Abstract
Leaves of trees experience the maximum brunt of exposure and undergo certain changes in physiological traits responding to air pollution, and then, the specific leaf traits can be the indicators of air pollution in an area. However, due to the diversity of sources, the composition of air pollutants is very complex. This makes it difficult to predict air pollution using physiological differentiation of leaves. The purpose of this investigation was to examine potential of Ligustrum lucidum Ait. leaf measurement as a method to predict the air pollutants in Luoyang, China. Leaves of roadside L. lucidum were studied from the city center with serious air pollution to relatively unpolluted areas. Leaf size, stomatal traits, and non-structural carbohydrate were measured. The particulate and gaseous pollutants (including sulfur dioxide, nitrogen dioxide, carbon monoxide, and ozone) were investigated too. The results showed that the leaf area and soluble sugar content decreased, while the aspect ratio of leaves increased in heavily polluted areas. As pollution increased, the stomatal traits in different crown positions were changed differently. No significant correlation was found between ozone content and the measured traits of leaves. The responses found in the physiological differentiation of the leaves reflect acclimation to air pollution. The soluble sugar content of the leaves could be used to indicate the short-term stress of air pollution, the area, and aspect ratio of leaves are indicative of the long-term stress due to air pollution. Therefore, physiological traits of L. lucidum leaves appeared to be significant predictive factors for the air pollutants in urban areas.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rapid economic growth, industrialization, and urbanization have resulted in deterioration of air quality in developing countries like China (Alnawaiseh et al., 2015; Angelevska et al., 2021). Action and improvement needs to be done to counteract this increasingly prevalent health risk, for instance, alternatives to crop residue burning (Kumar & Singh, 2021), green transition from coal to other energy resources (Neru, 2021), planning road transportation, and shifting from private car to public transportation based on reducing the impact of road transportation on air quality (Abdulkareem et al., 2020; Angelevska et al., 2021). Besides these air pollution mitigation measures, urban greening is an effective and long-term strategy of controlling air pollution in urban areas (Grote et al., 2016; Nowak et al., 2006; Wang, 2014). Plants can clean the air, block the particles in haze, and absorb toxic gases (Pugh et al., 2012). But when the pollutants in the air far exceed the carrying capacity of plants, it impacts plant growth negatively (Ozolincius et al., 2005; Yue et al., 2017). Due to their unique position in plants, the leaves change first under different air pollution stress. Firstly, although leaves have the function of capturing dust, leaves are saturated when the dust particle concentration is too high, affecting the plant respiration (Bae et al., 2009). Secondly, in haze weather, the dust particles in the air cover the sunlight, thus affecting photosynthesis, which is not conducive to the plant growth (Lin et al., 2019). Finally, the toxic substances in the air can impair the growth and development of the leaves (Schiffgens-Gruber & Lütz, 1992; Steubing et al., 1989), and even lead to the plant death in serious pollution (Saltan et al., 2020).
Ligustrum lucidum Ait., a native evergreen species of China, is cultivated in many southern and southwestern provinces (Wang, 1961). It is also common in North Korea, India, and Nepal. L. lucidum is widely distributed in the landscape because of its dense branches and leaves, strong germination, and low requirements for soil and climate (FRPS, 1992). Ligustrum lucidum is an ornamental and roadside tree species, which is highly resistant to sulfur dioxide and other toxic gases, and can also tolerate high levels of dust and smoke pollution (Graziani et al., 2019; Liang et al., 2008; Rossini Oliva & Valdés, 2004).
The strong tolerance of plants to polluted air may be obtained by changing the physiological, ecological, or structural characteristics. For instance, the leaf area became smaller (Khan et al., 1990), the contents of pigment and sugar in leaves of plants decreased (Yousafzai et al., 2018), while ascorbic acid and sulfate increased in the areas with high air pollution (Appalasamy et al., 2017; Irerhievwie et al., 2014; Sharma & Tripathi, 2009). Therefore, leaf-level physiological traits are often used as a valid method for studying air pollution (Terekhina & Ufimtseva, 2020). However, the source of air pollution is diverse, including thermal power plants (Zhu et al., 2016), industrial enterprises (Graziani et al., 2019), emissions of motor vehicles (Alnawaiseh et al., 2015; Dyvak et al., 2018), and, often to a certain extent, crop residue burning (Kumar & Singh, 2021; Li et al., 2021). Due to the diversity of sources, the composition of air pollutants is very complex (Gibergans-Baguena et al., 2020; Zhang et al., 2012).This makes it difficult to predict air pollution using physiological differentiation of leaves. The aim of this work was to assess the impact of air pollutants from the urban environment on the physiological traits of L. lucidum leaves, and which traits should be used to assess the quality of the urban environment.
Materials and methods
Study area
This study was carried out in Luoyang, a tourist city in the center area of China. Luoyang is famous for its 5000 years of civilization, 4000 years of city building, and 1500 years of capital building. Unfortunately, the air quality in Luoyang is deteriorated in recent years. According to the data released by Luoyang Environmental Protection Department, PM2.5 (i.e., fine particles with aerodynamic diameter ≤ 2.5 µm), one of the most important parameters reflecting the air pollution level, reached through 879 µg/m3 on January 4, 2016, which was due to be the large-scale dust storm that day and local industrial pollutants (Tian et al., 2018). Luoyang used to be one of the most important old industrial bases in China. The industrial production brought enormous economic benefits, but also produced a lot of pollutants (Wang et al., 2010). Additionally, the air quality in Luoyang continue to deteriorate due to the urbanization spread, in particular the rapid increase of motor vehicles, which also led to a rise in exhaust emissions (Zhang et al., 2012).
Sampling sites
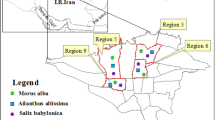
Sampling was conducted along Wangcheng Avenue in Luoyang, China. Wangcheng Avenue is a road with a high level of urbanization and traffic. The roadside trees are dominated by L. lucidum. Six sampling sites were selected along the road (Fig. 1). South of the intersection of Wangcheng Avenue and Zhongzhou Road (Site 1) is situated in the city center with a high density (> 2600 pcu/h) and low speed (10–15 km/h) of traffic. North of the intersection of Wangcheng Avenue and Yiluo Road (Site 2) is situated in the suburbs with about 1100 pcu/h traffic density and 40 km/h speed of vehicles. North of Yichuan County (Site 3) is situated in a newly developed area of the County with high traffic density (about 900 pcu/h) and 60 km/h speed of vehicles. South of Yichuan County (Site 4) is closer to two coal power plants in the east of Yichuan County with about 350 pcu/h traffic density and 70 km/h speed of vehicles. Minggao Town (Site 5) and Tianhu Town (Site 6) were chosen as controls and are about 50 km and 60 km away from the city center respectively, making them the least polluted sites in a sparsely populated area with low traffic density (about 180 pcu/h) and 80 km/h speed of vehicles, absence of industry around farmland.
Air quality monitoring
The air quality in the six sampling sites was investigated using micro air quality monitor (OSEN-AQMS, Osen Purification Technology Co., Ltd., Shenzhen, China) for a period of one month (May 2017). Data were collected at a height of 2 m and in 3-h intervals between 06:00 to 20:00 h (local time) for each day. The monitored particulate and gaseous pollutants included particles with aerodynamic diameter ≤ 2.5 µm (PM2.5), particles with aerodynamic diameter ≤ 10 μm (PM10), sulfur dioxide (SO2), nitrogen dioxide (NO2), carbon monoxide (CO), and ozone (O3). The mass concentration value of each pollutant is then converted into the air quality index (AQI) values using the Chinese Standard HJ 633–2012 (China SAotPsRO, 2012), which equals the American standard (US-EPA, 2003). The highest AQI value for the individual pollutants is the AQI value for that day in the study area.
Leaf size measurements
Sampling was conducted on May 31, 2017. At each sampling site, three healthy trees with similar size were chosen (Table 1). With the help of a curved arm aerial vehicle, the fresh leaves of the current year’s branches were collected carefully from the upper, middle, and lower crown facing the street side. The leaves were sealed in plastic bags and were stored in a refrigerator (0–4 °C) until transporting them back to the laboratory. Ten leaves were sampled from each crown position (i.e., 30 leaves per tree). Nine of the leaves were used for stomatal imprints, the others for leaf size and non-structural carbohydrate.
The leaf area, length, and width of leaves were measured with a image computer analysis system (TDY5.2, Tianyu Science and Technology Co., Ltd., Beijing, China). The aspect ratio of leaves was calculated as the ratio between leaf length and width (Fascella et al., 2014).
Stomatal trait measurements
Stomata in lower epidermis were selected to measure the traits in this study. Two imprints closed to both sides of the main leaf veins from the lower epidermis were taken using the clear nail polish method (Hilu & Randall, 1984). An average of 30 stomata was measured per imprint. Using the image computer analysis system (TDY5.2, Tianyu Science and Technology Co., Ltd., Beijing, China), the following stomatal traits were measured: length and width of the guard cells, length and width of stomata, and stoma density.
Non-structural carbohydrate measurements
The leaf area measured leaves were dried in an oven at 105 °C for 30 min, 70 °C for 2–3 days, and then were ground to pass through 80-mesh. The content of soluble sugar and starch was determined by the modified phenol sulfuric acid method (Buysse & Merckx, 1993). The powdered samples were extracted with 80% v/v ethanol. The extracted solution was used for determining the soluble sugar content with a spectrophotometer (722S, Jinghua Instrument Co., Shanghai, China) at a wavelength of 485 nm. The residues were hydrolyzed in 39.2 mol/l perchloric acid solution to measure the starch content identically. Total non-structure carbohydrate (TNC) was the sum of soluble sugar and starch content (Zhang et al., 2013).
Statistical analysis
Differences in leaf traits among the sampling sites were evaluated by analyses of variance (ANOVA), with the significance assessed at the alpha value < 0.05. Tests of the normality and homogeneity of variances were performed using the Shapiro–Wilk and Levene tests. The Pearson’s correlation among leaf traits and air pollutants including CO, NO2, SO2, O3, PM2.5, and PM10 were determined (Yousafzai et al., 2018). All analyses were performed using the IBM SPSS Statistics software (Version 24.0, International Business Machines Corporation, Armonk, New York, United States).
Results and discussion
Air quality
Table 2 showed the mean values for each pollutant measured. The highest AQI was observed in the city center (Site 1), whereas the lowest AQI was found at a location far away from the city center (Site 6). From Site 1 to Site 6, the AQI tended to decrease. Site 4 showed the second highest AQI after Site 1. Results from ANOVA analysis showed that there was a significant difference for AQI between each site (p < 0.05) except Site 2 and Site 3. From the city center to the outside, PM2.5, PM10, and CO showed similar patterns of change to the AQI; the sites with the high traffic density had the high concentration value of pollutants, which confirmed that the vehicular traffic was an active source of pollution (Dyvak et al., 2018). The maximum values of NO2 and SO2 were found at Site 4 which confirmed that air quality of the site was affected by the additional complex mixture of pollutants from the two nearby coal power plants (Shon et al., 2019). Site 4, which was located 5 km southwest of the two power stations, had relatively higher SO2 and NO2 levels compared to the other Sites and attributed this to strong NE winds (Chen, 1998; Liu et al., 2018). The amount of O3 at all sites varied from 87 to 167 µg/m3, as shown in Table 2, is higher than the estimated background ozone level of 70 µg/m by World Health Organization (WHO, 2006). O3 was found in high concentrations > 100 mg/kg (air quality guideline) at the sites except Site 2 and Site 6, which might be harmful to public health. The causes of O3 formation are complex. For instance, the amount of O3 may occasionally exceeded the guideline value due to natural causes (Kim et al., 2021). However, Site 1 showed high O3 concentrations, up to 167 µg/m3 which exceeded the Interim target-1 (167 µg/m3) level established by WHO (2006). Such high concentrations of O3 would result in middle level ambient pollution in the city center, which is believed to be influenced by traffic exhaust, energy consumption, and industrial emission changes besides meteorological factors. This is confirmed by the analysis of O3 sources in many cities (Unger et al., 2020; Yan et al., 2021).
Leaf size
The leaf area of L. lucidum at the Site 1 and 2 with serious pollution was smaller than those at sites with higher air quality (Table 3). Our study was consistent with the observations of previous studies; i.e., air pollution led to the reduction of leaf area (Cotrozzi, 2019; Khan et al., 1990; Pandey, 2005). Although the leaf area did not show a significant difference among the sites (Table 3), there was a significant negative correlation with PM2.5, PM10, CO, and NO2 relating to the crown positions (Table 4). Maher et al. (2008) confirmed that air pollutants increased with height in the crown by detecting the concentrations of heavy metals in the leaves. The uneven distribution of air pollutants in the crown may affect the growth and development of leaves. However, the variation of leaf area in crown may also be due to the long-term evolution of plants (Nikolov et al., 2019). Thereafter, to understand the relationship between leaf area and air pollutants, there are many challenges ahead. The variation of leaf area at Site 4 indicated negative responses in leaf size exposed to coal-fired power plant emission consistent with the studies by Jones et al. (1987), suggesting the leaf area are indicators of potential in monitoring air pollution. A controlled experiment made by Trlica et al. (1985) indicated that leaves possibly were injured by coal-fired power plant emission, and the leaf injury increased near the plant. Many studies have demonstrated that the leaf area can be used as a measure of the severity of air pollution. Deng et al. (1981), for instance, showed that leaf area of Robinia pseudoacacia could be used as bio-indicators for SO2 pollution in Shenyang, China. However, some studies have shown that the leaf area increases due to air pollution. For instance, In the works of Chen et al. (2015), leaf area was considerably larger in a site with high concentration PM 2.5 than in the control site. The adaptive change of leaf area caused by air pollution has complex species dependence and is also related to the stress mechanism of one or more air quality parameters (Wuytack et al., 2011). Clearly, the effect of air pollution on leaf area is related to the type of pollutants..
Interestingly, the aspect ratio of leaves in the areas with serious pollution is larger than that at the Site 5 and Site 6 with better air quality (Table 3). These indicated the leaves became narrow due to air pollution. Large leaves are more likely to retain dust (Chaturvedi et al., 2013). Narrowing leaves in a polluted site could help reduce the risk of being covered by dust (Leonard et al., 2016). Results from correlation analysis showed that the aspect ratio of leaves in the lower crown had a significant positive correlation with AQI, PM2.5, and PM10 of sites (p = 0.05), suggesting the aspect ratio of leaves in the lower crown could be an indicator for particulate matters rather than for other air pollutants.
Stomatal traits
Stomatal traits responding to air pollutants of the sampling sites varied among the crown positions (Tables 5 and 6). Despite differences in stomatal traits of leaves in whole crown or in middle crown, the traits did not reveal a regular pattern of change among sites (Table 5) and did not show significant correlation with air quality parameters, except for guard cell width in upper crown (Table 6). However, the size (length and width) and density of stomata in the upper crown at the polluted sites were much smaller than the Site 5 and Site 6 with better air quality. The reduction in size and density of stomata could be considered as a favorable adaptation as it was might be important for preventing the capture of pollutants (Hoshika et al., 2015). However, the first emergency response of stomata to the stress of air pollutants may be the closure of some stomata, which in turn reduces the stomatal conductance of leaves. Consequently, this study demonstrates the inability of using these L. lucidum stomatal traits (except guard cell width in upper crown) to monitor air pollutants concentration.
The size of guard cell was larger in leaves from the upper crown at the polluted sites than in those of leaves at the Sites 5 and 6 with better air quality, except for guard cell length at site 3 (Table 5). Furthermore, the guard cell length from the upper crown showed a significant positive correlation with AQI, PM10, PM2.5, and CO content of the sites (Table 6). The guard cells are primarily and directly affected by the stream of the pollutants entering the stomata, as the stomata are open (Christodoulakis, 1993). Ozone, for example, acts directly on guard cells by altering the activity of ion channels in the guard cell plasma membrane (Torsethaugen et al., 1999). Thus, increasing the size of guard cells was good for combating the air pollution (Iqbal et al., 2010a).
Following air quality of sampling sites, the stomatal traits of leaves in lower crown revealed an opposite pattern of change compared with that in upper crown (Table 5). For example, there was a marked increase in guard cell size of leaves in upper crown, whereas a slight decrease was observed in lower crown of L. lucidum with the increase of pollutant concentrations in sampling sites. This study suggested that the response of leaf stomata to air pollutants may also depend on the height and crown structure of trees. Under the influence of tree height, crown structure, and wind speed, the distribution of air pollutants in the canopy was heterogeneous (Albaugh et al., 1992). Wang (2014) found the vertical distribution of atmospheric particle concentration was related to tree height in the main roadside. However, it was difficult to accurately measure the distribution of pollutants due to the complexity in the array of leaves and branches and the complex process of air flow within a crown (Bache, 1979; Lin & Khlystov, 2012). Detailed estimates of the effects of crown position on pollutants collection by leaves still require a lot of work to be done.
Non-structural carbohydrates
The soluble sugar content in leaves at the polluted sites was generally lower than in those of leaves at the site with better air quality, except for the soluble sugar content at Site 3. The lowest soluble sugar content in leaves was in the Site 1 (Table 7). This was in agreement with Liang et al. (2008). Their study revealed a loss of soluble sugar in all tested species including L. lucidum at polluted sites. Inhibitory action of pollutants on photosynthesis in leaves was believed to reduce availability of soluble sugar (Iqbal et al., 2010b; Irerhievwie et al., 2014). The inhibitory effect included blocking the light by suspended particulate matter in the air and stomatal occlusion due to the deposition on leaf surface (Bae et al., 2009). This was revealed in this study by the negative correlation between the soluble sugar content in leaves and particle content in air (Table 8). Some studies have observed that a decreased soluble sugar was linked to various toxic gases (He, 2002; Ito et al., 1985). However, most of these studies were carried out using high pollutant concentrations comparing to that in real atmosphere. We reported here that the soluble sugar in leaves showed no significant correlation with SO2, NO2, and O3 concentration at the sites. The soluble sugar in leaves showed a significant negative correlation with CO concentration at the sites. However, the significant correlation was insufficient to draw conclusions on the effect of CO on soluble sugar. The leaves could not only absorb CO from the atmosphere (Bidwell & Fraser, 1972), but also produce CO themselves (Fischer & Ttge, 1978).Thus, we think the decreased soluble sugar may be an indicator for early damage of L. lucidum leaves, which, among other stress factors, reacted sensitively to the increasing load of particulate matter pollutants.
Our results showed the starch content was much higher in lower crown layer at Site 4 than that at other sites (Table 7). An unusual increase of starch content at polluted Site 4 could be due to much higher SO2 and NO2 in the site which closer the two coal power plants. NO2 and SO2 are two types of major pollutants from coal power plants (Sharma & Tripathi, 2009; Zhu et al., 2016). Steubing et al. (1989) found that carbohydrate metabolism in Allium ursinum leaves was altered by fumigation with moderate doses of SO2, NO2, and O3, leading to starch accumulation in the leaves. Schiffgens-Gruber and Lütz (1992) also reported an increase in the starch contents in spruce leaves after treatment with gas mixtures of O3, NO2, and SO2. However, according to Bücker and Ballach (1992), air pollution reduced the starch contents in the leaves of trees. These findings of the effects of NO2 and SO2 on starch content in leaves are inconsistent, therefore implying the complexity of starch responding to air pollution. The Pearson’s correlation analysis in our study showed the starch content in leaves did not show significant correlation with any air quality parameters of the sites (Table 8). Starch was one of the main substances stored in plants for long term (Zeeman et al., 2010). In the process of plant growth, the conversion between starch and soluble sugar is always going on. Whereas soluble sugar was generally reduced, both decreased and increased starch may be found in leaves (Bücker & Ballach, 1992; Fialho & Bücker, 1996). Also, those of starch in the leaves displayed unstable seasonal variations, compared with the soluble sugar content (Liu et al., 2019). In conclusion, the starch content in leaves of L. lucidum is not a good predictor of the air pollutants in Luoyang urban areas.
The content of TNC in leaves of L. lucidum at Site 1 was low, especially in the lower crown layer, which was significantly lower than that at the other sites (Table 7). Air pollution reduced the content of TNC, which is consistent with the research results of Cao et al. (2017) on Machilus ichangensis and Taxus wallichiana. The difference of TNC response to air pollutants between crown layers was related to the daylighting of leaves. According to previous studies (Legner et al., 2014; Li et al., 2020, Masarovicová & štefančík, 1990), crown structure can influence the growth of trees by influencing the proportion of sun and shade leaves. There was no significant correlation between TNC content and air quality parameters (Table 8), which corresponded to starch content. Bücker and Ballach (1992) also found that the response pattern of TNC stored in trees to pollution stress is the same as that of starch. This may be due to the dominance of starch in TNC (83% in this study).
Non structural carbohydrate stored in trees, like starch, has been demonstrated to confer resilience to environmental stress in the short term (Dong et al., 2018; Thalmann & Santelia, 2017). However, the observed changes in non structural carbohydrates in leaves during pollution stress result from multiple activities: flowering (Iqbal et al., 2010b), fruiting (Mesaa et al., 2016), and seasonal growth (Ramírez-Briones et al., 2017). Only one data collection was conducted in this study, which makes the knowledge still fragmentary and hinders our full understanding of the relationship between non structural carbohydrates and air pollutants. To evaluate how leaves respond to interacting factors, a more clearly view of the non structural carbohydrate change during air pollution stress will require more repetitive experiments conducted under different growth conditions.
Conclusions
Air pollution has affected growth of leaves in the L. lucidum. Leaf area and soluble sugar content decreased in trees in polluted area, while aspect ratio of leaves increased. The changes in the stomatal traits with the increase of environmental pollution depend on the sampling position within crown. It is concluded that the area, aspect ratio, and soluble sugar content of leaves and guard cell width of stomas can be used as indicators of air pollution in Luoyang city, but the sampling position in the crown might need to be considered. It is also suggested that such changes in the L. lucidum leaves of polluted areas could be of great significance in determining the level of air pollution and the types of air pollutants.
Data availability
The datasets supporting the conclusions of this article are included within the article.
References
Abdulkareem, M. N., Alsaidi, L. R., Yazid, M. R. M., Borhan, M. N., & Mahdi, M. S. (2020). Traffic congestion: Shift from private car to public transportation. Civil Engineering Journal, 6(8), 1547–1554. https://doi.org/10.28991/cej-2020-03091566
Albaugh, T., Mowry, F., & Kress, L. (1992). A field chamber for testing air pollution effects on mature trees. Journal of Environmental Quality, 21, 476–485. https://doi.org/10.2134/jeq1992.00472425002100030028x
Alnawaiseh, N. A., Hashim, J. H., & Md Isa, Z. (2015). Relationship between vehicle count and particulate air pollution in Amman. Jordan. Asia-Pacific Journal of Public Health, 27(2), 1742–1751. https://doi.org/10.1177/1010539512455046
Angelevska, B., Atanasova, V., & Andreevski, I. (2021). Urban air quality guidance based on measures categorization in road transport. Civil Engineering Journal, 7(2), 253–267. https://doi.org/10.28991/cej-2021-03091651
Appalasamy, M., Varghese, B., Ismail, R., & Sershen, N. (2017). Responses of Trichilia dregeana leaves to sulphur dioxide pollution: A comparison of morphological, physiological and biochemical biomarkers. Atmospheric Pollution Research, 8(4), 729–740. https://doi.org/10.1016/j.apr.2017.01.005
Bache, D. H. (1979). Particle transport within plant canopies—I. A Framework for Analysis. Atmospheric Environment, 13, 1257–1262. https://doi.org/10.1016/0004-6981(79)90080-5
Bae, K. K., Lee, D. K., & Park, Y. D. (2009). Effects of artificial asian dust on photosynthesis, respiration, growth and stoma in five tree species. Journal of Korean Forestry Society, 24(3), 317. https://doi.org/10.1016/0041-0101(86)90158-3
Bidwell, R., & Fraser, D. (1972). Carbon monoxide uptake and metabolism by leaves. Canadian Journal of Botany, 50, 1435–1439. https://doi.org/10.1139/b72-174
Bücker, J., & Ballach, H. J. (1992). Alterations in carbohydrate levels in leaves of Populus due to ambient air pollution. Physiologia Plantarum, 86(4), 512–517. https://doi.org/10.1111/j.1399-3054.1992.tb02163.x
Buysse, J. A. N., & Merckx, R. (1993). An improved colorimetric method to quantify sugar content of plant tissue. Journal of Experimental Botany, 44(10), 1627–1629. https://doi.org/10.1093/jxb/44.10.1627
Cao, J., Zhan, C., Hao, Y., & He, S. (2017). Differential responses in non-structural carbohydrates of Machilus ichangensis Rehd. et Wils. and Taxus wallichiana Zucc. Var. chinensis (Pilg.) Florin seedlings to elevated ozone. Forests, 8(9), 1–12. https://doi.org/10.3390/f8090323
Chaturvedi, R. K., Prasad, S., Rana, S., Obaidullah, S. M., Pandey, V., & Singh, H. (2013). Effect of dust load on the leaf attributes of the tree species growing along the roadside. Environmental Monitoring and Assessment, 185(1), 383–391. https://doi.org/10.1007/s10661-012-2560-x
Chen, J. (1998). Analysis of the climatic pattern and the single station index of the situation before the high wind in Luoyang city. Sichuan Meteorology, 18(4), 16–19. [in Chinese].
Chen, Z., Shang, H., Cao, J., & Yu, H. (2015). Effects of ambient ozone concentrations on contents of nonstructural carbohydrates in Phoebe bournei and Pinus massoniana seedlings in subtropical China. Water, Air, Soil Pollution, 226(9), 1–8. https://doi.org/10.1007/s11270-015-2555-7
China SAotPsRo. (2012). Technical requlation on ambient air quality index (on trial), HJ 633–2012, Beijing, China. [in Chinese]
Christodoulakis, N. S. (1993). Air pollution effects on the guard cells of the injury resistant leaf of Laurus nobilis L. Bulletin of Environmental Contamination & Toxicology, 51(3), 471–478.
Cotrozzi, L. (2019). Leaf demography and growth analysis to assess the impact of air pollution on plants: A case study on alfalfa exposed to a gradient of sulphur dioxide concentrations. Atmospheric Pollution Research, 11, 1–29.
Deng, S., Jiang, Z., & Dong, H. (1981). Evaluation of air pollution in Shenyang by the change of Robinia pseudoacacia leaf area. Journal of Liaoning University (natural Sciences Edition), 1, 68–72. [in Chinese].
Dong, S., Zhang, J., & Beckles, D. M. (2018). A pivotal role for starch in the reconfiguration of 14C-partitioning and allocation in Arabidopsis thaliana under short-term abiotic stress. Scientific Reports, 8(1), 9314. https://doi.org/10.1038/s41598-018-27610-y
Dyvak, M., Darmorost, I., Shevchuk, R., Manzhula, V., & Kasatkina, N. (2018). Correlation analysis traffic intensity of the motor vehicles and the air pollution by their harmful emissions, 14th International Conference on Advanced Trends in Radioelecrtronics, Telecommunications and Computer Engineering (TCSET). Lviv-Slavske, Ukraine, pp. 855–858. https://doi.org/10.1109/TCSET.2018.8336331
Fascella, G., Darwich, S., & Rouphael, Y. (2014). Validation of a leaf area prediction model proposed for rose. Chilean Journal of Agricultural Research, 73(1), 73–76. https://doi.org/10.4067/S0718-58392013000100011
Fialho, R., & Bücker, J. (1996). Changes in levels of foliar carbohydrates and myo-inositol before premature leaf senescence of Populus nigra induced by a mixture of O3 and SO2. Canadian Journal of Botany, 74, 965–970. https://doi.org/10.1139/b96-120
Fischer, K., & Ttge, U. (1978). Light-dependent net production of carbon monoxide by plants. Nature, 275, 740–741. https://doi.org/10.1038/275740a0
FRPS, E. Co. (1992). Flora Reipublicae Popularis Sinicae (FRPS). Ligustrum lucidum, 61. Science Press, Beijing
Gibergans-Baguena, J., Hervada-Sala, C., & Jarauta-Bragulat, E. (2020). The quality of urban air in Barcelona: A new approach applying compositional data analysis methods. Emerging Science Journal, 4(2), 113–121. https://doi.org/10.28991/esj-2020-01215
Graziani, N., Mateos, A., Silva, J., Ramos, S., Homem, V., Ratola, N., & Carreras, H. (2019). Estimation of urban POP and emerging SVOC levels employing Ligustrum lucidum leaves. Atmospheric Pollution Research, 10(5), 1524–1530. https://doi.org/10.1016/j.apr.2019.04.010
Grote, R., Samson, R., Alonso, R., Amorim, J. H., Cariñanos, P., Churkina, G., Fares, S., Thiec, D. L., Niinemets, Ü., & Mikkelsen, T. N. (2016). Functional traits of urban trees: Air pollution mitigation potential. Frontiers in Ecology & the Environment, 14(10), 543–550. https://doi.org/10.1002/fee.1426
He, T. G. (2002). The impact of SO2 pollution upon the dissoluble sugar and the content of chlorophyll of lamina. Journal of Huaibei Industry Teachers College, 23(2), 55–57. [in Chinese].
Hilu, K., & Randall, J. (1984). Convenient method for studying grass leaf epidermis. Taxon, 33(3), 413–415. https://doi.org/10.2307/1220980
Hoshika, Y., Watanabe, M., Kitao, M., Häberle, K. H., Grams, T. E., Koike, T., & Matyssek, R. (2015). Ozone induces stomatal narrowing in European and Siebold’s beeches: A comparison between two experiments of free-air ozone exposure. Environmental Pollution, 196, 527–533. https://doi.org/10.1016/j.envpol.2014.07.034
Iqbal, M., Jura-Morawiec, J., Włoch, W., & Mahmooduzzafar (2010a). Foliar characteristics, cambial activity and wood formation in Azadirachta indica A. Juss. as affected by coal-smoke pollution. Flora, 205(1), 61–71. https://doi.org/10.1016/j.flora.2008.12.003
Iqbal, M., Mahmooduzzafar, Nighat, F., & Khan, P. R. (2010b). Photosynthetic, metabolic and growth responses of Triumfetta rhomboidea to coal-smoke pollution at different stages of plant ontogeny. Journal of Plant Interactions, 5(1), 11–19. https://doi.org/10.1080/17429140902996165
Irerhievwie, G. O., Akpoghelie, J. O., & Esiefarienrhe, E. (2014). Evaluation of some plant species for soluble sugar and air pollution tolerance index in Oleh Metropolis, Isoko South L. G. A., Delta State, Nigeria. Journal of Emerging Trends in Engineering and Applied Sciences, 5(5), 323–328
Ito, O., Okano, K., Kuroiwa, M., & Totsuka, T. (1985). Effects of NO2 and O3 alone or in cmbination on kidney bean plants ( Phaseolus vulgaris L.): Growth, partitioning of assimilates and root activities. Journal of Experimental Botany, 36, 652–662. https://doi.org/10.1093/jxb/36.4.652
Jones, H., Noggle, J., & McDuffie, C. (1987). Effects of Emissions from a coal-fired power plant on soybean production. Journal of Environmental Quality, 16(4), 96–306. https://doi.org/10.2134/jeq1987.00472425001600040002x
Khan, A. M., Pandey, V., Shukla, J., Singh, N., Yunus, M., Singh, S. N., & Ahmad, K. J. (1990). Effect of thermal power plant emissions on Catharanthus roseus L. Bulletin of Environmental Contamination and Toxicology, 44(6), 865–870. https://doi.org/10.1007/BF01702176
Kim, H., Lee, D., Ngan, F., Kim, B., Kim, S., Bae, C., & Yoon, J-H. (2021). Synoptic weather and surface ozone concentration in South Korea. Atmospheric Environment, 244, 117985
Kumar, P., & Singh, R. K. (2021). Selection of sustainable solutions for crop residue burning: An environmental issue in northwestern states of India. Environment, Development and Sustainability, 23(3), 3696–3730. https://doi.org/10.1007/s10668-020-00741-x
Legner, N., Fleck, S., & Leuschner, C. (2014). Within-canopy variation in photosynthetic capacity, SLA and foliar N in temperate broad-leaved trees with contrasting shade tolerance. Trees- Structure and Function, 28(1), 263–280. https://doi.org/10.1007/s00468-013-0947-0
Leonard, R. J., Mcarthur, C., & Hochuli, D. F. (2016). Particulate matter deposition on roadside plants and the importance of leaf trait combinations. Urban Forestry & Urban Greening, 20, 249–253. https://doi.org/10.1016/j.ufug.2016.09.008
Li, J., Wang, X., Wang, Z. -H., Wang, B., Wang, C. -Z., Deng, M. -F., & Liu, L. -L. (2020). Effects of ozone and aerosol pollution on photosynthesis of poplar leaves. Chinese Journal of Plant Ecology, 44(8), 854–863. [in Chinese].
Li, T., Dai, Q., Bi, X., Wu, J., & Zhang, Y. (2021). Size distribution and chemical characteristics of particles from crop residue open burning in North China. Journal of Environmental Sciences, 109(30), 66–76. https://doi.org/10.1016/j.jes.2021.02.019
Liang, S. -Y., Hu, H. -B., & Xia, S. -G. (2008). Effects of air pollution on physio-biochemical characteristics of three street trees. China Forestry Science & Technology, 22(3), 29–32. [in Chinese].
Lin, M. Y., & Khlystov, A. (2012). Investigation of ultrafine particle deposition to vegetation branches in a wind tunnel. Aerosol Science & Technology, 46(4), 465–472. https://doi.org/10.1080/02786826.2011.638346
Lin, X. -Y., Li, Y. -H., Li, H. -M., & Song, Q. -W. (2019). Dust retention and photosynthesis of eight species of Arbor trees. Fujian Journal of Agricultural Sciences, 34(3), 313–318. [in Chinese].
Liu, M., Wang, Z., Li, Q., Xiao, W., & Song, X. (2019). Photosynthesis, ecological stoichiometry, and non-structural carbohydrate response to simulated nitrogen deposition and phosphorus addition in Chinese fir forests. Forests, 10, 1068. https://doi.org/10.1007/s11104-021-04831-1
Liu, Y., Xu, X., Huang, M., & Liu, Q. (2018). Characteristics of change in concentrations of atmospheric pollutants in Luoyang, He′nan Province. Research of Soil & Water Conservation, 25(1), 178–182. [in Chinese].
Maher, B., Moore, C., & Matzka, J. (2008). Spatial variation in vehicle-derived metal pollution identified by magnetic and elemental analysis of roadside tree leaves. Atmospheric Environment, 42, 364–373. https://doi.org/10.1016/j.atmosenv.2007.09.013
Masarovicová, E., & štefančík, L. (1990). Some ecophysiological features in sun and shade leaves of tall beech trees. Biologia Plantarum, 32(5), 374–387. https://doi.org/10.1007/BF02898503
Mesaa, K., Serra, S., Masia, A., Gagliardi, F., Bucci, D., & Musacchi, S. (2016). Seasonal trends of starch and soluble carbohydrates in fruits and leaves of ‘Abbe Fetel’ pear trees and their relationship to fruit quality parameters. Sciencia Horticulturae, 211, 60–69. https://doi.org/10.1016/j.scienta.2016.08.008
Neru, V. (2021). Romania’s coal-fired power plants efficiency and pollution in the context of the European green deal. Theoretical and Applied Economics, XXVIII, 117–134.
Nikolov, L. A., Runions, A., Das Gupta, M., & Tsiantis, M. (2019). Chapter Five—Leaf development and evolution. In: Grossniklaus U (Editor), Current topics in developmental biology. Academic Press, pp. 109–139. https://doi.org/10.1016/bs.ctdb.2018.11.006
Nowak, D., Crane, D., & Stevens, J. (2006). Air pollution removal by urban trees and shrubs in the United States. Urban Forestry & Urban Greening, 4(3), 115–123. https://doi.org/10.1016/j.ufug.2006.01.007
Ozolincius, R., Stakenas, V., & Serafinaviciute, B. (2005). Meteorological factors and air pollution in Lithuanian forests: Possible effects on tree condition. Environmental Pollution, 137(3), 587–595. https://doi.org/10.1016/j.envpol.2005.01.044
Pandey, J. (2005). Evaluation of air pollution phytotoxicity downwind of a phosphate fertilizer factory in India. Environmental Monitoring and Assessment, 100(1), 249–266. https://doi.org/10.1007/s10661-005-6509-1
Pugh, T., Mackenzie, A., Whyatt, D., & Hewitt, C. N. (2012). Effectiveness of green infrastructure for improvement of air quality in urban street canyons. Environmental Science & Technology, 46(14), 7692–7699. https://doi.org/10.1021/es300826w
Ramírez-Briones, E., Rodríguez-Macías, R., Salcedo-Pérez, E., Martínez-Gallardo, N., Tiessen, A., Molina-Torres, J., Délano-Frier, J. P., & Zañudo-Hernández, J. (2017). Seasonal variation in non-structural carbohydrates, sucrolytic activity and secondary metabolites in deciduous and perennial Diospyros species sampled in Western Mexico. PLoS ONE, 12(10), e0187235. https://doi.org/10.1371/journal.pone.0187235
Rossini Oliva, S., & Valdés, B. (2004). Ligustrum lucidum Ait. f. leaves as a bioindicator of the air-quality in a Mediterranean city. Environmental monitoring and assessment, 96(1–3), 221–232. https://doi.org/10.1023/B:EMAS.0000031729.62970.fa
Saltan, N., Svyatkovskaya, E., & Trostenyuk, N. (2020). Fluctuating asymmetry of leaves of Betula pubescens Ehrh. for assessment of pollution of the urban environment of the Kola North. KnE Life Sciences, 37–43. https://doi.org/10.18502/kls.v5i1.6017
Schiffgens-Gruber, A., & Lütz, C. (1992). Ultrastructure of mesophyll cell chloroplasts of spruce needles exposed to O3, SO2 and NO2 alone and in combination. Environmental and Experimental Botany, 32(3), 243–254. https://doi.org/10.1016/0098-8472(92)90007-O
Sharma, A. P., & Tripathi, B. D. (2009). Biochemical responses in tree foliage exposed to coal-fired power plant emission in seasonally dry tropical environment. Environmental Monitoring and Assessment, 158(1), 197–212. https://doi.org/10.1007/s10661-008-0573-2
Shon, Z.-H., Kang, M., Park, G., & Bae, M. (2019). Impact of temporary emission reduction from a large-scale coal-fired power plant on air quality. Atmospheric Environment: X, 5, 100056. https://doi.org/10.1016/j.aeaoa.2019.100056
Steubing, L., Fangmeier, A., Both, R., & Frankenfeld, M. (1989). Effects of SO2, NO2, and O3 on population development and morphological and physiological parameters of native herb layer species in a beech forest. Environmental Pollution, 58, 281–302. https://doi.org/10.1016/0269-7491(89)90140-1
Terekhina, N., & Ufimtseva, M. (2020). Leaves of trees and shrubs as bioindicators of air pollution by particulate matter in Saint Petersburg. Geography, Environment, Sustainability, 13(1), 224–232. https://doi.org/10.24057/2071-9388-2019-65
Thalmann, M., & Santelia, D. (2017). Starch as a determinant of plant fitness under abiotic stress. The New Phytologist, 214(3), 943–951. https://doi.org/10.1111/nph.14491
Tian, Y., Li J., Liu, M., & Wang, J. (2018). Research of PM2.5 pollution condition in Luoyang city based on principal component regression. Journal of Luoyang Institute of Science and Technology (Natural Science Edition), 28(1), 84–88. [in Chinese]
Torsethaugen, G., Pell, E. J., & Assmann, S. M. (1999). Ozone inhibits guard cell K+ channels implicated in stomatal opening. Proceedings of the National Academy of Sciences of the United States of America, 96(23), 13577–13582. https://doi.org/10.1073/pnas.96.23.13577
Trlica, M., Child, R., & Bauerle, B. (1985). Leaf injury and elemental concentrations in vegetation near a coal-fired power plant. Water, Air, and Soil Pollution, 24(4), 375–396. https://doi.org/10.1007/BF00282489
Unger, N., Zheng, Y., Yue, X., & Harper, K. (2020). Mitigation of ozone damage to the world’s land ecosystems by source sector. Nature Climate Change, 10(4), 134–137. https://doi.org/10.1038/s41558-019-0678-3
US-EPA. (2003). Air quality index: A guide to air quality and your health. EPA-454/K-03–002. https://doi.org/10.1109/eurocon.2017.8011246
Wang, C. -W. (1961). The forests of China. Maria Moors Cabot Foundation Publ, 5. Harvard Univ., Cambridge, Mass
Wang, X. (2014). The spatial and temporal distributions of atmospheric particulate matter in road protection forest, Chinese Academy of Forestry, Beijing. [in Chinese]
Wang, X. G., Li, G., & Tao, P. (2010). The analysis on hydrochemistry environment evolvement of pore groundwater in shallow aquifer in LuoYang city, 2010 4th International Conference on Bioinformatics and Biomedical Engineering, Chengdu, China. https://doi.org/10.1109/icbbe.2010.5517942
WHO. (2006). WHO air quality guidelines for particulate matter, ozone, nitrogen dioxide and sulfur dioxide. Global Update. 2005 Summary of Risk Assessment WHO.
Wuytack, T., Wuyts, K., Van Dongen, S., Baeten, L., Kardel, F., Verheyen, K., & Samson, R. (2011). The effect of air pollution and other environmental stressors on leaf fluctuating asymmetry and specific leaf area of Salix alba L. Environmental pollution, 159(10), 2405e2411. https://doi.org/10.1016/j.envpol.2011.06.037
Yan, Y., Zheng, H., Kong, S., Lin, J., Yao, L., Wu, F., Cheng, Y., Niu, Z., Zheng, S., Zeng, X., Wu, J., Zheng, M., Liu, M., Ni, R., Chen, L., Chen, N., Xu, K., Liu, D., & Qi, S. (2021). On the local anthropogenic source diversities and transboundary transport for urban agglomeration ozone mitigation. Atmospheric Environment, 245(6), 118005. https://doi.org/10.1016/j.atmosenv.2020.118005
Yousafzai, A., Durani, A., Hameedi, A., Mohammadi, M. H., Durrani, H., & Safiullah, K. (2018). Effects of air pollution on chlorophyll content of urban trees leaves. International Journal of Biology Research, 3(1), 287–291. https://doi.org/10.22271/biology
Yue, X., Unger, N., Harper, K., Xia, X., Liao, H., Zhu, T., Xiao, J., Feng, Z., & Li, J. (2017). Ozone and haze pollution weakens net primary productivity in China. Atmospheric Chemistry & Physics, 17(9), 6073–6089. https://doi.org/10.5194/acp-17-6073-2017
Zeeman, S. C., Kossmann, J., & Smith, A. M. (2010). Starch: Its metabolism, evolution, and biotechnological modification in plants. Annual Review of Plant Biology, 61, 209–234. https://doi.org/10.1146/annurev-arplant-042809-112301
Zhang, H., Wang, C., & Wang, X. (2013). Spatial variations in non-structural carbohydrates in stems of twelve temperate tree species. Trees- Structure and Function, 28(1), 77–89. https://doi.org/10.1007/s00468-013-0931-8
Zhang, Y., Xu, J., Zhao, X., & Wang, D. (2012). Analysis of influencing factors of air quality in Luoyang city based on grey relation. Journal of Henan University of Science and Technology: Natural Science, 33, 100–104. [in Chinese].
Zhu, W., Li, N., Huang, Z., Yin, X., Huang, X., Qin, Y., & Zheng, J. (2016). Emission characteristics of thermal power in Guangdong province and influence on atmospheric environment. Research of Environmental Sciences, 29(6), 810–818. [in Chinese].
Acknowledgements
The authors thank Zhongzhou Li (Green Office, Luoyang City Administration Bureau, China) and Yanmei Shen (Department of Afforestation, Luoyang Forestry Bureau, China) for their helpful suggestions, and the students of Henan University of Science and Technology for harvesting and collecting the leaf samples.
Funding
Research grants from the Natural Science Foundation of China (31000265).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by Xiping Zhao, Pingping Guo, and Yongqiang Yang. Data analysis was performed by Haixin Peng. The first draft of the manuscript was written by Xiping Zhao, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, X., Guo, P., Yang, Y. et al. Effects of air pollution on physiological traits of Ligustrum lucidum Ait. leaves in Luoyang, China. Environ Monit Assess 193, 530 (2021). https://doi.org/10.1007/s10661-021-09338-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-021-09338-x