Abstract
The Indo-Pacific is an area of intense ecological interest, not least because of the region’s rich biodiversity. Important insights into the origins, evolutionary history, and maintenance of Indo-Pacific reef faunas depend upon the analysis of faunal occurrences derived from detailed stratigraphic sections. We investigated Neogene origination and extinction patterns derived from a combination of new coral occurrences and previously published records from the central Indo-West Pacific Ocean (cIWP, Indonesia, Papua New Guinea and Fiji). Two faunal turnover events were observed. In the first, an increase in generic richness of Scleractinia from the cIWP during the middle Miocene (17–14 Ma) coincided with both large-scale sea level fluctuations and the great Mid-Miocene collision event. We raise the hypothesis that Mid-Miocene origination was facilitated by habitat and population fragmentation associated with tectonism and sea level fall. The second, subsequent, turnover event was characterized by an overall lowering of generic diversity throughout the late Miocene and Pliocene (7–3 Ma), and was followed by a pronounced pulse of extinction at the Pliocene–Pleistocene boundary (~2.6 Ma). With the exception of the onset of Pleistocene sea-level cycles and the onset of northern hemisphere glaciation around 2.5 Ma, which might explain increased extinction during this time interval, there are no tectonic, eustatic, climatic or oceanographic events that neatly coincide with this second episode of Neogene coral taxonomic turnover. Our results reveal a total of 62 genera, including synonyms, from the Miocene to the Pleistocene. Neither episode of turnover among coral genera is exactly coincident with turnover in the Atlantic thus regional environmental change is found to drive Neogene reef dynamics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The evolutionary history of coral reefs over the past 65 My has been characterized by large-scale shifts in biodiversity associated with major tectonic events (Renema et al. 2008). The last of these shifts left a biodiversity hotspot in the Neogene of the Indo-Pacific region that endures to the present. While major tectonic events surely play a role in the large-scale evolution of tropical biodiversity, the drivers of regional biodiversity and evolutionary turnover is largely unstudied in the Indo-Pacific region. Indeed, evolution of the hard corals, or Scleractinia, has focused largely on sample locations in the northern hemisphere (e.g. the Atlantic, Caribbean and East Pacific), and much is known about the origins and dynamics of reefs in the western Atlantic (Budd and Johnson 1999; Budd et al. 1996, 1998; Johnson 2001; Johnson et al. 1995; O’Dea et al. 2007). In contrast, there is a paucity of similar information available for the Indo-Pacific (though see Renema et al. 2008), despite the fact that the Indo-Pacific is presently the global centre for marine biodiversity (Barber 2009; Hoeksema 2007; Roberts et al. 2002).
Scleractinian corals, the dominant frame-builder on modern coral reefs, first became important in the shallow seas of the Tethys during the Mesozoic, where they quickly evolved into a pan-tropical fauna (Potts 1983). Most modern families and genera were present by the middle Cenozoic. During the Oligocene (~34–23 Ma), closure of the Tethys (Rogl and Steininger 1984; Rogl 1999; Bunje and Lindberg 2007) meant that the original Tethyan fauna divided into the two unique faunas of the modern tropical Atlantic and the Indo-Pacific regions (Stehli and Wells 1971; Vaughan and Wells 1943; although see Harzhauser et al. 2002 for a comprehensive review of biogeographic events in the Paratethys/Mediterranean during the Oligocene–Miocene). Indo-Pacific coral communities had been isolated from the Tethyan pan-tropical fauna since sometime before the early Eocene, and the present faunal distribution of corals in the Indo-Pacific was established by the early Miocene (Rosen and Smith 1988). First appearances of Scleractinia in the Palaeocene Indo-Pacific (Wilson and Rosen 1998) were followed by a rapid increase in generic diversity in the region during the Neogene (Rosen 1988a, b; Rosen and Smith 1988; Wilson and Rosen 1998). Thus sometime from the late Oligocene/early Miocene, modern patterns of high coral diversity began to emerge in the Indo-West Pacific (Wilson and Rosen 1998).
Predicting the response of ecosystems to environmental change is difficult given both the enormous complexity of ecological interactions and the challenges of obtaining detailed stratigraphic/chronologic control of both drivers and ecological events (O’Dea et al. 2007). The added factors of lag times and non-linearity in ecosystem response to environmental change make correlation between evolutionary events and environmental drivers even more challenging, and increase difficulty in assigning causation (e.g. O’Dea and Jackson 2009). In this study, we begin to unravel the relationship between environment and evolution in Neogene Indo-Pacific coral reefs by making detailed observations of faunal change in a region of the cIWP encompassed by Indonesia, Papua New Guinea, and Fiji. We then discuss these changes in the context of documented physical changes. Specifically, we investigate coral generic richness and faunal turnover (patterns in origination and extinction) in the cIWP from the early Miocene to the early Quaternary covering from 17 to 1.8 Ma (see Bromfield and Renema 2011 for age limitations). We search for global (e.g. climate change) and regional (e.g. tectonic) environmental correlates of evolutionary change in reef corals. To enhance our environmental interpretations and to investigate the potential for common global drivers in the Indo-Pacific and Caribbean, we compare the timing and potential environmental drivers of corresponding evolutionary patterns in the cIWP with those previously documented in the Neogene and early Quaternary in the West Atlantic.
Materials and methods
Study sites
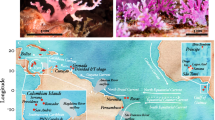
We sampled Neogene strata from three cIWP sites (Fig. 1), including the Salayar Limestone from Salayar Island in Indonesia (Fig. 1a); the Yalam Limestone from East New Britain, Papua New Guinea (P.N.G.; Fig. 1b); and the Daliconi and Futuna Limestone members of the Tokelau Limestone Group from Vanua Balevu in the Lau Group, Fiji (Fig. 1c). In these locations, rock formations with coral bearing reef deposits are uplifted and accessible above sea level. Each locality has been described within a precise chronological framework: corals from the Salayar Limestone grew between 5.8 and 1.4 Ma, those from the Yalam limestone grew between 14.8 and 12.3 Ma and those from the Tokelau Limestone Group grew between 16.9 and 6.7 Ma (Bromfield and Renema 2011; and references therein). Sedimentary beds from all localities formed in similar depositional environments, either in the intermediate or lower reef slope, in water depths ranging from 10 to 30 m (Table 1: Bromfield and Renema 2011).
Regional maps showing the location of sampled sections. a Salayar, South Sulawesi, with the Benteng West section (ISS 1) and Toto section (ISS 2); b East New Britain, Papua New Guinea (P.N.G.), with the Ramanadu section (P.N.G. 1) and Malasait section (P.N.G. 2); c Vanua Balevu, Lau Group, Fiji, with the Daliconi section (FLV 1), Namalata section (FLV 2) and Wai-Ruku section (FLV 3). From Bromfield and Renema (2011)
The Salayar Limestone is a massive coralliferous limestone and calcarenite, with intercalations of marl and calcareous sandstone (Koswara et al. 1994; Sukamto and Supriatna 1982; Wilson 2002). The Yalam Limestone is a bioclastic limestone with thickly interbedded clayey biomicrite and chalky limestone (Lindley 1988). The Tokelau Limestone is a sporadically coralliferous, massive, recrystallised biogenic limestone with local volcanogenic debris (Rodda 1994).
Sampling
Compilations of coral genera were derived from three new sampling locations in the central Indo-West Pacific Ocean (cIWP). At each location, replicate sections were established and stacked horizontal belt transects were used to characterize genus abundance patterns of coral communities at each section. In Indonesia, we sampled four transects in two sections: in Fiji, we sampled three transects in two sections and four transects at a third section: and in New Britain we sampled four transects in one section and three transects in a second section (Fig. 2). Corals were collected along time standardised transects (1 h per 20 m) and transect length (Table 2) was determined by the extent of the exposed section. All corals encountered within 1 m above and below the transect tape were collected for subsequent identification, and the number of taxa per section are given in Table 2. The number of stacked transects was determined by the thickness and accessibility of each geological section. This sampling design, with multiple stacked transects per location, is essential for calculating stratigraphic range when fossil horizons are separated by unequal temporal intervals (see Marshall 1990, 1994).
Taxonomic classification of genera followed Vaughan and Wells (1943), Wells (1956), Reyes-Bonilla (2002), and Baron-Szabo (2006), with the addition of microskeletal information provided by Chevalier and Beauvis (1987). Further fossil identification was made with the aid of the Neogene Marine Biota of Tropical America (NMITA) database (Budd et al. 2006), and the Scleractinia of Eastern Australia, Parts 1–5 (Veron and Pichon 1976, 1979, 1982; Veron et al. 1977; Veron and Wallace 1984). Specimens were compared with type specimens of Neogene Indo-Pacific corals held at the Naturalis Museum, Leiden, Netherlands.
Data analysis
We constructed taxonomic accumulation curves to establish the adequacy of our sampling effort. Accumulation curves were generated using presence/absence of genera in the Mao Tau estimator of EstimateS 8 (Colwell et al. 2004) with 999 random permutations to define curves for samples from each location.
To examine generic richness throughout the Neogene, we calculated two diversity indices, Chao 2 (Chao 1984, 1987) and the incidence based coverage estimate (ICE mean) (Chao et al. 2000; Chazdon et al. 1998), for our new collections, and we plotted these through time using EstimateS (Colwell 2005). For each analysis, the number of runs was set to 1 and samples were arranged in ascending order of age [age values were assigned using published Sr isotopic ages (Bromfield and Renema 2011)]. From this we assigned the predicted richness values to the appropriate time interval of the sample. Values for Chao2 were calculated using the classic algorithm in EstimateS (Colwell et al. 2004). The Chao2 index augments the observed number of specimens according to the number of unique or duplicate genera within the dataset (Colwell and Coddingham 1996). Upper and lower 95% confidence intervals were generated for the Chao2 index. In contrast, the ICE mean normalises collections according to the presence or absence of rare genera, and tends to provide a higher estimate of richness than other methods (Colwell 2005; Colwell et al. 2004).
Literature survey
In order to investigate the history of faunal turnover in coral genera in the cIWP during the Neogene and early Quaternary, and to establish the first and last appearances of coral genera, field data were combined with information on coral occurrences from Indonesia, Papua New Guinea and Fiji, derived from the literature (Dana 1846; Felix 1898, 1913, 1915, 1921; Gerth 1921a, b, 1923, 1925, 1930; Hoffmeister 1942; Lamarck 1801, 1816; Martin 1880; Michelin 1840–1847; Milne Edwards and Haime 1848; Orbigny 1849; Osberger 1946; Quelch 1886; Reuss 1867; Umbgrove 1939, 1942, 1945, 1946a, b, 1950; Veron and Kelly 1988; Veron and Pichon 1976, 1979, 1982; Veron et al. 1977; Veron and Wallace 1984; Verrill 1865; Wilson 1995; Wilson and Rosen 1998). The records of fossil cIWP corals have been checked for synonyms by comparing notes and diagrams from the published literature, and in some cases by comparing type specimens with published records, to avoid duplication of generic ranges. The combined data was used to construct range charts for Scleractinia in the cIWP, using the “long range” method that extends the range of a taxon to the end of the stage in which it occurred (see Johnson 2001). Appendix 1 is a checklist of coral genera identified from our new collections, along with those reported previously in the literature.
The combined data was also used to plot the generic first and last appearance datums (FAD’s and LAD’s) to investigate temporal patterns in origination and extinction. FADs and LADs are endpoints of time intervals spanning taxon occurrences in the fossil record, and provide the taxon range (Sadler 2004). Extended taxon ranges were plotted in 2 Myr bins. For example, a genus known to have existed in the Langhian (middle Miocene; 15.97–13.65 Ma), would be allocated a position in the 17–15 Ma, and 15–13 Ma bins. This has the effect of smoothing the data; that is, capturing the important patterns, while eliminating noise. Combining prior records of generic occurrences from the Indo-Pacific, with our own new dataset helps to minimize the potential for observed patterns to result from overprinting of sampling locality and taphonomy and not be representative of Neogene diversity trends.
Results
The highest number of genera were collected from Indonesia with 30 along 8 transects. Twenty-three genera along 7 transects were collected from both Fiji and P.N.G. (Fig. 3). The genus accumulation curve for samples collected from P.N.G. appears to level after 6 transects, while the curves for samples collected from Indonesia and Fiji have a slightly shallower incline towards their asymptotes, indicating slight under-representation of genera (Fig. 3).
Estimated generic richness fluctuated between 30 and 40 (95% CI—50) from 17 to 14 Ma (Fig. 4). Richness then remained constant, at ~38 (95% CI—50) genera for much of the rest of the Miocene, until a peak in generic richness of ~40 (95% CI—70) occurred ~7–5 Ma. This was followed by a decline over the Miocene-Pliocene boundary that continued until 1.8 Ma. After the beginning of the Pleistocene, the number of genera rapidly increased to ~45 (95% CI—120).
Using the combination of literature-based and field-based generic compilations, we found two episodes of faunal turnover in the central Indo-West Pacific region (Fig. 5). The first was an increase in origination during the middle Miocene, when fourteen new genera arose between 17 and 14 Ma (Fig. 5a). The second was an episode of stepwise origination of new coral genera and the simultaneous extinction of others during the late Miocene and Pliocene (7–3 Ma), followed by the extinction of genera at the Pliocene–Pleistocene boundary (~2.6 Ma: Fig. 5b).
Range charts of 62 Miocene to early Quaternary coral genera, from the central Indo-West Pacific (Indonesia, P.N.G. and Fiji), arranged to highlight a first appearances and b last appearances. Graph compiled using data on generic occurrences collated from the literature, shown as faint grey lines (see “Methods” for references), and from specimens collected during this study, shown as solid black lines. Numbers on the x-axis correspond to the list of genera given in the appendix. Pli Pliocene and Q Quaternary
Nine new coral genera arose after the end of the Miocene, ~5 Ma, while five genera became extinct during the late Miocene, between 11 and 5 Ma, and 12 became extinct during the Pliocene, between 5 and ~2 Ma (Fig. 6b). Sampling effort shows a positive relationship with generic diversity (Fig. 6a), so under-sampled times (including the gap between 9 and 7 Ma) need to be targeted to verify the observed turnover events.
Evolutionary turnover in Neogene to early Quaternary coral genera from the central Indo-West Pacific during the last 17 million years; a the number of occurrences of genera relative to the number of specimens collected during this study, and b first and last appearances of coral genera in the west-central Indo-Pacific (Indonesia, P.N.G., and Fiji) through time, compiled from the literature (see in-text for references), and from our new collections. Ple Pleistocene
Discussion
By expanding the range of some genera (for example Madracis, Turbinaria and Ctenella), we demonstrate that some genera in the cIWP may have had an older and more widespread distribution than previously thought. The precise chronostratigraphic and biostratigraphic framework of this study makes the patterns shown here more robust than was previously possible, although we recognise that they are likely to underestimate the magnitude of events.
Taxonomic turnover in the central Indo-West Pacific
The evolutionary history of coral reef organisms over broad time scales is generally related to one or more physical factors: tectonic, eustatic, climatic or oceanographic (TECO) (Rosen 1984). In this study, we recognize two episodes of taxonomic turnover in the central Indo-West Pacific Ocean. The first was an increase in origination during the early to middle Miocene (17–14 Ma) and the second an episode of stepwise origination with contemporaneous extinctions during the late Miocene and Pliocene (7–3 Ma) (Figs. 4a, 5b). The second event was followed by the increased extinction of genera at the Pliocene–Pleistocene boundary (~2.6 Ma: Figs. 4b, 5b). The limited geographic coverage of our analysis makes it difficult to ascribe individual coral turnover events to coincident environmental factors, and the limited temporal resolution makes it difficult to accurately infer the timing of regional events. However, our results do lend themselves to a discussion of the concomitant TECO framework in the Indo-Pacific region, in order to determine the environmental context of these turnover events.
The early Miocene (23–16 Ma) of the Indo-Pacific region, was characterized by an order-of-magnitude expansion of shallow-carbonate areas through extensive reef development (Wilson 2008). The peak of the Middle Miocene Climatic Optimum (MMCO) occurred at 15 Ma (Fig. 7) and was charactaerized by warm temperatures and minimal global ice (Warny et al. 2009). However, there followed a long-term cooling period from 14.7 to 13.9 Ma, with fluctuating “Icehouse” conditions (Holbourn et al. 2005, 2007). These changes in climatic conditions are associated with increased coral origination in the latter phases of the middle Miocene, with lowered or variable sea level reducing genetic exchange among populations. Another important TECO event coincident with the first turnover event in the early to middle Miocene (17–14 Ma), was initiation of the so-called ‘Great Mid-Miocene collision event’ around 15 Ma, a progressive collision between the Australian/New Guinea component of Gondwana and the Asian component of Laurasia (Charlton 2000). This collision likely began as far back as 25 Ma, and continued progressively throughout the Miocene (Hall and Wilson 2000) but there is some agreement that a significant component of this collision event occurred 15–14 Ma (Elburg et al. 2002 and references therein). It resulted in greater habitat differentiation and again, potential fragmentation of established coral populations (Pandolfi 1993). We thus hypothesize that our middle Miocene origination event was facilitated by tectonically-induced fragmentation of shallow water habitat in areas already replete with reef growth.
Major taxonomic turnover and environmental events in Neogene to Quaternary coral reefs of the central Indo-West Pacific Ocean and Caribbean Sea. Plot includes geological timescale (Gradstein et al. 2004), Oxygen isotope curve (Zachos et al. 2001) as a proxy for sea surface temperature, sea level curves (Haq et al. 1987), and significant climatic and tectonic events of the Neogene. Light grey bars represent permanent ice sheets, while thin black bars represent ephemeral ice formation at the poles. Ple Pleistocene
The second episode of faunal turnover in the cIWP comprised the origination of new coral genera and the extinction of others during the late Miocene and Pliocene (7–3 Ma), and was followed by a pronounced pulse of extinction at the Pliocene–Pleistocene boundary (~2.6 Ma), which probably continued into the Pleistocene (see Veron and Kelly 1988). With the exception of the onset of Pleistocene sea-level cycles and northern hemisphere glaciation around 2.5 Ma, which might explain increased extinctions during this time interval, there are no TECO events that neatly coincide with this second episode of Neogene coral taxonomic turnover. Restriction of the Indonesian Throughflow (ITF) likely began in the early Miocene but peaked from 10 Ma to between 5 and 4 Ma (Kuhnt et al. 2004). Then during the Pliocene Climatic Optimum (PCO: 4.5–3 Ma), climate was considerably warmer than present, with global temperatures up to 3°C higher than modern temperatures (Ravelo et al. 2004; Wara et al. 2005), CO2 concentrations ~30% higher than pre-industrial levels (Filipelli and Flores 2009; Raymo et al. 1996), and sea level up to 20 m higher than present (Ravelo et al. 2004). But polythermal conditions changed to dryer, colder conditions from the middle to late Pliocene ~5–2 Ma (Rebesco and Camerlenghi 2008), culminating in the formation of extensive ice sheets in the Northern Hemisphere by ~2.5 Ma (Mudelsee and Raymo 2005; Raymo 1994; Wara et al. 2005). However, in the equatorial Pacific, sea surface temperatures apparently remained constant during this period of cooling. This was also a time of rapid sea-level fluctuation (Naish and Wilson 2009).
It is tempting to postulate that taxonomic turnover in the cIWP occurred initially as a response to changing tectonic regimes (see Hall 2001, 2002) throughout the Cenozoic, and later as a response to rapid and repeated sea level and sea surface temperature fluctuations in the Pliocene (Rosen 1988a, b), and further to sea-level fluctuations resulting from Pleistocene glacial cycles (Mora et al. 2003). However, these interpretations must remain as working hypotheses for the evolutionary history of Indo-Pacific coral reefs, which must be further explored through greater spatial and temporal sampling of reef faunas. Finding drivers for this second episode of faunal turnover must await finer-scale taxonomy, broader sampling and higher resolution environmental chronologies than are presently available.
Comparison with the Caribbean Sea
The biological response of the Neogene biota of the Caribbean to environmental change is well known (Budd et al. 1996; Coates et al. 1992; Jackson and Johnson 2000; Johnson and Kirby 2006; Klaus et al. 2008; O’Dea et al. 2007). Three major coral turnover events have been documented in the Caribbean, but they are not strictly coincident with those from the Indo-Pacific. The first event occurred between 24 and 16 Ma, with numerous genera undergoing range restriction and regional extinction (Edinger and Risk 1994, 1995; Stehli and Wells 1971). The second event was characterized by heightened origination among genera in the late Miocene ~10 Ma (Collins et al. 1996) and the third was a pulse of extinctions in the Pliocene–Pleistocene between 4 and 1 Ma (Budd et al. 1994; Jones and Hasson 1985). During this third event, no new genera arose, and even though numerous genera once again underwent regional extinction and range migration, many are still extant in the Indo-Pacific.
Similar to potential factors governing turnover in the middle Miocene of the cIWP, turnover in the reef fauna of the Caribbean Sea between 24 and 16 Ma has also been attributed to tectonically derived local changes in ocean circulation and upwelling in the Miocene (Edinger and Risk 1994, 1995). In the late Miocene, the initial constriction of the American Seaway began to increase the carbonate content of the sediments and induced origination among reef corals (Collins et al. 1996). Final closure of the Panama seaway in the Pliocene then affected global thermohaline circulation and contributed to a reorganisation of oceanic and atmospheric systems (Filipelli and Flores 2009). Thus, the extinctions from 4 to 1 Ma in the Neogene reef fauna of the Caribbean Sea have been attributed to collapse in productivity as a result of the closure of the Isthmus of Panama (Jackson et al. 1996; O’Dea and Jackson 2009) in the Pliocene–Pleistocene, though environmental change per se, remains an insufficient explanation (O’Dea et al. 2007). O’Dea and Jackson (2009) found that species of bryozoans that failed to adjust their reproductive strategies and morphologies in response to the decline in productivity suffered an immediate decline, then lingered in the environment for 2–1 My, which illustrates the observed lag in time between the onset of environmental change and the onset of extinctions in the region (Budd and Johnson 1999). It is interesting to note that in the Caribbean, while the corals were hard hit, the record of their demise does not explain the biogeographic history of the region well. It was necessary to examine other organisms (molluscs; calcareous algae) at the level of species, and with a clear understanding of their biology and life histories (see Jackson et al. 1995; Jackson and Johnson 2000; O’Dea et al. 2007; O’Dea and Jackson 2009) before a clear picture of the region could be developed.
The timing of turnover events in the Caribbean Sea (e.g. Budd 2000; Budd et al. 1996; Jackson and Johnson 2000; Johnson 2001) and those found in the present study of the cIWP are not temporally concordant, so that turnover events cannot have been a response to mechanisms operating on a global scale. While both Indo-Pacific and Caribbean reefs were characterized by multiple turnover episodes and substantial variability in generic diversity from the Neogene to the early Quaternary, independent regional tectonic and climatic drivers appear to have controlled these events. Future work on Indo-Pacific reefs using multiple taxa at a variety of levels, from family down to species level, should help to clarify the extent to which evolutionary trends in the two oceans are the result of similar drivers.
References
Barber PH (2009) The challenge of understanding the coral triangle. J Biogeogr 36:1845–1846
Baron-Szabo RC (2006) Corals of the K/T-boundary: scleractinian corals of the suborders Astrocoeniina, Faviina, Rhipidogyrina and Amphiastraeina. J Syst Palaeontol 4(1):1–108
Bromfield K, Renema W (2011) Comparison of Sr/Sr isotope and biostratigraphic ages of uplifted fossil reefs in the Indo-Pacific: Indonesia, Papua New Guinea and Fiji. Aust J Earth Sci 58(1):61–87
Budd AF (2000) Diversity and extinction in the cenozoic history of Caribbean reefs. Coral Reefs 19(1):25–35
Budd AF, Johnson KG (1999) Origination preceding extinction during late cenozoic turnover of Caribbean reefs. Paleobiology 25(2):188–200
Budd AF, Stemann TA, Johnson KG (1994) Stratigraphic distributions of Neogene to Recent Caribbean reef corals: a new compilation. J Paleontol 68:951–959
Budd AF, Johnson KG, Stemann TA (1996) Plio-pleistocene turnover and extinctions in the Caribbean reef-coral fauna. In: Jackson JBC, Budd AF, Coates A (eds) Evolution and environment in tropical America. The University of Chicago Press, Chicago, pp 168–204
Budd AF, Petersen R, McNeill DF (1998) Stepwise faunal change during evolutionary turnover: a case study from the Neogene of Curacao, Netherlands Antilles. Palaios 13:170–188
Budd AF, Foster CTJ, Adrain T, Dawson JP (2006) Neogene marine biota of tropical America. World wide website at http://nmita.geology.uiowa.edu
Bunje PME, Lindberg DR (2007) Lineage divergence of a freshwater snail clade associated with post-Tethys marine basin development. Mol Phylogenet Evol 42(2):373–387
Chao A (1984) Non-parametric estimation of the number of classes in a population. Scand J Stat 11:265–270
Chao A (1987) Estimating the population size for capture-recapture data with unequal catchability. Biometrics 43(4):783–791
Chao A, Hwang W-H, Chen Y-C, Kuo C-Y (2000) Estimating the number of shared species in two communities. Statistica Sinica 10:227–246
Charlton TR (2000) Tertiary evolution of the eastern Indonesian collision complex. J Asian Earth Sci 18(5):603–631
Chazdon RL, Colwell RK, Denslow JS, Guariguata MR (1998) Statistical methods for estimating species richness of woody regeneration in primary and secondary rain forests of NE Costa Rica. In: Dallmeier F, Comiskey JA (eds) Forest biodiversity research, monitoring and modelling: conceptual background and old world case studies. Parthenon Publishing, Paris, pp 285–309
Chevalier JP, Beauvis L (1987) Systematique. In: Grasse P-P (ed) Traite de Zoologie: Cnidaires. Masson
Coates A, Jackson JBC, Collins LS, Cronin TM, Dowsett HJ, Bybell LM, Jung P, Obando JA (1992) Closure of the Isthmus of Panama: the near-shore marine record of Costa Rica and western Panama. Geol Soc Am Bull 104:814–828
Collins LS, Budd AF, Coates A (1996) Earliest evolution associated with closure of the tropical American Seaway. Proc Natl Acad Sci USA 93:6069–6072
Colwell RK (2005) EstimateS: statistical estimation of species richness and shared species from samples. Version 7.5. user’s guide and application published at http://purl.oclc.org/estimates
Colwell RK, Coddingham JA (1996) Estimating terrestrial biodiversity through extrapolation. In: Hawksworth DL (ed) Biodiversity: measurement and estimation. Chapman and Hall, London
Colwell RK, Mao CX, Chang J (2004) Interpolating, extrapolating, and comparing incidence-based species accumulation curves. Ecology 85(10):2717–2727
Dana JD (1846) Zoophytes. Volume VII of the United States exploring expedition. During the years 1838, 1839, 1840, 1841, 1842. Under the command of Charles Wilkes. Lea and Blanchard, Philadelphia
Edinger EN, Risk MJ (1994) Oligocene-Miocene extinction and geographic restriction of Caribbean corals: roles of turbidity, temperature, and nutrients. Palaios 9(6):576–598
Edinger EN, Risk MJ (1995) Preferential survivorship of brooding corals in a regional extinction. Paleobiology 21(2):200–219
Elburg MA, Van Leeuwen T, Foden J, Muhardjo (2002) Origin of geochemical variability by arc–continent collision in the Biru Area, Southern Sulawesi (Indonesia). J Petrol 43(4):581–606
Felix J (1898) Beitrage zur Kenntniss der Astrocoeninae. Deut geol Ges Zeitschr 1:247–256 pl 11
Felix J (1913) Die fossilen Anthozoen aus der Umgegend von Trinil. Palaeontographica 60:311–365
Felix J (1915) Jungtertiäre und quartäre Anthozoën von Timor und Obi I. Theil. Paläontologie von Timor 2:3–45
Felix J (1921) Fossile Anthozoën von Borneo. Paläontologie von Timor 9(15):1–61
Filipelli GM, Flores J-A (2009) From the warm pliocene to the cold pleistocene: a tale of two oceans. Geology 37(10):959–960
Gerth H (1921a) Coelenterata. Sammlungen des geologischen Reichs-Museums in Leiden. Neue Folge 1(2):387–445
Gerth H (1921b) Die Anthozoen der Dyas von Timor. Paläontologie von Timor 9:67–147
Gerth H (1923) Die Anthozoenfauna des Jungtertiärs von Borneo. Serie 1. Sammlungen des geologischen Reichs-Museums in Leiden. Neue Folge 10:37–136
Gerth H (1925) Jungtertiäre Korallen von Nias, Java und Borneo, nebst einer Uebersicht über die as dem Känozoikum des Indischen Archipels bekannten Arten. Leidsche Geologische Mededeelingen 1(1):22–82
Gerth H (1930) The evolution of reefcorals during the Cenozoic period. In: Proceedings of the fourth pacific congress, Java, 1929 2(a):333–350
Gradstein FM, Ogg JG, Smith AG, Agterberg FP, Bleeker W, Cooper RA, Davydov V, Bibbard P, Hinnov LA, House MR, Lourens LJ, Luterbacher H-P, McArthur JM, Melchin MJ, Robb LJ, Shergold J, Villeneuve M, Wardlaw BR, Ali JR, Brinkhuis H, Hilgen FJ, Hooker J, Howarth RJ, Knoll AH, Laskar J, Monechi S, Powell J, Plumb KA, Raffi I, Rohl U, Sanfilippo A, Schmitz B, Shackleton NJ, Shields GA, Strauss H, Van Dam J, Veizer J, van Kolfschoten Th, Wilson D (2004) A geological time scale. Cambridge University Press, Cambridge
Hall R (2001) Cenozoic reconstructions of SE Asia and the SW Pacific: changing patterns of land and sea. In: Metcalfe I, Smith JMB, Moorwood M, Davidson ID (eds) Faunal and floral migrations and evolution in SE Asia-Australasia. Swets & Zeitlinger Publishers, Lisse, pp 35–56
Hall R (2002) Cenozoic geological and plate tectonic evolution of SE Asia and the SW Pacific. J Asian Earth Sci 20(4):353–434
Hall R, Wilson MEJ (2000) Neogene sutures in Eastern Indonesia. J Asian Earth Sci 18:781–808
Haq B, Hardenbol J, Vail PR (1987) Chronology of fluctuating sea level since the Triassic. Science 235:1156–1166
Harzhauser M, Piller WE, Steininger FF (2002) Circum-Mediterranean oligo-Miocene biogeographic evolution-the gastropods’ point of view. Palaeogeogr Palaeoclimatol Palaeoecol 183(1-2):103–133
Hoeksema BW (2007) Delineation of the Indo-Malayan centre of maximum marine biodiversity: the coral triangle. In: Renema W (ed) Biogeography, time and place: distributions, barriers and islands. Springer, Dordrecht, pp 117–178
Hoffmeister JE (1942) Fossil corals from Viti Levu, Fiji. Bernice P. Bishop Museum-Bulletin 119:154–157
Holbourn A, Kuhnt W, Schulz M, Erlenkeuser H (2005) Impacts of orbital forcing and atmospheric carbon dioxide on Miocene ice-sheet expansion. Nature 438(7067):483–487
Holbourn A, Kuhnt W, Schulz M, Flores J-A, Andersen N (2007) Orbitally-paced climate evolution during the middle Miocene “Monterey” carbon-isotope excursion. Earth Planet Sci Lett 261:534–550
Jackson JBC, Johnson KG (2000) Life in the last few million years. Paleobiology 26(4):221–235
Jackson JBC, Jung P, Coates AG, Collins LS (1995) Diversity and extinction of tropical American molluscs and emergence of the Isthmus of Panama. Science 260:1624–1626
Jackson JBC, Budd AF, Pandolfi JM (1996) The shifting balance of natural communities? In: Jablonski D, Erwin DH, Lipps JH (eds) Evolutionary paleobiology. The University of Chicago Press, Chicago, pp 89–122
Johnson KG (2001) Middle Miocene recovery of Caribbean reef corals: new data from the Tamana formation, Trinidad. J Paleontol 75(3):513–526
Johnson KG, Kirby MX (2006) The Emprador limestone rediscovered: early Miocene corals from the Culebra Formation, Panama. J Paleontol 80(2):283–293
Johnson KG, Budd AF, Stemann TA (1995) Extinction selectivity and ecology of Neogene Caribbean reef corals. Paleobiology 21(1):52–73
Jones D, Hasson P (1985) History and development of the marine invertebrate faunas separated by the Central American isthmus. In: Stehli FG, Webb S (eds) The Great American biotic interchange. Plenum Press, New York, pp 325–355
Klaus JS, McNeill DF, Budd AF, Johnson KG (2008) Assessing community change in miocene to pliocene coral assemblages of the Northern Dominican Republic. In: Nehm RH, Budd AF (eds) Evolutionary stasis and change in the Dominican Republic Neogene. Springer Science + Business Media B.V.
Koswara A, Panggabean H, Baharuddin DS (1994) Geological map of the Bonerate Sheet, South Sulawesi, Scale 1:250,000. Geological Survey Indonesia, Bandung
Kuhnt W, Holbourn A, Hall R, Zuvela M, Kase R (2004) Neogene history of the Indonesian throughflow. Continent–ocean interactions within East Asian marginal seas. Geophys Monogr Ser 149:299–320
Lamarck JB (1801) Systeme des animaux sans vertebres. Paris
Lamarck JB (1816) Histoire naturelle des animaux sans vertèbres. Paris
Lindley D (1988) Early cainozoic stratigraphy and structure of the Gazelle Peninsula, east New Britain: an example of extensional tectonics in the New Britain arc-trench complex. Aust J Earth Sci 35:231–244
Marshall CR (1990) Confidence intervals on stratigraphic ranges. Paleobiology 16(1):1–10
Marshall CR (1994) Confidence intervals on stratigraphic ranges: partial relaxation of the assumption of randomly distributed fossil horizons. Paleobiology 20(4):459–469
Martin K (1880) Corallia, Foraminifera. In: Martin K (ed) Die Tertiärschichten auf Java. Nach den entdeckungen von Fr. Junghuhn. Brill, Leiden, pp 132–164
Michelin H (1840–1847) Iconographie zoophytologique. Description par localités et terrains des ploypiers fossiles de France. Paris
Milne Edwards H, Haime J (1848) Recherches sur les polypiers; deuxième mémoire. Monographie des Turbinolides. Annu Soc Natl Paris Zoölogie 3(9):211–344
Mora C, Chittaro PM, Sale PF, Kritzer JP, Ludsin SA (2003) Patterns and processes in reef fish diversity. Nature 421:933–936
Mudelsee M, Raymo ME (2005) Slow dynamics of the Northern Hemisphere glaciation. Paleoceanography 20:PA4022
Naish TR, Wilson GS (2009) Constraints on the amplitude of Mid-Pliocene (3.6–2.4 Ma) eustatic sea-level fluctuations from the New Zealand shallow-marine sediment record. Philos Trans R Soc A 367(1886):169–188
O’Dea A, Jackson JBC (2009) Environmental change drove macroevolution in cupuladriid bryozoans. Proc R Soc B 276:3629–3634
O’Dea A, Jackson JBC, Fortunato H, Smith T, D’Croz L, Johnson KG, Todd JA (2007) Environmental change preceded Caribbean extinction by 2 million years. Proc Natl Acad Sci USA 104(13):5501–5506
Orbigny AD (1849) Note sur des polpiers fossiles. Victor Masson, Paris
Osberger R (1946) Research on fossil corals from Java. Geological Dept. Faculty of Science, University of Indonesia, Bandung
Pandolfi JM (1993) A review of the tectonic history of New Guinea and its significance for marine biology, vol 2. In: Proceedings of the seventh international reef symposium, pp 718–727
Potts DC (1983) Evolutionary disequilibrium among Indo-Pacific corals. Bull Mar Sci 33(3):619–632
Quelch JJ (1886) Report on the reef-corals collected by H.M.S. challenger during the years 1873–76. Report on the scientific results of the voyage of the H.M.S. Challenger during the years 1873–76 (Zoology). no. 16, p 208
Ravelo AC, Andreasen DH, Lyle M, Lyle AO, Wara MW (2004) Regional climate shifts caused by gradual global cooling in the Pliocene epoch. Nature 429:263–267
Raymo ME (1994) The initiation of Northern Hemisphere glaciation. Annu Rev Earth Planet Sci 22:353–383
Raymo ME, Grant B, Horowitz M, Rau GH (1996) Mid-Pliocene warmth: stronger greenhouse and stronger conveyor. Mar Micropaleontol 27:313–326
Rebesco M, Camerlenghi A (2008) Late Pliocene margin development and mega debris flow deposits on the Antarctic continental margins: evidence of the onset of the modern Antarctic ice sheet? Palaeogeogr Palaeoclimatol Palaeoecol 260:149–167
Renema W, Bellwood DR, Braga JC, Bromfield K, Hall R, Johnson KG, Lunt P, Meyer CP, McMonagle LB, Morley RJ, O’Dea A, Todd JA, Wesselingh FP, Wilson ME, Pandolfi JM (2008) Hopping hotspots: global shifts in marine biodiversity. Science 321:654–657
Reuss AE (1867) Über fossile Korallen der Insel Java. Reise der Oesterreichischen Fregatte Novara um die Erde in den Jahren 1857, 1858, 1859. Geologischer Theil 2:165–185
Reyes-Bonilla H (2002) Checklist of valid names and synonyms of stony corals (Anthozoa: Scleractinia) from the eastern Pacific. J Nat Hist 36:1–13
Roberts CM, McClean CJ, Veron JEN, Hawkins JP, Allen GR, McAllister DE, Mittermeier CG, Schueler FW, Spalding M, Well F, Vynne C, Werner TB (2002) Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295(5558):1280–1284
Rodda P (1994) Geology of Fiji. In: Stevenson AJ, Herzer RH, Ballance PF (eds) Geology and submarine resources of the Tonga-Lau-Fiji region. SOPAC technical bulletin, Suva. pp 131–151
Rogl F (1999) Mediterranean and Paratethys. Facts and hypotheses of an Oligocene to Miocene paleogeography (short overview). Geologica Carpathica 50(4):339–349
Rogl F, Steininger FF (1984) Neogene Paratethys, Mediterranean and Indo-Pacific seaways. In: Brenchley PJ (ed) Fossils and climate. Wiley, New York, pp 171–200
Rosen BR (1984) Reef coral biogeography and climate through the late cainozoic; just islands in the sun or a critical pattern of islands. In: Brenchley PJ (ed) Fossils and climate. Wiley, New York, pp 201–262
Rosen BR (1988) Progress, problems and patterns in the biostratigraphy of reef corals and other tropical marine organisms. Helgolander Meeresuntersuchungen 42:269–301
Rosen BR, Smith AB (1988) Tectonics from fossils? Analysis of reef-coral and sea-urchin distributions from late cretaceous to recent, using a new method. In: Audley-Charles MG, Hallam A (eds) Gondwana and Tethys. Geol Soc Lond Special Publ, pp 275–306
Sadler PM (2004) Quantitative biostratigraphy—achieving finer resolution in global correlation. Annu Rev Earth Planet Sci 32:187–213
Stehli FG, Wells JW (1971) Diversity and age patterns in hermatypic corals. Syst Zool 20:115–126
Sukamto R, Supriatna S (1982) The geology of the Ujung Pandang, Benteng and Sinjai quadrangles, Sulawesi. In Geological Research and Development Centre
Umbgrove JHF (1939) Miocene corals from flores (East Indies). Leidse Geologische Mededelingen 11:62–67
Umbgrove JHF (1942) Corals from asphalt deposits of the island Buton (East Indies). Leidse Geologische Mededelingen 29–38:12–13
Umbgrove JHF (1945) Corals from the upper Miocene of Tjisande, Java. In: Proceedings of the Koninklijke Nederlandse Akademie van Wetenschappen (1990), vol 48, pp 340–344
Umbgrove JHF (1946a) Corals from a lower Pliocene patch reef in Central Java. J Paleontol 20(6):521–542
Umbgrove JHF (1946b) Evolution of reef corals in East Indies since Miocene time. Bull Am Assoc Petrol Geol 30(1):23–31
Umbgrove JHF (1950) Corals from the Putjangan Beds (Lower Pleistocene) of Java. J Paleontol 24(6):637–651
Vaughan TW, Wells JW (1943) Revision of the suborders families, and genera of the scleractinia. Geol Soc Am
Veron JEN, Kelly R (1988) Species stability in reef corals of Papua New Guinea and the Indo Pacific. Association of Australian Palaeontologists, Moorooka
Veron JEN, Pichon M (1976) Scleractinia of Eastern Australia Part 1. Australian Government Printing Service, Canberra
Veron JEN, Pichon M (1979) Scleractinia of Eastern Australia Part 3. Australian National University Press, Canberra
Veron JEN, Pichon M (1982) Scleractinia of Eastern Australia Part 4. Australian National University Press, Canberra
Veron JEN, Wallace CC (1984) Scleractinia of Eastern Australia Part 5. Australian National University Press, Canberra
Veron JEN, Pichon M, Wijsman-Best M (1977) Scleractinia of Eastern Australia Part 2. Australian Government Publishing Service, Canberra
Verrill AE (1865) List of polyps and corals sent by the Museum of Comparative Zoology to other institutions in exchange, with annotations. Bull Mus Compar Zool 1:29–60
Wara MW, Ravelo AC, Delaney ML (2005) Permanent El Nino-like conditions during the Pliocene warm period. Science 309:758–761
Warny SA, Askin RA, Hannah MJ, Mohr BAR, Raine JI, Harwood DM, Florindo F (2009) Palynomorphs from a sediment core reveal a sudden remarkably warm Antarctica during the middle Miocene. Geology 37(10):955–958
Wells JW (1956) Scleractinia. Pp. F328-F444. In: Moore RC, (ed) Treatise on invertebrate paleontology. Geological Society of America and University of Kansas Press
Wilson MEJ (1995) The Tonasa limestone formation, Sulawesi, Indonesia: development of a tertiary carbonate platform. University of London, London
Wilson MEJ (2002) Cenozoic carbonates in Southeast Asia: implications for equatorial carbonate development. Sed Geol 147:295–428
Wilson MEJ (2008) Global and regional influences on equatorial shallow-marine carbonates during the Cenozoic. Palaeogeogr, Palaeoclimatol, Palaeoecol 265(3–4):262–274
Wilson MEJ, Rosen BR (1998) Implications of paucity of corals in the Paleogene of SE Asia: plate tectonics or centre of origin? In: Hall RH (ed) Biogeography and geological evolution of SE Asia. Backbuys Publishers, Leiden, pp 165–195
Zachos JC, Pagani M, Sloan L, Thomas E, Billups K (2001) Trends, rhythms and aberrations in global climate 65 Ma to present. Science 292:686–693
Acknowledgments
We acknowledge financial contributions from American Paleontology Society (Steven J. Gould Award for Paleontology 2007); Paleontological Research Institute (Student Award for Paleontology 2005); Commonwealth Scientific Research Organisation (CSIRO) Wealth form the Oceans Flagship (2005–2008); University of Queensland Joint Research Scholarship (UQJRS) 2005–2009, Centre for Marine Studies and the Australian Research Council Centre of Excellence for Coral Reef Studies. We thank the Leiden Natural History Museum for access to collections; Professor Hugh Davies and the University of Papua New Guinea for access to the Pliocene coral collection; James Zachos for access to climate data; Jim Robbins for facilitating research visas in Papua New Guinea and the Papua New Guinean Government for allowing us to conduct research in P.N.G.; Kabira Dive, New Britain for accommodation and logistical support; Dr. Suharsono, Research Centre for Oceanography, Indonesian Institute of Science (LIPI); the Indonesian Government for allowing us to conduct research in Indonesia; Sarah Grimes and the Pacific Islands Applied Geoscience Commission (SOPAC); and the Fijian Government for allowing us to conduct research in Fiji.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bromfield, K., Pandolfi, J.M. Regional patterns of evolutionary turnover in Neogene coral reefs from the central Indo-West Pacific Ocean. Evol Ecol 26, 375–391 (2012). https://doi.org/10.1007/s10682-011-9483-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-011-9483-9