Abstract
The ability to recover to original states after disturbances makes macroinvertebrates useful tools for assessing the impacts of pesticides. Many studies showed that direct exposure to pesticides decreases macroinvertebrate richness and alters their composition. The main objective of this study was to assess recovery patterns in macroinvertebrate communities after pesticide application in irrigated rice fields. We analyzed short-term temporal dynamics of macroinvertebrate communities after application of the herbicides bispyribac-sodium and clomazone and the insecticide chlorantraniliprole, over the rice-growing season in southern Brazil. We selected three conventional rice fields and the recovery of macroinvertebrate communities was also compared with three adjacent natural ponds. The study was developed from November 2011 to February 2012 (rice-growing season). Five macroinvertebrate collections were carried out 3, 7, 14, 38, and 60 days after pesticide application (November 25). Rice fields showed lower richness and abundance than ponds in the period immediately after pesticide application, and recovery rates in the richness of macroinvertebrate communities were more conspicuous as pesticide residuals dissipated from the fields. Macroinvertebrate community structure in rice fields also became more similar to natural ponds as pesticide traces were scarcer. However, macroinvertebrate abundance patterns were not related to pesticide concentrations in the fields. Our results supported the general hypothesis on the negative effects of pesticide application on macroinvertebrate community in irrigated rice fields, although other environmental features (e.g., length of the flooded period) also contributed to explain temporal dynamics in the macroinvertebrate communities from irrigated rice fields.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pesticides are a significant component of modern rice technology, and contamination by pesticides is one of the main environmental problems caused by rice production (Barrigossi et al. 2004; Parsons et al. 2010). Pesticides are primarily used to prevent, control, or eliminate pests (target organisms), with little consideration to the effects on nontarget ones. The indiscriminate use of these agrochemicals indirectly affects nontarget organisms (Bambaradeniya and Amerasinghe 2003; Parsons et al. 2010). Nontarget organisms are, however, involved in the maintenance of soil fertility and nutrient cycling and compete with parasites of rice pests and vectors of human and animal diseases, and the maintenance of biodiversity within rice systems is essential for their agronomic sustainability (Bambaradeniya and Amerasinghe 2003; Schmidt et al. 2015).
Brazil is the largest consumer market of pesticides worldwide since the last 10 years, with a 190% growth in pesticide use over the last decade (Albuquerque et al. 2016; Rigotto et al. 2014). Brazil is also the largest rice producer in the Americas, and the state of Rio Grande do Sul accounts for up to 60% of the rice production, almost entirely in the irrigated system (CONAB 2015; IRGA 2013). The high rice production has been largely associated with the use of pesticides and chemical fertilizers (Parsons et al. 2010).
Rice fields have been recognized worldwide as having considerable conservation value for many aquatic species, including plants, invertebrates, and vertebrates (Katayama et al. 2015; Lupi et al. 2013; Maltchik et al. 2011, 2017; Rolon and Maltchik 2010; Stenert et al. 2012). Irrigated rice fields are important habitats for the development of aquatic macroinvertebrates (Stenert et al. 2012). However, the macroinvertebrate community is the most vulnerable group to pesticide application in rice fields, particularly the benthic organisms (Lim 2005). Many studies showed that the use of pesticides is a major contributor to the reduction of macroinvertebrate richness in these agroecosystems (Bambaradeniya and Amerasinghe 2003; Mesléard et al. 2005; Wilson et al. 2008; Rizo-Patrón et al. 2013).
Nonlethal effects of pesticides include changes in development, reproduction, and behavior of macroinvertebrates, as well as alterations in their ecological sensitivity (Rizo-Patrón et al. 2013). Punctual effects of pesticides on particular invertebrate populations were also investigated. Morphological deformities in Chironomidae larvae have been suggested as indicators of exposure to pesticides (Gagliardi and Pettigrove 2013). Madden et al. (1992) showed a positive relationship between the concentration of the insecticide DDT and the percentage of deformed mouthparts in Chironomidae larvae. Furthermore, pesticide application may also alter the sex ratios of chironomids in rice fields, probably due to differences between males and females in predation vulnerability or pesticide sensitivity (Takamura 1996). Other studies reported the acute toxicity of pesticides to certain aquatic invertebrate species (Rossaro and Cortesi 2013; Suárez-Serrano et al. 2010).
On the other hand, no negative effects of the pesticides on macroinvertebrate richness and density were observed in rice fields of southern Brazil, considering the application of a single dose in the recommended period (Wandscheer et al. 2017). Accordingly, Dalzochio et al. (2016a) showed that pesticide-free (organic) and pesticide-treated (conventional) rice fields had similar macroinvertebrate richness and abundance in southern Brazil. However, some functional traits of aquatic insects were associated to the pesticide-free rice fields, like the presence of genera sensitive to pesticides, and more active and predatory insects, when compared to pesticide-treated rice fields (Dalzochio et al. 2016b). Heckman (2005) showed that predatory taxa, such as Odonata and Heteroptera, were the most abundant aquatic insects in the pesticide-free rice fields of Southeast Asia.
The ability to recover to original states after disturbances makes macroinvertebrates useful tools for assessing impacts of pesticides, and this group has thus been extensively used for environmental monitoring in rice fields (Rosenberg and Resh 1993; Leitão et al. 2007; Wilson et al. 2008; Melo et al. 2015; Rizo-Patrón et al. 2013). Due to connectivity with natural systems, the effect of pesticides on macroinvertebrates has been analyzed in combination with water chemistry in streams, wetlands, and rice ditches (Brock et al. 2009, 2010; Melo et al. 2015). However, short-term responses of macroinvertebrate communities to application of pesticides have not been compared between rice fields and natural wetlands over the rice-growing season.
The main objective of this study was to assess patterns in the recovery of macroinvertebrate communities after pesticide application in irrigated rice fields over the rice-growing season. We examined variation in richness, abundance, and community structure after application of the herbicides bispyribac-sodium and clomazone and the insecticide chlorantraniliprole and compared macroinvertebrate recovery in rice fields with communities from natural wetlands.
Materials and methods
Study area
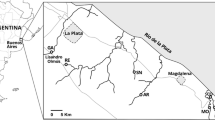
The study area is located in Capivari do Sul municipality (30°08′49″ S–50°30′48″ W), in the Coastal Plain region of Rio Grande do Sul state, extreme southern Brazil (Fig. 1). The climate in the region is humid subtropical, with annual average temperature of ~ 17.5 °C and average annual precipitation of ~ 1250 mm (Rambo 2000). Wetlands and rice fields are common systems in the Coastal Plain (Maltchik et al. 2003). The rice-growing area in Capivari do Sul municipality was ~ 16,000 ha (IRGA 2013).
Sampling design
The rice cultivation cycle in southern Brazil comprises the following phases: fallow phase, soil preparation, rice-growing season, and post-harvest. The fallow phase represents the period when the agricultural land remains without rice culture, and the soil preparation represents the period when the soil is prepared for planting. In the soil preparation and post-harvest periods, the rice fields are drained.
The study was developed during the rice-growing season in southern Brazil (from November 2011 to February 2012). We randomly selected a subset of three conventional rice fields and three intermittent natural ponds from a larger set of 60 rice fields and 12 natural ponds located in the studied farm. Natural ponds with similar characteristics to the rice fields (area of approximately 1 ha, water depth of ~ 40 cm, and intermittent hydrological regime) corresponded to the larger set of ponds. Rice fields were ~ 600 m equidistant and ponds were ~ 1 km distant from the fields (Fig. 1) to minimize spatial autocorrelation. Rice fields were irrigated at the beginning of rice emergence, and the surface water was present only in the growing season (during 3 months). Natural ponds had a reduction in their surface flooding area greater than 70% of the original area in December 2011, which coincided with summer. These natural systems showed an initial recovery in their surface flooding area by January 2012.
The pesticides used in rice fields were the commercial herbicides bispyribac-sodium (Nominee–SC400) and clomazone (Gamit Star-EC500) and the insecticide chlorantraniliprole (Altacor-WG350). Pesticide application was carried out at the beginning of rice emergence, and the doses used were 1 L/ha of clomazone, 100 mL/ha of bispyribac-sodium, and 30 g/ha of chlorantraniliprole. The herbicides and insecticides are classified as highly and moderately toxic to aquatic organisms, respectively (product manufacturer information).
Sampling methods
Five macroinvertebrate collections were carried out 3, 7, 14, 38, and 60 days after pesticide application (November 25, 2011). One water sample was collected (1 L) from each sampling site for the analysis of pesticide concentration at the Laboratory of Organic Compounds and Metal Analysis of the Federal University Foundation of Rio Grande (FURG). The following water physicochemical variables were measured in situ with a water quality meter (HORIBA U-50): water temperature (WT), pH, total dissolved solids (TDS), electrical conductivity (EC), dissolved oxygen (DO), and water turbidity (NTU).
Five macroinvertebrate samples were taken from each sampling site with an aquatic frame dip-net (30 in cm width; 250 μm in mesh size). Each sample was represented by a 1-m sweep, posteriorly fixed in situ with 10% formaldehyde. In the laboratory, samples were sieved and macroinvertebrates were collected under a stereomicroscope. Individuals were classified to family or genus level, according to specialized bibliography (Fernández and Domínguez 2009). Macroinvertebrates were preserved in 80% alcohol and archived in the reference collection of the Laboratory of Ecology and Conservation of Aquatic Ecosystems of UNISINOS.
Data analysis
Macroinvertebrate communities in rice fields and ponds
Variation in macroinvertebrate richness and abundance between rice fields and ponds over the study period was tested using generalized linear mixed-effects models (GLMM). Fixed effects were environment type (rice field and pond) and days after pesticide application (continuous). Sampling site was included as a random factor in all models to control for pseudoreplication due to repeated sampling. Tests for overdispersion and zero-inflation effects were conducted to corroborate model assumptions and improve accuracy, as the outcome variables (richness and abundance) were count data. Models were then built with negative binomial distribution, log link function, and Laplace approximation procedures. All possible combinations of fixed effects were tested and compared against null models (intercept only). Posteriorly, model selection was performed using the conditional Akaike information criterion (AICc) to control for small sample sizes (Burnham and Anderson 2002). Only models with delta AIC < 2.0 were kept for further inference. Estimated parameters and their respective 95% confidence intervals were used to predict richness and abundance across the study period. Trends in richness and abundance over the study period were fitted with LOESS method.
Variation in the aquatic macroinvertebrate community structure between rice fields and ponds over the cultivation period was assessed with a nonmetric multidimensional scaling analysis (NMDS) ordination diagram. Significance of differences in community structure between environment type and days after pesticide application was tested with a nonparametric multivariate analysis of variance followed by 9999 permutations using sampling site as blocking factor (two-way randomized-block PERMANOVA). Both analyses were based on Bray-Curtis distance matrices generated from a log-transformed matrix of the fauna and computed using the vegan package (Oksanen et al. 2016).
Relationships between pesticides and water physicochemical variables
We summarized variation in pesticide concentration in rice fields and water physicochemical variables in both rice fields and ponds over the study period with principal component analysis (PCA) by correlation. We selected the axes that represented a significant portion of the explained variance in each dataset according to the Kaiser-Guttman criterion. In the pesticide dataset, only the first axis was retained, while in the water physicochemical variable datasets in both rice fields and ponds, the two first axes were retained.
The relationship between pesticide concentration and variation in water physicochemical variables was assessed through co-inertia analysis (Dray et al. 2003). This symmetrical approach computes eigenvectors that describe the common structure of two or several datasets. We used the selected PCA axes that summarized variation in each dataset (pesticide and water physicochemical matrices). After checking that the row weights of the axes of both datasets are similar, co-inertia analysis retained the canonical axes that maximized the co-structure of the datasets. The significance of the relationship between datasets (RV coefficient) was tested by 999 permutations. Co-inertia analysis was computed using package ade4 (Dray and Dufour 2007). As no significant relationship was detected (RV = 0.22; P value = 0.11), the influence of pesticides and water physicochemical variables on macroinvertebrate communities in rice fields was separately assessed.
Influence of pesticides and water physicochemical variables on macroinvertebrate communities
The influence of pesticides on the richness and abundance of macroinvertebrate communities in rice fields was tested with GLMM with negative binomial distribution. The selected PCA axes of the pesticide dataset were used as predictors, and sampling site as random factor. Model assumptions and inference were assessed using the same procedures previously described in the first item of this subsection.
The influence of water physicochemical variables on macroinvertebrate richness and abundance was assessed using the same approach. Analyses were separately conducted for rice field and pond datasets, using the selected PCA axes of each environmental dataset as predictors. The goodness-of-fit of the GLMM for rice field analyses (pesticide and water physicochemical datasets) was improved with zero-inflated models and for analysis of richness in ponds with Gaussian distribution. GLMM were constructed using the lme4 and glmmADMB packages (Bates et al. 2015; Skaug et al. 2016).
The influence of water physicochemical variables (DO, EC, NTU, pH, TDS, and WT) on macroinvertebrate community structure in rice fields and ponds was assessed by fitting the water physicochemical variables’ dataset to the previously generated NMDS ordination diagrams of the fauna of each environment (envfit function of vegan package; 999 permutations). All analyses were computed in the statistical environment R version 3.3.2 (Oksanen et al. 2016).
Results
Relationships between pesticides and water physicochemical variables
Concentrations of pesticides showed a clear temporal trend over the study period. Concentrations sharply decreased from the 3rd to 14th day after application, and residual concentrations were present until the end of the study (60th day; Fig. 2). The first PCA axis of the pesticide concentration summarized 94% of the dataset and it was positively related to all pesticides. The PCA axes of the water physicochemical variables of rice fields explained 73% of the variation in the dataset. The first component summarized the temporal variation in the variables, and it was positively related to electrical conductivity, total dissolved solids, dissolved oxygen, and water temperature. The PCA of the pond water physicochemical variables summarized 71% of the dataset and it was positively related to DO (Table 1).
Macroinvertebrate communities in rice fields and wetlands
We collected 3682 individuals from 30 macroinvertebrate taxa in rice fields (Supplementary Material 1). Abundance ranged from 969 to 1512 individuals and richness from 7 to 10 taxa per rice field over the cultivation period (Supplementary Material 1). Insecta was the predominant group (61.3% of the individuals collected from 28 taxa). Chironomidae and Oligochaeta were the most frequent taxa, represented 33.5 and 26.8% of the individuals, respectively (Supplementary Material 1).
In ponds, 6149 individuals from 38 taxa were collected (Supplementary Material 1). Abundance ranged from 759 to 3974 individuals and richness from 7 to 17 taxa per pond over the cultivation period. Aquatic insects were also the most abundant macroinvertebrates in natural ponds (42.3% of the individuals from 30 taxa), corresponding to sampled. Dogielinotidae and Glossiphoniidae were the most abundant frequent taxa in natural ponds (21.7 and 18.7% of the individuals, respectively; Supplementary Material 1).
The NMDS ordination diagrams displayed a clear segregation in the composition of aquatic macroinvertebrate communities between rice fields and ponds, as well as among samples from first and ultimate days after application of pesticides (Fig. 3a). Indeed, significant differences between environment type and days after application of pesticides were detected by PERMANOVA (environment type, pseudo-F(1,17) = 4.47; R2 = 0.13; P value = 0.01; days after application, pseudo-F(4,17) = 1.97; R2 = 0.23; P value = 0.002). The interaction between environment type and days was not significant (P value = 0.2). Macroinvertebrate composition was more similar between rice fields and natural ponds in the late period of the study (Fig. 3a). While some macroinvertebrate taxa were more associated with natural ponds, (such as Corbiculidae, Hydrobiidae, and Pomacea), other were more associated to rice fields (Oligochaeta, Glossiphoniidae, and Tipulidae) (Fig. 3b).
NMDS ordination plot of aquatic macroinvertebrate communities in rice fields and wetlands over the study period. Samples are identified according to days after pesticide application. a Overall ordination (R, rice field samples; W, wetland samples). b Ordination with taxa scores (codes for the taxa are listed in Supplementary Material 1)
Influence of pesticides and water physicochemical variables on macroinvertebrate communities
Model selection showed that there was only one top model for richness and abundance of macroinvertebrate communities. Estimated coefficients in the best model revealed that environment type, number of days after pesticide application, and their interaction were useful to explain variations in the richness and abundance of macroinvertebrates (Table 2). At the beginning of the study period, richness and abundance were higher in ponds than in rice fields, and then, similar values of richness and abundance were found in both environments at the end of the study period (Fig. 4a). Richness and abundance increased over the study period (Figs. 4a, b).
The influence of pesticide concentration in rice fields was detected only on the richness of aquatic macroinvertebrate communities. The best-fit model included the effects of variation in concentration summarized by PCA. However, no clear effect of pesticide was detected on macroinvertebrate abundance in rice fields. The model with the intercept alone was more efficient in explaining variation rather than days and pesticides (Table 3).
Best-fit models for the influence of water physicochemical variables on macroinvertebrate communities in rice fields showed that richness rather than abundance was significantly explained by the water dataset (Table 4). In ponds, both richness and abundance of macroinvertebrate communities were best explained by water physicochemical variables (Table 4).
Different water physicochemical variables were related to macroinvertebrate communities in each environment. Electrical conductivity, total dissolved solids, and water turbidity were significantly associated with variation of macroinvertebrate communities in rice fields (P value < 0.05; Fig. 5a). In turn, dissolved oxygen was the only variable associated with macroinvertebrate communities in ponds (P value < 0.05; Fig. 5b).
Discussion
Rice fields are man-made ecosystems with high biodiversity of aquatic macroinvertebrates (Rizo-Patrón et al. 2013; Stenert et al. 2012). Temporal succession of aquatic communities over the rice-growing period has been analyzed by several researchers (Dalzochio et al. 2016a, b; Leitão et al. 2007; Bambaradeniya and Amerasinghe 2003; Suhling et al. 2000). Our study evidenced short-term temporal dynamics (i.e., 60 days) of macroinvertebrate communities after pesticide application in irrigated rice fields. The temporal patterns found in rice fields were related to the period immediately posterior to pesticide application. Our main findings include significant trends of recovery in richness and increased similarity in macroinvertebrate community structure with natural ponds associated with reductions in pesticide concentration in rice fields.
Macroinvertebrate richness varied between ponds and rice fields and over the study period. This relationship was more conspicuous when interaction between both environments was included. Richness was lower in rice fields than in ponds at the beginning of the growing season, immediately after pesticide application in rice fields. In addition, macroinvertebrate richness was influenced by pesticide concentration in the fields. These results corroborate previous results that indicate negative effects of pesticides on macroinvertebrate richness in rice fields.
Pesticides negatively impacted macroinvertebrate community, and lower macroinvertebrate richness was observed in rice fields under more intensive use of pesticides (Rizo-Patrón et al. 2013; Wilson et al. 2008). However, many studies take into consideration experiments developed over the whole growing season rather than short-term responses. Short-term responses of macroinvertebrate to pesticide application are not consensual across the literature (Baumart and Santos 2010; Mesléard et al. 2005). Our result was similar to those observed by Wilson et al. (2008), who detected lower richness at the beginning of the growing season in pesticide-treated rice fields. The negative impacts of pesticides are generally related to their concentration (Brock et al. 2010). Schulz and Liess (1999) showed that pesticide toxicity to aquatic organisms decreases over the cultivation cycle. Baumart and Santos (2010) and Baumart et al. (2011) reported that some macroinvertebrate taxa recolonized rice fields at the end of the crop cycle. In our study, pesticide concentration decreased to residual levels over the growing season, which coincided with higher macroinvertebrate richness. An increase in macroinvertebrate richness in rice fields was also recorded in the absence of pesticide application in European rice fields (Leitão et al. 2007).
Macroinvertebrate abundance varied over the study period in rice fields and ponds, though similar patterns in both environments were not observed. However, macroinvertebrate abundance in rice fields was not related to pesticide concentration. A recent study has reported that pesticide application did not affect neither density nor richness of macroinvertebrates in rice fields of southern Brazil, considering the application of a single dose in the recommended period (Wandscheer et al. 2017). However, negative effects of pesticides on the abundance of the benthic fauna after pesticide use were recurrently reported in rice fields. In general, communities tended to recover over the cultivation period (Dalzochio et al. 2016a, b; Baumart and Santos 2010; Brock et al. 2010; Mesléard et al. 2005). Gagneten (2002) observed that herbicide effect on the benthic community may be associated with toxic effects on feeding resources in the community. Changes in trophic structure of communities led to differential responses of some taxa to pesticide application (Dalzochio et al. 2016b; Baumart and Santos 2010; Wilson et al. 2008; Leitão et al. 2007; Mesléard et al. 2005). Thus, the increase in overall abundance found in our study might have not reflected the individual responses of each macroinvertebrate taxa to pesticide application.
One factor that probably best explained the abundance patterns in both environments is the role of habitat permanence on aquatic macroinvertebrate communities. Abundance of invertebrates in lentic sites generally increases with water permanence, as more species can complete their life cycles (Tarr et al. 2005). The length of the flooded period has special importance in structuring rice field macroinvertebrates (Lupi et al. 2013; Suhling et al. 2000). In our study, abundance continuously increased in rice fields over the study period, i.e., it increased with permanence. Ponds, in turn, showed a distinct pattern. Abundance decreased between the second and third sampling period, probably affected by a natural hydric stress. However, the macroinvertebrate community showed a rapid recovery when the surface water re-flooded.
Recovery in macroinvertebrate community structure in rice fields was also evident, as shown by PERMANOVA and ordination diagrams. Rice field communities sampled at the beginning of the cultivation period (after pesticide application) were different from those in the late period. The decrease in the composition dissimilarity between ponds and rice fields was associated with the decrease in the concentrations of pesticides applied on rice fields. Changes in the community structure of macroinvertebrates following pesticide use have been recorded in the literature (Baumart and Santos 2010; Wilson et al. 2008; Leitão et al. 2007). Moreira et al. (2004) described how differently each pesticide acts on the biota, including the selectivity process of each pesticide. Many studies showed a reduction of more sensitive organisms and a prevalence of generalist and tolerant individuals in rice fields affected by pesticides (Dalzochio et al. 2016b; Rizo-Patrón et al. 2013). In the study rice fields, Chironomidae and Oligochaeta were the predominant taxa, and Oligochaeta and Glossiphoniidae were related to the early periods of the cultivation. Chironomidae and many Annelida were frequently assigned as pollutant-resistant taxa and found in rice fields subject to pesticides (Molozzi et al. 2006; Mesléard et al. 2005; Suhling et al. 2000). These taxa are often the first macroinvertebrates to colonize the most varied aquatic ecosystems and display great dispersal ability and fast development, which guarantee habitat colonization and establishment in rice fields (Bambaradeniya and Amerasinghe 2003).
Pesticide concentration did not affect water physicochemical variables in rice fields. The influence of herbicides and insecticides on water variables is not always straightforward, because pesticides may have had their action hindered by adverse environmental conditions and management practices (Baumart and Santos 2010; Molozzi et al. 2007). For all pesticides analyzed, although residual concentrations were present over the study, they were much reduced and probably ineffective to influence water physicochemical variables.
Differences in the hydrological characteristics between rice fields and ponds during the study period probably accounted for the influence of distinct water physicochemical variables on the fauna of both environments. In rice fields, electrical conductivity and water turbidity were more related to the early periods of cultivation, a common pattern in rice fields (Baumart and Santos 2010; Leitão et al. 2007; Molozzi et al. 2006). The hydric stress to which ponds were subject might have influenced the water physicochemical variables, as hydroperiod is generally correlated to changes in water chemistry (Wissinger et al. 1999). Dissolved oxygen, for instance, was significantly related to the fauna in ponds during at the beginning of the study period, when invertebrate abundance was lower. Intermittent sites are generally subject to oxygen saturation during drought, which is a significant constraint to invertebrate fauna (Batzer and Wissinger 1996).
Conclusions
Short-term responses of macroinvertebrate communities to application of the herbicides bispyribac-sodium and clomazone and the insecticide chlorantraniliprole corroborated previous results that show that pesticides negatively affected macroinvertebrate community in irrigated rice fields. Recovery rates in the richness of macroinvertebrate communities were more conspicuous as pesticide residuals dissipated from the rice fields. Macroinvertebrate community structure in rice fields also became more similar to natural ponds as pesticide traces were scarcer, probably due to differential sensitivity of macroinvertebrate taxa to pesticides.
However, changes in the macroinvertebrate community structure over the study period should be not attributed only to the direct and indirect effects of pesticides, as macroinvertebrate abundance was not related to pesticide concentration in rice fields. Other environmental features beside pesticide application also affected macroinvertebrate communities over time, for instance, natural droughts, management practices (e.g., length of flooded period), and the natural colonization process since richness and abundance increased over the study period in both ponds and rice fields. Moreover, the negative effects of agrochemicals on macroinvertebrate community should also be included in future ecotoxicological studies. These features hindered extrapolations regarding cause-effect relationship between pesticide contamination and their effects on natural adjacent aquatic systems.
References
Albuquerque, A. F., Ribeiro, J. S., Kummrow, F., Nogueira, A. J. A., Montagner, C. C., & Umbuzeiro, G. A. (2016). Pesticides in Brazilian freshwaters: a critical review. Environmental Science: Processes & Impacts, 18(7), 779–787. https://doi.org/10.1039/c6em00268d.
Bambaradeniya, C. N. B., & Amerasinghe, F. P. (2003). Biodiversity associated with the rice field agroecosystem in Asian countries: a brief review. Columbus: International Water Management Institute.
Barrigossi, J. A. F., Lanna, A. C., & Ferreira, E. (2004). Agrotóxicos no cultivo do arroz no Brasil: análise do consumo e medidas para reduzir o impacto ambiental negativo. Circular Técnica 67. Santo Antônio de Goiás: Embrapa Arroz e Feijão. Available online at <https://www.agencia.cnptia.embrapa.br/Repositorio/circ_67_000fyufbxtc02wx5ok076raloqwxcbwj.pdf> Accessed on June 2016.
Bates, D., Maechler, M., Bolker, B., & Walker, S. (2015). Fitting linear mixed-effects models using lme4. Journal of Statistical Software, 67, 1–48.
Batzer, D. P., & Wissinger, S. A. (1996). Ecology of insect communities in nontidal wetlands. Annual Review of Entomology, 41(1), 75–100. https://doi.org/10.1146/annurev.en.41.010196.000451.
Baumart, J. S., & Santos, S. (2010). The impact of herbicides on benthic organisms in flooded rice fields in southern Brazil. In A. Kortekamp (Ed.), Herbicides and environment (pp. 369–382). Rijeka: InTech.
Baumart, J., Dalosto, M., & Santos, S. (2011). Effects of carbofuran and metsulfuron-methyl on the benthic macroinvertebrate community in flooded ricefields. Acta Limnologica Brasiliensia, 3, 138–144.
Brock, T. C. M., Roessink, I., Belgers, J. D. M., Bransen, F., & Maund, S. J. (2009). Impact of a benzoyl urea insecticide on aquatic macroinvertebrates in ditch mesocosms with and without non-sprayed sections. Environmental Toxicology and Chemistry, 28(10), 2191–2205. https://doi.org/10.1897/09-010.1.
Brock, T. C. M., Belgers, J. D. M., Roessink, I., Cuppen, J. G. M., & Maund, S. J. (2010). Macroinvertebrate responses to insecticide application between sprayed and adjacent nonsprayed ditch sections of different sizes. Environmental Toxicology and Chemistry, 29(9), 1994–2008. https://doi.org/10.1002/etc.238.
Burnham, K. P., & Anderson, D. R. (2002). Model selection and multimodel inference: a practical information-theoretic approach. New York: Springer.
CONAB (Companhia Nacional de Abastecimento). (2015). A Cultura do Arroz. Available online at <http://www.conab.gov.br> Accessed on October 2016.
Dalzochio, M. S., Baldin, R., Stenert, C., & Maltchik, L. (2016a). Can organic and conventional agricultural systems affect wetland macroinvertebrate taxa in rice fields? Basic and Applied Ecology, 17(3), 220–229. https://doi.org/10.1016/j.baae.2015.10.009.
Dalzochio, M. S., Baldin, R., Stenert, C., & Maltchik, L. (2016b). How does the management of rice in natural ponds alter aquatic insect community functional structure? Marine and Freshwater Research, 67(11), 1644–1654. https://doi.org/10.1071/MF14246.
Dray, S., & Dufour, A. B. (2007). The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software, 22, 1–20.
Dray, S., Chessel, D., & Thioulouse, J. (2003). Co-inertia analysis and the linking of ecological data tables. Ecology, 84(11), 3078–3089. https://doi.org/10.1890/03-0178.
Fernández, H. R., & Domínguez, E. (2009). Guide for the determination of the benthic arthropods of South America. Tucumán: Fundación Miguel Lillo.
Gagliardi, B., & Pettigrove, V. (2013). Removal of intensive agriculture from the landscape improves aquatic ecosystem health. Agriculture, Ecosystems and Environment, 176, 1–8. https://doi.org/10.1016/j.agee.2013.05.020.
Gagneten, A. M. (2002). Efectos del herbicida paraquat sobre el zooplancton. Iheringia Série Zoologia, 92(3), 47–56. https://doi.org/10.1590/S0073-47212002000300005.
Heckman, C. W. (2005). The evolution from marsh to rice field. In C. H. Fernando, F. Göltenboth, & J. Margraf (Eds.), Aquatic ecology of rice fields (pp. 1–69). Ontario: Volumes Publishing.
IRGA (Instituto Rio Grandense do Arroz). (2013). Acompanhamento da Semeadura do Arroz Irrigado no Rio Grande do Sul – Safra 2012/2013. Available online at <http://www.irga.rs.gov.br> Accessed on October 2016.
Katayama, N., Osawa, T., Amano, T., & Kusumoto, Y. (2015). Are both agricultural intensification and farmland abandonment threats to biodiversity? A test with bird communities in paddy-dominated landscapes. Agriculture, Ecosystems & Environment, 214, 21–30. https://doi.org/10.1016/j.agee.2015.08.014.
Leitão, S., Pinto, P., Pereira, T., & Brito, M. F. (2007). Spatial and temporal variability of macroinvertebrate communities in two farmed Mediterranean rice fields. Aquatic Ecology, 41(3), 373–386. https://doi.org/10.1007/s10452-007-9082-6.
Lim, R. P. (2005). Impact of pesticides on the aquatic biota in ricefields. In C. H. Fernando, F. Göltenboth, & J. Margraf (Eds.), Aquatic ecology of rice fields (pp. 435–462). Ontario: Volumes Publishing.
Lupi, D., Rocco, A., & Rossaro, B. (2013). Benthic macroinvertebrates in Italian rice fields. Journal of Limnology, 72, 184–200.
Madden, C., Suter, P., Nicholson, B., & Austin, A. (1992). Deformities in chironomid larvae as indicators of pollution (pesticide) stress. Netherlands Journal of Aquatic Ecology, 26(2-4), 551–557. https://doi.org/10.1007/BF02255289.
Maltchik, L., Costa, E. S., Becker, C. G., & Oliveira, A. E. (2003). Inventory of wetlands of Rio Grande do Sul (Brazil). Pesquisas Botânica, 53, 89–100.
Maltchik, L., Rolon, A. S., Stenert, C., Machado, I. F., & Rocha, O. (2011). Can rice field channels contribute to biodiversity conservation in Southern Brazilian wetlands? Revista de Biología Tropical, 59(4), 1895–1914.
Maltchik, L., Stenert, C., & Batzer, D. P. (2017). Can rice field management practices contribute to the conservation of species from natural wetlands? Lessons from Brazil. Basic and Applied Ecology, 18, 50–56. https://doi.org/10.1016/j.baae.2016.10.002.
Melo, S., Stenert, C., Dalzochio, M. S., & Maltchik, L. (2015). Development of a multimetric index based on aquatic macroinvertebrate communities to assess water quality of rice fields in southern Brazil. Hydrobiologia, 742(1), 1–14. https://doi.org/10.1007/s10750-014-1957-7.
Mesléard, F., Garnero, S., Beck, N., & Rosecchi, E. (2005). Uselessness and indirect negative effects of an insecticide on rice field invertebrates. Comptes Rendus Biologies, 328(10-11), 955–962. https://doi.org/10.1016/j.crvi.2005.09.003.
Molozzi, J., Pinheiro, A., & Silva, M. R. (2006). Qualidade da água em diferentes estádios de desenvolvimento do arroz irrigado. Pesquisa Agropecuária Brasileira, 41, 139–1398.
Molozzi, J., Hepp, L. U., & Dias, A. S. (2007). Influence of rice crop on the benthic community in Itajaí Valley (Santa Catarina, Brazil). Acta Limnologica Brasiliensia, 19, 383–392.
Moreira, M. R. S., Mucci, J. L. N., & Abakerli, R. B. (2004). Monitoramento dos resíduos de Carbofuran em área de produção de arroz irrigado, Taubaté, São Paulo. Arquivos do Instituto Biológico, 71, 221–226.
Oksanen, J. F., Blanchet, F. G., Friendly, M., Kindt, R., Legendre, P., McGlinn, D., Minchin, P. R., O’Hara, R. B., Simpson, G. L., Solymos, P., Henry, M., Stevens, H., Szoecs, E., & Wagner, H. (2016). Vegan: community ecology package. Available online at <https://cran.r-project.org/package=vegan> Accessed on December 2016.
Parsons, K. C., Mineau, P., & Renfrew, R. B. (2010). Effects of pesticide use in rice fields on birds. Waterbirds, 33(sp1), 193–218. https://doi.org/10.1675/063.033.s115.
Rambo, B. (2000). A fisionomia do Rio Grande do Sul: Ensaio de Monografia Natural. São Leopoldo: Unisinos.
Rigotto, R. M., Vasconcelos, D. P., & Rocha, M. M. (2014). Pesticide use in Brazil and problems for public health. Cadernos de Saúde Pública, 30(7), 1360–1362. https://doi.org/10.1590/0102-311XPE020714.
Rizo-Patrón, F. V., Kumarb, A., Colton, M. B. C., Springer, M., & Trama, F. A. (2013). Macroinvertebrate communities as bioindicators of water quality in conventional and organic irrigated rice fields in Guanacaste, Costa Rica. Ecological Indicators, 29, 68–78. https://doi.org/10.1016/j.ecolind.2012.12.013.
Rolon, A. S., & Maltchik, L. (2010). Does flooding of rice fields after cultivation contribute to wetland plant conservation in southern Brazil? Applied Vegetation Science, 13(1), 26–35. https://doi.org/10.1111/j.1654-109X.2009.01046.x.
Rosenberg, D., & Resh, V. (1993). Freshwater biomonitoring and benthic macroinvertebrates. New York: Chapman & Hall.
Rossaro, B., & Cortesi, P. (2013). The effects of tricyclazole treatment on aquatic macroinvertebrates in the field and in laboratory. Journal of Entomological and Acarological Research, 45(e23), 128–136.
Schmidt, A., Auge, H., Brandl, R., Heong, K. L., Hotes, S., Settele, J., Villareal, S., & Schädler, M. (2015). Small-scale variability in the contribution of invertebrates to litter decomposition in tropical rice fields. Basic and Applied Ecology, 16(8), 674–680. https://doi.org/10.1016/j.baae.2015.01.006.
Schulz, R., & Liess, M. (1999). Validity and ecological relevance of an active in situ bioassay using Gammarus pulex and Limnephilus lunatus. Environmental Toxicology and Chemistry, 18(10), 2243–2250. https://doi.org/10.1002/etc.5620181018.
Skaug, H., Fournier, D., Bolker, B., Magnusson, A., & Nielsen, A. (2016). Generalized Linear Mixed Models using ‘AD Model Builder’. R package version 0.8.3.3. Available online at <http://glmmadmb.r-forge.r-project.org> Accessed on December 2016.
Stenert, C., Maltchik, L., & Rocha, O. (2012). Diversity of aquatic invertebrates in rice fields in southern Brazil. Neotropical Biology and Conservation, 7, 67–77.
Suárez-Serrano, A., Ibáñez, C., Lacorte, S., & Barata, C. (2010). Ecotoxicological effects of rice field waters on selected planktonic species: comparison between conventional and organic farming. Ecotoxicology, 19(8), 1523–1535. https://doi.org/10.1007/s10646-010-0537-5.
Suhling, F., Befeld, S., Hausler, M., Katzur, K., Lepkojus, S., & Mesleard, F. (2000). Effects of pesticide applications on macroinvertebrate density and biomass in ricefields in Rhone-delta, France. Hydrobiologia, 431(1), 69–79. https://doi.org/10.1023/A:1004006422334.
Takamura, K. (1996). Changes in sex ratios of chironomid imagines from ricefield waters. Archiv für Hydrobiologie, 135, 413–421.
Tarr, T. L., Baber, M. J., & Babbit, K. J. (2005). Macroinvertebrate community structure across a wetland hydroperiod gradient in southern New Hampshire, USA. Wetlands Ecology and Management, 13(3), 321–334. https://doi.org/10.1007/s11273-004-7525-6.
Wandscheer, A. C. D., Marchesan, E., Santos, S., Zanella, R., Silva, M. F., Londero, G. P., & Donato, G. (2017). Richness and density of aquatic benthic macroinvertebrates after exposure to fungicides and insecticides in rice paddy fields. Anais da Academia Brasileira de Ciências, 89(1), 355–369. https://doi.org/10.1590/0001-3765201720160574.
Wilson, A. L., Watts, R. J., & Stevens, M. M. (2008). Effects of different management regimes on aquatic macroinvertebrate diversity in Australian rice fields. Ecological Research, 23(3), 565–572. https://doi.org/10.1007/s11284-007-0410-z.
Wissinger, S. A., Whiteman, H. H., Sparks, G. B., Rouse, G. L., & Brown, W. S. (1999). Foraging trade-offs along a predator-permanence gradient in subalpine wetlands. Ecology, 80, 2102–2116.
Acknowledgements
We would like to thanks Dr. Claudio Mario Mundstock and Graziela Scherer from IRGA for helpful comments in the beginning of research. We are grateful to all private land owners that authorized the use of their rice fields for our research. We declare that the data collection complied with Brazilian current environmental laws (Sistema de Autorização e Informação em Biodiversidade-SISBIO number 24882-2).
Funding
This research was supported by funds from UNISINOS (number 02.00.023/00-0), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; grant number 473123-2010-0), and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS, PqG 02/2011). LM and CS hold Research Productivity Grants from CNPq. ICMF held a scholarship from Instituto Riograndense do Arroz (IRGA). MMP and DSK held a scholarship from FAPERGS/CAPES. NK was supported by a JSPS KAKENHI grant (number 25830154).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 118 kb)
Rights and permissions
About this article
Cite this article
Stenert, C., de Mello, Í.C.M.F., Pires, M.M. et al. Responses of macroinvertebrate communities to pesticide application in irrigated rice fields. Environ Monit Assess 190, 74 (2018). https://doi.org/10.1007/s10661-017-6425-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-6425-1