Abstract
This paper describes the prevalence of indicator and pathogenic bacterial groups in water and sediments in OSZ—offshore, ISZ—inshore, IEZ—inner estuary, and UEZ—upper estuary along the river Chapora, central west coast of India, which is influenced by anthropogenic inputs. The abundance of indicator bacterial groups such as total coliforms and Escherichia coli–like organisms in water ranged from non-detectable (ND) to 103 colony-forming units (CFU)/mL. In contrast, their abundance in the sediments was six orders magnitude higher than water (ND to 109 CFU/g). The abundance of potential pathogenic bacteria in water and sediment samples ranged from ND to 103 CFU/mL and from ND to 109 CFU/g respectively, with Shigella-like organisms (SHLO) being the most abundant. In the surface waters, SHLO and Pseudomonas aeruginosa–like organisms (PALO) and in bottom waters, Vibrio parahaemolyticus–like organism and PALO increased progressively from OSZ to UEZ. In contrast, Proteus/Klebsiella-like organisms (PKLO) showed a reverse trend. Amongst all four zones, IEZ was the most contaminated in terms of the higher abundance of indicator and potential pathogenic bacterial populations as corroborated by significantly lower water quality index value. Principal component analysis performed using physico-chemical variables and bacterial groups to reduce data set variability revealed that a different set of parameters contributed differently to the total variation in each zone. Considering the eco-sensitivity of the river Chapora, the results of the present study call for precautionary measures to minimize the degree of anthropogenic inputs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Estuaries are highly productive unique ecosystems and serve as breeding grounds and as a niche for various marine species, euryhaline species (McLusky and Elliott 2004; Nybakken and Bertness 2005), and a wide variety of fish and shellfish species. Estuaries support the coastal economy in the form of fishing, aquaculture, transport, tourism activities, etc. Owing to growing population and exploitation pressure, these ecosystems are increasingly becoming threatened habitats in most parts of the world. The physical, biological, and geochemical attributes of the vulnerable estuarine systems may get altered to a significant extent due to anthropogenic activities (Shenai-Tirodkar et al. 2016a). The importance of the major estuaries, bays, and inshore waters of Goa in terms of productivity and their relevance to fish in different life stages has been well-recognized (Ansari et al. 1995). Previous studies on the water quality and microbial contamination in major riverine (Ganga river, Dash et al. 2010; Mishra et al. 2009; Baghel et al. 2005; Hoogly river, Batabyal et al. 2014; Gola river, Chandra et al. 2006; Cauvery river, Kumarasamy et al. 2009) and bay-estuarine systems (west coast of India, Nagvenkar and Ramaiah 2009; Borade et al. 2015) indicated the prevalence of high concentration of pathogenic bacteria. Endowed with a variety of finfish and shellfish species predominantly in oyster beds (Shenai-Tirodkar et al. 2016b), the Chapora bay-estuarine system of Goa serves an ideal model site for assessing the prevalence of indicator and potential pathogenic bacteria which is impacted by anthropogenic inputs.
The Central Pollution Control Board (CPCB) has classified the stretch of the river Chapora from Morjim to Pernem (~ 32 km) under priority V (biochemical oxygen demand levels range between 3.5 and 5.2 mg/L), and the stretch of the River Chapora between Morjim and Alorna fort falling under saline zone has been classified as SWII (for bathing, contact water sports, and commercial fishing) (RRC 2019). Elevated concentrations of heavy metals (cadmium and copper) in the tissues of edible oysters inhabiting the estuarine stretches of the Chapora have been reported by Shenai-Tirodkar et al. (2016b). Such a trend may be due to anthropogenic activities and is likely to pose potential health risks to humans via consumption of shellfish. In addition to heavy metal contamination, the prevalence of potential pathogenic bacteria in water and sediment associated with anthropogenic inputs could also pose a severe threat to public health and is of paramount importance. Despite of bay-estuarine system of the river Chapora being a significant coastal area for commercial fishing and tourism activities, its vulnerability to anthropogenic inputs and the prevalence of indicator and pathogenic bacterial groups have not been studied. With this background, the present study was undertaken to assess the abundance and types of physiological groups of indicator and potential pathogenic bacteria in water and sediments in different bay-estuarine zones of the river Chapora. The relative importance of physico-chemical parameters such as temperature (Temp), pH, total suspended solids (TSS), turbidity, dissolved oxygen (DO), biochemical oxygen demand (BOD), NO3− and PO43−, and their interrelationship in influencing the water quality have also been addressed. The present results serve as baseline data to gain an understanding of the influence of environmental variables on the prevalence of indicator and pathogenic bacteria in the different zones along the Chapora bay-estuarine system. This study could further aid in initiating environmental protective measures.
Materials and methods
Study area and sampling
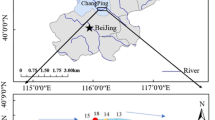
The river Chapora, located between the latitudes (15° 42′ 42.3″ and 15° 35′ 53.1″ N) and longitudes (73° 51′ 31.5″ and 73° 42′ 01.3″ E), is one of the nine major rivers of Goa flowing from the east (Western Ghats) to the west (Arabian Sea) and draining into the Arabian Sea near the Morjim/Chapora beach after traversing a distance of 32 km. The total basin area of the Chapora River is 255 km2 (RRC 2019). Before draining into the Arabian Sea, it forms an integral part of the lifeline in Goa as it supports the agricultural, fishery, and tourism sector. Sampling for the present study was carried out during March 2017 just before the start of the peak of the tourism season. Surface and bottom water samples were collected from 22 stations (C1–C22) encompassing different saline zones (offshore (OSZ) = C1–C7; inshore (ISZ) = C8–C13; inner estuary (IEZ) = C14–C18; upper estuary (UEZ) = C19–C22) covering the stretch of ~ 20 km of the river Chapora (Fig. 1). The depths of the sampled stations ranged between 2 m at UEZ and 17 m at OSZ.
Physico-chemical analysis
At each sampling station, water temperature and salinity were measured using a portable CTD profiler (model Seacat-26, Sea Bird Electronics Inc. USA). A Niskin water sampler was used to collect surface (~ 1 m) and bottom waters from 22 pre-determined stations from the study area. A portable pH meter (Eutech Tutor) was used to measure the pH of the water samples onboard. Turbidity was measured using a portable turbidimeter (Eutech TBDW-1000). Concentrations of total suspended solids (TSS), dissolved oxygen (DO), biochemical oxygen demand (BOD), total phosphorous (TP), and total nitrogen (TN) were measured following standard analytical methods (APHA 1989; Strickland and Parsons 1972).
Bacteriological analysis
For bacteriological analysis, surface (~ 1 m) and bottom water (30 cm above bottom) samples were collected using Niskin water sampler and then transferred to sterile polypropylene bottles. Sediment samples were collected by van Veen grab and transferred into clean zip-lock bags. Immediately after collection, both water and sediment samples were kept in an icebox and transported to the laboratory for further analysis. In the laboratory, for enumeration of total bacterial counts (TBC), 1 mL of water and 1 g of sediment was fixed with buffered formalin (2% final concentration). The fixed sample was then stained with acridine orange stain as a fluorochrome (0.01%) for 5 min, followed by filtration on 0.22-μm nuclepore black polycarbonate filter paper (Millipore). The bacterial counts were enumerated by examining the filter papers using epifluorescence microscopy, according to Hobbie et al. (1977). For enumerating the abundance of indicator bacteria such as total coliforms (TC) and Escherichia coli–like organisms (ECLO), and pathogenic bacteria like Shigella-like organisms (SHLO), Salmonella-like organisms (SLO), Proteus/Klebsiella-like organisms (PKLO), Vibrio-like organism (VLO), V. parahaemolyticus–like organism (VPLO), V. cholera–like organism (VCLO), Pseudomonas aeruginosa–like organism (PALO), and Streptococcus faecalis–like organism (SFLO), 0.1 mL of water sample was directly spread plated onto selective media in triplicate (Hi-media, India) (Table 1). The media plates were incubated at 37 °C for 24–36 h. For estimation of bacterial abundance in sediments, 1 g of sediment sample was serially diluted using 50% sterile seawater to 10−4, 10−5, and 10−6 dilution. From these dilutions, 0.1 mL was spread plated onto selective media in triplicate and then incubated as mentioned above. The final count of colonies in water and sediment was expressed in terms of CFU/mL and CFU/g of dry sediment, respectively (CFU = colony forming units).
Water quality index
The water quality index (WQI) values at the 22 stations along the Chapora bay-estuary were elucidated using the National Sanitation Foundation Water Quality Index (Oram 2010) by incorporating the values of physico-chemical and biological parameters (Temp, pH, DO, BOD, TSS, turbidity, TP, TN, and E. coli counts). Resultant scores of 90–100, 70–90, 50–70, 25–50, and 0–25 correspond to excellent, good, medium, bad, and very bad grades of water quality, respectively.
Statistical analysis
To reveal the main factors that influenced the water quality in the four zones along the Chapora bay-estuary, principal component analysis (PCA) was carried out using PRIMER version 6 (Clarke and Warwick 2001). Prior to analysis, physico-chemical parameters were square-root transformed, and bacterial values were log10 (x + 1) transformed. The variables with a loading of ≥ 0.35 were considered for interpretation of the data.
Results
Physico-chemical parameters
The mean surface temperature of 29.60 ± 0.48 °C was at OSZ and a minimum of 28.17 ± 2.27 °C was recorded at ISZ (Table 2). Generally, the temperature of the bottom water was slightly lower than that of the surface water. The mean pH values ranged from 6.04 ± 1.42 in the bottom waters of ISZ to 7.64 ± 0.18 in the surface waters of UEZ. The values of salinity progressively dropped from OSZ (33.43 ± 3.10) to UEZ (12.95 ± 4.49). The content of DO in surface waters was slightly more than the bottom water at all the zones, ranging from 6.77 ± 0.53 mg/L to 7.53 ± 0.31 mg/L. The BOD values showed an opposite trend with bottom waters having higher values than the surface waters. The highest BOD of 3.55 ± 2.31 mg/L was at the OSZ and lowest of 0.50 ± 0.48 mg/L at the ISZ. The lowest TSS was at UEZ while higher values were recorded at OSZ and ISZ. At IEZ and UEZ, the turbidity values in the bottom waters were almost twice those recorded at the surface. Concentrations of TP did not show any significant variation, while the concentrations of TN recorded at ISZ, IEZ, and UEZ were twice that at OSZ (Table 2).
Total bacterial population
The abundance of the total bacterial population (TBC) was of an order of 106 cells/mL in both surface and bottom waters (Fig. 2). However, in the sediments, the TBC ranged between 2.04 (C5; OSZ) and 297 × 108 cells/g (C17; IEZ) of dry weight. The TBC in sediment samples was two to four orders magnitude higher than that in water (Fig. 2). On an average, the water samples at OSZ (3.87 ± 1.1 × 106 cells/mL) and sediments from IEZ (6.31 ± 1.07 × 109 cells/g) harboured relatively more bacterial abundance compared with the other zones.
Indicator and potential pathogenic bacteria
The abundance of different indicators and potential pathogenic bacterial populations in the water and sediments of the four bay-estuarine zones showed a significant spatial variation (Fig. 3 and Fig. 4). A substantial variation in the abundance of all groups of pathogenic bacteria was discernible (Fig. 4). The abundance of bacterial groups, TC, ECLO, SHLO, SLO, PKLO, VLO, VPLO, VCLO, PALO, and SFLO, ranged from ND to 103 CFU/mL in all water samples. On the other hand, abundances of these bacterial groups in sediments varied from 106 to 109, ND to 108, 109, ND to 105, 107, 106, ND to 108, ND to 108, ND to 107, and ND to 107 CFU/g, respectively.
Abundance of indicator bacterial groups (TC, total coliforms and ECLO, E. coli–like organisms in (a) surface water, (b) bottom water, and (c) sediment at different bay estuarine zones (OSZ, offshore; ISZ, inshore; IEZ, inner estuary; UEZ, upper estuary) of the river Chapora. Vertical bars indicate mean ± standard deviation
Variation (%) in pathogenic bacterial populations at different zones (OSZ, offshore; ISZ, inshore; IEZ, inner estuary; UEZ, upper estuary) in the Chapora bay-estuarine system. (a) Surface water, (b) bottom water, and (c) sediment. SHLO, Shigella-like organism; SLO, Salmonella-like organisms; PKLO, Proteus/Klebsiella-like organisms; VLO, Vibrio-like organisms; VPLO, Vibrio parahaemolyticus–like organisms; VCLO, Vibrio cholera–like organisms; PALO, Pseudomonas aeruginosa–like organisms; SFLO, Streptococcus faecalis–like organisms
The SHLO group was found to be the most dominant in almost all the 4 zones in water and sediment samples ranging from 12% in ISZ bottom waters to 84% in OSZ sediments (Fig. 4). PALO contributed up to 35% and 25% gradually increasing from OSZ to UEZ in surface and bottom waters, respectively. Similarly, SHLO in the surface waters and VPLO in the bottom increased progressively from OSZ to UEZ. PKLO showed an opposite trend in surface water, which contributed from 11% in UEZ to 28% in OSZ. However, in the bottom waters, it showed a fluctuating pattern (10 to 29%). VCLO contributed fairly in both water and sediment of all zones contributing significantly up to 34% in ISZ of sediments. VLO and SLO were almost absent in all the sediment samples; however, a minor fraction of SFLO (up to 9%) and VLO (up to 19%) contributed in all the zones of water and sediment samples. Overall, the mean abundances of indicator and potentially pathogenic bacteria in ISZ and IEZ were noticeably higher compared with the other zones. In the OSZ, the pathogenic abundance was an order lower than the other 3 zones. Surface and bottom waters showed similar patterns of abundance of TC and ECLO. In the OSZ, abundances of TC and ECLO were 1–2 orders magnitude lower compared with the other three zones (Fig. 3). Finally, amongst all four zones, IEZ was the most contaminated in terms of the higher abundance of indicator as well as potential pathogenic bacterial populations.
Water quality index
Indices of water quality determined by using the parameters listed (Table 2) indicated the value of the samples to be in the range of 67–80 indicating the water to be of either ‘medium’ or ‘good’ quality with a water quality trend of UEZ > IEZ > OSZ > ISZ (Table 3). The surface waters exhibited comparatively poor index values than the bottom waters. But individually, when only ECLO counts were taken into consideration, the water quality index at all the stations was in the range of ND to 22 indicating ‘very bad’ quality of the water.
Principal component analysis
Principal component analysis (PCA) was performed to evaluate the probable relationship between the physico-chemical parameters and bacterial groups. Based on eigen coefficients represented by each component in Tables 4 and 5, the total variation was reduced to three components for further consideration. In the surface waters, all the parameters at the OSZ showed an equal role in PC1 while a variation of 23.3% was observed in PC2, showing higher loading for bacterial parameters like ECLO and SFLO in the OSZ (Table 4). On the other hand, in ISZ, TP, VCLO, and SFLO, factors played an important role in the variation of 37.1% in PC1, and PC2 accounted for 29.9% variation with SHLO and VLO. PC2 of the IEZ constituted of 26.4% variance only by SFLO whereas, at the UEZ, 40.9% variation was seen due to DO, and 33.9% variation was contributed by TP and TC. In the bottom waters (Table 5), OSZ showed PC1 accounting for 34.3% variation by TC and VPLO, and PC2 accounted for 23.9% variation due to SLO and VCLO. At the ISZ, DO, pH, and SFLO were the only factors responsible for 19.3% variation in PC2. At the IEZ, PC1 accounted for 40.1% variation with VLO. While at the UEZ, PC2 contributed a 28.9% variation due to SLO and TSS.
Discussion
Microbial analysis of the culturable population of indicator and potential pathogenic bacteria is essential to study the impact of contamination levels in the area. The Chapora bay-estuarine system, considered a model coastal ecosystem, has a dense coastal population and is a popular commercial fishing port besides being a tourist spot on the west coast of India. CPCB has classified the Chapora River under priority V (BOD levels, 3.5–5.2 mg/L). Although recorded concentrations of DO and BOD meet the prescribed statutory requirement, the levels of faecal coliforms (FC) exceed the prescribed limits (RRC 2019). Our study area comprised four zones along the estuarine river stretch right from the mouth of the river Chapora (OSZ) until the upper reaches of the estuary (UEZ) covering a stretch of ~ 20 km. Most of the tourism and recreational activities are concentrated in the ISZ. The luxuriant growth of mangrove vegetation and agricultural lands lies on both the sides of IEZ and UEZ zones. Discharge of untreated domestic sewage mostly near the southern bank area and direct discharge of waste mainly consisting of stormwater drains from individual houses into the river Chapora have been reported (RRC 2019). The DO concentrations are reliant on the variation in chemical, biological, and microbial factors (Yang et al. 2007). In the present study, the level of DO in all stations was within the normal range (Table 2). Similarly, the turbidity values were also within an optimum range. High values of turbidity mostly occur due to the turbulent flow which results in mixing of the silt and sand at the bottom of the river. The mean value of BOD in the present study was 3.55 ± 2.31 mg/L. The values are in accordance with those reported earlier for saline stretches of the river Chapora (RRC 2019). According to EPA (1996), the BOD values of 5 mg/L or more are considered severely polluted. Slightly lower BOD values recorded in the present study highlight the importance of currents in regulating the rate of decomposition (Sanap et al. 2006). The nitrate and phosphate levels were within the standard limits.
In addition to physico-chemical parameters, bacterial activity plays an essential role in mediating the cycling of organic matter. In the present study, the bacterial load in the sediments of the Chapora estuary ranged from 2.04 × 108 to 2.97 × 1010 cells/g. Similar results have been reported from other coastal and estuarine regions of the west coast of India (Ramaiah et al. 2002; Ramaiah et al. 2004; Khandeparker et al. 2015) (Table 6). The sediment samples showed 2 to 4 orders magnitude higher bacterial abundance than water samples suggesting a bacterial affinity for sediment attachment (Liss et al. 1996). The texture of the sediments in the study area was predominantly fine sand with various proportions of silt and clay. The organic matter in the sediment was also moderate and did not vary spatially. The organic matter derived from both anthropogenic and natural sources was medium to high (data not shown). Finer grained sediments rich in organic matter have been reported to harbour rich bacterial load. Furthermore, the residence time of bacteria in water is comparatively lower than in sediments (Mohandass et al. 2000). Furthermore, high availability of nutrients and organic matter along with protection from stressors such as temperature and salinity fluctuation support higher bacterial load in sediments (Craig et al. 2004).
Coliform group of bacteria has widely been used as an indicator for sewage and faecal contamination of water bodies (Tallon et al. 2005). Pathogenic bacteria in waters are usually much lower in numbers and much harder to isolate and identify. Therefore, TC is mostly used as a general indicator of potential contamination. But coliforms are also found in soil and may be transported to water bodies thus resulting in overestimation of their abundance. To overcome this, faecal coliforms could instead be used to assess faecal contamination as these are only present in the intestinal tracts of humans and other animals and thus can be more specific in indicating faecal pathogenic contamination (Snozzi et al. 2001). Faecal coliforms such as E. coli are often used to evaluate recent faecal pollution as they cannot survive for a long time outside the intestines (Jang et al. 2017). Although E. coli may not be pathogenic per se, however, their occurrence suggests the potential risk of co-occurrence of other faecal bacteria, which can be very pathogenic like Salmonella spp. or the hepatitis A virus (Odonkor and Ampofo 2013). It is universally accepted that the high numerical abundance of coliforms in water bodies directly relates to high level of faecal contamination (Fujioka 2002). The acceptable limit for TC and ECLO is ≤ 500 and 100 CFU/100 mL, respectively. In the present study, the TC counts were in the range of 3.75 ± 0.42 to 15.02 ± 0.34 × 104 CFU/100 mL and 4.03 ± 0.44 to 11.78 ± 0.31 × 104 CFU/100 mL in surface and bottom waters, respectively. On the other hand, the ECLO counts ranged from 0.58 ± 0.13 to 8.54 ± 0.19 × 104 CFU/100 mL and 1.13 ± 0.12 to 6.4 ± 0.14 × 104 CFU/100 mL in surface and bottom waters, respectively. These values far exceed the acceptable limits and reflect the degree of anthropogenic inputs into the Chapora bay-estuarine system.
The results of the present study indicate that the water samples of the Chapora bay-estuarine system (Table 7) harboured relatively lower pathogenic bacterial load than adjoining the Mandovi and Zuari estuary, Goa (Nagvenkar and Ramaiah 2009), Hoogly River (Batabyal et al. 2014), River Ganga (Dash et al. 2010; Mishra et al. 2009; Baghel et al. 2005), Mumbai waters (Ramaiah et al. 2002), Gola River (Chandra et al. 2006), and Cauvery River (Kumarasamy et al. 2009) but were higher by 1–2 orders reported from the Veraval coast in Gujarat (Borade et al. 2015). The sediments of the river Chapora harboured 3–5 orders higher pathogenic counts than the Veraval coast (Borade et al. 2015) and the creeks along the Mumbai coast (Ramaiah et al. 2004). Zone-wise, a higher abundance of potential pathogenic bacteria was observed in ISZ and IEZ compared with the OSZ and UEZ. Such a trend could be due to the resuspension of sediment-associated bacteria into the water column under the prevailing pattern of tidal currents as the nearly well-mixed conditions were observed in ISZ and IEZ (Pers Comm. Seelam Jayakumar).
The determined WQI in the present study showed that the waters of the Chapora bay-estuarine system belonged either to ‘medium’ or ‘good’ categories of water quality. However, the water quality slips into category ‘very bad’ when only ECLO counts were considered for estimating the WQI. This implies that although the concentrations of physico-chemical parameters are within acceptable limits, the actual threat of faecal contamination is often masked. Lowest values of WQI recorded in ISZ reflect the poor water quality due to increased anthropogenic activities. Similarly, PCA also indicated that ISZ was most affected by the pathogenic counts at a loading of ≥ 0.35 in the surface waters (Table 4).
In the present study, the counts for pathogenic groups were 2 to 4 orders magnitude higher in sediment samples than in water samples. Pathogenic groups such as ECLO, VCLO, VPLO, SHLO, SLO, PALO, and SFLO are the main cause of waterborne diseases (diarrhoea, dehydration, cholera, enteric fever, dysentery, Vibrio illness, food poisoning, enteritis, enterocolitis, etc.). In the present study, water samples from all zones showed low number (up to 103 orders) of potential pathogenic bacteria. However, in the sediment samples, higher counts of potential pathogenic bacteria (up to 109) were observed with SHLO dominant in all four zones. Similar findings have also been reported by Alm et al. (2003) and Luna et al. (2010) who postulated that the sediments could act as a source for faecal and pathogenic origin of microorganism and virulent strain of bacteria. This suggests that the sediments of the Chapora estuary serve as a reservoir for potential pathogenic bacteria.
Conclusion
Presence of high level of pathogenic bacteria in water bodies and aquatic sediments is a serious concern for human health as sediments can undergo resuspension due to natural (tidal currents) and anthropogenic disturbance. The present study on the Chapora bay estuary reveals that the sediments have a high load of pathogenic bacteria which may be due to anthropogenic inputs in the form of untreated/partially treated sewage, faecal contamination, and a large amount of domestic waste in the water from the surrounding human settlement as observed during physical survey (RRC 2019). The water quality index reveals the estuarine system to be ‘good’ at most stations and ‘medium’ at few stations; however, the actual situation of the high bacterial load is often masked. Furthermore, the revaluation of WQI by incorporating only ECLO counts significantly dropped the grading of water quality to ‘poor’ at all stations. Hence, it is essential to monitor the individual factors involved in the WQI as it can under/overestimate the actual finding. The present study showed the dominance of SHLO amongst those from the other groups, namely SLO, PKLO, VLO, VPLO, VCLO, PALO, and SFLO, in the sediments indicating their probable role in disease during sediment resuspension. The zones ISZ and IEZ were relatively more polluted compared with the OSZ and UEZ due to the lower abundance of indicator and potentially pathogenic bacteria in the latter zones. The coastal area in proximity with the former zones harboured a dense human population as compared with that of the latter. The comparison of the above findings will help in decision making while selecting safer zones for the promotion of tourism and fishing activities. Also, the baseline data and inference from this study can prompt environmental measures needed for the restoration of the coastal-estuarine ecosystem and its resources, thus maintaining an environmentally friendly ecosystem to support a healthy and economically viable coastal population.
Data availability
Not applicable
References
Alm, E. W., Burke, J., & Spain, A. (2003). Fecal indicator bacteria are abundant in wet sand at freshwater beaches. Water Research, 37(16), 3978–3982. https://doi.org/10.1016/S0043-1354(03)00301-4.
Ansari, Z. A., Chatterji, A., Ingole, B. S., Sreepada, R. A., Rivonkar, C. U., & Parulekar, A. H. (1995). Community structure and seasonal variation of an inshore demersal fish community at Goa, west coast of India. Estuarine, Coastal and Shelf Science, 41(5), 593–610. https://doi.org/10.1016/0272-7714(95)90029-2.
APHA. (1989). Standard methods for the examination of water and wastewater (17th ed.). Washington, D.C: American Public Health Association.
Baghel, V. S., Gopal, K., Dwivedi, S., & Tripathi, R. D. (2005). Bacterial indicators of faecal contamination of the Gangetic river system right at its source. Ecological Indicators, 5(1), 49–56. https://doi.org/10.1016/j.ecolind.2004.09.002.
Batabyal, P., Einsporn, M. H., Mookerjee, S., Palit, A., Neogi, S. B., Nair, G. B., & Lara, R. J. (2014). Influence of hydrologic and anthropogenic factors on the abundance variability of enteropathogens in the Ganges estuary, a cholera endemic region. Science of the Total Environment, 472, 154–161. https://doi.org/10.1016/j.scitotenv.2013.10.093.
Borade, S., Dhawde, R., Maloo, A., Gajbhiye, S. N., Ram, A., & Dastager, S. G. (2015). Assessment of enteric bacterial indicators and correlation with physico-chemical parameters in Veraval coast, India. Indian Journal of Geo-Marine Sciences, 44(4), 501–507 http://nopr.niscair.res.in/handle/123456789/34727.
Chandra, R., Singh, S., & Raj, A. (2006). Seasonal bacteriological analysis of Gola river water contaminated with pulp paper mill waste in Uttaranchal, India. Environmental Monitoring and Assessment, 118(1–3), 393–406. https://doi.org/10.1007/s10661-006-1508-4.
Clarke, K. R., & Warwick, R. M. (2001). Change in marine communities: an approach to statistical analysis and interpretation (2nd ed.). Plymouth: PRIMER-E.
Craig, D. L., Fallowfield, H. J., & Cromar, N. J. (2004). Use of microcosms to determine persistence of Escherichia coli in recreational coastal water and sediment and validation with in situ measurements. Journal of Applied Microbiology, 96(5), 922–930. https://doi.org/10.1111/j.1365-2672.2004.02243.x.
Dash, R. R., Prakash, E. B., Kumar, P., Mehrotra, I., Sandhu, C., & Grischek, T. (2010). River bank filtration in Haridwar, India: removal of turbidity, organics and bacteria. Hydrogeology Journal, 18(4), 973–983. https://doi.org/10.1007/s10040-010-0574-4.
EPA (Environmental Protection Agency) (1996). Quality criteria for waters, Washington, DC.
Fujioka, R. (2002). Microbial indicators of marine recreational water quality. In C. J. Hurst, R. L. Crawford, G. Knudsen, M. J. McIneney, & L. D. Stetzenbach (Eds.), Manual of environmental microbiology (2nd ed., pp. 234–243). Washington DC: American Society for Microbiology Press.
Hobbie, J. E., Daley, R. J., & Jasper, S. (1977). Use of nuclepore filters for counting bacteria by fluorescence microscopy. Applied Environmental Microbiology, 33(5), 1225–1228.
Jang, J., Hur, H. G., Sadowsky, M. J., Byappanahalli, M. N., Yan, T., & Ishii, S. (2017). Environmental Escherichia coli: ecology and public health implications-a review. Journal of Applied Microbiology, 123(3), 570–581. https://doi.org/10.1111/jam.13468.
Khandeparker, L., Anil, A. C., Naik, S. D., & Gaonkar, C. C. (2015). Daily variations in pathogenic bacterial populations in a monsoon influenced tropical environment. Marine Pollution Bulletin, 96(1–2), 337–343. https://doi.org/10.1016/j.marpolbul.2015.04.051.
Kumarasamy, P., Vignesh, S., Arthur James, R., Muthukumar, K., & Rajendran, A. (2009). Enumeration and identification of pathogenic pollution indicators in Cauvery, South India. Research Journal of Microbiology, 4(12), 540–549. https://doi.org/10.3923/jm.2009.540.549.
Liss, S. N., Droppo, I. G., Flannigan, D. T., & Leppard, G. G. (1996). Floc architecture in wastewater and natural riverine systems. Environmental Science & Technology, 30(2), 680–686. https://doi.org/10.1021/es950426r.
Luna, G. M., Vignaroli, C., Rinaldi, C., Pusceddu, A., Nicoletti, L., Gabellini, M., Danovaro, R., & Biavasco, F. (2010). Extraintestinal Escherichia coli carrying virulence genes in coastal marine sediments. Applied Environmental Microbiology, 76(17), 5659–5668. https://doi.org/10.1128/AEM.03138-09.
McLusky, D. S., & Elliott, M. (2004). The estuarine ecosystem: ecology, threats and management (3rd ed.). Oxford: Oxford University Press 216 pp.
Mishra, A., Mukherjee, A., & Tripathi, B. D. (2009). Seasonal and temporal variations in physico-chemical and bacteriological characteristics of River Ganga in Varanasi. International Journal of Environmental Research, 3(3), 395–402. https://doi.org/10.12944/CWE.2.2.08.
Mohandass, C., Nair, S., Achuthankutty, C. T., & Bharathi, P. A. (2000). Pollution monitoring of coastal and estuarine areas: I. Bacterial indicators along the south Gujarat coast. Indian Journal of Marine Sciences, 29, 43–47.
Nagvenkar, G. S., & Ramaiah, N. (2009). Abundance of sewage-pollution indicator and human pathogenic bacteria in a tropical estuarine complex. Environmental Monitoring and Assessment, 155(1–4), 245–256. https://doi.org/10.1007/s10661-008-0432-1.
Nybakken, J. W., & Bertness, M. D. (2005). Estuaries and salt marshes. In J. W. Nybakken & M. D. Bertness (Eds.), Marine biology: an ecological approach (Vol. 6, pp. 1–579). San Francisco: Pearson/Benjamin Cummings.
Odonkor, S. T., & Ampofo, J. K. (2013). Escherichia coli as an indicator of bacteriological quality of water: an overview. Microbiology Research, 4, 2. https://doi.org/10.4081/mr.2013.e2.
Oram, B. P. G. (2010). The water quality index. Water research Center. https://www.water-research.net/waterqualityindex/waterqualityindex.html Accessed 15 September 2018.
Ramaiah, N., Kenkre, V. D., & Verlecar, X. N. (2002). Marine environmental pollution stress detection through direct viable counts of bacteria. Water Research, 36(9), 2383–2393. https://doi.org/10.1016/s0043-1354(01)00435-3.
Ramaiah, N., Kolhe, V., & Sadhasivan, A. (2004). Abundance of pollution indicator and pathogenic bacteria in Mumbai waters. Current Science, 87(4), 435–439.
RRC. (2019). River Rejuvenation Committee (RRC), Goa. River Rejuvenation Action Plan-Chapora River. The River Rejuvenation Committee Government of Goa (India), 33 pp.
Sanap, R. R., Mohite, A. K., Pingle, S. D., & Gunale, V. R. (2006). Evaluation of water qualities of Godawari River with reference to physico-chemical parameters, Dist. Nasik (MS), India. Pollution Research, 25(4), 775.
Shenai-Tirodkar, P. S., Gauns, M. U., & Ansari, Z. A. (2016a). Evaluation of surface water and sediment quality in Chicalim Bay, Nerul Creek, and Chapora Bay from Goa coast, India– a statistical approach. Environmental Monitoring and Assessment, 188(8), 472. https://doi.org/10.1007/s10661-016-5445-6.
Shenai-Tirodkar, P. S., Gauns, M. U., & Ansari, Z. A. (2016b). Concentrations of heavy metals in commercially important oysters from Goa, central-west coast of India. Bulletin of Environmental Contamination and Toxicology, 97(6), 813–819. https://doi.org/10.1007/s00128-016-1956-7.
Snozzi, M., Ashbolt, N. J., & Grabow, W. O. K. (2001). Indicators of microbial water quality. In L. Fewtrell & J. Bartram (Eds.), Water quality guidelines, standards and health: assessment of risk and risk management for water-related infectious disease (pp. 289–316). London: IWA Publishing.
Strickland, J. D., & Parsons, T. R. (1972). A practical handbook of seawater analysis, Vol. 167, (2nd ed., pp. 310). Ottawa: Bulletin of the Fisheries Research Board of Canada.
Tallon, P., Magajna, B., Lofranco, C., & Leung, K. T. (2005). Microbial indicators of faecal contamination in water: a current perspective. Water, Air, and Soil Pollution, 166(1–4), 139–166. https://doi.org/10.1007/s11270-005-7905-4.
Yang, H. J., Shen, Z. M., Zhang, J. P., & Wang, W. H. (2007). Water quality characteristics along the course of the Huangpu River (China). Journal of Environmental Sciences, 19(10), 1193–1198. https://doi.org/10.1016/s1001-0742(07)60195-8.
Acknowledgements
Authors express their gratitude to the Director of the CSIR-National Institute of Oceanography (CSIR-NIO), Goa, for the research and analytical facilities provided for carrying out this work. This is CSIR-NIO contribution No.__6544__.
Code availability
Not applicable
Author information
Authors and Affiliations
Contributions
1) Nitisha Sangodkar—Conceptualization, Formal analysis, software analysis and writing original draft.
2) Maria Judith Gonsalves—conceptualization, supervision, review and editing.
3) Yogini Shanbhag—Formal analysis and writing original draft.
4) R.A. Sreepada—Conceptualization, supervision, review and editing.
5) Delcy Nazareth— Formal analysis and software analysis.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Consent to participate
Not applicable
Ethics approval
Not applicable
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sangodkar, N., Gonsalves, M.J., Shanbhag, Y. et al. Prevalence of indicator and potential pathogenic bacterial groups in the Chapora bay-estuarine system, Goa, central west coast of India. Environ Monit Assess 192, 397 (2020). https://doi.org/10.1007/s10661-020-08368-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-020-08368-1