Abstract
The purpose of this study was to verify the longitudinal distribution of phytoplankton biomass in two subtropical Brazilian reservoirs in the State of Paraná and investigate intervening factors on changes in phytoplankton biomass according to functional groups. In the Capivari and Segredo reservoirs, samples were obtained every 3 months during 2002, along a longitudinal axis (fluvial, transition, and lacustrine zones) at different depths. One hundred and eighteen taxa were identified, with Chlorophyceae as the most specious group. During the study period, both reservoirs had mostly low biomass values (less than 1 mm3 l−1). The short retention time of these reservoirs constituted the principal limiting factor to phytoplankton development. Biomass values above 1 mm3 l−1 were observed in the Capivari fluvial zone in March and in the Segredo lacustrine zone in December, with dominance by Microcystis aeruginosa Kütz (LM) and Anabaena circinalis Rab. (H1), respectively. Vertical and horizontal gradients of analyzed abiotic variables and phytoplankton biomass were observed. Considering the phytoplankton biomass values, both reservoirs were oligotrophic for the duration of the study. The Canonical Correspondence Analysis (CCA) evidenced temporal and spatial gradients of phytoplankton biomass; nevertheless, it did not follow the classic model proposed for deep reservoirs, since higher biomass was registered in the lacustrine zone during some months and in fluvial zones during other months. Distinct functional groups of phytoplankton characterized both studied reservoirs. Capivari Reservoir was best characterized by LM and Y groups, indicative of its greater water column stability and higher phosphorus concentration, whereas Segredo Reservoir was principally characterized by the MP functional group, indicative of its greater mixing zone extension and higher nitrate concentration. The obtained results also evidenced the influence of morphometric conditions and watershed purposes as important structuring factors of phytoplankton biomass in these reservoirs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Longitudinal changes on reservoir morphology and current flow result in different mixing zone depths and alterations in light availability and nutrient concentrations, which promote the occurrence of vertical and longitudinal gradients of abiotic variables and phytoplankton in these systems (Kimmel et al., 1990). In general, phytoplankton from deep reservoirs present a longitudinal distribution pattern with greater concentrations in the transition zone of the reservoirs. In the fluvial zone, there is light limitation despite nutrient availability, and afterwards, the relative fertility of the mixing zone decreases towards the dam because nutrient supply, introduced by advective processes, reduces with distance from the river inflow. Therefore, phytoplankton production becomes more dependent on in situ nutrient regeneration (Thornton et al., 1990; Armengol et al., 1999; Tundisi et al., 1999).
The phytoplankton in reservoirs is also influenced by regional climate, hydrological and geomorphological regime, size and watershed purposes, trophic chain structure, water retention time, and pulses produced as level variations resulting from reservoir operations (Tundisi et al., 1999).
Currently, the maintenance of sustainability of reservoirs for multiple purposes is a challenge, mainly due to the increased demand for potable water and conservation of ecosystems and their respective watersheds. However, changes in natural river systems resulting from reservoir construction have intensified problems related to water quality, such as the increase in harmful algae blooms (Calijuri et al., 2002). Predicting and mitigating the deterioration of this resource requires information about the structure and dynamics of nutrients and communities present in these ecosystems.
The delimitation of spatial gradients in reservoirs allows for identification of parts of reservoirs that are adequate for different purposes, such as fishing and water supply. By readily indicating environmental conditions and exhibiting conservative characteristics superior to those of physical and chemical variables, taxonomic and functional phytoplankton groups can be efficient in characterization of these gradients, as well as for the detection of undesirable environmental scenarios (Nogueira, 2000; Reynolds et al., 2002).
Because of the absence of studies on longitudinal distribution of phytoplankton in subtropical reservoirs, this study investigated the occurrence of a longitudinal gradient in two subtropical Brazilian reservoirs and whether the transition zone constitutes the most productive zone for phytoplankton biomass. This study also verified the efficiency of phytoplankton functional groups in the characterization of longitudinal gradients in reservoirs.
Materials and methods
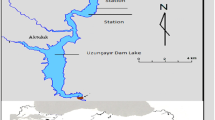
Reservoirs from two hydroelectric power stations (Fig. 1) situated in Southern Brazil (State of Paraná) present distinct age and morphometry. Both present water retention time of about 50 days, i.e., they are classified in Category B according to the classification proposed by Straškraba (1999). Regional climate is characterized as subtropical humid (Maack, 1981), with average annual precipitation of 1,900 mm and average temperatures between 12 and 16°C in winter, and between 20 and 23°C in summer (Júlio et al., 2005). Segredo Reservoir is the second in the series of reservoirs located at the Iguaçu River (Júlio et al., 2005). The Iguaçu River is 1,600 km long and its watershed covers about 72,000 km2 (Eletrosul, 1978).
Capivari Reservoir, located at the Capivari River (Governador Parigot de Souza Hydroelectric Power Station) is considered a headwater reservoir in relation to its position along the river’s longitudinal axis. Although it is considered a littoral watershed reservoir, the Capivari River is a tributary of the Ribeira de Iguape River, which had its water dammed and diverted to the Parana littoral plain by the Cachoeira River bed, also known as Capivari-Cachoeira (Júlio et al., 2005). Table 1 summarizes some available information for both reservoirs.
Samples were obtained every 3 months in 2002, during dry (June and September) and rainy seasons (March and December). Samples were taken at the fluvial (F), transition (T) and lacustrine (L) zones in each reservoir from the following levels: at the subsurface (S); above the lower boundary of the mixing zone (Z M); above the euphotic zone boundary (Z eu); and near the bottom (Z max). The limnological variables, abundance of zooplankton groups and phytoplankton attributes analyzed and their respective methodologies are listed in Table 2. Zooplankton data is available in Velho et al. (2005).
The mixing zone (Z M) was estimated according to the temperature profile of the water column, and the Z eu:Z M ratio was used as a light availability index in the mixing zone (Jensen et al., 1994). Dominant phytoplankton species were considered to be those occurring in biovolume greater than 10% of the total sample biovolume (Huszar & Caraco, 1998). Dominant taxa at epilimnium were grouped in functional groups (FG) according to Huszar et al. (2000), Reynolds et al. (2002) and Padisák et al. (2006).
Using the PC-ORD program (McCune & Mefford, 1999), Canonical Correspondence Analysis (CCA) (Ter Braak, 1986) was used to determine spatial and temporal gradients, and to estimate the environmental variables determining fluctuations in biovolume, dynamics of phytoplankton species, and the influence of zooplankton abundance.
Results
In both studied environments, mean values of air and water temperatures were similar, although Capivari Reservoir presented the lowest mean values, registered in September. Precipitation values were higher from January to March and from October to December 2002, but did not exhibit remarkable seasonality during the study period (Figs. 2a, b). Greater pluviometric indexes occurred in Segredo Reservoir regions (Fig. 2b). As for mean concentrations of dissolved oxygen, we did not observe clear differences between reservoirs during the whole study (Table 3).
Precipitation values in Capivari (a) and Segredo (b) reservoirs. Sampling days are indicated by arrows. Values of maximum depth (Z max), above the euphotic zone boundary (Z eu), above the lower boundary of the mixing zone (Z M) and euphotic zone and mixing zone ratio (Z eu:Z M), recorded in different zones (F: fluvial, T: transition, L: lacustrine) in Capivari (c) and Segredo (d) reservoirs, during study period
Vertical gradients of temperature were verified, as well as differences in euphotic zone depth in both reservoirs. Capivari Reservoir was stratified during the entire study period, except in the fluvial zone in September. In this reservoir, considering the longitudinal gradient, a subtle increase in water transparency from the river towards the dam was registered in March and September, and an accentuated increase of mixing layer depth was seen in June and September (Fig. 2c).
In Segredo Reservoir, a clear distinction among different zones in relation to mixing regime was observed, except during June (Fig. 2d). In this reservoir, a striking vertical temperature gradient was registered in the transition and lacustrine zones. A longitudinal water transparency gradient, which was lower throughout the reservoir, was not observed except in December, when Z eu:Z M ratio was high in the transition and lacustrine zones. In September and especially in December, there was higher light availability in the mixing zone. Throughout the entire study, Segredo Reservoir displayed the highest mean values of TN and NO3 and the highest N:P ratios, especially in September (Table 3 and Fig. 2d).
For both reservoirs, among analyzed abiotic variables, nutrient concentrations (mainly NH4 concentrations) and pluviometric precipitation presented higher variation coefficients (Table 3) during the study period, due to greater differences in nutrient concentrations between the surface and the bottom. In both reservoirs, higher nutrient concentrations were observed in deeper layers, especially in the lacustrine zone. Capivari Reservoir presented higher mean values of electric conductivity, alkalinity, turbidity, and NH4, TP and SRP concentrations, as well as a higher Z eu:Z M ratio. Higher values of these variables were recorded in September in the three zones of the reservoir, except the turbidity, whose higher values were registered in December in the fluvial zone, and the Z eu:Z M ratio, which was higher in June in the same zone (Table 3 and Fig. 2c).
Considering the analyzed nutrients (TP, PO4, TN, NO3), turbidity, and dissolved oxygen, and the decrease in values of these variables towards the dam, Capivari Reservoir exhibited an evident longitudinal gradient. Segredo Reservoir exhibited a less evident longitudinal gradient, and displayed high similarity in values of the above-mentioned variables between the fluvial and transition zones. More details about limnological features of these reservoirs were described by Pagioro et al. (2005).
From the total samples, 118 taxa were identified, and Chlorophyceae (35.6%), Bacillariophyceae (21.2%), and Cyanobacteria (18.6%) were the most specious groups. Segredo Reservoir presented 62 exclusive taxa from a total of 108 surveyed taxa, while 56 taxa were observed in Capivari Reservoir, 10 of which were exclusive to this environment.
Values of phytoplankton biomass were low in both reservoirs, less than 3 mm3 l−1, during the whole study period (Fig. 3a, b). Higher values were observed in the epilimnetic layer. As for longitudinal gradients, Capivari Reservoir showed a decrease in biomass values from the river towards the dam in March and June, and an increase in December. In September, the highest values were recorded in the transition zone. Segredo Reservoir displayed a biomass increase along its longitudinal axis during most of the study period, except in June when the highest values occurred in the transition zone.
Values of phytoplankton biovolume registered in Capivari (a) and Segredo (b) reservoirs, in different zones (F: fluvial, T: transition, L: lacustrine) and depths (s: sub-surface, Z eu: euphotic zone, Z M: inferior limit of mixing zone, Z max: near the bottom; coincidence depth of Z eu and Z M: Z eu = Z M, coincidence depth of Z M and Z max: Z M = Z max) during study period
In Capivari Reservoir, phytoplankton biovolume values ranged from 0.002 mm3 l−1 near the bottom of the lacustrine zone in March, to 2.6 mm3 l−1 at the subsurface of the fluvial zone in the same month (Fig. 3a). Cyanobacteria, Cryptophyceae, and Bacillariophyceae were the greatest contributors to biovolume. For Segredo Reservoir, values of phytoplankton biovolume ranged from 0.009 mm3 l−1 near the bottom of the lacustrine zone in March to 2.3 mm3 l−1 at subsurface of the lacustrine zone in December. Cyanobacteria and Bacillariophyceae were the dominant groups (Fig. 3b).
Six functional groups were observed, and in some cases co-dominance of species belonging to different groups at the same period was also observed. In Capivari Reservoir, LM, B, Y, and X1 groups were observed, and in Segredo Reservoir, LM, X1, MP, and H1 groups were observed (Table 4).
Canonical Correspondence Analysis (CCA) showed significant scores for the first two canonical axes (P < 0.05), according to the Monte Carlo test (Fig. 4a, b). The first two axes together explained 24% of the total data variability. The first CCA axis discriminated both reservoirs, and separated to the left of the diagram, with greater scores dispersion, the Segredo Reservoir, influenced by higher TN concentrations (−0.86), NO3 (−0.85), higher N:P ratio (−0.71) and higher biomass values of Anabaena circinalis, A. solitaria, Aulacoseira spp., and Urosolenia eriensis. Capivari Reservoir was discriminated to the right of the diagram, influenced by higher values of alkalinity (0.82), pH (0.73), conductivity (0.84), air temperature (0.55), greater abundance of rotifer (0.73), copepodites (0.73), and Calanoida adults (0.64) (Table 5), as well as higher biovolume values of Microcystis aeruginosa, Discostella stelligera, and Cryptomonas spp.
Scores dispersion of reservoirs and zones (a), scores of phytoplankton species biovolume, abiotic variables and zooplankton abundance (b) in the first two CCA axes, in different zones (F: fluvial, T: transition, L: lacustrine) and months in Capivari and Segredo reservoirs, during study period. Abundance of rotifers (rot), Calanoida copepodites (ccal), Calanoida adults (acal), subsurface (s), inferior limit of mixing zone (Z M), and other codes in Table 3
The second CCA axis evidenced a temporal gradient for Segredo Reservoir and particularly discriminated December from the other months, as seen in the lower left portion of the diagram, influenced by a greater Z eu:Z M ratio (0.6). In this same month, a longitudinal distribution was observed, with discrimination between the transition and lacustrine zones. For Capivari Reservoir, the CCA diagram evidenced low scores dispersion and, in relation to longitudinal distribution, the fluvial zone in September was discriminated from the transition and lacustrine zones.
Discussion
The low values of phytoplankton biomass registered in the studied reservoirs, even in the epilimnetic layer with great light availability, can be explained by low water retention time, as seen in other reservoirs (Straškraba, 1999; Tundisi et al., 1999; Pivato et al., 2006). In relation to trophic level, according to the Vollenweider (1968) criteria and considering values of phytoplankton biomass, both reservoirs are oligotrophic (<3 mm3 l−1), although, in relation to total phosphorus concentrations, they were predominated by mesotrophic conditions.
The total number of taxa observed in Capivari and Segredo reservoirs is comparable to those registered in other Brazilian reservoirs located in the Central and Southern regions of the country (Nogueira, 2000; Calijuri et al., 2002; Silva et al., 2005). Meanwhile, for Segredo Reservoir, the total number of taxa identified in this study was less than those observed in this environment in 1994–1995, when 463 taxa were registered (Ludwig et al., 1997), which can be explained by the lower sampling effort used in the present study.
In both reservoirs, we observed the classic model of longitudinal distribution of limnological variables described by Thornton et al. (1990). This was more evident in Capivari Reservoir and can be attributed to different morphological features of the reservoirs, since both presented similar retention time. The high flow of Segredo Reservoir, expressed by the greater extension of mixing zone, when compared to Capivari Reservoir, probably hindered the delimitation of transition zone, which displayed similar characteristics as the fluvial zone during most of the study period.
Regarding phytoplankton biovolume, the reservoirs were more distinct from each other than among analyzed zones. The absence of remarkable longitudinal gradients for phytoplankton in most of the months can be related to low Z eu:Z M ratios verified in Segredo Reservoir during most of the study period. In Capivari Reservoir, this absence of clear distribution can be ascribed to the accentuated process of nutrient settlement, occurring since the fluvial zone, due to permanent water column stratification, which limited phytoplankton development in the whole reservoir.
Although a clear seasonality of phytoplankton biomass was not observed in the studied reservoirs, considering the precipitation, greater concentrations of rain in March and December can favor the increase in phytoplankton biomass in Capivari and Segredo reservoirs, respectively, due to greater input of allochtonous nutrients. The increase in biomass during the rainy season associated with increases in nutrient concentrations was also recorded by Silva et al. (2005) in the lacustrine zone from Segredo Reservoir, and Nogueira (2000) in Jurumirim Reservoir.
The dominance of cryptophyceans and diatoms during most of the study period in Capivari Reservoir reflected the hydrodynamic environment, which favors the nanoplanktonic species with high reproductive rate that recycle the nutrients in the epilimnetic layer (Reynolds, 1997; Reynolds et al., 2002). Dominance of Cyanobacteria during the rainy season and Bacillariophyceae during the dry season in Segredo Reservoir, was influenced by seasonal temperature variations observed between these periods, with higher diatom biomass during the coldest months (June and September), as already verified by Ludwig et al. (1997) and Silva et al. (2005), in this same environment. Diatoms contributed more to biomass in Segredo Reservoir by virtue of being adapted to high current flow and low light availability (Reynolds, 1997) and, probably, due to inoculum addition coming from the upstream stretch and from tributaries where this group is commonly found (Ludwig et al., 1997).
The dominance of Aulacoseira granulata var. granulata and Aulacoseira granulata var. angustissima (MP) in Segredo Reservoir, was associated with higher nutrient concentrations (especially nitrogen), lower water temperatures and Z eu:Z M ratios, and conditions of total water column mixing or when the extension of the mixing zone was greater than 5 m. A. granulata is common in tropical and subtropical reservoirs where, during thermal stratification periods, it lies in sediment, which constitutes a defensive and advantageous mechanism (Reynolds, 1999).
Aulacoseira ambigua is grouped by Reynolds et al. (2002) in FG C in conditions of total water column mixing, as it is sensitive to stratification. However, in the present study, A. ambigua var. ambigua f. spiralis occurred together with A. granulata var. granulata and Aulacoseira granulata var. angustissima in Segredo Reservoir, which suggests that these species have similar environmental requirements. Based on that, we opted to establish these meroplanktonic species in FG MP (Padisák et al., 2006).
We suggest a review of Urosolenia eriensis insertion, originally belonging to FG A, which characterizes transparent and oligotrophic lakes (Reynolds et al., 2002), since in the present study, this species was co-dominant with A. granulata var. granulata and A. ambigua var. ambigua f. spiralis in mesotrophic conditions and high turbidity. Moreover, the record of these meroplanktonic species in conditions similar to those seen in this study, as observed by Train & Rodrigues (1998) and Bovo-Scomparin & Train (in press), corroborates their insertion into FG MP.
Anabaena circinalis (H1) dominance at the epilimnium of Segredo Reservoir in December, especially in the transition and lacustrine zones, can be explained by the occurrence of water column stratification and high nutrient and light availability in the mixing zone. Blooms of this species are common in conditions of thermal stratification (Westwood & Ganf, 2004; Bovo-Scomparin & Train, in press).
Although Anabaena species have low ability to change their vertical position in the water column, the presence of aerotopes and a mucilaginous sheath allow filaments to be maintained in euphotic zone for long periods; consequently, these species can be favored in conditions of nutrient availability in this water layer (Nogueira, 2000; Reynolds et al., 2002). Another factor that probably favored Anabaena circinalis in December in Segredo Reservoir is its competitive ability to reduce the pressure of zooplankton predation due to its large size and toxicogenic potential (Carmichael, 1997).
High values of Microcystis aeruginosa (LM) biomass observed in Capivari Reservoir during stratification conditions are ascribed to the ability of this species to regulate its position in the water column due to the presence of a mucilaginous sheath and aerotopes (Ganf, 1974; Reynolds, 2007). M. aeruginosa was also favored by high temperature, low N:P ratios, and nutrient and light availability in the mixing zone. Other authors have also verified the favorability of the aforementioned conditions for Cyanobacteria (Fujimoto et al., 1997; Marinho & Huszar, 2002; Bovo-Scomparin & Train, in press).
The dominance of Cryptomonas sp., belonging to the Y functional group, in Capivari Reservoir in favorable conditions of total phosphorus and lower light availability in the mixing zone denotes its opportunistic feature (Reynolds, 1997; Rodrigues et al., 2005; Train et al., 2005; Pivato et al., 2006). The occurrence of Synechocystis aquatilis concomitant with Cryptomonas sp. in mesotrophic conditions, and also in reservoirs’ fluvial zones, enables the insertion of this taxum into FG X1, as suggested by Huszar et al. (2000).
Discostella stelligera (B) was dominant in Capivari Reservoir in mesotrophic conditions and the mixing zone up to 6 m. Although diatoms, in general, are favored in low temperatures (Reynolds, 1997; Silva et al., 2005), the biomass of Discostella stelligera was probably limited by the short retention time of this reservoir, and not by high temperatures registered in December when its biomass values were greater, indicating the eurythermic character of this species.
Zooplankton communities can negatively affect phytoplankton by cell consumption, or positively by nutrient recycling (Grigorszky et al., 1998). This relationship is dependent on several factors, including the composition and morphophysiological features of algae (James & Forsyth, 1990). For the studied reservoirs, we observed that greater abundance of rotifers, copepodites, and Calanoida adults were correlated to the biomass of nanoplanktonic species with high reproductive rate. Bonecker et al. (2007) also verified a correlation between these two communities in other reservoirs from Southern Brazil.
Functional groups of phytoplankton were efficient indicators of environmental conditions, as observed in other environments (Huszar et al., 2000; Reynolds et al., 2002; Pivato et al., 2006; Bovo-Scomparin & Train, in press), and can be used to indicate limnological variables of different reservoir zones and characterize reservoirs with distinct age, morphometry, and watershed purposes. Capivari Reservoir was characterized mainly by LM and Y functional groups, indicative of its greater water column stability and higher phosphorus concentration, while Segredo Reservoir was principally characterized by the MP functional group, indicative of its greater mixing zone extension and higher nitrate concentration. The functional group H1 also efficiently characterized the transition and lacustrine zones of Segredo Reservoir during December, when water column stratification, high Z eu:Z M ratio and high nutrient availability were observed.
The occurrence of a longitudinal gradient with higher phytoplankton biomass in the transition zone was only observed in Segredo Reservoir in June, and in Capivari Reservoir in September. This absence of remarkable longitudinal gradients for phytoplankton can be related to low Z eu:Z M ratios verified in Segredo Reservoir during most of the study period, whereas in Capivari Reservoir, this result was associated with the accentuated process of nutrient settlement which occurred since the fluvial zone. The obtained results also evidenced the influence of morphometric conditions and watershed purposes as important structuring factors of phytoplankton biomass in these reservoirs.
References
American Public Health Association, 1995. Standard Methods for the Examination of Water and Wastewater, 19th ed. APHA, Washington DC.
Armengol, J., J. C. Garcia, M. Comerma, M. Romero, J. Dolz, M. Roura, B. H. Han, A. Vidal & K. Simek, 1999. Longitudinal processes in canyon type reservoirs: The case of Sau (N.E. Spain). In Tundisi, J. G. & M. Straškraba (eds), Theoretical Reservoir Ecology and its Applications. International Institute of Ecology, Brazilian Academy of Sciences and Backhuys Publishers, São Carlos, 313–345.
Bonecker, C. C., M. Y. Nagae, M. C. M. Bletller, L. M. F. Velho & F. A. Lansac-Tôha, 2007. Zooplankton biomass in tropical reservoirs in southern Brazil. Hydrobiologia 579: 115–123.
Bottrell, H. H., A. Duncan, Z. M. V. Gliwicz, E. Grygierek, A. Herzig, A. Hillbricht-Ilkowska, H. Kurazawa, P. Larsson & T. Weglenska, 1976. A review of some problems in zooplankton production studies. Norwegian Journal of Zoology 24: 419–456.
Bovo-Scomparin, V. M. & S. Train. Long-term variability of the phytoplankton community in an isolated floodplain lake of the Ivinhema River State Park, Brazil. Hydrobiologia (in press).
Calijuri, M. C., A. C. A. Dos Santos & S. Jati, 2002. Temporal changes in the phytoplankton community structure in tropical and eutrophic reservoir (Barra Bonita, SP – Brazil). Journal of Plankton Research 24: 617–634.
Carmichael, W. W., 1997. The cyanotoxins. Advances in Botanical Research 27: 211–256.
Edler, L., 1979. Recommendations for Marine Biological Studies in the Baltic Sea: Phytoplankton and Chlorophyll. UNESCO, Paris (UNESCO, Working Group 11, Baltic Marine Biologists).
Eletrosul, 1978. O impacto ambiental da ação do homem sobre a natureza – rio Iguaçu, Paraná, Brasil – reconhecimento da ictiofauna, modificações ambientais e usos múltiplos dos reservatórios. Florianópolis.
Fujimoto, N., R. Sudo, N. Shugiura & Y. Inamori, 1997. Nutrient-limited growth of Microcystis aeruginosa and Phormidium tenue and competition under various N:P supply ratios and temperatures. Limnology and Oceanography 42: 250–256.
Ganf, G. G., 1974. Diurnal mixing and vertical distribution of phytoplankton in a shallow equatorial lake. Journal of Ecology 62: 611–629.
Giné, M. F., H. Bergamin, E. A. G. Zagatto & B. F. Reis, 1980. Simultaneous determination of nitrite and nitrate by flow injection analysis. Analytica Chimica Acta 114: 191–197.
Golterman, H. L., R. S. Clymo & M. A. M. Ohstad, 1978. Methods for Physical and Chemical Analysis of Freshwater. Blackwell Scientific Publication, Oxford.
Grigorszky, I., S. Nagy, A. Tóth, C. Máthé, Z. Müller & G. Borbély, 1998. Effect of large and of small-bodied zooplankton on phytoplankton in a eutrophic oxbow. Journal of Plankton Research 20: 1989–1995.
Huszar V. L. M. & N. F. Caraco, 1998. The relationship between phytoplankton composition and physical–chemical variables: A comparison of taxonomic and morphological–functional descriptors in six temperate lakes. Freshwater Biology 40: 679–696.
Huszar, V. L. M., L. H. S. Silva, M. M. Marinho, P. Domingos & C. L. Sant`Anna, 2000. Cyanoprokaryote assemblages in eight productive tropical Brazilian waters. Hydrobiologia 424: 67–77.
James M. R. & D. J. Forsyth, 1990. Zooplankton–phytoplankton interactions in a eutrophic lake. Journal of Plankton Research 12: 455–472.
Jensen, J. P., E. Jeppesen, K. Olrik & P. Kristensen, 1994. Impact of nutrients and physical factors on a shift from Cyanobacterial to Chlorophyte dominance in shallow Danish lakes. Canadian Journal of Fisheries and Aquatic Science 51: 1692–1699.
Júlio, H. F., Jr., S. M. Thomaz, A. A. Agostinho & J. D. Latini, 2005. Distribuição e caracterização dos reservatórios. In Rodrigues, L., S. M. Thomaz, A. A. Agostinho & L. C. Gomes (eds), Biocenoses em reservatórios: padrões espaciais e temporais. RIMA, São Carlos, 1–16.
Kimmel, B. L., O. T. Lind & L. J. Paulson, 1990. Reservoir primary production. In Thornton, K. W., B. L. Kimmel & F. E. Paine (eds), Reservoir Limnology: Ecological Perspectives. Wiley, New York, 133–193.
Ludwig, T. A. V., I. T. Cecy, L. H. Riberiro, T. L. Flores, D. Atab & S. Silva, 1997. Phytoplankton and some abiotics parameters in Segredo hydroeletric power plant reservoir during one year (Parana State, south Brazil). Internationale Vereinigung fuer Theoretische und Angewandte Limnologie Verhandlugen 26: 520–521.
Maack, R., 1981. Geografia física do Estado do Paraná. José Olympio, Rio de Janeiro.
Mackereth, F. Y. H., J. R. Heron & J. F. Tailing, 1978. Water analysis: Some revised methods for limnologists. Freshwater Biological Association Scientific Publication.
Marinho, M. M. & V. L. M. Huszar, 2002. Nutrient availability and physical conditions as controlling factors of phytoplankton composition and biomass in a tropical reservoir (Southeastern Brazil). Archiev für Hydrobiologie 153: 443–468.
McCune, B. & M. J. Mefford, 1999. PC-ORD. Multivariate Analysis of Ecological Data, Version 4.0. MjM Software Design, Gleneden Blach, Oregon.
Nogueira, M. G., 2000. Phytoplankton composition, dominance and abundance as indicators of environmental compartmentalization in Jurumirim Reservoir (Paranapanema River), São Paulo, Brazil. Hydrobiologia 431: 115–128.
Padisák, J., G. Borics, I. Grigorszky & E. Soroczki-Pinter, 2006. Use of phytoplankton assemblages for monitoring ecological status of lakes the Water Framework Directive: The assemblage index. Hydrobiologia 553: 1–14.
Pagioro, T. A., M. C. Roberto, S. M. Thomaz, S. A. Pierini & M. Taka, 2005. Zonação longitudinal das variáveis limnológicas abióticas em reservatórios. In Rodrigues, L., S. M. Thomaz, A. A. Agostinho & L. C. Gomes (eds), Biocenoses em reservatórios: Padrões espaciais e temporais. RIMA, São Carlos, 39–56.
Pivato, B. M., S. Train & L. C. Rodrigues, 2006. Dinâmica nictemeral das assembléias fitoplanctônicas em um reservatório tropical (reservatório de Corumbá, Estado de Goiás, Brasil), em dois períodos do ciclo hidrológico. Acta Scientiarum 28: 19–29.
Reynolds, C. S., 1997. Vegetation process in the pelagic: A model for ecosystem theory. Ecology Institute, Oldendorf.
Reynolds, C. S., 1999. Phytoplankton assemblages in reservoirs. In Tundisi, J. G. & M. Straskraba (eds), Theoretical Reservoir Ecology and its Applications. International Institute of Ecology, Brazilian Academy of Sciences and Backhuys Publishers, São Carlos, 439–456.
Reynolds, C. S., 2007. Variability in the provision and function of mucilage in phytoplankton: Facultative responses to the environment. Hydrobiologia 578: 37–45.
Reynolds, C. S., V. L. M. Huszar, C. Kruk, L. Naselli-Flores & S. Melo, 2002. Towards a functional classification of the freshwater phytoplankton. Journal of Plankton Research 24: 417–428.
Rodrigues, L. C., S. Train, B. M. Pivato, V. M. Bovo, P. A. F. Borges & S. Jati, 2005. Assembléias fitoplanctônicas de trinta reservatórios do Estado do Paraná. In Rodrigues, L., S. M. Thomaz, A. A. Agostinho & L. C. Gomes (eds), Biocenoses em reservatórios: padrões espaciais e temporais. RIMA, São Carlos, 57–72.
Silva, C. A., S. Train & L. C. Rodrigues, 2005. Phytoplankton assemblages in a Brazilian subtropical cascading reservoir system. Hydrobiologia 537: 99–109.
Straškraba, M., 1999. Retention time as a key variable of reservoir limnology. In Tundisi, J. G. & M. Straškraba (eds), Theoretical Reservoir Ecology and its Applications. International Institute of Ecology, Brazilian Academy of Sciences and Backhuys Publishers, São Carlos, 385–410.
Ter Braak, C. J. F., 1986. Canonical correspondence analysis: A new eigenvector technique for multivariate direct gradient analysis. Ecology 67: 1167–1179.
Thornton, K. W., B. L Kimmel & F. E. Paine, 1990. Reservoir Limnology: Ecological Perspectives. Wiley, New York.
Train, S., S. Jati, L. C. Rodrigues & B. M. Pivato, 2005. Distribuição Espacial e Temporal do Fitoplâncton em Três Reservatórios da Bacia do Rio Paraná. In Rodrigues, L., S. M. Thomaz, A. A. Agostinho & L. C. Gomes (eds), Biocenoses em reservatórios: padrões espaciais e temporais. RIMA, São Carlos, 73–85.
Train, S. & L. C. Rodrigues, 1998. Temporal fluctuations of the phytoplankton community of the Baía River, in the upper Paraná River floodplain, Mato Grosso do Sul, Brazil. Hydrobiologia 361: 125–134.
Tundisi, J. G., T. Matsumura-Tundisi & O. Rocha, 1999. Theoretical basis for reservoir management. In Tundisi, J. G. & M. Straškraba (eds), Theoretical Reservoir Ecology and its Applications. International Institute of Ecology, Brazilian Academy of Sciences and Backhuys Publishers, São Carlos, 505–528.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen phytoplankton-methodic. Mitteilungen Internationale Vereinigung für Theoretische und Angewandte Limnologie 9: 1–38.
Velho, L. F. M., F. A. Lansac-Tôha & C. C. Bonecker, 2005. Distribuição longitudinal da comunidade zooplanctônica em reservatórios. In Rodrigues, L., S. M. Thomaz, A. A. Agostinho & L. C. Gomes (eds), Biocenoses em reservatórios: Padrões espaciais e temporais. RIMA, São Carlos, 129–136.
Vollenweider, R. A., 1968. Scientific fundamentals of the eutrophication of lakes and flowing water, with particular reference to phosphorus and nitrogen as factors in eutrophication. Tech. Rep. OECD Paris, DAS/CSI/58-27, 1–159.
Westwood, K. J. & G. G. Ganf, 2004. Effect of mixing patterns and light dose on growth of Anabaena circinalis in a turbid, lowland river. River Research Application 20: 115–126.
Wetzel, R. G. & G. E. Likens, 2000. Limnological Analysis. Springer-Verlag, New York.
Acknowledgements
The authors are grateful to the Núcleo de Pesquisas em Limnologia, Ictiologia e Aqüicultura (Nupélia) of Universidade Estadual de Maringá, for logistic support; to researchers from Nupélia Limnology and Zooplankton Laboratories; to Agência Nacional de Águas (ANA) and Companhia Paranaense de Energia (COPEL); to CNPq/PRONEX for financial support; to CAPES for a scholarship granted to the first author, and to anonymous reviewers for suggestions on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: L. Naselli-Flores
Rights and permissions
About this article
Cite this article
Borges, P.A.F., Train, S. & Rodrigues, L.C. Spatial and temporal variation of phytoplankton in two subtropical Brazilian reservoirs. Hydrobiologia 607, 63–74 (2008). https://doi.org/10.1007/s10750-008-9367-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-008-9367-3