2002) in the Eastern Tropical Pacific Ocean. Moreover, in western North Pacific (WNP) Ocean, El Niño and La Niña events from 1995 to 2004 were also reported to impact the recruitment rates of the winter–spring spawning cohort of neon flying squid (Chen et al., 2007; Cao et al., 2009). The results of these studies revealed that El Niño/La Niña resulted to the subsequent increase/decrease of squid recruitment, through variability in the environmental conditions on its spawning grounds. Another recent work on the variability of suitable habitats from July to November, 1998–2009 for winter–spring cohort of neon flying squid, however, revealed opposite impacts such that El Niño/La Niña resulted in low/high squid habitat quality (Yu et al., 2015). While there are a number of studies reporting the effects of ENSO events to the winter–spring cohort of neon flying squid, information on the impacts of similar climate episodes to summer potential habitats of the autumn spawning cohort was limited. Previous studies on the autumn cohort have investigated the effects of large-scale climate shifts from 1979 to 2006, manifested in basin-wide changes in sea surface temperature (SST) and sea level anomaly, to squid stock recruitment (Ichii et al., 2011) and explored the relationships between decadal climate phase shifts, such as the Pacific Decadal Oscillation, to its abundance (Igarashi et al., 2015).
Therefore, in the present study, we challenged to address this potential research gap by examining the inter-annual variability in the spatial patterns of summer potential habitat for the autumn cohort in relation to the ENSO-driven environmental changes from June to July, 1999–2010. During this period, the ENSO conditions further comprised the conventional El Niño (Eastern Pacific, EP)/La Niña episodes and the recently reported Central Pacific (CP) El Niño (Ashok & Yamagata, 2009), thereby providing a platform to explore the impacts of the two El Niño flavors to squid potential habitat. It has long been recognized that El Niño events display a broad array of amplitudes, triggers, patterns, and life cycles; however, the cognizance that the ENSO’s impacts can be sensitive to this event-to-event diversity is driving a renewed interest (Capotondi et al., 2014). Here, we have designed our analyses to elucidate the potential effects of this event-to-event diversity on squid fishery resource in central North Pacific. To do so, we constructed monthly squid habitat models using regression-based generalized additive models (GAMs), combining Oceanic Niño Index (ONI) during the squid spawning and nursery periods and parsimonious suite of environmental factors selected based on the findings of our recent studies (Alabia et al., 2015a, b, c), to predict the abundance-based potential squid habitat. Our earlier results from effort-based (presence-only) squid habitat model showed the high environmental importance of SST, sea surface salinity (SSS), and sea surface height (SSH) on the formation of seasonal potential habitat and pelagic hotspots for neon flying squid in the North Pacific. These environmental factors were reported to affect the distributions and abundance of marine species (Muhling et al., 2011; Vijai et al., 2014) and were also considered as indicators of oceanographic features, critical for foraging and transport (Godø et al., 2012; Mugo et al., 2014).
As neon flying squid exhibits a life-span of 1 year (Yatsu et al., 1997; O’Dor, 1998), our assumption is that the summer potential squid habitats are directly affected by the fluctuations in recruitment levels from preceding spawning and nursery periods, which in turn, coincide with the peak of the ENSO signals. While the effects of ENSO to the marine resources were widely recognized, species-specific biological responses to these events were also known to vary across the spatial and temporal scales and as such could provide contemporary insights imperative for fisheries management. Hence, the aim of the present study is to characterize the impacts of ENSO-driven environmental changes to potential habitat for autumn cohort of the neon flying squid in central North Pacific. We hypothesize that summer potential squid habitats have different responses to CP- and EP-El Niño-driven environmental conditions.
Materials and methods
Fishery-dependent data
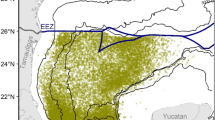
The squid fishery dataset was collected and provided by the Aomori Prefecture Industrial and Technology Research Center and included information on fishing date (month, day and year), squid fishing locations (longitude and latitude), and catch per unit effort (CPUE, in raw catch ton/vessel/day) from June to July, 1999–2010. All the fishing vessels in operation during the 12-year period recorded positive squid catches, and hence, all the CPUEs have non-zero values. Here, we used CPUE as relative index of squid abundance, assumed as relative proxies of stock level (Nishikawa et al., 2014) and adult squid potential habitat, and defined as areas of moderate–high predicted squid abundance. Japanese commercial squid jigging fishery in summer operates offshore, covering the CP waters from 170° E and 160° W and 36°–46° N (Fig. 1), and primarily targets the autumn spawning cohort. Almost all of the commercial vessels in the database were categorized as medium class size (138–183 GT) vessels, which were further equipped with an average of 24 ± 5 automatic jigging machines.
Map showing the spawning and feeding habitats and migration patterns of seasonal spawning cohorts of neon flying squid in literature (Ichii et al., 2011). Region exploited by Japanese commercial squid jigging fishery is shown in the light gray-filled area, and slanted line-filled box corresponds to Niño 3.4 region, where SST anomalies for Oceanic Niño Index (ONI) were computed
Oceanographic variables
The oceanographic datasets used for the habitat model construction comprised the daily SST from the optimally interpolated AVHRR-OI SST and altimetry-derived SSH from the AVISO website (ftp://ftp.aviso.oceanobs.com). SSS was obtained from the multi-variate ocean variational estimation system for the WNP, developed by Meteorological Research Institute, Japan Meteorological Agency (Usui et al., 2006). To examine the potential effects of ENSO-mediated environmental changes to squid’s spawning and feeding habitats, ONI provided by NOAA Climate Prediction Center (http://www.cpc.ncep.noaa.gov/products/analysis_monitoring/ensostuff/ensoyears.shtml) was also used as one of the model covariates. The ONI is defined as the 3-month running mean of the ERSST.v4 SST (Huang et al., 2015) anomalies in the Niño 3.4 region (120°–170° W, 5° N–5° S) and is based on the centered 30-year base period (1981–2010). Periods of below and above the ±0.5°C SST anomaly (SSTA) threshold for a minimum of five consecutive overlapping seasons were considered as La Niña and El Niño events, respectively. El Niño events were further classified as either EP (canonical) or CP (dateline) El Niño based on the location of the maximum SSTAs in the tropical North Pacific (Ashok & Yamagata, 2009; Kug et al., 2009). Monthly Reynolds and Smith OISST version 2 anomalies with respect to the 1971–2000 base period were obtained from Columbia University IRI/LDEO data library (http://iridl.ldeo.columbia.edu/). These data were mapped to examine the spatial SSTA distribution under representative ENSO events. The monthly averaged datasets of sea-viewing wide field-of-view sensor chlorophyll-a (Chl-a) products, used as proxy for food availability (Ichii et al., 2009; Robinson et al., 2013), were obtained from NOAA ERDDAP (http://coastwatch.pfeg.noaa.gov) website and were used to examine the meridional shifts of transition zone Chl-a front (TZCF) under the different ENSO periods.
Generalized additive model (GAM) construction
Regression-based statistical model approaches, such as GAM, are among the widely used analytical tools for marine species distribution modeling (Howell et al., 2010; Forney et al., 2012). GAM is an empirical approach that maximizes the log-likelihood by replacing the linear form of likelihood-based regression model by a sum of smooth functions and was proven useful for uncovering the nonlinear covariate effects (Hastie & Tibshirani, 1986). In the present study, the oceanographic variables, seasonally averaged ONI values from the autumn to winter (September–February) of the year prior to squid’s recruitment into the fishery (succeeding summer; June–August) and the log-transformed fishery-dependent squid abundance information (CPUE) were used to develop the monthly GAMs for June and July, 1999–2010, to separately examine the effects of the ENSO-mediated environmental variability to the inter-annual pattern of the squid potential habitats in summer following specific ENSO periods. Separate GAMs for the summer months (June and July) were constructed to examine the individual contributions of the oceanographic and climate variables to the predicted squid abundance and potential habitats. Moreover, logarithmic transformation of squid CPUE was implemented as raw the fishery data were asymmetrical. Model fitting and predictions were performed using the mgcv package version 1.7-29 (Wood, 2011) in R software version 3.2.1 (R Core Team, 2015). The GAMs applied to elucidate the potential squid habitat stakes the form
where g is the link function, lnCPUE is the log-transformed CPUE (response variable), α 0 is the model constant, s is the smooth function for each of the environmental predictors, and ε is the random error. As the squid CPUE assumes a continuous distribution, we used the identity link function of Gaussian model. The resulting abundance-based squid habitat predictions were then re-transformed to raw CPUE values for subsequent mapping, where time–latitude CPUE predictions were used to examine the spatio-temporal potential squid habitat patterns. All the plotting routines were performed using an open-source mapping package, generic mapping tools version 4.5.9 (Wessel et al., 2013).
Spatio-temporal correlation analyses
Cross-correlation analyses between the ONI and environmental variables (SSTA, SSH and SSS) were carried out to examine the significant temporal leads and lags in the spatial responses of environmental parameters to the ENSO-mediated environmental changes on the squid feeding grounds. All the environmental data used for the analyses were initially low-pass filtered using the 3-month running mean similar to the temporal smoothing of the ONI data. The putative spawning, nursery, and feeding zones of autumn cohort and respective timings (Yatsu et al., 1998; Ichii et al., 2006) in the study region are summarized in Table 1.
Results
ENSO episodes and squid abundance
From 1999 to 2010, a total of four El Niño and five La Niña events have transpired based on the ONI time series (Fig. 2A). The El Niño events were further categorized into two moderate–strong CP- (\(\overline{\text{ONI}}\) ≥+1°C for 10 months) and two weak–moderate EP-El Niño (+0.5 ≥ \(\overline{\text{ONI}}\) > +1°C, from 5 to 7 months). Furthermore, three out of the five La Niña recorded were also characterized by high ONI magnitude (\(\overline{\text{ONI}}\) <−1°C) persisting over long duration (10–21 months) and with ENSO-neutral periods, varying from 5 to 16 months interspersed in between the ENSO episodes. The actual squid CPUE during the 12-year period also reflected the inter-annual fluctuations (Fig. 2B) likely related to remote ENSO forcing experienced during their spawning and nursery seasons (autumn–winter, September–February), which generally correspond to the months when ENSO develops and reaches maximum strength (Stuecker et al., 2013). Relative squid abundance was highest in following summers of La Niña-dominated spawning and nursery years, moderately high during EP-El Niño and ENSO-neutral and lowest during CP-El Niño (Fig. 2B). The linear regression between the annual averages of autumn–winter ONI and squid CPUE of following summers also yielded significant negative correlations (R = −0.51, P < 0.1, n = 12) that accounted for at least 26% of the variance observed in the squid summer abundance from 1999 to 2010.
Time series of A 3-month running average of the Oceanic Niño Index (ONI) with dotted line corresponding to ±0.5°C SST anomaly threshold and B annually averaged summer (June–July) CPUE, coded based on ENSO conditions during the spawning and nursery periods (autumn–winter), preceding the recruitment into fishery (summer) from 1999 to 2010. Dot-filled periods correspond to La Niña events, and gray and black-filled years correspond to Central Pacific (CP) and Eastern Pacific (EP) El Niño, respectively
Environmental and climate effects on squid potential habitats
Figure 3A, B shows the smooth functions of each environmental and climate predictor derived from the monthly base models, capturing the optimal environmental ranges characterizing the summer potential squid habitat. All the environmental covariates showed significant effects on squid’s relative abundance based on the F-statistics (i.e., the ratio between the explained and unexplained variances), with the highest values derived for ONI and SST, suggesting the high relative contribution of these factors to the potential squid habitat (Table 2). The smooth curves of environmental variables on the squid feeding grounds further revealed different responses between June (Fig. 3A) and July (Fig. 3B) as the squid habitat shifted from the northern part of TZ and towards the SAFZ as the summer season progresses. From June to July, SST ranges on the squid fishing regions shifted 1°C warmer with the highest fraction found in the waters with SST from 11 to 18°C, showing positive effects on the relative squid abundance between 11 and 13.5°C in June and between 12 and 15°C in July. Meanwhile, SSS and SSH ranges dropped by as much as 0.4 psu and 12 cm, respectively, from June to July. However, the ONI showed similar bimodal curves in both models, where the squid spawning and nursery periods under the moderate to strong La Niña/El Niño resulted in positive/negative impacts on the squid abundance, with magnitudes higher in July relative to June (Fig. 3A, B, rightmost panel).
Spatial and temporal patterns of squid potential habitats
Based on the GAM-derived spatial and temporal distributions of the summer potential habitat (i.e., defined as regions of moderate–high relative squid abundance) on squid's putative feeding grounds (Fig. 4), the highest degree of spatial variability in the potential habitats also occurred off the SAFZ (40°–46° N) with a prominent increase from 1999 to 2001 and from 2005 to 2008 under the long La Niña periods and alternating periods of the EP-El Niño and La Niña, respectively. However, during the autumn–winter periods under CP-El Niño events of 2002–2003 and 2009–2010, the squid potential habitat exhibited the largest reduction which also coincided with years of the lowest observed relative squid abundance. Furthermore, the distribution patterns of observed squid abundance in summer revealed apparent shifts in the latitudinal distribution centroids, such that the summers following La Niña generally showed the mean distributions farther to the north relative to ENSO-neutral and El Niño periods, when the distribution centroids were located south of the subarctic boundary at 40° N. These northward displacements, captured on both the actual squid abundance and predicted abundance-based squid habitats during La Niña-preceded summers, could provide higher potential for the squids to exploit the more productive waters of the SAFZ, thereby generating better foraging opportunities.
Time–latitude plot of the annually averaged predicted squid abundance from June to July, 1999–2010. ENSO conditions from autumn to winter were indicated in the north axis, and latitudinally averaged observed summer squid abundance was overlain on the plot. The red line corresponds to latitudinal centroids of squid distribution for summer of each year
Environmental patterns during the different ENSO flavors
Figure 5A–D shows the spatial distributions of the SSTAs and meridional positions of the TZCF corresponding to the 0.2 mg/m3 Chl-a contour (Polovina et al., 2001) in the North Pacific Ocean averaged from autumn to winter and summer of the representative ENSO events. During the low magnitude EP-El Niño (Fig. 5A, left panel), the temperate central North Pacific area between 30° and 50° N showed predominantly positive SSTAs, with spawning and nursery grounds for squid’s autumn cohort characterized by the minimal SST departures from the 30-year base period, while the summer following this event (Fig. 5A, right panel) highlighted the presence of warm squid feeding grounds flanked by cold water masses on western and eastern parts of the basin. However, during CP-El Niño condition (Fig. 5B, left panel), the subtropical (20°–30° N) and subarctic (40°–50° N) regions of the North Pacific experienced colder than normal autumn–winter periods with extensive regions of negative SSTAs encroaching past the squid spawning and nursery grounds off the TZ. Similarly, the summer following the CP-El Niño (Fig. 5B, right panel) showcased the predominantly warm waters on the squid feeding grounds with cold water parcel displaced further north. During the ENSO-neutral (Fig. 5C, left panel) and La Niña (Fig. 5D, left panel) episodes, on the other hand, the central North Pacific Ocean was characterized by warmer waters that extended throughout the TZ, with the highest magnitude of warming observed in the latter. The summers following the ENSO-neutral (Fig. 5C, right panel) and La Niña (Fig. 5D, right panel), moreover, revealed the highest and lowest extents of warming relative to other ENSO periods, respectively. Despite the prominent spatial changes in autumn–winter and summer SST patterns during different ENSO flavors, the mean geographical locations of TZCF feature remained proximal to the spawning, nursery, and feeding grounds of the squid’s autumn cohort (Fig. 5A–D). However, the highest southward TZCF displacement in autumn–winter and summer occurred during CP-El Niño (34.32° N) and La Niña (39.70° N) periods, respectively. The southward meridional shifts in mean seasonal TZCF positions were coincident with the cooling patterns on the squid spawning and nursery grounds as well as squid feeding regions. The highest northward TZCF shifts, moreover, were observed during EP-El Niño from autumn–winter (36.63°N) and CP-El Niño-preceded summer (41.50°N).
Spatial SST anomaly distributions for the representative autumn–winter and summer periods during A EP-El Niño, B CP-El Niño, C ENSO-neutral, and D La Niña. Overlain are seasonal mean (autumn–winter and summer) positions of the TZCF (black lines) and squid spawning, nursery, and feeding grounds during the succeeding summer (region enclosed by green lines), as described in the present study (Table 1)
Further analyses of temporal lags between the ONI and SSTAs on squid feeding grounds (Fig. 6A) revealed that the squid feeding grounds subsequently cooled/warmed between 2 and 7 months after an El Niño/La Niña occurrence. It is also worth noting that the significant negative lags between ONI and feeding ground SSTA of periods between 8 and 12 months were also apparent, suggesting that the warming/cooling of squid feeding grounds leads to the occurrence of El Niño/La Niña, after 8 to 12 months. The significant negative spatial correlations between ONI and SSH (Fig. 6B) were also evident, with positive temporal lag that slightly advances with periods from 4 to 8 months, suggesting that the impacts of ENSO events to SSH on squid foraging grounds occurred slightly later than that observed in SSTA signals. However, the SSS on the same region highlighted significant positive correlations with shorter positive temporal lags (5–6 months) relative to SSTA and SSH temporal patterns (Fig. 6C). All positive temporal lags corresponding to periods when ENSO-mediated environmental changes exerted significant impacts on the squid feeding conditions, for all the environmental variables peaked at 5 months.
Temporal (left panels) and spatial (right panels) relationships between Oceanic Niño Index (ONI) and A SST anomaly, B SSH, and C SSS on the squid feeding grounds based on the highest significant spatial (P = 0.05) and temporal (5 month lag) relationships, derived from cross-correlation analyses with a 12-month lag. Broken lines on the lagged time series plots (left panels) represent significance at 95% confidence level
Discussion
The present study analyzed the impacts of recent ENSO conditions to potential summer habitat for the autumn cohort of neon flying squid in the central North Pacific, through the possible ENSO-regulated environmental changes on the spawning, nursery, and adult foraging grounds, deduced from abundance-based habitat predictions of the GAMs during a 12-year period. The summer potential squid habitats were largely influenced by ONI from autumn to winter, suggesting that the ENSO events significantly impacted the prevailing oceanographic conditions on the squid spawning and nursery grounds, consequently affecting potential recruitment into commercial fisheries in the following summers. For most short-lived species such as squids, recruitment success dominates the annual changes in stock abundance and is heavily dependent on the physical and biological environment within their spawning and nursery grounds (Pierce et al., 2008; Rosa et al., 2011). In the same manner, associated changes in recruitment of squid species under anomalous environments have led to the apparent changes in fishery yields (Brodeur et al., 2006; Thiaw et al., 2011).
Based on the results of our study, it appears that despite the favorable foraging conditions on the squid spawning and nursery grounds during CP-El Niño (2009), marked with the highest southward TZCF displacement, the pronounced drop in the SST presumably resulted in adverse effects on the pre-recruit survival, leading to a subsequent reduction in summer potential squid habitats relative to other ENSO periods. The substantial southward displacement of the TZCF during the CP-El Niño relative to EP-El Niño consequently advects more productive yet colder waters from the northern latitudes farther down south (Ayers & Lozier, 2010), past the autumn cohort’s spawning, and nursery grounds on the TZ. Following these results, it is most likely that the recruitment of the autumn cohort could be highly dependent on the critical balance between the availability of optimal spawning grounds and quality of pre-recruit’s feeding environments, with the former likely to be more limiting than the latter under an ENSO-dominated condition. This is further supported by the observation that the putative spawning and nursery grounds for autumn cohort of neon flying squids were generally found proximal to the TZCF frontal feature under all the representative ENSO events, thereby providing the squids access to the productive environment of central North Pacific with moderate–high Chl-a (Alabia et al., 2015b).
Moreover, the potential squid habitat distributions in summer were also affected by the environmental changes on squid’s feeding grounds following the ENSO events, primarily from associated changes in SST, with the second highest F-statistics in all monthly models, followed by SSS and SSH, which are oceanographic indicators of water masses and mesoscale activity, respectively. The leading negative CCF coefficients between the ONI and SSTA, ONI and SSH, and ONI and SSS on the putative squid foraging grounds, suggested that the ENSO-mediated environmental changes impose significant yet delayed effects on thermohaline properties and mesoscale activities on the squid feeding regions. The El Niño-preceded summers were marked by colder and saltier waters with weaker mesoscale activity, in contrast to the La Niña-preceded summers, where the warmer and calmer conditions in the latter potentially enhance biological productivity on the SAFZ, as the primary producers are likely to remain above the sun-lit zone (Sasaoka et al., 2002). These environmental conditions create favorable feeding states for higher trophic species including squids, on the nutrient-rich yet light-limited subarctic region.
While the environmental conditions during CP-El Niño resulted in possible deleterious effects on the spawning and nursery habitats for the autumn cohort of neon flying squids, these conditions, meanwhile, could be favorable for the winter–spring squid spawners off the CP, creating better foraging opportunities in the latter’s otherwise nutrient limited (Ichii et al., 2009), yet warmer spawning and nursery regions on the southern end of the subtropical zone (Vijai et al., 2014). As such, investigation on this potential gap should be examined in the future as existing information to date was only limited to the winter–spring cohort west of the dateline (Chen et al., 2007; Yu et al., 2015). Nonetheless, our findings highlighted that ENSO-regulated environmental changes revealed significant and dynamic impacts on summer potential habitats for the autumn cohort of neon flying squids in central North Pacific. More so, while we reported the impacts of CP- and EP-El Niño-driven environmental changes to major squid habitats, it is imperative to recognize that the CP-El Niño events transpiring from 1999 to 2010 were stronger in magnitude and longer in duration than the EP-El Niños and may dampen the environmental effects of the latter to squid habitats in the study region. Thus, future studies should also explore the impacts of El Niño flavors of comparable strength to summer potential squid habitats.
Finally, the differences in the spatial responses of the potential squid habitats under the quite diverse flavors of ENSO further underpin the importance of developing robust strategies for managing squid resources under a rapidly changing climate. As potential impacts of ENSO-driven environmental changes between the seasonal squid cohorts are likely to vary with ENSO events, current management schemes will also need to be restructured to adapt to the predicted changes in squid distribution and abundance. Depending on the prevailing ENSO episode, the targeted squid fishery is likely to shift between seasonal cohorts to optimize both the economic and ecological profits among stakeholders and as such could potentially translate to alterations in the timing and duration of squid resource exploitation in the central North Pacific. Moreover, in a broader context, the apparent quick responses of neon flying squids to the drastic ENSO-mediated environmental signals (owing to their relatively short life-span and fast growth rates), as manifested in large inter-annual fluctuations in their relative abundance, further accentuate their paramount potential as ecosystem sentinels and productivity integrators (Ichii et al., 2011), under the impending threats of climate change. Thus, monitoring the spatial and temporal squid habitat distribution patterns is likely to facilitate and advance our understanding of the complex ecological impacts of the abrupt climatic changes.
References
Alabia, I. D., S.-I. Saitoh, R. Mugo, H. Igarashi, Y. Ishikawa, N. Usui, M. Kamachi, T. Awaji & M. Seito, 2015a. Seasonal potential fishing ground prediction of neon flying squid (Ommastrephes bartramii) in the western and central North Pacific. Fisheries Oceanography 24(2): 190–203.
Alabia, I. D., S.-I. Saitoh, R. Mugo, H. Igarashi, Y. Ishikawa, N. Usui, M. Kamachi, T. Awaji & M. Seito, 2015b. Identifying pelagic habitat hotspots of neon flying squid in the temperate waters of the central North Pacific. PLoS One 10(11): e0142885.
Alabia, I. D., S.-I. Saitoh, H. Igarashi, Y. Ishikawa, N. Usui, M. Kamachi, T. Awaji & M. Seito, 2015c. Future projected impacts of ocean warming to potential squid habitat in western and central North Pacific. ICES Journal of Marine Science. doi:10.1093/icesjms/fsv203.
Anderson, C. I. H. & P. G. Rodhouse, 2001. Life cycles, oceanography and variability: ommastrephid squid in variable oceanographic environments. Fisheries Research 54(1): 133–143.
Ashok, K. & T. Yamagata, 2009. Climate change: the El Nino with a difference. Nature 461(7263): 481–484.
Ayers, J. M. & M. S. Lozier, 2010. Physical controls on the seasonal migration of the North Pacific transition zone chlorophyll front. Journal of Geophysical Research: Oceans 115(C5): 1–11.
Bower, J. R. & T. Ichii, 2005. The red flying squid (Ommastrephes bartramii): a review of recent research and the fishery in Japan. Fisheries Research 76(1): 39–55.
Brodeur, R. D., S. Ralston, R. L. Emmett, M. Trudel, T. D. Auth & A. J. Phillips, 2006. Anomalous pelagic nekton abundance, distribution, and apparent recruitment in the northern California Current in 2004 and 2005. Geophysical Research Letters 33(22): L22S08.
Cao, J., X. J. Chen & Y. Chen, 2009. Influence of surface oceanographic variability on abundance of the western winter–spring cohort of neon flying squid Ommastrephes bartramii in the NW Pacific Ocean. Marine Ecology Progress Series 381: 119–127.
Capotondi, A., A. T. Wittenberg, M. Newman, E. Di Lorenzo, J.-Y. Yu, P. Braconnot, J. Cole, B. Dewitte, B. Giese, E. Guilyardi, F.-F. Jin, K. Karnauskas, B. Kirtman, T. Lee, N. Schneider, Y. Xue & S.-W. Yeh, 2014. Understanding ENSO diversity. Bulletin of the American Meteorological Society 96(6): 921–938.
Chen, X. J., X. H. Zhao & Y. Chen, 2007. Influence of El Niño/La Niña on the western winter–spring cohort of neon flying squid (Ommastrephes bartramii) in the northwestern Pacific Ocean. ICES Journal of Marine Science 64(6): 1152–1160.
Coll, M., J. Navarro, R. J. Olson & V. Christensen, 2013. Assessing the trophic position and ecological role of squids in marine ecosystems by means of food-web models. Deep Sea Research Part II: Topical Studies in Oceanography 95: 21–36.
Doney, S. C., M. Ruckelshaus, J. Emmett Duffy, J. P. Barry, F. Chan, C. A. English, H. M. Galindo, J. M. Grebmeier, A. B. Hollowed, N. Knowlton, J. Polovina, N. N. Rabalais, W. J. Sydeman & L. D. Talley, 2012. Climate change impacts on marine ecosystems. Annual Review of Marine Science 4(1): 11–37.
Forney, K., M. Ferguson, E. Becker, P. Fiedler, J. Redfern, J. Barlow, I. Vilchis & L. Ballance, 2012. Habitat-based spatial models of cetacean density in the eastern Pacific Ocean. Endangered Species Research 16(2): 113–133.
Godø, O. R., A. Samuelsen, G. J. Macaulay, R. Patel, S. S. Hjøllo, J. Horne, S. Kaartvedt & J. A. Johannessen, 2012. Mesoscale eddies are oases for higher trophic marine life. PLoS One 7(1): e30161.
Hastie, T. & R. Tibshirani, 1986. Generalized additive models: rejoinder. Statistical Science 1(3):314–318. doi:10.1214/ss/1177013609.
Hoegh-Guldberg, O. & J. F. Bruno, 2010. The impact of climate change on the world’s marine ecosystems. Science 328(5985): 1523–1528.
Holmgren, M., M. Scheffer, E. Ezcurra, J. R. Gutiérrez & G. M. J. Mohren, 2001. El Niño effects on the dynamics of terrestrial ecosystems. Trends in Ecology and Evolution 16(2): 89–94.
Howell, E. A., D. R. Hawn & J. J. Polovina, 2010. Spatiotemporal variability in bigeye tuna (Thunnus obesus) dive behavior in the central North Pacific Ocean. Progress in Oceanography 86(1–2): 81–93.
Huang, B., V. F. Banzon, E. Freeman, J. Lawrimore, W. Liu, T. C. Peterson, T. M. Smith, P. W. Thorne, S. D. Woodruff & H.-M. Zhang, 2015. Extended reconstructed sea surface temperature version 4 (ERSST.v4). Part I: upgrades and intercomparisons. Journal of Climate 28(3): 911–930.
Ichii, T., K. Mahapatra, T. Watanabe, A. Yatsu, D. Inagake & Y. Okada, 2002. Occurrence of jumbo flying squid Dosidicus gigas aggregations associated with the countercurrent ridge off the Costa Rica Dome during 1997 El Niño and 1999 La Niña. Marine Ecology Progress Series 231: 151–166.
Ichii, T., K. Mahapatra, H. Okamura & Y. Okada, 2006. Stock assessment of the autumn cohort of neon flying squid (Ommastrephes bartramii) in the North Pacific based on past large-scale high seas driftnet fishery data. Fisheries Research 78(2–3): 286–297.
Ichii, T., K. Mahapatra, M. Sakai & Y. Okada, 2009. Life history of the neon flying squid: effect of the oceanographic regime in the North Pacific Ocean. Marine Ecology Progress Series 378: 1–11.
Ichii, T., K. Mahapatra, M. Sakai, T. Wakabayashi, H. Okamura, H. Igarashi, D. Inagake & Y. Okada, 2011. Changes in abundance of the neon flying squid Ommastrephes bartramii in relation to climate change in the central North Pacific Ocean. Marine Ecology Progress Series 441: 151–164.
Igarashi, H., T. Ichii, M. Sakai, Y. Ishikawa, T. Toyoda, S. Masuda, N. Sugiura, K. Mahapatra & T. Awaji, 2015. Possible link between interannual variation of neon flying squid (Ommastrephes bartramii) abundance in the North Pacific and the climate phase shift in 1998/1999. Progress in Oceanography. doi:10.1016/j.pocean.2015.03.008.
Kug, J.-S., F.-F. Jin & S.-I. An, 2009. Two types of El Niño events: cold tongue El Niño and warm pool El Niño. Journal of Climate 22(6): 1499–1515.
Latif, M. & N. S. Keenlyside, 2009. El Niño/Southern Oscillation response to global warming. Proceedings of the National Academy of Sciences of USA 106(49): 20578–20583.
Martins, R. S. & J. A. A. Perez, 2007. The ecology of loliginid squid in shallow waters around Santa Catarina Island, southern Brazil. Bulletin of Marine Science 80(1): 125–145.
McPhaden, M. J., 1999. Genesis and evolution of the 1997–98 El Niño. Science 283(5404): 950–954.
Mori, J., T. Kubodera & N. Baba, 2001. Squid in the diet of northern fur seals, Callorhinus ursinus, caught in the western and central North Pacific Ocean. Fisheries Research 52(1–2): 91–97.
Mugo, R. M., S.-I. Saitoh, F. Takahashi, A. Nihira & T. Kuroyama, 2014. Evaluating the role of fronts in habitat overlaps between cold and warm water species in the western North Pacific: a proof of concept. Deep Sea Research Part II: Topical Studies in Oceanography 107: 29–39.
Muhling, B. A., S.-K. Lee, J. T. Lamkin & Y. Liu, 2011. Predicting the effects of climate change on bluefin tuna (Thunnus thynnus) spawning habitat in the Gulf of Mexico. ICES Journal of Marine Science 68(6): 1051–1062.
Nishikawa, H., H. Igarashi, Y. Ishikawa, M. Sakai, Y. Kato, M. Ebina, N. Usui, M. Kamachi & T. Awaji, 2014. Impact of paralarvae and juveniles feeding environment on the neon flying squid (Ommastrephes bartramii) winter–spring cohort stock. Fisheries Oceanography 23(4): 289–303.
O’Dor, R. K., 1998. Can understanding squid life-history strategies and recruitment improve management? South African Journal of Marine Science 20(1): 193–206.
Pierce, G., V. Valavanis, A. Guerra, P. Jereb, L. Orsi-Relini, J. Bellido, I. Katara, U. Piatkowski, J. Pereira, E. Balguerias, I. Sobrino, E. Lefkaditou, J. Wang, M. Santurtun, P. Boyle, L. Hastie, C. MacLeod, J. Smith, M. Viana, A. González & A. Zuur, 2008. A review of cephalopod–environment interactions in European seas. In Valavanis, V. (ed.), Essential Fish Habitat Mapping in the Mediterranean. Developments in Hydrobiology, Vol. 203 (pp 49–70), Springer, Netherlands.
Polovina, J. J., E. Howell, D. R. Kobayashi & M. P. Seki, 2001. The transition zone chlorophyll front, a dynamic global feature defining migration and forage habitat for marine resources. Progress in Oceanography 49(1–4): 469–483.
R Core Team, 2015. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna.
Robinson, C. J., J. Gómez-Gutiérrez & D. A. S. de León, 2013. Jumbo squid (Dosidicus gigas) landings in the Gulf of California related to remotely sensed SST and concentrations of chlorophyll a (1998–2012). Fisheries Research 137: 97–103.
Rodhouse, P. G., 2001. Managing and forecasting squid fisheries in variable environments. Fisheries Research 54(1): 3–8.
Rosa, A. L., J. Yamamoto & Y. Sakurai, 2011. Effects of environmental variability on the spawning areas, catch, and recruitment of the Japanese common squid, Todarodes pacificus (Cephalopoda: Ommastrephidae), from the 1970s to the 2000s. ICES Journal of Marine Science: Journal du Conseil 68(6): 1114–1121.
Sakurai, Y., H. Kiyofuji, S. Saitoh, T. Goto & Y. Hiyama, 2000. Changes in inferred spawning areas of Todarodes pacificus (Cephalopoda: Ommastrephidae) due to changing environmental conditions. ICES Journal of Marine Science 57(1): 24–30.
Sasaoka, K., S.-I. Saitoh, I. Asanuma, K. Imai, M. Honda, Y. Nojiri & T. Saino, 2002. Temporal and spatial variability of chlorophyll-a in the western subarctic Pacific determined from satellite and ship observations from 1997 to 1999. Deep Sea Research Part II: Topical Studies in Oceanography 49(24–25): 5557–5576.
Seki, M. P., 1993. The role of neon flying squid, Ommastrephes bartramii, in the North Pacific pelagic food web. Bulletin of the International North Pacific Commission 53: 207–215.
Stuecker, M. F., A. Timmermann, F.-F. Jin, S. McGregor & H.-L. Ren, 2013. A combination mode of the annual cycle and the El Nino/Southern Oscillation. Nature Geoscience 6(7): 540–544.
Thiaw, M., D. Gascuel, D. Thiao, O. T. Thiaw & D. Jouffre, 2011. Analysing environmental and fishing effects on a short-lived species stock: the dynamics of the octopus Octopus vulgaris population in Senegalese waters. African Journal of Marine Science 33(2): 209–222.
Tian, Y., K. Nashida & H. Sakaji, 2013. Synchrony in the abundance trend of spear squid Loligo bleekeri in the Japan Sea and the Pacific Ocean with special reference to the latitudinal differences in response to the climate regime shift. ICES Journal of Marine Science. doi:10.1093/icesjms/fst015.
Usui, N., S. Ishizaki, Y. Fujii, H. Tsujino, T. Yasuda & M. Kamachi, 2006. Meteorological Research Institute multivariate ocean variational estimation (MOVE) system: some early results. Advances in Space Research 37(4): 806–822.
Vijai, D., M. Sakai, Y. Kamei & Y. Sakurai, 2014. Spawning pattern of the neon fling squid Ommastrephes bartramii (Cephalopoda: Oegopsida) around the Hawaiian Islands. Scientia Marina 78(4): 9.
Waluda, C. M. & P. G. Rodhouse, 2006. Remotely sensed mesoscale oceanography of the Central Eastern Pacific and recruitment variability in Dosidicus gigas. Marine Ecology Progress Series 310: 25–32.
Waluda, C., P. Rodhouse, G. Podestá, P. Trathan & G. Pierce, 2001. Surface oceanography of the inferred hatching grounds of Illex argentinus (Cephalopoda: Ommastrephidae) and influences on recruitment variability. Marine Biology 139(4): 671–679.
Wang, H.-J., R.-H. Zhang, J. Cole & F. Chavez, 1999. El Niño and the related phenomenon Southern Oscillation (ENSO): the largest signal in interannual climate variation. Proceedings of the National Academy of Sciences of USA 96(20): 11071–11072.
Watanabe, H., T. Kubodera, T. Ichii & S. Kawahara, 2004. Feeding habits of neon flying squid Ommastrephes bartramii in the transitional region of the central North Pacific. Marine Ecology Progress Series 266: 173–184.
Wessel, P., W. H. F. Smith, R. Scharroo, J. Luis & F. Wobbe, 2013. Generic mapping tools: improved version released. EOS, Transactions American Geophysical Union 94(45): 409–410.
Wood, S. N., 2011. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. Journal of the Royal Statistical Society: Series B (Statistical Methodology) 73(1): 3–36.
Yatsu, A., S. Midorikawa, T. Shimada & Y. Uozumi, 1997. Age and growth of the neon flying squid, Ommastrephes bartramii, in the North Pacific Ocean. Fisheries Research 29(3): 257–270.
Yatsu, A., N. Mochioka, K. Morishita & H. Toh, 1998. Strontium/calcium ratios in statoliths of the neon flying squid, Ommastrephes bartramii (Cephalopoda), in the North Pacific Ocean. Marine Biology 131(2): 275–282.
Yu, W., X. Chen, Q. Yi, Y. Chen & Y. Zhang, 2015. Variability of suitable habitat of western winter–spring cohort for neon flying squid in the northwest Pacific under anomalous environments. PLoS One 10(4): e0122997.
Acknowledgments
This work was supported by the Research on Climate Change Adaptation (RECCA) Project of the Grant-in-Aid from Japan’s Ministry of Education, Culture, Sports, Science and Technology (MEXT). The authors are also grateful to Aomori Prefectural Industrial Technology Research Center for providing the squid fishery data and NOAA coast watch for environmental data used in analyses. The authors also thank Cesar L. Villanoy for very helpful discussion on correlation and time series analyses. We are equally grateful to the referees for the constructive comments that significantly improved the contents of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Vasilis Valavanis
Rights and permissions
About this article
Cite this article
Alabia, I.D., Saitoh, SI., Hirawake, T. et al. Elucidating the potential squid habitat responses in the central North Pacific to the recent ENSO flavors. Hydrobiologia 772, 215–227 (2016). https://doi.org/10.1007/s10750-016-2662-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2662-5