Abstract
Bisphenol-A (BPA) is a potential endocrine disruptor besides being associated with oxidative damage in several vertebrate classes. In the present study we investigated oxidative effects in erythrocytes and sperm cells as well as spermatic quality in Danio rerio exposed to 14 days at BPA concentrations of 2, 10 and 100 μg/L. Organelles structure, reactive species of oxygen (ROS) and lipoperoxidation (LPO) on erythrocytes and sperm cells were measured by flow cytometry and spermatic parameters were analyzed by the computer-assisted sperm analysis (CASA) system. For both cell types, when compared with control BPA treatment induced a significant increase in ROS and LPO production causing the membrane fluidity disorder, loss of membrane integrity and mitochondrial functionality. Furthermore, it was found a significant increase in DNA fragmentation in erythrocytes of zebrafish BPA exposed. Regarding the spermatic quality, results showed lower sperm motility in animals exposed to BPA, and alterations on velocity parameters of spermatozoa. Thus, the present study concludes that BPA affects the oxidative balance of both cell types, and that can directly affects the reproductive success of the adult Danio rerio. The sensitivity of erythrocytes to oxidative damage induced by BPA was similar to sperm cells, indicating a potential use of blood cells as indicators of oxidative damage present in fish sperm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Bisphenol-A (BPA) is an industrial chemical, which has been widely used in the manufacture of several products, such as polycarbonates, epoxy resins, thermal papers and dental sealants (Bermudez et al. 2010; Riu et al. 2011). It is one of the chemicals with the highest volume of production worldwide, according to estimates will be produced around 8.4 million tons of BPA by 2018 (Gran View Research 2015) and over 100,000 tons of this compound are released annually into the environment (Myridakis et al. 2016). BPA migrates to the environment mainly through processes such as the manufacture of plastics, incomplete removal during treatment of wastewater and leaching of waste discarded of BPA-based materials (Im and Löffler 2016). As a result, BPA has been detected in several environmental matrices such as soil, sediments, groundwater, surface waters, atmosphere and food (Kang et al. 2006; Careghini et al. 2015). In China, BPA was detected in rivers, groundwater and even seawater samples at concentrations up to 16 μg/L (Huang et al. 2012). Some authors defined about 12 µg/L or lower as the environmentally relevant concentration in surface waters (Flint et al. 2012) but in a review of data reported in Europe, Asia and North America, it was revealed that BPA was found in surface water at an average 56 µg/L (Corrales et al. 2015). BPA levels in hazardous waste landfill leachates in Japan were between 1.3 and 17,200 μg/L; mean concentration was 269 μg/L (Yamamoto et al. 2001). Thus, the concentrations used in the present study were chosen according with concentrations of this contaminant found in the environment. Studies have shown that BPA is an endocrine disruptor in several vertebrate classes (Vandenberg et al. 2009; Bhandari et al. 2015) and also is capable of generating oxidative stress by increasing the amount of reactive oxygen species (ROS) in the brain, liver, testis and epididymis and of reducing mitochondrial function in mammals (Nakagawa and Tayama 2000; Bindhumol et al. 2003; Chitra et al. 2003). The mechanism of action of this compound in oxidative damage is not entirely clear, but livers from rats exposed for 30 days to BPA (0.2, 2 and 20 μg/kg/day) demonstrated a decrease in important antioxidant enzymes such as superoxide dismutase-1 (SOD-1) and catalase (CAT) (Bindhumol et al. 2003). In fish, BPA was also related to oxidative stress via reduction of antioxidant defenses, even though further studies of the activity of this compound as a pro-oxidant in these animals are needed (Hulak et al. 2013). In general, studies of fish exposed to BPA at concentrations found in the environment are still scarce; thus, there is little information on the reproductive development and cellular stress at these concentrations in fish. Danio rerio fish has an interesting model in ecotoxicology, since it has been used over the years due to its advantages as a fast reproductive cycle and easy maintenance (Shrader et al. 2003; Coe et al. 2009; Bambino and Chu 2017). Sperm cells are vulnerable to the action of endocrine disrupters, since fish spermatozoa, for example, have limited ability to adjust to physico-chemical changes in their external environment (Lahnsteiner et al. 2004). Similar to mammalian spermatozoa, fish spermatozoa contain high levels of polyunsaturated fatty acids, which are particularly susceptible to ROS-induced lipid peroxidation (Vernet et al. 2004). In mammals the effect of ROS on spermatozoa is well characterized, it may cause lipid peroxidation of spermatozoa membranes, loss of motility and infertility (Sikka 2001). Thus, the oxidative stress in fish is able to cause irreversible reproductive damage, like loss of viable spermatozoa and sperm motility (Li et al. 2010). In this way, studies of sperm parameters and oxidative stress against environmental contamination are necessary, since changes in these parameters can lead to infertility and cause important reproductive problems in affected populations (Schulz et al. 2010). A comparative study of proteomics in fish has shown that, in contrast to mammals, most proteins present in seminal plasma are blood proteins (Dietrich et al. 2014). However, these cells have completely different metabolisms and functions, which can lead to a difference in sensitivity to different contaminants including BPA. The analysis of oxidative parameters in blood cells after exposure to contaminants has been a good biomarker for several species, since they perform key functions such as transport of nutrients and oxygen and immune defense (Shimada and Yamauchi 2004; Çimen 2008; Cocci et al. 2017). In vertebrates, the most abundant cell types in the blood are erythrocytes, which are nucleated cells in fish and most vertebrates, except for mammals. Some studies analyzing erythrocytes from BPA exposure were recently performed with humans and demonstrated that there was an increase in ROS, lipid peroxidation and changes in the main enzymes of the antioxidant defense system (Maćczak et al. 2017a, 2017b). Considering the potential of blood cells as biomarkers of several environmental contaminants, together with the sensitivity of the gonadal cells to endocrine disrupters, they were chosen as a study model to verify the action of BPA (2, 10 and 100 μg/L) on the animals’ redox balance. This study aimed at analyzing comparatively ROS and organelle alterations in erythrocytes and spermatic cells. In addition, knowing that oxidative stress can cause severe reproductive damage, was analyzed the spermatic quality of zebrafish Danio rerio exposed to BPA, so as to generate a discussion about reproductive changes caused by this compound and its impact on the wildlife.
Methodology
Exposure and sampling
Bisphenol-A exposure was performed at the laboratory of Toxicology at the Federal University of Rio Grande FURG (Rio Grande, RS, Brazil). Photoperiodic lighting was 12-h light and 12-h dark, at 26 ± 1 °C and 70% oxygen saturation. The male adults zebrafish were fed twice times a day with commercial feed (Supervit®) ad libitum. For the exposure, the animals (weight of 0.5826 ± 0.1161 g) were randomly divided into four groups respecting the proportion of 1 g animal per liter of water: a control group and three groups at BPA (Sigma-Aldrich Co., St. Louis, MO, USA) concentrations of 2, 10 and 100 μg/L (or 0.008, 0.04 and 0.4 µM). In each group, the fish were arranged in triplicate with 12 animals in each replicate, totaling 36 animals per experimental group. Fish were exposed to BPA for 14 days, in agreement with the guidelines issued by the OECD (OECD 1993), through continuous flow regulated by programmed peristaltic pumps. After two-week exposure, all animals were euthanized according Experimental Animal Ethic Committee (CEUA – FURG—Process number 23116.000355/2016-72). The tail was sectioned posteriorly to the genital orifice, and blood was placed in 500 μL of fetal bovine serum (FBS) and the gonads were also stored for flow cytometer.
Flow cytometer analysis
Attune® Acoustic Focusing Flow Cytometer (Applied Biosystems) was used to detect the cells population. For the erythrocytes and sperm cells, removal cell debris and other cellular types was based on the FSC x SSC scatter plots (Petrunkina et al. 2005; Piehler et al. 2006) and was eliminated by staining cells with Hoechst 33342 at concentration of 16.2 µM (Sigma-Aldrich Co., St. Louis, MO, USA). A total of 20,000 erythrocytes and 10,000 spermatozoa per sample with flow of 200 event/s were analyzed.
Concentration of reactive oxygen species (ROS)
ROS concentration was determined by the fluorescent dye 2’7-dichlorofluorescein diacetate at final concentration of 1.0 µM, which emits green fluorescence when oxidized by intracellular ROS and IP (7.3 µM final concentration). Only the median intensity of green fluorescence of the erythrocytes and living sperm (IP-) was used as measurement (Domínguez-Rebolledo et al. 2011).
Lipoperoxidation (LPO)
Analysis of erytrocytes and spermatozoa lipoperoxidation was evaluated with final concentration of 1 μM of lipid peroxidation sensor Bodipy C11 (Hagedorn et al. 2012) in 100 μL sample. It was incubated for 2 h at room temperature (20 °C). The rate of lipoperoxidation was calculated by the median intensity of green fluorescence (peroxidized lipid)/median green fluorescence intensity + median red fluorescence (non-peroxidized lipid) * 100.
Fluidity of plasma membrane
Plasma membrane fluidity was analyzed by hydrophobic merocyanine 540 dye (M540) at final concentration of 2.7 M (Sigma-Aldrich Co., St. Louis, MO, USA) and YO-PRO, which fluoresces green, at final concentration of 0.1 M (Invitrogen, Eugene, OR, USA). Only live cells (YO-PRO negative) were selected and classified into high fluidity cells (high M540 concentration) and low fluidity cells (low M540 concentration) (Gillan et al. 2005).
DNA fragmentation
Only for the erythrocytes, DNA integrity was assessed by the chromatin structure assay (SCSA). To verify this parameter, 10 μL of FBS blood cells was added to 5 μL of TNE (0.01 M Tris-HCl, 0.15 M NaCl, 0.001 M EDTA, pH 7.2), 10 μL 1X Triton (Triton X-100, 1%) (v/v) at 30-second intervals. The acridine orange dye was added and incubated for 30 s not exceeding 2 min to read at room temperature (22 °C). The erythrocyte cells were classified as whole (green) and fragmented (orange/red) DNA. The DNA fragmentation index was calculated by median red/medium fluorescence green + red fluorescence.
Assessment of sperm motility by computer-assisted semen analysis (CASA)
To analyze sperm motility, semen diluted in Milli-Q water was placed on slides under coverslips and analyzed by CASA (Chyb et al. 2001). Resulting images were reproduced and efficiently and objectively analyzed by the Sperm Class Analyzer (SCA) software to assess overall motility parameters, progressive motility, straight line velocity (VSL), curvilinear velocity (VCL) and VAP (Verstegen et al. 2002). Determination of time of motility after sperm activation was based on the time of complete arrest of the progressive movement of the spermatozoa, in agreement with the method described by (Sorensen 1979). Every image (n = 10) was analyzed using the standard settings for fish by SCA. Sperm was considered immotile when velocity was <10 m/s. Although SCA simultaneously assessed more than 15 sperm motility end points, for brevity only VCL, VSL, and VAP were considered for further analysis, since similar effects were observed for all end points. To determine these velocities, every individual sperm cell (n = at least 1000 sperm) was followed throughout the 10 images and a sperm trajectory was calculated.
Statistical analysis
In this study, descriptive data mean ± SEM were generated for every dependent variable: ROS, LPO, plasma membrane fluidity, DNA fragmentation, total sperm motility and progressive sperm motility. For all dependent variables, normality was tested by the Shapiro–Wilk test. Subsequently, the Kruskal Wallis test for nonparametric data was used because no variable exhibited normal distribution. Statistix® 2009 software was used for the analyses.
Results
Throughout the experiment, there was no mortality of animals, a fact that indicates that BPA concentration was sublethal. Moreover, neither behavioral not feeding abnormalities were observed in the animals. This study found alterations in erythrocytes and sperm cells, especially at concentrations of 10 and 100 μg/L of BPA. A significant increase in the ROS in erythrocytes of the animals exposed to 10 and 100 μg/L (P < 0.05) and in sperm cells only to 10 μg/L (P < 0.05) was observed (Fig. 1a, b). Besides increase of lipoperoxidation was demonstrated at concentrations of 10 and 100 μg/L (P < 0.05) in both cell types, when compared with the control groups (Fig. 2a, b). However, fish erythrocytes exposed to 100 μg/L of BPA had higher levels (P < 0.05) of LPO than those treated with 10 μg/L of BPA. In contrast, the sperm cells presented more LPO at the concentration of 10 μg/L than in 100 μg/L (P < 0.05).
Production of reactive oxygen species (ROS) in erythrocytes (a) and sperm cells (b) of Danio rerio exposed for 14 days to BPA (2, 10 and 100 µg/L) and the respective control groups (0.0 µg/L). The values are means ± SEM (n = 12). The different letters represent significant differences among treatments at the same exposure periods (P < 0.05)
Lipoperoxidation (LPO) in erythrocytes (a) and sperm cells (b) of Danio rerio exposed for 14 days to BPA (2, 10 and 100 µg/L) and the respective control groups (0.0 µg/L). The values are means ± SEM (n = 12). The different letters represent significant differences among treatments at the same exposure periods (P < 0.05)
For the plasma membrane fluidity, both cell types were changed, where there was an increase in fluidity in erythrocytes at concentrations of 10 and 100 µg/L (P < 0.05). Already for the lower concentration in the erythrocytes (2 µg/L), there was a decrease of the plasma membrane fluidity, by comparison with the control group (P < 0.05). For sperm cells, there was an increase in membrane fluidity in relation to the control, but these cells were shown to be less sensitive to this parameter when compared to blood cells (P < 0.05) (Fig. 3a, b). DNA fragmentation analysis was performed only for erythrocytes, and an increase was observed to 10 and 100 μg/L in comparison to the control group (P < 0.05) (Fig. 4).
Membrane fluidity (%) in erythrocytes (a) and sperm cells (b) of Danio rerio exposed for 14 days to BPA (2, 10 and 100 µg/L) and the respective control groups (0.0 µg/L). The values are means ± SEM (n = 12). The different letters represent significant differences among treatments at the same exposure periods (p < 0.05)
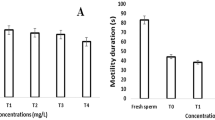
The sperm quality data analyzed by the computer-assisted sperm analysis (CASA) system showed that total motility and progressive motile spermatozoa were decreased at BPA concentrations of 2, 10 and 100 μg/L (P < 0.05) (Fig. 5a, b). The analysis also showed that distance average path (DAP), curved line distance (DCL), straight line distance (DSL), mean velocity (VAP), linearity (LIN), equilibrium (WOB) and flagellar beating frequency (BCF) had significantly higher values at concentrations of 10 and 100 μg/L (P < 0.05) by comparison with the control (Table 1).
Sperm motility total (a) and progressive (b) in Danio rerio exposed for 14 days to BPA (2, 10 and 100 µg/L) and the respective control groups (0.0 µg/L). The values are means ± SEM (n = 12). The different letters represent significant differences among treatments at the same exposure periods (P < 0.05)
Discussion
The constant presence of Bisphenol A (BPA) in different types of samples in humans (Calafat et al. 2008) and its short half-life in the organism indicates that human exposure to BPA is permanent (Huang et al. 2012) Bisphenol A in addition to its endocrine-disrupting chemical (EDC) properties is also a non-persistent organic chemical (NPOC) that disturbs semen quality (see review by Dziewirska et al. 2018). Erythrocytes are considered important biological models for toxicity screening, since they play a key role in the transport of O2 and CO2, essential for respiration and maintenance of nutrient metabolism in fish (Çimen 2008; Farag and Alagawany 2018). Hence, non nucleated red blood cells (mammals) and nucleated red blood cells (non-mammal vertebrates) are used to study the effect of oxidative stress caused by endocrine disruptors and other environmental pollutants (Shimada and Yamauchi 2004; Maćczak et al. 2017a; Cocci et al. 2017). Consequently, carry out research on the effects of chemicals on red blood cells can be an advantageous model because the cells are relatively easy to obtain, can also be used in vitro or in vivo research and are sensitive biomarkers to chemical-induced damage. The chemical used in this study was BPA in three concentrations (2, 10 and 100 μg/L or 0.0087, 0.0438 and 0.438 μM) which we consider to be environmentally relevant. We denominate environmentally relevant concentrations those that are close to the average of the actual concentrations found in the environment (Flint et al. 2012).
In the present study we observed in the red blood cells a significant oxidative stress response and DNA fragmentation. In the same way, we studied the sperm cells of these fish and then were observed a response quite similar to what we had recorded in the red blood cells. Spermatogenesis is a complex process controlled from higher centers, Hypothalamus and Pituitary; and locally by the Sertoli and Leydig cells, among others (Schulz et al. 2010). Considering this complexity can deduce that exogenous chemicals acting in one or more these places can cause spermatogenesis impairment. In fact, several studies have shown that BPA at environmental concentrations caused reproductive toxicity in males of Danio rerio (Chen et al. 2015; Li et al. 2016) as well as similar effects were recorded in other classes of vertebrates and is therefore characterized as a potential EDC (Vandenberg et al. 2009).
It is known that BPA is a chemical capable of generating oxidative stress by reducing antioxidant defenses in various tissues and in different classes of vertebrates (Chitra et al. 2003; Kourouma et al. 2014), which may explain the generation of ROS in the erythrocytes and sperm cells of fish exposed to concentrations of 10 and 100 µg/L of BPA. In aerobic organisms, reactive oxygen species (ROS) are formed as natural products of oxygen metabolism and have important functions in cellular signaling systems (Devasagayam et al. 2004). However, excess ROS can overcome the levels of antioxidant defense and cause damage in macromolecules, impairing cellular homeostasis (Li et al. 2010). The presence of contaminants such as BPA in the cell can trigger oxidative stress directly, through its metabolites, through interaction with transcription factors, among other pathways (Hassan et al. 2012). However, the mechanism of action by which BPA can generate ROS increase is unclear despite its action (in vitro and in vivo) as an oxidizing agent in several studies and in different cell types (Hulak et al. 2013; Hamed and Abdel-Tawwab 2017). In abalone Haliotis diversicolor, BPA was able to alter signaling pathways related to energy metabolism and stress responses, impairing its metamorphosis (Liu et al. 2011). Furthermore, in common carp, for example, similar concentrations of BPA resulted in ROS generation in the liver, as well as disturbances in their immune response (Qiu et al. 2016). Larvae of rare minnow Gobiocypris rarus exposed to BPA for seven days presented inflammatory effects resulting from increased ROS production, accompanied by immunosuppression (Tao et al. 2016). (Michałowicz et al. 2015) demonstrated that exposure to BPA in human blood cells is capable of causing decrease in cell viability due to ROS generation and consequent lipoperoxidation in cells exposed to this chemical. The LPO results observed in our study showed significant oxidative damage in erythrocytes and sperm cells of fish exposed to 10 and 100 μg/L of BPA when compared to the control group. Besides that, in erythrocytes the LPO displayed a significant decrease P < 0.05) in fish exposed to 2 μg/L of BPA. This pattern of response fits into the non-monotonic curve response characteristics that are observed after exposure to various hormones and endocrine disrupters (Vandenberg et al. 2013).
Thus, the increase of lipoperoxidation in exposed fish may be a consequence of the high levels of ROS generated by the presence of the compound. Red blood cells are cells that are relatively vulnerable to lipid peroxidation, which makes them a good biological membrane model to analyze oxidative stress and to lipoperoxidation against different xenobiotics (Farag and Alagawany 2018). Increased levels of lipoperoxidation may be responsible for decrease in plasma membrane integrity, which in turn affects cell permeability and may cause enzymatic inactivation and DNA damage (Domínguez-Rebolledo et al. 2011). In the present study an increase in membrane fluidity in erythrocytes and sperm cells exposed to BPA was found. Changes in membrane fluidity are generally related to changes in lipid composition caused by various types of cell injury such as membrane lipid peroxidation or internal cell damage (Sergent et al. 2009). Besides that, the sperm plasma membrane, as the first barrier, may also be more sensitive to different types of pollutants. This idea was demonstrated by (Harayashiki et al. 2013; Lopes et al. 2014) in Poecilia vivipara and Danio rerio spermatozoa, respectively, treated by different glyphosate concentrations. Finally, several studies have shown that BPA has a genotoxic and mutagenic potential, and may lead to DNA molecule breakdown, DNA adduct formation among other similar effects (Tiwari et al. 2012; Jalal et al. 2018). In the present study, an increase in the DNA fragmentation erythrocytes of the animals exposed to 10 and 100 μg/L of BPA was observed in relation to the control group. The DNA fragmentation caused by exposure to BPA can be explained mainly by the generation of oxidative stress which in turn can damage the DNA molecule, as observed in other studies (Lv et al. 2017; Gao et al. 2018). Moreover, it is known that some BPA metabolites are capable of interacting with the DNA and consequently generate damage that can lead to the breakdown of the molecule (Mokra et al. 2017; Zhao et al. 2018). In vitro studies with acute exposure to low doses of BPA also altered DNA integrity and promoted the generation of ROS in a fish cell line (Hulak et al. 2013). Other DNA damage, such as methylation, was found in rare minnow exposed to relatively low concentrations of BPA (Zhang et al. 2017). Other contaminants, such as endosulfan, cause DNA damage as well as generation of ROS in Danio rerio (Shao et al. 2012). Fragmentation or loss of DNA integrity may induce chromosomal aberrations and mutations, and in the long run cause more severe effects such as apoptosis, cancer and irreversible toxic changes (Rocco et al. 2012). Thus, the results obtained showed that the environmentally relevant concentrations of BPA can generate excess ROS and consequent effects on the plasma membrane and blood and sperm cells, and DNA damage on erythrocytes. From this, parameters related to sperm quality were also evaluated in order to evaluate the possible impact of these alterations on the performance of fish reproductive system. Semen quality is considered a biomarker of early warning to reproductive disorders and within its various parameters, sperm motility is one of the most important characteristics to be evaluated because it is a prerequisite for fertilization (Rurangwa et al. 2004). Findings of this study showed that a decreased of sperm motility, total and progressive, on zebrafish exposed to 10 and 100 µg/L of BPA. When male fathead minnows, Pimephales promelas, were exposed to BPA concentrations of 16 μg/L or higher ones, their gonads significantly reduced the number of mature spermatozoa and increased the number of immature spermatozoa in seminiferous tubules (Sohoni et al. 2001). A study of goldfish, Carassius auratus, also found decrease in motility in sperm exposed to BPA concentrations, similar to the one described by this study after a 90-day exposure period (Hatef et al. 2012). Parameters related to spermatic velocity, straightness, linearity, balance, lateral displacement amplitude and frequency of flagellar beats were significantly higher at concentrations of 10 and 100 μg/L when compared with the control and the lowest BPA concentration (2 μg/L). Others endocrine disruptors, nonylphenol and atrazine, also increased sperm velocity parameters in mammals at environmental concentrations in which this contaminant is considered environmental (Duan et al. 2016; Saalfeld et al. 2018). Under physiological conditions and normal sperm density, fish semen has molecular inhibitors whose roles include regulation of spermatogenesis, removal of immature and damaged sperm and stimulation of sperm velocity (Inaba et al. 1998; Dzyuba and Cosson 2014). Results of increasing sperm velocity in animals exposed to BPA may be due to decrease in the number of viable cells at the 10 and 100 µg/L of BPA (decrease motility -%), and, consequently, dilution of inhibitors found in semen.
Although the evidence demonstrates adverse effects of BPA on sperm quality, their mechanisms are not well understood yet. Sperm maturation in fish, for example, is regulated by cell signaling with increased cAMP and pH (Cosson 2004) and sperm motility depends on the ATP content (Cosson 2010). The reduction in sperm motility after treatment with BPA can be explained in several ways. Most reported effects of BPA on vertebrates may be attributed to its activity as an estrogen receptor agonist, but this compound has also caused ROS generation and cellular oxidative stress (Crain et al. 2007). Sperm motility may be the most sensitive indicator of oxidative stress, since high ROS levels are able to inhibit one or more oxidative phosphorylation and/or glycolysis enzymes, limiting ATP generation (Lamirande and Gagnon 1995). In vitro studies of environmentally relevant BPA concentrations in fish spermatozoa demonstrated decrease in sperm motility and sperm velocity associated with increased ATP levels, oxidative stress and lipoperoxidation (Hulak et al. 2013). Therefore, more biochemical studies are needed, since oxidative stress may influence the sperm quality of these animals and reduce the motility of their spermatozoa. Toxin-induced oxidative stress is the most common cause of damage to sperm (Pasqualotto et al. 2000).
Conclusion
Considering this scenario, our results allow us to conclude that zebrafish Danio rerio, exposed to environmentally relevant BPA concentrations, currently found in the aquatic environment, induced higher ROS amount, as well as structural damage caused by lipoperoxidation in both red blood cells and sperm cells indicating oxidative stress. Still, as a possible consequence of these cellular damages was observed a decrease in sperm quality, which can lead to important effects that compromise their reproductive success. The sensibility of red blood cells and sperm cells was similar, suggesting a potential use of the red blood cells as indicators of oxidative damages present in the sperm of the animal. Although wild fish are more genetically variable than the laboratory strains, it is known that this difference is small and does not invalidate the use of laboratory strains in ecotoxicological studies (Coe et al. 2009; Bambino and Chu 2017). Changes in key aspects of zebrafish sperm physiology from exposure to BPA suggest the possibility of reproductive disturbances which may affect the survival capacity of populations in BPA-contaminated environments. The ubiquity of BPA in different environments raises concern about the risks of preserving different populations multigenerational exposure to this chemical. Some studies have shown that gonadal damage in fish similar to the can lead to decrease in the fertility of the animals and consequent decrease in the population abundance (Baker et al. 2014; Akhter et al. 2018). de Kermoysan et al. (2013) demonstrated that exposure to BPA concentrations, similar of the present study, experiment caused damage to fish Gasterosteus aculeatus gonads as well as an increase in the proportion of immature males in subsequent generations in a chronic mesocosm assay. Thus, the results of present study indicate that BPA in environmentally relevant concentrations has the potential to cause oxidative damage in blood and sperm cells of zebrafish Danio rerio, which could in the long term provide irreversible changes in the fish populations.
References
Akhter A, Rahaman M, Suzuki R-T et al. (2018) Next-generation and further transgenerational effects of bisphenol A on zebrafish reproductive tissues. Heliyon 4:e00788. https://doi.org/10.1016/j.heliyon.2018.e00788
Baker TR, Peterson RE, Heideman W (2014) Using zebrafish as a model system for studying the transgenerational effects of dioxin. Toxicol Sci 138:403–411. https://doi.org/10.1093/toxsci/kfu006
Bambino K, Chu J (2017) Zebrafish in toxicology and environmental health. In: Current topics in developmental biology. pp 331–367. https://doi.org/10.1016/bs.ctdb.2016.10.007
Bermudez DS, Gray LE, Wilson VS (2010) Modeling the interaction of binary and ternary mixtures of estradiol with Bisphenol A and Bisphenol AF in an in vitro estrogen-mediated transcriptional activation assay (T47D-KBluc). Toxicol Sci 116:477–487. https://doi.org/10.1093/toxsci/kfq156
Bhandari RK, Deem SL, Holliday DK et al. (2015) Effects of the environmental estrogenic contaminants bisphenol A and 17α-ethinyl estradiol on sexual development and adult behaviors in aquatic wildlife species. Gen Comp Endocrinol 214:195–219. https://doi.org/10.1016/j.ygcen.2014.09.014
Bindhumol V, Chitra KC, Mathur PP (2003) Bisphenol A induces reactive oxygen species generation in the liver of male rats. Toxicology 188:117–124. https://doi.org/10.1016/S0300-483X(03)00056-8
Calafat AM, Ye X, Wong L-Y et al. (2008) Exposure of the U.S. population to bisphenol A and 4-tertiary-octylphenol: 2003–2004. Environ Health Perspect 116:39–44. https://doi.org/10.1289/ehp.10753
Careghini A, Mastorgio AF, Saponaro S, Sezenna E (2015) Bisphenol A, nonylphenols, benzophenones, and benzotriazoles in soils, groundwater, surface water, sediments, and food: a review. Environ Sci Pollut Res 22:5711–5741. https://doi.org/10.1007/s11356-014-3974-5
Chen J, Xiao Y, Gai Z et al. (2015) Reproductive toxicity of low level bisphenol A exposures in a two-generation zebrafish assay: evidence of male-specific effects. Aquat Toxicol 169:204–214. https://doi.org/10.1016/j.aquatox.2015.10.020
Chitra KC, Latchoumycandane C, Mathur PP (2003) Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 185:119–127
Chyb J, Kime DE, Szczerbik P et al. (2001) Computer-assisted analysis (CASA) of common Carp Cyprinus Carpio l. spermatozoa motility in the presence of cadmium. Arch Pol Fish 9:173–181
Çimen MYB (2008) Free radical metabolism in human erythrocytes. Clin Chim Acta 390:1–11. https://doi.org/10.1016/j.cca.2007.12.025
Cocci P, Capriotti M, Mosconi G, Palermo FA (2017) Effects of endocrine disrupting chemicals on estrogen receptor alpha and heat shock protein 60 gene expression in primary cultures of loggerhead sea turtle (Caretta caretta) erythrocytes. Environ Res 158:616–624. https://doi.org/10.1016/J.ENVRES.2017.07.024
Coe TS, Hamilton PB, Griffiths AM et al. (2009) Genetic variation in strains of zebrafish (Danio rerio) and the implications for ecotoxicology studies. Ecotoxicology 18:144–150. https://doi.org/10.1007/s10646-008-0267-0
Corrales J, Kristofco LA, Steele WB et al. (2015) Global assessment of Bisphenol A in the environment. Dose-Response 13:155932581559830. https://doi.org/10.1177/1559325815598308
Cosson J (2010) Frenetic activation of fish spermatozoa flagella entails short-term motility, portending their precocious decadence. J Fish Biol 76:240–279. https://doi.org/10.1111/j.1095-8649.2009.02504.x
Cosson J (2004) The ionic and osmotic factors controlling motility of fish Spermatozoa. Aquac Int 12:69–85. https://doi.org/10.1023/B:AQUI.0000017189.44263.bc
Crain DA, Eriksen M, Iguchi T et al. (2007) An ecological assessment of Bisphenol-A: evidence from comparative biology Reprod Toxicol 24:225–239. https://doi.org/10.1016/j.reprotox.2007.05.008
de Kermoysan G, Joachim S, Baudoin P et al. (2013) Effects of bisphenol A on different trophic levels in a lotic experimental ecosystem. Aquat Toxicol 144–145:186–198. https://doi.org/10.1016/j.aquatox.2013.09.034
Devasagayam TPA, Tilak JC, Boloor KK et al. (2004) Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India 52:794–804
Dietrich MA, Arnold GJ, Nynca J et al. (2014) Characterization of carp seminal plasma proteome in relation to blood plasma. J Proteom 98:218–232. https://doi.org/10.1016/J.JPROT.2014.01.005
Domínguez-Rebolledo A, Martínez-Pastor F, Bisbal A et al. (2011) Response of thawed epidi dymal red deer spermatozoa to increasing concentrations of hydrogen peroxide, and importance of individual male variability. Reprod Domest Anim 46:393–403. https://doi.org/10.1111/j.1439-0531.2010.01677.x
Duan P, Hu C, Butler HJ et al. (2016) Effects of 4-nonylphenol on spermatogenesis and induction of testicular apoptosis through oxidative stress-related pathways. Reprod Toxicol 62:27–38. https://doi.org/10.1016/j.reprotox.2016.04.016
Dziewirska E, Hanke W, Jurewicz J (2018) Environmental non-persistent endocrine-disrupting chemicals exposure and reproductive hormones levels in adult men. Int J Occup Med Environ Health https://doi.org/10.13075/ijomeh.1896.01183
Dzyuba V, Cosson J (2014) Motility of fish spermatozoa: from external signaling to flagella response. Reprod Biol 14:165–175. https://doi.org/10.1016/J.REPBIO.2013.12.005
Farag MR, Alagawany M (2018) Erythrocytes as a biological model for screening of xenobiotics toxicity. Chem Biol Inter 279:73–83. https://doi.org/10.1016/j.cbi.2017.11.007
Flint S, Markle T, Thompson S, Wallace E (2012) Bisphenol A exposure, effects, and policy: a wildlife perspective. J Environ Manag 104:19–34. https://doi.org/10.1016/j.jenvman.2012.03.021
Gao M, Yang Y, Lv M et al. (2018) Oxidative stress and DNA damage in zebrafish liver due to hydroxyapatite nanoparticles-loaded cadmium. Chemosphere 202:498–505. https://doi.org/10.1016/j.chemosphere.2018.03.146
Gillan L, Evans G, Maxwell WMC (2005) Flow cytometric evaluation of sperm parameters in relation to fertility potential. Theriogenology 63:445–457. https://doi.org/10.1016/j.theriogenology.2004.09.024
Gran View Research (2015) Global Bisphenol A (BPA) market by application (appliances, automotive, consumer, construction, electrical & electronics) expected to reach USD 20.03 billion by 2020: Grand View Research, Inc. https://www.grandviewresearch.com/press-release/global-bisphenol-a-bpa-market. Accessed 28 Jun 2018
Hagedorn M, McCarthy M, Carter VL, Meyers SA (2012) Oxidative stress in zebrafish (Danio rerio) sperm. PLoS ONE 7:e39397. https://doi.org/10.1371/journal.pone.0039397
Hamed HS, Abdel-Tawwab M (2017) Ameliorative effect of propolis supplementation on alleviating bisphenol-A toxicity: growth performance, biochemical variables, and oxidative stress biomarkers of Nile tilapia, Oreochromis niloticus (L.) fingerlings. Comp Biochem Physiol Part—C Toxicol Pharm 202:63–69. https://doi.org/10.1016/j.cbpc.2017.08.001
Harayashiki CAY, Junior ASV, Machado AA, de S et al. (2013) Toxic effects of the herbicide Roundup in the guppy Poecilia vivipara acclimated to fresh water. Aquat Toxicol 142–143:176–184. https://doi.org/10.1016/j.aquatox.2013.08.006
Hassan ZK, Elobeid MA, Virk P, et al. (2012) Bisphenol a induces hepatotoxicity through oxidative stress in rat model. Oxid Med Cell Longev 2012. https://doi.org/10.1155/2012/194829
Hatef A, Alavi SMH, Abdulfatah A et al. (2012) Adverse effects of bisphenol A on reproductive physiology in male goldfish at environmentally relevant concentrations. Ecotoxicol Environ Saf 76:56–62. https://doi.org/10.1016/j.ecoenv.2011.09.021
Sohoni PCRT, Tyler CR, Hurd K et al. (2001) Reproductive effects of long-term exposure to Bisphenol A in the fathead minnow (Pimephales promelas) Environ Sci Technol 35:2917–2925
Huang YQ, Wong CKC, Zheng JS et al. (2012) Bisphenol A (BPA) in China: a review of sources, environmental levels, and potential human health impacts. Environ Int 42:91–99. https://doi.org/10.1016/j.envint.2011.04.010
Hulak M, Gazo I, Shaliutina A, Linhartova P (2013) In vitro effects of bisphenol A on the quality parameters, oxidative stress, DNA integrity and adenosine triphosphate content in sterlet (Acipenser ruthenus) spermatozoa. Comp Biochem Physiol—C Toxicol Pharm 158:64–71. https://doi.org/10.1016/j.cbpc.2013.05.002
Im J, Löffler FE (2016) Fate of Bisphenol A in terrestrial and aquatic environments. Environ Sci Technol 50:8403–8416. https://doi.org/10.1021/acs.est.6b00877
Inaba K, Morisawa S, Morisawa M (1998) Proteasomes regulate the motility of salmonid fish sperm through modulation of cAMP-dependent phosphorylation of an outer arm dyne in light chain. J Cell Sci 111(Pt 8):1105–1115
Jalal N, Surendranath AR, Pathak JL et al. (2018) Bisphenol A (BPA) the mighty and the mutagenic. Toxicol Rep 5:76–84. https://doi.org/10.1016/j.toxrep.2017.12.013
Kang J-H, Kondo F, Katayama Y (2006) Human exposure to bisphenol A. Toxicology 226:79–89. https://doi.org/10.1016/j.tox.2006.06.009
Kourouma A, Peng D, Chao Q et al. (2014) Bisphenol A induced reactive oxygen species (ROS) in the liver and affect epididymal semen quality in adults Sprague-Dawley rats. J Toxicol Environ Health Sci 6:103–112. https://doi.org/10.5897/JTEHS2014.0309
Lahnsteiner F, Mansour N, Berger B (2004) The effect of inorganic and organic pollutants on sperm motility of some freshwater teleosts. J Fish Biol 65:1283–1297. https://doi.org/10.1111/j.0022-1112.2004.00528.x
Lamirande EDe, Gagnon C (1995) Impact of reactive oxygen species on spermatozoa: a balancing act between beneficial and detrimental effects. Hum Reprod 10:15–21
Li D, Chen Q, Cao J et al. (2016) The chronic effects of lignin-derived bisphenol and bisphenol A in Japanese medaka Oryzias latipes. Aquat Toxicol 170:199–207. https://doi.org/10.1016/j.aquatox.2015.11.024
Li P, Li Z-H, Dzyuba B et al. (2010) Evaluating the impacts of osmotic and oxidative stress on Common Carp (Cyprinus carpio, L.) sperm caused by cryopreservation techniques. Biol Reprod 83:852–858. https://doi.org/10.1095/biolreprod.110.085852
Liu Y, Tam NFY, Guan Y et al. (2011) Acute toxicity of nonylphenols and bisphenol A to the embryonic development of the abalone Haliotis diversicolor supertexta. Ecotoxicology 20:1233–1245. https://doi.org/10.1007/s10646-011-0672-7
Lopes FM, Varela Junior AS, Corcini CD et al. (2014) Effect of glyphosate on the sperm quality of zebrafish Danio rerio. Aquat Toxicol 155:322–326. https://doi.org/10.1016/j.aquatox.2014.07.006
Lv Y, Lu S, Dai Y et al. (2017) Higher dermal exposure of cashiers to BPA and its association with DNA oxidative damage. Environ Int 98:69–74. https://doi.org/10.1016/j.envint.2016.10.001
Maćczak A, Cyrkler M, Bukowska B, Michałowicz J (2017a) Bisphenol A, bisphenol S, bisphenol F and bisphenol AF induce different oxidative stress and damage in human red blood cells (in vitro study). Toxicol In Vitro 41:143–149. https://doi.org/10.1016/j.tiv.2017.02.018
Maćczak A, Duchnowicz P, Sicińska P et al. (2017b) The in vitro comparative study of the effect of BPA, BPS, BPF and BPAF on human erythrocyte membrane; perturbations in membrane fluidity, alterations in conformational state and damage to proteins, changes in ATP level and Na+/K+ATPase and AChE activities. Food Chem Toxicol 110:351–359. https://doi.org/10.1016/j.fct.2017.10.028
Michałowicz J, Mokra K, Bak A (2015) Bisphenol A and its analogs induce morphological and biochemical alterations in human peripheral blood mononuclear cells (in vitro study). Toxicol In Vitro 29:1464–1472. https://doi.org/10.1016/j.tiv.2015.05.012
Mokra K, Kuźmińska-Surowaniec A, Woźniak K, Michałowicz J (2017) Evaluation of DNA-damaging potential of bisphenol A and its selected analogs in human peripheral blood mononuclear cells (in vitro study). Food Chem Toxicol 100:62–69. https://doi.org/10.1016/j.fct.2016.12.003
Myridakis A, Chalkiadaki G, Fotou M et al. (2016) Exposure of Preschool-Age Greek Children (RHEA Cohort) to bisphenol A, parabens, phthalates, and organophosphates. Environ Sci Technol 50:932–941. https://doi.org/10.1021/acs.est.5b03736
Nakagawa Y, Tayama S (2000) Metabolism and cytotoxicity of bisphenol A and other bisphenols in isolated rat hepatocytes. Arch Toxicol 74:99–105
OECD (1993) OECD Guidelines for the Testing of Chemicals. Test 203: Fish, Acute Toxicity Test. OECD Publishing
Pasqualotto FF, Sharma RK, Nelson DR et al. (2000) Relationship between oxidative stress, semen characteristics, and clinical diagnosis in men undergoing infertility investigation. Fertil Steril 73:459–464
Petrunkina AM, Volker G, Weitze K-F et al. (2005) Detection of cooling-induced membrane changes in the response of boar sperm to capacitating conditions. Theriogenology 63:2278–2299. https://doi.org/10.1016/j.theriogenology.2004.10.008
Piehler E, Petrunkina AM, Ekhlasi-Hundrieser M, Töpfer-Petersen E (2006) Dynamic quantification of the tyrosine phosphorylation of the sperm surface proteins during capacitation. Cytometry Part A 69A:1062–1070. https://doi.org/10.1002/cyto.a.20338
Qiu W, Chen J, Li Y et al. (2016) Oxidative stress and immune disturbance after long-term exposure to bisphenol A in juvenile common carp (Cyprinus carpio). Ecotoxicol Environ Saf 130:93–102. https://doi.org/10.1016/j.ecoenv.2016.04.014
Riu A, Maire A, Grimaldi M et al. (2011) Characterization of novel ligands of ER a, Er b, and PPAR c: the case of halogenated bisphenol A and their conjugated. Metabolites 122:372–382. https://doi.org/10.1093/toxsci/kfr132
Rocco L, Frenzilli G, Zito G et al. (2012) Genotoxic effects in fish induced by pharmacological agents present in the sewage of some Italian water-treatment plants. Environ Toxicol 27:18–25. https://doi.org/10.1002/tox.20607
Rurangwa E, Kime D, Ollevier F, Nash J (2004) The measurement of sperm motility and factors affecting sperm quality in cultured fish. Aquaculture 234:1–28. https://doi.org/10.1016/J.AQUACULTURE.2003.12.006
Saalfeld GQ, Varela Junior AS, Castro T et al. (2018) Low atrazine dosages reduce sperm quality of Calomys laucha mice. Environ Sci Pollut Res 25:2924–2931. https://doi.org/10.1007/s11356-017-0657-z
Schulz RW, de França LR, Lareyre JJ et al. (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Sergent O, Ekroos K, Lefeuvre-Orfila L et al. (2009) Ximelagatran increases membrane fluidity and changes membrane lipid composition in primary human hepatocytes. Toxicol In Vitro 23:1305–1310. https://doi.org/10.1016/j.tiv.2009.07.019
Shao B, Zhu L, Dong M et al. (2012) DNA damage and oxidative stress induced by endosulfan exposure in zebrafish (Danio rerio). Ecotoxicology 21:1533–1540. https://doi.org/10.1007/s10646-012-0907-2
Shimada N, Yamauchi K (2004) Characteristics of 3,5,3′-triiodothyronine (T3)-uptake system of tadpole red blood cells: effect of endocrine-disrupting chemicals on cellular T3 response. J Endocrinol 183:627–637. https://doi.org/10.1677/joe.1.05893
Shrader EA, Henry TR, Greeley MS, Bradley BP (2003) Proteomics in zebrafish exposed to endocrine disrupting chemicals. Ecotoxicology 12:485–488
Sikka SC (2001) Relative impact of oxidative stress on male reproductive function. Curr Med Chem 8:851–862
Sorensen AM (1979) Repro lab: a laboratory manual for animal reproduction. American Press, Boston, MA, USA
Tao S, Zhang Y, Yuan C et al. (2016) Oxidative stress and immunotoxic effects of bisphenol A on the larvae of rare minnow Gobiocypris rarus. Ecotoxicol Environ Saf 124:377–385. https://doi.org/10.1016/j.ecoenv.2015.11.014
Tiwari D, Kamble J, Chilgunde S et al. (2012) Clastogenic and mutagenic effects of bisphenol A: an endocrine disruptor. Mutat Res - Genet Toxicol Environ Mutagen 743:83–90. https://doi.org/10.1016/j.mrgentox.2011.12.023
Vandenberg LN, Hunt PA, Myers JP, vom Saal FS (2013) Human exposures to bisphenol A: mismatches between data and assumptions. Rev Environ Health 28:37–58. https://doi.org/10.1515/reveh-2012-0034
Vandenberg LN, Maffini MV, Sonnenschein C et al. (2009) Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption. Endocr Rev 30:75–95. https://doi.org/10.1210/er.2008-0021
Vernet P, Aitken R, Drevet J (2004) Antioxidant strategies in the epididymis. Mol Cell Endocrinol 216:31–39. https://doi.org/10.1016/j.mce.2003.10.069
Verstegen J, Iguer-Ouada M, Onclin K (2002) Computer assisted semen analyzers in andrology research and veterinary practice. Theriogenology 57:149–179
Yamamoto T, Yasuhara A, Shiraishi H, Nakasugi O (2001) Bisphenol A in hazardous waste landfill leachates. Chemosphere 42:415–418. https://doi.org/10.1016/S0045-6535(00)00079-5
Zhang T, Liu Y, Chen H et al. (2017) The DNA methylation status alteration of two steroidogenic genes in gonads of rare minnow after bisphenol A exposure. Comp Biochem Physiol Part - C Toxicol Pharm 198:9–18. https://doi.org/10.1016/j.cbpc.2017.05.001
Zhao H, Wei J, Xiang L, Cai Z (2018) Mass spectrometry investigation of DNA adduct formation from bisphenol A quinone metabolite and MCF-7 cell DNA. Talanta 182:583–589. https://doi.org/10.1016/j.talanta.2018.02.037
Acknowledgements
We would like to acknowledge the Fundação de Amparo à pesquisa Rio Grande do Sul (FAPERGS). ASV Jr (Process number 307195/2015-7) and CDC (Process number 306356/2014-7) are a research fellow from the Brazilian Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted (Experimental Animal Ethic Committee CEUA – FURG Process number 23116.000355/2016-72).
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Silveira, C.R., Varela Junior, A.S., Corcini, C.D. et al. Effects of Bisphenol A on redox balance in red blood and sperm cells and spermatic quality in zebrafish Danio rerio. Ecotoxicology 28, 913–922 (2019). https://doi.org/10.1007/s10646-019-02091-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-019-02091-5