Abstract
Marine sponges harbor dense microbial communities of exceptionally high diversity. Despite the complexity of sponge microbiota, microbial communities in different sponges seem to be remarkably similar. In this study, we used a subset of a previously established 454 amplicon pyrosequencing dataset (Schmitt and Taylor, unpublished data). Five Mediterranean sponges were chosen including the model sponge Aplysina aerophoba to determine the extent of uniformity by defining (i) the core microbial community, consisting of bacteria found in all sponges, (ii) the variable microbial community, consisting of bacteria found in 2–4 sponges, and (iii) the species-specific community, consisting of bacteria found in only one sponge. Using the enormous sequencing depth of pyrosequencing the diversity in each of the five sponges was extended to up to 15 different bacterial phyla per sponge with Proteobacteria and Chloroflexi being most diverse in each of the five sponges. Similarity comparison of bacteria on phylum and phylotype level revealed most similar communities in A. aerophoba and A. cavernicola and the most dissimilar community in Pseudocorticium jarrei. A surprising minimal core bacterial community was found when distribution of 97% operational taxonomic units (OTUs) was analyzed. Core, variable, and species-specific communities were comprised of 2, 26, and 72% of all OTUs, respectively. This indicates that each sponge contains a large set of unique bacteria and shares only few bacteria with other sponges. However, host species-specific bacteria are probably still closely related to each other explaining the observed similarity among bacterial communities in sponges.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The recent advent of next-generation sequencing technologies (Metzger, 2010) has led to exciting advances in the study of microorganisms in natural environments. Among the most significant has been the identification of a microbial “rare biosphere” (Sogin et al., 2006), which comprises a phylogenetically diverse set of organisms that are present only at very low abundances. An initial, 454 pyrosequencing-based survey of deep sea samples indicated that the resident bacterial communities were up to two orders of magnitude more complex than previously reported for any other environment (Sogin et al., 2006). While acknowledging the limitations of this technique, e.g., sequencing artifacts that led to inflation of diversity (Quince et al., 2009; Huse et al., 2010; Kunin et al., 2010) subsequent studies on plants and humans (Turnbaugh et al., 2009; Redford et al., 2010), soil (Roesch et al., 2007; Chou et al., 2010), and other marine habitats (Galand et al., 2009; Gilbert et al., 2009; Andersson et al., 2010) supported these initial conclusions.
Few habitats are better suited to exploration using next-generation sequencing approaches than the highly diverse microbial communities of marine sponges. Indeed, 454 pyrosequencing of 16S rRNA gene amplicons has already yielded much new information about microbes in Great Barrier Reef (Webster et al., 2010) and Red Sea (Lee et al., 2010) sponges. In the former study, more than 250,000 16S rRNA gene sequences were obtained from the Great Barrier Reef sponges Ianthella basta, Ircinia ramosa, and Rhopaloeides odorabile (Webster et al., 2010). A broad taxonomic range of bacteria were detected, including common sponge associates such as Acidobacteria, Actinobacteria, Chloroflexi, Proteobacteria, and Poribacteria as well as a number of taxa recorded from sponges for the first time (e.g., Deferribacteres, Tenericutes, candidate phylum WS3). Invariably, the “new” taxa were at low abundance, demonstrating the utility of pyrosequencing for uncovering the rare microbial biosphere of marine sponges. It is clear that this new technology offers much to the study of marine sponge microbiology.
In a previous, large-scale 454 amplicon pyrosequencing study (Schmitt and Taylor, unpublished data) we analyzed the diversity and community structure of the total sponge microbiota (obtained from 32 sponge species) as well as the global biogeography of sponge symbionts. Here we used a subset of the previous dataset to investigate in detail the diversity of bacteria associated with each of five Mediterranean sponges (Aplysina aerophoba, Aplysina cavernicola, Ircinia variabilis, Petrosia ficiformis, and Pseudocorticium jarrei). These sequencing data are also used to delineate the bacterial community into core, variable, and (host) species-specific subsets.
Materials and methods
Amplicon 454 pyrosequencing data were previously generated using a set of 32 sponge species (GenBank accession number SRP003545; Schmitt and Taylor, unpublished data). Briefly, high-quality DNA was extracted from three individuals of each species and a ca. 145 bp fragment of the 16S rRNA gene, including the hypervariable V3 region, was amplified using the modified primer pair 338f and 533r (338fdeg: ACW CCT ACG GGW GGC WGC AG, 533rdeg: TKA CCG CRG CTG CTG GCA C). Equal amounts of PCR products were pooled from all three individuals and amplicon libraries were sequenced with a 454 Life Sciences FLX pyrosequencer (University of Otago, Dunedin, New Zealand).
For this study, tag sequence data from the five Mediterranean sponges Aplysina aerophoba, Aplysina cavernicola, Ircinia variabilis, Petrosia ficiformis, and Pseudocorticium jarrei (collection: A. aerophoba: Croatia, 45°05′N, 13°38′E, all other sponges: France, 43°12′N, 5°21′E, all at a depth of 15 m) were extracted from the whole dataset and analyzed in greater detail. To avoid overestimating the true diversity due to erroneous tag reads as a result of sequencing errors and formation of homopolymers and chimeras (Reeder & Knight, 2009), the approach of Kunin et al. (2010) was followed: the tag sequences were end-trimmed based on quality scores at a stringency of 0.2% per base error probability using LUCY (Chou & Holmes, 2001). Unique sequences were identified with Mothur 1.9.0 (Schloss et al., 2009), aligned against a SILVA alignment (available at http://www.mothur.org/wiki/Alignment_database) using a kmer search and a Needleman algorithm, and then grouped into 97% OTUs based on uncorrected pairwise distance matrices with the furthest neighbor algorithm. A representative sequence (defined as implemented in Mothur) of each OTU was used for the taxonomic assignment using customized perl scripts similar to the approach used by Sogin et al. (2006) and Webster et al. (2010). For each tag sequence, a BLAST search (Altschul et al., 1990) was performed against a manually modified SILVA version 98 database. Pairwise global alignments were performed between each of the 10 best hits against the tag sequence using a Smith–Waterman algorithm. The most similar sequence to the tag sequence (or multiple sequences if within a range of 0.1% sequence divergence) was then used for assignment according to the RDP taxonomy implemented in the SILVA database. For assignment at phylum, class, order, family, and genus level, sequence similarity thresholds of 75, 80, 85, 90, and 95% were applied. In cases where the taxonomy of the most similar sequences was inconsistent, a majority rule was applied, and the tag sequence was only assigned if at least 60% of all reference sequences shared the same taxonomic annotation at the respective taxonomic level. All previously published, sponge-derived sequences in the SILVA reference database were labeled as such and it was noted when a tag sequence was assigned to a sponge-derived sequence. Based on the taxonomic assignment as well as on the presence/absence of each OTU in the sponges, Bray-Curtis similarities were calculated using the program PRIMER-6 (PRIMER-E, UK) and visualized as heatmaps using JColorGrid (Joachimiak et al., 2006). Bray-Curtis similarities were also used for unweighted pair-group average cluster analysis with PRIMER-6.
Results
Phylogenetic affiliation of OTUs
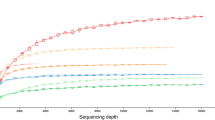
Overall, 831, 908, 709, 595, and 255 high-quality tag sequences and 133, 119, 111, 142, and 44 different OTUs were obtained from the sponges A. aerophoba, A. cavernicola, I. variabilis, P. ficiformis, and P. jarrei, respectively (Fig. 1). These OTUs were affiliated with 15 described bacterial phyla, 5 candidate phyla, and an unclassified bacterial lineage that was previously termed SAUL (sponge-associated unidentified lineage, Schmitt and Taylor, unpublished data) (Fig. 1, Table S1). A. aerophoba showed the highest phylum level diversity with 15 different bacterial phyla whereas P. jarrei revealed the lowest diversity with 8 bacterial phyla. The remaining three sponges all contained OTUs affiliated with 13 or 14 bacterial phyla. Acidobacteria, Actinobacteria, Chloroflexi, Nitrospira, Proteobacteria, and Spirochaetes as well as the candidate phylum Poribacteria were present in all five investigated sponges. In contrast, Chlamydiae, Lentisphaerae, and Planctomycetes, as well as the candidate phyla TM6 and TM7, were only found in one of the five sponge species. Most diverse in this dataset were the Chloroflexi and Proteobacteria with up to 53 and 45 different OTUs per host species, respectively. Acidobacteria, Actinobacteria, and Poribacteria were represented by an average of 11, 6, and 9.4 OTUs. All other phyla were less diverse with 7 or less different OTUs.
Bacterial community similarity among the five sponges
At phylum level, analysis of Bray-Curtis similarity values showed a high similarity (>75%) for bacterial communities present in A. aerophoba, A. cavernicola, P. ficiformis, and I. variabilis with the two Aplysina sponges having most similar bacterial communities (Fig. 2A). The bacterial community in P. jarrei was more dissimilar to the other sponges with a Bray-Curtis value of 60%. At phylotype level, the same similarity pattern was obtained with both Aplysina sponges having most similar bacterial communities and P. jarrei having the most dissimilar bacterial community, though with overall lower similarity values (Fig. 2B).
Phylum level (A) and phylotype level (B) similarity of bacterial communities among the five sponges. Heat map illustrating Bray-Curtis similarity values and cluster analysis of assigned OTUs on phylum level (A) as well as presence/absence of each OTU in sponges (B) are shown. Sponges are abbreviated in the order as shown on the left side
Distribution of OTUs within the five sponges
The bacterial communities of the 5 sponges were divided into the following three categories: (i) core community, represented by OTUs that are present in all 5 sponge species; (ii) variable community, represented by OTUs that are present in 2–4 sponge species; (iii) species-specific community, represented by OTUs that are present in a single sponge species (Fig. 3). The core community consisted of 2% of all OTUs and contained representatives of five different bacterial phyla. The variable community comprised 26% of all OTUs and included 11 different bacterial phyla and the SAUL lineage. The species-specific community was biggest with 72% of all OTUs and represented 18 bacterial phyla and the SAUL lineage. Core, variable, and species-specific OTUs were further divided into “Plus-OTUs” and “Minus-OTUs” depending on whether the respective tag sequences were assigned to a previously sponge-derived 16S rRNA gene sequence (Plus-OTU) or to a non-sponge-derived 16S rRNA gene sequence (Minus-OTU) during the taxonomic assignment. 89% of core OTUs were Plus-OTUs. The variable community comprised 75% Plus-OTUs whereas the species-specific community contained 69% Plus-OTUs.
Number of OTUs that comprise species-specific, variable, and core bacterial communities defined as presence of OTUs in 1 sponge, 2–4 sponges, and all 5 sponges, respectively. Phylogenetic composition and number of different OTUs per phylum is given for each of the three communities. Species-specific, variable and core OTUs are further divided into Plus- or Minus-OTUs depending on whether a previously sponge-derived 16S rRNA gene sequence was used for taxonomic assignment or a non-sponge-derived sequence
Discussion
Taxonomic richness of sponge microbiota
Aplysina aerophoba is a model sponge for the investigation of sponge-microbe associations. Studies on the morphological diversity of bacteria associated with A. aerophoba reach back to 1970s (Vacelet, 1975) and were later complemented by cultivation approaches (Hentschel et al., 2001; Pimentel-Elardo et al., 2003) and molecular studies on microbial diversity (Hentschel et al., 2002; Fieseler et al., 2004). Subsequently, different aspects of the association of A. aerophoba with diverse microbes were analyzed in greater detail such as the stability of the association (Friedrich et al., 2001), physiology of microbial symbionts (Bayer et al., 2008; Schlappy et al., 2010), disturbance of the association during sponge disease (Webster et al., 2008), and secondary metabolites (Ahn et al., 2003; Siegl & Hentschel, 2010).
In this study, a 454 amplicon pyrosequencing dataset obtained from A. aerophoba was re-analyzed together with tag sequence data from four other Mediterranean sponges (Fig. 1). From the 15 bacterial phyla found in A. aerophoba, 10 were discovered previously from this sponge (Hentschel et al., 2002; Fieseler et al., 2004), four were not known from this species until now while members of the Firmicutes were only detected in A. aerophoba by cultivation approaches (Hentschel et al., 2001; Pabel et al., 2003). The SAUL lineage was found in several other sponges before (Taylor et al., 2007; Kamke et al., 2010) but was not detected in A. aerophoba so far. The phylogenetic affiliation of the SAUL lineage is still unclear although previous phylogenetic analysis placed it into the Planctomycetes–Verrucomicrobia–Chlamydiae (PVC) superphylum (Wagner & Horn, 2006). Among the four newly found phyla are Deinococcus-Thermus and the candidate phylum TM7 which were also previously found in other sponges albeit always at low diversity (Schirmer et al., 2005; Thiel et al., 2007; Schmitt et al., 2008; Webster et al., 2010). In addition, two new candidate phyla, OS-K and OP10, were detected. These data show the great potential of amplicon pyrosequencing to discover less abundant members of bacterial communities that may be missed with conventional molecular methods, and therefore to extend our knowledge of bacterial diversity.
Similar to the results for A. aerophoba, some hitherto undetected phyla were also found within the other four Mediterranean sponges (Fig. 1). The microbial diversity within A. cavernicola was also studied before by molecular methods although not as extensively as in A. aerophoba (Friedrich et al., 1999; Thoms et al., 2003; Lafi et al., 2009). In this study, 13 different bacterial phyla were detected in A. cavernicola including two candidate phyla (Poribacteria, OP10), as well as the unclassified SAUL lineage. Particularly interesting was the finding of a Chlamydiae-affiliated OTU as this phylum has only been reported once from sponges investigated by conventional molecular methods (Zhu et al., 2008) but was found in a recent 454 amplicon pyrosequencing study in two more sponges (Webster et al., 2010). This might indicate that members of the Chlamydiae could actually be widespread among sponges but likely belong to the rare biosphere in many sponges and were therefore overlooked in most previous studies. Only few 16S rRNA gene sequence data are available for I. variabilis, either from cultivated bacteria (De Rosa et al., 2003) or from cyanobacteria-specific studies (Usher et al., 2004). Here, members of 14 different bacterial phyla were found including two candidate phyla (Poribacteria, OS-K) as well as Planctomycetes and Lentisphaerae. Although the latter two phyla have been found before in sponges (Taylor et al., 2007; Webster et al., 2010) they do not seem to be very diverse in these hosts. The respective 16S rRNA gene sequences recovered in this study do not match with other sponge-derived but with environmental 16S rRNA gene sequences and might therefore represent contaminants from seawater. The association of P. ficiformis with microbes has long been studied (Vacelet & Donadey, 1977; Wilkinson & Vacelet, 1979), however, many of the 16S rRNA gene sequences from this sponge available in public databases such as GenBank (http://www.ncbi.nlm.nih.gov) were derived from cultivation-based studies (Chelossi et al., 2007; Muscholl-Silberhorn et al., 2008). In this study, P. ficiformis contained members of 13 different bacterial phyla including two candidate phyla (Poribacteria, OP10) and the SAUL lineage. Two OTUs found in P. ficiformis were affiliated with the phylum Verrucomicrobia, a phylum that is only irregularly found in sponges. Interestingly, these two OTUs were identified as Plus-OTUs meaning their most similar sequences in the database were also retrieved from sponges. This also might indicate a closer relationship than previously thought. Finally, the microbiology of P. jarrei, a representative of the group Homoscleromorpha, is completely unknown so far. Here, we found eight different bacterial phyla including the candidate phyla Poribacteria and TM6, as well as the SAUL lineage.
Similarity among Mediterranean sponge-associated bacteria
The four sponges A. aerophoba, A. cavernicola, I. variabilis, and P. ficiformis exhibit more than 75% overlap between their microbial communities at phylum level (Fig. 2A), with 8 phyla being present in all four of these sponges (Fig. 1). With the exception of Firmicutes, all of these phyla are known to contain sponge-specific clusters and represent the typical bacterial profile of bacteriosponges (Hentschel et al., 2002; Taylor et al., 2007). Most diverse in all sponges are the phyla Proteobacteria and Chloroflexi, which is in agreement with previous data obtained from 16S rRNA gene clone libraries (Mohamed et al., 2008; Lee et al., 2009; Kamke et al., 2010). The similarity pattern on phylum level where both Aplysina sponges have most similar bacterial communities followed by P. ficiformis and I. variabilis was supported by the comparison of bacterial communities on phylotype level (Fig. 2B). The bacterial community in P. jarrei seems to differ more on both phylum level, with only 60% similarity, and phylotype level, with 20% similarity (Fig. 2). This might indicate a different bacterial profile in P. jarrei, but more likely reflects the much lower number of analyzed OTUs due to fewer 454 reads. It is therefore conceivable that the more abundant bacteria were detected in P. jarrei whereas the rare bacteria are underrepresented in the dataset which then has an effect on the comparison analysis.
Community structure of complex sponge microbiota
Core (presence in all sponges), variable (presence in 2–4 sponges), and species-specific (presence in only one sponge) communities were defined by determining the presence of each OTU in every sponge species (Fig. 3). The core community was rather small with only 2% of all OTUs found in all sponges. In stark contrast, the species-specific community comprised almost three quarters of all OTUs. Core, variable, and species-specific communities are phylogenetically diverse and not dominated by a single phylum (Fig. 3). Assuming that 97% sequence similarity is an approximate threshold for bacterial species, then these data suggest that sponges do not share a lot of their bacterial species. Instead each sponge species contains a large set of unique bacterial species. This seemingly contrasts with current understanding of microbiota in sponges that are believed to be highly similar (Hentschel et al., 2002; Olson & McCarthy, 2005; Hill et al., 2006; Taylor et al., 2007; Lee et al., 2009; Anderson et al., 2010). The hypothesis of a uniform bacterial community in sponges is mainly based on the finding of sponge-specific clusters, e.g., clusters of only sponge-derived 16S rRNA gene sequences from different sponge species and/or locations (Hentschel et al., 2002). However, these clusters are not limited by a sequence similarity threshold and, in fact, within-cluster similarity can be as low as 77% (Hentschel et al., 2002). Because of the short length of our tag sequences (ca. 145 bp) we could not determine sponge-specific clusters in our dataset. Instead, we used a similar designation into Plus- and Minus-OTUs depending on whether the respective 16S rRNA gene sequence was assigned to a sponge-derived (Plus-OTU) or a non-sponge-derived (Minus-OTU) 16S rRNA gene sequence in the database. Interestingly, we found a high percentage of Plus-OTUs ranging from 69% in the species-specific community to 89% in the core community. We therefore propose that different sponges might contain different bacterial species. But these species are still more closely related to each other than to, e.g., seawater bacteria as indicated by the high percentage of Plus-OTUs in our dataset and generally by sponge-specific clusters.
Symbiont transmission model revisited
In a previous study we proposed a symbiont transmission model including a combination of both vertical and horizontal transmission to maintain complex microbial consortia in sponges (Schmitt et al., 2008). Here, we provide an update including the current literature on the mechanism of formation of this specific association (Fig. 4). There are now several microscopic and molecular studies showing that bacterial symbionts in sponges are vertically transmitted through reproductive stages (Usher et al., 2001; Ereskovsky et al., 2005; Oren et al., 2005; Enticknap et al., 2006; DeCaralt et al., 2007; Maldonado, 2007; Schmitt et al., 2007a, b; Sharp et al., 2007; Steger et al., 2008; Lee et al., 2009; Webster et al., 2010). The alternative mechanism of horizontal or environmental transmission, e.g., uptake of symbionts from seawater, is much more difficult to prove and so far there is only indirect evidence for it. For P. ficiformis, a species that was also used in this study, the gametes do not contain any bacterial cells and it was therefore concluded that bacteria must be acquired from the environment by juveniles of each new generation (Maldonado & Riesgo, 2009). A similar argument was used for cyanobacterial cells in A. aerophoba and Chondrilla australiensis (Usher, 2008; Maldonado, 2009). Both species contain cyanobacteria in the mesohyl of adult sponges but these bacteria could not be detected at all in oocytes of A. aerophoba (Maldonado, 2009) and only in some gametes of C. australiensis (Usher, 2008). Strictly vertically transmitted bacterial symbionts co-speciate with their hosts which results in congruent phylogenies (Zientz et al., 2004). Such a co-speciation signal was not found in a comprehensive phylogenetic study of vertically transmitted symbionts in sponges and it was assumed that additional horizontal symbiont transmission might obscure a co-speciation pattern (Schmitt et al., 2008). Finally, a recent 454 amplicon pyrosequencing study detected sponge-specific microbes in seawater albeit at very low abundances (Webster et al., 2010). The authors speculated that sponge-specific microbes might in fact be present in seawater as part of the rare seawater biosphere which might serve as a seed bank for colonization of sponges. In this case, sponges might harvest these microbial lineages from seawater in addition to vertical transmission of symbionts. Altogether, these new results corroborate the theory that horizontal/environmental transmission of sponge symbionts occurs and that a combination of horizontal and environmental transmission maintains complex sponge microbes associations.
Update of the previously proposed microbial symbiont transmission model in sponge hosts hypothesizing a combination of vertical and horizontal transmission to maintain complex sponge-microbe associations. Figure amended, with permission, from Schmitt et al. (2008)
Conclusions
This study complements a previous large-scale study on whole sponge microbiome diversity and global sponge symbiont biogeography (Schmitt and Taylor, unpublished data) on a small-scale level including microbial diversity and community structure on a sponge species level. For this study the tag dataset from five Mediterranean sponges was chosen including the model sponge Aplysina aerophoba. Deep sequencing of these sponges revealed a previously unknown diversity including several phyla so far not known from these sponges. The sponge microbiome consists of very few core bacteria found in all five sponges and a majority of host species-specific bacteria found in only one sponge. However, these host species-specific bacteria are probably still closely related to each other explaining the observed similarity among bacterial communities in sponges. These important new results allow us to better assess and understand one of the most diverse and possibly most ancient symbiotic systems.
References
Ahn, Y. B., S. K. Rhee, D. E. Fennell, L. J. Kerkhof, U. Hentschel & M. M. Haggblom, 2003. Reductive dehalogenation of brominated phenolic compounds by microorganisms associated with the marine sponge Aplysina aerophoba. Applied and Environmental Microbiology 69: 4159–4166.
Altschul, S. F., W. Gish, W. Miller, E. W. Myers & D. J. Lipman, 1990. Basic local alignment search tool. Journal of Molecular Biology 215: 403–410.
Anderson, S. A., P. T. Northcote & M. J. Page, 2010. Spatial and temporal variability of the bacterial community in different chemotypes of the New Zealand marine sponge Mycale hentscheli. FEMS Microbiology Ecology 72: 328–342.
Andersson, A. F., L. Riemann & S. Bertilsson, 2010. Pyrosequencing reveals contrasting seasonal dynamics of taxa within Baltic Sea bacterioplankton communities. The ISME Journal 4: 171–181.
Bayer, K., S. Schmitt & U. Hentschel, 2008. Physiology, phylogeny and in situ evidence for bacterial and archaeal nitrifiers in the marine sponge Aplysina aerophoba. Environmental Microbiology 10: 2942–2955.
Chelossi, E., R. Pantile, R. Pronzato, M. Milanese & U. Hentschel, 2007. Bacteria with antimicrobial properties isolated from the Mediterranean sponges Chondrilla nucula and Petrosia ficiformis. Aquatic Microbial Ecology 49: 157–163.
Chou, H. H. & M. H. Holmes, 2001. DNA sequence quality trimming and vector removal. Bioinformatics 17: 1093–1104.
Chou, H. Y., N. Fierer, C. L. Lauber, J. G. Caporaso, R. Knight & P. Grogan, 2010. Soil bacterial diversity in the Arctic is not fundamentally different from that found in other biomes. Environmental Microbiology 12: 2998–3006.
De Rosa, S., M. Mitova & G. Tommonaro, 2003. Marine bacteria associated with sponge as source of cyclic peptides. Biomolecular Engineering 20: 311–316.
DeCaralt, S., M. J. Uriz & R. H. Wijffels, 2007. Vertical transmission and successive location of symbiotic bacteria during embryo development and larva formation in Corticium candelabrum (Porifera: Demospongiae). Journal of the Marine Biological Association of the United Kingdom 87: 1693–1699.
Enticknap, J. J., M. Kelly, O. Peraud & R. T. Hill, 2006. Characterization of a culturable alphaproteobacterial symbiont common to many marine sponges and evidence for vertical transmission via sponge larvae. Applied and Environmental Microbiology 72: 3724–3732.
Ereskovsky, A. V., E. Gonobobleva & A. Vishnyakov, 2005. Morphological evidence for vertical transmission of symbiotic bacteria in the viviparous sponge Halisarca dujardini Johnston (Porifera, Demospongiae, Halisarcida). Marine Biology 146: 869–875.
Fieseler, L., M. Horn, M. Wagner & U. Hentschel, 2004. Discovery of the novel candidate phylum “Poribacteria” in marine sponges. Applied and Environmental Microbiology 70: 3724–3732.
Friedrich, A. B., H. Merkert, T. Fendert, J. Hacker, P. Proksch & U. Hentschel, 1999. Microbial diversity in the marine sponge Aplysina cavernicola (formerly Verongia cavernicola) analyzed by fluorescence in situ hybridization (FISH). Marine Biology 134: 461–470.
Friedrich, A. B., I. Fischer, P. Proksch, J. Hacker & U. Hentschel, 2001. Temporal variation of the microbial community associated with the mediterranean sponge Aplysina aerophoba. FEMS Microbiology Ecology 38: 105–113.
Galand, P. E., E. O. Casamayor, D. L. Kirchman & C. Lovejoy, 2009. Ecology of the rare microbial biosphere of the Arctic Ocean. Proceedings of the National Academy of Sciences of the United States of America 106: 22427–22432.
Gilbert, J. A., D. Field, P. Swift, L. Newbold, A. Oliver, T. Smyth, P. J. Somerfield, S. Huse & I. Joint, 2009. The seasonal structure of microbial communities in the Western English Channel. Environmental Microbiology 11: 3132–3139.
Hentschel, U., M. Schmid, M. Wagner, L. Fieseler, C. Gernert & J. Hacker, 2001. Isolation and phylogenetic analysis of bacteria with antimicrobial activities from the Mediterranean sponges Aplysina aerophoba and Aplysina cavernicola. FEMS Microbiology Ecology 35: 305–312.
Hentschel, U., J. Hopke, M. Horn, A. B. Friedrich, M. Wagner, J. Hacker & B. S. Moore, 2002. Molecular evidence for a uniform microbial community in sponges from different oceans. Applied and Environmental Microbiology 68: 4431–4440.
Hill, M., A. Hill, N. Lopez & O. Harriott, 2006. Sponge-specific bacterial symbionts in the Caribbean sponge, Chondrilla nucula (Demospongiae, Chondrosida). Marine Biology 148: 1221–1230.
Huse, S., D. B. M. Welch, H. G. Morrison & M. L. Sogin, 2010. Ironing out the wrinkles in the rare biosphere through improved OTU clustering. Environmental Microbiology 12: 1889–1898.
Joachimiak, M. P., J. L. Weisman & B. C. H. May, 2006. JColorGrid: software for the visualization of biological measurement. BMC Bioinformatics 27: 7–225.
Kamke, J., M. W. Taylor & S. Schmitt, 2010. Activity profiles for marine sponge-associated bacteria obtained by 16S rRNA vs 16S rRNA gene comparisons. The ISME Journal 4: 498–508.
Kunin, V., A. Engelbrektson, H. Ochman & P. Hugenholtz, 2010. Wrinkles in the rare biosphere: pyrosequencing errors can lead to artificial inflation of diversity estimates. Environmental Microbiology 12: 118–123.
Lafi, F. F., J. A. Fuerst, L. Fieseler, C. Engels, W. W. L. Goh & U. Hentschel, 2009. Widespread distribution of Poribacteria in Demospongiae. Applied and Environmental Microbiology 75: 5695–5699.
Lee, O. O., P. Y. Chui, Y. H. Wong, J. R. Pawlik & P. Y. Qian, 2009. Evidence for vertical transmission of bacterial symbionts from adult to embryo in the Caribbean sponge Svenzea zeai. Applied and Environmental Microbiology 75: 6147–6156.
Lee, O. O., Y. Wang, J. Yang, F. F. Lafi, A. Al-Suwailem & P.-Y. Qian, 2010. Pyrosequencing reveals highly diverse and species-specific microbial communities in sponges from the Red Sea. The ISME Journal, epub ahead of print, doi:10.1038/ismej.2010.165.
Maldonado, M., 2007. Intergenerational transmission of symbiotic bacteria in oviparous and viviparous demosponges, with emphasis on intracytoplasmically-compartmented bacterial types. Journal of the Marine Biological Association of the United Kingdom 87: 1701–1713.
Maldonado, M., 2009. Embryonic development of verongid demosponges supports the independent acquisition of spongin skeletons as an alternative to the siliceous skeleton of sponges. Biological Journal of the Linnean Society 97: 427–447.
Maldonado, M. & A. Riesgo, 2009. Gametogenesis, embryogenesis, and larval features of the oviparous sponge Petrosia ficiformis (Haplosclerida, Demospongiae). Marine Biology 156: 2181–2197.
Metzger, M. L., 2010. Sequencing technologies—the next generation. Nature Reviews Genetics 11: 31–46.
Mohamed, N. M., V. Rao, M. T. Hamann, M. Kelly & R. T. Hill, 2008. Monitoring bacterial diversity of the marine sponge Ircinia strobilina upon transfer into aquaculture. Applied and Environmental Microbiology 74: 4133–4143.
Muscholl-Silberhorn, A., V. Thiel & J. F. Imhoff, 2008. Abundance and bioactivity of cultured sponge-associated bacteria from the Mediterranean sea. Microbial Ecology 55: 94–106.
Olson, J. B. & P. J. McCarthy, 2005. Associated bacterial communities of two deep-water sponges. Aquatic Microbial Ecology 39: 47–55.
Oren, M., L. Steindler & M. Ilan, 2005. Transmission, plasticity and the molecular identification of cyanobacterial symbionts in the Red Sea sponge Diacarnus erythraenus. Marine Biology 148: 35–41.
Pabel, C. T., J. Vater, C. Wilde, P. Franke, J. Hofemeister, B. Adler, G. Bringmann, J. Hacker & U. Hentschel, 2003. Antimicrobial activities and matrix-assisted laser desorption/ionization mass spectrometry of Bacillus isolates from the marine sponge Aplysina aerophoba. Marine Biotechnology 5: 424–434.
Pimentel-Elardo, S., M. Wehrl, A. B. Friedrich, P. R. Jensen & U. Hentschel, 2003. Isolation of planctomycetes from Aplysina sponges. Aquatic Microbial Ecology 33: 239–245.
Quince, C., A. Lanzen, T. P. Curtis, R. J. Davenport, N. Hall, I. M. Head, L. F. Read & W. T. Sloan, 2009. Accurate determination of microbial diversity from 454 pyrosequencing data. Nature Methods 6: 639–641.
Redford, A. J., R. M. Bowers, R. Knight, Y. Linhart & N. Fierer, 2010. The ecology of the phyllosphere: geographic and phylogenetic variability in the distribution of bacteria on tree leaves. Environmental Microbiology 12: 2885–2893.
Reeder, J. & R. Knight, 2009. The ‘rare biosphere’: a reality check. Nature Methods 6: 636–637.
Roesch, L. F., R. R. Fulthorpe, A. Riva, G. Casella, A. K. M. Hadwin, A. D. Kent, S. H. Daroub, F. A. O. Camargo, W. G. Farmerie & E. W. Triplett, 2007. Pyrosequencing enumerates and contrasts soil microbial diversity. The ISME Journal 1: 283–290.
Schirmer, A., R. Gadkari, C. D. Reeves, F. Ibrahim, E. F. DeLong & C. R. Hutchinson, 2005. Metagenomic analysis reveals diverse polyketide synthase gene clusters in microorganisms associated with the marine sponge Discodermia dissoluta. Applied and Environmental Microbiology 71: 4840–4849.
Schlappy, M. L., S. I. Schottner, G. Lavik, M. M. M. Kuypers, D. de Beer & F. Hoffmann, 2010. Evidence of nitrification and denitrification in high and low microbial abundance sponges. Marine Biology 157: 593–602.
Schloss, P. D., S. L. Westcott, T. Ryabin, J. R. Hall, M. Hartmann, E. B. Hollister, R. A. Lesniewski, B. B. Oakley, D. H. Parks, C. J. Robinson, J. W. Sahl, B. Stres, G. G. Thallinger, D. J. Van Horn & C. F. Weber, 2009. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Applied and Environmental Microbiology 75: 7537–7541.
Schmitt, S., M. Wehrl, N. Lindquist, J. B. Weisz & U. Hentschel, 2007a. Morphological and molecular analyses of microorganisms in Caribbean reef adult sponges and in corresponding reproductive material. In Porifera Research: Biodiversity, Innovation & Sustainability. Rio de Janeiro Museu Nacional, Buzios, Brazil: 561–568.
Schmitt, S., J. B. Weisz, N. Lindquist & U. Hentschel, 2007b. Vertical transmission of a phylogenetically complex microbial consortium in the viviparous sponge Ircinia felix. Applied and Environmental Microbiology 73: 2067–2078.
Schmitt, S., H. Angermeier, R. Schiller, N. Lindquist & U. Hentschel, 2008. Molecular microbial diversity survey of sponge reproductive stages and mechanistic insights into vertical transmission of microbial symbionts. Applied and Environmental Microbiology 74: 7694–7708.
Sharp, K. H., B. Eam, D. J. Faulkner & M. G. Haygood, 2007. Vertical transmission of diverse microbes in the tropical sponge Corticium sp. Applied and Environmental Microbiology 73: 622–629.
Siegl, A. & U. Hentschel, 2010. PKS and NRPS gene clusters from microbial symbiont cells of marine sponges by whole genome amplification. Environmental Microbiology Reports 2: 507–513.
Sogin, M. L., H. G. Morrison, J. A. Huber, D. Mark Welch, S. M. Huse, P. R. Neal, J. M. Arrieta & G. J. Herndl, 2006. Microbial diversity in the deep sea and the underexplored “rare biosphere”. Proceedings of the National Academy of Sciences of the United States of America 103: 12115–12120.
Steger, D., P. Ettinger-Epstein, S. Whalan, U. Hentschel, R. de Nys, M. Wagner & M. W. Taylor, 2008. Diversity and mode of transmission of ammonia-oxidizing archaea in marine sponges. Environmental Microbiology 10: 1087–1094.
Taylor, M. W., R. Radax, D. Steger & M. Wagner, 2007. Sponge-associated microorganisms: evolution, ecology, and biotechnological potential. Microbiology and Molecular Biology Reviews 71: 295–347.
Thiel, V., S. Leininger, R. Schmaljohann, F. Brummer & J. F. Imhoff, 2007. Sponge-specific bacterial associations of the Mediterranean sponge Chondrilla nucula (Demospongiae, Tetractinomorpha). Microbial Ecology 54: 101–111.
Thoms, C., M. Horn, M. Wagner, U. Hentschel & P. Proksch, 2003. Monitoring microbial diversity and natural product profiles of the sponge Aplysina cavernicola following transplantation. Marine Biology 142: 685–692.
Turnbaugh, P. J., M. Hamady, T. Yatsunenko, B. L. Cantarel, A. Duncan, R. E. Ley, M. L. Sogin, W. J. Jones, B. A. Roe, J. P. Affourtit, M. Egholm, B. Henrissat, A. C. Heath, R. Knight & J. I. Gordon, 2009. A core gut microbiome in obese and lean twins. Nature 457: 480–487.
Usher, K. M., 2008. The ecology and phylogeny of cyanobacterial symbionts in sponges. Marine Ecology and Evolutionary Perspective 29: 178–192.
Usher, K. M., J. Kuo, J. Fromont & D. C. Sutton, 2001. Vertical transmission of cyanobacterial symbionts in the marine sponge Chondrilla australiensis (Demospongiae). Hydrobiologia 461: 15–23.
Usher, K. M., J. Fromont, D. C. Sutton & S. Toze, 2004. The biogeography and phylogeny of unicellular cyanobacterial symbionts in sponges from Australia and the Mediterranean. Microbial Ecology 48: 167–177.
Vacelet, J., 1975. Etude en microscopie electronique de l’association entre bacteries et spongiaires du genre Verongia (Dictyoceratida). Journal De Microscopie 23: 271–288.
Vacelet, J. & C. Donadey, 1977. Electron-microscope study of association between some sponges and bacteria. Journal of Experimental Marine Biology and Ecology 30: 301–314.
Wagner, M. & M. Horn, 2006. The Planctomycetes, Verrucomicrobia, Chlamydiae and sister phyla comprise a superphylum with biotechnological and medical relevance. Current Opinion in Biotechnology 17: 241–249.
Webster, N. S., J. R. Xavier, M. Freckelton, C. A. Motti & R. Cobb, 2008. Shifts in microbial and chemical patterns within the marine sponge Aplysina aerophoba during a disease outbreak. Environmental Microbiology 10: 3366–3376.
Webster, N. S., M. W. Taylor, F. Behnam, S. Luecker, T. Rattei, S. Whalan, M. Horn & M. Wagner, 2010. Deep sequencing reveals exceptional diversity and modes of transmission for bacterial sponge symbionts. Environmental Microbiology 12: 2070–2082.
Wilkinson, C. R. & J. Vacelet, 1979. Transplantation of marine sponges to different conditions of light and current. Journal of Experimental Marine Biology and Ecology 37: 91–104.
Zhu, P., Q. Z. Li & G. Y. Wang, 2008. Unique microbial signatures of the alien Hawaiian marine sponge Suberites zeteki. Microbial Ecology 55: 406–414.
Zientz, E., T. Dandekar & R. Gross, 2004. Metabolic interdependence of obligate intracellular bacteria and their insect hosts. Microbiology and Molecular Biology Reviews 68: 745–770.
Acknowledgments
Research was supported by German Research Foundation (DFG) grants Schm2559/1-1 and 2-1 and a stipend in the program “Chancengleichheit” of the University of Wuerzburg to SS, DFG grant HE3299/1-3 to UH and University of Auckland FRDF grants 3609286 and 3622989 to MWT.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: M. Maldonado, X. Turon, M. A. Becerro & M. J. Uriz / Ancient animals, new challenges: developments in sponge research
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schmitt, S., Hentschel, U. & Taylor, M.W. Deep sequencing reveals diversity and community structure of complex microbiota in five Mediterranean sponges. Hydrobiologia 687, 341–351 (2012). https://doi.org/10.1007/s10750-011-0799-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-011-0799-9