Abstract
The objective of the present study was to monitor the levels of organochlorine pesticides HCB; α-, β-, γ-HCH; pp′DDE; op′DDT; and pp′DDT in blood serum of Veracruz, Mexico inhabitants. Organochlorine pesticides were analyzed in 150 blood serum samples that constituted that which remained after clinical analyses, using gas chromatography–electron-capture detection (GC-ECD). The results were expressed as milligrams per kilogram on fat basis and micrograms per liter on wet weight. Only the following pesticides were detected: p,p′-DDE was the major organochlorine component, detected in 100% of samples at mean 15.8 mg/kg and 8.4 μg/L; p,p′-DDT was presented in 41.3.% of monitored samples at mean 3.1 mg/kg and 1.4 μg/L; β-HCH was found in 48.6% of the samples at mean 4.9 mg/kg and 2.7 μg/L; op′DDT was determined to be in only 3.3% of monitored samples at mean 2.7 mg/kg and 1.4 μg/L. The pooled samples divided according to sex showed significant differences of β-HCH and pp′DDE concentrations in females. The samples grouped according to age presented the third tertile as more contaminated in both sexes, indicating age as a positively associated factor with serum organochlorine pesticide levels in Veracruz inhabitants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The widespread use of pesticides causes inevitable human exposure to their residues through different routes. Organochlorines are a group of pesticides that have provided great benefits in the eradication of various pest diseases in agriculture and in combating the propagation of vectors transmitting mortal diseases for humans. Used for these purposes in Mexico until 1999, DDT (1,1,1-trichloro-2,2-bis(4-chlorophenyl) ethane) and HCH (hexachlorocyclohexane) have been sprayed extensively in public health programs to combat the spread of disease-transmitting vectors. These pesticides are characterized by chemical stability, a lipophilic nature and a propensity to bioaccumulate in the environment and food chain. Through their persistence long after application and susceptibility to long-range transport routes, they remain in the environment as ubiquitous contaminants. Pesticides that have low or intermediate vapor pressure volatilize from the ground and surface soils, exposing humans that have not lived in contaminated areas or manipulated these compounds (Alegria et al. 2008; Wong et al. 2008). DDT, banned in developed countries, is still used today in many countries where malaria is a public health problem, such as in Africa, Asia, and Latin America (WHO 2006). Consistent with this usage history, populations of these countries generally should have higher tissue residue levels than in Europe and the USA (Perry et al. 2005). Nevertheless, inhabitants of areas where DDT was used in the past to combat malaria continue to be exposed to the vapors caused by their current volatilization.
One of the most important obstacles that hamper the routine application of human body monitoring on a large scale is the obvious need to collect human samples, often invasively. Blood has the undeniable advantage that it is in contact with all tissues and is in equilibrium with organs' fats and fatty tissues. Therefore, it has been used extensively to achieve various research and survey goals.
Due to its high lipid affinity, organochlorine pesticides are stored in the fats of tissues of most organs. They enter the circulatory system and are transported via the lipid and protein components of blood serum. The determination of serum levels are commonly used as biomarkers of exposure in epidemiologic studies evaluating the concentrations related to health effects (Sawada et al. 2010; Eskenazi et al. 2009). Except for a positive correlation with age (Patterson et al. 2009; Perry et al. 2005; Herrero-Mercado et al. 2010) and regional variability (Wolff et al. 2007), consistent determinants of serum organochlorine pesticide levels are not well established.
The city of Veracruz and its surroundings are endemic spaces for malaria. To prevent its propagation, these areas were treated with DDT at a rate of 2 g/m2 from 1956 to 1999. The subsequent contamination of the inhabitants is influenced by local environmental pollution, duration of exposure, age, diet, capacity for elimination by metabolism, and number of nursed infants (Brunetto et al. 1996; Czaja et al. 1997; Laden et al. 1999; Smith 1999). Thus, the monitoring of human tissues such as blood, serves as an indicator for understanding the biological specificity in the behavior of organochlorine pesticides in the tropical environment, as well as for assessing their participation in environmental pollution (Laden et al. 1999).
Few reports are available that establish the extent of organochlorine pesticide residue levels in areas where they were applied only for malaria or ectoparasites control. Moreover, there are scarce studies that implicate sanitary actions as a principal source of human exposure. The aim of the study was to determine organochlorine pesticide levels in blood serum of Veracruz inhabitants in order to establish environmental exposure to organochlorine pesticides.
Materials and methods
During 2010, 150 blood serum samples that constituted that which remained after clinical analyses were obtained from clinical analyses laboratories in Veracruz. The serum samples were stored in glass jars, immediately frozen, and kept at −25°C until analyzed.
Gas chromatography was conducted with a Varian model 3400CX (Palo Alto, CA, USA) equipped with a 63Ni electron-capture detector. The operating conditions were as follows: the capillary chromatography column from J&W Scientific (Folsom, CA, USA) was a DB-608 with a 30-m, 0.32-mm inner diameter (i.d.) and 0.83-μm film thickness; the temperature program was 193°C (7 min) to 250°C at 6°C/min, hold for 20 min; the carrier gas was nitrogen at 6.3 mL/min and a 1 μL aliquot was injected in a splitless mode.
Chemical analyses of organochlorine pesticides were performed according to a previously detailed method (Waliszewski and Szymczynski 1991; Waliszewski et al. 2004). A blood serum sample placed in a 25-mL tube with stopper was weighed and acetic acid in a ratio of 1:1 was added. The sample with acetic acid was left for 30 min to hydrolyze and liberate pesticides from complexes with endogenous substances of blood. Thereafter, organochlorine pesticide residues were extracted three times with 10-mL portions of a mixture of hexane and acetone (9:1). The extracts were collected in a 100-mL separatory funnel and washed twice with 25-mL portions of distilled water to remove traces of acetic acid, acetone, and water-soluble substances. The organic phase was collected in a 30-mL tube with stopper, and 2 mL of concentrated sulfuric acid was added. The contents were vigorously shaken for about 1 min and left for 3 min to ensure good phase separation. The hexane phase was passed through a sodium sulfate layer to a 50-mL round-bottomed flask. The sodium sulfate was rinsed several times with hexane. The extract with rinses was rotary evaporated to a few drops. The concentrate was transferred quantitatively with hexane to a 1-mL calibrated tube and the final volume was adjusted to 1.0 mL for qualitative and quantitative determinations with ECD gas chromatography.
All of the samples were analyzed for: HCB; α-, β-, γ-HCH; pp′DDE; pp′DDT; and op′DDT. The minimum detection limits for the organochlorine pesticide residues were as follows: 0.2 mg/kg on fat basis or 0.1 μg/L on wet weight. To determine the quality of the method, a recovery study was performed on ten spiked replicates of blank cow blood samples, which presented contamination levels below the detection limits. The fortification study, done at 2.0 mg/kg on fat basis and 1.0 μg/L on wet weight levels, showed mean recovery values from 85% to 92%. The standard deviation and coefficient of variation were below 10, indicating excellent repeatability of the method. Concentrated sulfuric acid used in the cleanup step degrades the ubiquitous phthalate esters that interfere in the GC-ECD identification of organochlorine pesticides, permitting their accurate determination (Waliszewski and Szymczynski 1990).
Total serum lipids were determined colorimetrically with phosphovanillin according to the method recommended by Hycel de Mexico using a commercial kit for clinical laboratories.
Statistical calculations were conducted using statistical software Minitab version 12. Concentrations of organochlorine pesticide (milligrams per kilogram on fat base and micrograms per liter on wet weight) were expressed as frequencies, arithmetic means (X) ± standard deviations (SD), medians, and geometric means (GM). The resulting concentrations were then used to determine the significance of categorical factors on pesticide levels by the variability among samples, pairing them to identify differences among means by applying the Student's t test between sexes and dividing these groups in tertiles to associate sex and age as a determinant factor of exposure.
Results and discussion
Routine biomonitoring may be desirable for the efficiency valuation of risk management options and efficacy of environmental and health policies. For pesticides, average exposures may not reflect peak exposures arising through infrequent exposure episodes. Repeated sampling of high-exposure subjects provides more insight into the true nature of these episodes and of their toxicological consequences. Invasively collected matrices for human biomonitoring, obtained as the remainder part of clinical analyses, are toxicologically relevant alternatives for many of the biomarkers currently determined. Moreover, a well-informed choice of matrix can provide an added value for human biomonitoring, offering opportunities to study additional aspects of exposure to organochlorine pesticide residues and effects of short- and long-term toxicokinetics, change of exposure over time, or monitoring of selected communities (Smolders et al. 2009).
During the blood serum monitoring study 150 blood serum samples (75 from females and 75 from males) were collected in which only the presence of β-HCH, pp′DDE, op′DDT, and pp′DDT were detected, thus only these pesticides are discussed. The summarized results of organochlorine pesticides levels from a total 150 monitored samples, expressed as frequencies, ranges, mean, and standard deviations of mean (SD), medians, and geometric means (GM) expressed on lipid base (milligrams per kilogram) are presented in Table 1 and on wet weight (micrograms per liter) in Table 2. The age of participants ranged from 2 to 85 years with a mean of 43 years. One hundred percent of analyzed blood serum samples contained pp′DDE at mean concentrations expressed as 15.8 mg/kg on lipid base and 8.4 μg/L wet weights. This compound is followed by β-HCH that had concentrations of 4.9 mg/kg and 2.7 μg/L and was found in 48.6% of the samples. The analysis of pp′DDT and op′DDT indicated lower concentrations. pp′DDT was presented in 41.3% of total blood serum monitored at 3.1 mg/kg and 1.4 μg/L and op′DDT in 3.3% of samples at 2.7 mg/kg and 1.4 μg/L. Calculated pp′DDE/pp′DDT ratios which evaluate the age of exposure, shows high mean value 18.9. This indicates antique exposures and the predominance of pp′DDE as the principal contaminant of Veracruz inhabitants, caused by previous metabolization of the insecticide pp′DDT. Moreover, the values show a decreased trend in concentrations of insecticide pp′DDT in the Veracruz environment. Our pp′DDE/pp′DDT ratio was higher than the values 11.5 and 12.7, calculated by Hansen et al. (2009) in Vietnam produced by lower pp′DDE levels. During the monitoring, we detected 12 persons with extreme contamination: pp′DDE levels that reached 154.2 mg/kg and 65.4 μg/L and pp′DDT levels of 48.2 mg/kg and 18.8 μg/L. These results are explained by the existence in Veracruz of specific points, contaminated with the insecticide DDT and alimentary habits of persons that expose them at a higher rate to organochlorine pesticide residues.
To observe the possible influence of sex as a discriminatory factor for organochlorine pesticide levels, the pooled sample was divided according to sex of donors (75 females and 75 males). The results are presented in Tables 3 and 4 on fat basis and Tables 5 and 6 on wet weight. The calculated mean ages for females (43.3 years) and for males (43.3 years) show no differences between mean ages between sexes (p > 0.05). In order to look for differences in organochlorine pesticide levels between sexes, the samples were paired. The results demonstrate significant differences between sexes for β-HCH mean concentrations (p < 0.05). Other organochlorine pesticide concentrations expressed on fat basis and wet weight indicated no statistical differences (p > 0.05) between sexes, including the pp′DDE/pp′DDT ratios. In general, during the monitoring study, sex was not observed to be a determinant factor for organochlorine pesticide contamination level. This fact expresses the uniform extend of inhabitants' exposure caused only by living in the urban Veracruz environment.
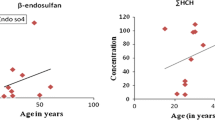
To determine if organochlorine pesticide levels in human blood serum can depend on age of the monitored person, each sex group was further divided according to age into an ordered distribution of three parts, each containing a third of the population and the mean and median tertiles were calculated. The results obtained are presented in Table 7 for females and in Table 8 for males. The t test applied to compute differences among three tertiles of females shows that the mean concentrations of β-HCH levels are not different (p > 0.05). The mean pp′DDE did not exhibit a difference between the first and second tertiles (p > 0.05), whereas between the first and third and second and third, the differences are statistically significant (p < 0.05). op′DDT and pp′DDT did not present differences among means when comparing the three tertiles. op′DDT was detected only in the third tertile that corresponds to older participants. The conclusion of a similar degree of exposure to insecticide pp′DDT can be explained by its ban since 1999 in Veracruz and the stepwise disappearance of the insecticide from the environment. Comparison of the three tertiles revealed that total DDT has different (p < 0.05) mean concentrations, which was influenced by pp′DDE levels. The pp′DDE/pp′DDT ratio increases significantly for the mean from the second to third tertile and this difference is statistically significant (p < 0.05). The results display an increase in organochlorine pesticide concentrations with age (especially pp′DDE) caused by its storage in human adipose tissue over the total life. The pesticide is stored, partitions between adipose fats and blood lipids, and circulates in the whole human body (Herrero-Mercado et al. 2011).
Analyzing the mean differences for all organochlorine pesticide levels among males three tertiles, only statistical differences (p < 0.05) between first and third, and second and third pp′DDE and Σ-DDT tertiles levels were found. β-HCH, op′DDT, and pp′DDT concentrations among three tertiles showed differences that were not statistically significant among concentrations, indicating similar exposure rates for Veracruz male inhabitants. The pp′DDE/pp′DDT ratio did not increase significantly for the mean from the first to second and third tertile (p < 0.05). Moreover, the ratios compared between sexes are higher for the males.
The comparison of pp′DDE and pp′DDT levels between female and male tertiles, grouped according to age, is presented for pp′DDE in Fig. 1 and for pp′DDT in Fig. 2. Applying the t test to compare both sexes, the only significant difference (p < 0.05) for the pp′DDE in the first tertile between female and male was obtained.
Blood concentrations of organochlorine pesticides integrate exposures from all pathways of exposure as well as over time. Hence, with few exceptions, these serum concentrations differentiate neither the route of exposure nor the time period of exposure. Possible exceptions include the ratio of pp′DDE/pp′DDT, which yields information regarding the time since the last DDT exposure. The values are high, indicating residual sources of the pesticide DDT coming from past applications which was previously metabolized to DDE that is more persistent and is stored principally in the body.
Table 9 presents the comparison of organochlorine pesticide levels between monitored females and previously monitored pregnant women (Herrero-Mercado et al. 2010). The comparison reveals statistically significant differences (p < 0.05) between means of pp′DDE and total Σ-DDT concentrations. The comparison, delimited only to the first tertile of monitored females (21 years) and previously monitored pregnant women (26 years), pointed out that there were no statistically significant differences (p > 0.05) among all organochlorine pesticide levels of both groups. The result of no differences can be justified because both populations correspond to younger women who have a shorter time of exposure that does not permit store of organochlorine pesticides at higher levels in adipose tissue.
The measurement of an environmental chemical in a person's blood does not by itself mean that an insecticide causes disease. Advances in analytical methods have allowed the measurement of low levels of environmental chemicals in a person's blood. Studies must also consider other factors, such as duration of exposure expressed in the age of a person (Patterson et al. 2009; Eskenazi et al. 2009; Ibarluzea et al. 2010) that present in the study a close correlation with organochlorine pesticide levels. Thus, obtained results from the monitoring study were computed applying multiple regression analyses. The obtained results (r 2 > 90%) indicate that age has a linear relationship with an increase in serum organochlorine pesticide levels. Our result are supported by similar conclusions from other studies (Dirtu et al. 2006; Lino and da Silveira 2006; Lee et al. 2007). Accumulation of these pesticides in aged people revealed that these pesticides are more slowly eliminated in our environment and are difficult to eliminate from the human body. Once accumulated in adipose tissue they remain in the human body for life. Comparing third tertile between sexes, higher concentrations correspond to female, especially pp′DDE, and that is in agreement with observations of Dirtu et al. (2006) and Koepke et al. (2004) Park et al. (2005) who determined that this difference is caused by different body fat composition between sexes and different alimentary habits.
References
Alegria, H., Wong, F., Jantunen, L. M., Bidleman, T. F., Salvador-Figueroa, M., Gold-Bouchot, G., et al. (2008). Organochlorine pesticides and PCBs in air of southern México (2002–2004). Atmospheric Environment, 42(38), 8810–8818.
Brunetto, R., Leon, A., Burguerra, J. L., & Burguerra, M. (1996). Levels of DDT residues in human milk of Venezuelan women from various rural populations. Science of the Total Environment, 186, 203–207.
Czaja, K., Ludwicki, J. K., Góralczyk, K., & Strucinski, P. (1997). Effect of age and number of deliveries on mean concentration of organochlorine compounds in human breast milk in Poland. Bulletin of Environmental Contamination and Toxicology, 59, 407–413.
Dirtu, A. C., Cernat, R., Dragan, D., Mocanu, R., Van Grieken, R., Neels, H., & Covaci, A. (2006). Organohalogenated pollutants in human serum from Iassy, Romania and their relation with age and gender. Environment International 32(6): 797–803.
Eskenazi, B., Chevrier, J., Goldman Rosas, L., Anderson, H. A., Bornman, M. S., Bouwman, H., et al. (2009). The Pine River Statement: Human health consequences of DDT use. Environmental Health Perspectives, 117(9), 1359–1367.
Hansen, S., Odland, J. O., Phi, D. T., Nieboer, E., & Sandanger, T. M. (2009). Maternal levels of organochlorines in two communities in southern Vietnam. Science of the Total Environment, 408(2), 225–232.
Herrero-Mercado, M., Waliszewski, S. M., Caba, M., Martínez-Valenzuela, C., Gómez Arroyo, S., Villalobos Pietrini, R., et al. (2011). Organochlorine pesticide gradient levels among maternal adipose tissue, maternal blood serum and umbilical blood serum. Bulletin of Environmental Contamination and Toxicology. doi:10.1007/s00128-011-0204-4.
Herrero-Mercado, M., Waliszewski, S. M., Valencia-Quintana, R., Caba, M., Hernández-Chalate, F., García-Aguilar, E., et al. (2010). Organochlorine pesticide levels in adipose tissue of pregnant women in Veracruz, Mexico. Bulletin of Environmental Contamination and Toxicology, 84, 652–656.
Ibarluzea, J., Alvarez-Pedrerol, M., Guxens, M., Marina, L. S., Basterrechea, M., Lertxundi, A., et al. (2010). Sociodemographic, reproductive and dietary predictors of organochlorine compounds levels in pregnant women in Spain. Chemosphere, 19. doi:10.1016/j.physletb.2003.10.071.
Koepke, R., Warner, M., Petreas, M., Cabria, A., Danis, R., Hernandez-Avila, M., et al. (2004). Serum DDT and DDE levels in pregnant women of Chiapas, Mexico. Archives of Environmental Health, 59(11), 559–565.
Laden, F., Neas, L. M., Spiegelman, D., Hankinson, S. E., Willett, W. C., Ireland, K., et al. (1999). Predictors of plasma concentrations of DDE and PCBs in a group of U.S. women. Environmental Health Perspectives, 107, 75–81.
Lee, S. A., Dai, Q., Zheng, W., Gao, Y. T., Blair, A., Tessari, J. D., et al. (2007). Association of serum concentration of organochlorine pesticides with dietary intake and other lifestyle factors among urban Chinese women. Environment International, 32(6), 797–803.
Lino, C. M., & da Silveira, M. I. (2006). Evaluation of organochlorine pesticides in serum from students in Coimbra, Portugal: 1997–2001. Environmental Research, 102(3), 339–351.
Park, M. J., Lee, S. K., Yang, J. Y., Kim, K. W., Lee, S. Y., Lee, W. T., et al. (2005). Distribution of organochlorines and PCB congeners in Korean human tissues. Archives of Pharmacal Research, 28(7), 829–838.
Patterson, D. G., Wong, L. Y., Turner, W. E., Caudill, S. P., Dipietro, E. S., McClure, P. C., et al. (2009). Levels in the U.S. population of those persistent organic pollutants (2003–2004) included in the Stockholm Convention or in other long-range transboundary air pollution agreements. Environmental Science and Technology, 43, 1211–1218.
Perry, M. J., Ouyang, F., Korrick, S., Venners, S. A., Altshul, L., Xu, X., et al. (2005). Body mass index and serum 1,1,1-trichloro-2,2- bis(p-chlorophenyl)ethane in nulliparous Chinese women. Cancer Epidemiology, Biomarkers & Prevention, 14(10), 2433–2438.
Sawada, N., Iwasaki, M., Inoue, M., Itoh, H., Sasazuki, S., Yamaji, T., et al. (2010). Plasma organochlorines and subsequent risk of prostate cancer in Japanese men: A nested case–control study. Environmental Health Perspectives, 118(5), 659–665.
Smith, D. (1999). Worldwide trends in DDT levels in human breast milk. International Journal of Epidemiololgy, 28, 179–188.
Smolders, R., Schramm, K.-W., Nickmilder, M., & Schoeters, G. (2009). Applicability of non-invasively collected matrices for human biomonitoring. Environmental Health, 8, 1–10.
Waliszewski, S. M., Gómez-Arroyo, S., Carvajal, O., Villalobos-Pietrini, R., & Infanzón, R. M. (2004). Uso del ácido sulfúrico en las determinaciones de plaguicidas organoclorados. Revista Internacional de Contaminación Ambiental, 20(4), 185–192.
Waliszewski, S. M., & Szymczynski, G. A. (1990). Determination of phthalate esters in human semen. Andrologia, 22, 69–73.
Waliszewski, S. M., & Szymczynski, G. A. (1991). Persistent organochlorine pesticides in blood serum and whole blood. Bulletin of Environmental Contamination and Toxicology, 46, 803–809.
WHO. (2006). Indoor residual spraying: Use of indoor residual spraying for scaling up global malaria control and elimination: WHO position statement. Geneva: Global Malaria Programme, World Health Organization.
Wolff, M. S., Anderson, H. A., Britton, J. A., & Rothman, N. (2007). Pharmacokinetic variability and modern epidemiology—The example of dichlorodiphenyltrichloroethane, body mass index, and birth cohort. Cancer Epidemiology, Biomarkers & Prevention, 16(10), 1925–1930.
Wong, F., Alegria, H., Jantunen, L. M., Bidleman, T. F., Salvador-Figueroa, M., Gold-Bouchot, G., et al. (2008). Organochlorine pesticides in soil and air of southern Mexico: Chemical profiles and potential for soil emissions. Atmospheric Environment, 42(37), 7737–7745.
Acknowledgments
The authors express their appreciation to Winston Smith of the Peace Corps México for his help in editing this article and we thank the Laboratory of Environmental Engineering from Institute of Enginery, University of Veracruz for analytical support in total lipid determinations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waliszewski, S.M., Caba, M., Herrero-Mercado, M. et al. Organochlorine pesticide residue levels in blood serum of inhabitants from Veracruz, Mexico. Environ Monit Assess 184, 5613–5621 (2012). https://doi.org/10.1007/s10661-011-2366-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-011-2366-2