Abstract
Phytophthora parasitica causes serious damage to a broad spectrum of agriculturally important crops and natural ecosystems. To investigate plant responses to P. p arasitica, differential gene expressions between inoculated and mock-treated Nicotiana benthamiana leaves were analyzed by RNA-Seq approach. A total of 5375 and 3614 N. b enthamiana genes were found to be upregulated and downregulated, respectively. Infection with P. p arasitica triggered massive metabolic reprogramming in the inoculated tissues. Genes related to photosynthesis, starch biosynthesis, and nitrogen assimilation were suppressed while sucrose degrading genes were induced. Notably, plant defense responses were activated, reflected by larger number of upregulated JA and ET signaling genes, receptor-like kinases, pathogenesis-related genes, and transcription factors. Collectively, these results provide broad insights into N. b enthamiana defense mechanisms against P. p arasitca and advance our understanding of plant-Phytophthora interactions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past several centuries, the agricultural productivity was severely threatened by pathogen diseases, leading to vast economic losses every year. Plants have evolved complex mechanisms to recognize and induce defense responses to protect themselves from the threats of pathogens (Jones and Dangl 2006). The induced defenses include two layers: the first layer is triggered upon recognition of pathogen-associated molecular patterns (PAMPs) (Boller and He 2009), and the second layer of defenses is activated by special recognition of effectors from pathogens (Jones and Dangl 2006). In both layers, plants can induce defense-related physical changes containing cytoskeletal reorganization, callose depositions and cell wall thickening. Meanwhile, some biochemical responses such as generation of reactive oxygen species, production of salycylic acid, jasmonic acid or ethylene signaling compounds, are activated to perturb infection (Jones and Dangl 2006; Chisholm et al. 2006). In addition, energy balance is also critical to fight off pathogens (Scheideler et al. 2002). Therefore, changes in the transcriptomes of plants are usually significant and distinct during interactions with different pathogens (Rojas et al. 2014).
Phytophthora is an important plant pathogen genus belonging to oomycetes, and most species cause severe threats to crops and natural ecosystems (Kroon et al. 2012; Kamoun et al. 2015). Among these species, P. i nfestans and P. s ojae are two notorious pathogens with narrow host ranges, while many other Phytophthora species infect a broad spectrum of plants (Attard et al. 2008). As a consequence, an increasing attention has focused on a representative species, P. p arasitica Dastur (syn. Phytophthora nicotianae Breda de Haan). This pathogen is capable of infecting many plant species, including Arabidopsis thaliana and Nicotiana tabacum (Meng et al. 2014). It causes severe foliar, fruit, root and crown rots on solanaceous crops, as well as horticultural and fruit trees. P. p arasitica has emerged as an ideal pathogen to study because of its economic importance and broad host ranges. However, its interactions with plants have not been extensively studied.
Nicotiana b enthamiana is an important model plant for studying molecular plant-pathogen interactions (Goodin et al. 2008). Firstly, it was originally found to be infected by diverse plant viruses, leading to development of virus-induced gene silencing (VIGS), a widely used technology to decipher functions of plant genes (Purkayastha and Dasgupta 2009). Secondly, N. b enthamiana is also susceptible to a vast number of other pathogens including bacteria, fungi and oomycetes, giving rise to a system to study plant innate immunity mechanisms (Kamoun et al. 1998; Bos et al. 2010). Thirdly, due to the large leaves and its susceptibility to diverse pathogens, N. b enthamiana has been adopted as the host for transient expression using either engineered viruses or syringe-infiltration of Agrobacterium tumefaciens (Chapman et al. 1992; Ma et al. 2012). Recently, the draft genomic sequence of N. b enthamiana was released (Bombarely et al. 2012), increasingly heightening its usefulness for plant-pathogen interaction researches.
Many researchers have focused on the pathosystems of interactions between Phytophthora and model plant N. b enthamiana. Combined with great wealth of N. b enthamiana, including its natural ecotypes, genomic resources, and vast genetic and molecular tools, researchers could excavate more basic knowledge on plant immune system in response to Phytophthora attack. The majority of basic knowledge about PAMP perception and PAMP-triggered immunity (PTI), effector-triggered immunity (ETI), transcriptional regulation networks, energy arrangement, and hormone pathway signaling could be greatly accelerated. On the other side, the functions of diverse Phytophthora genes have been studied by robust genetic tools in N. b enthamiana (Dou et al. 2008; Liu et al. 2011; Chen et al. 2015), which also accelerates the functional study of virulence-related genes in Phytophthora. The study of this pathosystem is expected to facilitate understanding of Phytophthora pathogenesis and plant susceptibility or resistance responses, and ultimately lead to the development of novel strategies to control Phytophthora diseases.
In this study, RNA-Seq technology was applied to investigate the dynamic changes of the N. b enthamiana transcriptome in response to P. p arasitica. This approach allowed us to globally examine the expression changes of genes involved in primary and secondary metabolisms, defense-related pathways, and transcriptional regulation. These results could gain molecular insights into plant genes underlying the immune responses against P. p arasitica.
Materials and methods
Materials and inoculation procedures
Phytophthora parasitica isolate 025 was grown on V8 agar and cultured at 25 °C in the dark. N. b enthamiana seedlings were grown in a growth chamber at 25 °C under fluorescent white light in a 16:8-h light/dark cycle. Six-week-old N. b enthamiana leaves were detached and immersed in suspensions containing 105 zoospores of P. p arasitica. The leaves were collected at 3, 6, 9, and 12 hours post inoculation (hpi), and then P. p arasitica mycelia and N. b enthamiana cell death were visualized by staining with trypan blue staining method. Briefly, the trypan blue stock solution was prepared by mixing 10 g of phenol, 10 mL of glycerol, 10 mL of lactic acid, 10 mL of distilled water, and 0.02 g of trypan blue. The inoculated leaves in trypan blue solution were boiled in a water bath for 2 min and then incubated for 1 day. After destaining in saturated chloral hydrate solution (2.5 g chloral hydrate/1 mL water) for 2 days, the leaves were equilibrated with 70 % glycerol for photography.
Sample preparation and sequencing
The N. b enthamiana leaves at 6 hpi and the mock-treated leaves were selected for RNA-Seq sequencing. Total RNA was isolated using the PureLink RNA mini kit (Invitrogen) according to the manufacturer’s instructions and then treated with DNase I (RNase free, TaKaRa) to remove genomic DNA contaminations. The RNA samples from three biological replicates for each sample were pooled and sequenced with 100 bp paired-end reads on Illumina HiSeq 2000 platform.
Reads mapping and annotations
The produced clean reads were mapped to the N. b enthamiana V0.4.4 reference genome (https://solgenomics.net/organism/Nicotiana_benthamiana/genome) and P. p arasitica genome (https://www.broadinstitute.org) using TopHat (Trapnell et al. 2009) software with default parameters. Only the reads that could be uniquely mapped to the N. b enthamiana genome were used for subsequent processing. The retained reads were quantified using Cufflinks v1.0.3 program (Trapnell et al. 2010), and expression level of each gene was calculated by normalizing to the fragment per kilobase of exon per million mapped reads (FPKM) value. To filter out weakly expressed genes, only genes with FPKM larger than one were included in the analysis. Differentially expressed genes were identified using the GFOLD algorithm (Feng et al. 2012), which was biologically meaningful for single replicate experiments. Genes with four fold change (GFOLD > 1 or < −1) were considered differentially expressed between two samples. TransDecoder tool was applied to identify potential coding regions within novel transcripts. MapMan ontology tool was used to obtain an overview of tobacco genes involved in metabolic pathways, in which a plant-specific ontology classifies genes into well defined hierarchical categories and were denominated BINs (Thimm et al. 2004). Tobacco genes were assigned to BINs using the Mercator automated annotation pipeline. The enriched Mapman BINs were identified using Fisher’s exact test.
Validation of RNA-Seq data by qRT-PCR
To validate the RNA-Seq results, qRT-PCR was performed for six transcription factors which were induced by more than four fold after P. p arasitica infection. First strand cDNA was synthesized using an iScript cDNA Synthesis kit (Takara Bio DRR036A). N. b enthamiana ef1-α (Genbank accession no. AF120093.1) was used as an endogenous control to normalize the expression data. Quantitative real-time PCR thermal cycler conditions and reaction mixtures were performed according to the manufacturer’s instructions (SYBR® Premix Ex Taq™). The endpoint was used in the real-time PCR quantification (Livak and Schmittgen 2001), △Ct (for cycle threshold) relative quantification with reference gene normalization was performed. At least three biologically independent experiments were carried out. Pearson correlation coefficient was calculated between the fold change value of the qRT-PCR analysis and RNA-Seq analysis.
Results
P. p arasitica could infect N. b enthamiana
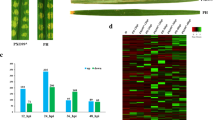
Phytophthora parasitica is a pathogen with broad host ranges (Meng et al. 2014). To test whether it could infect the model plant N. b enthamiana, the detached leaves of N. b enthamiana were immersed in P. p arasitica zoospore suspensions and phenotypes were observed. The necrosis symptoms was visually observed at 9 hpi. The leaves began to darken and wilt at 12 hpi. To investigate the infection processes, the trypan blue-stained leaves were observed. At 3 hpi, most of cysts germinated and the hyphae began to spread among plant cells. At 6 hpi, the hyphae extensively observed and could touch almost all the plant cells. Then, the necrosis occurred in only limited areas at 9 hpi and the majority of plant tissues developed necrosis at 12 hpi (Fig. 1). The results suggest that P. p arasitica is able to infect N. b enthamiana, making this pathosystem a good model to study interactions of plants and Phytophthora pathogens.
Overview of the transcriptome
Two pool samples including the inoculated and mock-treated N. b enthamiana leaves at 6 hpi were analyzed using RNA-Seq approach. Based on illumina HiSeq 2000 platform, approximately 65.6 and 52.8 million paired-end reads were produced from the two samples. The reads were then aligned onto the N. b enthamiana V0.4.4 reference genome using TopHat software, resulting in 91.5 and 85.1 % mapped reads, respectively (Table 1). Most of the mapped reads (95.6 and 95.7 %) were uniquely aligned to the genome, and were used for further analysis. To get the expression value of each N. b enthamiana gene in inoculated and mock-treated samples, the fragment per kilobase of exon per million mapped reads (FPKM) value was calculated for each gene. Of the 76,379 predicted genes in the N. b enthamiana genome (Bombarely et al. 2012), 35,180 (46.1 %) genes were expressed using FPKM larger than 1 as the cutoff for gene expression, and 26,041 genes among these were detectable in the both samples.
To evaluate the gene expression, ten housekeeping genes including tubulin, actin, and ef1-α (elongation factor 1-α) (Supplementary Table 1) were selected as described previously (Liu et al. 2012). Based on the comparison of the two samples, the log2 fold changes for the housekeeping genes ranged from −0.20 (NbS00023178g0001 coding for ef1-α) to 0.21 (NbS00033002g0006 coding for TIP41-like protein) (Supplementary Table 1), indicating that none of these housekeeping genes was significantly altered. The results reflect that the transcript data meets the requirements for the following transcriptome analysis.
Identification of novel transcripts
We observed that a small number of reads could be mapped in regions of the N. b enthamiana genome which has not yet been annotated, indicating that potential new genes could be predicted based on RNA-Seq data. In total, we identified 7547 novel transcripts based on the following filtering criteria: (1) transcript length longer than 300 bp; (2) The FPKM value was larger than 1 in at least one sample; (3) the novel transcript was at least 300 bp away from an annotated gene.
Based on previous studies, novel transcripts seem to have fewer exons and less protein-coding genes compared with the annotated genes (Bruno et al. 2010; Lu et al. 2013). Here, we found that 98.5 % of the novel transcripts had no more than three exons while this number was 57.7 % in the annotated genes (Fig. 2a). Averagely, the novel transcripts had ~1.3 exons per transcript while the annotated genes were ~4.3. The average size of the novel transcripts was 1048 bp. In total, only 1481 transcripts were predicted to encode open reading frames >100 amino acids, suggesting that others may be non-coding RNAs. As Fig. 2b shows, 1126 transcripts with >100 amino acids had blast hits in the NCBI NR database (E-value ≤1 × 10−5) and could be considered as novel protein-coding genes. More than half of these were matched to uncharacterized proteins or hypothetical proteins, while the rest had sequence similarity to polyproteins, resistance proteins, ubiquitin proteins, auxin-induced proteins and other functional proteins. Thus, the results added a large amount of unpredicted protein-encoding genes and non-coding RNAs in N. b enthamiana.
Features of novel transcripts. a Comparison of exon numbers between novel transctipts and annotated genes. b Distribution of potential coding protein length associated with corresponding blast hits in NCBI NR database. The novel transcripts which encoded open reading frames greater than 100 amino acids were used for blast analysis against NCBI NR database. The longer potential proteins had higher probability to be homologs to the known proteins
Differentially expressed genes
For further identification of differentially expressed genes (DEGs) between the two samples, four fold-change was applied with GFOLD algorithm (GFOLD > 1 or GFOLD < −1), which gives more biologically meaningful results when no replicate is available. A total of 8989 (11.8 %) DEGs were identified, including 5375 upregulated and 3614 downregulated genes in the inoculated sample (Supplementary Table 2). We used the MapMan tool to investigate how P. p arasitica infection affected the expressions of N. b enthamiana genes. The results reflected dynamic transcript changes in photosynthesis, sucrose degradation, nitrogen assimilation and defense-related pathways (Supplementary Table 3), which will be described in the following parts.
Primary and secondary metabolisms
Phytopathogen infection usually alters plant photosynthesis process (Berger et al. 2007). Here, we found that 208 N. b enthamiana genes associated with photosynthesis were found to be differentially downregulated while only one gene was upregulated (Supplementary Table 4). This phenomenon was particularly evident for genes encoding proteins in photosystem I and photosystem II reaction centres, including 58 downregulated genes participated in light harvesting complex and 53 downregulated photosystem subunit genes (Supplementary Table 4). Moreover, 43 DEGs were identified as significantly downregulated in calvin cycle, which referred to the light-independent reactions in photosynthesis. These results indicated that the photosynthetic rates of N. b enthamiana were largely reduced by the infection of P. p arasitica.
The reduction of photosynthetic metabolism and the simultaneously increased cellular demands during the defense response often initiate the transition from source status to sink status in infected tissue (Bolton 2009). Eleven genes involved in sucrose degradation were differentially expressed, in which eight were upregulated (Supplementary Table 5). For examples, three genes encoding cell wall invertases were highly induced, especially for NbS00010282g0017, whose FPKM value was over 500-folds increased. Two genes encoding hexokinases were also induced, which were reported to sense soluble hexoses (Bolton 2009). At the same time, seven upregulated hexose transporters were identified, suggesting that the plants need to transport hexoses into the cells to fulfill the energy and carbon requirements for defense responses. Conversely, the expressions of genes associated with sucrose and starch biosynthesis were significantly reduced (Supplementary Table 5).
In response to P. p arasitica infection, N. b enthamiana amino acid metabolism pathways also seemed to be differentially regulated. Genes involved in biosynthesis of asparagine, methionine, serine, and tyrosine were upregulated (Supplementary Table 6). By contrast, other amino acids including proline, isoleucine, and histidine were reduced by the observation of upregulated degrading genes (Supplementary Table 6). It is likely that the above accumulated or reduced amino acids were rearranged to participate in plant defense, and more comprehensive studies are needed to prove it. The catabolism of some amino acids may result in the release of ammonium and intermediate compounds to energy-generating pathways such as the tricarboxylic acid cycle. Nitrogen assimilation, where the inorganic nitrogen is assimilated into glutamine and glutamate, is important for plant development. However, this process was impaired in infected N. b enthamiana. Genes encoding critical enzymes for nitrogen assimilation were reduced, presented by two nitrate reductase, three nitrite reductases, and two glutamine synthetases (Supplementary Table 6).
Despite the variation of primary metabolism, plant secondary metabolism also changes based on the induction of defense programmes. In this study, a cohort of DEGs involved in secondary metabolism were identified in infected N. b enthamiana. Genes related to the biosynthesis of terpenoids, lignins and alkaloids were highly induced (Supplementary Table 7). It was previously reported that the lignin pathway was activated due to the importance of lignification and reinforcement of cell walls in the response of plants to pathgen infection (Miedes et al. 2014). Most N. b enthamiana lignin biosynthetic genes, especially six phenylalanine ammonia-lyases which played an important role in cell wall lignification, were upregulated, indicating that lignin was deposited in infected regions to avoid the spread of the pathogen.
Defense signaling pathways
Salicylic acid (SA), jasmonic acid (JA) and ethylene (ET) are three major defense hormones, which mediate plant defense responses to pathogens (Lopez et al. 2008; Spoel and Dong 2008). We identified 55 DEGs involved in ET-signaling pathway, and 44 of these coding for ethylene response factors were upregulated after P. p arasitica infection (Supplementary Table 8). Meanwhile, four 1-aminocyclopropane-1-carboxylate (ACC) oxidases and one ACC synthase were identified as upregulated in infected N. b enthamiana. The ACC oxidases and synthase are known to play important roles in ET biosynthesis (Wang et al. 2002). The hormone JA oftern acts synergistically with ET to trigger the expression of defense-related proteins. Four jasmonate ZIM-domain genes, which acted as negative regulators in the JA-signalling pathway, were identified to be downregulated. Meanwhile, the majority of JA biosynthesis genes, such as lipoxygenases and allene oxidase synthases, were upregulated (Supplementary Table 8). Paradoxically, no gene associated with SA-signaling pathway was differentially expressed, and genes involved in regulating synthesis of SA were downregulated. These results reflected that attack by P. p arasitica induced constitutive activation of ET and JA, but not SA signaling pathway.
In response to pathogens, the ability to perceive extracellular molecules and then rapidly initiate defense responses is integral to the basal plant resistance (Goff and Ramonell 2007). N. b enthamiana possesses a large superfamily of receptor-like kinases (RLKs) and receptor-like proteins (RLPs) to recognize pathogenic molecules, including PAMPs. Remarkly, 253 genes belonging to RLKs or RLPs were identified to be differentially expressed in our study, and 168 among these were upregulated (Supplementary Table 9). We discovered 56 upregulated LRR-RLKs, which comprise the largest class of RLKs. One of the best-characterized LRR-RLK was the FLAGELLIN SENSITIVE 2 (FLS2) protein, an RLK that was reported to be involved in the perception of bacterial flagellin in Arabidopsis thaliana (Gomez-Gomez and Boller 2000). Five homologs of FLS2 were identified to be highly induced in infected N. b enthamiana, suggesting similar functions as in A. t haliana. In addition, other classes of RLKs including wall associated kinases, S-locus receptor kinases, wheat LRK10-like kinases, and legume lectin kinases were mainly upregulated after P. p arasitica inoculation (Supplementary Table 9). These results indicated that different RLK classes may play important or overlapping roles in pathogen recognition and plant defense.
Defense-related genes
Pathogenesis-related (PR) genes are produced by plants to protect themselves against invading pathogens and accumulate mainly in the infected and surrounding tissues (Sels et al. 2008). Currently, PR genes were classified into 17 families based on their properties and functions. At least 140 PR genes belonging to 15 families were identified to be differentially expressed in infected N. b enthamiana (Supplementary Table 9). Most PR family members, including chitinases, thaumatin-like proteins, β-1,3-glucanases, and peroxidases, were induced. Chitinases and β-1,3-glucanases are two critical hydrolytic enzymes that play an important role in defense reaction against pathogen by degrading cell walls (Jongedijk et al. 1995). We found 13 induced β-1,3-glucanases from PR-2 family, and 16 induced chitinases from PR-3, PR-4, PR-8 and PR-11 families. Intriguingly, 22 upregulated PR-9 family genes coded for peroxidases, which were involved in cell wall lignification and enhanced resistance in plants. On the opposite, several PR families, involing subtilisin-like proteases, lipid transfer proteins and germin-like proteins, were downregulated after P. p arasitica infection (Supplementary Table 9).
Other putative defense-related genes, such as two NADPH oxidases involved in reactive oxygen species production in response to pathogens, six kunitz trypsin inhibitors with antifungal activity, were upregulated (Supplementary Table 9). These results revealed that the N. b enthamiana defense responses were active once infected by P. p arasitica.
Transcriptional regulators
Identification of transcription factors (TFs) expressed in a specific stage provides a foundation for understanding the transcriptional regulatory networks underlying the development, structure and function of the stage. Based on family assignment rules on the Plant Transcription Factor Database (http://planttfdb.cbi.pku.edu.cn), we identified 1428 TFs expressed in at least one of the two samples, and 504 of these showed differential expression, covering 40 families (Supplementary Table 10). Among the 504 TFs, 235 showed upregulation while the left 269 showed downregulation in infected N. b enthamiana (Fig. 3). Ethylene responsive factors (ERFs), involving in biotic and abiotic stress responses, composed the largest TF family in N. b enthamiana. A total of 68 ERFs were induced after P. p arasitica infection, suggesting activation of the transcription of defense-related genes. WRKY transcription factors are the second large family of regulatory proteins, which participate in regulating defense gene expression (Jing et al. 2009). We identified 50 upregulated WRKY TFs, potentially had partly redundant functions in regulating resistance to P. p arasitica. Furthermore, 30 downregulated genes were identified to encode bHLH TFs (Supplementary Table 10), acting as negative regulators of JA-mediated defense. In addition to the above three major TF families associated with plant defense, we also identified other induced TFs such as MYB, HSF, and GRAS TFs, although most of their functions were unclear.
Summary of differentially expressed transcription factors. The 504 differentially expressed transcription factors were distributed in 40 families according to family assignment rules from Plant Transcription Factor Database. The x-axis represented 40 transcription factor families. The proportions of upregulated and downregulated genes within a family were represented by blue and orange bars, respectively. The number on the top of each bar represented downregulated gene number while the number on the bottom represented upregulated gene number
To validate the differentially expressed TFs identified by RNA-Seq analysis, three ERF and three WRKY TFs involved in defense responses were evaluated by qRT-PCR. All of these selected TFs were shown to be upregulated in infected N. b enthamiana by RNA-Seq analysis (Supplementary Table 10). The validation results revealed that the genes’ differential expressions were positively correlated with qRT-PCR (r = 0.8, Fig. 4), which highlighted the accuracy and reproducibility of the RNA-Seq analysis in this study.
Validation of RNA-Seq data by qRT-PCR. The relative expression levels of six transcription factors were verified by qRT-PCR. Error bars represented the SD for three independent experiments. The y-axis represented the relative expression level. ‘r’ refers to the Pearson correlation coefficient of gene expression changes between RNA-Seq and qRT-PCR
Discussion
The molecular mechanisms related to the interactions between Phytophthora and plants are largely unknown. In this study, the model plant N. b enthamiana was applied to investigate the plant responses triggered by the inoculation of P. p arasitica using RNA-Seq technology. We found that expression profiles of N. b enthamiana genes involved in metabolism and defense pathways were altered in the infection stage. The majority of genes involved in sucrose degradation, secondary metabolism, and defense-related pathways were upregulated. In contrast, genes related to photosynthesis, starch biosynthesis, and nitrogen assimilation were downregulated. These results expanded our general understanding of plant immune responses against P. p arasitica.
Plants rely on intricate defense responses to protect themselves from the attack of pathogens. The induction of defense responses is cost-intensive, which causes increasing energy demands (Bolton 2009). Therefore, plants induce desense-related pathways while downregulate other metabolic pathways to establish a favorable energy balance for defense. In agreement with this notion, the decrease in photosynthesis has been broadly reported upon the threats of pathogens (Teixeira et al. 2014; Chandran et al. 2010). In the current study, large number of N. b enthamiana genes involved in photosynthesis were differentially downregulated after infection. Among them, most genes encoding proteins in photosystem I and photosystem II reaction centres were repressed, suggesting impairment of photosynthesis in N. b enthamiana. Moreover, it has been reported that the role of primary metabolism is to provide cellular energy demands for defense responses (Bolton 2009). Remarkably, we identified upregulated genes associated with sucrose degradation, which support energy and carbon requirements for defense responses. In this process, three cell wall invertases participated in cleaving sucorse into glucose and fructose were highly induced. Moreover, seven hexose transporters which transport the resulting hexoses into the plant cells were also upregulated. These results reflected that infected plant tissues appear to use alternative sources to obtain energy.
In general terms, plant resistance against biotrophic pathogens is mainly controlled by SA-dependent signalling pathways. On the other hand, resistance to necrotrophic pathogens involves JA and ET signalling pathways (Glazebrook 2005). In our study, P. p arasitica was a hemibiotrophic pathogen and it was still in biotrophic stage during the early infection we investigated. However, we didn’t find any difference in the expression of SA-signaling genes. In contrast, totally 55 genes involved in ET-signaling pathway were highly expressed, and majority of these were ethylene response factors. Meanwhile, most genes involved in JA biosynthesis were also upregulated. These results suggested that JA and ET signalling pathways were activated in inoculated N. b enthamiana leaves. In addition, plants produce a large family of RLKs to perceive different signals in the presence of pathogens. Among these, LRR-RLKs are the best-studied RLKs which recognize pathogen elicitors and participate in plant defense. In current study, we identified 56 upregulated LRR-RLKs, and five of these were FLS2-type receptors. The upregulation of FLS2 receptors suggest the recognization of well-conserved protein flagellin from pathogens and activation of plant initiate immunity. In response to pathogen attack, plants produce another large family of PR proteins to protect themselves. Several PR subfamilies, including chitinases, thaumatin-like proteins, β-1,3-glucanases, and peroxidases, were found to be upregulated in our study. Notably, β-1,3-glucanases participate in degradation of pathogen cell walls while peroxidases contribute to plant cell wall lignification. The upregulation of these PR proteins reflects that N. b enthamiana fights against P. p arasitica infection in different ways.
In conclusion, we present the dynamic changes of the N. b enthamiana transcriptome in response to P. p arasitica, including metabolisms, defense-related pathways, and transcriptional regulation. The above results would provide a broader view of knowledge underlying plant immune responses against P. p arasitica.
References
Attard A, Gourgues M, Galiana E, Panabieres F, Ponchet M, Keller H (2008) Strategies of attack and defense in plant-oomycete interactions, accentuated for Phytophthora parasitica Dastur (syn. P. Nicotianae Breda de Haan). J Plant Physiol 165(1):83–94
Berger S, Sinha AK, Roitsch T (2007) Plant physiology meets phytopathology: plant primary metabolism and plant-pathogen interactions. J Exp Bot 58(15–16):4019–4026
Boller T, He SY (2009) Innate immunity in plants: an arms race between pattern recognition receptors in plants and effectors in microbial pathogens. Science 324(5928):742–744
Bolton MD (2009) Primary metabolism and plant defense-fuel for the fire. Mol Plant Microbe In 22(5):487–497
Bombarely A, Rosli HG, Vrebalov J, Moffett P, Mueller LA, Martin GB (2012) A draft genome sequence of Nicotiana benthamiana to enhance molecular plant-microbe biology research. Mol Plant Microbe Interact 25(12):1523–1530
Bos JIB, Prince D, Pitino M, Maffei ME, Win J, Hogenhout SA (2010) A functional genomics approach identifies candidate effectors from the aphid species Myzus persicae (Green Peach Aphid). PLoS Genet 6(11):e1001216
Bruno VM, Wang Z, Marjani SL, Euskirchen GM, Martin J, Sherlock G, Snyder M (2010) Comprehensive annotation of the transcriptome of the human fungal pathogen Candida albicans using RNA-seq. Genome Res 20(10):1451–1458
Chandran D, Inada N, Hather G, Kleindt CK, Wildermuth MC (2010) Laser microdissection of Arabidopsis cells at the powdery mildew infection site reveals site-specific processes and regulators. Proc Natl Acad Sci USA 107(1):460–465
Chapman S, Kavanagh T, Baulcombe D (1992) Potato virus X as a vector for gene expression in plants. Plant J 2(4):549–557
Chen XR, Li YP, Li QY, Xing YP, Liu BB, Tong YH, Xu JY (2015) SCR96, a small cysteine-rich secretory protein of Phytophthora cactorum, can trigger cell death in the Solanaceae and is important for pathogenicity and oxidative stress tolerance. Mol Plant Pathol. doi:10.1111/mpp.12303
Chisholm ST, Coaker G, Day B, Staskawicz BJ (2006) Host-microbe interactions: shaping the evolution of the plant immune response. Cell 124(4):803–814
Dou D, Kale SD, Wang X, Chen Y, Wang Q, Jiang RH, Arredondo FD, Anderson RG, Thakur PB, McDowell JM, Wang Y, Tyler BM (2008) Conserved C-terminal motifs required for avirulence and suppression of cell death by Phytophthora sojae effector Avr1b. Plant Cell 20(4):1118–1133
Feng J, Meyer CA, Wang Q, Liu JS, Shirley Liu X, Zhang Y (2012) GFOLD: a generalized fold change for ranking differentially expressed genes from RNA-seq data. Bioinformatics 28(21):2782–2788
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Goff KE, Ramonell KM (2007) The role and regulation of receptor-like kinases in plant defense. Gene Regul Syst Biol 1:167–175
Gomez-Gomez L, Boller T (2000) FLS2: an LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol Cell 5(6):1003–1011
Goodin MM, Zaitlin D, Naidu RA, Lommel SA (2008) Nicotiana benthamiana: its history and future as a model for plant-pathogen interactions. Mol Plant Microbe Interact 21(8):1015–1026
Jing SJ, Zhou X, Song Y, Yu DQ (2009) Heterologous expression of OsWRKY23 gene enhances pathogen defense and dark-induced leaf senescence in Arabidopsis. Plant Growth Regul 58(2):181–190
Jones JDG, Dangl JL (2006) The plant immune system. Nature 444(7117):323–329
Jongedijk E, Tigelaar H, Vanroekel JSC, Bresvloemans SA, Dekker I, Vandenelzen PJM, Cornelissen BJC, Melchers LS (1995) Synergistic activity of chitinases and beta-1,3-glucanases enhances fungal resistance in transgenic tomato plants. Euphytica 85(1–3):173–180
Kamoun S, van West P, Vleeshouwers VGAA, de Groot KE, Govers F (1998) Resistance of Nicotiana benthamiana to Phytophthora infestans is mediated by the recognition of the elicitor protein INF1. Plant Cell 10(9):1413–1425
Kamoun S, Furzer O, Jones JD, Judelson HS, Ali GS, Dalio RJ, Roy SG, Schena L, Zambounis A, Panabieres F, Cahill D, Ruocco M, Figueiredo A, Chen XR, Hulvey J, Stam R, Lamour K, Gijzen M, Tyler BM, Grunwald NJ, Mukhtar MS, Tome DF, Tor M, Van Den Ackerveken G, McDowell J, Daayf F, Fry WE, Lindqvist-Kreuze H, Meijer HJ, Petre B, Ristaino J, Yoshida K, Birch PR, Govers F (2015) The Top 10 oomycete pathogens in molecular plant pathology. Mol Plant Pathol 16(4):413–434
Kroon LP, Brouwer H, de Cock AW, Govers F (2012) The genus Phytophthora anno 2012. Phytopathology 102(4):348–364
Liu T, Ye W, Ru Y, Yang X, Gu B, Tao K, Lu S, Dong S, Zheng X, Shan W, Wang Y, Dou D (2011) Two host cytoplasmic effectors are required for pathogenesis of Phytophthora sojae by suppression of host defenses. Plant Physiol 155(1):490–501
Liu D, Shi L, Han C, Yu J, Li D, Zhang Y (2012) Validation of reference genes for gene expression studies in virus-infected Nicotiana benthamiana using quantitative real-time PCR. PLoS ONE 7(9):e46451
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408
Lopez MA, Bannenberg G, Castresana C (2008) Controlling hormone signaling is a plant and pathogen challenge for growth and survival. Curr Opin Plant Biol 11(4):420–427
Lu X, Chen D, Shu D, Zhang Z, Wang W, Klukas C, Chen LL, Fan Y, Chen M, Zhang C (2013) The differential transcription network between embryo and endosperm in the early developing maize seed. Plant Physiol 162(1):440–455
Ma L, Lukasik E, Gawehns F, Takken FL (2012) The use of agroinfiltration for transient expression of plant resistance and fungal effector proteins in Nicotiana benthamiana leaves. Methods Mol Biol 835:61–74
Meng Y, Zhang Q, Ding W, Shan W (2014) Phytophthora parasitica: a model oomycete plant pathogen. Mycology 5(2):43–51
Miedes E, Vanholme R, Boerjan W, Molina A (2014) The role of the secondary cell wall in plant resistance to pathogens. Front Plant Sci 5:358
Purkayastha A, Dasgupta I (2009) Virus-induced gene silencing: a versatile tool for discovery of gene functions in plants. Plant Physiol Bioch 47(11–12):967–976
Rojas CM, Senthil-Kumar M, Tzin V, Mysore KS (2014) Regulation of primary plant metabolism during plant-pathogen interactions and its contribution to plant defense. Front Plant Sci 5:17
Scheideler M, Schlaich NL, Fellenberg K, Beissbarth T, Hauser NC, Vingron M, Slusarenko AJ, Hoheisel JD (2002) Monitoring the switch from housekeeping to pathogen defense metabolism in Arabidopsis thaliana using cDNA arrays. J Biol Chem 277(12):10555–10561
Sels J, Mathys J, De Coninck BM, Cammue BP, De Bolle MF (2008) Plant pathogenesis-related (PR) proteins: a focus on PR peptides. Plant Physiol Bioch 46(11):941–950
Spoel SH, Dong X (2008) Making sense of hormone crosstalk during plant immune responses. Cell Host Microbe 3(6):348–351
Teixeira PJ, Thomazella DP, Reis O, do Prado PF, do Rio MC, Fiorin GL, Jose J, Costa GG, Negri VA, Mondego JM, Mieczkowski P, Pereira GA (2014) High-resolution transcript profiling of the atypical biotrophic interaction between Theobroma cacao and the fungal pathogen Moniliophthora perniciosa. Plant Cell 26(11):4245–4269
Thimm O, Blasing O, Gibon Y, Nagel A, Meyer S, Kruger P, Selbig J, Muller LA, Rhee SY, Stitt M (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37(6):914–939
Trapnell C, Pachter L, Salzberg SL (2009) TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25(9):1105–1111
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28(5):511–515
Wang KL, Li H, Ecker JR (2002) Ethylene biosynthesis and signaling networks. Plant Cell 14(Suppl):S131–S151
Acknowledgments
This research was supported by grants from the National Natural Science Foundation of China (31501589) and China Postdoctoral Science Foundation (2015M571769).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shen, D., Chai, C., Ma, L. et al. Comparative RNA-Seq analysis of Nicotiana benthamiana in response to Phytophthora parasitica infection. Plant Growth Regul 80, 59–67 (2016). https://doi.org/10.1007/s10725-016-0163-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-016-0163-1