Abstract
Grain yield in rice is largely determined by grain size. Grain Size 3 (GS3) is a major quantitative trait locus for grain size. The C–A natural variation in the second exon of GS3 was reported to play an important role in regulating grain length in rice. Here we evaluate GS3 alleles among 303 germplasm accessions. The GS3A allele was predominant in xian/indica (XI) accessions, whereas geng/japonica (GJ) accessions mainly carried GS3C. The GS3 allele affected the grain length significantly in XI, while its function was minimal in GJ, indicating that introduction of GS3 alleles might be useful to modify grain length in XI breeding programs, but not in GJ breeding. The association between GS3 alleles and seed weight was not significant in any of the individual subpopulations, suggesting that the contribution of GS3 to grain weight could be slight in terms of different subspecies. To develop an effective marker for GS3, a penta-primer amplification-refractory mutation system (PARMS) marker exploiting a single-base mutation (C–A) was developed, which entailed lower cost and less time than other available markers, and should be useful for fine marker-assisted selection of grain length in XI accessions breeding.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Grain size, which is specified by grain length, width and length-to-width ratio, is a highly important quality trait and an important target for selection during domestication and breeding in rice (Fitzgerald et al. 2009; Xing and Zhang 2010; Sun et al. 2018). Long and slender grain is preferred in China for xian/indica rice because of its desirable appearance (Xu and Chen 2016; Huang and Qian 2017; Chen et al. 2023). GRAIN SIZE 3 (GS3), encoding a Gγ subunit (Group III) of the heterotrimeric G protein, has been identified as a major QTL for grain length. In addition, GS3 participates in stigma exsertion (Takano-Kai et al. 2011) and alkaline tolerance (Zhang et al. 2023). Natural variants of GS3 have been shown to boost grain yield (Fan et al. 2006), in particular, with a single nucleotide polymorphism (SNP) between C and A in the second exon (Fan et al. 2009; Takano-Kai et al. 2009). With technological advances in functional genomics, great progress has been achieved in clarifying the molecular mechanisms that determine seed size (Song et al. 2007; Weng et al. 2008; Li et al. 2011; Qi et al. 2012; Wang et al. 2012, 2015, 2019; Zhang et al. 2012; Duan et al. 2015; Hu et al. 2015; Xu et al. 2018; Zhao et al. 2018). Among them, the GS3 locus also provides an informative system for studying the evolutionary processes underlying rice domestication and breeding (Mao et al. 2010). However, the functional effect of natural variants of GS3 has been described only in particular rice cultivars; its role in determining the grain size phenotype in different genetic backgrounds remains be confirmed.
The GS3 gene has five exons and encodes 232 amino acids with a specific organ-size regulation (OSR) domain near the N terminus, a tumor necrosis factor receptor/nerve growth factor receptor-family cysteine-rich domain, and a von Willebrand factor type C module (Fan et al. 2006). The GS3 gene functions as a negative regulator of grain size: the OSR domain is both necessary and sufficient for functioning as a negative regulator (Mao et al. 2010). A nonsense mutation (C–A) is found in the second exon of GS3 shared among the long length-grain varieties but not in varieties with smaller grains. The C–A mutation causes a 178-amino acid truncation at the C terminus, resulting in part of the OSR domain and all the other three domains deleted (Mao et al. 2010). Besides the C–A mutation, a further three polymorphic loci were discovered in the second intron, the last intron and the final exon of GS3. It is worth noting that the C–A mutation is a highly effective QTL that explains one third of the grain length variation in rice (Wang et al. 2010). In this study, we evaluated the natural variation of GS3 alleles among 303 germplasm accessions, including 81 xian/indica (XI) rice types and 222 geng/japonica (GJ) types to investigate the correlation between GS3 alleles and grain length and weight. We also developed a novel intragenic marker that provides a valuable tool for the evaluation and use of the GS3 gene in screening germplasm accessions and in breeding new varieties.
Materials and methods
Rice accessions and cultivars
We used 81 XI rice accessions and 222 GJ accessions in this study (Table S1). The XI accessions consisted of rice cultivars approved in the southern provinces of China, including Chinese landraces and cultivated varieties. The GJ accessions consisted of typical cultivated varieties, a small number of rice strains and 31 varieties approved by Shandong Crop Varieties Examination and Approval Committee in the last five years. In addition, the Indian cultivars Pokkali, Kasalath, and Dular and seven aus accessions were included in this study (Table S2).
Marker design and analysis
The penta-primer amplification-refractory mutation system (PARMS) marker is designed based on the C–A polymorphism in the second codon of GS3, as described in previously (Fan et al. 2009), and comprised the primer pair PARMS-GS3-F1 (GAAGGTGACCAAGTTCATGCTCTGCCTCCAGATGCTGA) and PARMS-GS3-F2 (GAAGGTCGGAGTCAACGGATTCTGCCTCCAGATGCTGC) plus PARMS-GS3-R-Common (TGCATGGTAAGAGTAAAGACGAGA). The primers GS3-F (AAATATCCCTCAGACATCAC) and GS3-R (CACTCAAAAAGCTTGCAC) amplified the C–A mutation region and were designed from the two allelic sequences of rice varieties ‘9311’ and ‘Nipponbare’.
We carried out PARMS amplification PCR in a total volume of 10 μL containing 1 μL genomic DNA, 0.15 μL F primers, 0.4 μL R primers and 5 μL PARMS mix using an Applied Biosystems Q5 Real-Time PCR System according to the manufacturer’s instructions. The PCR conditions were: 15 min at 94 °C for denaturation, followed by 10 cycles of 94 °C for 20 s and 65 °C (with the temperature for each cycle decreased by 0.8 °C) for 1 min, then 28 cycles of 94 °C for 20 s and 57 °C for 1 min, and finally by 1 min at ambient temperature. For the developed marker, the PCR reaction contained 2 μL (40 ng) genomic DNA, 1 μL each primer (10 μM), 10 μL 2 × Taq PCR MasterMix and 6 μL ddH2O. The PCR profile was 3 min at 94 °C for denaturation, followed by 32 cycles of 94 °C for 30 s, 55 °C for 30 s, and 72 °C for 1 min, then 3 min at 72 °C for extension. The products were detected by agarose gel electrophoresis and sequenced by the Qingke Biological Technology Co. Ltd.
Measurements of grain traits
Harvested paddy rice was air-dried and stored at room temperature before testing. Ten fully-filled grains were randomly chosen from each plant were divided into three equal groups. Each group was lined up length-wise along a vernier caliper to measure total grain length. Grain weight was calculated on the basis of 1000 grains. As described in the textbook ‘The Principle and Method of Testing New Rice Varieties’, the grain length and weight were selected as per the following table (Tables 1 and 2):
Statistical analysis
Duncan’s test was performed to compare the means of seed traits for different allelic groups or cultivars/lines using SPSS 19.0. T-tests were performed or F statistic estimated for each allelic group using Excel 2007.
Results
Distribution of GS3 alleles in rice subspecies from China
As mentioned above, the nonsense mutation (C–A) causes a truncated protein with part of the OSR domain and all the other three domains are deleted (Fig. 1A). To facilitate the genetic improvement of GS3 in XI and GJ accessions in China, alleles of GS3 were identified in 303 germplasm accessions using Fan’s CAPS marker (Fan et al. 2009). We firstly analyzed the distribution of GS3 in 81 XI accessions. The GS3A allele was found in 59 out of the 81 XI accessions (73%), and the GS3C allele in 22 out of 81 accessions (27%) (Fig. 1B). We then analyzed the distribution of GS3 in GJ accessions: 176 out of the 222 GJ accessions (80%) carried the GS3C allele, and only 46 accessions (20%) carried the GS3A allele (Fig. 1C). These results indicate that GS3 was selected independently of subspecies. It was apparent that the distribution of GS3 alleles was opposite between XI and GJ accessions. The distribution was broadly in keeping with the phenotype of grain length, in that long and slender grain is prevalent in XI rice and short round grain in GJ cultivars. Interestingly, all the aus accessions carried the GS3C allele (Fig. 1D). In view of the aus rice is mainly distributed across South Asia, mainly concentrated in the Indian subcontinent. It seems likely that GS3A allele was the result of later artificial selection in indica rice breeding in China.
Correlation between GS3 alleles and grain length
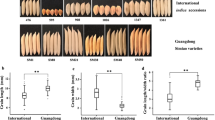
To investigate whether there was a functional difference between XI and GJ accessions, caused by the GS3 genotype, We measured their ten-seed length in all the 303 accessionsare (Fig. 2A,B; Supplementary Table 1). In XI accessions, the grain lengths of the GS3C allele mainly fell into the range of 5- to 7-class and most of them were below 6-class. In contrast, the grain lengths of GS3A accessions fell into the 6- to 9-class and most of them were 7-class and above (Fig. 2C). The average grain lengths of the 59 accessions carrying GS3A were significantly longer than that 22 accessions that carried GS3C (Fig. 2D). However, in GJ accessions, there was no significant difference in average grain length between GS3C and GS3A alleles (Fig. 2D), as shown by that the grain lengths of accessions carrying either the GS3C allele (176 accessions) or the GS3A allele (46 accessions) mainly fell into 5- to 6-class (Fig. 2E). As a result, the grain lengths of accessions carrying the GS3A allele in GJ accessions were significantly shorter than those in XI accessions (Fig. 2E). We deduced that the function of the GS3A allele in regulating grain length was less pronounced in GJ than in XI accessions.
the grain length levels between XI and GJ accessions. A–B The overall trend of the grain length in random accessions: A XI accessions; B GJ accessions; C–D The situation of grain length levels: C indica cultivars; D japonica cultivars; E The average 10-grain length in 303 accessions. Student’s t test was used to generate the P values
In conclusion, the SNP (C–A) in the second exon of GS3 was confirmed to be highly associated with grain length in Chinese accessions, consistent with the previous report that the coding sequence TGC occurred in almost all the short-grain group and the TGA (premature coding stop) in the long-grain group (Fan et al. 2009; Wang et al. 2010). Interestingly, this association was different between XI and GJ accessions. The functional effect of the GS3 alleles was significant in XI but unproductive in GJ accessions.
To further confirm the effects of the SNP variation on indica–japonica differentiation, statistical analysis was carried out. There was a highly significant strong correlation between grain length and allele (C–A) in XI cultivars (P < 0.01), whereas the correlation in GJ accessions was weaker (correlation coefficient 0.068; Table 3). These results further confirmed that the natural variation (C–A) was subjected to artificial selection in XI but not in GJ.
Correlation between GS3 alleles and grain weight
Numerous studies have shown that the rice GS3 gene is associated with not only grain length but also grain weight (Takano-Kai et al. 2009). The GS3 gene has a significant negative effect on seed length and weight. However, previous studies were based on a single cultivar or transgenic lines, and it remains unclear whether the correlation between GS3 alleles and grain weight is observed more widely in XI and GJ accessions. For this purpose, we selected all 303 accessions from XI and GJ, and measured their 1000-grain weight. The distribution of grain weights overlapped between accessions that carried GS3C and GS3A in either XI or GJ subspecies. The grain weight ranges of XI accessions carrying GS3A were concentrated at level 4–6, while those with GS3C were at level 3–5 (Fig. 3A, C). In GJ accessions, the grain weights were mostly at level 4–6, regardless of allele (Fig. 3B, D). There was no significant difference in average weight between GS3A and GS3C, in either XI or in GJ (Fig. 3E). Further, statistical analysis showed that the correlation coefficient was 0.248 between GS3C and GS3A alleles in XI, and 0.214 in GJ, which meant there was only a weak correlation between grain weight and allele (C–A) in XI and GJ cultivars (Table 4).
the distribution of the GS3 haplotypes and grain weight levels. A–B the overall trend of the grain weight in random accessions: A indica cultivars; B japonica cultivars; C–D the situation of grain weight levels: C indica cultivars; D japonica cultivars; E the average 1000-grain weight in 303 cultivars. Student’s t test was used to generate the P values and there is no significant differences between indica GS3(C) and indica GS3(A), japonica GS3(C) and joponica GS3(A)
Development of intragenic markers for GS3 gene
The SNP (C–A) was confirmed to be highly associated with grain length in the Chinese rice accessions. Based on the two alleles, several markers have been developed, such as a CAPS marker (Fan et al. 2009), linked marker (Huang et al. 2013), and tetra-primer ARMS marker (Wu et al. 2019). However, both enzyme digestion and electrophoresis entail significant time and cost. To develop an effective marker for GS3, we chose to focus on the C–A mutation variation and designed a penta-primer amplification refractory mutation system (PARMS) marker, which would entail lower cost and take less time than would other available markers. Specifically, the sequence containing the C nucleobase (causing short grains) was matched and combined with HEX fluorescence and showed as red dots. The sequence with the A nucleobase is matched with FAM fluorescence and shown blue dots. Meanwhile, if the sample being tested is heterozygous, the result is shown fluorescent green (Fig. 4A, B). To confirm the specificity and accuracy of this primer, the GS3 marker of Wu (Wu et al. 2019) was used to test 11 random samples from 303 accessions: the results confirmed exact agreement with our new PARMS marker (Fig. 4C). Further, next-generation sequencing was used to characterize and confirm the amplification fragment. Together, these results confirmed that this PARMS primer would be useful as a functional marker in selecting for improvement of rice grain length (Fig. 4D).
The PARMS marker development and validation for GS3. A the schematic diagram of design principle about GS3 PARMS primers; B The allelic discrimination plot of GS3. The homozygous Allele1 (red dots) indicated GS3A type, Allele2 (blue dots) indicated GS3C type. If fluorescent green dots are presented, these are heterozygous. The black dots indicated blank control; C the Tetra-primer ARMS marker (Wu et al. 2019). The electrophoretic bands of 720 bp and 308 bp indicate GS3A and GS3C type. Here are Heixiangnuo, Shengdao19, Nonghan1, Runnong11, Shengdao22, Huageng5, Lindao16, Neixiang2B, Shuhui527, Yixiang1B and Suxiu867 from left to right; D The sequencing validation of natural variation
Discussion
In the previous study, the GS3 gene had a major effect on rice grain size and the C–A mutation in the second exon of GS3 was functionally associated with enhanced grain length and weight (Mao et al. 2010; Yan et al. 2011). However, this result was found based on a specific cultivar background. This study has evaluated the functional effect of different alleles of GS3 on grain length and weight between between XI and GJ subspecies.
First, the distribution of the GS3 gene alleles (C–A mutation) between XI and GJ accessions was not completely dependent on indica–japonica differentiation. Specifically, the XI accessions mostly carried the GS3A allele whereas the GJ accessions have largely the GS3C allele, which probably explains the phenotypic difference, namely that long and slender grain in XI while short round grain in GJ accessions (Fig. 1B, C). Rice is the largest cereal crop planted throughout the world. It is widely believed that Oryza indica originated in Southeast Asia and South Asia countries such as India (Khush 1997; Ikehashi 2009). In this study, we analyzed ten typical ancient accessions from India, including the cultivars Pokkali, Kasalath and Dular. None of them carried GS3A allele. However, the GS3A allele was predominant in XI accessions from China. We therefore deduced that the GS3A genotype was the result of later artificial selection in XI subspecies (Fig. 1D).
Second, the GS3A allele affected the grain length significantly in XI accessions: the average grain length of accessions that carried GS3A was significantly larger than in lines that carried GS3C (Fig. 2E). Also, there was a strong and significant correlation between grain length and allele (C–A) in XI cultivars (P < 0.001; Table 3). Interestingly, the effect of the GS3A allele on grain length was minimal in in GJ accessions (Fig. 2D, E). Why does GS3A alleles function differentially in regulating grain length between XI and GJ rice? As shown in the previous study, GS3, although having no direct function in promoting or inhibiting grain length, regulates grain length by blocking the interaction of DEP1 (Dense panicle 1) and GGC2 (G protein gamma subunit) with RGB1 (G protein beta subunit) (Sun et al. 2018). The GS3 and DEP1 proteins interact directly with the conserved keratin-like domain of MADS transcription factors, and function as cofactors to enhance OsMADS1 (MADS-domain transcription factor) transcriptional activity, thereby regulating grain size (Liu et al. 2018). In addition, GL3.3 (Grain length and weight) protein interacts with GS3 and leads to extra-long grains in rice (Xia et al. 2018). The net outcome of the functional interactions among these genes thus determines grain size (Sun et al. 2018). Considering multiple factors interacting with GS3, we speculate that factors acting downstream of GS3 probably differentially function to regulate grain length between XI and GJ rice. In addition, it has been reported that grain size is determined by a complex regulatory network (Ren et al. 2023). It is possible that other regulatory pathways determining grain length have antagonistic effects with GS3A pathway on grain length in GJ accessions. However, our results suggest that introduction of GS3 alleles would be directly useful to modify grain length in XI breeding programs, but would be of doubtful value in GJ breeding programs. Further research will be needed to understand the biological functions of GS3 more fully.
Third, numerous studies have identified the GS3 gene as being associated with grain weight (Fan et al. 2006; Takano-Kai et al. 2009; Yan et al. 2011), which is different from our results in this study. We found the association between GS3 alleles and seed weight was not significant in any of the individual subpopulations (Fig. 3; Table 4). Considering that grain weight is affected by grain length, grain width, grain thickness and grain filling, we speculate that it might be difficult to increase rice yield by genetic improvement using GS3 alleles. Finally, the intragenic marker for the GS3 gene that we developed in this study could be of great use in evaluating the genotype of germplasm accessions and guiding the improvement of grain length in XI cultivars in the future (Fig. 4).
Conclusion
We identified GS3 alleles and analyzed the distribution of GS3 in 81 XI and 222 GJ accessions. The results showed that GS3 was selected independently of subspecies and the distribution was broadly in keeping with the phenotype of grain length, in that long and slender grain is prevalent in XI rice and short round grain in GJ cultivars. Perhaps GS3A allele was the result of later artificial selection in indica rice breeding in China. The average grain lengths of the 59 accessions carrying GS3A were significantly longer than that 22 accessions that carried GS3C in XI accessions, however, there was no significant difference in average grain length between GS3C and GS3A alleles in GJ accessions. The functional effect of the GS3 alleles on grain length was significant in XI but unproductive in GJ accessions. For the grain weight, there was no significant difference in average weight between GS3A and GS3C, in either XI or in GJ. To develop an effective marker for GS3, a PARMS marker was designed, which would entail lower cost and take less time than would other available markers. Taken together, the analysis of GS3 alleles helps gain insight into the molecular mechanisms involved in grain size in rice, and GS3A alleles in XI accessions may provide a new opportunity to improve rice grain size breeding.
References
Chen RJ, Xiao N, Lu Y, Tao TY, Huang QF, Wang ST, Wang ZC, Chuan ML, Bu Q, Lu Z, Wang HY, Su YZ, Ji Y, Ding JH, Gharib A, Liu HX, Zhou Y, Tang SZ, Liang GH, Zhang HG, Yi CD, Zheng XM, Cheng ZK, Xu Y, Li PC, Xu CW, Huang JL, Li AH, Yang ZF (2023) A de novo evolved gene contributes to rice grain shape difference between Indica and Japonica. Nat Commun 14(1):5906
Duan PG, Ni S, Wang JM, Zhang BL, Xu R, Wang YX, Chen HQ, Zhu XD, Li YH (2015) Regulation of OsGRF4 by OsmiR396 controls grain size and yield in rice. Nat Plants 2:15203
Fan CC, Xing YY, Mao HL, Lu TT, Han B, Xu CG, Li XH, Zhang QF (2006) GS3, a major QTL for grain length and weight and minor QTL for grain width and thickness in rice, encodes a putative transmembrane protein. Theor Appl Genet 112:1164–1171
Fan C, Yu S, Wang C, Xing Y (2009) A causal C-A mutation in the second exon of GS3 highly associated with rice grain length and validated as a functional marker. Theor Appl Genetics 118:465–472
Fitzgerald MA, McCouch SR, Hall RD (2009) Not just a grain of rice: the quest for quality. Trends Plant Sci 14(3):133–139
Hu J, Wang YX, Fang YX, Zeng LJ, Xu J, Yu HP, Shi ZY, Pan JJ, Zhang D, Kang SJ, Zhu L, Dong GJ, Guo LB, Zeng DL, Zhang GH, Xie LH, Xiong GS, Li JY, Qian Q (2015) A rare allele of GS2 enhances grain size and grain yield in rice. Mol Plant 8:1455–1465
Huang HY, Qian Q (2017) Progress in genetic research of rice grain shape and breeding achievements of long-grain shape and good quality Japonica Rice. Chin J Rice Sci 31(6):665–672 (in Chinese)
Huang RY, Jiang LR, Zheng JS, Wang TS, Wang HC, Huang YM, Hong ZL (2013) Genetic bases of rice grain shape: so many genes, so little known. Trends Plant Sci 18(4):218–226
Ikehashi H (2009) Why are there Indica type and Japonica type in rice History of the studies and a view for origin of two types. Rice Sci 16(1):1–13
Khush GS (1997) Origin, dispersal, cultivation and variation of rice. Plant Mol Biol 35(1):24–34
Li YB, Fan CC, Xing YZ, Jiang HY, Luo LJ, Sun L, Shao D, Xu CJ, Li XH, Xiao JH, He YQ, Zhang QF (2011) Natural variation in GS5 plays an important role in regulating grain size and yield in rice. Nat Genet 43:1266–1269
Liu Q, Han RX, Wu K, Zhang JQ, Ye YF, Wang SS, Chen JF, Pan YJ, Li Q, Xu XP, Zhou JW, Tao DY, Wu YJ, Fu XD (2018) G-protein βγ subunits determine grain size through interaction with MADS-domain transcription factors in rice. Nat Commun 9(1):852
Mao HL, Sun SY, Yao JL, Wang CR, Yu SB, Xu CG, Li XH, Zhang QF (2010) Linking differential domain functions of the GS3 protein to natural variation of grain size in rice. Proc Natl Acad Sci USA 107:19579–19584
Qi P, Lin YS, Song XJ, Shen JB, Huang W, Shan JX, Zhu MZ, Jiang LW, Gao JP, Lin HX (2012) The novel quantitative trait locus GL3.1 controls rice grain size and yield by regulating Cyclin-T1;3. Cell Res 22:1666–1680
Ren DY, Ding CQ, Qian Q (2023) Molecular bases of rice grain size and quality for optimized productivity. Sci Bull 68(3):314–350
Song XJ, Huang W, Shi M, Zhu MZ, Lin HX (2007) A QTL for rice grain width and weight encodes a previously unknown RING-type E3 ubiquitin ligase. Nat Genet 39:623–630
Sun SY, Wang L, Mao HL, Shao L, Li XH, Xiao JH, Ouyang YD, Zhang QF (2018) A G-protein pathway determines grain size in rice. Nat Commun 9(1):851
Takano-Kai N, Jiang H, Kubo T, Sweeney M, Matsumoto T, Kanamori H, Padhukasahasram B, Bustamante C, Yoshimura A, Doi K, McCouch S (2009) Evolutionary history of GS3 a gene conferring grain length in rice. Genetics 182(4):1323–1334
Takano-Kai N, Doi K, Yoshimura A (2011) GS3 participates in stigma exsertion as well as seed length in rice. Breed Sci 61(3):244–250
Wang CR, Chen S, Yu SB (2010) Functional markers developed from multiple loci in GS3 for fine marker-assisted selection of grain length in rice. Theor Appl Genet 122:905–913
Wang SK, Wu K, Yuan QB, Liu XY, Liu ZB, Lin XY, Zeng RZ, Zhu HT, Dong GJ, Qian Q, Zhang GQ, Fu XD (2012) Control of grain size, shape and quality by OsSPL16 in rice. Nat Genet 44:950–954
Wang YX, Xiong GS, Hu J, Jiang L, Yu H, Xu J, Fang YX, Zeng LJ, Xu EB, Xu J, Ye WJ, Meng XB, Liu RF, Chen HQ, Jing YH, Wang YH, Zhu XD, Li JY, Qian Q (2015) Copy number variation at the GL7 locus contributes to grain size diversity in rice. Nat Genet 47:944–948
Wang AH, Hou QQ, Si LZ, Huang XH, Luo JH, Lu DF, Zhu JJ, Shangguan YY, Miao JS, Xie YF, Wang YC, Zhao Q, Feng Q, Zhou CC, Li Y, Fan DL, Lu YQ, Tian QL, Wang ZX, Han B (2019) The PLATZ transcription factor GL6 affects grain length and number in rice. Plant Physiol 180:2077–2090
Weng JF, Gu SH, Wan XY, Gao H, Guo T, Su N, Lei CL, Zhang X, Cheng ZJ, Guo XP, Wang JL, Jiang L, Zhai HQ, Wan JM (2008) Isolation and initial characterization of GW5, a major QTL associated with rice grain width and weight. Cell Res 18:1199–1209
Wu H, Gao LJ, Huang J, Gao J, Qing DJ, Dai GX, Liang HF, Zhou WY (2019) Development and application of functional molecular marker of grain length and grain weight major gene GS3 in rice. Southwest Chin J Agric Sci 32(06):1211–1215 (in Chinese)
Xia D, Zhou H, Liu R, Dan WH, Li PB, Wu B, Chen JX, Wang LQ, Gao GJ, Zhang QL, He YQ (2018) GL3.3, a novel QTL encoding a GSK3/SHAGGY-like kinase, epistatically interacts with GS3 to produce extra-long grains in rice. Mol Plant 11(5):754–756
Xing YZ, Zhang QF (2010) Genetic and molecular bases of rice yield. Annu Rev Plant Biol 61:421–442
Xu ZJ, Chen WF (2016) Research progress and related problems on japonica Super rice in northern China. Sci Agric Sin 49(2):239–250 (in Chinese)
Xu R, Duan PG, Yu HY, Zhou ZK, Zhang BL, Wang RC, Li J, Zhang GZ, Zhuang SS, Lyu J, Li N, Chai TY, Tian ZX, Yao SG, Li YH (2018) Control of grain size and weight by the OsMKKK10-OsMKK4-OsMAPK6 signaling pathway in rice. Mol Plant 11:860–873
Yan S, Zou GH, Li SJ, Wang H, Liu HQ, Zhai GW, Guo P, Song HM, Yan CJ, Tao YJ (2011) Seed size is determined by the combinations of the genes controlling different seed characteristics in rice. Theor Appl Genet 123:1173–1181
Zhang XJ, Wang JF, Huang J, Lan HX, Wang CL, Yin CF, Wu YY, Tang HJ, Qian Q, Li JY, Zhang HS (2012) Rare allele of OsPPKL1 associated with grain length causes extra-large grain and a significant yield increase in rice. Proc Natl Acad Sci USA 109:21534–21539
Zhang HL, Yu FF, Xie P, Sun SY, Qiao XH, Tang SY, Chen CX, Yang S, Mei C, Yang DK, Wu YR, Xia R, Li X, Lu J, Liu YX, Xie XW, Ma DM, Xu X, Liang ZW, Feng ZH, Huang XH, Yu H, Liu GF, Wang YC, Li JY, Zhang QF, Chen C, Ouyang YD, Xie Q (2023) A Gγ protein regulates alkaline sensitivity in crops. Science 379(6638):8eade8416
Zhao DS, Li QF, Zhang CQ, Zhang C, Yang QQ, Pan LX, Ren XY, Lu J, Gu MH, Liu QQ (2018) GS9 acts as a transcriptional activator to regulate rice grain shape and appearance quality. Nat Commun 9:1240
Acknowledgements
This work was financially supported by Strategic Priority Research Program of the Chinese Academy of Sciences (XDA24030101-6), the Shandong Provincial Natural Science Foundation (ZR2020QC112) and the Science and Technology Innovation Program of Shandong Academy of Agricultural Sciences (CXGC2023A04).
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peng, YB., Du, CY., He, YN. et al. Natural variation of Grain size 3 allele differentially functions in regulating grain length in xian/indica and geng/japonica rice. Euphytica 220, 44 (2024). https://doi.org/10.1007/s10681-024-03293-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-024-03293-7