Abstract
This study examines the population structure and regeneration of Brazil nut tree (Bertholletia excelsa) populations at sites subjected to extractive activities in the Capanã Grande Lake Extractive Reserve (Rio Madeira, Amazonas, Brazil). We analyze correlations between three regeneration levels (seedling, sapling and juvenile density) and seven possible explicatory variables related to vegetation structure and human action. The tested vegetation structure variables include adult tree density, crown area, basal area, canopy openness, and fruits opened by agoutis. We also test the time of human use and distance from residences as possible explicatory variables. The density values of the Brazil nut stands in the region are high (12.5 tree/ha), with 60 % of trees having a diameter at breast height (DBH) > 80 cm. The mean potential regeneration (24.8 seedlings/ha), pre-established regeneration (4.4 saplings/ha) and established regeneration (3.2 juveniles/ha) values are high compared to other Brazil nut tree populations and tend to be greater in the “intensively used” stands, defined as sites where human presence during the year is more frequent. The time of human use and the density of adult Brazil nut trees are the variables that best correlate with established regeneration (density of juveniles). The number of juveniles/adult Brazil nut trees is five times higher in intensively used stands than in those moderately used. These results suggest that extractive activities and disturbances in the understory caused by human presence may favor (intentionally or not) regeneration of Brazil nut trees in more-frequently used stands. The data support the concept, frequently suggested in the literature, that aggregations of Brazil nut trees could be a consequence of the activities of Amerindian populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Brazil nut tree (Bertholletia excelsa Bonpl., Lecythidaceae) is a tropical species that has a wide and patchy distribution throughout the Amazon Basin and the Guyanas in non-flooded (terra firme) forests (Mori and Prance 1990). It is a large, emergent tree that can reach a height of more than 50 m and a diameter at breast height (DBH) of greater than 300 cm (Zuidema and Boot 2002; Salomão 2009; Scoles and Gribel 2011, 2012).
The fruit is a lignified sphere with a diameter of 10 to 12 cm and a weight of 0.5 to 2.5 kg, and contains 10 to 25 seeds. The seeds (the commercially available Brazil nuts) are edible, have a wedge-like shape, and are covered by a protective lignified shell (Mori and Prance 1990; Mori 1992). Most individuals of reproductive age have DBHs > 40 cm (Zuidema and Boot 2002), whereas the most productive trees have DBHs between 100 and 150 cm (Wadt et al. 2005; Kainer et al. 2007). Their longevity and high survival rates under natural conditions allow these trees to produce nuts for hundreds of years (Zuidema and Boot 2002).
One characteristic of the population structure of the Brazil nut tree is that it forms stands of between 75 and 150 trees with DBHs > 10 cm (Peres and Baider 1997), at densities (5–20 trees ha−1) that are generally higher than those of other tree species in the Amazon rainforest. Such stands are interspersed with areas of forest lacking Brazil nut trees or showing densities of this species as low as one tree per 6 ha (Mori and Prance 1990). Most studies of Brazil nut populations have reported a low proportion of non-reproductive trees (DBH < 40 cm; Salomão 1991; Nepstad et al. 1992; Scoles and Gribel 2012) and a predominance of trees with intermediate size (80 cm < DBH < 160 cm; Zuidema and Boot 2002; Peres et al. 2003; Wadt et al. 2008; Scoles and Gribel 2012).
B. excelsa produces an edible seed (Brazil nuts) that is one of the principal non-timber forest products (NTFP) commercialized in the Amazon region and is an important economic resource for the human populations that inhabit the Amazon rain forest (Mori 1992; Clay 1997; Ortiz 2002; Stoian 2004; [IBCE] Instituto Boliviano de Comercio Exterior 2010). Extraction in natural forests is the principal source of Brazil nut seeds (Ortiz 1995; Clay 1997; Stoian 2004), despite the possibility of cultivation of Brazil nuts in plantations and agroforests (Fernandes and Alencar 1993; Souza et al. 2008; Costa et al. 2009; Scoles et al. 2011, 2014).
The dispersal of the seeds of the Brazil nut tree begins with the fall of mature fruits during the rainy season. B. excelsa is the only species in the family Lecythidaceae with a functionally indehiscent fruit (i.e., it does not open spontaneously when mature) (Mori and Prance 1990). Consequently, seeds are inserted into the lignified fruit and dispersion depends on an external agent. In addition to humans, agoutis are the only animals capable of opening the hard fruit wall with regularity and efficiency (Huber 1910; Ortiz 1995; Peres and Baider 1997; Tuck Haugaasen et al. 2010, 2012).
The natural dispersion of Brazil nut tree seeds is complex. Agoutis are seed predators and seed dispersion occurs indirectly when they bury the seeds for future consumption (“scatter-hoarding”). Only when the seed buried will germination occur, as long as it remains undisturbed by agoutis. In addition to being inefficient, agouti seed dispersion occurs only over short distances, and fruit dispersal and seed burial at distances greater than 50 m from the source tree is rare (Peres and Baider 1997; Tuck Haugaasen et al. 2010, 2012).
Once germination has occurred, the survival and development of the seedlings are supported by favorable light conditions in large gaps or below canopy disturbances in the forest. The Brazil nut tree is a heliophytic species (Salomão 1991; Scoles et al. 2011, 2014) that behaves as a long-lived light-demanding climax species (Swaine and Whitmore 1988); it cannot develop in understory conditions (Kainer et al. 1998; Myers et al. 2000; Peña-Claros et al. 2002; Scoles et al. 2011, 2014).
Generally, Brazil nut harvesters intensively extract the seeds of B. excelsa in accessible productive stands of trees since harvesting more distant stands requires a larger investment of time. Harvesters also hunt animals such as agoutis, especially when harvesting requires extended periods in the forest away from the community (Ortiz 2002; personal observations by the authors).
The harvest of NTFP is generally considered a promissory activity that has a low environmental impact and that represents an important source of income for traditional Amazon communities; it is an alternative to activities such as cattle ranching and farm monoculture. Therefore, NTFP harvesting may potentially reconcile conservation and social development in the Amazon region (Fearnside 1989; Allegretti 1994; Anderson 1994; Ruiz-Pérez et al. 2004). However, extractive activities have also been criticized by economists because of the lack of control over production, harvest unpredictability, difficulty in meeting growing demands and a low level of processing and technology (Homma 1993; Amin 1997; Clement 2006).
The ecological sustainability of Brazil nut harvesting has been analyzed in several studies (Mori 1992; Viana et al. 1998; Peres et al. 2003; Silvertown 2004; Wadt et al. 2008; Scoles and Gribel 2011, 2012; Ribeiro et al. 2014), some of which have reached contrasting conclusions. With the exception of Peres et al. (2003) and Silvertown (2004)), these studies found no evidence that traditional extractive methods damage the regeneration of Brazil nut stands. In general, the studies show that the relative scarcity of trees of non-reproductive age (juveniles) and the low recruitment rate are not related to harvesting intensity. Furthermore, Scoles and Gribel (2011), comparing populations located in two distinct sites (Trombetas and the Madeira River Region), found a positive influence of human activities on the rejuvenation and density of B. excelsa stands. In the State of Acre, southwest of the Amazon, Wadt et al. (2008) concluded that within the current harvest context regeneration is sufficient for population persistence at the research sites. Additionally, Ribeiro (2011) highlighted the possibility that current management by humans has contributed to the maintenance and formation of Brazil nut stands in the indigenous territory of the Xingu River. In the Trombetas River valley in Pará State, the low observed regeneration rates were independent of both harvest intensity and the dispersive activity of agoutis (Scoles and Gribel 2012).
The balance between the negative effects of human activities on regeneration (removal of seeds by harvesting, hunting of the main natural seed disperser) and putative positive influences (“release” of seeds from the lignified fruits, unintentional dispersion of seeds, increased sunlight in the understory by small disturbances) is still an open question for understanding the structure and dynamics of the exploited Brazil nut stands. In this context, this study examines how regeneration (seedling, sapling and juvenile density) of Brazil nut trees is correlated with vegetation structure and human presence in the stands. The main issues we address are: 1) the ecological variables that best correlate with the natural regeneration of B. excelsa, and 2) the differences observed in the population structures of B. excelsa with respect to the two types of human use patterns encountered in the study area (moderate and intensive use of stands).
Material and Methods
Study Site
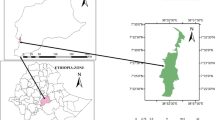
This study was conducted at Capanã Grande Lake, which is located on the left bank of the Madeira River, in the municipality of Manicoré, State of Amazonas, Brazil. In 2004, the study area was incorporated into a federal conservation unit called the ‘Capanã Grande’ Lake Extractive Reserve).
The stands of Brazil nut trees investigated in this study are situated around the central portion of Capanã Grande Lake on the western margin of the Madeira River (Fig. 1), near the riverine communities of Nossa Senhora de Sta. Civita (30 families), Ponta do Campo (39 families), São Sebastião de Cumã (10 families), and São José de Cumã (28 families), a total of 581 inhabitants (ICMbio 2013). The local communities are composed of descendants of Amerindians who mixed with European migrants during colonial times. The communities are concentrated around nucleated settlements, but scattered households are also dispersed around the lake shores. Productive forest areas (‘castanhais,’ ‘seringais’) tend to be exploited by different family groups. The livelihoods of the residents are based on fishing, hunting, extraction of forest products (rubber, fibers, nuts, fruits, resin, etc.) and subsistence agriculture (mainly manioc crops). The Brazil nut stands were mainly visited during the wet season for nut collection. The more accessible stands (generally those closer to the communities) were visited frequently, and they consequently exhibit understories that are more frequently disturbed by trails and clearings.
The climate of the region is classified in the Köppen system as humid tropical without a dry season (Af), with average temperatures between 24° and 26 °C, relative humidity greater than 80 % and average annual precipitation of approximately 2500 mm (PROJETO RADAMBRASIL 1978; Martorano et al. 2004; Alvares et al. 2013).
Study Design
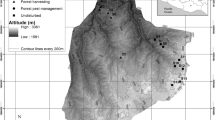
Data were collected in 10 sampling transects. Each transect measured 1000 × 50 m (5 ha), with a central line that approximately corresponded to a section of the trails used by Brazil nut harvesters. In each transect, a maximum perpendicular distance of 25 m on either side of the central line was allotted for the detection of Brazil nut trees. Compared to the other sites, the Laranjal site had a smaller sample size (800 × 50 m = 4 ha) because of the reduced length of the harvester trail at this site. Therefore, the total sampled area of all the transects was 49 ha. Ten 25 × 10 m subplots that were randomly positioned within the transects were used for the collection of data that required more intensive observations, such as the identification of seedlings, saplings, and fruits on the forest floor.
Human Influence Indicators
A selection of 10 transects was made after surveying and mapping stands of Brazil nut trees in the study region with the help of local harvesters. Each selected transect was representative of Brazil nut stands harvested by one to three families from the neighboring communities. In all of the study sites, Brazil nut seed harvest activity occurs every year.
We used two quantitative indicators to measure the extent of human influence on the stands of Brazil nut trees. The first was the ‘time of human use’ in the Brazil nut tree stands. This indicator quantified the number of days each family spent in the stand conducting different activities (nut collection, rubber tapping, fiber extraction, perambulation, etc.) over the course of a year. The data to estimate this indicator were obtained from 20 interviews with the local families that used each stand (1–3 interviews per site). The second indicator, ‘distance from the nearest residence,’ quantified the linear distance between the core area of the stand being investigated and the nearest human residence, obtained from GPS measurements. This variable is considered a putative indicator of human influence in the stands, as areas near human residences tend to be more disturbed by anthropogenic activities.
Based on interviews with local harvesters and personal observations, two types of Brazil nut tree stands were considered for comparative analyses: stands of ‘moderate use’ and ‘intensive use’ (Table 1). Moderately used stands are characterized as being used by harvesters during 2–4 months per year that are almost always restricted to the period of the Brazil nut harvest. In general, access to these stands is limited by the period of flooding. These stands are located in forest areas with low levels of human influence and with little or no forest fragmentation. Six transects were in this group, three of which were localized near streams on the left bank of the Capanã Grande Lake: Taracuá (Inajá stream), Castalhalzinho, and Porto Alto (Cumã stream). The Lembrança, São Julião, and Fraternidade stands were located near flooded forests and were accessible by canoe only during the high-water period. The distance between these sites and the nearest human residence varied between 1.44 km (Fraternidade) and 8.75 km (Taracuá).
Intensively used stands were generally located close the human communities and were easily accessed by land during the dry season and by river during the wet season. These stands are fragmented forests that are surrounded by crops and disturbed vegetation that promote border effects. In addition to the collection of fruits from the Brazil nut trees, other activities, such as rubber tapping of Hevea brasiliensis, trail opening, extractive removal of forest products, and selective logging are frequently conducted in these areas, especially during the dry season. We estimated the time of human use of these stands to be 300 days annually. In spite of the managed appearance of the forest, residents of the communities near these stands affirmed that the stands of Brazil nut trees were spontaneous and that humans had never intentionally planted seedlings of B. excelsa in these forests.
Population Structure
For each Brazil nut tree with a DBH > 10 cm, the DBHs and the lengths of two perpendicular branches of the crown were measured. The individual crown area was calculated by multiplying the elliptical surface area of the crown by the crown form index. The principal branches measured during the initial data collection served as the radii of the ellipse. Based on our field observations, the crown form index was assigned one of four possible proportional values: 1 = intact crown, 0.75 = crown missing one principal branch, 0.5 = crown in the shape of a half-moon, and 0.25 = crown containing only one principal branch. The B. excelsa crown area per hectare was estimated by summing the individual crown areas and dividing by the area of the sampling transect. Basal area per hectare was calculated by summing the cross-sectional areas (=π/4*DBH2) of all B. excelsa trees with DBH > 10 cm and then dividing by the area of the sampling transect.
Stems of B. excelsa with DBH values between 10 and 40 cm were categorized as juveniles or non-reproductive trees. Small plants (DBH < 10 cm) were categorized as seedlings (<1.3 m) or saplings (>1.3 m). Seedlings, having only recently germinated, generally still have the hypocotyl (seed carbohydrate reserve) attached at the base of the plant, whereas for saplings, the hypocotyl is completely absorbed (Myers et al. 2000; Zuidema and Boot 2002). Individual saplings and seedlings found within 25 m × 10 m subplots and up to 5 m on each side of the central line of the transect were registered. Sapling and seedling densities for each sampled transect were obtained by the average density found in the subplots and along the central transect line.
Calculation of Canopy Openness
Canopy openness was estimated indirectly by analyzing photographic images of the forest canopy (Engelbrecht and Herz 2001) and counting non-obscured pixels as a proportion of the total number of pixels within a grayscale range of 0 to 256. The photographs were taken with a Ricoh GX100 digital camera with a wide-angle lens (19 mm), placed at a height of 1.30 m above the ground on a tripod in conditions of indirect sun exposure in the early morning and/or late afternoon.
In each sampling unit, 20 images of the canopy were made: 10 were taken from the central position of the 25 × 10 m subplots, and 10 were taken at random positions located at least 50 m from each other (Nicotra et al. 1999). The digital images were processed using Miramón 6.0 image analysis software, calculating the percentage of the area of each photograph that was not obscured by vegetation. The proportions of unobscured pixels were arcsine-transformed (Zar 1999), and the resulting value was used as the measure of canopy openness.
Estimation of Agouti Activity
The number of Brazil nut fruits opened and gnawed by agoutis in each transect was used as an indirect estimate of the dispersive and/or predatory activities of these animals. Sampling for fruits that showed evidence of having been gnawed by agoutis was conducted using two methods: by counting the fruits found within 5 m on each side of the central transect line (10 × 1.000 m) and by counting the fruits within ten 25 m × 10 m subplots. The agouti activity estimate was the weighted average of the gnawed fruit density using the two methods.
Regeneration Indicators
Natural regeneration of B. excelsa populations was evaluated on three levels: potential, pre-established, and established regeneration. Seedling density was used to estimate the regeneration potential in the studied transects. In this study, regeneration was qualified as ‘potential,’ as the mortality rates of this age group under natural conditions are high (Myers et al. 2000; Zuidema and Boot 2002); for this reason, seedling presence does not guarantee the future establishment of Brazil nut trees.
In contrast, juvenile density (DBH 10 ≥ 40 cm) was considered an indicator of established regeneration, as this age group has already developed a lignified structure that allows for vertical growth up into the higher strata of the sub-canopy or even to the canopy. The proportion of juveniles was calculated by taking into consideration the ratio between the number of non-reproductive trees (DBH 10 ≥ 40 cm) and the total number of trees with DBH > 10 cm. Finally, sapling density was estimated as an indicator of pre-established regeneration, which is an intermediate situation between potential and established regeneration. Despite the high survival rate of this group due to the absorption of the hypocotyl, saplings generally inhabit the understory and therefore have not reached the higher strata of the forest canopy structure.
Data Analyses
We calculated descriptive statistics (average, standard deviation, minimum and maximum) for each analyzed variable. We tested possible correlations between regeneration indicators, ecological variables of the Brazil nut stands and human influence indicators. Multiple regression analyses were then performed between each of the three dependent variables (the regeneration indicators) and the independent variables (the human influence indicators and the ecological variables). When the multiple regressions yielded non-significant equations, paired correlations were used to analyze the possible associations between each independent variable and the dependent variables. In the case of more than one significant result between the independent and dependent variables, a partial correlation analysis was used to identify a stronger relationship between them. The type of regression used depended on the sample distribution of the variables analyzed: Pearson’s test was used when the distribution was normal and the variables had a linear relationship; in other cases, Spearman’s test was used. The Kruskal-Wallis test was used to compare differences in canopy openness among the ten sampling transects. Comparative analyses between the two types of stands (moderate- and intensive-use stands) were conducted using t-test (for variables exhibiting normal distribution) or Kolmogorov-Smirnov (K-S; for variables without normal distribution) (Zar 1999).
Results
Population Structure of Brazil Nut Trees (DBH > 10 cm)
A total of 609 Brazil nut trees with DBH > 10 cm were found in the 49-ha area surveyed; there were 452 potentially reproductive (DBH > 40 cm) trees and 157 non-reproductive juveniles (DBH < 40 cm). The overall density of all the Brazil nut trees with DBH > 10 cm was 12.5 ± 7.8 trees ha−1 (9.3 ± 4.6 adults ha−1 and 3.2 ± 3.4 juveniles ha−1). Among trees with DBH > 10 cm, the mean DBH was 73.1 ± 44.0 cm, the total average basal area was 7.1 ± 2.5 m2 ha−1 and the total area of crown coverage was 3385 ± 1351 m2 ha -1, which represents a 33.8 % occupation of the forest canopy by Brazil nut trees.
The transect with the largest number of non-reproductive juveniles was Vitoriano (9.8 juveniles ha−1). In the three transects near the stream, there was an absence of juveniles in the stands of Brazil nut trees. Considering all individuals with DBH > 10 cm, the proportion of juveniles in the study area was 25.8 %, and five transects had >25 % juveniles, with a maximum value of 40.5 % in the Vitoriano transect (Table 1).
We identified 158 small plants (DBH < 10 cm) of B. excelsa; there were 123 seedlings and 35 saplings. The average density of the seedlings was 24.8 ± 19.9 seedlings ha−1 (maximum 64 and minimum 8). The average density of the saplings was 4.4 ± 5.3 ha−1 (maximum 13.1 and minimum 0). Sixteen percent of seedlings were found in small bunches of B. excelsa fruits opened and left by harvesters, and 4 % of seedlings and 17 % of saplings had more than one stem growing at their bases. The majority of saplings with this growth form were found in areas of secondary forest. Seedling density was significantly greater than sapling density using a paired t-test (p = 0.003).
Canopy Openness
In general, the light availability at the understory level in the transects was very low and variable. Across all the samples (n = 200), the canopy openness averaged 6.6 %, with a maximum of 30.2 % and a minimum of 2.3 %. In the vast majority of samples (95 %), the understory had less than 10 % openness, with most samples (81.1 %) being between 4 and 8 %. Only 1 % of the samples demonstrated characteristics typical of a forest gap, with canopy openness > 20 %. There were no significant differences in canopy openness among the samples from the ten sampled transects (Kruskal-Wallis test, α = 0.05, p = 0.700).
Agouti Activity
A total of 388 Brazil nut fruits that had been opened and gnawed by agoutis were found in the 10 transects, with 96 near the central lines of the transects and 292 in the 100 subplots. The estimated average agouti activity throughout all the transects was 11.1 ± 3.6 fruits opened and gnawed per ha (maximum 33.0 and minimum 1.0).
The estimated agouti activity was not statistically related to any independent variable, although there was a negative trend associated with time of human use (r = −0.620, R 2 = 0.275, p = 0.056).
Establishment of Small Plants (DBH < 10 cm) and Canopy Openness
There was no significant correlation, using Spearman’s correlation test, between the number of seedlings and canopy openness (r s = 0.009, p = 0.931) or between the number of saplings and canopy openness (r s = −0.122, p = 0.228). With respect to the sub-plots, 40 % had small plants (DBH < 10 cm), and this 40 % was further divided into 30 % with only seedlings, 8 % with only saplings, and 2 % with both plant types.
The majority of small plants (100 % of saplings and 76.4 % of seedlings) were found in sub-plots exposed to low light incidence (≤10 %; Fig. 2). Nevertheless, 15.9 % of seedlings were found in subplots with a relatively open understory (canopy openness > 20 %). No significant differences were found with respect to canopy openness between subplots with and without saplings/seedlings (K-S test, p = 0.269). However, 10 of the 20 saplings found along the central line of the transects occurred in a small patch of secondary forest in a single transect.
Regeneration Potential
Multiple regression analyses of seedling density (dependent variable) and eight independent variables (adult tree density, juvenile density, crown area, basal area, agouti activity, time of human use, distance of residence, and canopy openness) showed no significant results. When paired comparisons were made, the only significant correlation was a positive relationship between seedling density and time of human use, although this relationship had a weak R 2 value (0.40). The distance between the nearest human residence and agouti activity showed a weak negative tendency but was non-significant (Table 2).
Pre-established Regeneration
Multiple regression analysis was conducted for sapling density (dependent variable) and eight independent variables (adult tree density, juvenile density, crown area, basal area, agouti activity, time of human use, distance of residence and canopy openness); this analysis showed no significant results. Similarly, there were no significant results in paired comparisons (Table 2).
Established Regeneration
Multiple regression analysis was also performed for juvenile density (dependent variable) and the seven remaining independent predictive variables. The results of this multiple regression were not significant; thus, the data were subsequently analyzed using a paired correlation analysis. These analyses showed that established regeneration was correlated significantly and positively with time of human use, total crown area and adult tree density (Table 2). The partial correlation test revealed that the strongest correlation was found between juvenile density and adult density when controlling for time of human use (r = 0.69, R 2 = 0.48, p = 0.026). The distance between a Brazil nut stand and the nearest human residence showed a slight negative tendency, although it was statistically non-significant (Table 2).
Differences Between Moderate- and Intense-Use Brazil Nut Stands
Populations of B. excelsa in intense-use stands showed greater densities of trees with DBH 10–110 cm, i.e., adult trees (DBH > 40 cm), non-reproductive juveniles (DBH 10–40 cm), and saplings and seedlings (DBH < 10 cm), than moderate-use sites. In contrast, the sites that were considered moderate-use had a greater density only for the DBH > 110 cm size class. The basal area and total crown area were variables that did not show significant differences between the two types of stands (Table 3).
The diameter class distributions were significantly different between the two groups (K-S test, D = 0.415, p < 0.0001). In moderate-use sites, 56.5 % of trees were in the 60–140 cm DBH class, with a peak at 120–130 cm. Within this group, the three transects near streams had the highest concentration of trees in the intermediate DBH class (62.4 %, 100–150 cm DBH). In contrast, in intensive-use stands, the diameter class distribution had a higher concentration of smaller stems, with 65.4 % of trees showing DBH < 70 cm (Fig. 3).
With respect to ecological variables, canopy openness was nearly equal at all sites sampled, but the density of fruits opened and gnawed by agoutis was significantly higher in the moderate-use stands (Table 3). In relation to regeneration indicators, intensive-use stands showed higher values than moderate-use stands, although only juvenile density showed a significant difference (Table 3). The number of B. excelsa juveniles/adult trees was significantly higher in the intense-use stands than in the moderate-use stands (t-test, p = 0.010).
Discussion
Potential and Pre-established Regeneration
There are few demographic studies of B. excelsa that include the identification and quantification of seedlings in natural conditions. In the north of Bolivia, and in the indigenous reserve of Pinkaiti (Xingú River valley, Pará, Brazil), the estimated regeneration potentials (25–50 seedlings ha−1; Zuidema and Boot 2002; Baider 2000) were similar to those of the present study. In the Acre River valley, in contrast, the densities were considerably lower (3.2 to 5.8 seedlings ha−1; Wadt et al. 2008). Comparing the ratios between the number of seedlings and the number of adults, the sites with the highest proportion of seedlings were located in Bolivia and in the indigenous reserve of Pinkaiti (Xingú River valley). With the exception of the latter site, which is still largely unexplored, the research sites in Bolivia, Acre, and the present study are intensively used by the nearby communities. In the region of the Trombetas River, where tree stands are typically distant from human communities, the seedling density and the ratio of seedlings to adults was lower than in the Capanã Grande area (Scoles and Gribel 2011, 2012). For Ribeiro et al. (2014), the high seedling density in harvested sites in the forest area of the Kayapó territory in the southeastern region of the Amazon seems to be a consequence of seed dispersal by harvesters along trails. These data as a whole suggest that the accessibility of the stands to traditional communities does not negatively affect the regenerative potential of the B. excelsa population, despite the higher frequency of human visits and more intensive seed removal in these areas.
The establishment of seedlings in the Capanã Grande Lake region seems not to be as dependent on natural dispersion by agoutis as has been suggested by various studies (Peres and Baider 1997; Zuidema and Boot 2002). In fact, there appears to be no correlation between the number of seedlings and dispersal activities by agoutis; even if there were a correlation, it would be negative, as the stands with greater seedling densities had low densities of gnawed fruits. Moreover, 15.4 % of seedlings were identified near piles of fruits opened by harvesters, which points to a greater human influence on B. excelsa regeneration in the study area.
The average sapling density at Capanã Grande Lake was slightly higher than has been observed in other stands of Brazil nut trees with a history of extractive harvesting, such as Nova Esperança (2.7 saplings ha−1; Viana et al. 1998), Vale do Rio Acre (0.7–1.8 saplings ha−1; Wadt et al. 2008) and Trombetas River (1.0 ± 3.1 saplings ha−1, Scoles and Gribel 2012), despite the low density (<1 saplings ha−1) in five of the ten sample sites in the present study. There was an elevated sapling density in two intensive-use stands (Laranjal and Vale Quem Tem), both of which showed low agouti activity.
In this study, as in Nova Esperança, Acre (Viana et al. 1998), no evidence was found to support a higher density of seedling or saplings in understory areas with greater light availability (forest gaps). Nevertheless, 50 % of the saplings found near the central line were situated in a small area with characteristics of secondary forest (low canopy height, high-density understory), which probably had greater light availability in the recent past. At this same site, the saplings showed a growth form with multiple shoots growing from the same stump, which may be due to damage followed by resprouting or multiple germination from the same fruit, as has been previously observed in seedlings.
The lack of significant correlations found in the comparative analyses at Capanã Grande suggests that human activities have no negative effect on seedling establishment, despite the annual seed removal by fruit harvesters and the low agouti activity observed in the most-frequented stands.
Established Regeneration
Among significant correlations between established regeneration and potentially predictive variables, three variables showed a positive correlation (Table 3), namely 1) the time of human use; 2) the total crown area, an indirect indicator of Brazil nut tree productivity (Wadt et al. 2005; Tonini et al. 2008); and 3) the density of adult trees. The partial correlations show that adult tree density is the main predictive variable for established regeneration in the stands of Capanã Grande Lake. There is, therefore, no evidence that human extractive activities, including the harvest of seeds, have detrimental effects on the density of juveniles, in contrast to the conclusion of Peres et al. (2003) and the suggestions of Silvertown (2004)). Furthermore, the number of juveniles per adult tree was five times higher in the intense-use stands than in the moderate-use stands (0.5 and 0.1 juveniles adult−1, respectively; Table 3). This result suggests the possibility of positive feedback between human activity and B. excelsa regeneration, with the more frequented stands exhibiting faster rejuvenation and consequently being more productive and more frequently visited in the long term.
In denser stands, and especially in those more intensely used, the percentage of juveniles was high (21.2 to 40.5 %), with values that are higher than the average for the entire study area and that approach the values from areas of the Amazon with the highest regeneration rates (Peres et al. 2003; Scoles and Gribel 2012). A strong correlation between adult density and established regeneration has been documented for tropical trees (Ghazoul 2005). In absolute terms, Capanã Grande Lake presents the highest average juvenile density yet reported (3.2 juveniles ha−1), with values greater than at Pinkaiti (1.5 juveniles ha−1; Baider 2000) or at Nova Esperança, Acre (1.3 juveniles ha−1; Viana et al. 1998).
The greater density of juvenile and adult trees in the most frequented sites near human communities at Capanã Grande Lake suggests that traditional extractive activities and low-intensity disturbances in the understory caused by human presence in the forest may influence the demographic structure of Brazil nut tree stands. Similar situations were also described in Pereira (1994) at Tefé Lake, State of Amazonas, in Ribeiro et al. (2014) at Xingú Valley, State of Pará, in Sousa et al. (2014) at Caxiuanã National Forest, State of Pará, and at Sapucuá Lake, State of Pará (Scoles and Aragão unpublished data). In these regions, stands of Brazil nut trees under intensive forest management had younger B. excelsa populations, while Brazil nut stands in areas more distant from human communities, where the only activity was Brazil nut collection, showed a tendency towards a low proportion of young B. excelsa trees.
In sum, despite of the removal of seeds by harvesters, established regeneration appears to be guaranteed in the most-visited Brazil nut stands in the study area. In contrast, the transects with the greatest evidence of rodent activity and with less-frequent human presence (moderately used stands) exhibited much lower established regeneration.
Concluding Remarks
The demographic and ecological data collected from stands of B. excelsa in the Capanã Grande Lake area suggest that high regeneration rates of this species can occur in association with a high frequency of human activities. The statistical analysis suggests that the adult tree density of B. excelsa and the frequency of human presence in the stands positively influenced the regeneration of B. excelsa in the region. We hypothesize that unintentional dispersion of seeds by harvesters during collection, storage and transport and the long-term, low-intensity disturbances in the understory caused by human activities in the forest, such as clearing, trail opening, border disturbance, extractive removal of forest products, and selective logging, are likely factors that facilitate the regeneration of this heliophytic species, thereby compensating for the loss of seeds by harvesting. The well-recognized seed dispersal function of agoutis seems to be less important in stands with greater human presence. Therefore, the findings of this study suggest that the high frequency of human activity in stands of Brazil nut trees and in neighboring areas over prolonged periods likely explains the demographic structure, dominated by non-reproductive and young adult trees, observed in many B. excelsa stands throughout the Amazon (Pereira 1994; Paiva et al. 2011; Sousa et al. 2014; Scoles and Aragão unpublished data).
Finally, this study highlights the symbiotic relationship between Brazil nut trees and human populations in the Amazon region, supporting the idea, frequently suggested in the literature, that the current distribution of B. excelsa trees may be a consequence of the historical actions of Amerindian populations in the Amazonian forest (Ducke 1946; Müller et al. 1980; Posey 1985; Balée 1989; Scoles and Gribel 2011). This work also calls attention to the important role of contemporary traditional communities in the maintenance and rejuvenation of B. excelsa populations.
References
Allegretti, M. H. (1994). Reservas extrativistas: parâmetros para uma política de desenvolvimento sustentável na Amazônia. In Anderson, A. B., et al. (eds.), O Destino da Floresta: Reservas Extrativistas e Desenvolvimento Sustentável na Amazônia. Relume-Dumara, Rio de Janeiro, pp. 17–47.
Alvares, C. A., Stape, J. L., Sentelhas, P. C., Goncalves, J. L. M., and Sparovek, G. (2013). Köppen’s climate classification map for Brazil. Meteorologische Zeitschrift 22(6): 711–728.
Amin, M. M. O. (1997). O extrativismo como fator de empobrecimento da economia do Pará. In Ximenes, T. (Org.), Perspectivas de Desenvolvimento Sustentável (uma contribuição para Amazônia 21). UFPA/NAEA/UNAMAZ, Belém, Pará, Brasil, pp. 177–209.
Anderson, A. B. (1994). Extrativismo vegetal e Reservas Extrativistas: limitações e oportunidades. In Anderson, A. B., et al. (eds.), O Destino da Floresta: Reservas Extrativistas e Desenvolvimento Sustentável na Amazônia. Relume-Dumara, Rio de Janeiro, pp. 227–245.
Baider, C. (2000). Demografia e ecologia de dispersão de frutos de Bertholletia excelsa Humb. and Bonpl. (Lecythidaceae) em castanhais naturais silvestres da Amazônia Oriental. Ph.D. thesis. Instituto de Biociências da Universidade de São Paulo, São Paulo, Brazil.
Balée, W. (1989). The culture of Amazonian forests. In Posey, D. A., Balee, W. (eds.), Resource Management in Amazonia: Indigenous and Folk Strategies. New York Botanical Gardens, pp. 1–21.
Clay, J. W. (1997). Brazil nuts: the use of a keystone species for conservation and development. In Freese, C. H. (ed.), Harvesting Wild Species: Implications for Biodiversity and Conservation. John Hopkins University Press, Baltimore, pp. 246–282.
Clement, C. R. (2006). A lógica do mercado e o futuro da produção extrativista, in VI Simpósio Brasileiro de Etnobiologia e Etnoecologia, Sessão 5: O (neo) extrativismo é viável socioambientalmente? Sociedade Brasileira de Etnobiologia e Etnoecologia. Universidade Federal do Rio Grande do Sul, Porto Alegre, Brasil.
Costa, J. R., Castro, A. B. C., Wandelli, E. V., Coral, S. C. T., and Souza, S. A. G. (2009). Aspectos silviculturais da castanha-do-brasil (Bertholletia excelsa) em sistemas agroflorestais na Amazônia Central. Acta Amazonica 39(4): 843–850.
Ducke, A. (1946). Plantas de cultura precolombiana na Amazônia Brasileira. Notas sobre as espécies ou formas espontâneas que supostamente lhes teriam dado origem. Boletim Técnico do Instituto Agronómico do Norte 8: 3–24.
Engelbrecht, B. M. J., and Herz, H. M. (2001). Evaluation of different methods to estimate understory light conditions in tropical forest. Journal osf Tropical Forest 17: 207–224.
Fearnside, P. M. (1989). Extractive reserves in Brazilian Amazonia. An opportunity to maintain forest under sustainable use. Bioscience 39: 387–393.
Fernandes, N. P., and Alencar, J. C. (1993). Desenvolvimento de árvores nativas em ensaios de espécies. 4. Castanha-do-brasil (Bertholletia excelsa H. B. K.), dez anos após o plantio. Acta Amazonica 23(2–3): 191–198.
Ghazoul, J. (2005). Pollen and seed dispersal among dispersed plants. Biological Reviews 80: 413–443.
Homma, A. K. O. (1993). Extrativismo Vegetal na Amazônia: Limites e Oportunidades. Embrapa, Centro de Pesquisas Agropecuárias do Tropico Úmido.
Huber, J. (1910). Mattas e madeiras amazônicas. Boletim do Museu Paraense de História Natural 6: 91–225.
[IBCE] Instituto Boliviano de Comercio Exterior 2010 (14 January 2014; http://www.ibce.do.bov).
[ICMBIO] Instituto Chico Mendes de Conservação da Biodiversidade (2013). Plano de Manejo Participativa da Reserva Extrativista do Lago Capanã Grande. ICMBIO, Manicoré.
Kainer, K. A., Duryea, M. L., Costa de Macedo, N., and Williams, K. (1998). Brazil nut seedling establishment and autoecology in an extractive reserve in Acre, Brazil. Ecological Applications 8(2): 397–410.
Kainer, K. A., Wadt, L. H. O., and Staudhammer, C. L. (2007). Explaining variation in Brazil nut fruit production. Forest Ecology and Management 250(2007): 244–255.
Martorano, L. G., Nechet, D., Manzatto, C. V., Rebello, E. R. G., and Bertolossi, R. (2004). Pluviometric variations as subsidiary information for agricultural planning in the Amazon. ISCO 2004—13th International Soil Conservation Organization Conference – Brisbane, July 2004, paper 781, pp. 1–4.
Mori, S. A. (1992). The Brazil-nut industry: past, present and future. In Plotkin, M., and Famolare, L. (eds.), Sustainable Harvest and Marketing of Rain Forest Products. Island Press, Washington, D.C, pp. 241–251.
Mori, S. A., and Prance, G. T. (1990). Taxonomy, ecology, and economy botany of Brazil nut (Bertholletia excelsa Humb. e Bonpl.: Lecythidaceae). Advances in Economic Botany 8: 130–150.
Müller, C. H., Rodrigues, I. A., Müller, A. A., and Müller, N. R. M. (1980). Castanha do Brasil: Resultados de Pesquisas. EMBRAPA, Centro de Pesquisas Agropecuária do Trópico Úmido, Belém.
Myers, G., Newton, A. C., and Melgarejo, O. (2000). The influence of canopy gap size on natural regeneration of Brazil nut (Bertholletia excelsa) in Bolivia. Forest Ecology and Management 127: 119–128.
Nepstad, D., Brown, I., Luz, L., Alechandre, A., and Viana, V. (1992). Biotic impoverishment of Amazonian forests by rubber tappers, loggers, and cattle ranchers. In Nepstad, D., and Schwartzman, S. (eds.), Non-Timber Products from Tropical Forests Evaluation of Conservation and Development Strategy. Advances in Economic Botany 9. New York Botanic Garden, New York, pp. 1–14.
Nicotra, A. B., Chazdon, R. B., and Iriarte, S. V. B. (1999). Spatial heterogeneity of light and woody seedling regeneration in tropical wet forest. Ecology 80(6): 1908–1926.
Ortiz, E. G. (1995). Survival in a nutshell. The Americas 47: 7–12.
Ortiz, E. G. (2002). Brazil nuts (Bertholletia excelsa). In Shanley, P., Pierce, A. R., Laird, S. A., and Guillen, A. (eds.), Tapping the Green Market: Certification and Management of Non-timber Forest Products. Earthsan Publications Ltd, London, pp. 61–74.
Paiva, P. M., Guedes, M. C., and Funi, C. (2011). Brazil nut conservation through shifting cultivation. Forest Ecology and Management 261: 508–514.
Peña-Claros, M. R., Boot, G. A., Dorado-Lora, J., and Zonta, A. (2002). Enrichment planning of Bertholletia excelsa in secondary forest in the Bolivian Amazon: effect of cutting line width on survival, growth and crown traits. Forest Ecology and Management 161: 159–168.
Pereira, H. S. (1994). Manejo agroflorestal da castanheira (Bertholletia excelsa H.B.K.) na região do Lago do Tefé (AM). Rev. U.A. Série Ciências Agrárias 3: 11–32.
Peres, C. A., and Baider, C. (1997). Seed dispersal, spatial distribution and population structure of Brazilnut trees (Bertholletia excelsa) in southeastern Amazonia. Journal of Tropical Ecology 13: 595–616.
Peres, C. A., Baider, C., Zuidema, P. A., Wadt, L. H. O., Kainer, K. A., Gomes-Silva, D. A. P., Salomão, R. P., Simões, L. L., Franciosi, E. R. N., Cornejo Valverde, F., Gribel, R., Shepard Jr., G. H., Kanashiro, M., Coventry, P., Yu, D. W., Watkinson, A. R., and Freckleton, R. P. (2003). Demographic threats to the sustainability of Brazil nut exploitation. Science 302: 2112–2114.
Posey, D. A. (1985). Indigenous management of tropical forest ecosystems: the case of Kayapó Indians of the Brazilian Amazon. Agroforestry Systems 3: 139–158.
RADAMBRASIL (1978). Folha SB.20 Purus: Geologia, Geomorfologia, Pedologia, Vegetação, uso Potencial da Terra. Departamento Nacional da Produção Mineral, Rio de Janeiro.
Ribeiro, M. B. N. (2011). Ecologia, manejo e sustentabilidade da exploração da castanha-da-Amazônia (Bertholletia excelsa) pelos índios Kayapó, sudeste da Amazônia. PhD Thesis. Instituto Nacional de Pesquisas da Amazônia, Manaus, Brazil.
Ribeiro, M. B. N., Jerozolimski, A., Robert, P., and Magnusson, W. E. (2014). Brazil nut stock and harvesting at different spatial scales in southeastern Amazonia. Forest Ecology and Management 319: 67–74.
Ruiz-Pérez, M., Belcher, B., Achdiawan, R., Alexiades, M., Aubertin, C., Caballero, J., Campbell, B., Clement, C., Cunningham, T., Fantini, A., Foresta, H., García Fernández, C., Gautam, K. H., Martínez, P. H., Jong, W., Kusters, K., Kutty, M. G., López, C., Fu, M., Martínez Alfaro, M. A., Nair, T. K. R., Ndoye, O., Ocampo, R., Rai, N., Ricker, M., Schreckenberg, K., Shackleton, S., Shanley, P., Sunderland, T., and Youn, Y. (2004). Markets drive the specialization strategies of forest peoples. Ecology and Society 9(2): 4.
Salomão, R. P. (1991). Estrutura e densidade de Bertholletia excelsa H. e B. (‘Castanheira’) nas regiões de Carajás, e Marabá, Estado do Para. Boletim do Museu Paraense Emilio Goeldi Serie Botânica 7: 47–68.
Salomão, R. P. (2009). Densidade, estrutura e distribuição espacial da castanheira-do-brasil (Bertholletia excelsa H. and B.) em dois platôs de floresta ombrófila densa na Amazônia setentrional brasileira. Boletim do Museu Paraense Emilio Goeldi Ciências Naturais 4: 11–25.
Scoles, R., and Gribel, R. (2011). Population structure of Brazil nut (Bertholletia excelsa, Lecythidaceae) stands in two areas with different occupation histories in the Brazilian Amazon. Human Ecology 39: 455–464.
Scoles, R., and Gribel, R. (2012). The regeneration of Brazil nut trees in relation to nut harvest intensity in the Trombetas River valley of Northern Amazonia, Brazil. Forest Ecology and Management 265: 71–81.
Scoles, R., Klein, G. N., and Gribel, R. (2011). Crescimento e sobrevivência de Bertholletia excelsa Bonpl. (castanheira) em diferentes condições ambientais na região do rio Trombetas, Oriximiná, Pará. Boletim Museu Paraense Emílio Goeldi. Ciencias Naturais 6(3): 273–293.
Scoles, R., Klein, G. N., and Gribel, R. (2014). Crescimento e sobrevivência de castanheira (Bertholletia excelsa Bonpl., Lecythidaceae) plantada em diferentes condições de luminosidade após seis anos de plantio na região do rio Trombetas, Oriximiná, Pará, Boletim Museu Paraense Emílio Goeldi. Ciencias Naturais 9(2): 321–336.
Silvertown, J. (2004). Sustainability in a nutshell. TRENDS in Ecology and Evolution 19(6): 276–277.
Sousa, D. G., Almeida, S. S., and Amaral, D. D. (2014). Estrutura de uma população manejada da castanheira (Bertholletia excelsa) na Floresta Nacional de Caxiuanã, Pará. Boletim Museu Paraense Emílio Goeldi. Ciencias Naturais 9(2): 353–370.
Souza, C. R., Lima, R. M. B., Azevedo, C. P., and Rossi, L. M. B. (2008). Desempenho de espécies florestais de uso múltiplo. Scientia Forestalis 36(77): 7–14.
Stoian, D. (2004). Cosechando lo que cae: la economía de la castaña (Bertholletia excelsa H.B.K.) en la Amazonía boliviana. In Alexiades, M. N., and Shanley, P. (eds.), Productos Forestales, Medios de Subsistencia y Conservación: Estudios de Caso Sobre Sistemas de Manejo de Productos Forestales no Maderables, vol. 3. CIFOR, América Latina, pp. 89–116.
Swaine, M. D., and Whitmore, T. C. (1988). On the definition of ecological species groups in tropical rain forest. Vegetatio 75: 81–86.
Tonini, H., Kaminski, P. E., and Costa, P. (2008). Relação da produção de sementes de castanha-do-brasil com características morfométricas da copa e índice de competição. Pesquisa Agropecuária Brasileira 43: 1509–1516.
Tuck Haugaasen, J. M., Haugaasen, T., Peres, C. A., Gribel, R., and Wegge, P. (2010). Brazil nut seed dispersal by scatter-hoarding rodents in a central Amazonian forest. Journal of Tropical Ecology 26: 251–262.
Tuck Haugaasen, J. M., Haugaasen, T., Peres, C. A., Gribel, R., and Wegge, P. (2012). Fruit removal and natural seed dispersal of the Brazil nut tree (Bertholletia excelsa) in Central Amazonia, Brazil. Biotropica 44(2): 205–210.
Viana, V. M., Mello, R. A., Moraes, L. M., and Mendes, N. T. (1998). Ecologia e manejo de populações de castanha-do-Pará em reservas extrativistas Xapurí, Estado do Acre. In Gascon, C., and Mountinho, P (eds.), Floresta Amazônica: Dinâmica, Regeneração e Manejo. Instituto Nacional de Pesquisas da Amazônia, pp. 277–292.
Wadt, L. H. O., Kainer, K. A., and Gomes-Silva, D. A. P. (2005). Population structure and nut yield of a Bertholletia excelsa stand in Southwestern Amazonia. Forest Ecology and Management 211: 371–384.
Wadt, L. H. O., Kainer, K. A., Staudhammer, C. L., and Serrano, R. O. P. (2008). Sustainable forest use in Brazilian extractives reserve: natural regeneration of Brazil in exploited populations. Biological Conservation 141: 332–346.
Zar, J. H. (1999). Biostatical Analysis, 4th edn. Prentice Hall.
Zuidema, P. A., and Boot, R. G. A. (2002). Demography of the Brazil nut tree (Bertholletia excelsa) in the Bolivian Amazon: impact of seed extraction on recruitment and population dynamics. Journal of Tropical Ecology 18: 1–31.
Acknowledgments
The authors thank the following entities for financial and logistic support: Programa de Áreas Protegidas da Amazônia (ARPA), Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Programa Beca of the Instituto Internacional de Educação do Brasil and the Projeto ‘Banco do Germoplasma da Castanheira’ of INPA/ MRN / ICMBio. Research grant to RG was provided by CNPq, proccess no. 308137/2011-6. We thank Salvador Pueyo and David Bertran for suggestions related to the data analysis and Paulo M. Alencastro Graça for designing the map. We also thank two anonymous reviewers for constructive comments on an earlier version of this manuscript. Our most sincere thanks to all the families of the communities involved in the fieldwork done for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scoles, R., Gribel, R. Human Influence on the Regeneration of the Brazil Nut Tree (Bertholletia excelsa Bonpl., Lecythidaceae) at Capanã Grande Lake, Manicoré, Amazonas, Brazil. Hum Ecol 43, 843–854 (2015). https://doi.org/10.1007/s10745-015-9795-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10745-015-9795-4