Abstract
We have designed a cell culture system for thoracic neurons of adult Locusta migratoria that enables the establishment of functional synapses in vitro. Patch-clamp recordings revealed three different neuron classes. About half of the neurons (47%) had unexcitable somata with outward and no inward conductance. The other half generated either single (37%) or multiple action potentials (18%) and differed mainly in lower outward conductance. Selectively stained motor neurons were analyzed to demonstrate varied physiological properties due to culture conditions. Using paired patch clamp recordings we demonstrate directly synaptic transmission in morphologically connected neurons in vitro. Presynaptic stimulation resulted in postsynaptic potentials in 42 pairs of neurons tested, independent of the type of neuron. According to pharmacological experiments most of these synapses were either glutamatergic or GABAergic. In addition to these chemical synapses, electrical synapses were found. With the demonstration of synapse formation in cell culture of adult locust neurons, this study provides the basis for the future analysis of more defined insect neuronal circuits in culture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Locusts are an established animal model to study neuroethological phenomena, the function of neuroendocrine cells, and the function of sensory systems. In the locust nervous system, many neurons can be individually identified, a prerequisite for a detailed analysis of particular neurons (Hoyle and Burrows 1973; Comer and Robertson 2001). Most valuable aspects of how the locust central nervous system generates behavior have been gained over the years (for review see Burrows 1996), but we are still a long way from understanding this ‘‘simple’’ nervous system. Potential interactions of neurons inflicting or masking neuronal activity are challenging studies in vivo. To reduce complexity for electrophysiological investigations one may use semi-intact preparations, isolated ganglia, or primary cell culture.
Since the first studies of locust neurons in culture (Giles and Usherwood 1985a, b), most studies concentrated on the influence of culture media on survival, growth and morphology of the cultured neurons (Pfahlert and Lakes-Harlan 1997; Vanhems et al. 1990; Kirchhof and Bicker 1992), or they study particular neurons with regard to ionic currents or receptors for neuroactive agents (Beadle 2006; Heck et al. 2009; Pfahlert and Lakes-Harlan 2008; Rössler and Bickmeyer 1993; Benkenstein et al. 1999; Brone et al. 2003; Laurent et al. 1993; Heidel and Pflüger 2006). A few attempts were made to analyze neuronal networks in vitro on the basis of morphology and histochemistry (Shefi et al. 2002; Melville et al. 2003; Torkkeli et al. 2005; Shefi et al. 2005; Anava et al. 2009a). The formation of functional synapses was demonstrated in neuronal cultures of honeybees, the fruit fly, crickets and the locust frontal ganglion (Lee and O’Dowd 1999; Lee et al. 2003; Su and O’Dowd 2003; Küppers-Munther et al. 2004; Fuchs et al. 2007; Reska et al. 2008; Oh et al. 2008).
Perhaps the most ambitious goal for an in vitro approach is to establish a functional network consisting of identified neurons of known in vivo connectivity. For such an approach neurons would have to be identified, transferred individually to the culture system, induced to grow out neurites, and to establish functional synaptic connections. Next to protocols for long-term culture, methods for labeling neuron populations prior to isolation have been established (Kloppenburg and Hörner 1998; Heidel and Pflüger 2006; Pfahlert and Lakes-Harlan 2008). First attempts to grow neurons on multi-electrode arrays for non-invasive long-term recording (Fuchs et al. 2007; Greenbaum et al. 2009) as well as techniques for guiding neurites by patterning the substrate yielded promising results (Reska et al. 2008; Göbbels et al. 2010).
In this study we show that locust neurons can also establish functional synapses in vitro.
For this, we developed a cell culture system for adult locusts neurons based on published protocols for primary neuronal cell culture and we evaluated the morphology and the electrophysiological properties on single cell level. We recorded from identified motor neurons, which allowed us a rough comparison with published in vivo data. By pair-wise patch-clamp recordings we show chemical as well as electrical synaptic connections. Using transmitter antagonists we evaluated the chemical synapses with respect to the transmitters glutamate and γ-aminobutyric acid (GABA).
Methods
Animals
In all experiments adult male and female Locusta migratoria were used during the first week after their final moult. The animals were reared in crowded breeding colonies. They were kept in a 12 h/12 h dark-light cycle and fed with wheat seedlings and wheat bran.
Preparation
Prior to dissection the insects were kept in a plastic tube and chilled on ice until they did not show movement anymore. Legs, head, the abdominal segments and the intestine were removed. Two parasagittal longitudinal sections isolated the mid-sternal region containing the thoracic ganglia. Mounted dorsal side up in a wax-coated dish, the preparation was submerged in ice-cold medium (modified Leibovitz L-15 medium, see below). The meso- and metathoracic ganglia were isolated and fixed in a wax-coated dish. All following steps were performed under sterile conditions. After removal of tracheae and fatty tissue, ganglia were washed three times in fresh medium and the neurolemma was removed with fine forceps. The cell dissociation and culture was performed as follows (modified from Kirchhof and Bicker 1992; Lapied et al. 1993; Smith and Howes 1996): the medium was exchanged with enzyme solution (2 mg/ml dispase (Gibco) dissolved in Hank`s Balanced Salt solution; all chemicals were obtained from Sigma if not stated otherwise) for 30 min at 29°C. The enzyme treatment was stopped by washing (three times) with ice cold medium.
Ganglia of two locusts were pooled, transferred to reaction vessels (2 ml) by pipettes (200 μl, both Biozym Scientific GmbH, Germany) and dissociated by trituration. Both, reaction vessels and pipettes, were siliconized to avoid cell loss. The cell suspension was subsequently centrifuged (400 g for 2 min). The supernatant was partly removed and the cells were resuspended in the remaining volume (200–400 μl). Cells were plated on pretreated coverslips (50 μl cell suspension/cover slip containing typically 100–200 neurons) and they were subsequently allowed to adhere to the surface for 1 h at 29°C. After gently adding 3 ml pre-warmed medium to each culture dish, neurons were incubated at 29°C and 95% humidity. Every third day half of the medium was exchanged with fresh medium.
Retrograde staining of motor neurons
In one set of experiments subsets of neurons were labeled by retrograde staining before cell isolation (Kloppenburg and Hörner 1998; Heidel and Pflüger 2006; Pfahlert and Lakes-Harlan 2008). Thoracic ganglia were dissected as described above, but care was taken that the main leg nerve (nerve 5) of the metathoracic ganglion remained as long as possible. The distal stumps of right and left nerves were isolated in small Vaseline troughs, their ends were cut and immersed in buffer solution (internal solution, see “Electrophysiology”). In order to osmotically open axons, troughs were filled with distilled water for 1 min. Subsequently, dye solution was applied (1% (w/v) dextran tetramethylrhodamine, 3,000 MW, Molecular Probes, Eugene, OR, USA in buffer solution) over night at 4°C. The anterior lateral ventral parts of the ganglia that contain the motor neuron somata innervating the hind leg muscles (Hoyle and Burrows 1973) were isolated and cultured as described above.
Surface pretreatment
Glass cover slips (Ø 12 mm; VWR International GmbH, Germany) were cleaned and made hydrophilic by treatment with 20% H2SO4 (20 min at 80°C). After extensive rinsing with purified water (MilliQ water, 18 MΩ) they were disinfected in 70% analytical grade ethanol washed again twice in MilliQ water to remove ethanol residues, and dried. A 50 μl drop of concanavaline A solution (Con A, 200 μg/ml in distilled water) was placed on each cover slip and allowed to react for 1 h at room temperature. Afterwards the cover slips were washed twice with MilliQ water and dried again.
Culture medium
The cell culture medium was Leibovitz L-15 medium (Leibovitz 1963), which was supplemented with proline (35 mg/ml), imidazole (6 mg/ml), l-glutamine (0.292 mg/ml), and HEPES (6 mg/ml). A mixture of penicilline (100 units/ml) and streptomycine (100 μg/ml) as well as fungizone (0.25 μg/ml, Invitrogene) and a combination of the neurotrophic factors insulin (5 μg/ml) and 20-hydroxyecdysone (20-HE, 5 μg/ml) was added. Osmolarity and pH were adjusted (~390 mOsmol/kg by equal shares of glucose/fructose and pH 7.0 by NaOH).
Electrophysiology
Before whole-cell patch-clamp experiments, culture medium was replaced by normal salt solution (NSS) containing (in mg/ml): 8.766 NaCl, 0.373 KCl, 0.222 CaCl2 and 2.383 HEPES (~390 mOsm/kg adjusted by equal fractions of glucose/fructose and pH 7.0 adjusted by NaOH).
Electrodes were pulled from borosilicate glass (o.d. 1.5 mm, i.d. 0.86 mm; Sutter Instrument Co., Novato, USA) using a Flaming/Brown micropipette puller (P-97; Sutter Instruments Co.) resulting in a resistance of 2–11 MΩ. They were filled with internal solution containing (in mg/ml): 0.292 NaCl, 11.183 KCl, 0.011 CaCl2, 0.190 MgCl2, 1.102 Na2-ATP, 0.380 EGTA and 10 HEPES (~390 mOsm/kg adjusted by equal fractions glucose/fructose and pH 7.0 adjusted by NaOH).
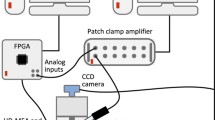
Whole-cell patch-clamp recordings were made using an EPC9/3 amplifier controlled by TIDA 5.x software (both HEKA Elektronik, Germany). The data were sampled with 25 kHz and lowpass-filtered at 10 kHz with a Bessel filter. The liquid junction potential was calculated using JPCalcW 1.0 software (P.H. Barry, University of New South Wales, Sydney, Australia and Axon Instruments Inc. California, USA) to be at most 3.7 mV and, hence, was not taken into account in our experiments.
Series resistance and capacities were measured and compensated with the internal routine of the patch-clamp amplifier. Data were discarded if series resistance was higher than 25 MOhm and a ratio of membrane resistance to series resistance was lower ten.
We considered only recordings from neurons with a resting membrane potential VR less than −35 mV and leak conductivity S Leak below 3 nS.
Electrophysiological experiments were performed under optical control (Olympus IX 50 inverted microscope equipped with epifluorescence and phase-contrast). To identify rhodamine labelled motor neurons, a mercury short arc lamp (HBO 50, Osram) was used (excitation: 546 nm, emission 575 nm). All experiments were performed at room temperature.
Pharmacology
For pharmacological experiments either the whole bath solution was exchanged by test solution or—in case of recordings from connected neurons—cells were perfused using a voltage controlled perfusion system (DAD-8VC; ALA scientific instruments, Inc.). Command voltages were applied by a voltage output channel of the ECP-9/3 amplifier and controlled by the TIDA software. An air pressure of 3.5–7 Pa produced a constant solution stream, which was set near the cells by the manifold (200 μm diameter). Test solutions were based on NSS by substituting glucose/fructose with the particular substances. The following reagents and concentrations were used: glutamate (100 μM), 2-amino-5-phosphonovalerate (AP-5, 100 μM), 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX, 5 μM), GABA (50 or 100 μM) and Picrotoxin (PTX, 100 nM).
Statistical analysis
Quantified results are expressed as mean ± standard deviation and assessed for significance using the unpaired Student’s t-test. Significance level was assigned at a probability p < 0.05.
Results
Morphology
Freshly dissociated neurons adhered within the first 2 h on the substrates. After 4 days in vitro (DIV) the neurons were differentiated and showed distinct morphologies, which remained almost unchanged for the rest of the culture period (up to 4 weeks). Typically neurite lengths ranged between twofold and fivefold of the soma diameter which was 10–120 μm. Several morphological types of neurons were observed (Fig. 1), which we classified as unipolar, bipolar and multipolar. Unipolar neurons were characterized by a single long primary neurite with branchings at its distal end (Fig. 1a). Bipolar neurons (Fig. 1b) typically possessed two neurites. When compared to those of the unipolar neurons these neurites were shorter and much stouter. Multipolar neurons (Fig. 1c) had numerous delicate neurites that formed a corona around the soma. Neurons with larger soma diameter tended to develop fewer and shorter neurites. Axons and dendrites could not be identified by morphological criteria. Neurites of adjacent neurons appeared often to form small morphologically interconnected networks of two to three neurons (Fig. 1d). We studied the electrophysiological parameters of the cells typically between DIV 2 and DIV 14.
Electrophysiology of unidentified thoracic neurons
Since we were recording between DIV 2 and 14, we initially checked for age-dependent effects. We noticed only slightly more hyperpolarized VR (DIV2 −39.4 ± 4.5 n = 34; DIV8 −41.3 ± 6.6 n = 8) and no further obvious effects on the distribution of response types.
The electrophysiological properties of cultured locust neurons were characterized by whole cell patch-clamp recordings at their soma. During current clamp recordings a holding current between −50 and −100 pA was applied resulting in a membrane potential (Vmem) of about −70 mV. Stimulation currents (Istim) between −200 pA and 1 nA (in 100 pA steps) were applied for 100 ms and the resulting voltage changes were monitored. Under these conditions three classes of neurons with different types of responses to the stimulus were observed: tonic firing neurons (TFN; 18%), phasic firing neurons producing just a single spike (PFN; 35%) and non-firing neurons (NFN; 47%; Fig. 2). Spontaneous activity was very rarely recorded. These physiological types did not correlate with the different cellular morphologies described above. TFN neurons fired bursts of action potentials (APs) when stimulated by current pulses (iThres = 4.5 ± 4.0 nA/mm2) with a spike frequencies ranging from 20 to 270 Hz. PFN neurons fired one AP (iThres = 11.8 ± 5.5 nA/mm2) even when depolarized with current pulses of increasing strength, or with current pulses over a prolonged time. The single AP of PFN neurons was occasionally followed by voltage oscillations. NFN neurons never generated APs. The membrane time constants τ did not differ significantly between the three types (TFN: 47 ± 29 ms; PFN: 27 ± 19 ms; NFN: 28 ± 13 ms; analyzed for n = 5 for each class).
Whole-cell patch-clamp recordings (current-clamp mode) of three exemplary neurons. A 200 pA stimulation current elicited a graded potential at the membrane of non-firing neuron (a). Phasic firing neurons (b) were able to generate one AP followed by sustained depolarization. In a few cases oscillation after the spike occurred. Tonic firing neurons (c) showed a series of APs during the depolarising pulse
Current density versus voltage relations were obtained from voltage-clamp recordings. Starting from a holding potential VH = −70 mV cells were hyperpolarized and depolarized in 10 mV steps (Fig. 3) and the sustained outward and peak inward currents were measured. The membrane currents were normalized to the membrane surface area assuming a specific membrane capacity of 1 μF/cm2 and the specific conductivities were calculated. The three classes of neurons differed in membrane conductivities (Fig. 3): TFN neurons and PFN showed similar maximal inward conductivities, while NFN showed marginal inward conductivities at VH ≥ 0 mV (Fig. 3a). The threshold voltage for eliciting inward directed ionic currents was lower for TFN neurons (VH ≥ −60 mV) than for PFN (VH ≥ −50 mV). The conductivities showed saturation with a maximum of about 0.3 μS/mm2.
Specific conductivity of neuron classes TFN, PFN, NFN. Data were averaged from five neurons arbitrarily chosen per class. See text for more details. The curves of different neurons were slightly shifted to show the standard deviation. a Non-firing neurons (NFN) showed no or even a low inward conductivity at Vm more positive than 0 mV (+70 mV than VH). In neurons, which were able to generate action potentials (APs), inward directed channels opened due to depolarizations above a certain threshold (Phasic firing neurons (PFN): −50 mV [+20 mV above VH]; Tonic firing neurons (TFN): −60 mV [+10 mV above VH]). Resulting conductivities reached a maximum of 0.7μS/mm2 (PFN: at VH = −20 mV; TFN: at VH = −30 mV) and decreased than slowly. b The threshold of outward directed ionic conductivities was at VH = −30 mV. Conductivities increased linear with depolarization. NFN showed larger outward conductivities (6.9 + −2.9 μS/mm2) than PFN (4.9 + −3.9 μS/mm2) and TFN (3.0 + −2.7 μS/mm2). The value of NFN and TFN differed significantly at VH = +70 mV
The sustained outward conductivities increased with depolarization above a threshold VH = −30 mV (Fig. 3b). This effect was significantly different between the three classes of neurons (p < 0.05 at VH + 70 mV). TFN showed the smallest specific outward conductivities gout compared to PFN and NFN (Table 1). The differences in these three classes of neurons were not related to cell size and electrophysiological state as VR, Cm and A were all in a normal range and did not differ significantly.
Electrophysiology of stained thoracic motor neurons
In order to determine whether the three physiological classes reflect different types of neurons or are artifacts caused by isolation and culture, defined neurons had to be cultured and to be examined with respect to their in vivo properties. For this, motor neurons innervating the hind legs were retrogradely labeled and cultured separately. All recorded motor neurons exhibited electrically active somata and generated single or multiple spikes in response to current injection. We saw a similar amount of PFN (3 out of 8) and TFN neurons (5 out of 8) but no NFN were found. Again, these neurons showed no significant differences in VR and Cm (Table 2).
Neuronal networks
When recording from single cultured cells, postsynaptic activity was often observed. For further evaluation of synapse formation in cell culture, pairs of morphologically connected neurons (see Fig. 1d) were investigated by simultaneous patch-clamp recordings (Vogt et al. 2003). Here, either Vmem of both neurons were simultaneously recorded when one of the neurons was stimulated by a current pulse or ionic currents when stimulated by voltage changes. Synaptic connectivity was studied by direct correlation of presynaptic and postsynaptic events. In 42 experiments correlated activity was observed. Here, we found chemical (n = 38) and electrical synapses (n = 4), which were discriminated by bidirectionality and signal transmission during hyperpolarization.
Chemical synapses
Typical current-clamp recordings of two synaptically connected neurons are shown in Fig. 4.
Simultaneous patch-clamp recording of two morphologically connected neurons (see inset in a). Stimulation of the first neuron (upper trace, a) with a 200pA depolarising current elicited three APs in this cell and correlated postsynaptic potentials in the second neuron (middle trace, a), while stimulating the second neuron (middle trace, b) did not influence the first neuron (upper trace, b). Hyperpolarization had no effect in any case
In this example, stimulation of one neuron resulted in graded postsynaptic potentials (PSPs) correlated in time to presynaptic APs (Fig. 4a). We also observed summation of PSPs during this synaptic activity (1st and 2nd AP). Their amplitudes were up to 10 mV. Latencies between the onset of presynaptic spikes and postsynaptic PSPs were around 1 ms. Hyperpolarizing presynaptic stimuli did not affect the postsynaptic neuron. Stimulation of the second neuron failed to elicit a response in the adjacent neuron (Fig. 4b).
In 47% of the investigated networks the presynaptic neuron was not spiking. In such cases signal transmission was observed by depolarizing the presynaptic neuron above a certain threshold without recording any presynaptic APs. The current-clamp recording shown in Fig. 5a did not exhibit APs in the stimulated neurons but postsynaptic potential changes occurred. The presynaptic neurons had to be depolarized to a threshold of around −35 mV. Further presynaptic depolarization increased the amplitude of postsynaptic potentials only within a small range before saturation occurred. The number and amplitude of postsynaptic events was not correlated to the presynaptic depolarization (Fig. 5a, d). As characteristic for chemical transmission, neither presynaptic hyperpolarization (Fig. 5a) nor stimulation of the second neuron triggered postsynaptic events (Fig. 5b).
Signal transmission between a pair of two non-spiking neurons (c). Depolarising current injections led to graded potentials in the first neuron (upper trace, a). These triggered postsynaptic potentials in the second neuron (middle trace, a). Neither stimulation of the second neuron (middle trace, b) nor hyperpolarization evoked a postsynaptic response (upper trace, b). d Determination of synaptic trigger threshold. The maximal amplitude of postsynaptic potentials was registered in dependency on the associated maximal presynaptic membrane potential. Passing a threshold of ~−35 mV led to postsynaptic potentials in a range of 2 mV up to 20 mV saturating at further presynaptic depolarization to more than 0 mV
Determination of neurotransmitters
To further elucidate the nature of synaptic transmission we tested the networks for the two prominent transmitters in thoracic ganglia of insects (Watson 1988; Watson and Laurent 1990; Watson and Schürmann 2002): glutamate and γ-aminobutyric acid (GABA). First, the neurons were perfused with the transmitter itself and subsequently treated with their respective blockers (6-cyano-7-nitriquino-2,3-dione (CNQX) and (1)-2-amino-5-phosphonopentanoic acid (AP-5) for glutamate; picrotoxin (PTX) for GABA while holding them at resting potential.
In case of glutamate, all 20 tested neurons responded to a concentration of 100 μM in various fashions. Responses consisted of a transient hyperpolarization followed by sustained depolarization (Fig. 6a, n = 1), transient depolarization (Fig. 6b, n = 11), sustained hyperpolarization (Fig. 6c, n = 7), or only a transient hyperpolarization (n = 1). Adding glutamate to a spontaneously active neuron increased the spike rate rapidly followed by a slow adaptation to the sustained stimulus (Fig. 6d). Concentrations of 5 μM CNQX or 100 μM AP-5 were determined to be sufficient to block such reactions (Fig. 6e, f). Determining reversal potentials (~+35 mV) gave evidence for cationic conductances triggered by glutamate (Fig. 6g).
Responses of neurons to glutamate. Glutamate elicited a variety of electrical responses like biphasic response with a transient hyperpolarization followed by a sustained depolarization (a), a transient depolarization (b) or a transient hyperpolarization followed by a sustained hyperpolarization (c). Application to a spontaneously active neuron increases the spike frequency (d). The glutamate responses could be blocked by AP-5 (e) and CNQX (f). Reversal potential (~+35 mV) indicates the involvement of cationic currents (g; n = 4)
In case of GABA all neurons responded with a hyperpolarization starting with a transient component during application of 50 or 100 μM GABA (Fig. 7a, n = 8). GABA was also able to completely abolish spontaneous activity (Fig. 7b). PTX at a concentration of 100 μM blocked these responses (Fig. 7c). A reversal potential for responses induced by GABA of ~5 mV (Fig. 7d) corresponds fairly well to the calculated Nernst potential for chloride ions (0.07 mV) under our conditions.
Responses of neurons to GABA. GABA hyperpolarized all tested cells (a) and blocked spiking in spontaneous active neurons (b). The response was blocked by PTX (c). Current/voltage relation revealed a reversal potential around +5 mV, which is near to the calculated reversal potential of Cl− (d; n = 4)
During paired recordings of neuronal networks applying the GABA blocker PTX (100 nM) lead to a complete inhibition of postsynaptic currents (n = 8), which was partially reversible (Fig. 8). In these experiments, neurons were also sensitive to stimulation with GABA. In three experiments neurons were sensitive to GABA and glutamate. Perfusion with AP-5/CNQX led to variable responses: either an increase of the number of PSPs or an increase of their amplitude or a slight reduction of their amplitude. Complete inhibition of synaptic transmission by AP-5/CNQX was not observed. In one experiment the neuron was neither sensitive to GABA nor glutamate and tested neurotransmitter antagonist did not affect the synaptic transmission.
Electrical synapses
In addition to signal transmission by transmitter release, electrical synapses were found in four experiments. To discriminate them from chemical synapses we tested for a short synaptic delay, bidirectionality and transmission of hyperpolarizing stimuli. Figure 9 shows simultaneous patch-clamp recordings of two neurons connected by an electrical synapse recorded in current-clamp mode. Current pulses between −200 pA and 1 nA were applied. Dependent on the stimulus amplitude, the stimulated cell reacted with graded voltage changes of variable amplitude. The second cell reacted in a similar fashion with potential changes of lower amplitude and without any time delay. Even hyperpolarizing stimuli led to postsynaptic hyperpolarizations (Fig. 9a). If the second cell was stimulated, the first one reacted as described before indicating bidirectional signal transmission between the two cells (Fig. 9b).
Whole-cell current clamp recording of an electrical synapse between a pair of nonspiking neurons (c). Stimulation of each of the neurons (upper trace, a and middle trace, b) led to postsynaptic potentials in the other cell even to hyperpolarising currents with no time delay (middle traces, a and upper trace, b). At higher presynaptic potentials the postsynaptic cells showed voltage-dependent rectifying behaviour. d Diagram of the normalised transjunctional conductance GJ. It is calculated according to Barrio et al. (1991)
In none of the observed electrical synapses the form of the presynaptic signal was mirrored exactly by the postsynaptic signal. In three experiments neurons in the networks were nonspikers with at least an overshooting peak. In one case the presynaptic neuron was a single spiker. Neither the AP nor the height of the presynaptic response was reflected by the postsynaptic response. The amplitude of the postsynaptic depolarization remained unchanged beyond a certain presynaptic potential level. This voltage-dependent rectifying effect is illustrated in Fig. 9d. The conductance of the gap junctions (gj) was calculated from the current/voltage relations monitored in voltage-clamp mode according to Barrio et al. (1991). The junctional conductance gJ was normalized to the extrapolated value at which the transjunctional voltage difference (VJ) was zero. Since both neurons were clamped to a Vmem of −70 mV VJ was zero at this potential.
Negative transjunctional voltages did not affect GJ (normalized gj) whereas voltage steps more positive than 20 mV (equivalent to a presynaptic Vmem = −50 mV) decreased the conductance of the gap junction until GJ ~ 0.3 (VJ = 220 mV).
Discussion
During the last two decades, the use of cell culture systems in insect neuroscience has increased rapidly. Cell cultures offer the possibility to characterize the properties of neurons while they are detached from their complex natural environment. Particular effects of culture parameters, transmitters or drugs can be studied by various techniques in cell cultures.
We are interested in the analysis of synaptic transmission between interconnected locust neurons on the level of single cells. Therefore, we tested and combined previous cell culture protocols for adult locust neurons to facilitate neuronal re-differentiation to functional networks. The neuronal characteristics were evaluated morphologically as well as electrophysiologically. In most of our experiments, a mixture of thoracic neurons was cultured and analyzed. Isolation procedure and culture conditions might affect different neuron types differently. Therefore, we also cultured a specific subpopulation for comparison. This also allows a rough comparison of their firing patterns under culture conditions with in vivo data. Morphologically connected neurons were tested for synaptic connectivity by double patch-clamp experiments. The existence of non-neuronal cells in our culture system was demonstrated previously using a special histological regime (Loesel et al. 2006; Gocht et al. 2009). However, the influence of non-neuronal cells onto the properties of cultured neurons was not investigated here.
Morphology
Neurons were dissociated from thoracic ganglia of adult locusts by enzymatic and mechanical treatment. Due to this preparation method, the neurons loose axons and dendrites and they retain at most stumps of their primary neurites of variable lengths. The neurons differentiated into three different morphological types: unipolar, bipolar, and multipolar. Unipolar neurons are typical for the insect CNS in vivo, bipolar and multipolar neurons are only known from the peripheral nervous system, with the bipolar morphology being characteristic for ciliary receptor cells. Thus bipolar and multipolar morphologies in the culture of central neurons must have been acquired during regeneration by formerly unipolar neurons. In our study a mixed population of thoracic neurons was cultured. So, a correlation of the morphology in culture and in vivo is not possible. Previous studies reported different findings on morphological changes in culture. For cultured thoracic interneurons and motorneurons a multipolar appearance was reported, which differed from the in vivo morphology (Pfahlert and Lakes-Harlan 2008). Only for cockroach dorsal unpaired neurons (DUM; Lapied et al. 1993) morphologies similar to those in vivo were described but Newland et al. (1993) demonstrated that these neurons in vitro may also display various morphologies that differ from their in vivo pattern. Kirchhof and Bicker (1992) have shown that the degree of axonal regeneration and cellular morphology of locust thoracic neurons in vitro was influenced by the length of the primary neurite stump which remains attached to the cell body after the dissociation process. When a long primary neurite stump remained this appeared to promote unipolar cell morphology, while short neurite stumps fostered bipolar or multipolar morphologies. So, the morphologies displayed in this and previous studies most likely reflect effects of the isolation procedures and do not correlate with particular neuron types (Pfahlert and Lakes-Harlan 2008; Göbbels and Bräunig, unpublished observations).
Electrophysiology
The recordings from the somata of cultured locust neurons revealed three types of neurons with different electrophysiological responses: tonic firing neurons (TFN), phasic firing neurons (PFN) and non-firing neurons (NFN) (Fig. 2). These three response types did not correlate with soma size and morphology. They were also not due to insufficient cell culture conditions (e.g. hypoxia) as VR, Cm and A were all in a similar range (see Table 1). The observed VR of the cultured neurons were in a similar range to previous findings under comparable culture conditions (Giles and Usherwood 1985b; Pfahlert and Lakes-Harlan 2008) as well to those in vivo (Goodman and Heitler 1979). Differences were found in the inward and outward ion conductivities. NFN neurons showed only marginal inward directed currents unlike neurons generating APs. PFN and TFN neurons had similar inward conductivities but the onset potential was slightly more positive for PFN. NFN exhibited largest outward conductivities gout, PFN intermediate and TFN lowest (Fig. 3b).
Surprisingly a large proportion of recorded neurons (53%) showed soma spikes and many neurons showed no spikes (47%). It is known from studies in vivo that the membrane of the soma is unable to generate APs in the great majority of all locust neurons. About 38% of locust neurons are unable to generate any AP and about 60% have spike generating zones at the distal end of their primary neurite only (for review see Burrows 1996). In locust thoracic ganglia, only a small fraction of neurons—mostly efferent neuromodulatory dorsal unpaired median neurons and other neurosecretory cells—generate soma spikes in vivo (Bräunig and Pflüger 2001; Bräunig, unpublished).
We can imagine different reasons for the observed distribution: (1) a change in the cellular properties due to isolation and culture, (2) our preparation favors the survival of particular neurons, (3) space clamp issues prevent the recording of APs at the soma. For this, we cultured identified motor neurons in a subset of experiments. Thus we could test to what extent they resemble neurons in vivo or whether they alter their properties. All of the recorded identified neurons showed either phasic or tonic firing. This phenomenon is similar to those described by Goodman and Heitler (1979) after axotomy in vivo: all somata were excitable meaning that a conversion from non-spiking properties to spiking properties had occurred.
A similar change in excitability was shown for cockroach motor neurons a few hours after isolation (Hancox and Pitman 1995) and for cricket giant interneurons (Kloppenburg and Hörner 1998).
In vivo studies of Goodman and Heitler (1979) on the non-spiking soma of the FETi motor neuron and Thomas (1984) on identified cockroach motor neurons show that the balancing of Na+ and K+ currents is important for the excitability of cells. After axotomy by isolation, neosynthesis of sodium channels at the soma or in newly generated processes close to it (Tribut et al. 1991; Kloppenburg and Hörner 1998) might disturb the balance resulting in higher excitability.
Interestingly, studies of non-spiking local interneurons in vivo (Laurent 1990) and in vitro (Laurent 1991; Laurent et al. 1993) showed no evidence for a change of membrane excitability and channel composition following culturing.
We found just slightly different values and onsets of inward conductivities for TFN and PFN and nearly no inward conductivities for NFN. Apart from probable newly synthesized Na+ channels we observed differences in the amount of outward directed currents which in turn could cause the different excitabilities (highest for NFN, lowest for TFN). A knockdown of potassium channel expression changed excitabilities of drosophila motor neurons from a phasic to a tonic firing behavior (Duch et al. 2008).
We believe that the distribution of different excitabilities is caused by a change of the amount of expressed ion channels in culture and not due to a favor for particular neurons or space clamp problems. Neurons, which were able to generate APs in vivo display now phasic or tonic firing patterns, while non-spiking neurons presumably keep their non-spiking behavior. Extensive branching might compromise voltage control of distance neurites leading to a misinterpretation in the firing pattern (Grunewald and Levine 1998; Bar-Yehuda and Korngreen 2008; Spruston et al. 1993). However, we did not find NFN in recordings of identified neurons. Thus it is unlikely, that the occurrence of NFN is caused by inappropriate voltage-control.
Neuronal networks
Neuronal cell cultures allow the study of the properties of neurons outside their complex natural environment, such that synaptic interactions between neurons of interest or complex processes like learning can be analyzed. In the last decades, many studies demonstrated the formation of neuronal connections in cultures of insect neurons on the basis of morphology (Shefi et al. 2002; Shefi et al. 2005; Küppers-Munther et al. 2004; Reska et al. 2008), characterization of transmitter receptors or postsynaptic events (Oh et al. 2008; Lee and O’Dowd 1999; Küppers-Munther et al. 2004; Beadle 2006). One study was showing synaptic connections by paired patch-clamp recordings in crickets (Reska et al. 2008).
Here, we demonstrate formation of synapses in cultures of locust neurons by direct correlation between presynaptic stimulation and postsynaptic events using paired patch recordings and tested the influence of the neurotransmitters GABA and glutamate. In most cases pairs of morphologically connected neurons were chosen, which had no contact to further cells. Simultaneous patch-clamp recordings showed synapses between two neurons in 42 experiments. Graded postsynaptic potentials and currents were recorded as a result of presynaptic stimulation mediated either by chemical or electrical synapses. Locusts have many neurons not capable to generate APs (NFN: ~35% in vivo estimated from Burrows (1996); ~47% in our culture). These nonspiking interneurons play in vivo an important role e. g. in motor networks (Burrows 1980; Burrows and Siegler 1978). Signal transmission is thus not inevitably dependent on presynaptic APs. We demonstrated postsynaptic events correlated with presynaptic AP and with presynaptic graded potentials. Immunohistochemical studies in the metathoracic ganglion of locusts showed that 35% of the neurons contain GABA and 20% contain glutamate (Watson 1988; Watson et al. 1993). This immunoreactivity is retained in culture (Pfahlert and Lakes-Harlan 2008). Thus it seemed reasonable to investigate whether chemical synapses use one or the other of these two transmitters. Our results show that GABA always elicited hyperpolarizing responses, which were probably chloride-driven and could be blocked by PTX.
The response to glutamate was more variable. We observed transient hyperpolarizations or depolarizations, biphasic responses or sustained hyperpolarizations. Obvious excitatory effects were measured, when flushing spontaneous active neurons. Even if the resting potential, at which the data were obtained, varied, the responses suggested the existence of more than one type of glutamate receptor. It was reported in previous work that insect neurons can exhibit glutamate-gated chloride channels and cationic channels (Sattelle 1992; Wafford et al. 1992; Raymond et al. 2000).
Ionic currents triggered by the application of the transmitter could be blocked by their particular antagonists. However, in recordings from neuron doublets postsynaptic answers could only be suppressed by the application of PTX and not by CNQX/AP-5, giving evidence for GABA-ergic transmission in these cases. Nevertheless several glutamate receptors are reported, which are insensitive to CNQX (Sattelle 1992) or which are even sensitive to PTX (Raymond et al. 2000; Janssen et al. 2007).
In addition to glutamate and GABA, acetylcholine (ACh) is an important transmitter in the insect nervous system. Even isolated thoracic neurons are sensitive to ACh application (Suter and Usherwood 1985; Benson 1992; Jackson et al. 2002). However, the ACh-receptor distribution in the thoracic ganglia is mainly restricted to neuropile regions receiving (mechano-)sensory input (Leitch et al. 1993). For mechanosensory transduction the role of ACh was demonstrated (Gauglitz and Pflüger 2001; Parker and Newland 1995). However, we have no sensory neurons in our culture due to the isolation procedure. To our knowledge, there is no evidence for cholinergic transmission between non-sensory neurons. Antisera directed against cholineacetyltransferase detected only very few immunoreactive somata in thoracic ganglia (Lutz and Tyrer 1987). Thus, we did not check for acetylcholine as transmitter. Since we also did not evaluate other neurotransmitters e.g. serotonine, final conclusion on all transmitters involved is not possible at present.
Evidence for gap junctions in the locust nervous system is sparse (O’Shea and Rowell 1976; Watson and Schürmann 2002; Anava et al. 2009b; Ganfornina et al. 1999). They are described in vivo in Drosophila (Trimarchi and Murphey 1997; Wu et al. 2011; Yaksi and Wilson 2010; Phelan et al. 2008) as well as in neuronal cultures (Oh et al. 2008).
In the present work, we found gap junctions in four cases. They were distinguished from chemical synapses by three criteria: no synaptic delay, bidirectionality and transmission of hyperpolarizing stimuli. All electrical synapses observed in the present work showed rectifying behavior to depolarizing stimuli (Fig. 9d).
In this work we present a proof of concept for analyzing small locust neuronal networks in culture. Neurons can be cultured over several weeks and they stay electrically active. The electrophysiological properties of the neurons may change due to isolation and cultivation. The most important finding of the present study is that neurons obtained from adult locusts regenerate neurites in culture and establish functional synaptic connections under cell culture conditions.
Together with recent work on neuronal patterning (Reska et al. 2008; Göbbels et al. 2010), demonstrating that guided growth in extremely low cell density cultures is possible, and work on neuron-electrode interfacing (Greenbaum et al. 2009) we believe, that cell cultures of locust neurons are a promising tool for the understanding of network activity. A next step would be to isolate identified neurons and look whether they re-establish functional connections that are known to exist in vivo.
References
Anava S, Greenbaum A, Ben Jacob E, Hanein Y, Ayali A (2009a) The regulative role of neurite mechanical tension in network development. Biophys J 96(4):1661–1670. doi:10.1016/j.bpj.2008.10.058
Anava S, Rand D, Zilberstein Y, Ayali A (2009b) Innexin genes and gap junction proteins in the locust frontal ganglion. Insect Biochem Mol Biol 39(3):224–233. doi:10.1016/j.ibmb.2008.12.002
Barrio LC, Suchyna T, Bargiello T, Xu LX, Roginski RS, Bennett MV, Nicholson BJ (1991) Gap junctions formed by connexins 26 and 32 alone and in combination are differently affected by applied voltage. Proc Natl Acad Sci USA 88(19):8410
Bar-Yehuda D, Korngreen A (2008) Space-clamp problems when voltage clamping neurons expressing voltage-gated conductances. J Neurophysiol 99(3):1127–1136. doi:10.1152/jn.01232.2007
Beadle DJ (2006) Insect neuronal cultures: an experimental vehicle for studies of physiology, pharmacology and cell interactions. Invert Neurosci 6(3):95–103. doi:10.1007/s10158-006-0024-0
Benkenstein C, Schmidt M, Gewecke M (1999) Voltage-activated whole-cell K + currents in lamina cells of the desert locust schistocerca gregaria. J Exp Biol 202(Pt 14):1939
Benson JA (1992) Electrophysiological pharmacology of the nicotinic and muscarinic cholinergic responses of isolated neuronal somata from locust thoracic ganglia. J Exp Biol 170:31
Bräunig P, Pflüger HJ (2001) The unpaired median neurons of insects. Adv Insect Physiol 28
Brone B, Tytgat J, Wang DC, Van KE (2003) Characterization of Na(+) currents in isolated dorsal unpaired median neurons of Locusta migratoria and effect of the alpha-like scorpion toxin BmK M1. J Insect Physiol 49(2):171
Burrows M (1980) The control of sets of motoneurones by local interneurones in the locust. J Physiol 298:213–233
Burrows M (1996) The neurobiology of an insect brain. Oxford University Press, Oxford
Burrows M, Siegler MV (1978) Graded synaptic transmission between local interneurones and motor neurones in the metathoracic ganglion of the locust. J Physiol 285:231–255
Comer CM, Robertson RM (2001) Identified nerve cells and insect behavior. Prog Neurobiol 63(4):409–439
Duch C, Vonhoff F, Ryglewski S (2008) Dendrite elongation and dendritic branching are affected separately by different forms of intrinsic motoneuron excitability. J Neurophysiol 100(5):2525–2536. doi:10.1152/jn.90758.2008
Fuchs E, Ayali A, Robinson A, Hulata E, Ben-Jacob E (2007) Coemergence of regularity and complexity during neural network development. Dev Neurobiol 67(13):1802–1814. doi:10.1002/dneu.20557
Ganfornina MD, Sanchez D, Herrera M, Bastiani MJ (1999) Developmental expression and molecular characterization of two gap junction channel proteins expressed during embryogenesis in the grasshopper Schistocerca americana. Dev Genet 24(1–2):137–150. doi:10.1002/(SICI)1520-6408(1999)24:1/2<137:AID-DVG13>3.0.CO;2-7
Gauglitz S, Pflüger HJ (2001) Cholinergic transmission via central synapses in the locust nervous system. J Comp Physiol A 187(10):825–836
Giles D, Usherwood PN (1985a) The effects of putative amino acid neurotransmitters on somata isolated from neurons of the locust central nervous system. Comp Biochem Physiol, C: Comp Pharmacol Toxicol 80(2):231–236
Giles DP, Usherwood PN (1985b) Locust nymphal neurones in culture: a new technique for studying the physiology and pharmacology of insect central neurones. Comput Biochem Physiol C 80(1):53
Göbbels K, Thiebes AL, van Ooyen A, Schnakenberg U, Bräunig P (2010) Low density cell culture of locust neurons in closed-channel microfluidic devices. J Insect Physiol 56(8):1003–1009. doi:10.1016/j.jinsphys.2010.05.017
Gocht D, Wagner S, Heinrich R (2009) Recognition, presence, and survival of locust central nervous glia in situ and in vitro. Microsc Res Tech 72(5):385–397. doi:10.1002/jemt.20683
Goodman CS, Heitler WJ (1979) Electrical properties of insect neurones with spiking and non-spiking somata: normal, axotomized, and colchicine-treated neurones. J Exp Biol 83:95
Greenbaum A, Anava S, Ayali A, Shein M, David-Pur M, Ben-Jacob E, Hanein Y (2009) One-to-one neuron-electrode interfacing. J Neurosci Methods 182(2):219–224. doi:10.1016/j.jneumeth.2009.06.012
Grünewald B, Levine RB (1998) Ecdysteroid control of ionic current development in Manduca sexta motoneurons. J Neurobiol 37(2):211–223
Hancox JC, Pitman RM (1995) Spontaneous bursting induced by convulsant agents in an identified insect neurone. Gen Pharmacol 26(1):195–204
Heck C, Kunst M, Härtel K, Hülsmann S, Heinrich R (2009) In vivo labeling and in vitro characterisation of central complex neurons involved in the control of sound production. J Neurosci Methods 183(2):202–212. doi:10.1016/j.jneumeth.2009.06.032
Heidel E, Pflüger HJ (2006) Ion currents and spiking properties of identified subtypes of locust octopaminergic dorsal unpaired median neurons. Eur J Neurosci 23(5):1189–1206. doi:10.1111/j.1460-9568.2006.04655.x
Hoyle G, Burrows M (1973) Neural mechanisms underlying behavior in the locust Schistocerca gregaria. I. Physiology of identified motorneurons in the metathoracic ganglion. J Neurobiol 4(1):3
Jackson C, Bermudez I, Beadle DJ (2002) Pharmacological properties of nicotinic acetylcholine receptors in isolated Locusta migratoria neurones. Microsc Res Tech 56(4):249–255. doi:10.1002/jemt.10028
Janssen D, Derst C, Buckinx R, Van den Eynden J, Rigo JM, Van Kerkhove E (2007) Dorsal unpaired median neurons of Locusta migratoria express ivermectin- and fipronil-sensitive glutamate-gated chloride channels. J Neurophysiol 97(4):2642–2650. doi:10.1152/jn.01234.2006
Kirchhof B, Bicker G (1992) Growth properties of larval and adult locust neurons in primary cell culture. J Comput Neurol 323(3):411
Kloppenburg P, Hörner M (1998) Voltage-activated currents in identified giant interneurons isolated from adult crickets Gryllus bimaculatus. J Exp Biol 201(17):2529
Küppers-Munther B, Letzkus JJ, Lüer K, Technau G, Schmidt H, Prokop A (2004) A new culturing strategy optimises Drosophila primary cell cultures for structural and functional analyses. Dev Biol 269(2):459
Lapied B, Tribut F, Sinakevitch I, Hue B, Beadle DJ (1993) Neurite regeneration of long-term cultured adult insect neurosecretory cells identified as DUM neurons. Tissue Cell 6(25):893
Laurent G (1990) Voltage-dependent nonlinearities in the membrane of locust nonspiking local interneurons, and their significance for synaptic integration. J Neurosci 10(7):2268
Laurent G (1991) Evidence for voltage-activated outward currents in the neuropilar membrane of locust nonspiking local interneurons. J Neurosci 11(6):1713
Laurent G, Seymour-Laurent KJ, Johnson K (1993) Dendritic excitability and a voltage-gated calcium current in locust nonspiking local interneurons. J Neurophysiol 69(5):1484–1498
Lee D, O’Dowd DK (1999) Fast excitatory synaptic transmission mediated by nicotinic acetylcholine receptors in Drosophila neurons. J Neurosci 19(13):5311
Lee D, Su H, O’Dowd DK (2003) GABA receptors containing Rdl subunits mediate fast inhibitory synaptic transmission in Drosophila neurons. J Neurosci 23(11):4625
Leibovitz A (1963) The growth and maintainance of tissue-cell cultures in free gas exchange with the atmosphere. Am J Hyg 78:173
Leitch B, Watkins BL, Burrows M (1993) Distribution of acetylcholine receptors in the central nervous system of adult locusts. J Comput Neurol 334(1):47–58. doi:10.1002/cne.903340104
Loesel R, Weigel S, Bräunig P (2006) A simple fluorescent double staining method for distinguishing neuronal from non-neuronal cells in the insect central nervous system. J Neurosci Methods 155(2):202–206. doi:10.1016/j.jneumeth.2006.01.006
Lutz EM, Tyrer NM (1987) Immunohistochemical localization of choline acetyltransferase in the central nervous system of the locust. Brain Res 407(1):173–179
Melville JM, Hoffman KL, Jarrard HE, Weeks JC (2003) Cell culture of mechanoreceptor neurons innervating proleg sensory hairs in Manduca sexta larvae, and co-culture with target motoneurons. Cell Tissue Res 311(1):117
Newland PL, Smith PJS, Howes EA (1993) Regenerating adult cockroach dorsal unpaired median neurons in vitro retain their in vivo membrane characteristics. J Exp Biol 179(1):323–329
Oh HW, Campusano JM, Hilgenberg LG, Sun X, Smith MA, O’Dowd DK (2008) Ultrastructural analysis of chemical synapses and gap junctions between Drosophila brain neurons in culture. Dev Neurobiol 68(3):281–294. doi:10.1002/dneu.20575
O’Shea M, Rowell CH (1976) The neuronal basis of a sensory analyser, the acridid movement detector system. II. response decrement, convergence, and the nature of the excitatory afferents to the fan-like dendrites of the LGMD. J Exp Biol 65(2):289–308
Parker D, Newland PL (1995) Cholinergic synaptic transmission between proprioceptive afferents and a hind leg motor neuron in the locust. J Neurophysiol 73(2):586–594
Pfahlert C, Lakes-Harlan R (1997) Responses of insect neurones to neurotrophic factors in vitro. Die Naturwissenschaften (84):163–165
Pfahlert C, Lakes-Harlan R (2008) Interneurons, motoneurons and sensory neurons of Locusta migratoria (Insecta: Orthoptera) in primary cell culture. Open J Entomol (2):6–13
Phelan P, Goulding LA, Tam JL, Allen MJ, Dawber RJ, Davies JA, Bacon JP (2008) Molecular mechanism of rectification at identified electrical synapses in the Drosophila giant fiber system. Curr Biol 18(24):1955–1960. doi:10.1016/j.cub.2008.10.067
Raymond V, Sattelle DB, Lapied B (2000) Co-existence in DUM neurones of two GluCl channels that differ in their picrotoxin sensitivity. NeuroReport 11(12):2695–2701
Reska A, Kuenzel T, Gasteier P, Schulte P, Moeller M, Offenhäusser A, Groll J (2008) Ultrathin coatings with change of reactivity over time enable functional in vitro networks of insect neurons. Adv Mater 20(14):2751–2755
Rossler W, Bickmeyer U (1993) Locust medial neurosecretory cells in vitro: morphology, electrophysiological properties and effects of temperature. J Exp Biol 183(1):323
Sattelle DB (1992) Receptors for L-glutamate and GABA in the nervous system of an insect (Periplaneta americana). Comp Biochem Physiol, C: Comp Pharmacol Toxicol 103(3):429–438
Shefi O, Golding I, Segev R, Ben-Jacob E, Ayali A (2002) Morphological characterization of in vitro neuronal networks. Phys Rev E Stat Nonlin Soft Matter Phys 66(2 Pt 1):021905
Shefi O, Golebowicz S, Ben-Jacob E, Ayali A (2005) A two-phase growth strategy in cultured neuronal networks as reflected by the distribution of neurite branching angles. J Neurobiol 62(3):361–368. doi:10.1002/neu.20108
Smith PJ, Howes EA (1996) Long-term culture of fully differentiated adult insect neurons. J Neurosci Methods 69(1):113
Spruston N, Jaffe DB, Williams SH, Johnston D (1993) Voltage- and space-clamp errors associated with the measurement of electrotonically remote synaptic events. J Neurophysiol 70(2):781–802
Su H, O’Dowd DK (2003) Fast synaptic currents in Drosophila mushroom body Kenyon cells are mediated by alpha-bungarotoxin-sensitive nicotinic acetylcholine receptors and picrotoxin-sensitive GABA receptors. J Neurosci 23(27):9246
Suter C, Usherwood PN (1985) Action of acetylcholine and antagonists on somata isolated from locust central neurons. Comp Biochem Physiol, C: Comp Pharmacol Toxicol 80(2):221–229
Thomas MV (1984) Voltage-clamp analysis of a calcium-mediated potassium conductance in cockroach (Periplaneta americana) central neurones. J Physiol 350:159–178
Torkkeli PH, Widmer A, Meisner S (2005) Expression of muscarinic acetylcholine receptors and choline acetyltransferase enzyme in cultured antennal sensory neurons and non-neural cells of the developing moth Manduca sexta. J Neurobiol 62(3):316–329. doi:10.1002/neu.20097
Tribut F, Lapied B, Duval A, Pelhate M (1991) A neosynthesis of sodium channels is involved in the evolution of the sodium current in isolated adult DUM neurons. Pflugers Arch 419(6):665–667
Trimarchi JR, Murphey RK (1997) The shaking-B2 mutation disrupts electrical synapses in a flight circuit in adult Drosophila. J Neurosci 17(12):4700–4710
Vanhems E, Delbos M, Girardie J (1990) Insulin and Neuroparsin promote neurite outgrowth in cultured locust CNS. Eur J Neurosci 2(9):776
Vogt AK, Lauer L, Knoll W, Offenhäusser A (2003) Micropatterned substrates for the growth of functional neuronal networks of defined geometry. Biotechnol Prog 19(5):1562
Wafford KA, Bai D, Sattelle DB (1992) A novel kainate receptor in the insect nervous system. Neurosci Lett 141(2):273–276
Watson AH (1988) Antibodies against GABA and glutamate label neurons with morphologically distinct synaptic vesicles in the locust central nervous system. Neuroscience 26(1):33–44
Watson AH, Laurent G (1990) GABA-like immunoreactivity in a population of locust intersegmental interneurones and their inputs. J Comput Neurol 302(4):761–767. doi:10.1002/cne.903020408
Watson AH, Schürmann FW (2002) Synaptic structure, distribution, and circuitry in the central nervous system of the locust and related insects. Microsc Res Tech 56(3):210
Watson AH, Burrows M, Leitch B (1993) GABA-immunoreactivity in processes presynaptic to the terminals of afferents from a locust leg proprioceptor. J Neurocytol 22(7):547–557
Wu CL, Shih MF, Lai JS, Yang HT, Turner GC, Chen L, Chiang AS (2011) Heterotypic gap junctions between two neurons in the drosophila brain are critical for memory. Curr Biol 21(10):848–854. doi:10.1016/j.cub.2011.02.041
Yaksi E, Wilson RI (2010) Electrical coupling between olfactory glomeruli. Neuron 67(6):1034–1047. doi:10.1016/j.neuron.2010.08.041
Acknowledgments
This work was in part supported by the EC project CICADA of the IST Programme FET Key Action Life Like Perception funded in FP 5. We are very grateful to M. Knierim-Grenzebach for her help and advice with neuronal cell culture. We thank A. Reska for her initial work in the early phase of this project and M. Gebhardt and K. Göbbels for critical comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weigel, S., Schulte, P., Meffert, S. et al. Locust primary neuronal culture for the study of synaptic transmission. J Mol Hist 43, 405–419 (2012). https://doi.org/10.1007/s10735-012-9395-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-012-9395-1