Summary
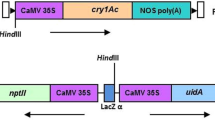
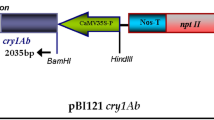
Semilooper resistant transgenic castor plants were produced through Agrobacterium-mediated genetic transformation method. Two castor cultivars, Jyothi and VP1 were transformed using the super-binary vector pTOK233 carrying gus A and hpt genes. Putative transformants were regenerated following selection on the hygromycin containing medium. GUS positive primary transformants, when subjected to Southern analysis, revealed stable integration of gus A into their genomes. In the T1 generation, a monogenic segregation ratio of 3 GUS positive: 1 GUS negative plants was observed. Furthermore, transformation experiments were carried out with the Agrobacterium pSB111 super-binary vector carrying a synthetic delta endotoxin gene cryIAb and the herbicide resistance gene bar both driven by cauliflower mosaic virus 35S promoter. Putative transformants were regenerated through selection on the phosphinothricin containing medium and Basta tolerant transformants were subjected to molecular analysis. PCR analysis revealed the presence of both bar and cryIAb genes in the Basta tolerant primary transformants. Southern analysis of PCR positive plants with cryIAb probe showed a 3 Kb band upon HindIII digestion and a > 6 Kb band with BamHI digestion, thus suggesting stable integration of cryIAb intact expression cassette and independent nature of the transformants. The primary transformants subjected to ELISA disclosed varied levels of Cry protein. These transgenics expressing cryIAb – when bioassayed against freshly hatched semilooper larvae – induced substantial (> 88%) insect mortality. Southern analysis of 2T1 plants revealed the presence of cryIAb gene, indicating stable inheritance of the transgene into the next generation. In T1, all the Southern-positive plants for cryIAb invariably exhibited tolerance to Basta, denoting co-segregation of both bar and cryIAb genes. Transgenics, expressing cryIAb exhibited ample resistance against the castor semilooper.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adang, M.J., E. Firoozabady, J. Klein, D. Deboer, V. Sekar, J.D. Kemp, E. Murray, T.A. Rocheleau, K. Rashka & G. Staffield, 1987. Application of a Bacillus thuringiensis crystal protein for insect control. In: C.J. Arntzen & C. Rayn (Eds.), Molecular Strategies for Crop Protection, pp. 345–353, Alan, R. Liss, New York.
Aldemita, R.R. & T.K. Hodges, 1996. Agrobacterium tumefaciens-mediated transformation of Japonica and Indica rice varieties. Planta. 199: 612–617.
Aronson, A.I., W. Beckman & P. Dunn, 1986. Bacillus thuringiensis and related insect pathogens. Microbiol. Rev. 50: 1–24.
Barteneva, R.V., 1986. Diseases and pests of castor and their control. In: V.A. Moshkin (Ed.), Castor, pp. 284, Oxonian Press Pvt Ltd. New Delhi.
Barton, K., H. Whiteley & N.S. Yang, 1987. Bacillus thuringiensis δ-endotoxin in transgenic Nicotiana tabacum provides resistance to lepidopteran insects. Plant Physiol. 85: 1103–1109.
Feitelson, J.S., J. Payne & L. Kim, 1992. Bacillus thuringiensis: insect and beyond. Bio/Technology 10: 271–275.
Finnegan, J. & D. McElroy, 1994. Transgenic inactivation: plants fight back! Bio/Technology 12: 883–888.
Gasser, C.S. & R.T. Fraley, 1989. Genetically engineering plants for crop improvement. Science. 244: 1293–1299.
Ghareyazie, B., F. Alinia, C.A. Menguito, L.G. Rubia, J.M. de Palma, E.A. Liwanag, M.B. Cohen, G.S. Khush & J. Bennet, 1997. Enhanced resistance to two stem borers in an aromatic rice containing a synthetic cryIA(b) gene. Mol. Breed. 3: 401–414.
Hiei, Y., S. Ohta, T. Komari & T. Kumashiro, 1994. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of boundaries of the T-DNA. Plant J. 6: 271–282.
Hood, E.E., G.L. Helmer, R.T. Fraley & M.D. Chilton, 1986. The hyper virulence of Agrobacterium tumefaciens A281 is encoded in a region of pTiBo542 outside of T-DNA. J. Bacteriol. 168: 1291–1301.
Jefferson, R.A., T.A. Kavanagh & M.W. Bevan, 1987. Gus fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6: 3901–3907.
Kolte, S.J., 1995. Insect pests of castor In: Castor diseases and crop improvement. Shipra publications, Delhi. India. pp: 95.
Lichtenstein, C. & J. Draper, 1985. Genetic engineering of plants. In: D.M. Glover (Ed.), DNA cloning, a practical perspective, vol II, pp. 67–140, IRL press, Washington DC.
Narayanan, E.S., 1959. Insect pests of castor. In: L.G. Kulkarni (Ed.),‘Castor’. Indian Central Oilseeds committee, Hyderabad, Chap, 13.
Ramesh, S., D. Nagadhara, I.C. Pasalu, A. Padma Kumari, N.P. Sarma, V.D. Reddy, & K.V. Rao, 2004. Development of stem borer resistant transgenic parental lines involved in the production of hybrid rice. J Biotechnol. 111: 131–141.
Reddy, K.R.K., N. Ramaswamy, & B. Bahadur, 1987. Cross incompatibility between Ricinus and Jatropha, Plant Cell Incompatibility Newsletter 19: 60–65.
Sambrook, J. & D.W. Russel, 2001. Molecular Cloning, a laboratory manual. 3ed. Vol. 1–3. Cold Spring Harbor laboratory Press, Cold spring harbor, New York.
Sanghai-Maroof, M.A., K.M. Soliman, R.A. Jorgensen & R.W. Allard, 1984. Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, population dynamics. Proc. Nat. Acad. Sci. USA. 81: 8014–8018.
Sathaiah, V. & T.P. Reddy, 1985. Seed protein profiles of Castor (Ricinus communis l.) and some Jatropha species, Genetica Agraria 39: 35–43.
Smith, R.H. & E.E. Hood, 1995. Agrobacterium tumefaciens transformation of monocotyledons. Crop Sci. 35: 301–309.
Sneh, B. & S. Gross, 1983. Biological control of the Egyptian cotton leafworm, Spodoptera littoralis (Lepidoptera, Noctuidae) in cotton and alfalfa fields, using a preparation of Bacillus thuringiensis ssp. entomocidus supplemented with adjuvants. Z. Angew. Entomol. 96: 418–424.
Stewart, Jr. C.N., M.J. Adang, J.N. All, H.R. Boerma, G. Cardineau, D. Tucker & W.A. Parrott, 1996. Genetic transformation, recovery and characterization of fertile soybean transgenic for a synthetic Bacillus thuringiensis cryIA(c) gene. Plant Physiol. 112: 121–129.
Sujatha, M. & T.P. Reddy, 1998. Differential cytokinin effects on the stimulation of in vitro shoot proliferation from meristematic explants of castor (Ricinus communis L.). Plant Cell Rep. 17: 561–566,.
Sujatha, M. & M. Sailaja, 2005. Stable genetic transformation of castor (Ricinus communis L.) via Agrobacterium tumefaciens-mediated gene transfer using embryo axes from mature seeds. Plant Cell Rep. 23: 803–810.
Vaeck, M., A. Reynaerts, H. Hoftey, S. Jansens, M. De Beuckleer, C. Dean, M. Zabeau, M. Van Montagu, & J. Leemans, 1987. Transgenic plants protected from insect attack. Nature. 327: 33–37.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Malathi, B., Ramesh, S., Rao, K.V. et al. Agrobacterium-mediated genetic transformation and production of semilooper resistant transgenic castor (Ricinus communis L.). Euphytica 147, 441–449 (2006). https://doi.org/10.1007/s10681-005-9043-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-005-9043-x