Abstract
Background
Refractory benign esophageal strictures (RBES) are defined as those that persist structurally and symptomatically after repeated dilation sessions. Small series have reported favorable outcomes after placement and subsequent removal of Polyflex™ self-expanding plastic stents (SEPS).
Aims
To characterize the outcomes after Polyflex™ stent placement in patients with RBES.
Methods
Outcomes of consecutive patients who underwent Polyflex™ stent placement for RBES between April 15, 2005 and November 20, 2006 were analyzed retrospectively. The etiology of the stricture, number of dilations prior to initial SEPS placement, size of stent placed, stricture resolution after SEPS removal, cases of stent migration, duration of SEPS placement, time to repeat dilation and/or SEPS replacement after stent removal, and complications were assessed.
Results
Twenty-three Polyflex™ stents were placed in 13 patients suitable for analysis. The majority of stenoses (11/13) were attributable to anastomotic strictures after curative esophagogastrectomy for esophageal neoplasms. All 11 patients in this group had satisfactory relief of dysphagia to solids determined clinically with indwelling SEPS placement. SEPS migration occurred in seven instances (30% of SEPS placed). Dysphagia remediation after subsequent SEPS removal was observed in only three patients (23%). Placement of a partially covered metal stent within a Polyflex™ stent was necessary in one case due to epithelial hyperplasia. The mean time for repeat dilation and/or SEPS reinsertion due to recurrent dysphagia after stent removal was 37 days (range 6–120 days).
Conclusions
Polyflex™ stents provided satisfactory palliation of dysphagia for RBES while in place. However, unplanned SEPS migration and dysphagia recurrence after stent removal were common.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The majority of benign esophageal strictures are effectively treated with endoscopic dilation [1]. Esophageal strictures that cannot be dilated to an adequate diameter for relief of dysphagia despite repeated dilation sessions or that require multiple dilations due to recurrence of dysphagia can be generally considered as refractory benign esophageal strictures (RBES) [2, 3]. The use of intralesional corticosteroid injections, electrocautery incisions, and endoscopic scissors for RBES has been reported and may reduce the rate of RBES recurrence [4–8].
Another proposed treatment of RBES is the placement of removable, covered self-expanding plastic stent (SEPS), such as the Polyflex™ esophageal stent (Willy Rüsch GmbH/Boston Scientific Corporation, Natick, MA). This stent is composed of a polyester mesh with a silicone covering and has the theoretical advantages compared with self-expanding metallic stents of being removable and decreased tissue ingrowth [9]. Two small studies from 2004 reported dysphagia improvement in over 70% of patients after Polyflex™ stent placement and removal [10, 11]. However, stent migration is not uncommon and higher failure rates have been reported more recently [12–17].
The aim of this study was to characterize treatment outcomes after Polyflex™ stent placement for management of recurrent dysphagia in a cohort of consecutive patients with RBES at our institution.
Methods
This was a retrospective cohort analysis. Outcomes of patients who underwent Polyflex™ stent placement for RBES between April 2005 and November 2006 at the Hospital of the University of Pennsylvania were analyzed. RBES was defined in this series as benign strictures that did not resolve clinically after three or more dilation sessions. Cases in which SEPS were placed for management of fistulas, leaks, or malignant stenoses were excluded.
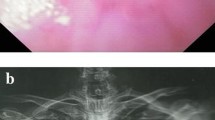
Dilations and stent placements were performed by three experienced endoscopists. All procedures were performed using either conscious sedation (midazolam, fentanyl) or propofol when necessary. Both Savary-Guilliard dilators (Cook Medical, Bloomington, IN) and CRE (Boston Scientific Corporation, Natick, MA) balloons were used for dilation up to 18–20 mm in diameter for esophageal dilations performed prior to consideration of SEPS placement. At time of SEPS placement, esophageal dilation was performed to 12–14 mm when necessary to allow passage of the Polyflex™ stent delivery device over a stiff guidewire. The SEPS were then deployed under fluoroscopic and endoscopic guidance. Placement was verified endoscopically and fluoroscopically after deployment and modified as indicated (Fig.1). Stent removal was performed using Olympus FG-47L-1 grasping forceps (Olympus America Incorporated, Center Valley, PA).
In the absence of stent migration or inability to remove the SEPS, the first stent was removed after approximately 6 weeks. If restenosis occurred after SEPS removal, subsequent stents were left in place for approximately 3 months followed by a maximum of approximately 6 months.
Relevant clinical information was obtained from patient charts as well as a computerized endoscopic database. For each patient, the stricture etiology, number of dilations prior to initial SEPS placement, size of stent(s) placed, stricture resolution after SEPS removal, stent migration, duration of SEPS placement, time to repeat dilation and/or SEPS replacement after stent removal, and complications were entered into a spreadsheet (Microsoft Excel, Microsoft Corporation, Redmond, WA) and analyzed. RBES resolution after SEPS placement and removal was defined as the patient reporting no recurrent symptoms of dysphagia and not requiring additional dilation or stent replacement.
For the entire cohort, the mean number of initial dilations, repeat dilations, as well as time to repeat dilation or stent replacement were calculated using the “average” function in Microsoft Excel.
This study was approved by the University of Pennsylvania Institutional Review Board.
Results
Thirteen patients met the study inclusion criteria, in whom a total of 23 SEPS were placed. A summary of stricture characteristics, interventions performed, and outcomes for each patient is outlined in Table 1.
Eleven of the 13 patients were male, and the mean age of this cohort was 66 years (range 47–82 years). Eleven of the 13 patients had RBES due to anastomotic strictures after curative esophagogastrectomy for esophageal cancer. Six of these 11 patients had received neoadjuvant chemoradiotherapy. All 11 patients in this group had satisfactory relief of dysphagia with the SEPS in place, as determined by patient self-reporting. Of the remaining two RBES, one was attributed to prolonged nasogastric tube placement during intensive care unit admission and the other was due to radiation therapy post laryngectomy for laryngeal carcinoma. There was suboptimal relief of dysphagia in the latter patient with indwelling SEPS placement.
The mean and median number of dilations prior to initial SEPS insertion was eight and seven, respectively (range 3–14). The 9-cm-long 18/23-mm-diameter Polyflex™ stent was used in most cases (19/23), and the 12-cm-long 21/25-mm-diameter Polyflex™ stent was used in the remaining four cases. Of the four cases in which the larger-diameter SEPS was used, two were placed after migration of the smaller-diameter SEPS. The number of SEPS placed per patient ranged from one to four.
Unplanned SEPS migration occurred in seven instances, specifically in 26% (5/19) and 50% (2/4) of the 9-cm 18/23-mm and 12-cm 21/25-mm stents placed, respectively (Fig.2). In all cases, the migrated stent was successfully removed with grasping forceps. Stricture resolution as determined endoscopically was observed in one out of the seven cases of SEPS migration and occurred after placement of the larger-diameter Polyflex™ stent. A new SEPS was placed in four cases and the old stent was repositioned in two cases; esophageal dilation was required for five of these cases. The migrated SEPS was removed without placement of a new stent in one case. For patients in whom stent migration was not observed or who were not lost to follow-up, the mean duration of SEPS insertion was 101 days (range 30–257 days).
In one case, a partially covered Ultraflex™ esophageal stent (Boston Scientific Corporation, Natick, MA) was placed within the Polyflex™ stent to palliate epithelial hyperplasia resulting from an indwelling 12-cm 21/25-mm Polyflex™ stent that had been in place for approximately 10 weeks.
Eventual stricture resolution after subsequent SEPS removal, determined clinically by resolution of dysphagia, was observed in only three patients (23%). Two patients whose dysphagia resolved had a 12-cm 21/25-mm Polyflex™ stent placed prior to removal, although stent migration occurred in both cases. Two of these patients required multiple SEPS prior to RBES resolution. Of the six patients who had received prior radiation therapy, one patient had resolution of RBES determined clinically. The relationship between stricture resolution and duration of stent insertion could not be analyzed because stent migration commonly occurred. The mean time for repeat dilation and/or SEPS reinsertion due to recurrent dysphagia after stent removal was 37 days (range 6–120 days).
Discussion
Initial studies of SEPS for the treatment of RBES reported encouraging results in terms of resolution of dysphagia with the SEPS in place as well as after subsequent removal [10, 11]. While most patients in our series experienced adequate relief of dysphagia while the SEPS was in place, over 75% of the patients did not have sustained relief of dysphagia once the SEPS was removed. One possible explanation is that the case selection in our series represented a more refractory cohort of benign esophageal strictures compared with those considered in prior studies. Another possibility is that the smaller-diameter (18/23 mm) Polyflex™ stent, placed more commonly in our patients, was less effective in achieving the tissue remodeling needed to promote durable stricture resolution after SEPS removal versus the larger-diameter (21/25 mm) Polyflex™ stent. However, the small numbers of patients treated with each precludes making any definitive association between the two. The small sample size was also inadequate to discern whether prior radiation therapy, which can result in fibrosis of submucosa and muscularis, was associated with failure of RBES to resolve after SEPS placement.
Similar suboptimal results after Polyflex™ stent removal in patients with RBES have been reported by Dua et al., who used inability to maintain luminal diameter of 14 mm despite dilation every 2–4 weeks as an entry criteria [14]. In this series, 40% of patients remained dysphagia free after stent removal and a stent migration rate of 22% was observed. RBES resolution rates as low as 12.5% after stent removal have been reported previously [16]. However, direct comparisons with previously published studies are potentially limited by the different definitions of RBES used. A recently published series from the Mayo Clinic also reported a low rate of long-term improvement of symptoms after SEPS removal in patients with benign esophageal strictures and a high rate of stent migration; however, the patients in this series were not noted to necessarily have RBES [15].
Stent migration occurred in approximately 30% of the patients in this series; all migrated stents were successfully recovered endoscopically. The significance of a higher percentage of larger-diameter Polyflex™ stents migrating compared with the smaller-diameter stents is unclear given the small number of larger-diameter Polyflex™ stents placed. Prior studies in patients with RBES have reported stent migration rates ranging from 7% to 85% of SEPS placed [10–12, 14, 16]. While no significant adverse sequelae have been reported due to SEPS migration, the possibility of bowel obstruction and perforation potentially exists. Asymptomatic Polyflex™ stent migration per rectum has been reported [13].
Significant epithelial hyperplasia occurred in one patient at the distal end of the Polyflex™ stent. This was surprising in that there was hope that the smooth covered surfaces of this stent would be apt to recruit a lesser degree of reactive epithelial hyperplasia. This phenomenon associated with Polyflex™ stent placement has been similarly reported by others [10, 12, 14, 15].
We did not observe significant complications in our series. However, perforation, tracheal compression, and aortoenteric fistula have been reported in the literature [11, 12, 14, 17–19].
In this cohort of patients with RBES, SEPS provided satisfactory palliation of dysphagia for a majority of patients while in place. However, unplanned stent migration, even with the larger-diameter stent, and stricture recurrence after stent removal commonly occurred. Clinically significant reactive epithelial hyperplasia was also observed. Exclusive use of larger-diameter (21/25 mm) Polyflex™ stents may yield more favorable results for durable RBES resolution. Standardizing entry criteria for patients undergoing endoprosthetic placement would potentially lead to more definitive conclusions in this regard [2].
An effective durable therapy for patients with RBES remains elusive. A discussion raising the consideration of long-term and potentially lifelong endoprosthetic placement may be necessary in some patients, since all esophageal strictures are not equally likely to respond [14, 20]. Further studies will be necessary to determine the optimal application of SEPS placement for the management of RBES in terms of stricture etiology, stent length and diameter, and duration of stent placement.
References
Marks RD, Richter JE. Peptic strictures of the esophagus. Am J Gastroenterol. 1993;88:1160–1173.
Kochman ML, McClave SA, Boyce HW. The refractory and the recurrent esophageal stricture: a definition. Gastrointest Endosc. 2005;62:474–475.
Shah JN. Benign refractory esophageal strictures: widening the endoscopist’s role. Gastrointest Endosc. 2006;63:164–167.
Lee M, Kubik CM, Polhamus CD, Brady CE 3rd, Kadakia SC. Preliminary experience with endoscopic intralesional steroid injection therapy for refractory upper gastrointestinal strictures. Gastroinest Endosc. 1995;41:598–601.
Zein NN, Greseth JM, Perrault J. Endoscopic intralesional steroid injections in the management of refractory esophageal strictures. Gastrointest Endosc. 1995;41:596–598.
Ramage JI Jr, Rumalla A, Baron TH, et al. A prospective, randomized, double-blind, placebo-controlled trial of endoscopic steroid injection therapy for recalcitrant esophageal peptic strictures. Am J Gastroenterol. 2005;100:2419–2425.
Hordijk ML, Sersema PD, Tilanus HW, Kuipers EJ. Electrocautery therapy for refractory anastomotic strictures of the esophagus. Gastrointest Endosc. 2006;63:157–163.
Beilstein MC, Kochman ML. Endoscopic incision of a refractory esophageal stricture: novel management with an endoscopic scissors. Gastrointest Endosc. 2005;61:623–625.
Karbowski M, Schembre D, Kozarek R, Ayub K, Low D. Polyflex self-expanding, removable plastic stents: assessment of treatment efficacy and safety in a variety of benign and malignant conditions of the esophagus. Surg Endosc. 2008;22:1326–1333.
Repici A, Conio M, De Angelis C, et al. Temporary placement of an expandable polyester silicone-covered stent for treatment of refractory benign esophageal stricture. Gastrointest Endosc. 2004;60:513–519.
Evrard S, Le Moine O, Lazaraki G, Dormann A, El Nakadi I, Devière J. Self-expanding plastic stents for benign esophageal lesions. Gastrointest Endosc. 2004;60:894–900.
Triester SL, Fleischer DE, Sharma VK. Failure of self-expanding plastic stents in treatment of refractory benign esophageal strictures. Endoscopy. 2006;38:533–537.
Oh YS, Kochman ML. Polyflex esophageal stent migration with elimination per rectum. Gastrointest Endosc. 2007;66:633.
Dua KS, Vleggaar FP, Santharam R, Siersema PD. Removable self-expanding plastic esophageal stent as a continuous, non-permanent dilator in treating refractory benign esophageal strictures: a prospective two-center study. Am J Gastroenterol. 2008;103:2988–2994.
Holm AN, a Levy JG, Gostout CJ, Topazian MD, Baron TH. Self-expanding plastic stents in the treatment of benign esophageal conditions. Gastrointest Endosc. 2008;67:20–25.
Barthel JS, Kelley ST, Klapman JB. Management of persistent gastroesophageal anastomotic strictures with removable self-expandable polyester silicon-covered (Polyflex) stents: an alternative to serial dilation. Gastorintest Endosc. 2008;67:546–552.
Radecke K, Gerken G, Treichel U. Impact of a self-expanding, plastic esophageal stent on various esophageal stenoses, fistulas, and leakages: a single-center experience in 39 patients. Gastrointest Endosc. 2005;61:812–818.
Rogart J, Greenwald A, Rossi F, Barrett P, Aslanian H. Aortoesophageal fistula following Polyflex stent placement for refractory benign esophageal stricture. Endoscopy. 2007;39(Suppl 1):E321–E322.
Pennathur A, Chang AC, McGrath KM, et al. Polyflex expandable stents in the treatment of esophageal disease: initial experience. Ann Thorac Surg. 2008;85:1968–1973.
Kochman ML. Removable endoprostetics in the management of esophageal pathology: all strictures and fistulae are not created equal. Gastrointest Endosc. 2008;67:26–27.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oh, Y.S., Kochman, M.L., Ahmad, N.A. et al. Clinical Outcomes After Self-Expanding Plastic Stent Placement for Refractory Benign Esophageal Strictures. Dig Dis Sci 55, 1344–1348 (2010). https://doi.org/10.1007/s10620-010-1134-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-010-1134-4