Abstract
Brassinosteroids (BRs) are steroid hormones that are essential for plant growth and development. These hormones control the division, elongation and differentiation of various cell types throughout the entire plant life cycle. Over the past few decades, studies on BRs caught the attention of plant scientists due to their versatile ability in mitigating various environmental stresses. Additionally, BR also involved in maintaining the quality of postharvest produces, by enhancing their resistance against abiotic and biotic stress. Furthermore, BRs are non-toxic and eco-friendly; this aids its importance in coping with adverse environmental conditions without disturbing the balance of the ecosystem. Our review summarized the structural characteristic and distribution of BRs in plants, role in postharvest technology, biotic stress tolerance, improving resistance against pesticide, organic pollutant toxicity and nodule formation and mycorrhization. This review provides useful information on BRs and its effects on plant system that we believe could be useful in maintaining environmental sustainability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Brassinosteroid (BRs) are endogenous phytohormones that promote plant growth by regulating various physiological process including male sterility and the timing of senescence (Bajguz 2007). BRs were first extracted from the pollen of rape plant and since then their role in plant physiology has been studied extensively (Grove et al. 1979). The phytohormone has also been detected in flower buds, pollens, fruit, seeds and leaves (Bajguz and Piotrowska-Niczyporuk 2014). These compounds exist in their conjugated form with fatty acids and sugar, in a free form or as glucosides and sulphates (Hayat et al. 2003; Bajguz and Hayat 2009; Fahad et al. 2014, 2016a, b, c, d) and are highly important for normal plant development (González-García et al. 2011). However, these phytohormones are especially important in situations when plants are exposed to environmental stresses such as water-limiting conditions (Farnham 2001; Thelen 2006), plant defoliation (Battaglia et al. 2018, 2019a, b) and low soil fertility (Shapiro and Wortmann 2006; Barbieri et al. 2008; Kaya et al. 2015, 2018; Sonmez et al. 2009a, b), among others. When this occurs, plants respond to environmental stresses by switching from growth activation activities to suppressing adverse conditions (Bechtold and Field 2018; Feng et al. 2016) involving the use of hormones such as abscisic acid (ABA) (Yoshida et al. 2014; Zhu et al. 2017). However, evidence suggests that BRs may also play a role in controlling the balance between growth under normal and stressed conditions by regulating the expression of stress specific transcript machineries (Ye et al. 2017), activation of antioxidant enzymes (Kim et al. 2012; Lima and Lobato 2017; Tunc-Ozdemir and Jones 2017; Zou et al. 2018; Fahad et al. 2014, 2016a, b, c, d) and production of osmoprotectants (Fàbregas et al. 2018; Fahad et al. 2014, 2016a, b, c, d). Other reviews highlighted the importance of BRs in plant growth, crosstalk with other phytohormones and plant tolerance to abiotic stress in details (Fariduddin et al. 2014; Rajewska et al. 2016; Anwar et al. 2018; Banerjee and Roy Choudhury 2018; Fahad et al. 2014, 2016a, b, c, d). This review aimed to underline (a) the extent and mechanisms of BRs in structural characteristic and distribution of BRs in plants, postharvest biotechnology and resistance to biotic stress and, (b) the mechanism regarding organic pollutant, increase resistance to pesticide and effects on nodule formation and mycorrhization.
The occurrence of brassinosteroid in plants
The action mode of hormones or ‘phytohormones’, as commonly referred in the literature, implies a complex communication mechanism by which chemical messages are dispatched from cell to cell in order to generate a given response. Hormones are naturally occurring molecules that influence various functions in plants across different developmental stages (Bajguz 2007). With the progression of knowledge in plant science, a series of phytohormones were discovered and characterized, including ethylene, gibberellins (GA), cytokinins, ABA, and auxin (Piotrowska and Bajguz 2011; Arteca 2013; Moore 2012; Kumar et al. 2014). Brassinosteroids are polyhydroxylated sterol hormones representing a sixth class of plant hormones (Ali 2017; Fahad et al. 2014, 2016a, b, c, d). This class of hormone comprises more than 70 steroids with varying structural and physiological activities (Sadura and Janeczko 2014). BRs have revealed a great structural similarity with animal and insects’ steroidal hormones called ecdysteroids (Thummel and Chory 2002). Even though the physiological roles of BRs in plants are not well known, they are considered to have key roles in a wide spectrum of plant growth regulatory mechanisms (Alabadí and Blázquez 2009; Bajguz and Hayat 2009; Fahad et al. 2014, 2016a, b, c, d). In the early 1960s, Mitchell and his team, working at USDA Agricultural Research Center in Maryland, conducted novel research with this phytohormone. In their study, pollen crude extracts from around 60 species were tested as growth enhancer in Phaseolus vulgaris L., a plant compound that is now known considered to be an excellent source of plant growth-regulating compounds (Bajguz and Tretyn 2003a, b). Results showed that around half of the tested pollen extracts resulted in a significant enhancement of vegetative growth of Phaseolus vulgaris L. From these results, investigators ascribed this higher activity to a brand-new class of steroidal hormones that they called brassinosteroid. In 1972, Mitchell and his team observed that brassinosteroid were helpful in the enhancement of seed vigor and crop yield (Mitchell and Gregory 1972). However, Millborrow and Pryce (1973) proposed that the hormonal activity of pollen could be due to the other compounds such as gibberellins and other phytohormones. To clarify this ambiguity, about 225 kg of pollen from rapeseed (Brassica napus L.), was collected, processed to obtain an active crystalline material, and samples analyzed. Results showed that the presence of brassinolide (BL), an active constituent of brassinosteroid (Grove et al. 1979). BRs are also known for influencing stem elongation, cell division, seed growth, leaf development, disease resistance, xylem differentiation, photomorphogenesis, and leaf senescence and ameliorating response to stresses such as drought, salt, extreme temperatures, and others (Javid et al. 2011; Sasse 2003; Fariduddin et al. 2014). When the amount of BRs is naturally low or null, as occurs with some mutant plants, a series of vegetative and reproductive defects, including slower growth, dwarf phenotypes, photomorphogenesis in the dark, changed stomatal expansion and reduced male fertility can take place in the plant (Clouse et al. 1996; Szekeres et al. 1996). BRs have attracted huge attention since last decade due to its esteem-understood importance for plant development and a possible tool for plant improvement.
Recently, the knowledge regarding the biosynthesis and mode of action of BRs have been greatly advanced with the help of molecular and genetic techniques (Camoni et al. 2018; Ogweno et al. 2010; Singh and Shono 2005). All BRs can be distributed into three main categories based on carbon-number of each steroid molecule (i.e., C27, C28 and C29) (Fujioka and Yokota 2003). Besides grain pollen, the phytohormone can be found in roots, seeds, stems, leaves, and flowers (Bajguz and Tretyn 2003a, b). Young growing tissues like pollen contain larger amounts of BRs, ranging between 1 and 100 μg kg−1), compared to mature tissues such as shoots and leaves (range 0.01–0.1 μg kg−1) (Takatsuto 1994). Since the discovery of BL in 1979, around 70 BRs have been characterized in 64 plant species comprising 53 angiosperms (12 monocotyledons and 41 dicotyledons), 6 gymnosperms, 1 pteridophyte (Equisetum arvense), 1 bryophyte (Marchantia polymorpha) and 3 algae (Chlorella Vulgaris, Cystoseira myrica and Hydrodictyon reticulatum). However, a limited number (52) of BRs were described in terms of their biological activities in plants. Among all the BRs studied to present, Castasterone (CS), Brassinolide (BL), typhasterol (TY), 6-deoxoCS, teasterone (TE) and 28-norCS have shown to be broadly present in different plant species across different environments (Bajguz and Tretyn 2003a, b). The occurrence of BRs in algae has been rarely reported and up to now, only two C28 brassinosteroids (24-epiCS and 28-HomoCS) have been recorded in Hydrodictyon reticulatum (Camoni et al. 2018). Considering the latest literature reports 81 natural BRs were characterized which include five conjugates along with 137 BRs analogues and 8 metabolites (Liu et al. 2017). Considering the chemistry of brassinosteroids, it can be stated that BRs are polyhydroxy steroid lactone having a similar chemical arrangement (in terms of carbon skeleton) than brassinolide (BL). A significant structural diversity at cyclic and side chain level exists however, and this is responsible for vital metabolic alterations to and from two other highly active equivalents of brassinosteroid known as 24-Epibrassinolide (EBL) and 28-Homobrassinolide (HBL) (Fig. 1). In addition, BRs are completely safe (Esposito et al. 2011) and eco-friendly hormones (Kang and Guo 2011).
Major BRs found in plants (Anwar et al. 2018)
Brassinosteroids are derivatives of the 5-cholestane skeleton, with structural differences that result from the form and location of functional groups in the A and B rings and the side-chain (Fig. 2) (Fujioka and Yokota 2003). BRs exhibit vicinal hydroxyl groups with respect to the A-ring at C-2α and C-3α. BRs having α-hydroxyl, β-hydroxyl or ketone at C-3 position are the progenitor of BRs with 2α, 3α-vicinal hydroxyls. BR exhibiting 2α, 3β-, 2β, 3α- or 2β, 3β-vicinal hydroxyls possibly the precursors of 2α, 3α-vicinal hydroxyls. The two 2α, 3α-vicinal hydroxyl groups on the A-ring constitute overall structural attributes of most active BRs, including BL and CS (Fig. 3). The declining order of activity 2α, 3α > 2α, 3β > 2β, 3α > 2β, 3β demonstrated by the relationship between structure and activity proposes that α-oriented hydroxyl group at C-2 is crucial for better biological activity of BRs in plants (Zhu et al. 2019; Zou et al. 2020).
Different substituents in the A- and B-rings of naturally occurring Brassinosteroids (Clouse 2011)
BRs mode of action and its signal transduction mechanism
Various plant growth processes showed positive correlation to BRs application. While plant which are not capable to produce BR in enough amount or malfunctioning in the signal transduction processes of BR show several types of abnormal phenotypes development, indicating the importance of biosynthesis of BR and their role in several plant process. Numerous mechanisms take part in the BRs biosynthetic and signaling pathway have been recognized through utilizing numerous methods like molecular, genetics and genomic approach. BRs take part in several physiological attributes of plants (development of root, elongation of cell, development of anther and pollen, vascular differentiation, stem elongation etc.) and also help to regulate the function of these attributes under unfavorable conditions (Fahad et al. 2014, 2016a, b, c, d).
In numerous studies, innovative approaches have been identified to spot the accompanying constituents in the signaling pathway of BRs and amongst them, bioinformatics and screening tools (Nam and Li 2004); mass spectrometry (Hink et al. 2008) and proteomics (Tang et al. 2008) have delivered vital information in recognizing novel constituents. The novel constituents that played a key role in the biosynthesis of BRs and signal transduction comprise BRI-1interacting proteins such as BRI-1-associated receptor kinase 1 (BAK-1), BRI-1 kinase inhibitor 1 (BKI-1), transthyretinlike (TTL), and BR signaling kinases (BSKs) (Yang et al. 2011; Ayub et al. 2018; Derevyanchuk et al. 2016).
According to Kim et al. (2009) and Yang et al. (2011), that plasma-membrane which is localized in leucine rich-repeat (LRR)-receptor-like-kinase (RLK) BRI-1, which stands for brassinosteroid insensitive 1 (BRI-1), perceived the BRs. This further led to departure of BKI-1 (a negative regulator) from plasma membrane. Now an association formed by the BAK-1 with BRI-1 in order to make a complex of functional BR-receptor. After phosphorylation, BSK which is considered a positive substrate attached with BSU-1 through BRI-1 kinase and hence it enhances the BSU-1 performances, ensuring dephosphorylation and hampering of BIN-2.
Consequently, gathering of BIM1 (protein complex) and Myb30 is engaged by unphosphorylated BES-1/BZR-1, consequences to the development of varied transcriptional complexes. Then these complexes attach promoter areas of BR response gene and thus regulate its expression which eventually effect all the developmental processes like cell division and its elongation etc. BR biosynthesis is inhibit by the Brassinazole through oxidation of cathasterone to teasterone, which is catalyzed via CPD (Szekeres et al. 1996).
Brassinazole (BZ) as an inhibitor of BRs
Brassinazole is used to decrease the activity of BRs, as they effects are antagonist. As a result of this, BRs induce ripening in fruits and vegetables while BZ inhibits its ripening (Zhu et al. 2015a, b) and increase the postharvest life of horticultural products. According to a recent study, carotenoid contents were enhanced in BZR1 (Brassinazole resistant 1) mutant and genes responsible for ripening, accumulation of lycopene and ascorbic acid contents exhibited a decreasing trend (Liu et al. 2014). Additionally, the treatment of aqueous BZ (5 μ mol L−1) for 30 min inhibited ripening in persimmons (He et al. 2018). More fruit firmness, cellulose content, decreased ethylene production, cell wall degrading enzymes activity and the expression of ethylene biosynthesis related genes was reduced in BZ treated fruits. Application of BL could accelerate ripening by 5 days while application of BZ delayed ripening by 7 days in big green strawberry, when compared to untreated control (Chai et al. 2013). Pre-harvest application of BZ at the concentration of 5 µL influence the onset of ripening in grape berries (Symons et al. 2006).
Brassinosteroid role in postharvest biotechnology
Use of brassinosteroids (BRs) prior to the harvesting
The positive impacts of BRs in enhancing the yield and quality of horticultural products (Ali 2017) and the plant response to environmental stresses (Rajewska et al. 2016; Anwar et al. 2018; Banerjee and Roy Choudhury 2018) are of great importance. The main advantages of BRs are their non-toxic effects and eco-friendly nature (Hayat and Ahmad 2010). The postharvest quality of horticultural commodities mainly depends on pre-harvest factors, as quality can only be maintained, not improved, during storage (Arah et al. 2015).
In a study conducted by (Ali et al. 2006), the roots of 20 days tomato seedlings were treated with various doses (10−8, 10−7, 10−6, 10−5 M) of 28-Homobrossinolide (HBR), analogue of BR, at different time intervals (15, 30 and 45 min). The study revealed that application of 10−8 M of HBR at 15 min resulted in greater production of tomatoes fruits with higher lycopene and ß-carotene contents at ripening relative to control treatment (Ali et al. 2006). In another study, different concentrations of DI-31 and DI-100 analogues of BRs, along with seaweed extract and Tomex Amin were applied on peppers; results shown increased yield and net photosynthesis due to application of DI-31 BRs as compared to DI-100s. The control treatment showed similar effect as BRs on the pepper skin firmness and color, fresh weight and ripening index. However, peppers treated with BRs had higher antioxidant activity and total phenolic content, two characteristics that are important for enhancing postharvest life of peppers (Serna et al. 2012).
Brassinosteroids have also been utilized in grape (Vitis vinifera L.). Working with different vines of flame seedless cultivars of grape, utilized different level of BRs aqueous solutions (0, 0.1, 0.5 and 1 mg L−1) in a 12-year old own rooted grapevine. Results revealed that the length, width and weight of clusters were considerably increased by the applications of 0.5 and 1 mg L−1 BRs. Remarkably, reduction of decay development in grapes subjected to cold storage conditions at 3–4 °C and 90–95% relative humidity, and other important postharvest attributes such as fruit firmness, anthocyanins, total phenolic content, TSS content, and TA were strongly influenced when 0.5 mg L−1 BRs were applied to 12-year old own rooted grapevine (Champa et al. 2015).
Pre-harvest application of BRs has also shown positive effects to induct ripening in non-climacteric fruits such as grape and berry. Applications of BRs also enhance the Brix levels of berries (Symons et al. 2006). In a study conducted by Chai et al. (2013) BRs were sprayed on strawberry plants to evaluate its role in the ripening initiation. The study suggested that BRs treatments induced ripening in strawberries along with higher chlorophyll, carotenoid, soluble solids, starch contents and induced antioxidant enzymes activities (Chai et al. 2013). Positive results have also been reported following application of HBR. HBR applied to early season cultivar Tulare and mid-season cherry cultivars Bing, Lapins and Rainer, Mandava and Wang (2016) observed a decrease in the maturation time and an improvement in postharvest quality.
Postharvest application of BRs
Brassinosteroids can help to maintain postharvest quality and are applied for several purposes in postharvest storage of different horticultural products. Implications of BRs in tomato, persimmon, mango, jujube, peach, lime, grape, potato and asparagus for improving postharvest quality have been discussed here. Brassinosteroids have been used following harvesting to induce ripening in horticultural crops. For example, mature green tomatoes were applied with BR to induce ripening in a study conducted by Zhu et al. (2015a, b). With the application of BRs increased the soluble solids, ascorbates, lycopene, CO2 and ethylene production, but reduced the chlorophyll content when compared with control. In the case of persimmon, a study conducted by He et al. (2018) found that BRs increased the production of ethylene, respiration rate, the activity of enzymes responsible for the degradation of the cell wall that leads to softening, thus, accelerating the ripening process of persimmon fruits (He et al. 2018). Similarly, in mango fruits, BRs hastened the ripening process by enhancing the production of ethylene, respiration rate, softening of fruits and development of skin color without affecting the quality of ripened fruit (Zaharah and Singh 2010; Zaharah et al. 2012). In Jujube fruits, BRs significantly reduced the senescence by slowing down the production of ethylene and respiration rate. The BRs-treated fruits had more total suspended solids (TSS), titratable acidity (TA) and ascorbic acid content when compared to the untreated control (Zhu et al. 2010). In other cases, BRs could be applied with other products such as chitosan to improve overall results. According to Wu and Yang (2016) chitosan treatment along with BRs increased the shelf life of asparagus as they retarded the respiration rate, loss of water, retained higher ascorbates, phenols, antioxidant activity, while inhibiting the rise in malonaldehyde (MDA) and leakage of electrolyte of asparagus spears during storage.
In other cases, application of BL and EBL has also been utilized to induce fruit ripening. In a recent study, Ge et al. (2014) tested BL concentration of 0, 2, 5 and 10 µ mol L−1 to determine its effects on postharvest quality of peaches (Ge et al. 2014). Authors suggested that 5 µ mol L−1 was the optimum dose to slow down the rise in weight losses, decline in firmness, SSC, TA and increase ascorbic acid contents (Ge et al. 2014). The impacts of 24-epibrassinolide (EBL) on the maintenance of postharvest quality of Persian lime and Tahiti lime were investigated by Tavallali (2018), who suggested that EBL had efficient results on the maintenance of firmness and weight of limes. Application of EBL reduced the decline of chlorophyll a and h° values. The use of EBL treatment considerably raised the ascorbates, phenolic content, the activity of antioxidants and TA. In another study, different concentrations of EBL (0.1, 0.4 and 0.8 mg L−1) were tested on the clusters of grapes. Results showed that EBL treatment resulted in fruit firmness increase, and lower weight losses and postharvest deterioration (Liu et al. 2016). Also, the use of EBL at a dose of 0.021 mg dm−3 on potato tuber after harvesting successfully inhibited potato sprouting, one of the major problems in potato postharvest storage, by 36–38 days. Moreover, the use of EBL raised the production of ethylene after 1 and 7 days of storage by almost 150% and 300%, respectively (Korableva et al. 2002).
Brassinosteroid and the suppression of biotic stresses during crop cycle
The positive role of BRs is not limited to plant response under abiotic stresses but also against biotic challenges (Adie et al. 2007; Krishna 2003). For instance, the pathogenesis related (PR) genes have been reported in numerous studies for their role in enhancing the plant tolerance against biotic stress (Sugawara et al. 2016; Filipenko et al. 2013; Wu et al. 2016). In line with that, the Arabidopsis cpd mutant deficient in BR biosynthesis showed a remarkable decline in the expression of PR genes (PR1, PR2, and PR5), whereas, the high expression was observed in CPD over-expressed transgenic plants (Szekeres et al. 1996).
In potato plants, application of 24-epibrassinolide (EBL) decreased disease incidence caused by Phytophthora by up to 34% in different cultivars (Khripach et al. 1996). In barley, application of 24 EBL reduced leaf disease caused by Helminthosporium teres Sacc at the tillering phase (Volynets et al. 2012).
The use of brassinosteroids in rice and tobacco plants has the potential to induce resistance to different plant diseases by suppressing the negative effects of Maganoprothe grisea, Xanthomonas oryzae and Tobacco mosaic virus (TMV) respectively (Nakashita et al. 2003; Friedrichsen et al. 2002). However, the role of BRs could be greatly impacted by the time, method of application and the interaction between host and its pathogen (Korableva et al. 2002; Vardhini 2016). Previous research reported that the application of BR softened the potato tuber tissues and weakened the immune system of pants, but these results were observed 4 months after BR application, which could have had an influence in the decrease in efficiency of exogenous BR (Bajguz and Hayat 2009). Furthermore, the treated potato tubers after harvesting showed prolonged dormant characteristics, enhanced resistance to Phytophthora infestans, and increased biosynthesis of phenolic compounds. In other cases, BRs can stimulate the mycelia growth and sporulation intensity of P. infestans in potato tubers, thereby inducing susceptibility to the disease (Pascual Serrano et al. 2016). Conversely, research shows that BRs can enhance the resistance against P. infestans in post-harvested tubers and its dormancy period (Choudhary et al. 2012),while application of both BRs and Salicylic acid can increase resistance to the bacterial pathogen Xanthomonas oryzae pv. Oryzae and fungal pathogen Maganoprothe grisea in rice plants.
Studies have reported that the protective role of BR’s is dependent upon the growth stage of plant and pathogen (Bajguz and Hayat 2009). Moreover, potato growth parameters and especially yield was increased by up to 50% under biotic stress (Roth et al. 2000).
Summarizing, BRs application to crops have revealed a significant protective role against environmental stress that can have a profoudn detrimental impact crop biomass and yield (Ahmad et al. 2017). Research has demonstrated that exogenous application of BR have increased the growth, yield and reduced stress effects in different plant species. The molecular mechanisms of BR have shown promising role in crop plants under stressful environments; and the strategies mentioned in Table 1 can also be used for practical application in the agriculture sector.
Alternative to chemicals against biotic and abiotic stresses in postharvest products
Brassinosteroids are a potential viable solution to inhibit the negative effects of biotic and abiotic stresses that occur during the postharvest storage of fruits and vegetables with no hazardous effects on human health.
The effect of Brassinosteroids applications on plant diseases have been extensively studied. In a recent study conducted related to Brassinosteroids, EBL (0.1 mg L−1; 0.4 mg L−1, 0.8 mg L−1) was applied to grapes to study its impact on grey mold infestation. Results from this study showed that treatment stimulated the actions of enzymes related to defense and considerably reduced the severity of the disease, superoxide anion radical production and malonaldehyde (MDA) contents. Moreover, authors concluded that EBL was responsible for terminated grey mold and keeping the postharvest quality of grapes (Liu et al. 2016). In jujube and peaches, the application of BRs (5 µM) and BL (5 µ mol L−1) induced resistance against the Penicillium expansum and slowed down the senescence process in the plant (Zhu et al. 2010; Ge et al. 2014).
Different types of abiotic stresses can also impact quality and yield of fruits and vegetables during postharvest. For example, browning is a major problem in postharvest storage of mushroom. Research conducted showed that brassinolide (BL) application decreased not only the development of browning, but also reduced the phenolic content, polyphenol oxidase activity and maintained better postharvest quality in white button mushroom (Ding et al. 2009). Also, application of BL has resulted in a reduction in chilling injury in bell peppers. When this occurred, application of 15 µM of BL was more beneficial than application of 5 and 10 µM doses of BL during 18 days of cold storage at 3 °C (Wang et al. 2012). Chilling injury can also cause important economic losses in subtropical fruits like orange. Application of BRs (0.75 and 1.5 ppm) increased catalase and peroxidase activities in Washington navel oranges (Ghorbani and Pakkish 2014) indicating that the BRs induced resistance against chilling injury. Table 2 shows different examples of postharvest application of BRs and analogues.
Brassinosteroid role in reducing pesticide toxicity
Although the impact of chemical pesticide use in the control of pest has been widely documented for many plant species and different conditions, improper use has resulted in ecosystems imbalances (Barański et al. 2014). In plants, the toxic effects of pesticide can systemically damage the plant structure and its physiological activities by initiating necrosis and arrest the photosynthesis system and plant growth (Xia et al. 2009). Authors also reported alteration in plant photosynthesis due to pesticide toxicity. Additionally, pesticide application disturbs the rhizosphere activities, which in turn negatively affects the process of symbiosis between bacteria and plants (Kumar et al. 1996). Despite the potential damage to crop production and the agriculture sector as a whole, adverse effects following improper use of pesticides have been reported in human health through air and water contamination (Aktar et al. 2009; Bueno et al. 2008).
The involvement of brassinosteroid as a detoxification plant mechanism to mitigate adverse pesticide reactions has been poorly studies (Krishna 2003; Hatzios and Burgos 2004; Sharma et al. 2016; Kanwar et al. 2012; Özdemir et al. 2004). In this sense, Hatzios and Burgos (2004) suggested that BRs enhance the transcript level of various genes encoding P450 cytochrome along with intense glutathione metabolism that ignite the herbicide biodegradation. The detoxification process initiate after the plant gets in contact with the pesticide. In the phase I of this process, the P450 cytochrome, peroxidases and mono oxygenase trigger a series of metabolic reactions that start metabolizing the absorbed pesticide. In phase II, the glutathione S transferase (GST) and UDP glucosyl transferase (UGT) promote the conjugation of glutathione (GSH) glucose. Finally, sequestration of soluble pesticide molecules and subsequent storage in plant apoplasts and vacuoles occurs in phase III. Other studies (Xia et al. 2009; Müssig et al. 2002) also highlighted the importance of BRs in detoxifying pesticide toxicity by performing transcriptional analyses of Arabidopsis and cucumber BRs deficient mutants. Overall, these studies revealed that BRs plays an important role in regulating the detoxification genes such as genes encoding P450 mono oxygenase, UDP and GST.
Exogenous application of other phytohormones has also shown a potential to reduce toxicity from pesticides. For example, application of 24 epibrassinolide (EBL) in tomato plants significantly reduced the negative effects of chlorothalonil (CHT) by upregulating the expression of RESPIRATORY BURST OXIDASE HOMOLOG1 (ROBH1) that also enhanced the accumulation of H2O2 in apoplast. In contrast to that, ROBH1 silenced plants showed considerable reduction in the EBL mediated CHT metabolism (Hou et al. 2018). Additionally, GRXS16 gene silenced plants failed to maintain the EBL mediated enhanced accumulation of CHT because of the reduced glutathione content, GST, and its transcript (GST1). In this study, silenced tomato plants were also reported for higher CHT residues (Hou et al. 2018). Table 3 shows some of the recent reported studies on BRs mimic pesticide toxicity. However, additional research is needed to inspect the BRs induced pesticide detoxification mechanism.
BRs role in mitigating the phytotoxicity of organic pollutant
The recent boom in urbanization has led to an increase in production of industrial waste in the form of organic pollutants that pose a threat to human health and our ecosystems. Persistent organic pollutants (POPs) are extremely toxic to our health because of their carcinogenic and mutagenic properties (Rostami and Juhasz 2011). In plants, the effects of organic pollutants can also be important. For example, the cherry tomato has been exposed to Fluoranthene mist for 30 days, which caused a reduction in the net photosynthesis rate up to 37% by overproducing the harmful reactive oxygen species (ROS) (Oguntimehin et al. 2010). Other organic toxicants such as dichlorobenzene and trichlorobenzene have been reported for their role in hindering the growth of the maize plant by impairing the cell division process (San Miguel et al. 2012). On the other hand, BRs and its analogues have shown a tremendous ability to minimize the phytotoxicity caused by organic pollutants by reducing lipid peroxidation while enhancing antioxidant activities (Ahammed et al. 2013a, b). The foliar application of EBR (100 nM) has successfully alleviated the adverse effects of polychlorinated biphenyl (PCB) in tomato by decreasing the harmful ROS production, and increasing the Fv/Fm ratio, photosynthetic capacity and overall biomass production (Ahammed et al. 2013a, b). Ahammed et al. (2017) studied the impact of organic pollutants 2,4,6-trichlorophenol, chlorpyrifos and oxytetracyclinein cucumber plants. Results from this study revealed that these pollutants severely impaired plant root elongation by increasing the production of malondialdehyde (MDA), H2O2 and NO (nitric oxide) in roots. However, the exogenously applied EBR attenuated the production of H2O2, MDA and NO via enriched antioxidant activities, flavonoid contents, and secondary metabolites enzymes in roots. Additionally, EBR application induced the glutathione content, which resulted in reduced redox states (Ahammed et al. 2017).
Benzene is an airborne gaseous organic pollutant which induces cell damage both in plants and animals. In a recent study, the harmful effects of benzene on Chlorophytum comosum were significantly ameliorated by application of exogenous 24-epibrassinolide (Setsungnern et al. 2019). The 24-epibrassinolide treated plants exhibited better antioxidant enzymes activity and hindered the elicited oxidative stress and lipid peroxidation along with an increased expression of benzene detoxification genes for ascorbic acid (AsA), homogentisate phytyltransferase (HPT), and glutathione synthetase (GS). Moreover, higher photosynthetic activity and glutathione biosynthesis were also observed in 24-epibrassinolide treated plants (Setsungnern et al. 2019).
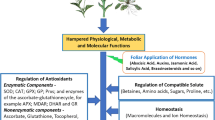
Polycyclic aromatic hydrocarbon (PAHs) is another major organic micro pollutant widely distributed in our environment. In a study conducted by Ahammed et al. (2012), tomato plants were exposed to 300 um of PAHs stress for 21 days triggered MDA production and photoinhibition while decreased net CO2 assimilation and total plant biomass (Ahammed et al. 2012). Conversely, foliar application of 24-epibrassinolide significantly reduced lipid peroxidation, boosted antioxidant enzymes activity, prevented the root cell from damage and upregulated the transcript abundance of detoxification genes such as CYP90b3, GSH1, and GST1 (Ahammed et al. 2012). Altogether, these results highlight the importance of BRs application to enhance resistance against the toxicity of organic pollutant by increasing the antioxidant activities, improving secondary metabolism and hindering the lipid peroxidation and harmful ROS (Fig. 4). Furthermore, BRs application could be an eco-friendly strategy to cope with the toxicity caused by organic pollutant in plants, which can in turn, limit the harmful effects these pollutants have on human health.
BRS regulate nodule and mycorrhiza formation
The infection of Leguminosae roots by rhizobacteria initiate the process of nodulation, which further convert the atmospheric nitrogen into ammonium to make it available for plant growth and development (Mylona et al. 2011). A specialized pathway known as autoregulation of the nodulation (AON) controls the number of nodules per plant (Reid et al. 2011). This metabolic pathway is controlled by Nodulation Autoregulation Receptor Kinase (NARK) in coordination with CLAVATA2 (CLV2) and/or KLAVIER (KLV). Another essential component in the AON pathway is the regulatory factor RDN1. Mutation in NARK, RDN1, KLV, and CLV2 genes have the potential to produce plants with a very high number of nodules (Miyazawa et al. 2010; Krusell et al. 2011; Foo et al. 2014). In this context, BRs has also been shown to be an important element during the nodule formation. In pea, a mutation in the BR biosynthetic enzymes LK and LKB severely affected the nodule formation process, suggested that BRs can be crucial in regulating the intensity of nodules (Ferguson et al. 2005). In another study, the BR and AON (nark 1K, clv2 1K, and rdn1 1K) double mutant exhibited superior nodulation ability similar to that of the single AON mutant, which indicates that BR is not the only responsible for the formation of nodules (Foo et al. 2014). However, exogenously applied Brassinazole (BZ) in soybean triggered the nodulation process, which may imply BRs actually works as an inhibitor during nodule formation (Terakado et al. 2005). For that reason, more studies are needed to unfold the opposite role of BRs and BZ during nodule formation in different plant species.
Arbuscular mycorrhiza fungi (AMF) is vital to make soil nutrients more readily available for plant utilization (Strack et al. 2003), especially phosphorus and nitrogen, two macronutrients that are required for plants in considerable amounts (Ketterings and Czymmek 2007; Kumar et al. 2019a, b; Czymmek et al. 2020). At present, research stressing the role of BRs in AMF symbiosis is scarce. In a study conducted by Foo et al. (2013), BR mutant lbk in pea inoculated with AMF showed no effect on plant mycorrhizal colonization. However, mutant plants in this study were leaky and, regardless of being a BR mutant, low levels of BR that may be enough for mycorrhizal activity were synthesized (Foo et al. 2013). In another study, tomato dX mutants malfunctioning in BR synthesis negatively affected the mycorrhization infection and colonization (Bitterlich et al. 2014a, b). The relative reduction in the level of different sugars required as energy supply during mycorrhization was also observed in plant mutants (Bitterlich et al. 2014a, b). In rice, the BRs mutant brd2-1 showed reduced mycorrhization along with a significant reduction in RNA accumulation of sucrose transporter 4 (OsSUT4) (Bitterlich et al. 2014b). The sucrose transporter SISUT2 regulate the process of mycorrhization by interacting with genes related to BR biosynthesis (Bitterlich et al. 2014a, b; Bitterlich et al. 2014b). Further efforts are required to investigate the importance of exogenous BRs application in inducing the mycorrhization process in plant.
Summary and further research directions
Brassinosteroid phytohormones are essential for plant growth, as they are involved in main plant antioxidant processes encompassing the regulation and enhancement of plant tolerance against different environmental stresses. In postharvest technology, brassinosteroids have shown great potential to be considered as a green and non-toxic management tool. As such, brassinosteroids may enhance the quality of postharvest fruits and vegetables by triggering the activities of proline, total phenolic compounds, soluble sugars, flavonoids, and ascorbic acid. Additionally, brassinosteroids have the potential to reduce pesticide toxicity by activating pesticide detoxification genes such as RBOH1. Moreover, exogenous application of brassinosteroids has effectively mitigated the toxicity of organic pollutant in several studies by restricting the activity of damaging reactive oxygen species and lipid peroxidation while enhancing the activity of antioxidant enzymes and secondary metabolism in plant cells. Furthermore, evidence shows that both nodule and mycorrhizal colonization in can be affected in brassinosteroids biosynthetic enzymes mutant showing the paramount involvement of brassinosteroids in these essential plant processes. However, a few other studies have reported the opposite role of brassinosteroids BRs in nodule and mycorrhizal colonization.
Finally, most of the available research with brassinosteroids has been conducted under lab-based conditions. Future research is required to examine the role of brassinosteroids in field conditions under a diverse set of environmental conditions and across different plant species. Research of this type will not only highlight the versatile role of brassinosteroids in mitigating stress and improving plant quality and productivity but will also provide with scientific “on-farm” evidence that could be used by farmers to decide whether they want to include these phytohormones as a management tool future agriculture.
References
Adie BA, Pérez-Pérez J, Pérez-Pérez MM, Godoy M, Sánchez-Serrano JJ, Schmelz EA, Solano R (2007) ABA is an essential signal for plant resistance to pathogens affecting JA biosynthesis and the activation of defenses in Arabidopsis. Plant Cell 19:1665–1681
Ahammed GJ, Gao C-J, Ogweno JO, Zhou Y-H, Xia X-J, Mao W-H, Shi K, Yu J-Q (2012) Brassinosteroids induce plant tolerance against phenanthrene by enhancing degradation and detoxification in Solanum lycopersicum L. Ecotoxicol Environ Saf 80:28–36
Ahammed G, Zhou Y, Xia X, Mao W, Shi K, Yu J (2013a) Brassinosteroid regulates secondary metabolism in tomato towards enhanced tolerance to phenanthrene. Biol Plant 57:154–158
Ahammed GJ, Ruan Y-P, Zhou J, Xia X-J, Shi K, Zhou Y-H, Yu J-Q (2013b) Brassinosteroid alleviates polychlorinated biphenyls-induced oxidative stress by enhancing antioxidant enzymes activity in tomato. Chemosphere 90:2645–2653
Ahammed GJ, He B-B, Qian X-J, Zhou Y-H, Shi K, Zhou J, Yu J-Q, Xia X-J (2017) 24-Epibrassinolide alleviates organic pollutants-retarded root elongation by promoting redox homeostasis and secondary metabolism in Cucumis sativus L. Environ Pollut 229:922–931
Ahmad H, Hayat S, Ali M, Ghani MI, Zhihui C (2017) Regulation of growth and physiological traits of cucumber (Cucumis sativus L.) through various levels of 28-homobrassinolide under salt stress conditions. Can J Plant Sci 98:132–140
Aktar W, Sengupta D, Chowdhury A (2009) Impact of pesticides use in agriculture: their benefits and hazards. Interdiscip Toxicol 2:1–12
Alabadí D, Blázquez MA (2009) Molecular interactions between light and hormone signaling to control plant growth. Plant Mol Biol 69:409
Albrecht C, Boutrot F, Segonzac C, Schwessinger B, Gimenez-Ibanez S, Chinchilla D, Rathjen JP, de Vries SC, Zipfel C (2012) Brassinosteroids inhibit pathogen-associated molecular pattern-triggered immune signaling independent of the receptor kinase BAK1. Proc Natl Acad Sci 109:303–308
Ali B (2017) Practical applications of brassinosteroids in horticulture—some field perspectives. Sci Hortic 225:15–21
Ali B, Hayat S, Hasan SA, Ahmad A (2006) Effect of root applied 28-homobrassinolide on the performance of Lycopersicon esculentum. Sci Hortic 110:267–273
Ali SS, Kumar GS, Khan M, Doohan FM (2013) Brassinosteroid enhances resistance to fusarium diseases of barley. Phytopathology 103:1260–1267
Ali SS, Gunupuru LR, Kumar GS, Khan M, Scofield S, Nicholson P, Doohan FM (2014) Plant disease resistance is augmented in uzu barley lines modified in the brassinosteroid receptor BRI1. BMC Plant Biol 14:219–227
Anwar A, Liu Y, Dong R, Bai L, Yu X, Li Y (2018) The physiological and molecular mechanism of brassinosteroid in response to stress: a review. Biol Res 51:46
Arah IK, Amaglo H, Kumah EK, Ofori H (2015) Preharvest and postharvest factors affecting the quality and shelf life of harvested tomatoes: a mini review. Intl J Agron
Arteca RN (2013) Plant growth substances: principles and applications. Springer, Berlin
Ayub RA, Reis L, Bosetto L et al (2018) Brassinosteroid plays a role on pink stage for receptor and transcription factors involved in strawberry fruit ripening. Plant Growth Regul 84:159–167. https://doi.org/10.1007/s10725-017-0329-5
Bajguz A (2007) Metabolism of brassinosteroids in plants. Plant Physiol Biochem 45:95–107
Bajguz A, Hayat S (2009) Effects of brassinosteroids on the plant responses to environmental stresses. Plant Physiol Biochem 47:1–8
Bajguz A, Piotrowska-Niczyporuk A (2014) Interactive effect of brassinosteroids and cytokinins on growth, chlorophyll, monosaccharide and protein content in the green alga Chlorella vulgaris (Trebouxiophyceae). Plant Physiol Biochem 80:176–183
Bajguz A, Tretyn A (2003a) The chemical characteristic and distribution of brassinosteroids in plants. Phytochemistry 62:1027–1046
Bajguz A, Tretyn A (2003b) The chemical structures and occurrence of brassinosteroids in plants. Brassinosteroids. Springer, Berlin, pp 1–44
Banerjee A, Roychoudhury A (2018) Interactions of Brassinosteroids with major phytohormones: antagonistic effects. J Plant Growth Regul 37:1025–1032
Barański M, Średnicka-Tober D, Volakakis N, Seal C, Sanderson R, Stewart GB, Benbrook C, Biavati B, Markellou E, Giotis C (2014) Higher antioxidant and lower cadmium concentrations and lower incidence of pesticide residues in organically grown crops: a systematic literature review and meta-analyses. Br J Nutr 112:794–811
Barbieri PA, Echeverría HE, Saínz Rozas HR, Andrade FH (2008) Nitrogen use efficiency in maize as affected by nitrogen availability and row spacing. Agron J 100:1094–1100. https://doi.org/10.2134/agronj2006.0057
Battaglia ML, Lee C, Thomason W (2018) Corn yield components and yield responses to defoliation at different row widths. Agron J 110:1–16. https://doi.org/10.2134/agronj2017.06.0322
Battaglia M, Lee C, Thomason W, Fike J, Sadeghpour A (2019a) Hail damage impacts on corn productivity: a review. Crop Sci 59:1–14. https://doi.org/10.2135/cropsci2018.04.0285
Battaglia ML, Lee C, Thomason W, Van Mullekom J (2019b) Effects of corn row width and defoliation timing and intensity on canopy light interception. Crop Sci. https://doi.org/10.2135/cropsci2018.05.0337
Bechtold U, Field B (2018) Molecular mechanisms controlling plant growth during abiotic stress. Oxford University Press, Oxford
Bitterlich M, Krügel U, Boldt-Burisch K, Franken P, Kühn C (2014a) Interaction of brassinosteroid functions and sucrose transporter SlSUT2 regulate the formation of arbuscular mycorrhiza. Plant Signal Behav 9:e970426
Bitterlich M, Krügel U, Boldt-Burisch K, Franken P, Kühn C (2014b) The sucrose transporter Sl SUT 2 from tomato interacts with brassinosteroid functioning and affects arbuscular mycorrhiza formation. Plant J 78:877–889
Bueno ADF, Bueno RCODF, Parra JRP, Vieira SS (2008) Effects of pesticides used in soybean crops to the egg parasitoid Trichogramma pretiosum. Cienc Rural 38:1495–1503
Camoni L, Visconti S, Aducci P, Marra M (2018) 14-3-3 proteins in plant hormone signaling: doing several things at once. Front Plant Sci 9:297
Chai Y-m, Zhang Q, Tian L, Li C-L, Xing Y, Qin L, Shen Y-Y (2013) Brassinosteroid is involved in strawberry fruit ripening. Plant Growth Regul 69:63–69
Champa WH, Gill M, Mahajan B, Aror N, Bedi S (2015) Brassinosteroids improve quality of table grapes (Vitis vinifera L.) cv. flame seedless. Trop Agric Res 26:368–379
Choudhary SP, Yu JQ, Yamaguchi-Shinozaki K, Shinozaki K, Tran L-SP (2012) Benefits of brassinosteroid crosstalk. Trends Plant Sci 17:594–605
Clouse SD (2011) Brassinosteroids. The Arabidopsis Book/American Society of Plant Biologists 9
Clouse SD, Langford M, McMorris TC (1996) A brassinosteroid-insensitive mutant in Arabidopsis thaliana exhibits multiple defects in growth and development. Plant Physiol 111:671–678
Czymmek K, Ketterings Q, Ros M, Battaglia M, Cela S, Crittenden S, Gates D, Walter T, Latessa S, Klaiber L, Albrecht G (2020) The New York Phosphorus Index 2.0. Agronomy Fact Sheet Series. Fact Sheet #110. Nutrient Management Spear Program. Cornell University Cooperative Extension. https://nmsp.cals.cornell.edu/publications/factsheets/factsheet110.pdf
Derevyanchuk M, Litvinovskaya R, Khripach V et al (2016) Brassinosteroid-induced de novo protein synthesis in Zea mays under salinity and bioinformatic approach for identification of heat shock proteins. Plant Growth Regul 78:297–305. https://doi.org/10.1007/s10725-015-0093-3
De Vos M, Van Oosten VR, Van Poecke RM, Van Pelt JA, Pozo MJ, Mueller MJ et al (2005) Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol Plant Microbe Interact 18:923–937. https://doi.org/10.1094/MPMI-18-0923
Ding Y, Zhu Z, Zhao J, Nie Y, Zhang Y, Sheng J, Meng D, Mao H, Tang X (2009) Effects of postharvest brassinolide treatment on the metabolism of white button mushroom (Agaricus bisporus) in relation to development of browning during storage. Food Bioprocess Technol 9:1327–1334
Esposito D, Komarnytsky S, Shapses S, Raskin I (2011) Anabolic effect of plant brassinosteroid. FASEB J 25:3708–3719
Fàbregas N, Lozano-Elena F, Blasco-Escámez D, Tohge T, Martínez-Andújar C, Albacete A, Osorio S, Bustamante M, Riechmann JL, Nomura T (2018) Overexpression of the vascular brassinosteroid receptor BRL3 confers drought resistance without penalizing plant growth. Nat Commun 9:4680
Fahad S, Hussain S, Matloob A, Khan FA, Khaliq A, Saud S, Hassan S, Shan D, Khan F, Ullah N, Faiq M, Khan MR, Tareen AK, Khan A, Ullah A, Ullah N, Huang J (2014) Phytohormones and plant responses to salinity stress: a review. Plant Growth Regul 75(2):391–404. https://doi.org/10.1007/s10725-014-0013-y
Fahad S, Hussain S, Saud S, Hassan S, Chauhan BS, Khan F et al (2016a) Responses of rapid viscoanalyzer profile and other rice grain qualities to exogenously applied plant growth regulators under high day and high night temperatures. PLoS ONE 11(7):159590. https://doi.org/10.1371/journal.pone.0159590
Fahad S, Hussain S, Saud S, Hassan S, Ihsan Z, Shah AN, Wu C, Yousaf M, Nasim W, Alharby H, Alghabari F, Huang J (2016b) Exogenously applied plant growth regulators enhance the morphophysiological growth and yield of rice under high temperature. Front Plant Sci 7:1250. https://doi.org/10.3389/fpls.2016.01250
Fahad S, Hussain S, Saud S, Hassan S, Tanveer M, Ihsan MZ, Shah AN, Ullah A, Nasrullah KF, Ullah S, AlharbyH NW, Wu C, Huang J (2016c) A combined application of biochar and phosphorus alleviates heat-induced adversities on physiological, agronomical and quality attributes of rice. Plant Physiol Biochem 103:191–198
Fahad S, Hussain S, Saud S, Khan F, Hassan S, Nasim W, Arif M, Wang F, Huang J (2016d) Exogenously applied plant growth regulators affect heat-stressed rice pollens. J Agron Crop Sci 202:139–150
Fariduddin Q, Yusuf M, Ahmad I, Ahmad A (2014) Brassinosteroids and their role in response of plants to abiotic stresses. Biol Plant 58:9–17
Farnham DE (2001) Row spacing, plant density, and hybrid effects on corn grain yield and moisture. Agron J 93:1049–1053
Feng W, Lindner H, Robbins NE, Dinneny JR (2016) Growing out of stress: the role of cell-and organ-scale growth control in plant water-stress responses. Plant Cell 28:1769–1782
Ferguson BJ, Ross JJ, Reid JB (2005) Nodulation phenotypes of gibberellin and brassinosteroid mutants of pea. Plant Physiol 138:2396–2405
Filipenko E, Kochetov A, Kanayama Y, Malinovsky V, Shumny V (2013) PR-proteins with ribonuclease activity and plant resistance against pathogenic fungi. Russ J Genet 3:474–480
Foo E, Ross JJ, Jones WT, Reid JB (2013) Plant hormones in arbuscular mycorrhizal symbioses: an emerging role for gibberellins. Ann Bot 111:769–779
Foo E, Ferguson B, Reid J (2014) The potential roles of strigolactones and brassinosteroids in the autoregulation of nodulation pathway. Ann Bot 113:1037–1045
Friedrichsen DM, Nemhauser J, Muramitsu T, Maloof JN, Alonso J, Ecker JR, Furuya M, Chory J (2002) Three redundant brassinosteroid early response genes encode putative bHLH transcription factors required for normal growth. Genetics 162:1445–1456
Fujioka S, Yokota T (2003) Biosynthesis and metabolism of brassinosteroids. Annu Rev Plant Biol 54:137–164
Furio R, Salazar S, Martínez-Zamora G, Coll Y, Hael-Conrad V, Díaz-Ricci J (2019) Brassinosteroids promote growth, fruit quality and protection against Botrytis on Fragaria x ananassa. Eur J Plant Pathol. https://doi.org/10.1007/s10658-019-01704-3
Gao W, Long L, Zhu LF, Xu L, Gao WH, Sun LQ, Liu LL, Zhang XL (2013) Proteomic and virus-induced gene silencing (VIGS) analyses reveal that gossypol, brassinosteroids, and jasmonic acid contribute to the resistance of cotton to Verticillium dahliae. Mol Cell Proteomics 12:3690–3703
Gao H, Kang L, Liu Q, Cheng N, Wang B, Cao W (2015) Effect of 24-epibrassinolide treatment on the metabolism of eggplant fruits in relation to development of pulp browning under chilling stress. J Food Sci Technol 52:3394–3401
Ge Y, Li C, Tang R, Sun R, Li J (2014) Effects of postharvest brassinolide dipping on quality parameters and antioxidant activity in peach fruit. In: Proceedings of III international symposium on postharvest pathology: using science to increase food availability, pp. 377–384
Ghorbani B, Pakkish Z (2014) Brassinosteroid enhances cold stress tolerance of Washington navel orange (Citrus sinensis L.) fruit by regulating antioxidant enzymes during storage. Agric Conspec Sci 79:109–114
González-García MP, Vilarrasa-Blasi J, Zhiponova M, Divol F, Mora-Garcia S, Russinova E, Cano-Delgado AI (2011) Brassinosteroids control meristem size by promoting cell cycle progression in Arabidopsis roots. Development 138:849–859. https://doi.org/10.1242/dev.057331
Grove MD, Spencer GF, Rohwedder WK, Mandava N, Worley JF, Warthen JD, Steffens GL, Flippen-Anderson JL, Cook JC (1979) Brassinolide, a plant growth-promoting steroid isolated from Brassica napus pollen. Nature 281:201–216
Hatzios KK, Burgos N (2004) Metabolism-based herbicide resistance: regulation by safeners. Weed Sci 52:454–467
Hayat S, Ahmad A (2010) Brassinosteroids: a class of plant hormone. Springer, Berlin
Hayat S, Ahmad A, Fariduddin Q (2003) Brassinosteroids: a regulator of 21st century. Springer, Berlin, pp 231–246
He Y, Li J, Ban Q, Han S, Rao J (2018) Role of brassinosteroids in persimmon (Diospyros kaki L.) fruit ripening. J Agric Food Chem 66:2637–2644
Hink MA, Shah K, Russinova E, De-Vries SC, Visser AJ (2008) Fluorescence fluctuation analysis of Arabidopsis thaliana somatic embryogenesis receptor-like kinase and brassinosteroid insensitive 1 receptor oligomerization. Biophys J 94:1052–1062
Hou J, Zhang Q, Zhou Y, Ahammed GJ, Zhou Y, Yu J, Fang H, Xia X (2018) Glutaredoxin GRXS16 mediates brassinosteroid-induced apoplastic H2O2 production to promote pesticide metabolism in tomato. Environ Pollut 240:227–234
Javid MG, Sorooshzadeh A, Moradi F, Modarres Sanavy SAM, Allahdadi I (2011) The role of phytohormones in alleviating salt stress in crop plants. Aust J Crop Sci 5:726
Kang Y, Guo S (2011) Role of brassinosteroids on horticultural crops. A class of plant hormone Springer, In Brassinosteroids, pp 269–288
Kanwar MK, Bhardwaj R, Arora P, Chowdhary SP, Sharma P, Kumar S (2012) Plant steroid hormones produced under Ni stress are involved in the regulation of metal uptake and oxidative stress in Brassica juncea L. Chemosphere 86:41–49
Kaya C, Ashraf M, Sonmez O (2015) Promotive effect of exogenously applied thiourea on key physiological parameters and oxidative defense mechanism in salt-stressed Zea mays L. plants. Turk J Bot 39(5):786–795
Kaya C, Akram NA, Ashraf M, Sonmez O (2018) Exogenous application of humic acid mitigates salinity stress in maize (Zea mays L.) plants by improving some key physico-biochemical attributes. Cereal Res Commun 46:67–78
Ketterings Q, Czymmek K (2007) Removal of phosphorus by field crops. Agronomy Fact Sheet Series. Fact Sheet #28. Nutrient Management Spear Program. Cornell University Cooperative Extension. https://nmsp.cals.cornell.edu/publications/factsheets/factsheet28.pdf
Khripach V, Zhabinskii V, Litvinovskaya R, Zavadskaya M, Savel'eva E, Karas I, Kilcchevskii A, Titova SA (1996) Titova S A method for protection of potato from phytophthorosis. Pat. Appl, BY, p 960346
Kim TW, Guan S, Sun Y, Deng Z, Tang W, Shang JX, Sun Y, Burlingame AL, Wang ZY (2009) Brassinosteroid signal transduction from cell-surface receptor kinases to nuclear transcription factors. Nat Cell Biol 11:1254–1260
Kim BH, Kim SY, Nam KH (2012) Genes encoding plant-specific class III peroxidases are responsible for increased cold tolerance of the brassinosteroid-insensitive 1 mutant. Mol Cells 34:539–548
Korableva N, Platonova T, Dogonadze M, Evsunina A (2002) Brassinolide effect on growth of apical meristems, ethylene production, and abscisic acid content in potato tubers. Biol Plant 45:39–43
Krishna P (2003) Brassinosteroid-mediated stress responses. J Plant Growth Regul 22:289–297
Krusell L, Sato N, Fukuhara I, Koch BE, Grossmann C, Okamoto S, Oka-Kira E, Otsubo Y, Aubert G, Nakagawa T (2011) The Clavata2 genes of pea and Lotus japonicus affect autoregulation of nodulation. Plant J 65:861–871
Kumar S, Mukerji KG, Lai R (1996) Molecular aspects of pesticide degradation by microorganisms. Crit Rev Microbiol 22:1–26
Kumar R, Khurana A, Sharma AK (2014) Role of plant hormones and their interplay in development and ripening of fleshy fruits. J Exp Bot 65:4561–4575
Kumar P, Lai L, Battaglia ML, Kumar S, Owens V, Fike J, Galbraith J, Hong CO, Faris R, Crawford R, Crawford J, Hansen J, Mayton H, Viands D (2019a) Impacts of nitrogen fertilization rate and landscape position on select soil properties in switchgrass field at four sites in the USA. CATENA 180:183–193
Kumar S, Lai L, Kumar P, Feliciano YMV, Battaglia ML, Hong CO, Owens VN, Fike J, Farris R, Galbraith J (2019b) Impacts of nitrogen rate and landscape position on soils and switchgrass root growth parameters. Agron J 111(3):1046–1059
Lima J, Lobato A (2017) Brassinosteroids improve photosystem II efficiency, gas exchange, antioxidant enzymes and growth of cowpea plants exposed to water deficit. Physiol Mol Biol Plants 23:59–72
Liu L, Jia C, Zhang M, Chen D, Chen S, Guo R, Guo D, Wang Q (2014) Ectopic expression of a BZR1-1D transcription factor in brassinosteroid signalling enhances carotenoid accumulation and fruit quality attributes in tomato. Plant Biotechnol J 12:105–115
Liu Q, Xi Z, Gao J, Meng Y, Lin S, Zhang Z (2016) Effects of exogenous 24-epibrassinolide to control grey mould and maintain postharvest quality of table grapes. Int J Food Sci Technol 51:1236–1243
Liu J, Zhang D, Sun X, Ding T, Lei B, Zhang C (2017) Structure-activity relationship of brassinosteroids and their agricultural practical usages. Steroids 124:1–17
Mandava B, Wang Y (2016) Effect of brassinosteroids cherry maturation, firmness and fruit quality. Acta Hort 1139:451–458
Milborrow B, Pryce R (1973) The brassins. Nature 243:46
Mitchell JW, Gregory LE (1972) Enhancement of overall growth, a new response to brassins. Nature 239:253–254
Miyazawa H, Oka-Kira E, Sato N, Takahashi H, Wu G-J, Sato S, Hayashi M, Betsuyaku S, Nakazono M, Tabata S (2010) The receptor-like kinase KLAVIER mediates systemic regulation of nodulation and non-symbiotic shoot development in Lotus japonicus. Development 137:4317–4325
Moore TC (2012) Biochemistry and physiology of plant hormones. Springer, Berlin
Müssig C, Fischer S, Altmann T (2002) Brassinosteroid-regulated gene expression. Plant Physiol 129:1241–1251
Mylona P, Pawlowski K, Bisseling T (2011) Symbiotic nitrogen fixation. Plant Cell 7:869
Nahar K, Kyndt T, Hause B, Höfte M, Gheysen G (2013) Brassinosteroids suppress rice defense against root-knot nematodes through antagonism with the jasmonate pathway. Mol Plant Microbe Interact 26:106–115
Nakashita H, Yasuda M, Nitta T, Asami T, Fujioka S, Arai Y, Sekimata K, Takatsuto S, Yamaguch I, Yoshida S (2003) Brassinosteroid functions in a broad range of disease resistance in tobacco and rice. Plant J 33:887–898
Nam KH, Li J (2004) The Arabidopsis transthyretin-like protein is a potential substrate of brassinosteroid-insensitive-1. Plant Cell 16:2406–2417
Oguntimehin I, Eissa F, Sakugawa H (2010) Negative effects of fluoranthene on the ecophysiology of tomato plants (Lycopersicon esculentum Mill): fluoranthene mists negatively affected tomato plants. Chemosphere 78:877–884
Ogweno JO, Hu WH, Song XS et al (2010) Photoinhibition-induced reduction in photosynthesis is alleviated by abscisic acid, cytokinin and brassinosteroid in detached tomato leaves. Plant Growth Regul 60:175–182. https://doi.org/10.1007/s10725-009-9439-z
Özdemir F, Bor M, Demiral T, Türkan İ (2004) Effects of 24-epibrassinolide on seed germination, seedling growth, lipid peroxidation, proline content and antioxidative system of rice (Oryza sativa L.) under salinity stress. Plant Growth Regul 42:203–211
Park S-H, Han K-S, Kim T-W, Shim J-K, Takatsuto S, Yokota T, Kim S-K (1999) In vivo and in vitro conversion of teasterone to typhasterol in cultured cells of Marchantia polymorpha. Plant Cell Physiol 40:955–960
Pascual Serrano D, Vera Pasamontes G, Girón Moreno R (2016) Modelos animales de dolor neuropático. Dolor Investigación Clínica & Terapéutica 31:70–76
Pinol R, Simón E (2009) Effect of 24-epibrassinolide on chlorophyll fluorescence and photosynthetic CO2 assimilation in Vicia faba plants treated with the photosynthesis-inhibiting herbicide terbutryn. J Plant Growth Regul 28:97–105
Piotrowska A, Bajguz A (2011) Conjugates of abscisic acid, brassinosteroids, ethylene, gibberellins, and jasmonates. Phytochemistry 72:2097–2112
Rajewska I, Talarek M, Bajguz A (2016) Brassinosteroids and response of plants to heavy metals action. Front Plant Sci 7:629
Reid DE, Ferguson BJ, Hayashi S, Lin Y-H, Gresshoff PM (2011) Molecular mechanisms controlling legume autoregulation of nodulation. Ann Bot 108:789–795
Rostami I, Juhasz AL (2011) Assessment of persistent organic pollutant (POP) bioavailability and bioaccessibility for human health exposure assessment: a critical review. Crit Rev Environ Sci Technol 41:623–656
Roth U, Friebe A, Schnabl H (2000) Resistance induction in plants by a brassinosteroid-containing extract of Lychnis viscaria L. Zeitschrift für Naturforschung C 55:552–559
Sadura I, Janeczko A (2014) Physiological and molecular mechanisms of brassinosteroid-induced tolerance to high and low temperature in plants. Biol Plantarum 40:333–342
San Miguel A, Faure M, Ravanel P, Raveton M (2012) Biological responses of maize (Zea mays) plants exposed to chlorobenzenes. Case study of monochloro-, 1, 4-dichloro-and 1, 2, 4-trichloro-benzenes. Ecotoxicology 21:315–324
Sasse JM (2003) Physiological actions of brassinosteroids: an update. J Plant Growth Regul 22:276–288
Schaller H (2003) The role of sterols in plant growth and development. Prog Lipid Res 42:163–175
Serna M, Hernández F, Coll F, Coll Y, Amorós A (2012) Brassinosteroid analogues effects on the yield and quality parameters of greenhouse-grown pepper (Capsicum annuum L.). Plant Growth Regul 68:333–342
Setsungnern A, Treesubsuntorn C, Thiravetyan P (2019) Exogenous 24-epibrassinolide enhanced benzene detoxification in Chlorophytum comosum via overexpression and conjugation by glutathione. Sci Total Environ 662:805–815
Shapiro CA, Wortmann CS (2006) Corn response to nitrogen rate, row spacing, and plant density in Eastern Nebraska. Agron J 98:529–535. https://doi.org/10.2134/agronj2005.0137
Sharma I, Bhardwaj R, Pati PK (2012) Mitigation of adverse effects of chlorpyrifos by 24-epibrassinolide and analysis of stress markers in a rice variety Pusa Basmati-1. Ecotoxicol Environ Saf 85:72–81
Sharma D, Kaur GS, Khera KS (2015) Triazophos-induced oxidative stress and histomorphological changes in ovary of female Wistar rats. Pestic Biochem Physiol 117:9–18. https://doi.org/10.1016/j.pestbp.2014.09.004
Sharma A, Thakur S, Kumar V, Kanwar MK, Kesavan AK, Thukral AK, Bhardwaj R, Alam P, Ahmad P (2016) Pre-sowing seed treatment with 24-epibrassinolide ameliorates pesticide stress in Brassica juncea L. through the modulation of stress markers. Front Plant Sci 7:1569
Sharma A, Kumar V, Bhardwaj R, Thukral AK (2017) Seed pre-soaking with 24-epibrassinolide reduces the imidacloprid pesticide residues in green pods of Brassica juncea L. Toxicol Environ Chem 99:95–103
Singh I, Shono M (2005) Physiological and molecular effects of 24-epibrassinolide, a brassinosteroid on thermotolerance of tomato. Plant Growth Regul 47:111. https://doi.org/10.1007/s10725-005-3252-0
Sonmez O, Pierzynski GM, Frees L, Davis B, Leikam D, Sweeney DW, Janssen KA (2009a) A field assessment tool for phosphorus losses in runoff in Kansas. J Soil Water Conserv 64(3):212–222
Sonmez O, Aydemir S, Kaya C (2009b) Mitigation effects of mycorrhiza on boron toxicity in wheat plants. N Z J Crop Hort Sci 37(2):99–104
Strack D, Fester T, Hause B, Schliemann W, Walter MH (2003) Arbuscular mycorrhiza: biological, chemical, and molecular aspects. J Chem Ecol 29:1955–1979
Sugawara T, Trifonova EA, Kochetov AV, Kanayama Y (2016) Expression of an extracellular ribonuclease gene increases resistance to Cucumber mosaic virus in tobacco. BMC Plant Biol 16:246
Symons GM, Davies C, Shavrukov Y, Dry IB, Reid JB, Thomas MR (2006) Grapes on steroids. Brassinosteroids are involved in grape berry ripening. Plant Physiol 140:150–158
Szekeres M, Németh K, Koncz-Kálmán Z, Mathur J, Kauschmann A, Altmann T, Rédei GP, Nagy F, Schell J, Koncz C (1996) Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in arabidopsis. Cell 85:171–182. https://doi.org/10.1016/S0092-8674(00)81094-6
Takatsuto S (1994) Brassinosteroids: distribution in plants, bioassays and microanalysts by gas chromatography—mass spectrometry. J Chromatogr 658:3–15
Takatsuto S, Kamuro Y, Watanabe T, Noguchi T, Kuriyama H (1996) Synthesis and plant growth promoting effects of brassinosteroid compound TS303. Proc Plant Growth Regul Soc Am 23:15–20
Tang W, Kim TW, Oses-Prieto JA, Sun Y, Deng Z, Zhu S, Wang R, Burlingame AL, Wang ZY (2008) BSKs mediate signal transduction from the receptor kinase BRI1 in Arabidopsis. Science 321:557–560
Tavallali V (2018) Vacuum infiltration of 24-epibrassinolide delays chlorophyll degradation and maintains quality of lime during cold storage. Acta Sci Polonorum-Hortorum Cultus 17:35–48
Terakado J, Fujihara S, Goto S, Kuratani R, Suzuki Y, Yoshida S, Yoneyama T (2005) Systemic effect of a brassinosteroid on root nodule formation in soybean as revealed by the application of brassinolide and brassinazole. Soil Sci Plant Nutr 51:389–395
Thapliyal VS, Rai PN, Bora L (2016) Influence of pre-harvest application of gibberellin and brassinosteroid on fruit growth and quality characteristics of pear (Pyrus pyrifolia (Burm.) Nakai) cv. Gola. J Appl Natl Sci 8(4):2305–2310
Thelen KD (2006) Interaction between row spacing and yield: why it works. Crop Manag. https://doi.org/10.1094/CM-2006-0227-03-RV
Thummel CS, Chory J (2002) Steroid signaling in plants and insects: common themes, different pathways. Genes Dev 16:3113–3129
Tunc-Ozdemir M, Jones AM (2017) BRL3 and AtRGS1 cooperate to fine tune growth inhibition and ROS activation. PLoS ONE 12:e0177400
Vardhini BV (2016) Enhancement of vegetables and fruits growth and yield by application of brassinosteroids under abiotic stresses. In: Azooz MM, Ahmad P (eds) Plant-environment interaction: responses and approaches to mitigate stress. New York, Wiley. https://doi.org/10.1002/9781119081005.ch7
Volynets A, Pshenichnaya L, Manzhelesova N, Morozik G, Khripach V (2012) The nature of protective action of 24-epibrassinolide on barley plants. In: Proceedings of proceedings-plant growth regulation society of america-annual meeting, pp. 133–137
Wang Q, Ding T, Gao L, Pang J, Yang N (2012) Effect of brassinolide on chilling injury of green bell pepper in storage. Sci Hortic 144:195–200
Wang Z, Jiang Y, Peng X, Xu S, Zhang H, Gao J, Xi Z (2017) Exogenous 24-epibrassinolide regulates antioxidant and pesticide detoxification systems in grapevine after chlorothalonil treatment. Plant Growth Regul 81:455–466
Winter J, Schneider B, Meyenburg S, Strack D, Adam G (1999) Monitoring brassinosteroid biosynthetic enzymes by fluorescent tagging and HPLC analysis of their substrates and products. Phytochemistry 51:237–242
Wu L, Yang H (2016) Combined application of carboxymethyl chitosan coating and brassinolide maintains the postharvest quality and shelf life of green asparagus. J Food Process Preserv 40:154–165
Wu J, Kim SG, Kang KY, Kim JG, Park SR, Gupta R, Kim YH, Wang Y, Kim ST (2016) Overexpression of a pathogenesis-related protein 10 enhances biotic and abiotic stress tolerance in rice. Plant Pathol J 32:552
Xia X-J, Wang Y-J, Zhou Y-H, Tao Y, Mao W-H, Shi K, Asami T, Chen Z, Yu J-Q (2009) Reactive oxygen species are involved in brassinosteroid-induced stress tolerance in cucumber. Plant Physiol 150:801–814
Yang CJ, Zhang C, Lu YN, Jin JQ, Wang XL (2011) The mechanisms of brassinosteroids’ action: from signal transduction to plant development. Mol Plant 4:588–600
Ye H, Liu S, Tang B, Chen J, Xie Z, Nolan TM, Jiang H, Guo H, Lin H-Y, Li L (2017) RD26 mediates crosstalk between drought and brassinosteroid signalling pathways. Nat Commun 8:14573
Yoshida T, Mogami J, Yamaguchi-Shinozaki K (2014) ABA-dependent and ABA-independent signaling in response to osmotic stress in plants. Curr Opin Plant Biol 21:133–139
Zaharah S, Singh Z (2010) Role of brassinosteroids in mango fruit ripening. In: Proceedings of XXVIII international horticultural congress on science and horticulture for people (IHC2010): International Symposium on 934; pp. 929–935.
Zaharah SS, Singh Z, Symons GM, Reid JB (2012) Role of brassinosteroids, ethylene, abscisic acid, and indole-3-acetic acid in mango fruit ripening. J Plant Growth Regul 31:363–372
Zhang DW, Deng XG, Fu FQ, Lin HH (2015) Induction of plant virus defense response by brassinosteroids and brassinosteroid signaling in Arabidopsis thaliana. Planta 241:875–885
Zhu Z, Zhang Z, Qin G, Tian S (2010) Effects of brassinosteroids on postharvest disease and senescence of jujube fruit in storage. Postharvest Biol Technol 56:50–55
Zhu F, Yun Z, Ma Q, Gong Q, Zeng Y, Xu J, Cheng Y, Deng X (2015a) Effects of exogenous 24-epibrassinolide treatment on postharvest quality and resistance of Satsuma mandarin (Citrus unshiu). Postharvest Biol Technol 100:8–15
Zhu T, Tan W-R, Deng X-G, Zheng T, Zhang D-W, Lin H-H (2015b) Effects of brassinosteroids on quality attributes and ethylene synthesis in postharvest tomato fruit. Postharvest Biol Technol 100:196–204
Zhu Y, Wang B, Tang K, Hsu C-C, Xie S, Du H, Yang Y, Tao WA, Zhu J-K (2017) An arabidopsis nucleoporin NUP85 modulates plant responses to ABA and salt stress. PLoS Genet 13:e1007124
Zhu Z, Liang H, Chen G et al (2019) Isolation of the brassinosteroid receptor genes and recharacterization of dwarf plants by silencing of SlBRI1 in tomato. Plant Growth Regul 89:59–71. https://doi.org/10.1007/s10725-019-00524-z
Zou LJ, Deng XG, Le Z, Zhu T, Tan WR, Muhammad A, Zhu LJ, Zhang C, Zhang DW, Lin HH (2018) Nitric oxide as a signaling molecule in brassinosteroid-mediated virus resistance to Cucumber mosaic virus in Arabidopsis thaliana. Physiol Plant 163:196–210
Zou L, Qu M, Zeng L et al (2020) The molecular basis of the interaction between Brassinosteroid induced and phosphorous deficiency induced leaf inclination in rice. Plant Growth Regul 91:263–276. https://doi.org/10.1007/s10725-020-00604-5
Acknowledgements
We apologize to the authors whose works are not cited because of space constraints.
Funding
The National Key Research and Development Program of China (No. 2016YFD0400605) and the Academic Backbone Project of Northeast Agricultural University (15XG23) supported this work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hussain, M.A., Fahad, S., Sharif, R. et al. Multifunctional role of brassinosteroid and its analogues in plants. Plant Growth Regul 92, 141–156 (2020). https://doi.org/10.1007/s10725-020-00647-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00647-8